Unravelling the Diversity of the Cyclopiazonic Acid Family of Mycotoxins in Aspergillus flavus by UHPLC Triple-TOF HRMS
Abstract
:1. Introduction
2. Results
2.1. Identification of Indole Cyclopiazonic Acid (CPA)-Type Derivatives
2.2. Identification of Oxindole CPA-Type Derivatives
2.3. Identification of Previously Unreported CPA-Type Derivatives
2.4. Screening of CPA-Type Alkaloids in Different A. flavus Strains
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Chemicals and Materials
5.2. Strains and Growth Conditions
5.3. Sample Preparation
5.4. UHPLC-qTOF-MS Analysis
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Holzapfel, C.W. The isolation and structure of cyclopiazonic acid, a toxic metabolite of Penicillium cyclopium Westling. Tetrahedron 1968, 24, 2101–2119. [Google Scholar] [CrossRef]
- Ohmomo, S.; Sugita, M.; Abe, M. Isolation of cyclopiazonic acid, cyclopiazonic acid imine and bissecodehydrocyclopiazonic acid from the cultures of Aspergillus versicolor (Vuill.) Tiraboshi. J. Agric. Chem. Soc. 1973, 47, 57–63. [Google Scholar]
- Luk, K.C.; Kobbe, B.; Townsend, J.M. Production of cyclopiazonic acid by Aspergillus flavus Link. Appl. Environ. Microbiol. 1977, 33, 211–212. [Google Scholar] [PubMed]
- Burdock, G.A.; Flamm, W.G. Review Article: Safety assessment of the mycotoxin cyclopiazonic acid. Int. J. Toxicol. 2000, 19, 195–218. [Google Scholar] [CrossRef]
- Frisvad, J.C. The connection between the Penicillia and Aspergillus and mycotoxins with special emphasis on misidentified isolates. Arch. Environ. Contam. Toxicol. 1989, 18, 452–467. [Google Scholar] [CrossRef] [PubMed]
- El-Banna, A.A.; Pitt, J.I.; Leistner, L. Production of mycotoxins by Penicillium species. Syst. Appl. Microbiol. 1987, 1, 42–46. [Google Scholar] [CrossRef]
- Dorner, J.W. Production of cyclopiazonic acid by Aspergillus tamarii Kita. Appl. Environ. Microbiol. 1983, 46, 1435–1437. [Google Scholar] [PubMed]
- Liu, X.; Walsh, C.T. Cyclopiazonic acid biosynthesis in Aspergillus sp.: Characterization of a reductase-like R* domain in cyclopiazonate synthetase that forms and releases cyclo-acetoacetyl-l-tryptophan. Biochemistry 2009, 48, 8746–8757. [Google Scholar] [CrossRef] [PubMed]
- Seshime, Y.; Juvvadi, P.R.; Tokuoka, M.; Koyama, Y.; Kitamoto, K.; Ebizuka, Y.; Fujii, I. Functional expression of the Aspergillus flavus PKS–NRPS hybrid CpaA involved in the biosynthesis of cyclopiazonic acid. Bioorg. Med. Chem. Lett. 2009, 19, 3288–3292. [Google Scholar] [CrossRef] [PubMed]
- Chang, P.K.; Ehrlich, K.C.; Fujii, I. Cyclopiazonic acid biosynthesis of Aspergillus flavus and Aspergillus oryzae. Toxins 2009, 1, 74–99. [Google Scholar] [CrossRef] [PubMed]
- Chang, P.K.; Ehrlich, K.C. Cyclopiazonic acid biosynthesis by Aspergillus flavus. Toxin Rev. 2011, 30, 79–89. [Google Scholar] [CrossRef]
- Seidler, N.W.; Jonaz, I.; Vegh, M.; Martonosi, A. Cyclopiazonic Acid is a specific inhibitor of the Ca2+-ATPase of sarcoplasmic reticulum. J. Biol. Chem. 1989, 264, 17816–17823. [Google Scholar] [PubMed]
- Lytton, J.; Westlin, M.; Hanleyll, M.R. Thapsigargin inhibits the sarcoplasmic or endoplasmic reticulum Ca-ATPase family of calcium pumps. J. Biol. Chem. 1991, 266, 17067–17071. [Google Scholar] [PubMed]
- Purchase, I.F. The acute toxicity of the mycotoxin cyclopiazonic acid to rats. Toxicol. Appl. Pharmacol. 1971, 18, 114–123. [Google Scholar] [CrossRef]
- Norred, W.P.; Morrissey, R.E.; Rilley, R.T.; Cole, R.J.; Dorner, J.W. Distribution, excretion, and skeletal muscle effects of the mycotoxin [14C]cyclopiazonic acid in rats. Food Chem. Toxicol. 1985, 23, 1069–1076. [Google Scholar] [CrossRef]
- Nishie, K.; Cole, R.J.; Dorner, J.W. Toxicity and neuropharmacology of cyclopiazonic acid. Food Chem. Toxicol. 1985, 23, 831–839. [Google Scholar] [CrossRef]
- Nishie, K.; Cole, R.J.; Dorner, J.W. Toxic effects of cyclopiazonic acid in the early phase of pregnancy in mice. Res. Commun. Chem. Pathol. Pharmacol. 1987, 55, 303–315. [Google Scholar] [PubMed]
- Antony, M.; Shukla, Y.; Janardhanan, K.K. Potential risk of acute hepatotoxicity of kodo poisoning due to exposure to cyclopiazonic acid. J. Ethnopharmacol. 2003, 87, 211–214. [Google Scholar] [CrossRef]
- Njobeh, P.B.; Dutton, M.F.; Koch, S.H.; Chuturgoon, A.; Stoev, S.; Seifert, K. Contamination with storage fungi of human food from Cameroon. Int. J. Food Microbiol. 2009, 135, 193–198. [Google Scholar] [CrossRef] [PubMed]
- Finoli, C.; Vecchio, A.; Galli, A.; Franzetti, L. Production of cyclopiazonic acid by molds isolated from Taleggio cheese. J. Food Prot. 1999, 62, 1198–1202. [Google Scholar] [CrossRef] [PubMed]
- Sosa, M.J.; Cordoba, J.J.; Diaz, C.; Rodriguez, M.; Bermudez, E.; Asensio, M.A.; Nunez, F. Production of cyclopiazonic acid by Penicillium commune isolated from dry-cured ham on ameatextract-based substrate. J. Food. Prot. 2002, 65, 988–992. [Google Scholar]
- Vaamonde, G.; Patriarca, A.; Fernandez Pinto, V.; Comerio, R.; Degrossi, C. Variability of aflatoxin and cyclopiazonic acid production by Aspergillus section Flavi from different substrates in Argentina. Int. J. Food Microbiol. 2003, 88, 79–84. [Google Scholar] [CrossRef]
- Dorner, J.W.; Cole, R.J.; Erlington, D.J.; Suksupath, S.; McDowell, G.H.; Bryden, W.L. Cyclopiazonic acid residues in milk and eggs. J. Agric. Food Chem. 1994, 42, 1516–1518. [Google Scholar] [CrossRef]
- Lee, Y.J.; Hagler, W.M.J. Aflatoxin and cyclopiazonic acid production by Aspergillus flavus isolated from contaminated maize. J. Food Sci. 1991, 56, 871–872. [Google Scholar] [CrossRef]
- Urano, T.; Trucksess, M.W.; Beaver, R.W.; Wilson, D.M.; Dorner, J.W.; Dowell, F.E. Co-occurrence of cyclopiazonic acid and aflatoxins in corn and peanuts. J. Off. Anal. Chem. Int. 1992, 75, 838–841. [Google Scholar]
- Heperkan, D.; Somuncuoglu, S.; Karbancioglu-Güler, F.; Mecik, N. Natural contamination of cyclopiazonic acid in dried figs and co-occurrence of aflatoxin. Food Control 2012, 23, 82–86. [Google Scholar] [CrossRef]
- Zorzete, P.; Baquiao, A.C.; Atayde, D.D.; Reis, T.A.; Goncalez, E.; Correa, B. Mycobiota, aflatoxins and cyclopiazonic acid in stored peanut cultivars. Food Res. Int. 2013, 52, 380–386. [Google Scholar] [CrossRef]
- Ezekiel, C.N.; Sulyok, M.; Somorin, Y.; Odutayo, F.I.; Nwabekee, S.U.; Balogun, A.T.; Krska, R. Mould and mycotoxin exposure assessment of melon and bush mango seeds, two common soup thickeners consumed in Nigeria. Int. J. Food Microbiol. 2016, 237, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Holzapfel, C.W.; Hutchinson, R.D.; Wilkins, D.C. The isolation and structure of two new indole derivatives from Penicillium cyclopium Westling. Tetrahedron 1970, 26, 5239–5246. [Google Scholar] [CrossRef]
- Lin, A.Q.; Lin, D.; Fang, Y.C.; Wang, F.Z.; Zhu, T.J.; Gu, Q.Q.; Zhu, W.M. iso-α-Cyclopiazonic acid, a new natural product isolated from the marine-derived fungus Aspergillus flavus C-F-3. Chem. Nat. Compd. 2009, 45, 677–680. [Google Scholar] [CrossRef]
- Lan, W.J.; Wang, K.T.; Xu, M.Y.; Zhang, J.J.; Lam, C.K.; Zhong, G.H.; Xu, J.; Yang, D.P.; Li, H.J.; Wang, L.Y. Secondary metabolites with chemical diversity from the marine-derived fungus Pseudallescheria boydii F19-1 and their cytotoxic activity. RSC Adv. 2016, 6, 76206–76213. [Google Scholar] [CrossRef]
- Kato, N.; Tokuoka, M.; Shinohara, Y.; Kawatani, M.; Uramoto, M.; Seshime, Y.; Fujii, I.; Kitamoto, K.; Takahashi, T.; Takahashi, S.; et al. Genetic safeguard against mycotoxin cyclopiazonic acid production in Aspergillus oryzae. ChemBioChem 2011, 12, 1376–1382. [Google Scholar] [CrossRef] [PubMed]
- Tokuoka, M.; Kikuchi, T.; Shinohara, Y.; Koyama, A.; Iio, S.; Kubota, T.; Kobayashi, J.; Koyama, Y.; Totsuka, A.; Shindo, H.; et al. Cyclopiazonic acid biosynthesis gene cluster gene cpaM is required for speradine A biosynthesis. Biosci. Biotechnol. Biochem. 2015, 79, 2081–2085. [Google Scholar] [CrossRef] [PubMed]
- Tsuda, M.; Mugishima, T.; Komatsu, K.; Sone, T.; Tanaka, M.; Mikami, Y.; Shiro, M.; Hirai, M.; Ohizumie, Y.; Kobayashi, J. Speradine A, a new pentacyclic oxindole alkaloid from a marine-derived fungus Aspergillus tamarii. Tetrahedron 2003, 59, 3227–3230. [Google Scholar] [CrossRef]
- Wang, N.; Hu, J.C.; Liu, W.; Wang, S.J. Cyclopiazonic Acid Compound, and Preparation and Application Thereof. CN Patent 103,183,666 A, 3 July 2013. [Google Scholar]
- Hu, X.; Xia, Q.W.; Zhao, Y.Y.; Zheng, Q.H.; Liu, Q.Y.; Chen, L.; Zhang, Q.Q. Speradines B-E, four novel tetracyclic oxindole alkaloids from the marine-derivied fungus Aspergillus oryzae. Heterocycles 2014, 89, 1662–1669. [Google Scholar]
- Hu, X.; Xia, Q.W.; Zhao, Y.Y.; Zheng, Q.H.; Liu, Q.Y.; Chen, L.; Zhang, Q.Q. Speradines F-H, three new oxindole alkaloids from the marine-derived fungus Aspergillus oryzae. Chem. Pharm. Bull. 2014, 62, 942–946. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Chena, C.; Wang, J.; Li, X.N.; Wei, G.; Guo, Y.; Yao, G.; Luo, Z.; Zhang, J.; Xue, Y.; et al. Penicamedine A, a highly oxygenated hexacyclic indole alkaloid from Penicillium camemberti. Chem. Biodivers. 2015, 12, 1547–1553. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Peng, J.; Wu, G.; Zhu, T.; Li, G.; Gu, Q.; Li, D. Speradines B-H, oxygenated cyclopiazonic acid alkaloids from the sponge-derived fungus Aspergillus flavus MXH-X104. Tetrahedron 2015, 71, 3522–3527. [Google Scholar] [CrossRef]
- Zhou, M.; Miao, M.M.; Du, G.; Li, X.N.; Shang, S.Z.; Zhao, W.; Lu, Z.H.; Yang, G.Y.; Che, C.T.; Hu, Q.F.; et al. Aspergillines A-E, highly oxygenated hexacyclic indole-tetrahydrofuran-tetramic acid derivatives from Aspergillus versicolor. Org. Lett. 2014, 16, 5016–5019. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Zhang, X.; Nong, X.; Wei, X.; Qi, S. Oxindole alkaloids from the fungus Penicillium commune DFFSCS026 isolated from deep-sea-derived sediments. Tetrahedron 2015, 71, 610–615. [Google Scholar] [CrossRef]
- Holzapfel, C.W.; Bredenkamp, M.W.; Snyman, R.M.; Boeyens, J.C.A.; Allen, C.C. Cyclopiamide, an isoindolo[4,6-cd]indole from Penicillium cyclopium. Phytochemistry 1990, 29, 639–642. [Google Scholar] [CrossRef]
- Abbas, H.K.; Zablotowicz, R.M.; Horn, B.W.; Phillips, N.A.; Johnson, B.J.; Jin, X.; Abel, C.A. Comparison of major biocontrol strains of non-aflatoxigenic Aspergillus flavus for the reduction of aflatoxins and cyclopiazonic acid in maize. Food Addit. Contam. 2011, 28, 198–208. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, K.F.; Mansson, M.; Rank, C.; Frisvad, J.C.; Larsen, T.O. Dereplication of microbial natural products by LC-DAD-TOFMS. J. Nat. Prod. 2011, 74, 2338–2348. [Google Scholar] [CrossRef] [PubMed]
- Losito, I.; Monaci, L.; Aresta, A.; Zambonin, C.G. LC-ion trap electrospray MS-MS for the determination of cyclopiazonic acid in milk samples. Analyst 2002, 127, 499–502. [Google Scholar] [CrossRef] [PubMed]
- Moldes-Anaya, A.S.; Asp, T.N.; Eriksen, G.S.; Skaar, I.; Rundberget, T. Determination of cyclopiazonic acid in food and feeds by liquid chromatography-tandem mass spectrometry. J. Chromatogr. A 2009, 1216, 3812–3818. [Google Scholar] [CrossRef] [PubMed]
- Klitgaard, A.; Iversen, A.; Andersen, M.R.; Larsen, T.O.; Frisvad, J.C.; Nielsen, K.F. Aggressive dereplication using UHPLC-DAD-QTOF: Screening extracts for up to 3000 fungal secondary metabolites. Anal. Bioanal. Chem. 2014, 406, 1933–1943. [Google Scholar] [CrossRef] [PubMed]
- Arroyo-Manzanares, N.; Diana di Mavungu, J.; Uka, V.; Gámiz-Gracia, L.; García-Campaña, A.M.; de Saeger, S. An integrated targeted and untargeted approach for the analysis of ergot alkaloids in cereals using UHPLC-hybrid quadrupole time-of-flight mass spectrometry. World Mycotoxin J. 2015, 8, 653–666. [Google Scholar] [CrossRef]
- Boettger, D.; Hertweck, C. Molecular diversity sculpted by fungal PKS-NRPS hybrids. ChemBioChem 2013, 14, 28–42. [Google Scholar] [CrossRef] [PubMed]
- Cary, J.W.; Uka, V.; Han, Z.; Buyst, D.; Harris-Coward, P.Y.; Ehrlich, K.C.; Wei, Q.; Bhatnagar, D.; Dowd, P.F.; Martens, S.L.; et al. An Aspergillus flavus secondary metabolic gene cluster containing a hybrid PKS-NRPS is necessary for synthesis of the 2-pyridones, leporins. Fungal Genet. Biol. 2015, 81, 88–97. [Google Scholar] [CrossRef] [PubMed]
- Arroyo-Manzanares, N.; Diana di Mavungu, J.; Uka, V.; Malysheva, S.V.; Cary, J.W.; Ehrlich, K.C.; Vanhaecke, L.; Bhatnagar, D.; de Saeger, S. Use of UHPLC high-resolution Orbitrap mass spectrometry to investigate the genes involved in the production of secondary metabolites in Aspergillus flavus. Food Addit. Contam. Part A 2015, 32, 1656–1673. [Google Scholar] [CrossRef] [PubMed]
- Royles, B.J.L. Naturally Occurring Tetramic Acids: Structure, Isolation, and Synthesis. Chem. Rev. 1995, 95, 1981–2001. [Google Scholar] [CrossRef]
- Spatz, J.H.; Welsch, S.J.; Duhaut, D.; Jäger, N.; Boursier, T.; Fredrich, M.; Allmendinger, L.; Ross, G.; Kolb, J.; Burdack, C.; et al. Tetramic acid derivatives via Ugi–Dieckmann reaction. Tetrahedron Lett. 2009, 50, 1705–1707. [Google Scholar] [CrossRef]
- Yang, Y.L.; Lu, C.P.; Chen, M.Y.; Chen, K.Y.; Wu, Y.C.; Wu, S.H. Cytotoxic polyketides containing tetramic acid moieties isolated from the fungus Myceliophthora thermophila: Elucidation of the relationship between cytotoxicity and stereoconfiguration. Chemistry 2007, 13, 6985–6991. [Google Scholar] [CrossRef] [PubMed]
- Abbas, H.K.; Wilkinson, J.R.; Zablotowicz, R.M.; Accinelli, C.; Abel, C.A.; Bruns, H.A.; Weaver, M.A. Ecology of Aspergillus flavus, regulation of aflatoxin production, and management strategies to reduce aflatoxin contamination of corn. Toxin Rev. 2009, 28, 142–153. [Google Scholar] [CrossRef]
- Ehrlich, K.C. Non-aflatoxigenic Aspergillus flavus to prevent aflatoxin contamination in crops: Advantages and limitations. Front. Microbiol. 2014, 5, 50. [Google Scholar] [CrossRef] [PubMed]
- Moore, G.G. Sex and recombination in aflatoxigenic Aspergilli: Global implications. Front. Microbiol. 2014, 5, 32. [Google Scholar] [CrossRef] [PubMed]
- Olarte, R.A.; Horn, B.W.; Dorner, J.W.; Monacell, J.T.; Singh, R.; Stone, E.A.; Carbone, I. Effect of sexual recombination on population diversity in aflatoxin production by Aspergillus flavus. Phytopathology 2012, 103, 8. [Google Scholar]
- Couto, S.R.; Sanroma, M.A. Application of solid-state fermentation to food industry-A review. J. Food Eng. 2006, 76, 291–302. [Google Scholar] [CrossRef]
- Ehrlich, K.C.; Cotty, P.J. An isolate of Aspergillus flavus used to reduce aflatoxin contamination in cottonseed has a defective polyketide synthase gene. Appl. Microbiol. Biotech. 2004, 65, 473–478. [Google Scholar] [CrossRef] [PubMed]
- Chang, P.K.; Horn, B.W.; Dorner, J.W. Sequence breakpoints in the aflatoxin biosynthesis gene cluster and flanking regions in nonaflatoxigenic Aspergillus flavus isolates. Fungal Genet. Biol. 2005, 42, 914–923. [Google Scholar] [CrossRef] [PubMed]
- Olarte, R.A.; Horn, B.W.; Dorner, J.W.; Monacell, J.T.; Singh, R.; Stone, E.A.; Carbone, I. Effect of sexual recombination on population diversity in aflatoxin production by Aspergillus flavus and evidence for cryptic heterokaryosis. Mol. Ecol. 2012, 21, 1453–1476. [Google Scholar] [CrossRef] [PubMed]
- Moore, G.G.; Singh, R.; Horn, B.W.; Carbone, I. Recombination and lineage-specific gene loss in the aflatoxin gene cluster of Aspergillus flavus. Mol. Ecol. 2009, 18, 4870–4887. [Google Scholar] [CrossRef] [PubMed]
- Nevzglyadova, O.V.; Gaivoronskii, A.A.; Artemov, A.V.; Smirnova, T.I.; Soidla, T.R. Detection of concealed “illegitimate” nuclei in tetrad analysis of the diploid progeny of heterokaryons in Saccharomyces cerevisiae. Russian J. Genet. 2001, 37, 617–623. [Google Scholar] [CrossRef]
- Vercauteren, A.; Boutet, X.; D’hondt, L.; van Bockstaele, E.; Maes, M.; Leus, L.; Chandelier, A.; Heungens, K. Aberrant genome size and instability of Phytophthora ramorum oospore progenies. Fungal Genet. Biol. 2011, 48, 537–543. [Google Scholar] [CrossRef] [PubMed]
- Kotsubei, A.; Gorgel, M.; Morth, J.P.; Nissen, P.; Andersen, J.L. Probing determinants of cyclopiazonic acid sensitivity of bacterial Ca2+-ATPases. FEBS J. 2013, 280, 5441–5449. [Google Scholar] [CrossRef] [PubMed]
- Cui, R.; Wang, Y.; Wang, L.; Li, G.; Lan, K.; Altmeyer, R.; Zou, G. Cyclopiazonic acid, an inhibitor of calcium-dependent ATPases withantiviral activity against human respiratory syncytial virus. Antivir. Res. 2015, 132, 38–45. [Google Scholar] [CrossRef] [PubMed]
- Kistamas, K.; Szentandrassy, N.; Hegyi, B.; Vaczi, K.; Ruzsnavszky, F.; Horvath, B.; Banyasz, T.; Nanasi, P.P.; Magyar, J. Changes in intracellular calcium concentration influence beat-to-beat variability of action potential duration in canine ventricular myocytes. J. Physiol. Pharmacol. 2015, 66, 73–81. [Google Scholar] [PubMed]
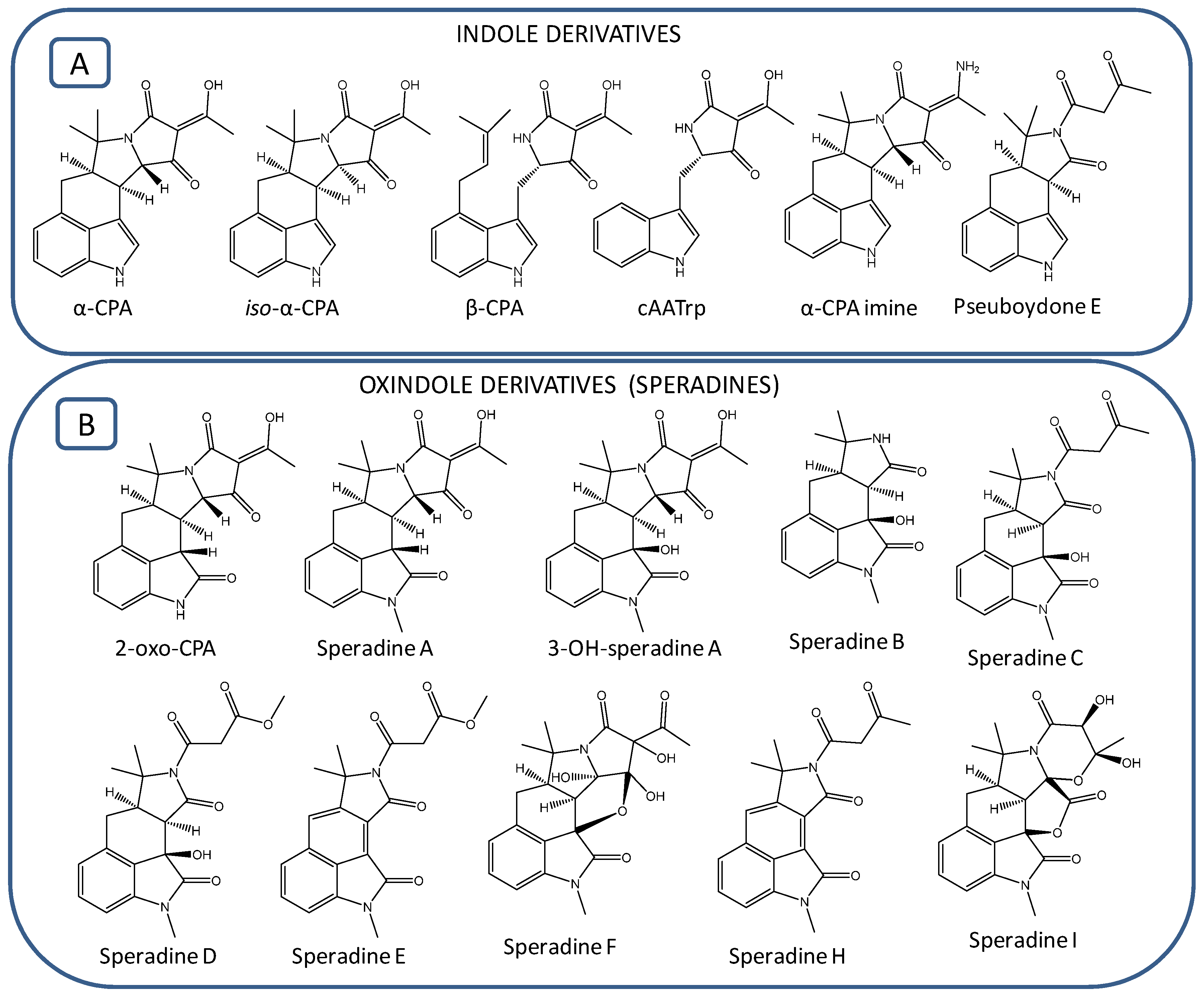

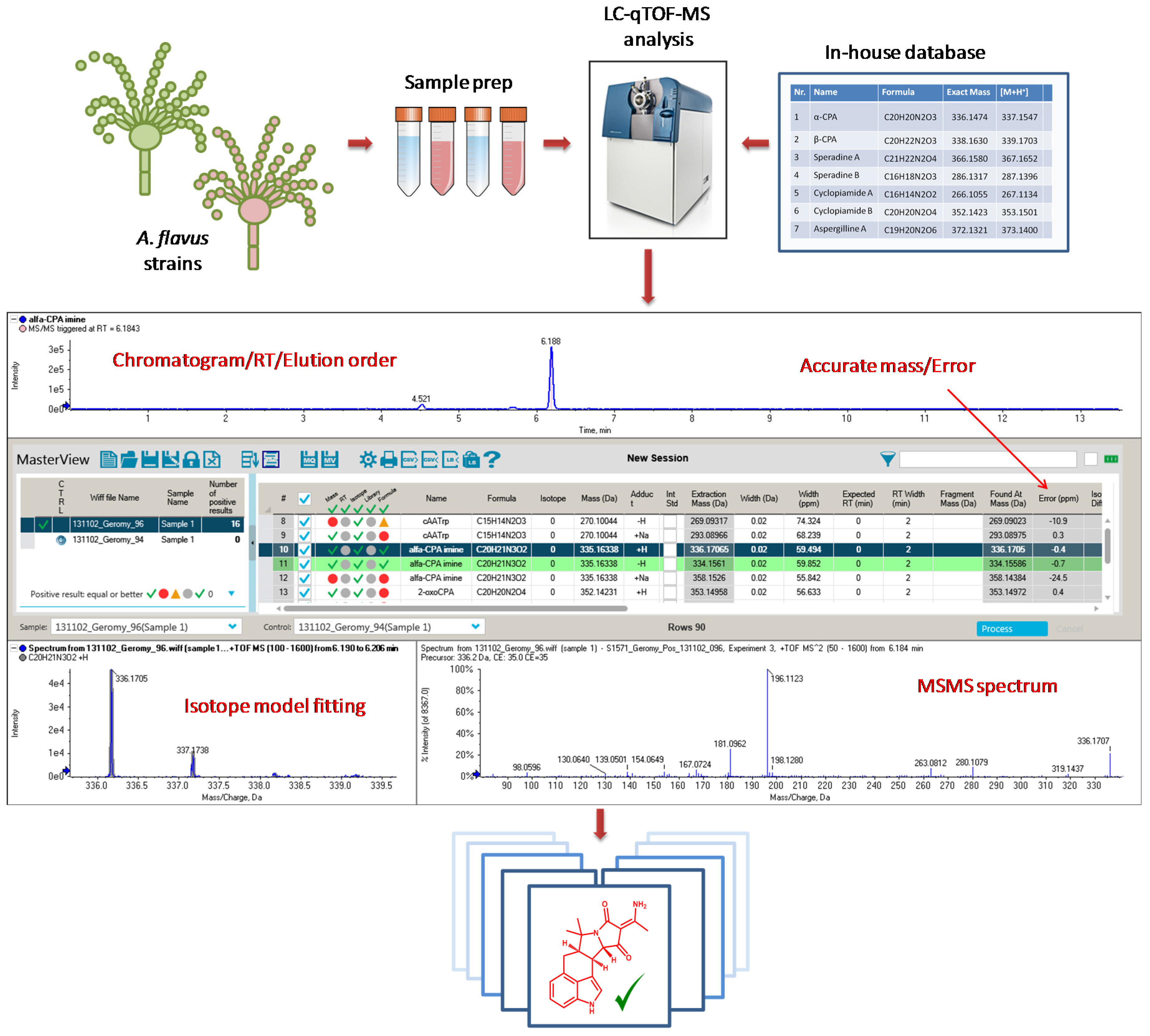

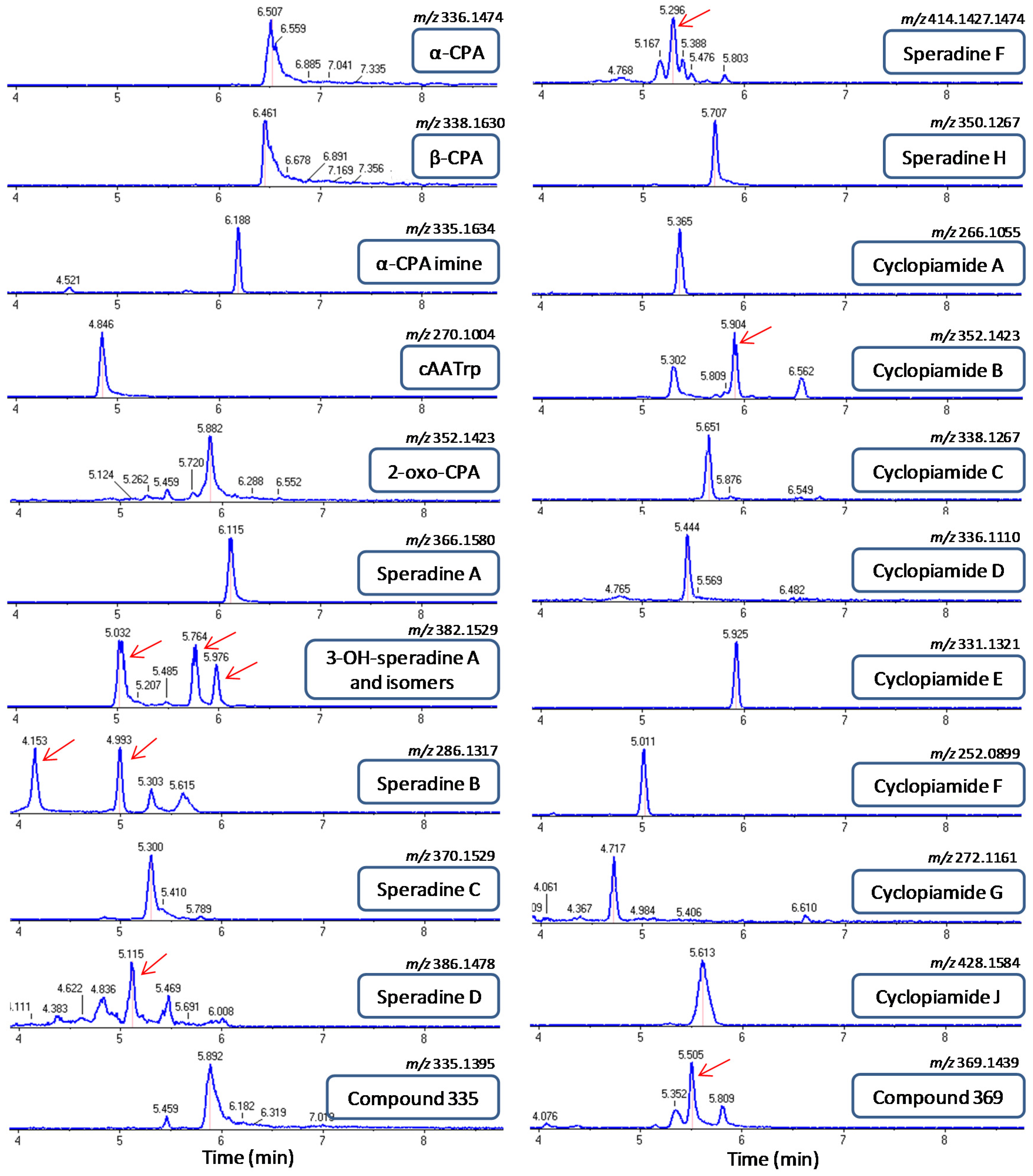
| Compound | Name | Formula | Exact Mass | Source | Reference |
|---|---|---|---|---|---|
| 1 | α-CPA | C20H20N2O3 | 336.1474 | Various Aspergillus and Penicillium species | [1,3,29] |
| 2 | Iso-α-CPA | C20H20N2O3 | 336.1474 | Aspergillus flavus | [30] |
| 3 | β-CPA | C20H22N2O3 | 338.1630 | Various Aspergillus and Penicillium species | [8,9,29] |
| 4 | cAATrp | C15H14N2O3 | 270.1004 | Various Aspergillus and Penicillium species | [8,9,10,11] |
| 5 | α-CPA imine | C20H21N3O2 | 335.1634 | Penicillium cyclopium | [29] |
| 6 | Pseuboydone E | C19H20N2O3 | 324.1474 | Pseudallescheria boydii | [31] |
| 7 | 2-oxoCPA | C20H20N2O4 | 352.1423 | Aspergillus oryzae | [32,33] |
| 8 | Speradine A | C21H22N2O4 | 366.1580 | A. tamarii, A. oryzae | [33,34] |
| 9 | 3-OH-speradine A | C21H22N2O5 | 382.1529 | A. tamarii | [35] |
| 10 | Speradine B | C16H18N2O3 | 286.1317 | A. oryzae, A. flavus | [36,39] |
| 11 | Speradine C | C20H22N2O5 | 370.1529 | A. oryzae, A. flavus | [36,39] |
| 12 | Speradine D | C20H22N2O6 | 386.1478 | A. oryzae | [36] |
| 13 | Speradine E | C20H18N2O5 | 366.1216 | A. oryzae | [36] |
| 14 | Speradine F (Pencamedine A) | C21H22N2O7 | 414.1427 | A. oryzae, P. commune, A. flavus, P. camemberti | [37,38,39,41] |
| 15 | Speradine H | C20H18N2O4 | 350.1267 | A. oryzae, P. commune | [37,41] |
| 16 | Speradine I | C21H22N2O7 | 414.1427 | A. flavus | [39] |
| 17 | Aspergilline A | C19H20N2O6 | 372.1321 | A. versicolor | [40] |
| 18 | Aspergilline B | C23H26N2O8 | 458.1689 | A. versicolor | [40] |
| 19 | Aspergilline C | C24H28N2O6 | 440.1947 | A. versicolor | [40] |
| 20 | Aspergilline D (Cyclopiamide I) | C21H24N2O7 | 416.1584 | A. versicolor, P. commune | [41,42] |
| 21 | Aspergilline E | C25H30N2O9 | 502.1951 | A. versicolor | [40] |
| 22 | Cyclopiamide A | C16H14N2O2 | 266.1055 | P. cyclopium, P. commune, A. flavus | [39,41,42] |
| 23 | Cyclopiamide B | C20H20N2O4 | 352.1423 | P. commune | [41] |
| 24 | Cyclopiamide C | C19H18N2O4 | 338.1267 | P. commune | [41] |
| 25 | Cyclopiamide D | C19H16N2O4 | 336.1110 | P. commune | [41] |
| 26 | Cyclopiamide E | C20H17N3O2 | 331.1321 | P. commune | [41] |
| 27 | Cyclopiamide F | C15H12N2O2 | 252.0899 | P. commune | [41] |
| 28 | Cyclopiamide G | C15H16N2O3 | 272.1161 | P. commune | [41] |
| 29 | Cyclopiamide H | C16H18N2O3 | 286.1317 | A. oryzae, P. commune, A. flavus | [36,39,41] |
| 30 | Cyclopiamide J | C22H24N2O7 | 428.1584 | P. commune | [41] |
| A. flavus Strains (SRRC) | CPA-Type Alkaloids | |||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| α-CPA | β-CPA | α-CPA Imine | cAATrp | 2-oxoCPA | Speradine A | 3-OH-Speradine A | Speradine B | Speradine C | Speradine D | Speradine F | Speradine H | Cyclopiamide A | Cyclopiamide B | Cyclopiamide C | Cyclopiamide D | Cyclopiamide E | Cyclopiamide F | Cyclopiamide G | Cyclopiamide J | Compound 335 | Compound 369 | |
| 0038 | ++ | ++ | ++ | + | ++ | + | - | - | - | - | ++ | - | - | - | ++ | + | - | + | + | - | ++ | ++ |
| 0141 | + | - | - | - | + | ++ | + | - | + | - | - | - | - | + | - | - | - | - | - | - | - | - |
| 0144 | +++ | ++ | - | ++ | ++ | + | + | - | - | - | - | - | - | ++ | ++ | + | - | + | + | - | ++ | ++ |
| 0150 | +++ | ++ | ++ | ++ | ++ | + | + | - | - | - | - | - | - | - | ++ | - | - | ++ | + | - | ++ | ++ |
| 0151 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| 0167 | +++ | ++ | + | ++ | ++ | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | ++ | ++ |
| 0283 | ++ | ++ | - | ++ | ++ | ++ | ++ | ++ | ++ | + | ++ | - | ++ | ++ | ++ | + | - | + | + | ++ | ++ | ++ |
| 0295 | ++ | ++ | - | ++ | ++ | ++ | +++ | ++ | ++ | + | ++ | ++ | ++ | ++ | + | + | ++ | + | + | ++ | ++ | ++ |
| 1000F | +++ | ++ | - | ++ | ++ | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | ++ | ++ |
| 1006 | ++ | ++ | ++ | ++ | + | ++ | ++ | - | - | - | - | - | - | + | + | + | - | + | - | - | ++ | ++ |
| 1020 | +++ | ++ | ++ | ++ | + | - | - | - | - | - | - | - | - | - | ++ | - | - | ++ | + | - | ++ | ++ |
| 1021 | ++ | ++ | + | + | ++ | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | ++ |
| 1055 | +++ | + | + | ++ | ++ | - | - | - | - | - | - | - | - | - | ++ | ++ | - | ++ | + | - | ++ | ++ |
| 1071 | ++ | ++ | - | - | + | - | - | - | - | - | - | - | - | - | + | - | - | + | - | - | + | + |
| 1098 | ++ | + | - | ++ | - | - | - | - | - | - | - | - | - | - | + | - | - | ++ | - | - | ++ | + |
| 1118 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| 1187 | ++ | ++ | - | ++ | ++ | - | - | - | - | - | - | - | - | - | ++ | - | - | - | - | - | ++ | ++ |
| 1299 | ++ | ++ | ++ | ++ | ++ | - | - | - | - | - | - | - | - | - | + | - | - | + | + | - | ++ | ++ |
| 1356 | ++ | - | ++ | ++ | ++ | ++ | - | - | - | - | - | - | - | - | + | - | - | ++ | + | - | ++ | ++ |
| 1357 | ++ | ++ | - | ++ | + | ++ | ++ | ++ | - | - | - | ++ | ++ | - | ++ | - | - | + | - | ++ | + | - |
| 1533 | ++ | ++ | - | ++ | ++ | - | - | - | - | - | - | - | - | - | ++ | - | - | + | + | - | ++ | ++ |
| 1534 | ++ | ++ | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - |
| 1540 | ++ | + | - | + | ++ | ++ | + | - | - | - | - | - | - | - | - | - | - | - | - | - | + | ++ |
| 1541 | ++ | - | - | ++ | - | - | - | - | - | - | - | - | - | - | ++ | - | - | - | - | - | - | - |
| 1543 | ++ | - | - | ++ | - | ++ | ++ | + | + | - | - | - | - | + | - | - | - | + | - | - | - | + |
| 1544 | ++ | ++ | - | ++ | ++ | ++ | ++ | ++ | ++ | ++ | ++ | ++ | ++ | ++ | + | - | ++ | ++ | - | ++ | ++ | ++ |
| 1545 | +++ | ++ | ++ | ++ | ++ | - | - | - | - | - | - | - | - | - | ++ | + | - | ++ | + | - | ++ | ++ |
| 1547 | ++ | ++ | - | ++ | - | ++ | ++ | ++ | ++ | - | ++ | - | ++ | ++ | - | - | - | - | - | ++ | ++ | - |
| 1552 | ++ | - | - | - | + | ++ | ++ | - | + | - | - | - | - | - | - | - | - | - | - | - | - | + |
| 1553 | ++ | + | + | + | ++ | - | - | - | - | - | ++ | - | - | - | ++ | - | - | ++ | + | - | ++ | ++ |
| 1554 | +++ | + | + | ++ | ++ | + | - | - | - | - | - | - | - | - | ++ | - | - | - | - | - | ++ | ++ |
| 1557 | ++ | ++ | - | ++ | + | ++ | ++ | ++ | ++ | - | ++ | ++ | ++ | ++ | - | - | ++ | - | - | ++ | ++ | - |
| 1558 | ++ | ++ | - | ++ | ++ | - | - | + | - | - | - | - | - | - | ++ | + | - | + | + | - | ++ | ++ |
| 1559 | +++ | ++ | + | ++ | ++ | - | - | - | - | - | - | - | - | - | ++ | - | - | ++ | + | - | ++ | ++ |
| 1565 | ++ | ++ | - | + | + | - | - | - | - | - | - | - | - | - | + | + | - | - | - | - | ++ | ++ |
| 1566 | +++ | ++ | - | + | ++ | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | ++ | ++ |
| 1568 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| 1571 | ++ | ++ | ++ | ++ | + | ++ | +++ | ++ | ++ | + | + | - | ++ | ++ | + | - | + | + | - | ++ | + | ++ |
| 1573 | +++ | ++ | - | + | + | - | - | - | - | - | - | - | - | - | ++ | + | - | + | + | - | ++ | ++ |
| 1574 | +++ | ++ | ++ | + | ++ | - | - | - | - | - | - | - | - | - | + | + | - | ++ | + | - | ++ | ++ |
| 1575 | ++ | ++ | - | ++ | - | ++ | ++ | ++ | ++ | - | ++ | ++ | ++ | ++ | - | - | ++ | - | - | ++ | ++ | - |
| 1576 | ++ | ++ | + | ++ | ++ | ++ | ++ | - | + | - | - | - | - | - | + | + | - | + | + | - | ++ | ++ |
| 1578 | +++ | ++ | ++ | ++ | ++ | - | - | - | - | - | - | - | + | - | ++ | + | - | + | - | - | ++ | ++ |
| 1591 | ++ | ++ | - | ++ | ++ | ++ | +++ | ++ | ++ | - | ++ | ++ | ++ | ++ | + | + | + | + | - | ++ | ++ | ++ |
| 1626 | ++ | ++ | + | ++ | ++ | ++ | ++ | - | + | - | - | - | - | - | ++ | + | - | + | + | - | ++ | ++ |
| 1637 | ++ | ++ | + | ++ | ++ | ++ | ++ | ++ | ++ | + | ++ | - | ++ | ++ | ++ | - | ++ | + | + | + | + | ++ |
| 2000 | ++ | - | - | ++ | ++ | - | - | - | - | - | - | - | - | - | ++ | - | - | - | - | - | + | ++ |
| 2001 | ++ | ++ | + | + | + | - | - | - | - | - | - | - | - | - | ++ | - | - | ++ | - | - | + | ++ |
| 2033 | +++ | ++ | ++ | + | ++ | - | - | - | - | - | - | - | - | - | ++ | + | - | + | + | - | ++ | ++ |
| 2035 | +++ | ++ | - | - | ++ | - | - | - | - | - | - | - | - | ++ | - | + | - | + | + | - | ++ | ++ |
| 2114 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| 2115 | +++ | ++ | ++ | ++ | ++ | - | - | - | - | - | - | - | - | - | - | - | - | + | + | - | ++ | - |
| 2118 | ++ | - | ++ | + | ++ | + | - | - | - | - | - | - | - | - | + | - | - | ++ | + | - | ++ | ++ |
| 2524 | +++ | ++ | - | + | ++ | - | - | - | + | - | - | - | - | - | ++ | + | - | + | + | - | ++ | ++ |
| 2711 | +++ | - | - | ++ | ++ | - | - | - | - | - | - | - | - | - | + | - | - | + | - | - | ++ | ++ |
© 2017 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Uka, V.; Moore, G.G.; Arroyo-Manzanares, N.; Nebija, D.; De Saeger, S.; Diana Di Mavungu, J. Unravelling the Diversity of the Cyclopiazonic Acid Family of Mycotoxins in Aspergillus flavus by UHPLC Triple-TOF HRMS. Toxins 2017, 9, 35. https://doi.org/10.3390/toxins9010035
Uka V, Moore GG, Arroyo-Manzanares N, Nebija D, De Saeger S, Diana Di Mavungu J. Unravelling the Diversity of the Cyclopiazonic Acid Family of Mycotoxins in Aspergillus flavus by UHPLC Triple-TOF HRMS. Toxins. 2017; 9(1):35. https://doi.org/10.3390/toxins9010035
Chicago/Turabian StyleUka, Valdet, Geromy G. Moore, Natalia Arroyo-Manzanares, Dashnor Nebija, Sarah De Saeger, and José Diana Di Mavungu. 2017. "Unravelling the Diversity of the Cyclopiazonic Acid Family of Mycotoxins in Aspergillus flavus by UHPLC Triple-TOF HRMS" Toxins 9, no. 1: 35. https://doi.org/10.3390/toxins9010035
APA StyleUka, V., Moore, G. G., Arroyo-Manzanares, N., Nebija, D., De Saeger, S., & Diana Di Mavungu, J. (2017). Unravelling the Diversity of the Cyclopiazonic Acid Family of Mycotoxins in Aspergillus flavus by UHPLC Triple-TOF HRMS. Toxins, 9(1), 35. https://doi.org/10.3390/toxins9010035









