1. Introduction
Ochratoxin A (OTA) was first characterised in South Africa [
1] but found application as a significant mycotoxin in epidemics of nephropathy in the Danish bacon industry, first noted by Larsen [
2] and seasonally prevalent in the 1960s and 1970s. Renal carcinogenicity was demonstrated experimentally in male mice after protracted exposure to OTA [
3]. A major study followed in the US National Toxicology Program [
4,
5], notably demonstrating renal carcinomas, particularly in male rats. The latter generated precautionary concern for natural OTA occurrence in some foodstuffs such as cereals, coffee and wine caused by fungal spoilage of agricultural products, and precipitated current human food safety regulations for the toxin in several parts of the world [
6]. A focus on the mechanisms of carcinogenicity was made in a European Commission project in the 2000s, confirming male rat sensitivity to continuous dietary OTA during most of the first half of life [
7,
8].
Meanwhile, following the deductions of Krogh [
9] that OTA’s causal nephropathic role in Danish porcine morbidity might point to an analogous role in the human disease Balkan endemic nephropathy (BEN), the urinary tract cancers commonly associated with the human disease were shown to be of urothelial origin [
10]. Soon after, the International Agency for Research on Cancer considered OTA as ‘possibly carcinogenic to humans’ [
11], although no rat or mouse renal tumours caused by OTA exposure had been shown experimentally to mimic human urothelial neoplasms, whether at urothelial sites or as renal cell carcinomas in renal parenchyma. However, rat kidney tumours may be either of renal cell or urothelial cell origin, but gross distortions of many experimental OTA kidneys preclude morphological diagnosis of their origin. Some experimental kidney tumours caused by OTA are accompanied by neoplasms in other organs (e.g., lung). These have always been assumed to be metastatic from the kidney [
4], but have never been subjected to contemporary clinical immunohistochemical scrutiny.
The present pilot study seeks for the first time to contribute to enhanced pathological definition of archived rat tumours, in response to chronic dietary exposure to OTA in the London studies [
12,
13], also through an application of clinical immunohistochemistry. Focus has been on large carcinomas from males caused by daily OTA intake in the range 30–300 µg/kg body weight, dosages much greater than in natural human exposure. Additionally, a prominent carcinoma among females of the NTP study [
4] has been included for immunohistochemical comparison. Histopathology in further sections of the other nine NTP female renal tumour cases, to complement general gender-response features already illustrated [
14], is also critically reviewed.
3. Discussion
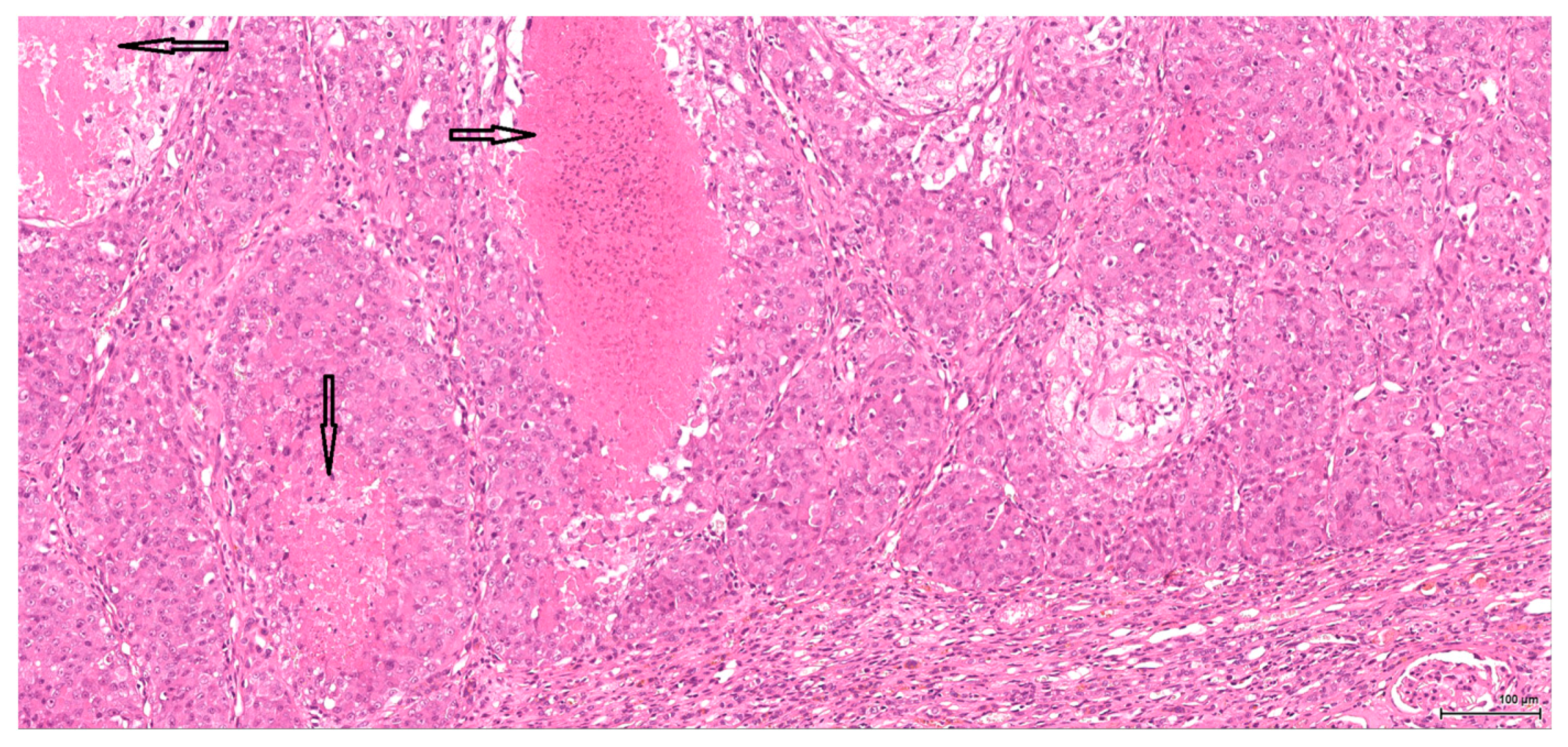
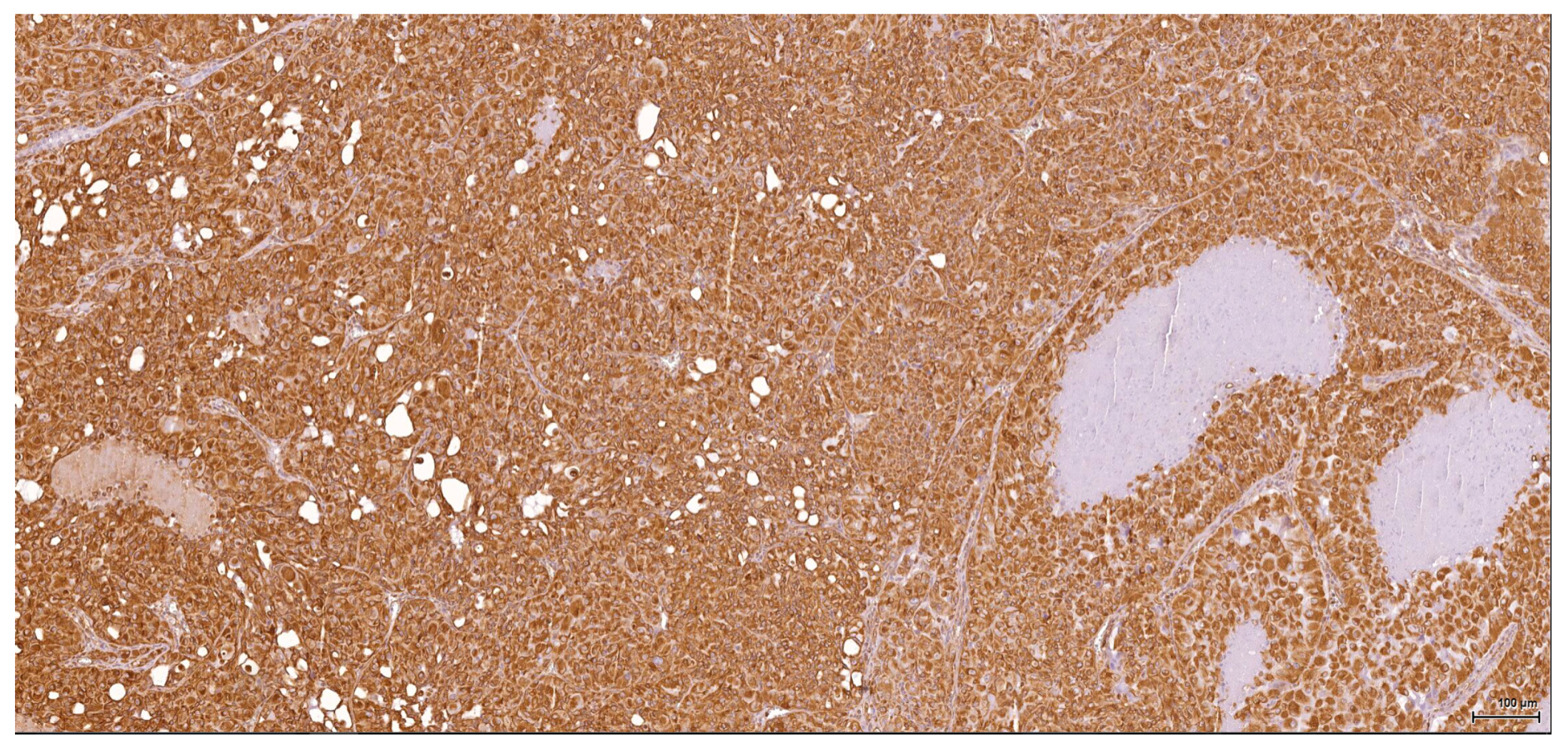
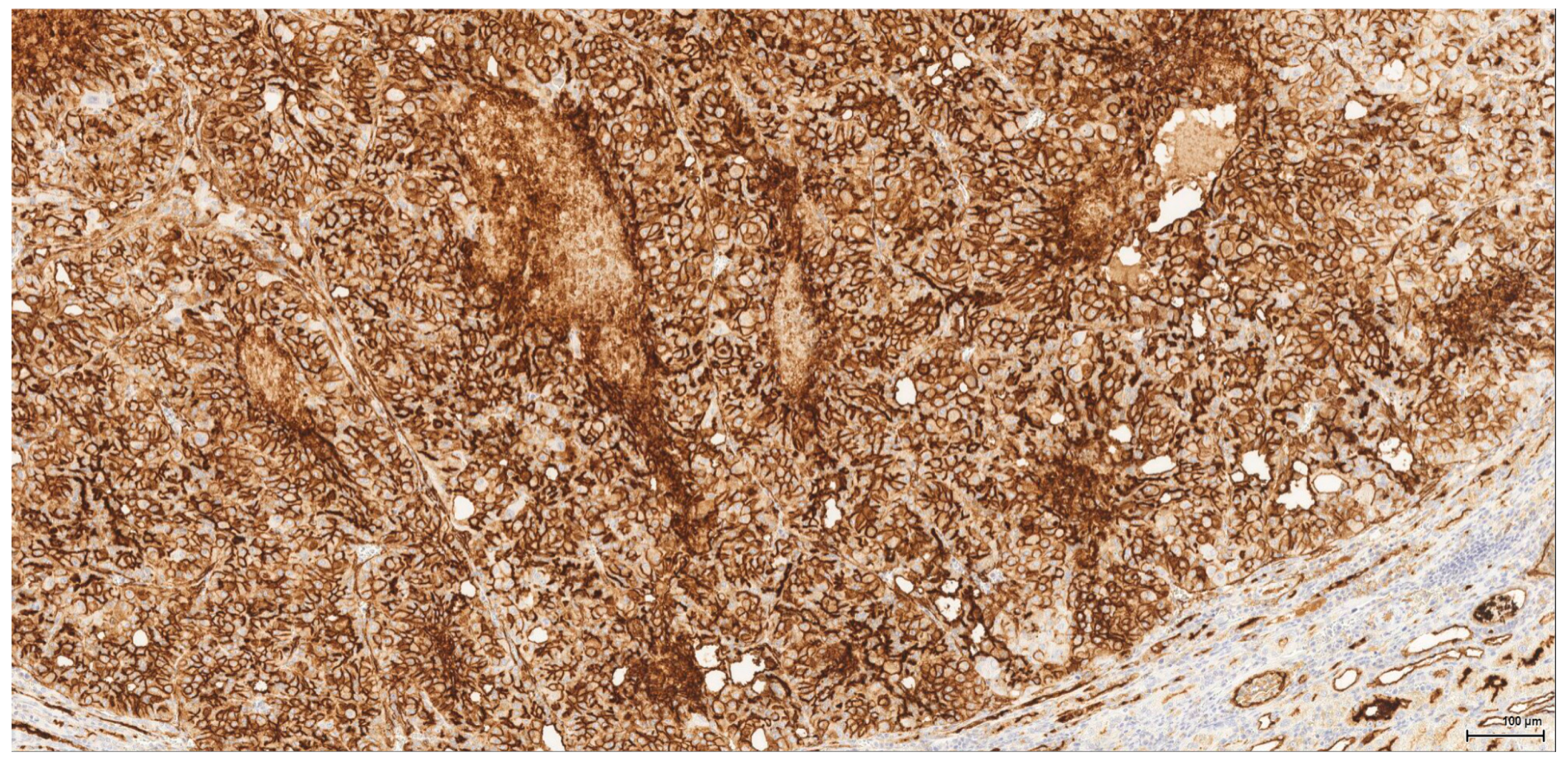
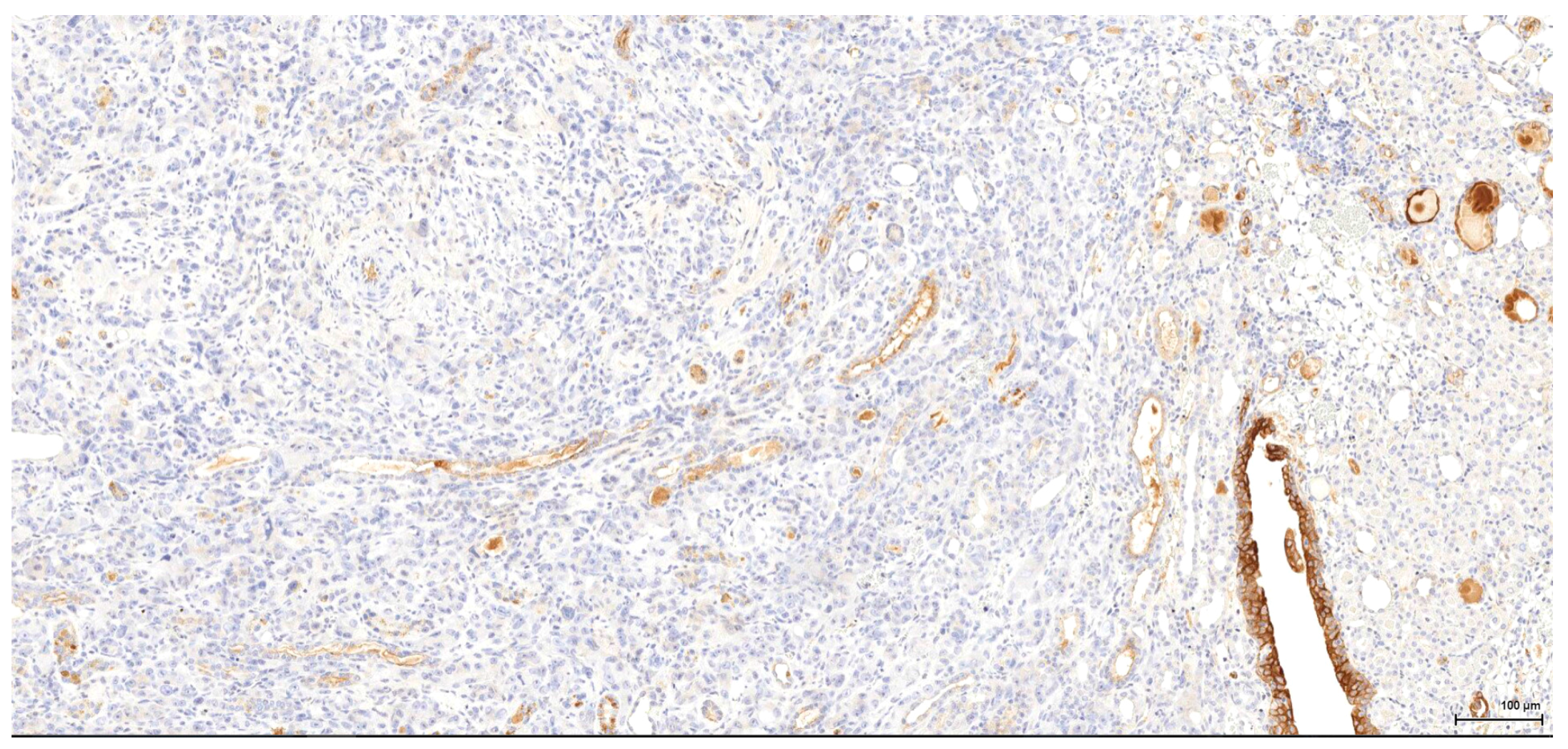
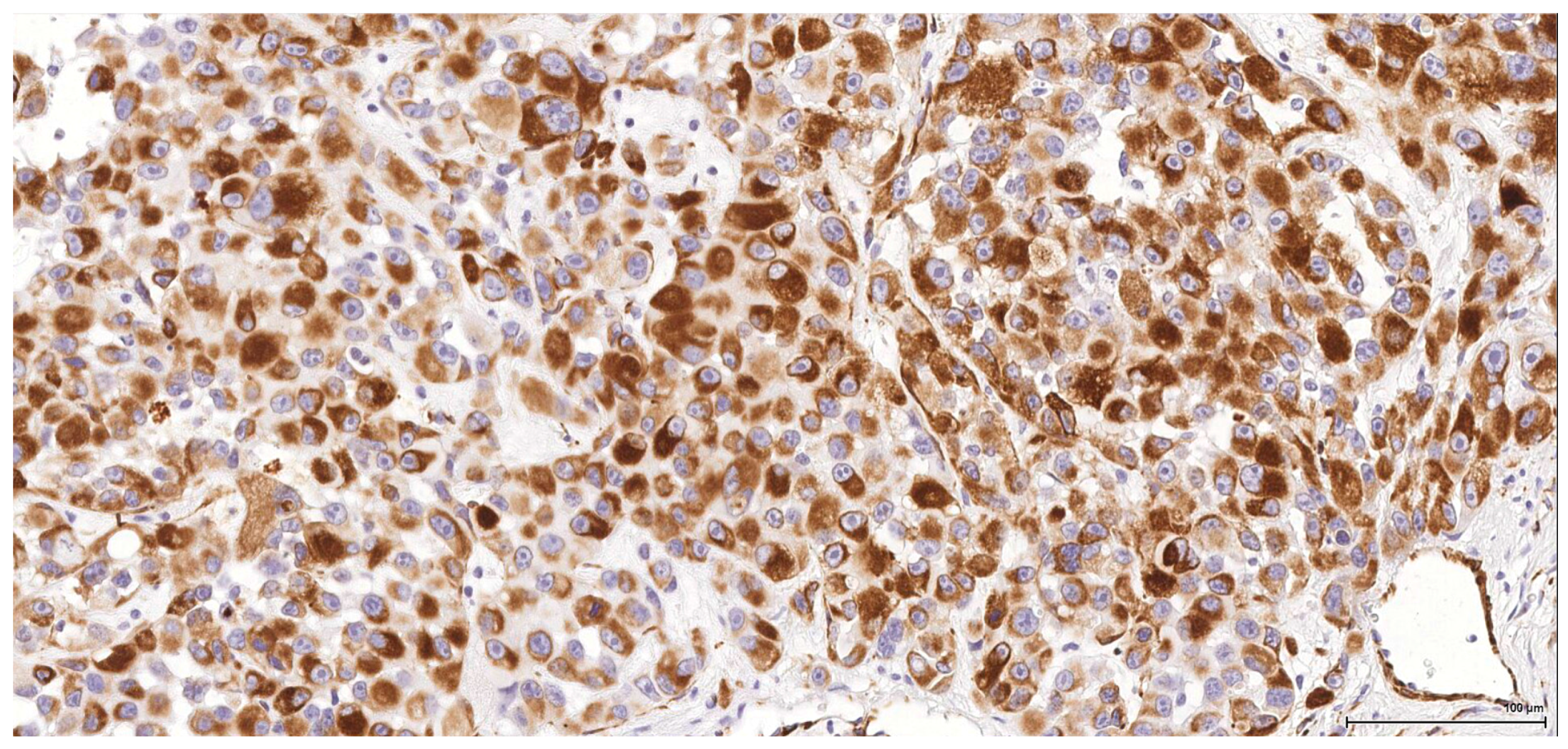
Historically, the renal cell origin has been difficult to establish and only the present IHC could prove it. It is important to emphasize that these tumours are of renal tubular cell origin and not urothelial, as proven by immunohistochemistry and suggested by the renal intraepithelial dysplastic lesion. Morphologically the renal cell nature is not immediately obvious, the cytoplasm being rather eosinophilic and only focally clear, as expected from a conventional clear cell renal cell carcinoma. Also, the nuclei are showing prominent nucleoli, visible on low power, indicating a high nuclear grade (Fuhrman). Likewise, the frequent association of a spindle cell component (sometimes massive or even exclusive) potentially hinders the renal cell origin assessment. However, immunohistochemically, the co-expression of vimentin and CD10 and the lack of reaction for CK7, CKHMW and p63 are supportive of this origin. In normal kidney, CD10 is strongly expressed by the cytoplasm and membrane of podocytes and proximal tubular cell brush borders; in renal cell carcinoma CD10 is usually positive in clear cell (including eosinophilic, granular cell variant) and papillary type renal cell carcinomas (RCC). In addition to this unequivocal immunohistochemical profile, an indirect criterion for non-urothelial cell origin is the fact that the sarcomatoid component association is rather uncommon for urothelial neoplasms; very few cases have been described in the literature [
15]. Sarcomatoid RCC is not very frequent either, but is still found in a significant percentage of RCC cases.
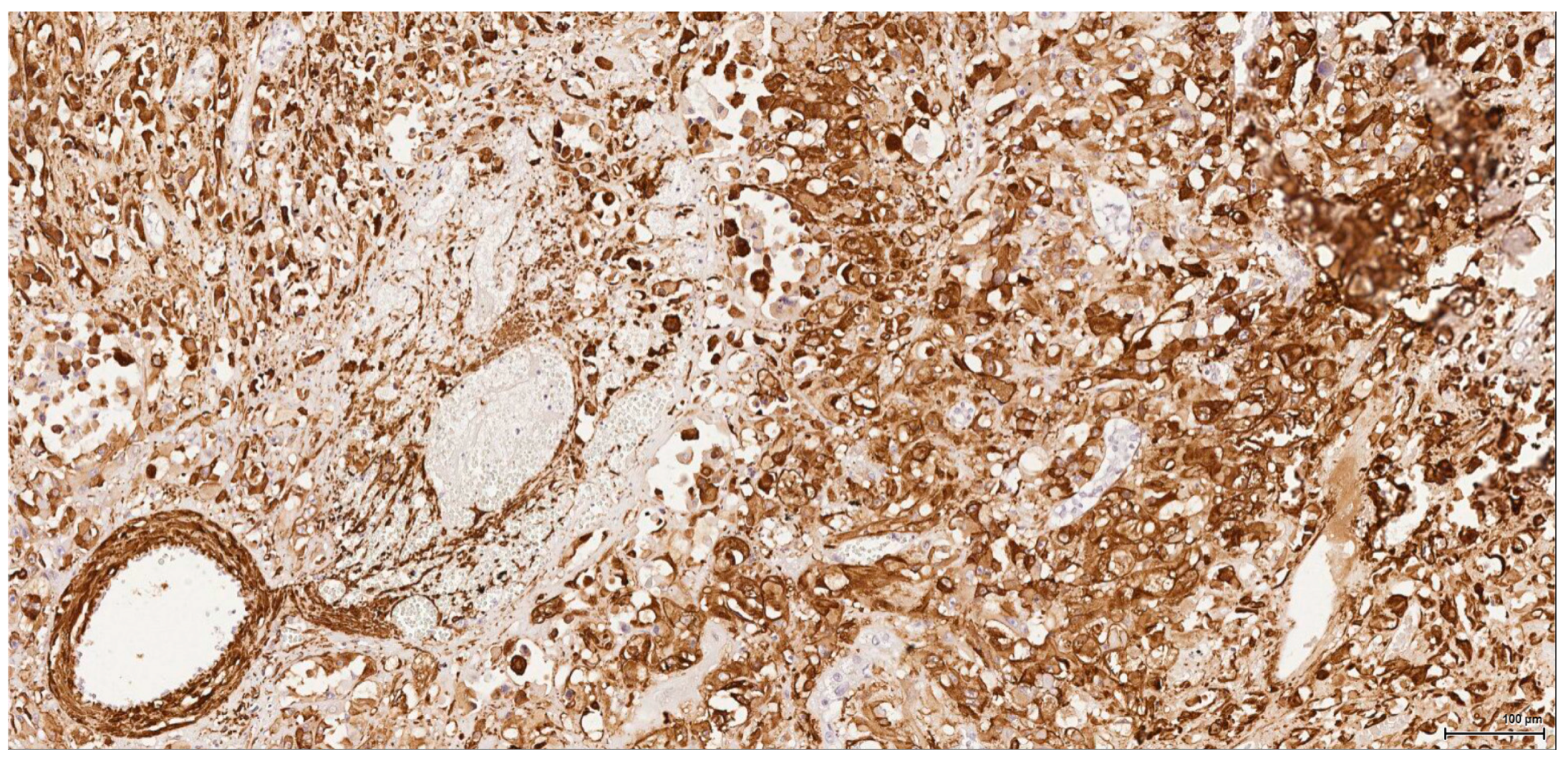
There is overall high grade cytology in both epithelioid and sarcomatoid areas, with high nuclear grade and necrosis; all these findings place these tumours in a poorly differentiated category. No classic conventional clear cell RCC picture has been identified; the epithelioid cells were mostly of eosinophilic granular cell type. When large amounts of spindle cell morphology are admixed, even merely associated or predominate, the degree of dedifferentiation is mirrored by the altered immune expression of CD10, which is lost. Instead, pan-CK expression occurs, which is variably co-expressed with vimentin, thus favouring the sarcomatoid nature of the tumour.
RCC with sarcomatoid features is not currently recognized as an entity as a specific type of renal parenchymal carcinoma by the most recent 2016 WHO classification of renal tumours, mainly because sarcomatoid areas can be found in all histologic subtypes of RCC [
16]. Collecting duct carcinoma exhibits the highest percentage of cases with sarcomatoid change ranging from 25% to 29% in separate series, whilst clear cell RCC displays sarcomatoid change in 5.2% to 8% of cases, compared with 2% to 9% by chromophobe RCC, and 1.9% to 5.4% by papillary RCC [
17].
Sarcomatoid RCC may arise out of a background of any histologic type of RCC as a manifestation of a final common dedifferentiation pathway. A pure sarcomatoid pattern is uncommon and, as recommended by the same previously mentioned guidelines, should be categorized as unclassified RCC (grade 4 unclassified carcinomas with a sarcomatoid component). The overwhelming sarcomatoid component found in these six tumours is striking because of its frequency and extent. OTA dose-factor difference does not seem to be crucial since, for example, within the same lot one tumour is organ-limited, small, nodular, well-demarcated, epithelioid and without spindle cell morphology, whilst the other one is a highly aggressive, metastasising tumour with sarcomatoid features.
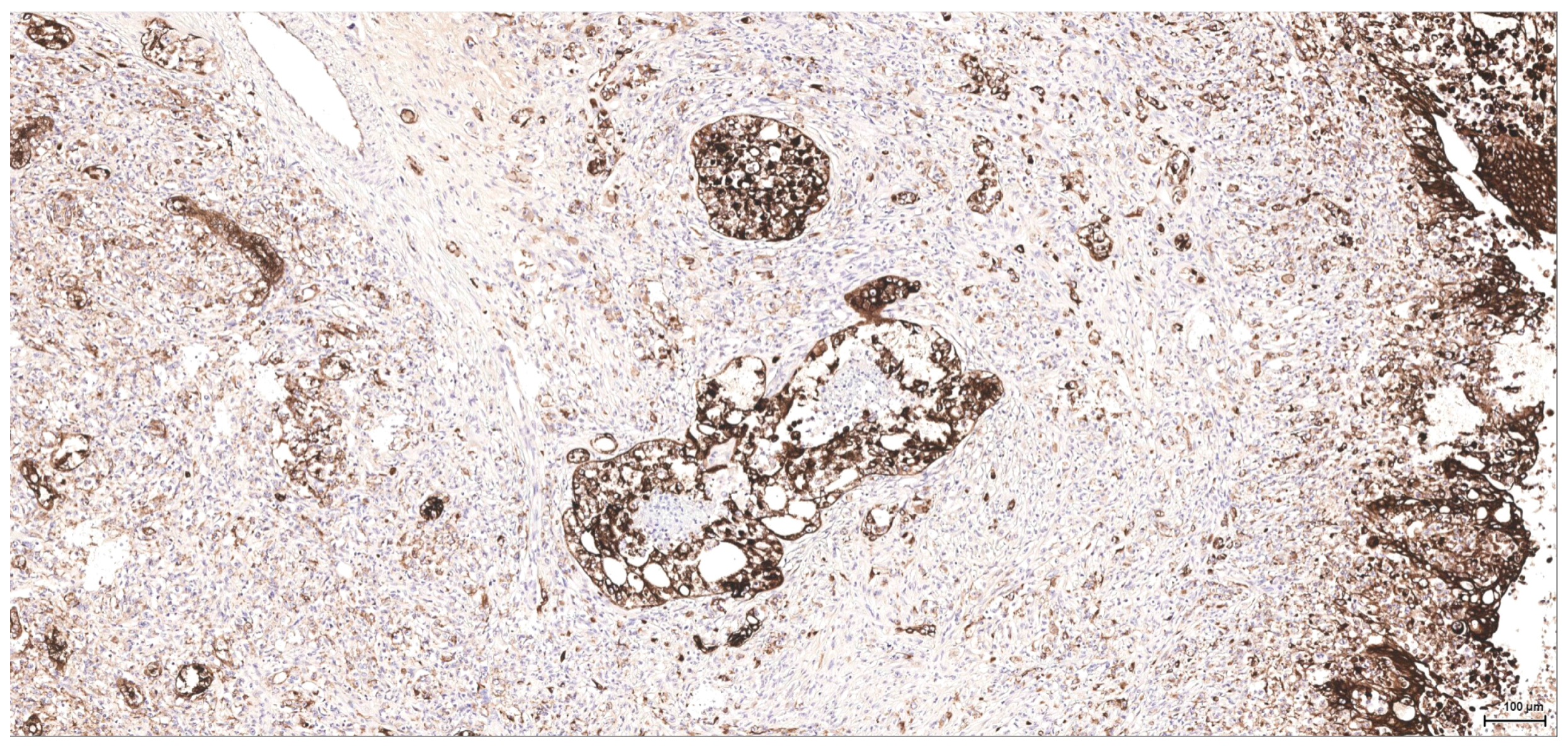
Rhabdoid differentiation in RCC refers to the development of neoplastic cells that morphologically resemble rhabdomyoblasts but differ in ultrastructural features and immunophenotype (being desmin negative). The similar immunophenotype of rhabdoid and non-rhabdoid foci supports the origin of the rhabdoid cells from renal cells. Areas of rhabdoid morphology do not represent metaplastic muscle differentiation [
16]. Bearing this in mind, the only case with focal rhabdoid morphology on a sarcomatoid background which does express desmin (but no SMA) might be interpreted as a heterologous differentiation along the rhabdomyosarcoma line arising in an otherwise unclassified RCC with sarcomatoid component.
Completely different in the present study is case 6, showing a spindle cell proliferation which is positive diffusely for SMA and focally for desmin. This indicates a pure mesenchymal origin, suggestive of leiomyosarcoma.
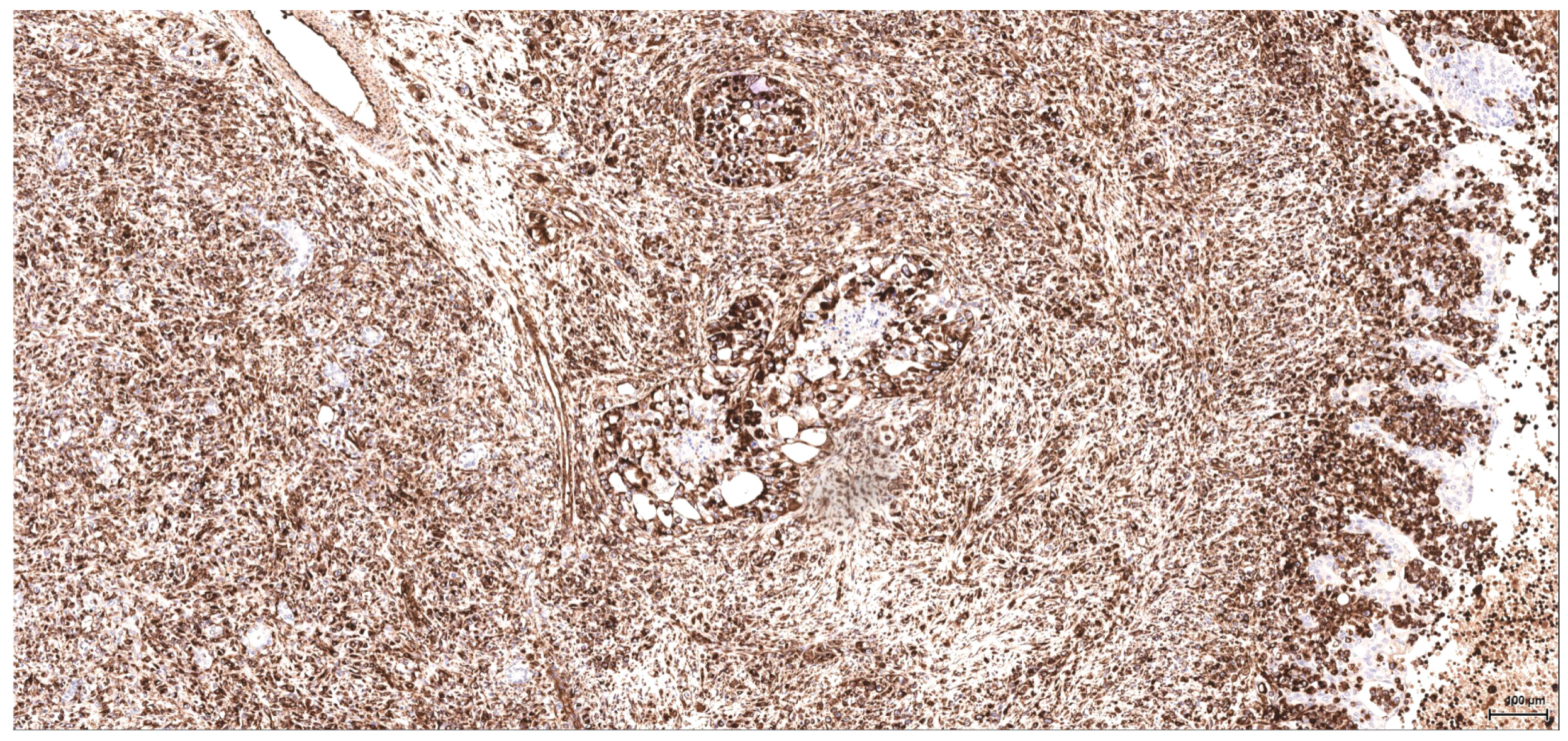
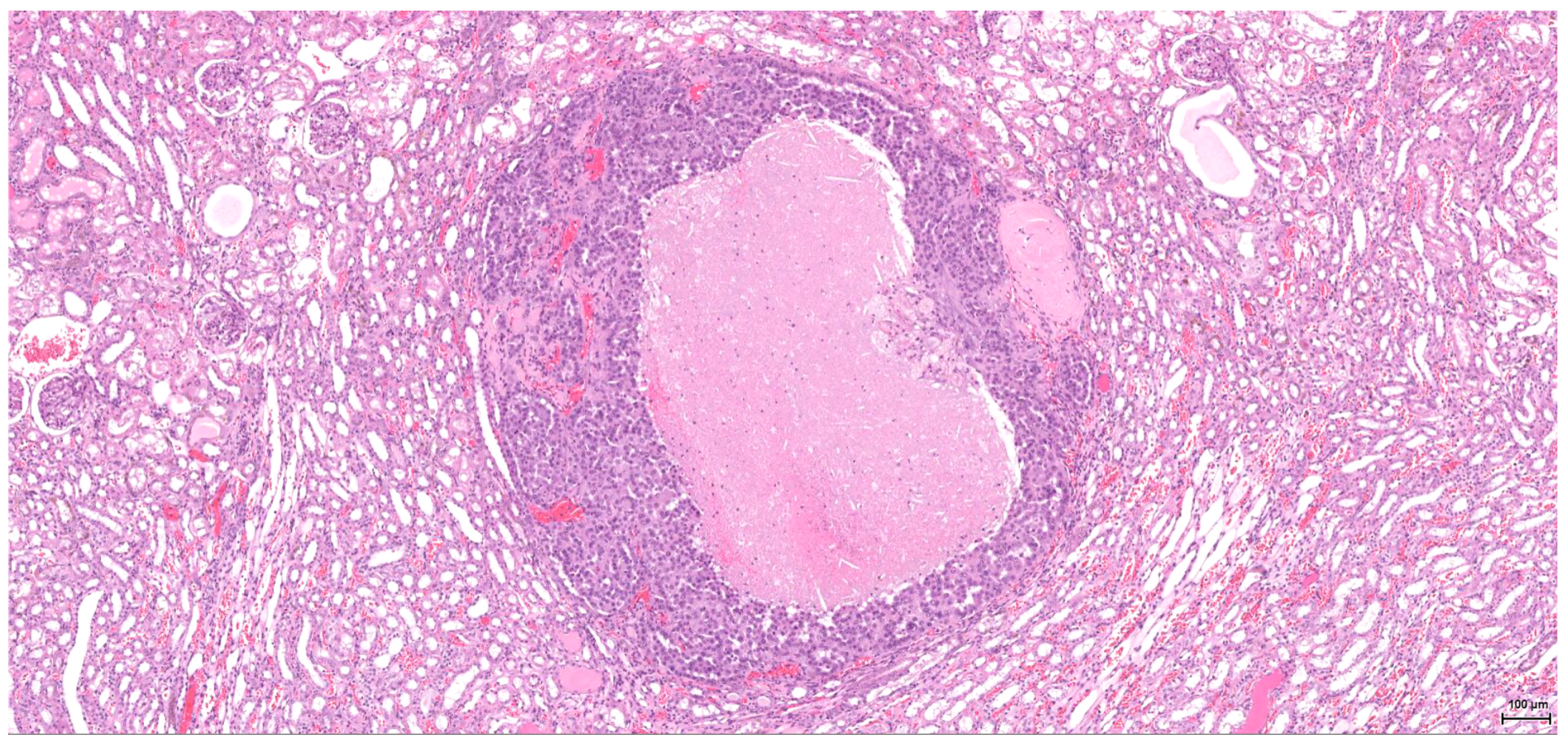
These poorly differentiated tumours have a highly aggressive course; they are usually very infiltrative, invading the surrounding local structures and metastasising at distant sites (liver, lung, mediastinum). The metastases have similar tumour morphology and are confirmed by the same immune profile. Consistently, all tumours at sites distant from a tumorous kidney were shown to have IHC profiles corresponding to the primary kidney neoplasm, confirming their metastatic status. This demonstrates the near-exclusive relationship between OTA and kidney concerning carcinogenicity in the rat, even after many months of exposure to the toxin.
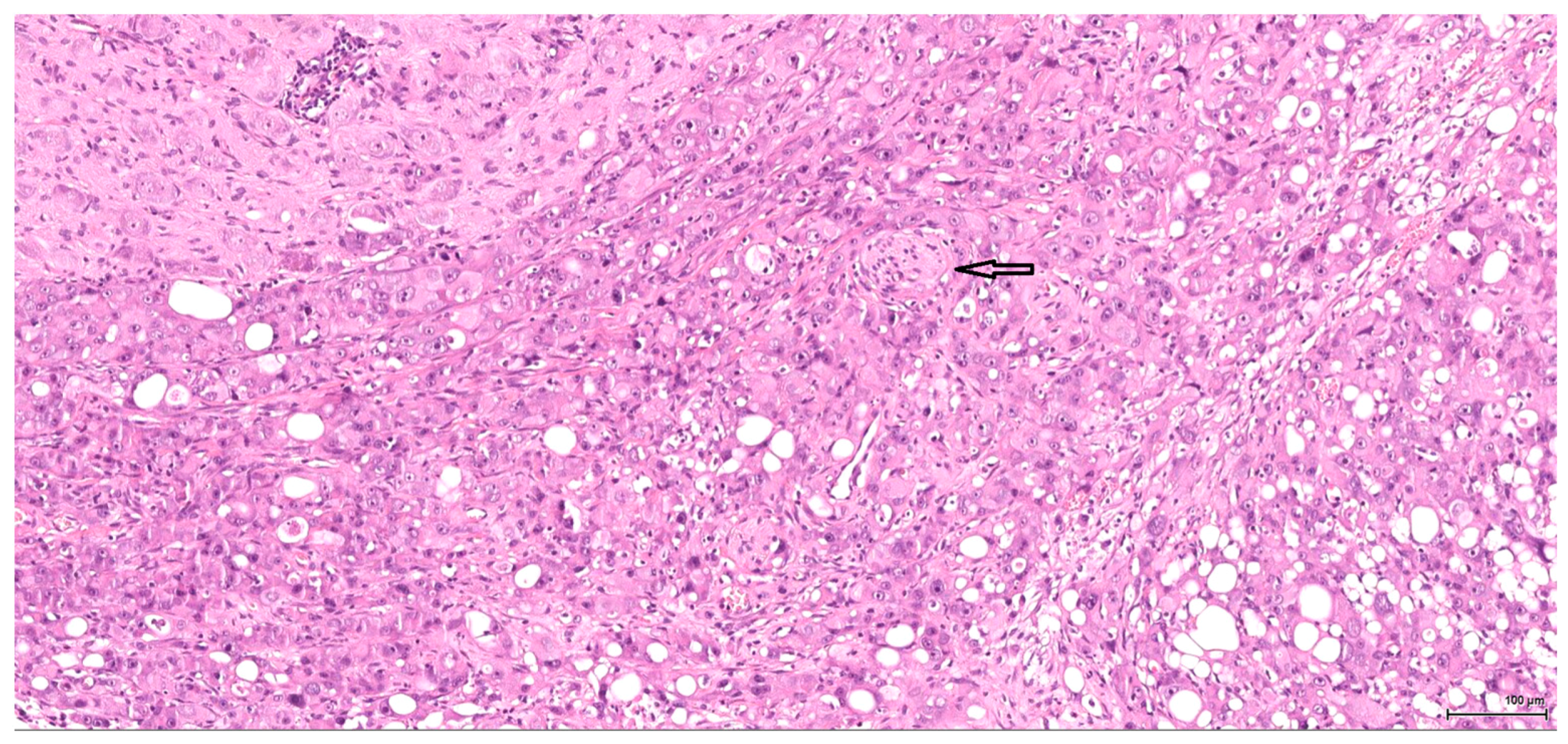
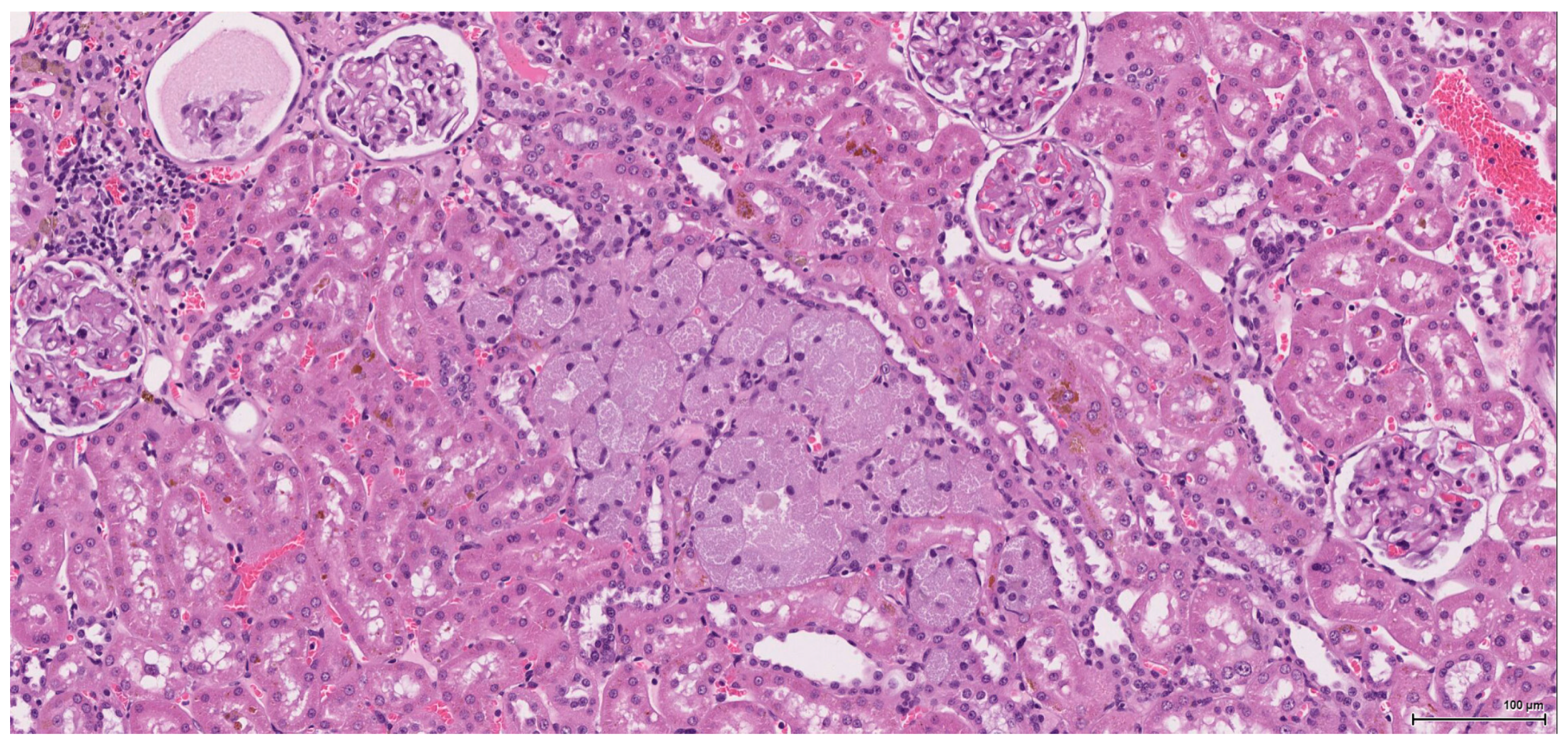
Dysplasia of tubular epithelium is probably a biologic precursor of at least some RCC, and it is also a strong support for the renal cell origin. In this study it was noticed within the adjacent renal parenchyma in four cases, ranging from mild to severe intraepithelial cytological atypia, similar to carcinoma in situ; interestingly, this lesion has been found occasionally in the contralateral kidney as well.
Premalignant precursor lesions of RCC are not well characterized, but intratubular epithelial dysplasia defined morphologically by crowded, enlarged and pleomorphic nuclei has been described by other authors [
18]. However, this finding remains understudied and outside the current recommendations for the pathological reporting of RCC. It warrants further study that will provide new insights into the pathogenesis, biologic behaviour, and natural history of RCC. In the present context it is worth mentioning that even the most subtle morphological changes (easily missed on H&E stain) could be potentially spotted by ribosomal p-S6 protein immunohistochemical expression, as demonstrated by one of our previous studies [
19]. Karyomegaly is presumed to result from nucleic acid replication without nuclear division. It is characterised by marked enlargement of nuclei observed predominantly in the proximal convoluted tubule cells and has been associated with chemical administration [
20]. Karyomegaly per se is considered a preneoplastic lesion and should be graded and reported. In our study karyomegaly, regarded as a dysplastic intraepithelial lesion, is a widespread change found in all cases, both male and female, and can be graded from mild to severe. The localisation is similar to that of the preneoplastic lesion below; sometimes one can follow a preneoplastic pathway, so to speak, along one collecting duct from the main tumour through the entire papilla.
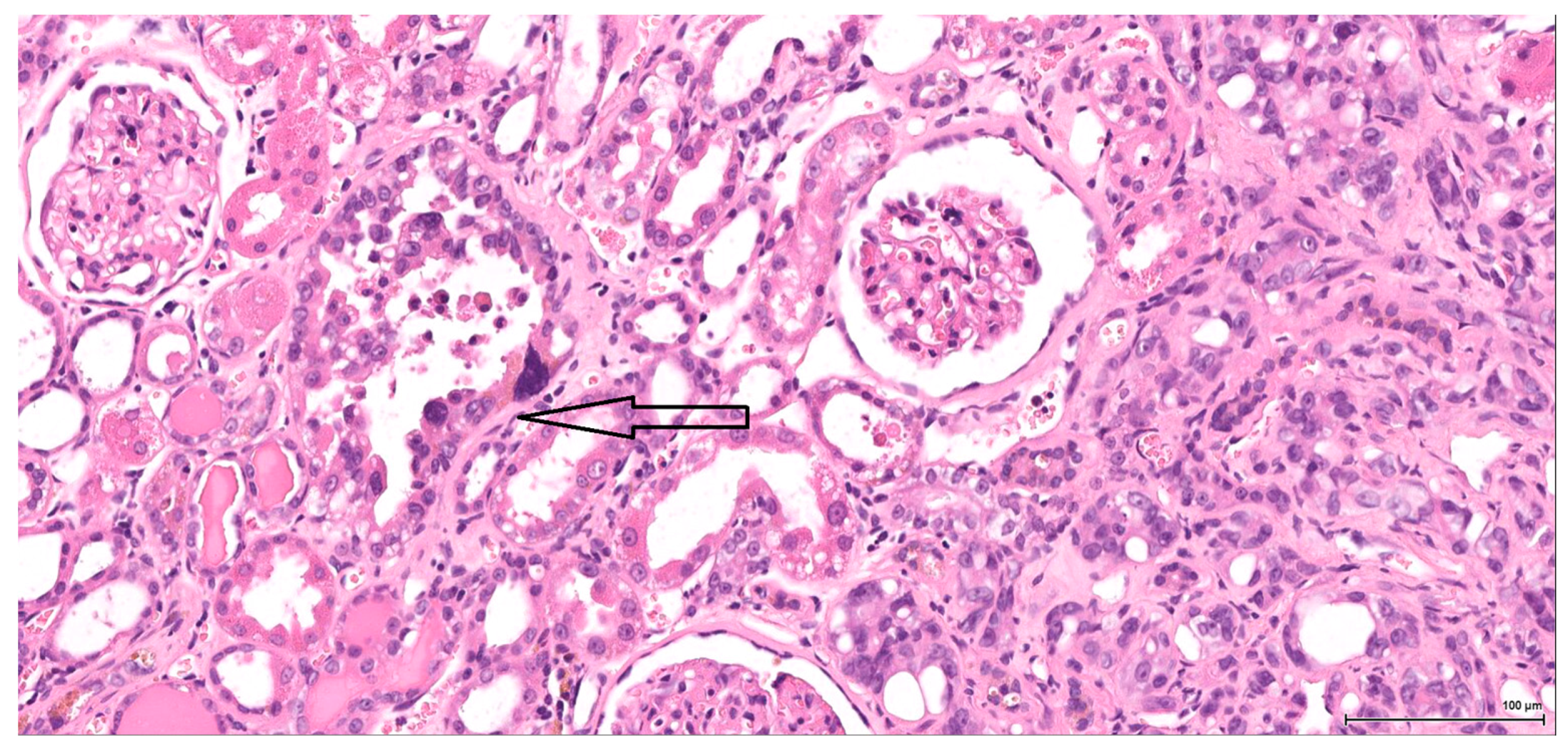
Atypical tubule hyperplasia is a tubule-confined epithelial proliferation of more than two to three cell layers, with variable degrees of cytoplasmic and nuclear pleiomorphism. It may occur spontaneously, in association with chronic progressive nephropathy, or as a result of chemical administration. Atypical tubule hyperplasia usually arises from the proximal convoluted tubule, but a distal or collecting duct origin is also possible. It is considered a putative preneoplastic lesion that is part of the continuum, leading to neoplasia [
20]. In our study, this proliferative (intra)tubular precursor lesion is seen either at the periphery of the tumour mass or as a minute lesion (0.1 mm) at a distant site.
Although only half of the elderly females in which renal tumour was recorded in the NTP study [
4] showed similar evidence in the sections provided for the present study, there is no cause to modify tumour incidence data. The small size of most neoplasms will readily affect ease of recognition in different section planes. Nevertheless, the present review confirms the marked rat gender difference in sensitivity to OTA as a renal carcinogen, and raises questions about the factor(s) affecting the limited proliferation of tumours in a female’s kidney during two years of gavage delivery of OTA. Nevertheless, occasional microscopic lesions in renal parenchyma, identified as ‘atypical tubule hyperplasia’, but not distorting the kidney, were probably not clinically significant for those animals.
Concerning the gender differential in the OTA dose-response, the perception of a mechanism involving OTA binding to male-specific rat (and presumably mouse) serum alpha 2u-globulins and augmented delivery to nephron epithelia via glomerular filtration, as in alpha 2u-nephropathy, has been described [
14]. It could make only the female rat dose-response renal carcinogenicity data potentially applicable to humans, since humans do not have these globulins. Unfortunately, exclusive temporary access to a commercial OTA primary antibody for that study has precluded subsequent confirmation.
The relevance of some experimental whole rat data to humans may be uncertain as a model, but for OTA the rat has been the best available. Studies on putative molecular tumorigenic mechanisms have in recent years used the sophisticated methodologies that have become available. However, no clear relevant picture has emerged to explain how tumour occurrence is mainly unilateral with highly focal origin in just a few of the thousands of separate nephrons, and that requires the insults from the ingestion of the mycotoxin well into the µg/kg b.w. range during most of the first year of life. OTA has been administered experimentally in vitro to tissue-cultured cells or kidney homogenates without knowledge of the relevant kidney epithelial cell exposure to OTA during in vivo tumorigenesis, because that is unknown. Previously, an analysis of several London OTA/rat carcinomas did not identify evidence of mutations in the rat orthologues of human familial renal cell carcinoma genes VHL and FLCN (Ricketts and Maher, personal communication), and genome-wide genetic sequencing studies are currently in progress. From the present immunohistochemical review of tumour heterogeneity a direct model for humans is uncertain, especially since a better affinity with spontaneous renal tumours arising in the Eker rat has recently been recognised also immunohistochemically [
20]. A comprehensive IHC study of all archived OTA/rat tumours from London dietary exposure experiments now seems desirable to quantify the spectrum of histopathologies within and across their experimental occurrence. This might be augmented from the NTP Archives, comparing its oral gavage delivery of OTA with the dietary protocol used in London. However, there remains the mysterious aetiology of complex histopathological findings in the bizarre NTP female rat studied here, set in the context of the markedly lower sensitivity of female rats and mice to OTA renal carcinogenicity. We also recommend preparing male mouse renal tumours, based on [
3], for full genetic analysis of an animal with a defined genome and using less of the expensive OTA.
Therefore, a provisional conclusion of the present pilot study is that the rat fails to provide any model support for an aetiological role for OTA in urothelial cancers sometimes associated with Balkan endemic nephropathy. Further, a predictive animal-model role for the rat in human RCC aetiology seems at present insecure. Notably in the particular case of the late Palle Krogh, whose premature demise from a metastasising RCC might have come from professional experimental exposure to OTA for several years, the primary renal tumour was diploid [
21] in marked histopathological contrast with the aneuploidy of OTA/rat carcinomas [
22]. This makes an aetiological connection between experimental rat and natural human difficult for OTA, particularly since experiment needs a vastly greater dosage of the mycotoxin than is naturally available.