Canopy Venom: Proteomic Comparison among New World Arboreal Pit-Viper Venoms
Abstract
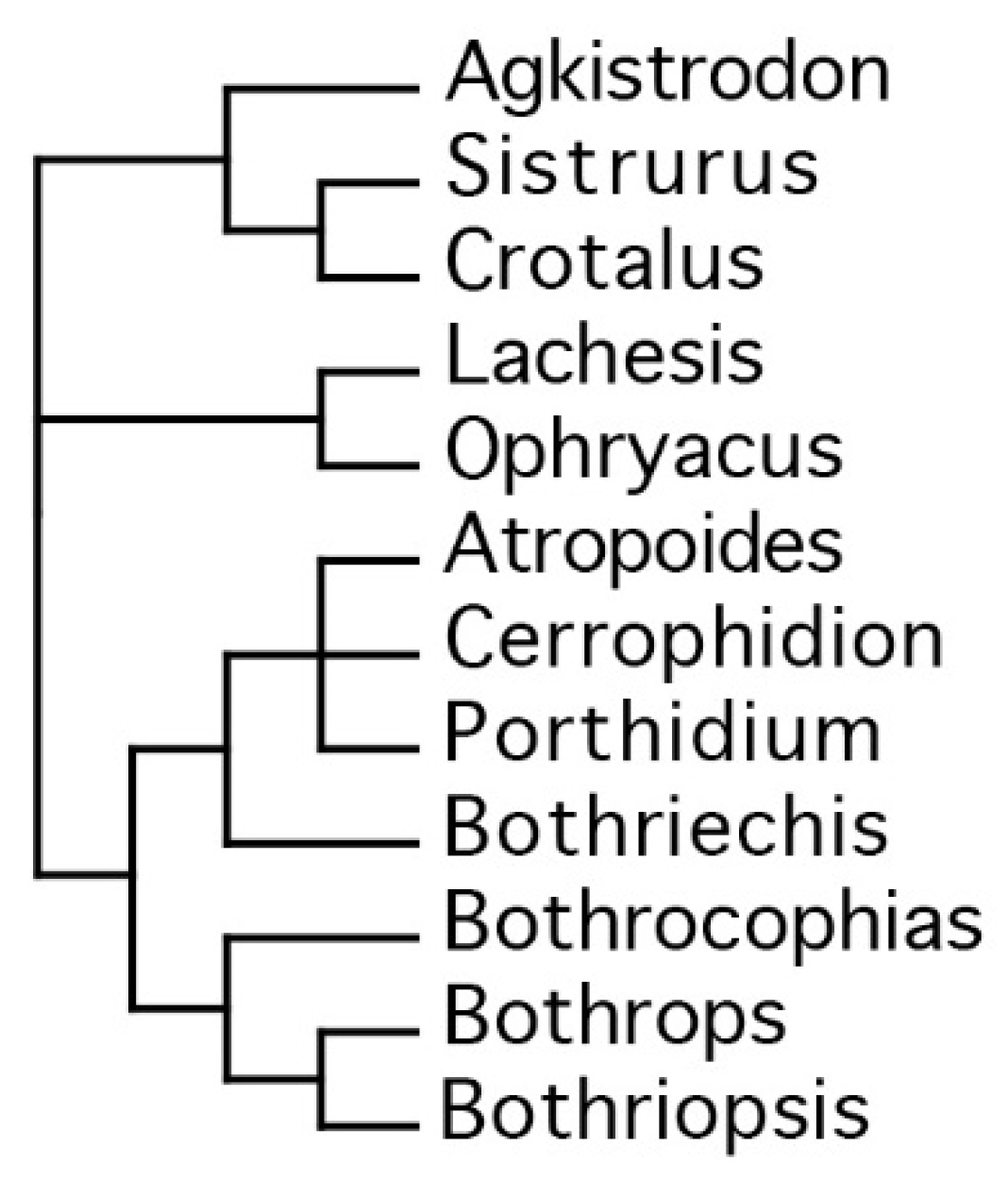
:1. Introduction
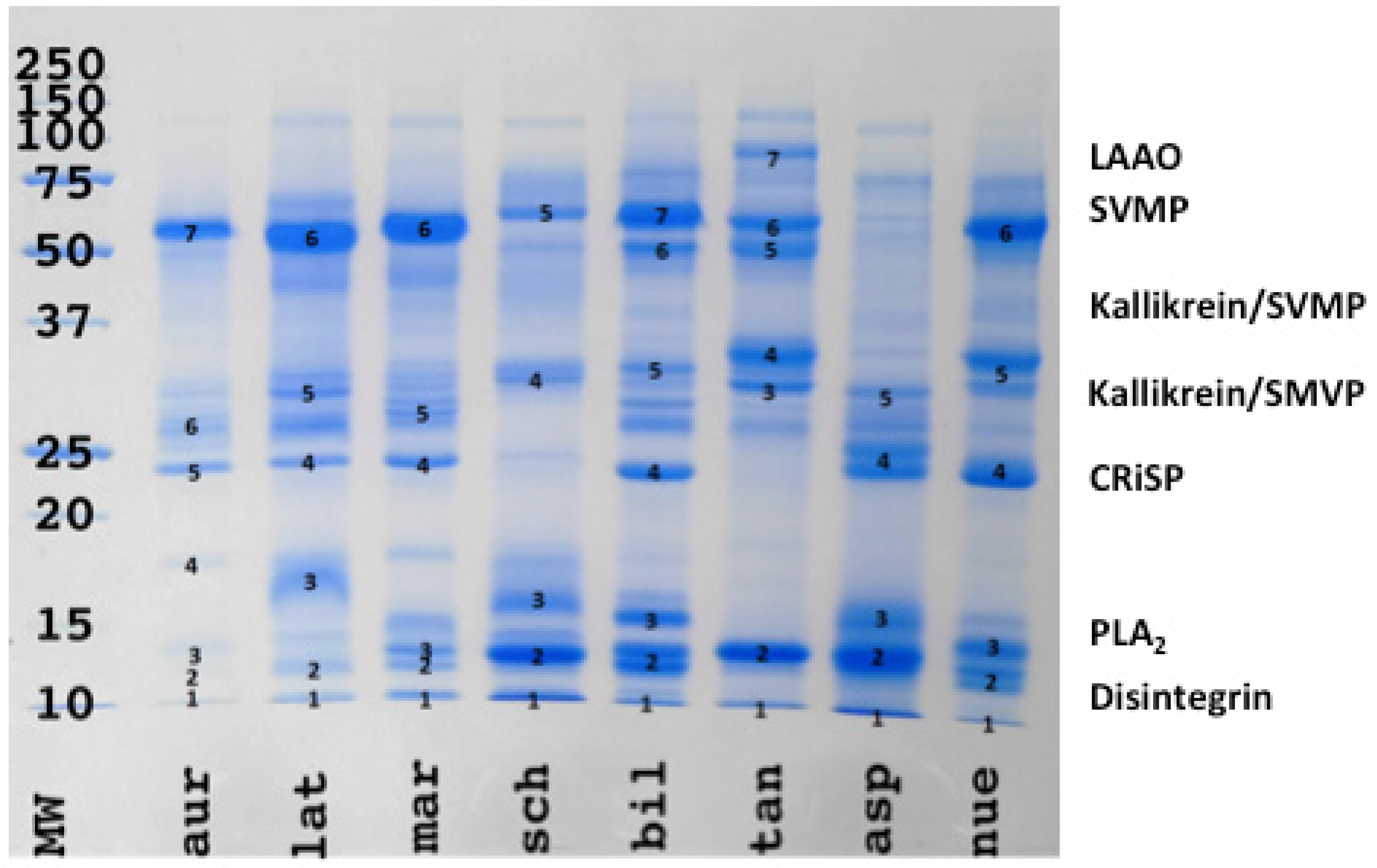
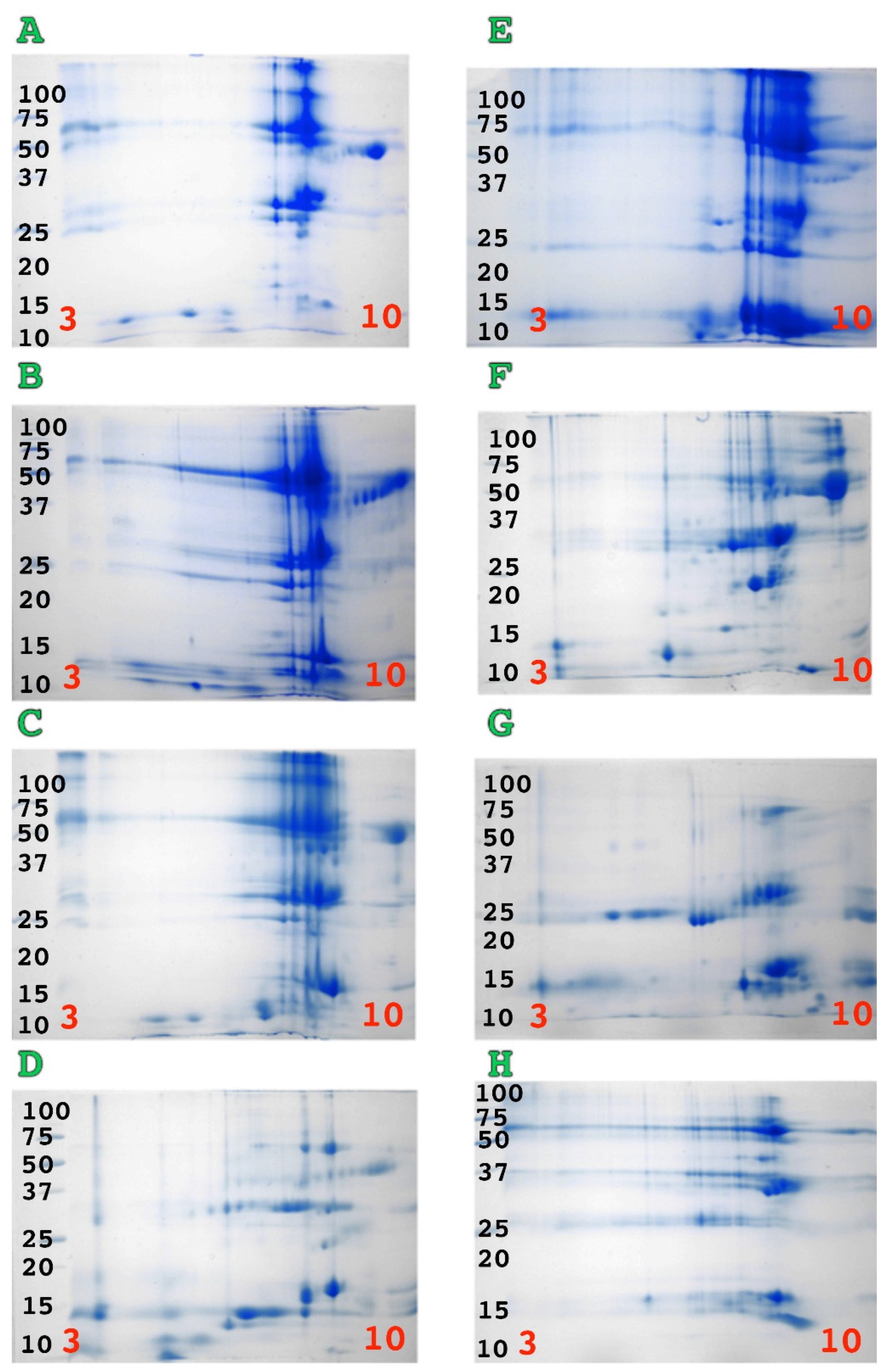
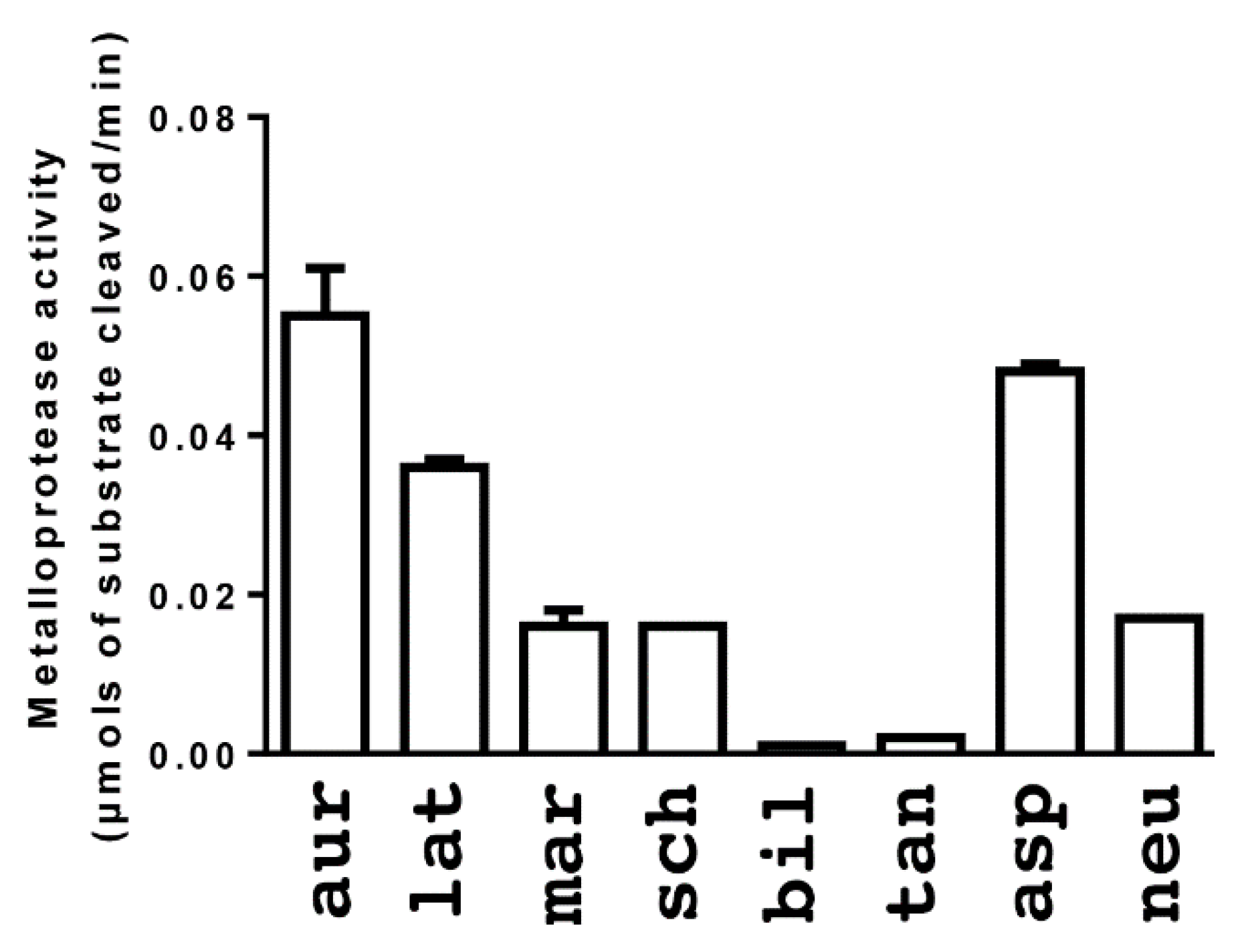
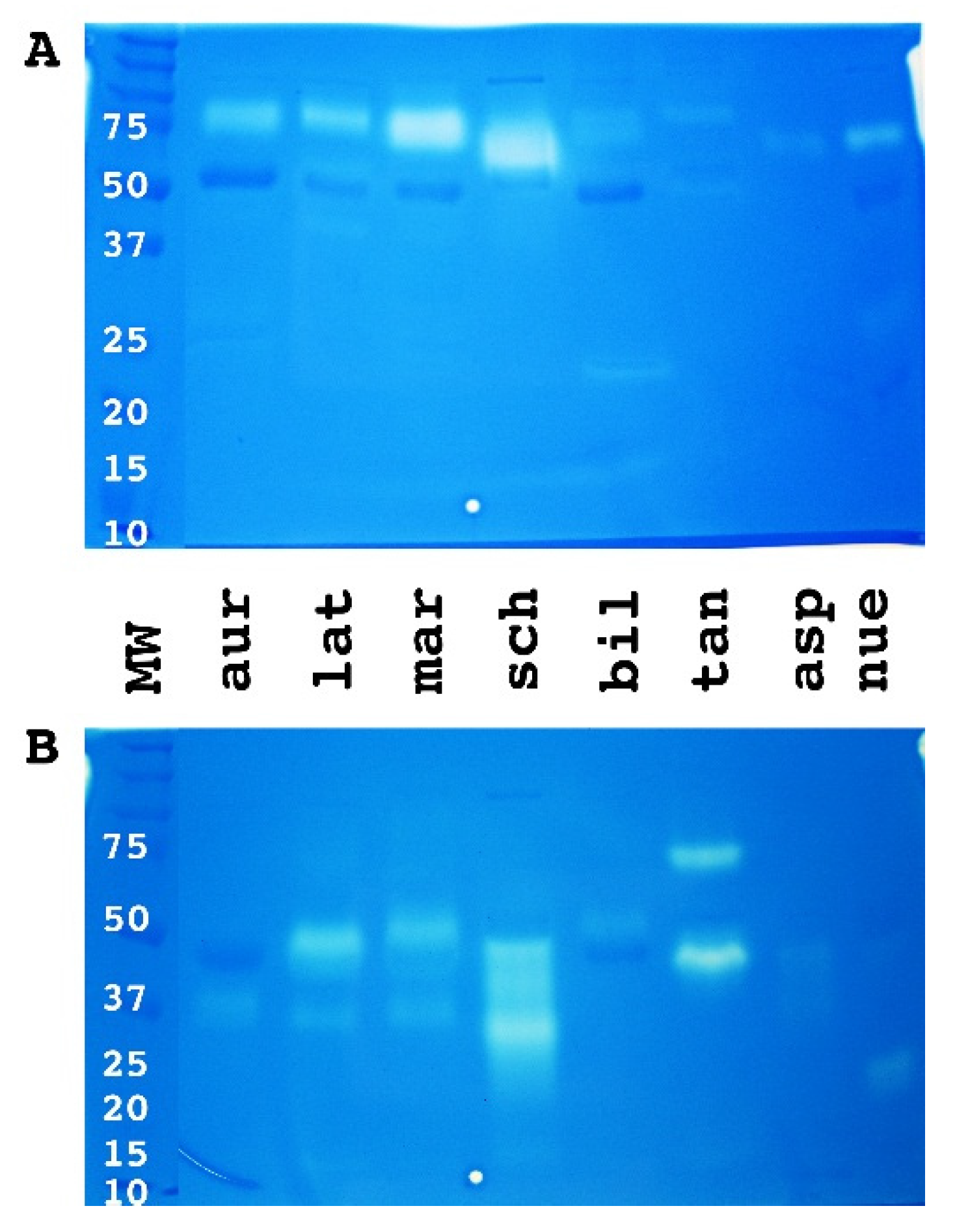
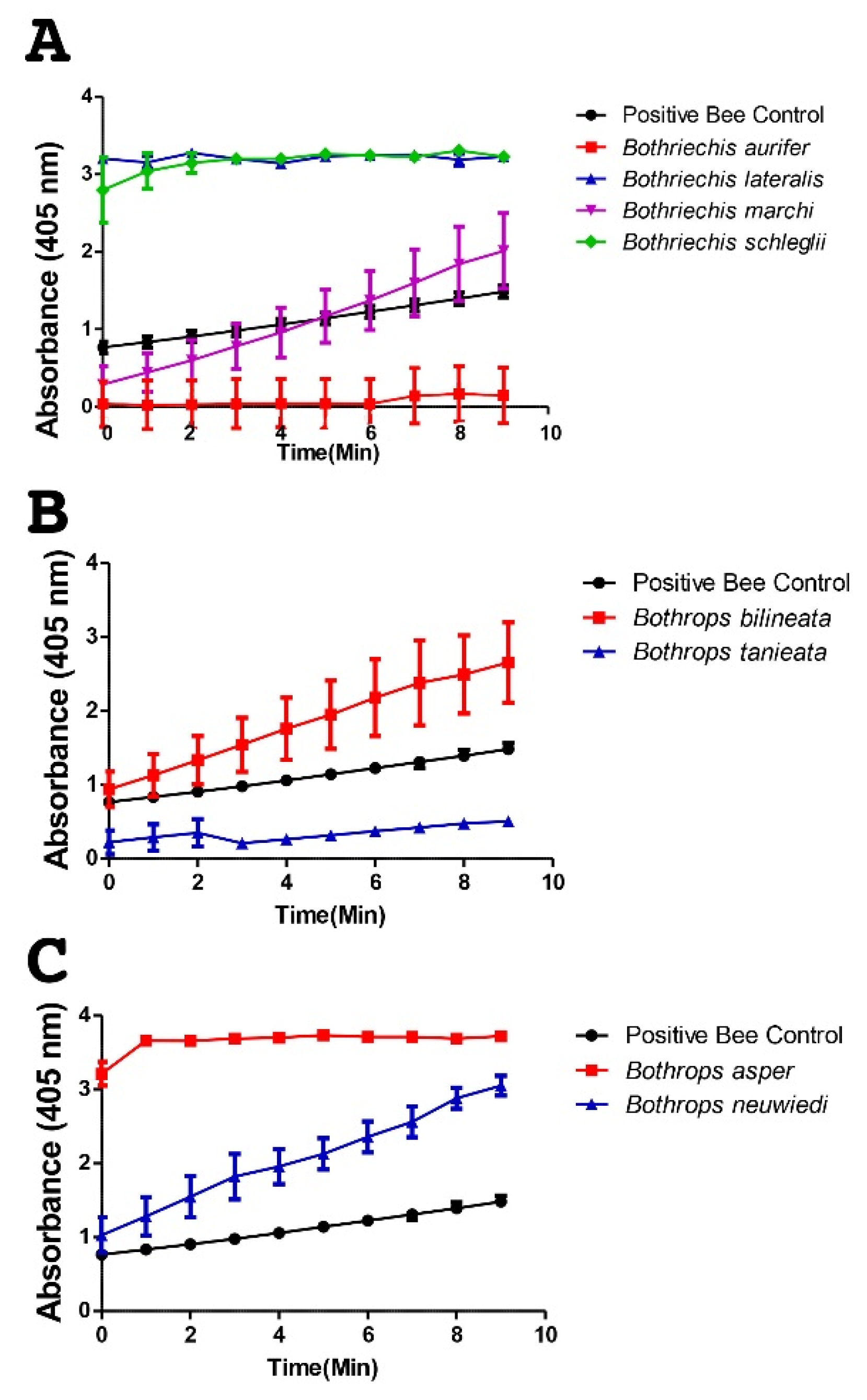
2. Results
3. Discussion
4. Experimental Section
4.1. Venom Collection
4.2. Proteomics
4.2.1. 1D and 2D Gels Using SDS PAGE Electrophoresis Methodology
1D SDS-PAGE
Mini 2D SDS-PAGE
First Dimension
Second Dimension
Liquid Chromatography-tandem Mass Spectrometry (LC-MS/MS) Analyses
Reduction and Alkylation
Enzymatic Digestion
Peptide Extraction
4.2.2. Shotgun Analysis
Reduction, Alkylation and Trypsinization
4.3. Bioactivity Testing
4.3.1. Fluorescent Determination of Metalloprotease Activity
4.3.2. Zymogram PAGE 10% and 12% Gels Bio-Rad
4.3.3. sPLA2 Assay Kit Cayman Activity
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Anaya, M.; Rael, E.D.; Lieb, C.S.; Perez, J.C.; Salo, R.J. Antibody Detection of Venom Protein Variation within a Population of the Rattlesnake Crotalus v. viridis. J. Herpetol. 1992, 26, 473–482. [Google Scholar] [CrossRef]
- Bush, S.P.; Green, S.M.; Moynihan, J.A.; Hayes, W.K.; Cardwell, M.D. Crotalidae polyvalent immune Fab (ovine) antivenom is efficacious for envenomations by Southern Pacific rattlesnakes (Crotalus helleri). Ann. Emerg. Med. 2002, 40, 619–624. [Google Scholar] [CrossRef] [PubMed]
- Casewell, N.R.; Wüster, W.; Vonk, F.J.; Harrison, R.A.; Fry, B.G. Complex cocktails: The evolutionary novelty of venoms. Trends Ecol. Evol. 2012, 28, 219–229. [Google Scholar] [CrossRef] [PubMed]
- Chippaux, J.-P.; Williams, V.; White, J. Snake venom variability: Methods of study, results and interpretation. Toxicon 1991, 29, 1279–1303. [Google Scholar] [CrossRef]
- Fry, B.G.; Casewell, N.R.; Wuster, W.; Vidal, N.; Young, B.; Jackson, T.N. The structural and functional diversification of the Toxicofera reptile venom system. Toxicon 2012, 60, 434–448. [Google Scholar] [CrossRef] [PubMed]
- Mackessy, S.P.; Baxter, L.M. Bioweapons synthesis and storage: The venom gland of front-fanged snakes. Zool. Anz. J. Comp. Zool. 2006, 245, 147–159. [Google Scholar] [CrossRef]
- Angulo, Y.; Escolano, J.; Lomonte, B.; Gutierrez, J.M.; Sanz, L.; Calvete, J.J. Snake venomics of Central American pitvipers: Clues for rationalizing the distinct envenomation profiles of Atropoides nummifer and Atropoides picadoi. J. Proteome Res. 2008, 7, 708–719. [Google Scholar] [CrossRef] [PubMed]
- Fry, B.G. From genome to “venome”: Molecular origin and evolution of the snake venom proteome inferred from phylogenetic analysis of toxin sequences and related body proteins. Genome Res. 2005, 15, 403–420. [Google Scholar] [CrossRef] [PubMed]
- Fry, B.G.; Undheim, E.A.; Ali, S.A.; Jackson, T.N.; Debono, J.; Scheib, H.; Ruder, T.; Morgenstern, D.; Cadwallader, L.; Whitehead, D.; et al. Squeezers and Leaf-cutters: Differential Diversification and Degeneration of the Venom System in Toxicoferan Reptiles. Mol. Cell Proteom. 2013, 12, 1881–1899. [Google Scholar] [CrossRef] [PubMed]
- Alape-Girón, A.; Sanz, L.; Escolano, J.; Flores-Díaz, M.; Madrigal, M.; Sasa, M.; Calvete, J.J. Snake venomics of the lancehead pitviper Bothrops asper: Geographic, individual, and ontogenetic variations. J. Proteome Res. 2008, 7, 3556–3571. [Google Scholar] [CrossRef] [PubMed]
- Ali, S.A.; Yang, D.; Jackson, T.N.; Undheim, E.A.; Koludarov, I.; Wood, K.; Jones, A.; Hodgson, W.C.; McCarthy, S.; Ruder, T. Venom proteomic characterization and relative antivenom neutralization of two medically important Pakistani elapid snakes (Bungarus sindanus and Naja naja). J. Proteom. 2013, 89, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Boldrini-Franca, J.; Correa-Netto, C.; Silva, M.M.; Rodrigues, R.S.; De La Torre, P.; Perez, A.; Soares, A.M.; Zingali, R.B.; Nogueira, R.A.; Rodrigues, V.M.; et al. Snake venomics and antivenomics of Crotalus durissus subspecies from Brazil: Assessment of geographic variation and its implication on snakebite management. J. Proteom. 2010, 73, 1758–1776. [Google Scholar] [CrossRef] [PubMed]
- Calvete, J.J.; Escolano, J.; Sanz, L. Snake venomics of Bitis species reveals large intragenus venom toxin composition variation: Application to taxonomy of congeneric taxa. J. Proteome Res. 2007, 6, 2732–2745. [Google Scholar] [CrossRef] [PubMed]
- Calvete, J.J.; Pérez, A.; Lomonte, B.; Sánchez, E.E.; Sanz, L. Snake venomics of Crotalus tigris: The minimalist toxin arsenal of the deadliest neartic rattlesnake venom. Evolutionary clues for generating a pan-specific antivenom against crotalid type II venoms. J. Proteome Res. 2012, 11, 1382–1390. [Google Scholar] [CrossRef] [PubMed]
- Calvete, J.J.; Sanz, L.; Cid, P.; de la Torre, P.; Flores-Díaz, M.; Dos Santos, M.C.; Borges, A.; Bremo, A.; Angulo, Y.; Lomonte, B. Snake venomics of the Central American rattlesnake Crotalus simus and the South American Crotalus durissus complex points to neurotoxicity as an adaptive paedomorphic trend along Crotalus dispersal in South America. J. Proteome Res. 2009, 9, 528–544. [Google Scholar] [CrossRef] [PubMed]
- Fernández, J.; Alape-Girón, A.; Angulo, Y.; Sanz, L.; Gutiérrez, J.M.; Calvete, J.J.; Lomonte, B. Venomic and antivenomic analyses of the Central American coral snake, Micrurus nigrocinctus (Elapidae). J. Proteome Res. 2011, 10, 1816–1827. [Google Scholar] [CrossRef] [PubMed]
- Fry, B.G.; Lumsden, N.G.; Wüster, W.; Wickramaratna, J.C.; Hodgson, W.C.; Kini, R.M. Isolation of a neurotoxin (α-colubritoxin) from a nonvenomous colubrid: Evidence for early origin of venom in snakes. J. Mol. Evol. 2003, 57, 446–452. [Google Scholar] [CrossRef] [PubMed]
- Fry, B.G.; Wickramaratna, J.C.; Hodgson, W.C.; Alewood, P.F.; Kini, R.; Ho, H.; Wüster, W. Electrospray liquid chromatography/mass spectrometry fingerprinting of Acanthophis (death adder) venoms: Taxonomic and toxinological implications. Rapid commun. Mass Spectrum. 2002, 16, 600–608. [Google Scholar] [CrossRef] [PubMed]
- Fry, B.G.; Wüster, W.; Ramjan, R.; Fadil, S.; Jackson, T.; Martelli, P.; Kini, R.M. Analysis of Colubroidea snake venoms by liquid chromatography with mass spectrometry: Evolutionary and toxinological implications. Rapid Commun. Mass Spectrom. 2003, 17, 2047–2062. [Google Scholar] [CrossRef] [PubMed]
- Aird, S.D.; Watanabe, Y.; Villar-Briones, A.; Roy, M.C.; Terada, K.; Mikheyev, A.S. Quantitative high-throughput profiling of snake venom gland transcriptomes and proteomes (Ovophis okinavensis and Protobothrops flavoviridis). BMC Genom. 2013, 14, 790. [Google Scholar] [CrossRef] [PubMed]
- Fry, B.G.; Roelants, K.; Winter, K.; Hodgson, W.C.; Griesman, L.; Kwok, H.F.; Scanlon, D.; Karas, J.; Shaw, C.; Wong, L. Novel venom proteins produced by differential domain-expression strategies in beaded lizards and gila monsters (genus Heloderma). Mol. Biol. Evol. 2010, 27, 395–407. [Google Scholar] [CrossRef] [PubMed]
- Fry, B.G.; Scheib, H.; de Azevedo, I.D.L.J.; Silva, D.A.; Casewell, N.R. Novel transcripts in the maxillary venom glands of advanced snakes. Toxicon 2012, 59, 696–708. [Google Scholar] [CrossRef] [PubMed]
- Fry, B.G.; Wroe, S.; Teeuwisse, W.; van Osch, M.J.; Moreno, K.; Ingle, J.; McHenry, C.; Ferrara, T.; Clausen, P.; Scheib, H. A central role for venom in predation by Varanus komodoensis (Komodo Dragon) and the extinct giant Varanus (Megalania) priscus. Proc. Natl. Acad. Sci. USA 2009, 106, 8969–8974. [Google Scholar] [CrossRef] [PubMed]
- Margres, M.J.; Aronow, K.; Loyacano, J.; Rokyta, D.R. The venom-gland transcriptome of the eastern coral snake (Micrurus fulvius) reveals high venom complexity in the intragenomic evolution of venoms. BMC Genom. 2013, 14, 531. [Google Scholar] [CrossRef] [PubMed]
- Rokyta, D.R.; Wray, K.P.; Lemmon, A.R.; Lemmon, E.M.; Caudle, S.B. A high-throughput venom-gland transcriptome for the Eastern Diamondback Rattlesnake (Crotalus adamanteus) and evidence for pervasive positive selection across toxin classes. Toxicon 2011, 57, 657–671. [Google Scholar] [CrossRef] [PubMed]
- Rokyta, D.R.; Wray, K.P.; Margres, M.J. The genesis of an exceptionally lethal venom in the timber rattlesnake (Crotalus horridus) revealed through comparative venom-gland transcriptomics. BMC Genom. 2013, 14, 394. [Google Scholar] [CrossRef] [PubMed]
- Terrat, Y.; Sunagar, K.; Fry, B.G.; Jackson, T.N.; Scheib, H.; Fourmy, R.; Verdenaud, M.; Blanchet, G.; Antunes, A.; Ducancel, F. Atractaspis aterrima toxins: The first insight into the molecular evolution of venom in side-stabbers. Toxins 2013, 5, 1948–1964. [Google Scholar] [CrossRef] [PubMed]
- Wagstaff, S.C.; Sanz, L.; Juárez, P.; Harrison, R.A.; Calvete, J.J. Combined snake venomics and venom gland transcriptomic analysis of the ocellated carpet viper, Echis ocellatus. J. Proteom. 2009, 71, 609–623. [Google Scholar] [CrossRef] [PubMed]
- Fry, B.G.; Scheib, H.; van der Weerd, L.; Young, B.; McNaughtan, J.; Ramjan, S.R.; Vidal, N.; Poelmann, R.E.; Norman, J.A. Evolution of an arsenal structural and functional diversification of the venom system in the advanced snakes (Caenophidia). Mol. Cell. Proteom. 2008, 7, 215–246. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez, J.M.; Sanz, L.; Escolano, J.; Fernández, J.; Lomonte, B.; Angulo, Y.; Rucavado, A.; Warrell, D.A.; Calvete, J.J. Snake Venomics of the Lesser Antillean Pit Vipers Bothrops caribbaeus and Bothrops lanceolatus: Correlation with Toxicological Activities and Immunoreactivity of a Heterologous Antivenom†. J. Proteome Res. 2008, 7, 4396–4408. [Google Scholar] [CrossRef] [PubMed]
- Lomonte, B.; Escolano, J.; Fernandez, J.; Sanz, L.; Angulo, Y.; Gutierrez, J.M.; Calvete, J.J. Snake venomics and antivenomics of the arboreal neotropical pitvipers Bothriechis lateralis and Bothriechis schlegelii. J. Proteome Res. 2008, 7, 2445–2457. [Google Scholar] [CrossRef] [PubMed]
- Sanz, L.; Gibbs, H.L.; Mackessy, S.P.; Calvete, J.J. Venom proteomes of closely related Sistrurus rattlesnakes with divergent diets. J. Proteome Res. 2006, 5, 2098–2112. [Google Scholar] [CrossRef] [PubMed]
- Tashima, A.K.; Sanz, L.; Camargo, A.C.; Serrano, S.M.; Calvete, J.J. Snake venomics of the Brazilian pitvipers Bothrops cotiara and Bothrops fonsecai. Identification of taxonomy markers. J. Proteom. 2008, 71, 473–485. [Google Scholar] [CrossRef] [PubMed]
- van der Weyden, L.; Hains, P.G.; Broady, K.W. Characterisation of the biochemical and biological variations from the venom of the death adder species (Acanthophis antarcticus, A. praelongus and A. pyrrhus). Toxicon 2000, 38, 1703–1713. [Google Scholar] [CrossRef]
- Castro, E.N.; Lomonte, B.; del Carmen Gutiérrez, M.; Alagón, A.; Gutiérrez, J.M. Intraspecies variation in the venom of the rattlesnake Crotalus simus from Mexico: Different expression of crotoxin results in highly variable toxicity in the venoms of three subspecies. J. Proteom. 2013, 87, 103–121. [Google Scholar] [CrossRef] [PubMed]
- Daltry, J.C.; Ponnudurai, G.; Shin, C.K.; Tan, N.-H.; Thorpe, R.S.; Wolfgang, W. Electrophoretic profiles and biological activities: Intraspecific variation in the venom of the Malayan pit viper (Calloselasma rhodostoma). Toxicon 1996, 34, 67–79. [Google Scholar] [CrossRef]
- Daltry, J.C.; Wüster, W.; Thorpe, R.S. Diet and snake venom evolution. Nature 1996, 379, 537–540. [Google Scholar] [CrossRef] [PubMed]
- Forstner, M.; Hilsenbeck, R.; Scudday, J. Geographic variation in whole venom profiles from the mottled rock rattlesnake (Crotalus lepidus lepidus) in Texas. J. Herpetol. 1997, 31, 277–287. [Google Scholar] [CrossRef]
- French, W.J.; Hayes, W.K.; Bush, S.P.; Cardwell, M.D.; Bader, J.O.; Rael, E.D. Mojave toxin in venom of Crotalus helleri (Southern Pacific Rattlesnake): Molecular and geographic characterization. Toxicon 2004, 44, 781–791. [Google Scholar] [CrossRef] [PubMed]
- Mackessy, S.P. Evolutionary trends in venom composition in the western rattlesnakes (Crotalus viridis sensu lato): Toxicity vs. tenderizers. Toxicon 2010, 55, 1463–1474. [Google Scholar] [CrossRef] [PubMed]
- Salazar, A.M.; Guerrero, B.; Cantu, B.; Cantu, E.; Rodríguez-Acosta, A.; Pérez, J.C.; Galán, J.A.; Tao, A.; Sánchez, E.E. Venom variation in hemostasis of the southern Pacific rattlesnake (Crotalus oreganus helleri): Isolation of hellerase. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2009, 149, 307–316. [Google Scholar] [CrossRef] [PubMed]
- Sunagar, K.; Undheim, E.A.; Scheib, H.; Gren, E.C.; Cochran, C.; Person, C.E.; Koludarov, I.; Kelln, W.; Hayes, W.K.; King, G.F. Intraspecific venom variation in the medically significant Southern Pacific Rattlesnake (Crotalus oreganus helleri): Biodiscovery, clinical and evolutionary implications. J. Proteom. 2014, 99, 68–83. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, J.A.; Glenn, J.L.; Straight, R.C.; Sites, J.W., Jr. Distribution and Genetic Variation in Venom A and B Populations of the Mojave Rattlesnake (Crotalus scutulatus scutulatus in Arizona. Herpetologica 1991, 47, 54–68. [Google Scholar]
- Menezes, M.C.; Furtado, M.F.; Travaglia-Cardoso, S.R.; Camargo, A.C.; Serrano, S.M. Sex-based individual variation of snake venom proteome among eighteen Bothrops jararaca siblings. Toxicon 2006, 47, 304–312. [Google Scholar] [CrossRef] [PubMed]
- Pimenta, D.C.; Prezoto, B.C.; Konno, K.; Melo, R.L.; Furtado, M.F.; Camargo, A.; Serrano, S.M. Mass spectrometric analysis of the individual variability of Bothrops jararaca venom peptide fraction. Evidence for sex-based variation among the bradykinin-potentiating peptides. Rapid Commun. Mass Spectrum. 2007, 21, 1034–1042. [Google Scholar] [CrossRef] [PubMed]
- Durban, J.; Pérez, A.; Sanz, L.; Gómez, A.; Bonilla, F.; Chacón, D.; Sasa, M.; Angulo, Y.; Gutiérrez, J.M.; Calvete, J.J. Integrated “omics” profiling indicates that miRNAs are modulators of the ontogenetic venom composition shift in the Central American rattlesnake, Crotalus simus simus. BMC Genom. 2013, 14, 234. [Google Scholar] [CrossRef] [PubMed]
- Guércio, R.A.; Shevchenko, A.; Shevchenko, A.; López-Lozano, J.L.; Paba, J.; Sousa, M.V.; Ricart, C.A. Ontogenetic variations in the venom proteome of the Amazonian snake Bothrops atrox. Proteome Sci. 2006, 4, 11. [Google Scholar] [CrossRef] [PubMed]
- López-Lozano, J.L.; de Sousa, M.V.; Ricart, C.A.O.; Chávez-Olortegui, C.; Sanchez, E.F.; Muniz, E.G.; Bührnheim, P.F.; Morhy, L. Ontogenetic variation of metalloproteinases and plasma coagulant activity in venoms of wild Bothrops atrox specimens from Amazonian rain forest. Toxicon 2002, 40, 997–1006. [Google Scholar] [CrossRef]
- Mackessy, S.P. Venom ontogeny in the Pacific rattlesnakes Crotalus viridis helleri and C. v. oreganus. Copeia 1988, 1988, 92–101. [Google Scholar] [CrossRef]
- Mackessy, S.P.; Williams, K.; Ashton, K.G. Ontogenetic variation in venom composition and diet of Crotalus oreganus concolor: A case of venom paedomorphosis? J. Inf. 2003, 2003, 769–782. [Google Scholar] [CrossRef]
- Saldarriaga, M.M.A.; Otero, R.; Núñez, V.; Toro, M.F.; Dı́az, A.; Gutiérrez, J.M.A. Ontogenetic variability of Bothrops atrox and Bothrops asper snake venoms from Colombia. Toxicon 2003, 42, 405–411. [Google Scholar] [CrossRef]
- Zelanis, A.; Tashima, A.K.; Rocha, M.M.; Furtado, M.F.; Camargo, A.C.; Ho, P.L.; Serrano, S.M. Analysis of the ontogenetic variation in the venom proteome/peptidome of Bothrops jararaca reveals different strategies to deal with prey. J. Proteome Res. 2010, 9, 2278–2291. [Google Scholar] [CrossRef] [PubMed]
- Mebs, D. Toxicity in animals. Trends in evolution? Toxicon 2001, 39, 87–96. [Google Scholar] [CrossRef]
- Sasa, M. Diet and snake venom evolution: Can local selection alone explain intraspecific venom variation? Toxicon 1999, 37, 249–252. [Google Scholar] [PubMed]
- Aird, S.D. Ophidian envenomation strategies and the role of purines. Toxicon 2002, 40, 335–393. [Google Scholar] [CrossRef]
- Barlow, A.; Pook, C.E.; Harrison, R.A.; Wüster, W. Coevolution of diet and prey-specific venom activity supports the role of selection in snake venom evolution. Proc. R. Soc. B Biol. Sci. 2009, 276, 2443–2449. [Google Scholar] [CrossRef] [PubMed]
- Brust, A.; Sunagar, K.; Undheim, E.A.; Vetter, I.; Yang, D.C.; Casewell, N.R.; Jackson, T.N.; Koludarov, I.; Alewood, P.F.; Hodgson, W.C. Differential evolution and neofunctionalization of snake venom metalloprotease domains. Mol. Cell. Proteom. 2013, 12, 651–663. [Google Scholar] [CrossRef] [PubMed]
- Gibbs, H.L.; Mackessy, S.P. Functional basis of a molecular adaptation: Prey-specific toxic effects of venom from Sistrurus rattlesnakes. Toxicon 2009, 53, 672–679. [Google Scholar] [CrossRef] [PubMed]
- Pawlak, J.; Mackessy, S.P.; Fry, B.G.; Bhatia, M.; Mourier, G.; Fruchart-Gaillard, C.; Servent, D.; Ménez, R.; Stura, E.; Ménez, A. Denmotoxin, a three-finger toxin from the colubrid snake Boiga dendrophila (Mangrove Catsnake) with bird-specific activity. J. Biol. Chem. 2006, 281, 29030–29041. [Google Scholar] [CrossRef] [PubMed]
- Sunagar, K.; Johnson, W.E.; O’Brien, S.J.; Vasconcelos, V.; Antunes, A. Evolution of CRISPs associated with toxicoferan-reptilian venom and mammalian reproduction. Mol. Biol. Evol. 2012, 29, 1807–1822. [Google Scholar] [CrossRef] [PubMed]
- Parkinson, C.L. Molecular systematics and biogeographical history of pitvipers as determined by mitochondrial ribosomal DNA sequences. Copeia 1999, 1999, 576–586. [Google Scholar] [CrossRef]
- Parkinson, C.L.; Campbell, J.A.; Chippindale, P.T.; Schuett, G. Multigene phylogenetic analysis of pitvipers, with comments on their biogeography. In Biology of the Vipers; Schuett, G.W., Höggren, M., Eds.; Eagle Mountain Publishing: Eagle Mountain, UT, USA, 2002; pp. 93–110. [Google Scholar]
- Kraus, F.; Mink, D.G.; Brown, W.M. Crotaline intergeneric relationships based on mitochondrial DNA sequence data. Copeia 1996, 1996, 763–773. [Google Scholar] [CrossRef]
- Wüster, W.; Peppin, L.; Pook, C.E.; Walker, D.E. A nesting of vipers: Phylogeny and historical biogeography of the Viperidae (Squamata: Serpentes). Mol. Phylogenetics Evol. 2008, 49, 445–459. [Google Scholar] [CrossRef] [PubMed]
- Castoe, T.A.; Parkinson, C.L. Bayesian mixed models and the phylogeny of pitvipers (Viperidae: Serpentes). Mol. Phylogenetics Evol. 2006, 39, 91–110. [Google Scholar] [CrossRef] [PubMed]
- Vidal, N.; Lecointre, G.; Vié, J.C.; Gasc, J.P. Molecular systematics of pitvipers: Paraphyly of the Bothrops complex. Comptes Rendus de l'Académie des Sciences-Series III-Sciences de la Vie 1997, 320, 95–101. [Google Scholar] [CrossRef]
- Castoe, T.A.; Daza, J.M.; Smith, E.N.; Sasa, M.M.; Kuch, U.; Campbell, J.A.; Chippindale, P.T.; Parkinson, C.L. Comparative phylogeography of pitvipers suggests a consensus of ancient Middle American highland biogeography. J. Biogeogr. 2009, 36, 88–103. [Google Scholar] [CrossRef]
- Cadle, J.E. Phylogenetic relationships among vipers: Immunological evidence. In Biology of the Pitvipers; Selva Natural History Book Publishers: Tyler, TX, USA, 1992; pp. 41–48. [Google Scholar]
- Salomão, M.; Wüster, W.; Thorpe, R. BBBSP MtDNA phylogeny of Neotropical pitvipers of the genus Bothrops (Squamata: Serpentes: Viperidae). Kaupia 1999, 8, 127–134. [Google Scholar]
- Salomão, M.; Wüster, W.; Thorpe, R.; Touzet, J. DNA evolution of South American pitvipers of the genus Bothrops (Reptilia: Serpentes: Viperidae). Venom. Snakes Ecol. Evol. Snakebite 1997, 70, 89–98. [Google Scholar]
- Werman, S.D. Phylogenetic Relationships of Central and South American Pitvipers of the Genus Bothrops (Sensu Lato): Cladistic Analyses of Biochemical and Anatomical Characters. In Biology Of the Pitvipers; Campbell, J.A., Brodie, E.D., Jr., Eds.; Selva: Tyler, TX, USA, 1992; pp. 21–40. [Google Scholar]
- Wüster, W.; Salomão, M.; Quijada-Mascareñas, J.A.; Thorpe, R.; Duckett, G.; Puorto, M.; Theakston, R.; Warrell, D. Origins and evolution of the South American pitviper fauna: Evidence from mitochondrial DNA sequence analysis. In Biology of the Vipers; Schuett, G.W., Höggren, M., Eds.; Eagle Mountain Publishing: Eagle Mountain, UT, USA, 2002; pp. 111–128. [Google Scholar]
- Schatti, B.; Kramer, E.; Touzet, J. Systematic remarks on a rare Crotalid snake from Ecuador, Bothriechis albocarinata (Shreve), with some comments on generic arrangement of Neotropical pitvipers. Ibid 1990, 97, 877–885. [Google Scholar] [CrossRef]
- Burger, W.L. Genera of Pitvipers (Serpentes: Crotalidae). PhD Thesis, University of Kansas, Lawrence, KS, USA, 1971. [Google Scholar]
- Campbell, J.A.; Lamar, W.W. Taxonomic status of miscellaneous Neotropical viperids, with the descriptions of a new genus. Occ. Pap. Mus. Tex. Tech. Univ. 1992, 153, 1–31. [Google Scholar]
- Fenwick, A.M.; Gutberlet, R.L., Jr.; Evans, J.A.; Parkinson, C.L. Morphological and molecular evidence for phylogeny and classification of South American pitvipers, genera Bothrops, Bothriopsis, and Bothrocophias (Serpentes: Viperidae). Zool. J. Linn. Soc. 2009, 156, 617–640. [Google Scholar] [CrossRef]
- Carrasco, P.A.; Mattoni, C.I.; Leynaud, G.C.; Scrocchi, G.J. Morphology, phylogeny and taxonomy of South American bothropoid pitvipers (Serpentes, Viperidae). Zool. Scr. 2012, 41, 109–124. [Google Scholar] [CrossRef]
- Otero, R.; Gutiérrez, J.; Mesa, M.A.B.; Duque, E.; Rodrı́guez, O.; Arango, J.L.; Gómez, F.; Toro, A.; Cano, F.; Rodrı́guez, L.M.A. Complications of Bothrops, Porthidium, and Bothriechis snakebites in Colombia. A clinical and epidemiological study of 39 cases attended in a university hospital. Toxicon 2002, 40, 1107–1114. [Google Scholar] [CrossRef]
- Rosenfeld, G. Symptomatology, pathology and treatment of snake bites in South America. Venom. Anim. Their Venoms 1971, 2, 345–384. [Google Scholar]
- Torrez, P.; Said, R.; Quiroga, M.; Duarte, M.; França, F. Forest pit viper (Bothriopsis bilineata bilineata) bite in the Brazilian Amazon with acute kidney injury and persistent thrombocytopenia. Toxicon 2014, 85, 27–30. [Google Scholar] [CrossRef] [PubMed]
- Warrell, D.A. Snake bite. Lancet 2010, 375, 77–88. [Google Scholar] [CrossRef]
- Calvete, J.J.; Sanz, L.; Angulo, Y.; Lomonte, B.; Gutierrez, J.M. Venoms, venomics, antivenomics. FEBS Lett. 2009, 583, 1736–1743. [Google Scholar] [CrossRef] [PubMed]
- Gibbs, H.L.; Sanz, L.; Chiucchi, J.E.; Farrell, T.M.; Calvete, J.J. Proteomic analysis of ontogenetic and diet-related changes in venom composition of juvenile and adult Dusky Pigmy rattlesnakes (Sistrurus miliarius barbouri). J. Proteom. 2011, 74, 2169–2179. [Google Scholar] [CrossRef] [PubMed]
- Juárez, P.; Sanz, L.; Calvete, J.J. Snake venomics: Characterization of protein families in Sistrurus barbouri venom by cysteine mapping, N-terminal sequencing, and tandem mass spectrometry analysis. Proteomics 2004, 4, 327–338. [Google Scholar] [CrossRef] [PubMed]
- Margres, M.J.; McGivern, J.J.; Seavy, M.; Wray, K.P.; Facente, J.; Rokyta, D.R. Comparative genotype-phenotype mapping reveals distinct modes of venom expression evolution in the sympatric eastern diamondback rattlesnake (Crotalus adamanteus) and eastern coral snake (Micrurus fulvius). Genetics 2014, 199, 165. [Google Scholar] [CrossRef] [PubMed]
- Margres, M.J.; McGivern, J.J.; Wray, K.P.; Seavy, M.; Calvin, K.; Rokyta, D.R. Linking the transcriptome and proteome to characterize the venom of the eastern diamondback rattlesnake (Crotalus adamanteus). J. Proteom. 2014, 96, 145–158. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Romero, G.; Rucavado, A.; Lazcano, D.; Gutiérrez, J.M.; Borja, M.; Lomonte, B.; Garza-García, Y.; Zugasti-Cruz, A. Comparison of venom composition and biological activities of the subspecies Crotalus lepidus lepidus, Crotalus lepidus klauberi and Crotalus lepidus morulus from Mexico. Toxicon 2013, 71, 84–95. [Google Scholar] [CrossRef] [PubMed]
- Massey, D.J.; Calvete, J.J.; Sánchez, E.E.; Sanz, L.; Richards, K.; Curtis, R.; Boesen, K. Venom variability and envenoming severity outcomes of the Crotalus scutulatus scutulatus (Mojave rattlesnake) from Southern Arizona. J. Proteom. 2012, 75, 2576–2587. [Google Scholar] [CrossRef] [PubMed]
- Pahari, S.; Mackessy, S.P.; Kini, R.M. The venom gland transcriptome of the Desert Massasauga Rattlesnake (Sistrurus catenatus edwardsii): Towards an understanding of venom composition among advanced snakes (Superfamily Colubroidea). BMC Mol. Biol. 2007, 8, 115. [Google Scholar] [CrossRef] [PubMed]
- Rokyta, D.R.; Wray, K.P.; McGivern, J.J.; Margres, M.J. The transcriptomic and proteomic basis for the evolution of a novel venom phenotype within the Timber Rattlesnake (Crotalus horridus). Toxicon 2015, 98, 34–48. [Google Scholar] [CrossRef] [PubMed]
- Correa-Netto, C.; Teixeira-Araujo, R.; Aguiar, A.S.; Melgarejo, A.R.; De-Simone, S.G.; Soares, M.R.; Foguel, D.; Zingali, R.B. Immunome and venome of Bothrops jararacussu: A proteomic approach to study the molecular immunology of snake toxins. Toxicon 2010, 55, 1222–1235. [Google Scholar] [CrossRef] [PubMed]
- Dias, G.S.; Kitano, E.S.; Pagotto, A.H.; Sant’anna, S.V.S.; Rocha, M.M.; Zelanis, A.; Serrano, S.M. Individual variability in the venom proteome of juvenile Bothrops jararaca specimens. J. Proteome Res. 2013, 12, 4585–4598. [Google Scholar] [CrossRef] [PubMed]
- Durban, J.; Juárez, P.; Angulo, Y.; Lomonte, B.; Flores-Diaz, M.; Alape-Girón, A.; Sasa, M.; Sanz, L.; Gutiérrez, J.M.; Dopazo, J. Profiling the venom gland transcriptomes of Costa Rican snakes by 454 pyrosequencing. BMC Genom. 2011, 12, 259. [Google Scholar] [CrossRef] [PubMed]
- Homsi-Brandeburgo, M.; Queiroz, L.; Santo-Neto, H.; Rodrigues-Simioni, L.; Giglio, J. Fractionation of Bothrops jararacussu snake venom: Partial chemical characterization and biological activity of bothropstoxin. Toxicon 1988, 26, 615–627. [Google Scholar] [CrossRef]
- Junqueira-de-Azevedo, I.L.; Bastos, C.M.V.; Ho, P.L.; Luna, M.S.; Yamanouye, N.; Casewell, N.R. Venom-related transcripts from Bothrops jararaca tissues provide novel molecular insights into the production and evolution of snake venom. Mol. Biol. Evol. 2015, 32, 754–766. [Google Scholar] [CrossRef] [PubMed]
- Kashima, S.; Roberto, P.G.; Soares, A.M.; Astolfi-Filho, S.; Pereira, J.O.; Giuliati, S.; Faria, M., Jr.; Xavier, M.A.; Fontes, M.R.; Giglio, J.R. Analysis of Bothrops jararacussu venomous gland transcriptome focusing on structural and functional aspects: I—gene expression profile of highly expressed phospholipases A 2. Biochimie 2004, 86, 211–219. [Google Scholar] [CrossRef] [PubMed]
- Mora-Obando, D.; Guerrero-Vargas, J.A.; Prieto-Sánchez, R.; Beltrán, J.; Rucavado, A.; Sasa, M.; Gutiérrez, J.M.; Ayerbe, S.; Lomonte, B. Proteomic and functional profiling of the venom of Bothrops ayerbei from Cauca, Colombia, reveals striking interspecific variation with Bothrops asper venom. J. Proteom. 2014, 96, 159–172. [Google Scholar] [CrossRef] [PubMed]
- Moura-da-Silva, A.M.; Furlan, M.S.; Caporrino, M.C.; Grego, K.F.; Portes-Junior, J.A.; Clissa, P.B.; Valente, R.H.; Magalhães, G.S. Diversity of metalloproteinases in Bothrops neuwiedi snake venom transcripts: Evidences for recombination between different classes of SVMPs. BMC Genet. 2011, 12, 94. [Google Scholar] [CrossRef] [PubMed]
- Serrano, S.M.; Oliveira, A.K.; Menezes, M.C.; Zelanis, A. The proteinase-rich proteome of Bothrops jararaca venom. Toxin Rev. 2014, 33, 169–184. [Google Scholar] [CrossRef]
- Sousa, L.F.; Nicolau, C.A.; Peixoto, P.S.; Bernardoni, J.L.; Oliveira, S.S.; Portes-Junior, J.A.; Mourão, R.H.V.; Lima-dos-Santos, I.; Sano-Martins, I.S.; Chalkidis, H.M. Comparison of phylogeny, venom composition and neutralization by antivenom in diverse species of Bothrops complex. PLoS Negl. Trop. Dis. 2013, 7, e2442. [Google Scholar] [CrossRef] [PubMed]
- Tashima, A.K.; Zelanis, A.; Kitano, E.S.; Ianzer, D.; Melo, R.L.; Rioli, V.; Sant’anna, S.S.; Schenberg, A.C.; Camargo, A.C.; Serrano, S.M. Peptidomics of three Bothrops snake venoms: Insights into the molecular diversification of proteomes and peptidomes. Mol. Cell. Proteom. 2012, 11, 1245–1262. [Google Scholar] [CrossRef] [PubMed]
- Valente, R.H.; Guimaraes, P.R.; Junqueira, M.; Neves-Ferreira, A.G.C.; Soares, M.R.; Chapeaurouge, A.; Trugilho, M.R.; Leon, I.R.; Rocha, S.L.; Oliveira-Carvalho, A.L. Bothrops insularis venomics: A proteomic analysis supported by transcriptomic-generated sequence data. J. Proteom. 2009, 72, 241–255. [Google Scholar] [CrossRef] [PubMed]
- Zelanis, A.; Andrade-Silva, D.; Rocha, M.M.; Furtado, M.F.; Serrano, S.M.; Junqueira-de-Azevedo, I.L.; Ho, P.L. A transcriptomic view of the proteome variability of newborn and adult Bothrops jararaca snake venoms. PLoS Negl. Trop. Dis. 2012, 6, e1554. [Google Scholar] [CrossRef] [PubMed]
- Angulo, Y.; Chaves, E.; Alape, A.; Rucavado, A.; Marı́, J.; Lomonte, B. Isolation and Characterization of a Myotoxic Phospholipase A 2 from the Venom of the Arboreal Snake Bothriechis (Bothrops) schlegelii from Costa Rica. Arch. Biochem. Biophys. 1997, 339, 260–266. [Google Scholar] [CrossRef] [PubMed]
- Fernández, J.; Lomonte, B.; Sanz, L.; Angulo, Y.; Gutiérrez, J.M.; Calvete, J.J. Snake venomics of Bothriechis nigroviridis reveals extreme variability among palm pitviper venoms: Different evolutionary solutions for the same trophic purpose. J. Proteome Res. 2010, 9, 4234–4241. [Google Scholar] [CrossRef] [PubMed]
- Kuch, U.; Mebs, D.; Gutiérrez, J.M.; Freire, A. Biochemical and biological characterization of Ecuadorian pitviper venoms (genera Bothriechis, Bothriopsis, Bothrops and Lachesis). Toxicon 1996, 34, 714–717. [Google Scholar] [CrossRef]
- Corasolla Carregari, V.; Stuani Floriano, R.; Rodrigues-Simioni, L.; Winck, F.V.; Baldasso, P.A.; Ponce-Soto, L.A.; Marangoni, S. Biochemical, Pharmacological, and Structural Characterization of New Basic Bbil-TX from Bothriopsis bilineata Snake Venom. BioMed Res. Int. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Floriano, R.S.; Carregari, V.C.; de Abreu, V.A.; Kenzo-Kagawa, B.; Ponce-Soto, L.A.; da Cruz-Hoefling, M.A.; Hyslop, S.; Marangoni, S.; Rodrigues-Simioni, L. Pharmacological study of a new Asp49 phospholipase A 2 (Bbil-TX) isolated from Bothriopsis bilineata smargadina (forest viper) venom in vertebrate neuromuscular preparations. Toxicon 2013, 69, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Porto, B.N.; Telli, C.A.; Dutra, T.P.; Alves, L.S.; Bozza, M.T.; Fin, C.A.; Thiesen, F.V.; Renner, M.F. Biochemical and biological characterization of the venoms of Bothriopsis bilineata and Bothriopsis taeniata (Serpentes: Viperidae). Toxicon 2007, 50, 270–277. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues-Simioni, L.; Floriano, R.S.; Rostelato-Ferreira, S.; Sousa, N.C.; Marangoni, S.; Ponce-Soto, L.A.; Carregari, V.C.; Hyslop, S. Presynaptic action of Bothriopsis bilineata smargadina (forest viper) venom in vitro. Toxicon 2011, 58, 140–145. [Google Scholar] [CrossRef] [PubMed]
- Changeux, J.-P.; Kasai, M.; Lee, C.-Y. Use of a snake venom toxin to characterize the cholinergic receptor protein. Proc. Natl. Acad. Sci. USA 1970, 67, 1241–1247. [Google Scholar] [CrossRef] [PubMed]
- Kamiguti, A.; Zuzel, M.; Theakston, R. Snake venom metalloproteinases and disintegrins: Interactions with cells. Braz. J. Med. Biol. Res. 1998, 31, 7. [Google Scholar] [CrossRef]
- Serrano, S.M.; Kim, J.; Wang, D.; Dragulev, B.; Shannon, J.D.; Mann, H.H.; Veit, G.; Wagener, R.; Koch, M.; Fox, J.W. The Cysteine-rich Domain of Snake Venom Metalloproteinases Is a Ligand for von Willebrand Factor A Domains role in substrate targeting*. J. Biol. Chem. 2006, 281, 39746–39756. [Google Scholar] [CrossRef] [PubMed]
- Singh, G.; Kuc, R.E.; Maguire, J.J.; Fidock, M.; Davenport, A.P. Novel Snake Venom Ligand Dendroaspis Natriuretic Peptide Is Selective for Natriuretic Peptide Receptor-A in Human Heart Down regulation of Natriuretic Peptide Receptor-A in Heart Failure. Circ. Res. 2006, 99, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Singh, G.; Maguire, J.; Kuc, R.; Skepper, J.; Fidock, M.; Davenport, A. Characterization of the snake venom ligand [125I]-DNP binding to natriuretic peptide receptor-A in human artery and potent DNP mediated vasodilatation. Br. J. Pharmacol. 2006, 149, 838–844. [Google Scholar] [CrossRef] [PubMed]
- Cushman, D.W.; Ondetti, M.A. History of the design of captopril and related inhibitors of angiotensin converting enzyme. Hypertension 1991, 17, 589–592. [Google Scholar] [CrossRef] [PubMed]
- Kemparaju, K.; Girish, K. Snake venom hyaluronidase: A therapeutic target. Cell Biochem. Funct. 2006, 24, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Koh, D.; Armugam, A.; Jeyaseelan, K. Snake venom components and their applications in biomedicine. Cell. Mol. Life Sci. CMLS 2006, 63, 3030–3041. [Google Scholar] [CrossRef] [PubMed]
- Marcinkiewicz, C. Functional characteristic of snake venom disintegrins: Potential therapeutic implication. Curr. Pharm. Des. 2005, 11, 815–827. [Google Scholar] [CrossRef] [PubMed]
- Pal, S.K.; Gomes, A.; Dasgupta, S.; Gomes, A. Snake venom as therapeutic agents: From toxin to drug development. Indian J. Exp. Biol. 2002, 40, 1353–1358. [Google Scholar] [PubMed]
- Patlak, M. From viper’s venom to drug design: Treating hypertension. FASEB J. 2004, 18, 421e–421e. [Google Scholar] [CrossRef]
- Swenson, S.; Ramu, S.; Markland, F.S. Anti-angiogenesis and RGD-containing snake venom disintegrins. Curr. Pharm. Des. 2007, 13, 2860–2871. [Google Scholar] [CrossRef] [PubMed]
- Fox, J.W.; Serrano, S.M. Approaching the golden age of natural product pharmaceuticals from venom libraries: An overview of toxins and toxin-derivatives currently involved in therapeutic or diagnostic applications. Curr. Pharm. Des. 2007, 13, 2927–2934. [Google Scholar] [CrossRef] [PubMed]
- Calvete, J.J. Antivenomics and venom phenotyping: A marriage of convenience to address the performance and range of clinical use of antivenoms. Toxicon 2010, 56, 1284–1291. [Google Scholar] [CrossRef] [PubMed]
- Chippaux, J.P. Snake-bites: Appraisal of the global situation. Bull. World Health Organ. 1998, 76, 515. [Google Scholar] [PubMed]
- Gutiérrez, J.M.; Theakston, R.D.G.; Warrell, D.A. Confronting the neglected problem of snake bite envenoming: The need for a global partnership. PLoS Med. 2006, 3, e150. [Google Scholar] [CrossRef] [PubMed]
- Fox, J.W.; Serrano, S.M. Structural considerations of the snake venom metalloproteinases, key members of the M12 reprolysin family of metalloproteinases. Toxicon 2005, 45, 969–985. [Google Scholar] [CrossRef] [PubMed]
- Casewell, N.R.; Wagstaff, S.C.; Harrison, R.A.; Renjifo, C.; Wüster, W. Domain loss facilitates accelerated evolution and neofunctionalization of duplicate snake venom metalloproteinase toxin genes. Mol. Biol. Evol. 2011, 28, 2637–2649. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez, J.M.; Lomonte, B.; Leon, G.; Alape-Giron, A.; Flores-Diaz, M.; Sanz, L.; Angulo, Y.; Calvete, J.J. Snake venomics and antivenomics: Proteomic tools in the design and control of antivenoms for the treatment of snakebite envenoming. J. Proteom. 2009, 72, 165–182. [Google Scholar] [CrossRef] [PubMed]
- Calvete, J.J. Proteomic tools against the neglected pathology of snake bite envenoming. Expert Rev. Proteom. 2011, 8, 739–758. [Google Scholar] [CrossRef] [PubMed]
- Campbell, J.A.; Lamar, W.W.; Brodie, E.D. The Venomous Reptiles of the Western Hemisphere; Comstock Publishing Associates: Ithaca, NY, USA, 2004. [Google Scholar]
- Martins, M.; Marques, O.A.; Sazima, I. Ecological and phylogenetic correlates of feeding habits in Neotropical pitvipers of the genus Bothrops. In Biology of the Vipers; Schuett, G.W., Höggren, M., Eds.; Eagle Mountain Publishing: Eagle Mountain, UT, USA, 2002; pp. 307–328. [Google Scholar]
- Mackessy, S. Venom composition in rattlesnakes: Trends and biological significance. In The Biology of Rattlesnakes; Hayes, W.K., Cardwell, M.D., Beaman, K.R., Bush, S.P., Eds.; Loma Linda University Press: Loma Linda, CA, USA, 2008; pp. 495–510. [Google Scholar]
- Gans, C. The feeding mechanism of snakes and its possible evolution. Am. Zool. 1961, 217–227. [Google Scholar] [CrossRef]
- Greene, H.W. Dietary correlates of the origin and radiation of snakes. Am. Zool. 1983, 23, 431–441. [Google Scholar] [CrossRef]
- Pough, F.H.; Groves, J.D. Specializations of the body form and food habits of snakes. Am. Zool. 1983, 23, 443–454. [Google Scholar] [CrossRef]
- Hampton, P.M.; Moon, B.R. Gape size, its morphological basis, and the validity of gape indices in western diamond-backed rattlesnakes (Crotalus atrox). J. Morphol. 2013, 274, 194–202. [Google Scholar] [CrossRef] [PubMed]
- Andrade, D.V.; Abe, A.S. Relationship of venom ontogeny and diet in Bothrops. Herpetologica 1999, 55, 200–204. [Google Scholar]
- MacArtney, J.M. Diet of the northern Pacific rattlesnake, Crotalus viridis oreganus, in British Columbia. Herpetologica 1989, 45, 299–304. [Google Scholar]
- Casasola, A.; Ramos-Cerrillo, B.; de Roodt, A.R.; Saucedo, A.C.; Chippaux, J.-P.; Alagón, A.; Stock, R.P. Paraspecific neutralization of the venom of African species of cobra by an equine antiserum against Naja melanoleuca: A comparative study. Toxicon 2009, 53, 602–608. [Google Scholar] [CrossRef] [PubMed]
- Ciscotto, P.H.; Rates, B.; Silva, D.A.; Richardson, M.; Silva, L.P.; Andrade, H.; Donato, M.F.; Cotta, G.A.; Maria, W.S.; Rodrigues, R.J. Venomic analysis and evaluation of antivenom cross-reactivity of South American Micrurus species. J. Proteom. 2011, 74, 1810–1825. [Google Scholar] [CrossRef] [PubMed]
| Toxin types | aur | lat | mar | sch | bil | tan | asp | neu |
|---|---|---|---|---|---|---|---|---|
| 5’ nuc | X | X | X | |||||
| BPP/CNP | X | X | X | X | ||||
| CRiSP | X | X | X | X | ||||
| Kallikrein | X | X | X | X | X | X | X | |
| Kunitz | X | |||||||
| Lectin | X | X | ||||||
| LAAO | X | X | X | X | X | X | X | |
| PLA2 | X | X | X | |||||
| PLB | X | |||||||
| PD | X | X | ||||||
| SVMP/Dis | X | X | X | |||||
| Usual TL (cm) | <70 | <80 | >80 | <60 | <70 | <100 | 120–180 | 60–70 |
| Max TL (cm) | 101 | 100 | 96.8 | 97.9 | 123 | 175 | 250 | 100 |
| Arboreal | Y | Y | Y | Y | Y | Y | N | N |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Debono, J.; Cochran, C.; Kuruppu, S.; Nouwens, A.; Rajapakse, N.W.; Kawasaki, M.; Wood, K.; Dobson, J.; Baumann, K.; Jouiaei, M.; et al. Canopy Venom: Proteomic Comparison among New World Arboreal Pit-Viper Venoms. Toxins 2016, 8, 210. https://doi.org/10.3390/toxins8070210
Debono J, Cochran C, Kuruppu S, Nouwens A, Rajapakse NW, Kawasaki M, Wood K, Dobson J, Baumann K, Jouiaei M, et al. Canopy Venom: Proteomic Comparison among New World Arboreal Pit-Viper Venoms. Toxins. 2016; 8(7):210. https://doi.org/10.3390/toxins8070210
Chicago/Turabian StyleDebono, Jordan, Chip Cochran, Sanjaya Kuruppu, Amanda Nouwens, Niwanthi W. Rajapakse, Minami Kawasaki, Kelly Wood, James Dobson, Kate Baumann, Mahdokht Jouiaei, and et al. 2016. "Canopy Venom: Proteomic Comparison among New World Arboreal Pit-Viper Venoms" Toxins 8, no. 7: 210. https://doi.org/10.3390/toxins8070210
APA StyleDebono, J., Cochran, C., Kuruppu, S., Nouwens, A., Rajapakse, N. W., Kawasaki, M., Wood, K., Dobson, J., Baumann, K., Jouiaei, M., Jackson, T. N. W., Koludarov, I., Low, D., Ali, S. A., Smith, A. I., Barnes, A., & Fry, B. G. (2016). Canopy Venom: Proteomic Comparison among New World Arboreal Pit-Viper Venoms. Toxins, 8(7), 210. https://doi.org/10.3390/toxins8070210







