Staphylococcus aureus Toxins and Diabetic Foot Ulcers: Role in Pathogenesis and Interest in Diagnosis
Abstract
:1. Introduction
2. DFI and Staphylococcus aureus
2.1. Clinical Aspects of DFI
2.2. Osteomyelitis
2.3. Matrix Metalloproteinases and DFI
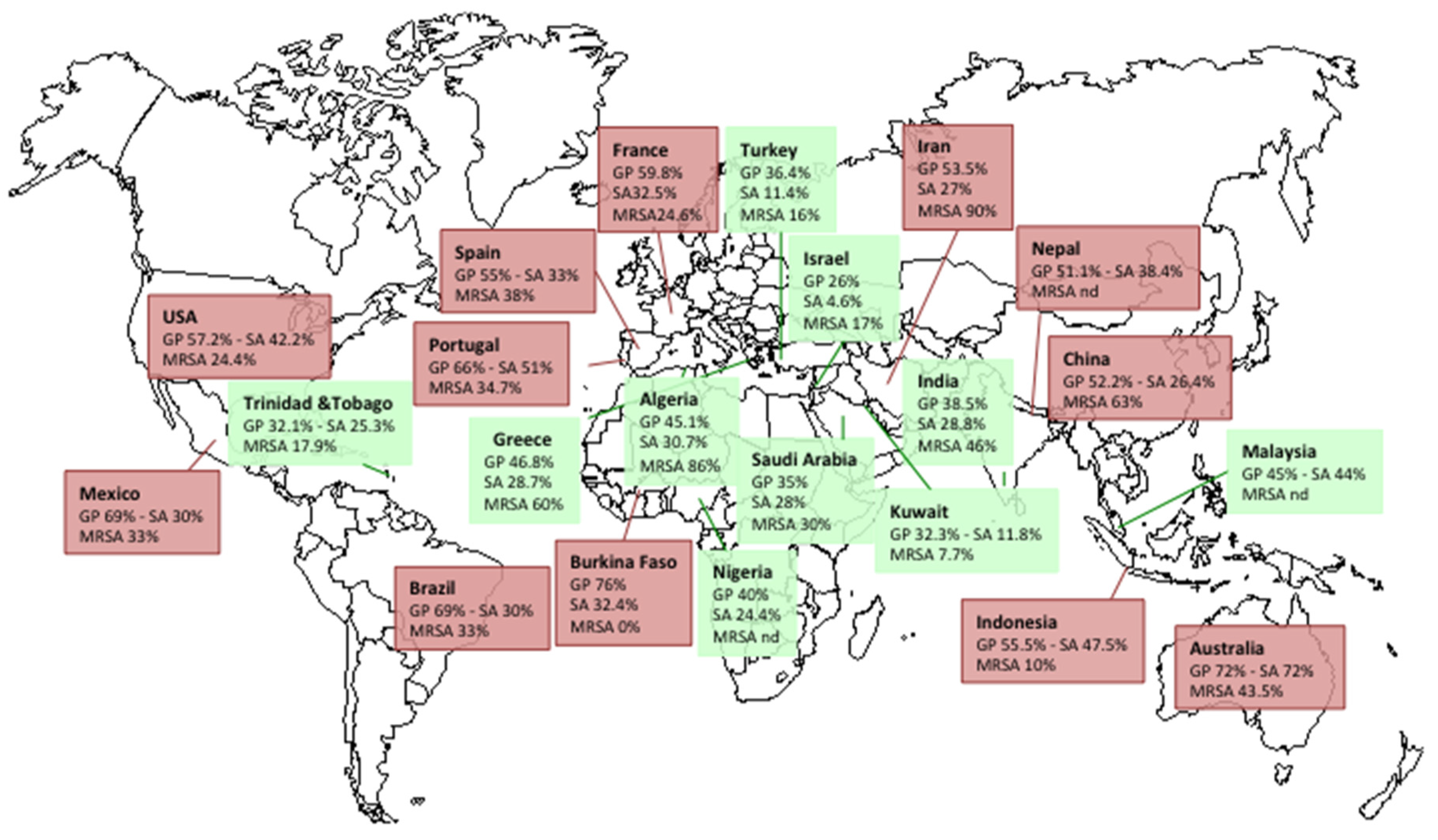
2.4. Prevalence of S. aureus in DFIs
2.5. Resistance of S. aureus in DFIs
2.6. Pathogenesis
3. Staphylococcus aureus Toxins in DFIs
3.1. Pore-Forming Toxins
3.1.1. α-Toxin
3.1.2. Phenol Soluble Modulins
3.1.3. The Bi-Component Leukotoxins
3.2. Exfoliative Toxins
3.3. Enterotoxins
3.3.1. Staphylococcal Enterotoxins and Enterotoxin-Like Toxins
3.3.2. Toxic Shock-Syndrome Toxin 1
3.4. Epidermal Cell Differentiation Inhibitors Toxins
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Singh, N.; Armstrong, D.G.; Lipsky, B.A. Preventing foot ulcers in patients with diabetes. JAMA 2005, 293, 217–228. [Google Scholar] [CrossRef] [PubMed]
- Prompers, L.; Huijberts, M.; Schaper, N.; Apelqvist, J.; Bakker, K.; Edmonds, M.; Holstein, P.; Jude, E.; Jirkovska, A.; Mauricio, D.; et al. Resource utilisation and costs associated with the treatment of diabetic foot ulcers. Prospective data from the Eurodiale Study. Diabetologia 2008, 51, 1826–1834. [Google Scholar] [CrossRef] [PubMed]
- Lipsky, B.A.; Berendt, A.R.; Deery, H.G.; Embil, J.M.; Joseph, W.S.; Karchmer, A.W.; LeFrock, J.L.; Lew, D.P.; Mader, J.T.; Norden, C.; et al. Diagnosis and treatment of diabetic foot infections. Clin. Infect. Dis. 2004, 39, 885–910. [Google Scholar] [CrossRef] [PubMed]
- Lavery, L.A.; Armstrong, D.G.; Wunderlich, R.P.; Tredwell, J.; Boulton, A.J. Diabetic foot syndrome: Evaluating the prevalence and incidence of foot pathology in Mexican Americans and non-Hispanic whites from a diabetes disease management cohort. Diabetes Care 2003, 26, 1435–1438. [Google Scholar] [CrossRef] [PubMed]
- Jeffcoate, W.J.; Lipsky, B.A.; Berendt, A.R.; Cavanagh, P.R.; Bus, S.A.; Peters, E.J.; van Houtum, W.H.; Valk, G.; Bakker, K. Unresolved issues in the management of ulcers of the foot in diabetes. Diabet. Med. 2008, 25, 1380–1389. [Google Scholar] [CrossRef] [PubMed]
- Lavery, L.A.; Armstrong, D.G.; Wunderlich, R.P.; Mohler, M.J.; Wendel, C.S.; Lipsky, B.A. Risk factors for foot infections in individuals with diabetes. Diabetes Care 2006, 29, 1288–1293. [Google Scholar] [CrossRef] [PubMed]
- Richard, J.L.; Lavigne, J.P.; Sotto, A. Diabetes and foot infection: More than double trouble. Diabetes Metab. Res. Rev. 2012, 28, 46–53. [Google Scholar] [CrossRef] [PubMed]
- Spichler, A.; Hurwitz, B.L.; Armstrong, D.G.; Lipsky, B.A. Microbiology of diabetic foot infections: From Louis Pasteur to “crime scene investigation”. BMC Med. 2015, 13, 2. [Google Scholar] [CrossRef] [PubMed]
- Lavigne, J.P.; Sotto, A.; Dunyach-Remy, C.; Lipsky, B.A. New molecular techniques to study the skin microbiota of diabetic foot ulcers. Adv. Wound Care 2015, 4, 38–49. [Google Scholar] [CrossRef] [PubMed]
- Edmonds, M. Infection in the neuroischemic foot. Int. J. Low. Extrem. Wounds 2005, 4, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Williams, D.T.; Hilton, J.R.; Harding, K.G. Diagnosing foot infection in diabetes. Clin. Infect. Dis. 2004, 39, S83–S86. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, D.G.; Lavery, L.A.; Sariaya, M.; Ashry, H. Leukocytosis is a poor indicator of acute osteomyelitis of the foot in diabetes mellitus. J. Foot Ankle Surg. 1996, 35, 280–283. [Google Scholar] [CrossRef]
- Eneroth, M.; Larsson, J.; Apelqvist, J. Deep foot infections in patients with diabetes and foot ulcer: An entity with different characteristics, treatments, and prognosis. J. Diabetes Complicat. 1999, 13, 254–263. [Google Scholar] [CrossRef]
- Eneroth, M.; Apelqvist, J.; Stenström, A. Clinical characteristics and outcome in 223 diabetic patients with deep foot infections. Foot Ankle Int. 1997, 18, 716–722. [Google Scholar] [CrossRef] [PubMed]
- Lipsky, B.A. International consensus group on diagnosing and treating the infected diabetic foot. A report from the international consensus on diagnosing and treating the infected diabetic foot. Diabetes Metab. Res. Rev. 2004, 20, S68–S77. [Google Scholar] [CrossRef] [PubMed]
- Richard, J.L.; Sotto, A.; Lavigne, J.P. New insights in diabetic foot infection. World J. Diabetes 2011, 15, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Messad, N.; Landraud, L.; Canivet, B.; Lina, G.; Richard, J.L.; Sotto, A.; Lavigne, J.P. Distribution of edin in Staphylococcus aureus isolated from diabetic foot ulcers. Clin. Microbiol. Infect. 2013, 19, 875–880. [Google Scholar] [CrossRef] [PubMed]
- Gardner, S.E.; Hillis, S.L.; Heilmann, K.; Segre, J.A.; Grice, E.A. The neuropathic diabetic foot ulcer microbiome is associated with clinical factors. Diabetes 2013, 62, 923–930. [Google Scholar] [CrossRef] [PubMed]
- Hatipoglu, M.; Mutluoglu, M.; Turhan, V.; Uzun, G.; Lipsky, B.A.; Turk-DAY Study Group; Sevim, E.; Demiraslan, H.; Eryilmaz, E.; Ozuguz, C.; et al. Causative pathogens and antibiotic resistance in diabetic foot infections: A prospective multi-center study. J. Diabetes Complicat. 2016, 30, 910–916. [Google Scholar] [CrossRef] [PubMed]
- Commons, R.J.; Robinson, C.H.; Gawler, D.; Davis, J.S.; Price, R.N. High burden of diabetic foot infections in the top end of Australia: An emerging health crisis (DEFINE study). Diabetes Res. Clin. Pract. 2015, 110, 147–157. [Google Scholar] [CrossRef] [PubMed]
- Lesens, O.; Desbiez, F.; Theïs, C.; Ferry, T.; Bensalem, M.; Laurichesse, H.; Tauveron, I.; Beytout, J.; Aragón-Sánchez, J.; Working Group on Diabetic Osteomyelitis. Staphylococcus aureus-related diabetic osteomyelitis: Medical or surgical management? A French and Spanish retrospective cohort. Int. J. Low. Extrem. Wounds 2015, 14, 284–290. [Google Scholar] [CrossRef] [PubMed]
- Serra, R.; Grande, R.; Butrico, L.; Rossi, A.; Settimio, U.F.; Caroleo, B.; Amato, B.; Gallelli, L.; de Franciscis, S. Chronic wound infections: The role of Pseudomonas aeruginosa and Staphylococcus aureus. Expert Rev. Anti-Infect. Ther. 2015, 13, 605–613. [Google Scholar] [CrossRef] [PubMed]
- Wertheim, H.F.; Melles, D.C.; Vos, M.C.; van Leeuwen, W.; van Belkum, A.; Verbrugh, H.A.; Nouwen, J.L. The role of nasal carriage in Staphylococcus aureus infections. Lancet Infect. Dis. 2005, 5, 751–762. [Google Scholar] [CrossRef]
- Tong, S.Y.; David, J.S.; Eichenberger, E.; Holland, T.L.; Fowler, V.G., Jr. Staphylococcus aureus infections: Epidemiology, pathophysiology, clinical manifestations, and management. Clin. Microbiol. Rev. 2015, 28, 603–661. [Google Scholar] [CrossRef] [PubMed]
- Lipsky, B.A.; Aragon-Sanchez, J.; Embil, J.; Kono, S.; Lavery, L.; Senneville, E.; Urbancic-Rovan, V.; Van Asten, S.; Peters, E.J.G.; International Working Group on the Diabetic Foot (IWGDF). IWGDF guidance on the diagnosis and management of foot infections in persons with diabetes. Diabetes Metab. Res. Rev. 2016, 32, 45–74. [Google Scholar] [CrossRef] [PubMed]
- Shaper, N.C. Diabetic foot ulcer classification system for research purposes: A progress report on criteria for including patients in research studies. Diabetes Metab. Res. Rev. 2004, 20, S90–S95. [Google Scholar] [CrossRef] [PubMed]
- Lipsky, B.A.; Berendt, A.R.; Cornia, P.B.; Pile, J.C.; Peters, E.J.; Armstrong, D.G.; Deery, H.G.; Embil, J.M.; Joseph, W.S.; Karchmer, A.W.; et al. 2012 Infectious Diseases Society of America clinical practice guideline for the diagnosis and treatment of diabetic foot infections. J. Am. Podiatr. Med. Assoc. 2013, 103, 2–7. [Google Scholar] [CrossRef] [PubMed]
- Lipsky, B.A. Osteomyelitis of the foot in diabetic patients. Clin. Infect. Dis. 1997, 25, 1318–1326. [Google Scholar] [CrossRef] [PubMed]
- Markanday, A. Diagnosing diabetic foot osteomyelitis: Narrative review and a suggested 2-step score-based diagnostic pathway for clinicians. Open Forum Infect. Dis. 2014, 1, ofu060. [Google Scholar] [CrossRef] [PubMed]
- Newman, L.G.; Waller, J.; Palestro, C.J.; Schwartz, M.; Klein, M.J.; Hermann, G.; Harrington, E.; Harrington, M.; Roman, S.H.; Stagnaro-Green, A. Unsuspected osteomyelitis in diabetic foot ulcers. Diagnosis and monitoring by leukocyte scanning with indium in 111 oxyquinoline. JAMA 1991, 266, 1246–1251. [Google Scholar] [CrossRef] [PubMed]
- Lazaro, J.L.; Izzo, V.; Meaume, S.; Davies, A.H.; Lobmann, R.; Uccioli, L. Elevated levels of matrix metalloproteinases and chronic wound healing: An updated review of clinical evidence. J. Wound Care 2016, 25, 277–287. [Google Scholar] [CrossRef] [PubMed]
- McCarty, S.M.; Cochrane, C.A.; Clegg, P.D.; Percival, S.L. The role of endogenous and exogenous enzymes in chronic wounds: A focus on the implications of aberrant levels of both host and bacterial proteases in wound healing. Wound Repair Regen. 2012, 20, 125–136. [Google Scholar] [CrossRef] [PubMed]
- Ren, Y.; Gu, G.; Yao, M.; Driver, V.R. Role of matrix metalloproteinases in chronic wound healing: Diagnostic and therapeutic implications. Chin. Med. J. 2014, 127, 1572–1581. [Google Scholar] [PubMed]
- Chang, M. Restructuring of the extracellular matrix in diabetic wounds and healing: A perspective. Pharmacol. Res. 2016, 107, 243–248. [Google Scholar] [CrossRef] [PubMed]
- Visse, R.; Nagase, H. Matrix metalloproteinases and tissue inhibitors of metalloproteinases: Structure, function, and biochemistry. Circ. Res. 2003, 92, 827–839. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Guo, S.; Yao, F.; Zhang, Y.; Li, T. Increased ratio of serum matrix metalloproteinase-9 against TIMP-1 predicts poor wound healing in diabetic foot ulcers. J. Diabetes Complicat. 2013, 27, 380–382. [Google Scholar] [CrossRef] [PubMed]
- Yager, D.R.; Zhang, L.Y.; Liang, H.X.; Diegelmann, R.F.; Cohen, I.K. Wound fluids from human pressure ulcers contain elevated matrix metalloproteinase levels and activity compared to surgical wound fluids. J. Investig. Dermatol. 1996, 107, 743–748. [Google Scholar] [CrossRef] [PubMed]
- Saito, S.; Trovato, M.J.; You, R.; Lal, B.K.; Fasehun, F.; Padberg, F.T., Jr.; Hobson, R.W., 2nd; Durán, W.N.; Pappas, P.J. Role of matrix metalloproteinases 1, 2, and 9 and tissue inhibitor of matrix metalloproteinase-1 in chronic venous insufficiency. J. Vasc. Surg. 2001, 34, 930–938. [Google Scholar] [CrossRef] [PubMed]
- Lobmann, R.; Ambrosch, A.; Schultz, G.; Waldmann, K.; Schiweck, S.; Lehnert, H. Expression of matrix-metalloproteinases and their inhibitors in the wounds of diabetic and non-diabetic patients. Diabetologia 2002, 45, 1011–1016. [Google Scholar] [CrossRef] [PubMed]
- Grinnell, F.; Zhu, M. Fibronectin degradation in chronic wounds depends on the relative levels of elastase, alpha1-proteinase inhibitor, and alpha2-macroglobulin. J. Investig. Dermatol. 1996, 106, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Tentolouris, N.; Petrikkos, G.; Vallianou, N.; Zachos, C.; Daikos, G.L.; Tsapogas, P.; Markou, G.; Katsilambros, N. Prevalence of methicillin-resistant Staphylococcus aureus in infected and uninfected diabetic foot ulcers. Clin. Microbiol. Infect. 2006, 12, 186–189. [Google Scholar] [CrossRef] [PubMed]
- Game, F.; Jeffcoate, W. MRSA and osteomyelitis of the foot in diabetes. Diabet. Med. 2004, 21, 16–19. [Google Scholar] [CrossRef] [PubMed]
- Dang, C.N.; Prasad, Y.D.; Boulton, A.J.; Jude, E.B. Methicillin-resistant Staphylococcus aureus in the diabetic foot clinic: A worsening problem. Diabet. Med. 2003, 20, 159–161. [Google Scholar] [CrossRef] [PubMed]
- Barshes, N.R.; Rodriguez-Barradas, M.C.; Bechara, C.F.; Pisimisis, G.; Young, E.J.; Kougias, P. Microbial isolates and their antimicrobial susceptibilities in inframalleolar foot infections. Surg. Infect. 2014, 15, 585–591. [Google Scholar] [CrossRef] [PubMed]
- Citron, D.M.; Goldstein, E.J.; Merriam, C.V.; Lipsky, B.A.; Abramson, M.A. Bacteriology of moderate-to-severe diabetic foot infections and in vitro activity of antimicrobial agents. J. Clin. Microbiol. 2007, 45, 2819–2828. [Google Scholar] [CrossRef] [PubMed]
- Lavery, L.A.; Harkless, L.B.; Felder-Johnson, K.; Mundine, S. Bacterial pathogens in infected puncture wounds in adults with diabetes. J. Foot Ankle Surg. 1994, 33, 91–97. [Google Scholar] [PubMed]
- Lavery, L.A.; Sariaya, M.; Ashry, H.; Harkless, L.B. Microbiology of osteomyelitis in diabetic foot infections. J. Foot Ankle Surg. 1995, 34, 61–64. [Google Scholar] [CrossRef]
- Djahmi, N.; Messad, N.; Nedjai, S.; Moussaoui, A.; Mazouz, D.; Richard, J.L.; Sotto, A.; Lavigne, J.P. Molecular epidemiology of Staphylococcus aureus strains isolated from inpatients with infected diabetic foot ulcers in an Algerian University Hospital. Clin. Microb. Infect. 2013, 19, E398–E404. [Google Scholar] [CrossRef] [PubMed]
- Richard, J.L.; Lavigne, J.P.; Got, I.; Hartemann, A.; Malgrange, D.; Tsirtsikolou, D.; Baleydier, A.; Senneville, E. Management of patients hospitalized for diabetic foot infection: Results of the French OPIDIA study. Diabetes Metab. 2011, 37, 208–215. [Google Scholar] [CrossRef] [PubMed]
- Al Benwan, K.; Al Mulla, A.; Rotimi, V.O. A study of the microbiology of diabetic foot infections in a teaching hospital in Kuwai. J. Infect. Public Health 2012, 5, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Akhi, M.T.; Ghotaslou, R.; Asgharzadeh, M.; Varshochi, M.; Pirzadeh, T.; Memar, M.Y.; Zahedi Bialvaei, A.; Seifi Yarijan Sofla, H.; Alizadeh, N. Bacterial etiology and antibiotic susceptibility pattern of diabetic foot infections in Tabriz, Iran. GMS Hyg. Infect. Control 2015, 10, Doc02. [Google Scholar] [CrossRef] [PubMed]
- Anvarinejad, M.; Pouladfar, G.; Japoni, A.; Bolandparvaz, S.; Satiary, Z.; Abbasi, P.; Mardaneh, J. Isolation and antibiotic susceptibility of the microorganisms isolated from diabetic foot infections in Nemazee Hospital, Southern Iran. J. Pathog. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Alavi, S.M.; Khosravi, A.D.; Sarami, A.; Dashtebozorg, A.; Montazeri, E.A. Bacteriologic study of diabetic foot ulcer. Pak. J. Med. Sci. 2007, 235, 681–684. [Google Scholar] [CrossRef]
- Raja, N.S. Microbiology of diabetic foot infections in a teaching hospital in Malaysia: A retrospective study of 194 cases. J. Microbiol. Immunol. Infect. 2007, 40, 39–44. [Google Scholar] [PubMed]
- Yoga, R.; Khairul, A.; Sunita, K.; Suresh, C. Bacteriology of diabetic foot lesions. Med. J. Malays. 2006, 61, 14–16. [Google Scholar]
- Martinez-Gomez Dde, A.; Ramirez-Almagro, C.; Campillo-Soto, A.; Morales-Cuenca, G.; Pagan-Ortiz, J.; Aguayo-Albasini, J.L. Diabetic foot infections. Prevalence and antibiotic sensitivity of the causative microorganisms. Enferm. Infecc. Microbiol. Clin. 2009, 27, 317–321. [Google Scholar] [PubMed]
- Perim, M.C.; Bordes Jda, C.; Celeste, S.R.; Orsolin Ede, F.; Mendes, R.R.; Mendes, G.O.; Ferreira, R.L.; Carreiro, S.C.; Pranchevicius, M.C. Aerobic bacterial profile and antibiotic resistance in patients with diabetic foot infections. Rev. Soc. Bras. Med. Trop. 2015, 48, 546–554. [Google Scholar] [CrossRef] [PubMed]
- Shanmugam, P.; Jeya, M.; Linda Susan, S. The bacteriology of diabetic foot ulcers, with a special reference to multidrug resistant strains. J. Clin. Diagn. Res. 2013, 7, 441–445. [Google Scholar] [CrossRef] [PubMed]
- Sugandhi, P.; Prasanth, D.A. Microbiological profile of bacterial pathogens from diabetic foot infections in tertiary care hospitals, Salem. Diabetes Metab. Syndr. 2014, 8, 129–132. [Google Scholar] [CrossRef] [PubMed]
- Mathangi, T.; Prabhakaran, P.; Rayappan, F.; Tilton, F. Isolation, molecular characterization and antibiogram of bacteria isolated from DFU. Int. J. Curr. Res. Acad. Rev. 2013, 1, 17–25. [Google Scholar]
- Sekhar, S.M.; Vyas, N.; Unnikrishnan, M.K.; Rodrigues, G.S.; Mukhopadhyay, C. Antimicrobial susceptibility pattern in diabetic foot ulcer: A pilot study. Ann. Med. Health Sci. Res. 2014, 4, 742–745. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.H.; Sun, Z.L.; Guo, Y.J.; Yang, B.Q.; Yuan, Y.; Wei, Q.; Ye, K.P. Meticillin-resistant Staphylococcus aureus isolated from foot ulcers in diabetic patients in a Chinese care hospital: Risk factors for infection and prevalence. J. Med. Microbiol. 2010, 59, 1219–1224. [Google Scholar] [CrossRef] [PubMed]
- Ding, Q.; Li, D.Q.; Wang, P.H.; Chu, Y.J.; Meng, S.Y.; Sun, Q. Risk factors for infections of methicillin-resistant Staphylococci in diabetic foot patients. Zhonhua Yi Xue Za Zhi 2012, 92, 228–231. [Google Scholar]
- Sharma, V.K.; Khadka, P.B.; Joshi, A.; Sharma, R. Common pathogens isolated in diabetic foot infection in Bir Hospital. Kathmandu Univ. Med. J. 2006, 4, 295–301. [Google Scholar]
- Islam, S.; Cawich, S.O.; Budhooram, S.; Harnarayan, P.; Mahabir, V.; Ramsewak, S.; Naraynsingh, V. Microbial profile of diabetic foot infections in Trinidad and Tobago. Prim. Care Diabetes 2013, 7, 303–308. [Google Scholar] [CrossRef] [PubMed]
- Mendes, J.J.; Marques-Costa, A.; Vilela, C.; Neves, J.; Candeias, N.; Cavaco-Silva, P.; Melo-Cristino, J. Clinical and bacteriological survey of diabetic foot infections in Lisbon. Diabetes Res. Clin. Pract. 2012, 95, 153–161. [Google Scholar] [CrossRef] [PubMed]
- Radji, M.; Putri, C.S.; Fauziyah, S. Antibiotic therapy for diabetic foot infections in a tertiary care hospital in Jakarta, Indonesia. Diabetes Metab. Syndr. 2014, 8, 221–224. [Google Scholar] [CrossRef] [PubMed]
- El-Tahawy, A.T. Bacteriology of diabetic foot. Saudi Med. J. 2000, 21, 344–347. [Google Scholar] [PubMed]
- Guira, O.; Tiéno, H.; Traoré, S.; Diallo, I.; Ouangré, E.; Sagna, Y.; Zabsonré, J.; Yanogo, D.; Traoré, S.S.; Drabo, Y.J. The bacterial microflora of diabetic foot infection and factors determining its spectrum in Ouagadougou (Burkina Faso). Bull. Soc. Pathol. Exot. 2015, 108, 307–311. [Google Scholar] [CrossRef] [PubMed]
- Unachukwu, C.N.; Obunge, O.K.; Odia, O.J. The bacteriology of diabetic foot ulcers in Port Harcourt, Nigeria. Niger. J. Med. 2005, 14, 173–176. [Google Scholar] [PubMed]
- Katz, D.E.; Friedman, N.D.; Ostrovski, E.; Ravid, D.; Amrami, N.; Avivi, D.; Mengesha, B.; Zaidenstein, R.; Lazarovitch, T.; Dadon, M.; et al. Diabetic foot infection in hospitalized adults. J. Infect. Chemother. 2016, 22, 167–173. [Google Scholar] [CrossRef] [PubMed]
- Cervantes-Garcia, E.; García-González, R.; Reséndiz-Albor, A.; Salazar-Schettino, P.M. Infections of diabetic foot ulcers with methicillin-resistant Staphylococcus aureus. Int. J. Low. Extrem. Wounds 2015, 14, 44–49. [Google Scholar] [CrossRef] [PubMed]
- Lelievre, H.; Lina, G.; Jones, M.E.; Olive, C.; Forey, F.; Roussel-Delvallez, M.; Nicolas-Chanoine, M.H.; Bébéar, C.M.; Jarlier, V.; Andremont, A.; et al. Emergence and spread in French hospitals of methicillin-resistant Staphylococcus aureus with increasing susceptibility to gentamicin and other antibiotics. J. Clin. Microbiol. 1999, 37, 3452–3457. [Google Scholar] [PubMed]
- Ertugrul, B.M.; Oncul, O.; Tulek, N.; Willke, A.; Sacar, S.; Tunccan, O.G.; Yilmaz, E.; Kaya, O.; Ozturk, B.; Turhan, O.; et al. A prospective, multi-center study: Factors related to the management of diabetic foot infections. Eur. J. Clin. Microbiol. Infect. Dis. 2012, 31, 2345–2352. [Google Scholar] [CrossRef] [PubMed]
- Leclercq, R. Epidemiological and resistance issues in multidrug-resistant staphylococci and enterococci. Clin. Microbiol. Infect. 2009, 15, 224–231. [Google Scholar] [CrossRef] [PubMed]
- Société de Pathologie Infectieuse de Langue Française. Management of diabetic foot infections. Short text. Med. Mal. Infect. 2007, 37, 1–25. [Google Scholar]
- American Diabetes Association. Consensus development conference on diabetic foot wound care. Diabetes Care 1999, 22, 1354–1360. [Google Scholar]
- Eleftheriadou, I.; Tentolouris, N.; Argiana, V.; Jude, E.; Boulton, A.J. Methicillin-resistant Staphylococcus aureus in diabetic foot infections. Drugs 2010, 70, 1785–1797. [Google Scholar] [CrossRef] [PubMed]
- Lagacé-Wiens, P.R.; Ormiston, D.; Nicolle, L.E.; Hilderman, T.; Embil, J. The diabetic foot clinic: Not a significant source for acquisition of methicillin-resistant Staphylococcus aureus. Am. J. Infect. Control 2009, 37, 587–589. [Google Scholar] [CrossRef] [PubMed]
- Pittet, D.; Hugonnet, S.; Harbarth, S.; Mourouga, P.; Sauvan, V.; Touveneau, S.; Perneger, T.V. Effectiveness of a hospital-wide programme to improve compliance with hand hygiene. Lancet 2000, 356, 1307–1312. [Google Scholar] [CrossRef]
- Zenelaj, B.; Bouvet, C.; Lipsky, B.A.; Uçkay, I. Do diabetic foot infections with methicillin-resistant Staphylococcus aureus differ from those with other pathogens? Int. J. Low. Extrem. Wounds 2014, 13, 263–272. [Google Scholar] [CrossRef] [PubMed]
- Richard, J.L.; Sotto, A.; Jourdan, N.; Combescure, C.; Vannereau, D.; Rodier, M.; Lavigne, J.P. Risk factors and healing impact of multidrug-resistant bacteria in diabetic foot ulcers. Diabetes Metab. 2008, 34, 363–369. [Google Scholar] [CrossRef] [PubMed]
- Aragón-Sánchez, J.; Lázaro-Martínez, J.L.; Quintana-Marrero, Y.; Hernández-Herrero, M.J.; García-Morales, E.; Cabrera-Galván, J.J.; Beneit-Montesinos, J.V. Are diabetic foot ulcers complicated by MRSA osteomyelitis associated with worse prognosis? Outcomes of a surgical series. Diabet. Med. 2009, 26, 552–555. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.; Sievert, D.M.; Hageman, J.C.; Boulton, M.L.; Tenover, F.C.; Downes, F.P.; Shah, S.; Rudrik, J.T.; Pupp, G.R.; Brown, W.J.; et al. Infection with vancomycin-resistant Staphylococcus aureus containing the vanA resistance gene. N. Engl. J. Med. 2003, 348, 1342–1347. [Google Scholar] [CrossRef] [PubMed]
- Dezfulian, A.; Aslani, M.M.; Oskoui, M.; Farrokh, P.; Azimirad, M.; Dabiri, H.; Salehian, M.T.; Zali, M.R. Identification and characterization of a high vancomycin-resistant Staphylococcus aureus harboring vanA gene cluster isolated from diabetic foot ulcer. Iran. J. Basic Med. Sci. 2012, 15, 803–806. [Google Scholar] [PubMed]
- Foster, T.J. Immune evasion by staphylococci. Nat. Rev. Microbiol. 2005, 3, 948–958. [Google Scholar] [CrossRef] [PubMed]
- Ambrosch, A.; Haefner, S.; Jude, E.; Lobmann, R. Diabetic foot infections: Microbiological aspects, current and future antibiotic therapy focusing on methicillin-resistant Staphylococcus aureus. Int. Wound J. 2011, 8, 567–577. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, R.; Cockayne, A.; Humphreys, H. Clinical and molecular aspects of the pathogenesis of Staphylococcus aureus bone and joint infections. J. Med. Microbiol. 1996, 44, 157–164. [Google Scholar] [CrossRef] [PubMed]
- Clarke, S.R.; Brummell, K.J.; Horsburgh, M.J.; McDowell, P.W.; Mohamad, S.A.; Stapleton, M.R.; Acevedo, J.; Read, R.C.; Day, N.P.; Peacock, S.J.; et al. Identification of in vivo-expressed antigens of Staphylococcus aureus and their use in vaccinations for protection against nasal carriage. J. Infect. Dis. 2006, 193, 1098–1108. [Google Scholar] [CrossRef] [PubMed]
- Weidenmaier, C.; Kokai-Kun, J.F.; Kristian, S.A.; Chanturiya, T.; Kalbacher, H.; Gross, M.; Nicholson, G.; Neumeister, B.; Mond, J.J.; Peschel, A. Role of teichoic acids in Staphylococcus aureus nasal colonization, a major risk factor in nosocomial infections. Nat. Med. 2004, 10, 243–245. [Google Scholar] [CrossRef] [PubMed]
- Burian, M.; Rautenberg, M.; Kohler, T.; Fritz, M.; Krismer, B.; Unger, C.; Hoffmann, W.H.; Peschel, A.; Wolz, C.; Goerke, C. Temporal expression of adhesion factors and activity of global regulators during establishment of Staphylococcus aureus nasal colonization. J. Infect. Dis. 2010, 201, 1414–1421. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.H.; Strickland, I.; Boguniewicz, M.; Leung, D.Y. Fibronectin and fibrinogen contribute to the enhanced binding of Staphylococcus aureus to atopic skin. J. Allergy Clin. Immunol. 2001, 108, 269–274. [Google Scholar] [CrossRef] [PubMed]
- Shi, S.; Zhang, X. Interaction of Staphylococcus aureus with osteoblasts. Exp. Ther. Med. 2012, 3, 367–370. [Google Scholar] [PubMed]
- Proctor, R.A.; von Eiff, C.; Kahl, B.C.; Becker, K.; McNamara, P.; Herrmann, M.; Peters, G. Small colony variants: A pathogenic form of bacteria that facilitates persistent and recurrent infections. Nat. Rev. Microbiol. 2006, 4, 295–305. [Google Scholar] [CrossRef] [PubMed]
- Tuchscherr, L.; Heitmann, V.; Hussain, M.; Viemann, D.; Roth, J.; von Eiff, C.; Peters, G.; Becker, K.; Löffler, B. Staphylococcus aureus small-colony variants are adapted phenotypes for intracellular persistence. J. Infect. Dis. 2010, 202, 1031–1040. [Google Scholar] [CrossRef] [PubMed]
- Von Eiff, C.; Becker, K.; Metze, D.; Lubritz, G.; Hockmann, J.; Schwarz, T.; Peters, G. Intracellular persistence of Staphylococcus aureus small-colony variants within keratinocytes: A cause for antibiotic treatment failure in a patient with darier’s disease. Clin. Infect. Dis. 2001, 32, 1643–1647. [Google Scholar] [CrossRef] [PubMed]
- Baumert, N.; von Eiff, C.; Schaaff, F.; Peters, G.; Proctor, R.A.; Sahl, H.G. Physiology and antibiotic susceptibility of Staphylococcus aureus small colony variants. Microb. Drug Res. 2002, 8, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Garcia, L.G.; Lemaire, S.; Kahl, B.C.; Becker, K.; Proctor, R.A.; Denis, O.; Tulkens, P.M.; Van Bambeke, F. Antibiotic activity against small-colony variants of Staphylococcus aureus: Review of in vitro, animal and clinical data. J. Antimicrob. Chemother. 2013, 68, 1455–1464. [Google Scholar] [CrossRef] [PubMed]
- Proctor, R.A.; Peters, G. Small colony variants in staphylococcal infections: Diagnostic and therapeutic implications. Clin. Infect. Dis. 1998, 27, 419–422. [Google Scholar] [CrossRef] [PubMed]
- Podbielska, A.; Galbowska, H.; Stelmach, E.; Mlynarczyk, G.; Olszewski, W.L. Slime production by Staphylococcus aureus and Staphylococcus epidermidis strains isolated from patients with diabetic foot ulcers. Arch. Immunol. Ther. Exp. 2010, 58, 321–324. [Google Scholar] [CrossRef] [PubMed]
- Bergamini, T.M.; Corpus, R.A.; Hoeg, K.L.; Brittian, K.R.; Peyton, J.C.; Cheadle, W.G. Immune regulation of bacterial biofilm graft infection. ASAIO J. 1994, 40, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Steinberg, D.; Poran, S.; Shapira, L. The effect of extracellular polysaccharides from Streptococcus mutans on the bactericidal activity of human neutrophils. Arch. Oral Biol. 1999, 44, 437–444. [Google Scholar] [CrossRef]
- Yasuda, H.; Ajiki, Y.; Aoyama, J.; Yokota, T. Interaction between human polymorphonuclear leucocytes and bacteria released from in vitro bacterial biofilm models. J. Med. Microbiol. 1994, 41, 359–367. [Google Scholar] [CrossRef] [PubMed]
- Bjarnsholt, T.; Kirketerp-Moller, K.; Jensen, P.O.; Madsen, K.G.; Philipps, R.; Krogfelt, K.; Hoiby, N.; Givskov, M. Why chronic wounds will not heal: A novel hypothesis. Wound Repair. Regen. 2008, 16, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Vandenesch, F.; Lina, G.; Henry, T. Staphylococcus aureus hemolysins, bi-component leukocidins, and cytolytic peptides: A redundant arsenal of membrane-damaging virulence factors? Front. Cell. Infect. Microbiol. 2012, 2, 12. [Google Scholar] [CrossRef] [PubMed]
- Grumann, D.; Nubel, U.; Broker, B.M. Staphylococcus aureus toxins—Their functions and genetics. Infect. Genet. Evol. 2014, 21, 583–592. [Google Scholar] [CrossRef] [PubMed]
- Otto, M. Staphylococcus aureus toxins. Curr. Opin. Microbiol. 2014, 17, 32–37. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Y.Q.; Willard, J.; Yeaman, M.R.; Cheung, A.L.; Bayer, A.S. Regulation of Staphylococcus aureus alpha-toxin gene (hla) expression by agr, sarA, and sae in vitro and in experimental infective endocarditis. J. Infect. Dis. 2006, 194, 1267–1275. [Google Scholar] [CrossRef] [PubMed]
- Valeva, A.; Walev, I.; Pinkernell, M.; Walker, B.; Bayley, H.; Palmer, M.; Bhakdi, S. Transmembrane beta-barrel of staphylococcal alpha-toxin forms in sensitive but not in resistant cells. Proc. Natl. Acad. Sci. USA 1997, 94, 11607–11611. [Google Scholar] [CrossRef] [PubMed]
- Sotto, A.; Richard, J.L.; Messad, N.; Molinari, N.; Jourdan, N.; Schuldiner, S.; Sultan, A.; Carrière, C.; Canivet, B.; Landraud, L.; et al. Distinguishing colonization from infection with Staphylococcus aureus in diabetic foot ulcers with miniaturized oligonucleotide arrays: A French multicenter study. Diabetes Care 2012, 35, 617–623. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Braughton, K.R.; Kretschmer, D.; Bach, T.H.; Queck, S.Y.; Li, M.; Kennedy, A.D.; Dorward, D.W.; Klebanoff, S.J.; Peschel, A.; et al. Identification of novel cytolytic peptides as key virulence determinants for community-associated MRSA. Nat. Med. 2007, 13, 1510–1514. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Diep, B.A.; Villaruz, A.E.; Braughton, K.R.; Jiang, X.; DeLeo, F.R.; Chambers, H.F.; Lu, Y.; Otto, M. Evolution of virulence in epidemic community-associated methicillin-resistant Staphylococcus aureus. Proc. Natl. Acad. Sci. USA 2009, 106, 5883–5888. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, S.S.; Joo, H.S.; Duong, A.C.; Dieringer, T.D.; Tan, V.Y.; Song, Y.; Fischer, E.R.; Cheung, G.Y.; Li, M.; Otto, M. Essential Staphylococcus aureus toxin export system. Nat. Med. 2013, 19, 364–367. [Google Scholar] [CrossRef] [PubMed]
- Surewaard, B.G.; de Haas, C.J.; Vervoort, F.; Rigby, K.M.; DeLeo, F.R.; Otto, M.; van Strijp, J.A.; Nijland, R. Staphylococcal alpha-phenol soluble modulins contribute to neutrophil lysis after phagocytosis. Cell. Microbiol. 2013, 15, 1427–1437. [Google Scholar] [CrossRef] [PubMed]
- Dumont, A.L.; Nygaard, T.K.; Watkins, R.L.; Smith, A.; Kozhaya, L.; Kreiswirth, B.N.; Shopsin, B.; Unutmaz, D.; Voyich, J.M.; Torres, V.J. Characterization of a new cytotoxin that contributes to Staphylococcus aureus pathogenesis. Mol. Microbiol. 2011, 79, 814–825. [Google Scholar] [CrossRef] [PubMed]
- Ventura, C.L.; Malachowa, N.; Hammer, C.H.; Nardone, G.A.; Robinson, M.A.; Kobayashi, S.D.; DeLeo, F.R. Identification of a novel Staphylococcus aureus two-component leukotoxin using cell surface proteomics. PLoS ONE 2010, 5, e11634. [Google Scholar] [CrossRef] [PubMed]
- Panton, P.N.; Valentine, F.C.O. Staphylococcal toxin. Lancet 1932, 219, 506–508. [Google Scholar] [CrossRef]
- Gravet, A.; Colin, D.A.; Keller, D.; Girardot, R.; Monteil, H.; Prevost, G. Characterization of a novel structural member, LukE-LukD, of the bi-component staphylococcal leucotoxins family. FEBS Lett. 1998, 436, 202–208. [Google Scholar] [CrossRef]
- Morinaga, N.; Kaihou, Y.; Noda, M. Purification, cloning and characterization of variant LukE-LukD with strong leukocidal activity of staphylococcal bi-component leukotoxin family. Microbiol. Immunol. 2003, 47, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, I.M.; Hartford, O.; Foster, T.; Tarkowski, A. Alpha-toxin and gamma-toxin jointly promote Staphylococcus aureus virulence in murine septic arthritis. Inf. Immun. 1999, 67, 1045–1049. [Google Scholar]
- Malachowa, N.; Whitney, A.R.; Kobayashi, S.D.; Sturdevant, D.E.; Kennedy, A.D.; Braughton, K.R.; Shabb, D.W.; Diep, B.A.; Chambers, H.F.; Otto, M.; et al. Global changes in Staphylococcus aureus gene expression in human blood. PLoS ONE 2011, 6, e18617. [Google Scholar] [CrossRef] [PubMed]
- Sotto, A.; Lina, G.; Richard, J.L.; Combescure, C.; Bourg, G.; Vidal, L.; Jourdan, N.; Etienne, J.; Lavigne, J.P. Virulence potential of Staphylococcus aureus strains isolated from diabetic foot ulcers. Diabetes Care 2008, 31, 2318–2324. [Google Scholar] [CrossRef] [PubMed]
- Boyle-Vavra, S.; Daum, R.S. Community-acquired methicillin-resistant Staphylococcus aureus: The role of Panton-Valentine leukocidin. Lab. Investig. 2007, 87, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Kaneko, J.; Kimura, T.; Kawakami, Y.; Tomita, T.; Kamio, Y. Panton-valentine leukocidin genes in a phage-like particle isolated from mitomycin C-treated Staphylococcus aureus V8 (ATCC 49775). Biosci. Biotechnol. Biochem. 1997, 61, 1960–1962. [Google Scholar] [CrossRef] [PubMed]
- Narita, S.; Kaneko, J.; Chiba, J.; Piemont, Y.; Jarraud, S.; Etienne, J.; Kamio, Y. Phage conversion of Panton-Valentine leukocidin in Staphylococcus aureus: Molecular analysis of a PVL-converting phage, phiSLT. Gene 2001, 268, 195–206. [Google Scholar] [CrossRef]
- Dupieux, C.; Blondé, R.; Bouchiat, C.; Meugnier, H.; Bes, M.; Laurent, S.; Vandenesch, F.; Laurent, F.; Tristan, A. Community-acquired infections due to Staphylococcus argenteus lineage isolates harbouring the Panton-Valentine leucocidin, France, 2014. Euro Surveill. 2015, 20, 21154. [Google Scholar] [CrossRef] [PubMed]
- Shallcross, L.J.; Fragaszy, E.; Johnson, A.M.; Hayward, A.C. The role of the Panton-Valentine leucocidin toxin in staphylococcal disease: A systematic review and meta-analysis. Lancet Infect. Dis. 2013, 13, 43–54. [Google Scholar] [CrossRef]
- Gillet, Y.; Issartel, B.; Vanhems, P.; Fournet, J.C.; Lina, G.; Bes, M.; Vandenesch, F.; Piemont, Y.; Brousse, N.; Floret, D.; et al. Association between Staphylococcus aureus strains carrying gene for Panton-Valentine leukocidin and highly lethal necrotising pneumonia in young immunocompetent patients. Lancet 2002, 359, 753–759. [Google Scholar] [CrossRef]
- Vandenesch, F.; Naimi, T.; Enright, M.C.; Lina, G.; Nimmo, G.R.; Heffernan, H.; Liassine, N.; Bes, M.; Greenland, T.; Reverdy, M.E.; et al. Community- acquired methicillin-resistant Staphylococcus aureus carrying Panton-Valentine leukocidin genes: Worldwide emergence. Emerg. Infect. Dis. 2003, 9, 978–984. [Google Scholar] [CrossRef] [PubMed]
- Shukla, S.K.; Karow, M.E.; Brady, J.M.; Stemper, M.E.; Kislow, J.; Moore, N.; Wroblewski, K.; Chyou, P.H.; Warshauer, D.M.; Reed, K.D.; et al. Virulence genes and genotypic associations in nasal carriage, community-associated methicillin- susceptible and methicillin-resistant USA400 Staphylococcus aureus isolates. J. Clin. Microbiol. 2010, 48, 3582–3592. [Google Scholar] [CrossRef] [PubMed]
- Kiliç, A.; Doğan, E.; Kaya, S.; Baysallar, M. Investigation of the presence of mecC and Panton-Valentine leukocidin genes in Staphylococcus aureus strains isolated from clinical specimens during seven years period. Mikrobiyol. Bulteni 2015, 49, 594–599. [Google Scholar] [CrossRef]
- Santosaningsih, D.; Santoso, S.; Budayanti, N.S.; Suata, K.; Lestari, E.S.; Wahjono, H.; Djamal, A.; Kuntaman, K.; van Belkum, A.; Laurens, M.; et al. Characterisation of clinical Staphylococcus aureus isolates harbouring mecA or Panton-Valentine leukocidin genes from four tertiary care hospitals in Indonesia. Trop. Med. Int. Health 2016, 21, 610–618. [Google Scholar] [CrossRef] [PubMed]
- Motamedi, H.; Rahmat Abadi, S.S.; Moosavian, S.M.; Torabi, M. The association of Panton-Valentine leukocidin and mecA genes in Methicillin-Resistant Staphylococcus aureus isolates from patients referred to Educational Hospitals in Ahvaz, Iran. Jundishapur J. Microbiol. 2015, 8, e22021. [Google Scholar] [CrossRef] [PubMed]
- Yu, F.; Liu, Y.; Lv, J.; Qi, X.; Lu, C.; Ding, Y.; Li, D.; Liu, H.; Wang, L. Antimicrobial susceptibility, virulence determinant carriage and molecular characteristics of Staphylococcus aureus isolates associated with skin and soft tissue infections. Braz. J. Infect. Dis. 2015, 19, 614–622. [Google Scholar] [CrossRef] [PubMed]
- Conceição, T.; Coelho, C.; de Lencastre, H.; Aires-de-Sousa, M. Frequent occurrence of oxacillin-susceptible mecA-positive Staphylococcus aureus (OS-MRSA) strains in two African countries. J. Antimicrob. Chemother. 2015, 70, 3200–3204. [Google Scholar] [PubMed]
- Chen, B.; Dai, X.; He, B.; Pan, K.; Li, H.; Liu, X.; Bao, Y.; Lao, W.; Wu, X.; Yao, Y.; Huang, S. Differences in Staphylococcus aureus nasal carriage and molecular characteristics among community residents and healthcare workers at Sun Yat-Sen University, Guangzhou, Southern China. BMC Infect. Dis. 2015, 15, 303. [Google Scholar] [CrossRef] [PubMed]
- Morgan, M.S. Diagnosis and treatment of Panton-Valentine leukocidin (PVL)-associated staphylococcal pneumonia. Int. J. Antimicrob. Agents 2007, 30, 289–296. [Google Scholar] [CrossRef] [PubMed]
- Rossney, A.S.; Shore, A.C.; Morgan, P.M.; Fitzgibbon, M.M.; O’Connell, B.; Coleman, D.C. The emergence and importation of diverse genotypes of methicillin-resistant Staphylococcus aureus (MRSA) harboring the Panton-Valentine leukocidin gene (pvl) reveal that pvl is a poor marker for community-acquired MRSA strains in Ireland. J. Clin. Microbiol. 2007, 45, 2554–2563. [Google Scholar] [CrossRef] [PubMed]
- Abdulgader, S.M.; Shittu, A.O.; Nicol, M.P.; Kaba, M. Molecular epidemiology of Methicillin-resistant Staphylococcus aureus in Africa: A systematic review. Front. Microbiol. 2015, 6, 348. [Google Scholar] [CrossRef] [PubMed]
- Bazzi, A.M.; Rabaan, A.A.; Fawarah, M.M.; Al-Tawfiq, J.A. Prevalence of Panton-Valentine leukocidin-positive methicillin-susceptible Staphylococcus aureus infections in a Saudi Arabian hospital. J. Infect. Public Health 2015, 8, 364–368. [Google Scholar] [CrossRef] [PubMed]
- Ohadian Moghadam, S.; Pourmand, M.R.; Mahmoudin, M.; Sadighian, H. Molecular characterization of methicillin-resistant Staphylococcus aureus: Characterization of major clones and emergence of epidemic clones of sequence type (ST) 36 and ST 121 in Tehran, Iran. FEMS Microbiol. Lett. 2015, 362, fnv043. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Liang, J.; Jiang, Y.; Wang, B.; Yuan, H.; Zhang, L.; Zhou, Y.; Xu, H.; Zhou, W. Molecular characteristics of community-acquired methicillin-resistant Staphylococcus aureus strains isolated from outpatients with skin and soft tissue infections in Wuhan, China. Pathog. Dis. 2016, 74, ftw026. [Google Scholar] [CrossRef] [PubMed]
- David, M.Z.; Acree, M.E.; Sieth, J.J.; Boxrud, D.J.; Dobbins, G.; Lynfield, R.; Boyle-Vavra, S.; Daum, R.S. Pediatric Staphylococcus aureus isolate genotypes and infections from the dawn of the Community-Associated Methicillin-Resistant S. aureus epidemic era in Chicago, 1994 to 1997. J. Clin. Microbiol. 2015, 53, 2486–2491. [Google Scholar] [CrossRef] [PubMed]
- Post, V.; Wahl, P.; Uçkay, I.; Ochsner, P.; Zimmerli, W.; Corvec, S.; Loiez, C.; Richards, R.G.; Moriarty, F. Phenotypic and genotypic characterisation of Staphylococcus aureus causing musculoskeletal infections. Int. J. Med. Microbiol. 2014, 304, 565–576. [Google Scholar] [CrossRef] [PubMed]
- Dunyach-Remy, C.; Courtais-Coulon, C.; DeMattei, C.; Jourdan, N.; Schuldiner, S.; Sultan, A.; Carrière, C.; Alonso, S.; Sotto, A.; Lavigne, J.P. Link between nasal carriage of Staphylococcus aureus and infected diabetic foot ulcers. 2016; Unpublished work. [Google Scholar]
- Stappers, M.H.T.; Hagen, F.; Reimnitz, P.; Mouton, J.W.; Meis, J.F.; Gyssens, I.C. Direct molecular versus culture-based assessment of Gram-positive cocci in biopsies of patients with major abscesses and diabetic foot infections. Eur. J. Clin. Microbiol. Infect. Dis. 2015, 34, 1885–1892. [Google Scholar] [CrossRef] [PubMed]
- Tristan, A.; Bes, M.; Meugnier, H.; Lina, G.; Bozdogan, B.; Courvalin, P.; Reverdy, M.E.; Enright, M.C.; Vandenesch, F.; Etienne, J. Global distribution of Panton-Valentine leukocidine positive methicillin-resistant Staphylococcus aureus, 2006. Emerg. Infect. Dis. 2007, 13, 594–600. [Google Scholar] [CrossRef] [PubMed]
- Antri, K.; Rouzic, N.; Dauwalder, O.; Boubekri, I.; Bes, M.; Lina, G.; Vandenesch, F.; Tazir, M.; Ramdani-Bouguessa, N.; Etienne, J. High prevalence of methicillin-resistant Staphylococcus aureus clone ST80-IV in hospital and community settings in Algiers. Clin. Microbiol. Infect. 2011, 17, 526–532. [Google Scholar] [CrossRef] [PubMed]
- Ben Nejma, M.; Mastouri, M.; Bel Hadj Jrad, B.; Nour, M. Characterization of ST80 Panton-Valentine leukocidin-positive community-acquired methicillin-resistant Staphylococcus aureus clone in Tunisia. Diagn. Microbiol. Infect. Dis. 2008, 77, 20–24. [Google Scholar] [CrossRef] [PubMed]
- Lamy, B.; Laurent, F.; Gallon, O.; Doucet-Populaire, F.; Etienne, J.; Decousser, J.W. Antibacterial resistance, genes encoding toxins and genetic background among Staphylococcus aureus isolated from community-acquired skin and soft tissue infections in France: A national prospective survey. Eur. J. Clin. Microbiol. Infect. Dis. 2012, 31, 1279–1284. [Google Scholar] [CrossRef] [PubMed]
- Durupt, F.; Mayor, L.; Bes, M.; Reverdy, M.E.; Vandenesch, F.; Thomas, L.; Etienne, J. Prevalence of Staphylococcus aureus toxins and nasal carriage in furuncles and impetigo. Br. J. Dermatol. 2007, 157, 1161–1167. [Google Scholar] [CrossRef] [PubMed]
- Issartel, B.; Tristan, A.; Lechevallier, S.; Bruyère, F.; Lina, G.; Garin, B.; Lacassin, F.; Bes, M.; Vandenesch, F.; Etienne, J. Frequent carriage of Panton-Valentine leucocidin genes by Staphylococcus aureus isolates from surgically drained abscesses. J. Clin. Microbiol. 2005, 43, 3203–3207. [Google Scholar] [CrossRef] [PubMed]
- Puzniak, L.A.; Quintana, A.; Wible, M.; Babinchak, T.; McGovern, P.C. Methicillin-resistant Staphylococcus aureus infection epidemiology and clinical response from tigecycline soft tissue infection trials. Diagn. Microbiol. Infect. Dis. 2014, 79, 261–265. [Google Scholar]
- Senneville, E.; Brière, M.; Neut, C.; Messad, N.; Lina, G.; Richard, J.L.; Sotto, A.; Lavigne, J.P.; French Study Group on the Diabetic Foot. First report of the predominance of clonal complex 398 Staphylococcus aureus strains in osteomyelitis complicating diabetic foot ulcers: A national French study. Clin. Microbiol. Infect. 2014, 20, O274–O277. [Google Scholar] [CrossRef] [PubMed]
- Alonzo, F., 3rd; Benson, M.A.; Chen, J.; Novick, R.P.; Shopsin, B.; Torres, V.J. Staphylococcus aureus leucocidin ED contributes to systemic infection by targeting neutrophils and promoting bacterial growth in vivo. Mol. Microbiol. 2012, 83, 423–435. [Google Scholar] [CrossRef] [PubMed]
- Feng, S.H.; Chu, Y.J.; Wang, P.H.; Jun, X.; Min, D.; Li, X.M. Risk factors and gene type for infections of MRSA in diabetic foot patients in Tianjin, China. Int. J. Low. Extrem. Wounds 2013, 12, 106–112. [Google Scholar] [CrossRef] [PubMed]
- Nhan, T.X.; Leclercq, R.; Cattoir, V. Prevalence of toxin genes in consecutive clinical isolates of Staphylococcus aureus and clinical impact. Eur. J. Clin. Microbiol. Infect. Dis. 2011, 30, 719–725. [Google Scholar] [CrossRef] [PubMed]
- Becker, K.; Friedrich, A.W.; Lubritz, G.; Weilert, M.; Peters, G.; von Eiff, C. Prevalence of genes encoding pyrogenic toxin superantigens and exfoliative toxins among strains of Staphylococcus aureus isolated from blood and nasal specimens. J. Clin. Microbiol. 2003, 41, 1434–1439. [Google Scholar] [CrossRef] [PubMed]
- Sila, J.; Sauer, P.; Kolar, M. Comparison of the prevalence of genes coding for enterotoxins, exfoliatins, Panton-Valentine leukocidin and Tsst-1 between methicillin-resistant and methicillin-susceptible isolates of Staphylococcus aureus at the University Hospital in Olomouc. Biomed. Pap. Med. Fac. Univ. Palacky Olomouc Czech Repub. 2009, 153, 215–218. [Google Scholar] [CrossRef] [PubMed]
- Spaulding, A.R.; Salgado-Pabon, W.; Kohler, P.L.; Horswill, A.R.; Leung, D.Y.; Schlievert, P.M. Staphylococcal and streptococcal superantigen exotoxins. Clin. Microbiol. Rev. 2013, 26, 422–447. [Google Scholar] [CrossRef] [PubMed]
- Vu, B.G.; Stach, C.S.; Salgado-Pabon, W.; Diekema, D.J.; Gardner, S.E.; Schlievert, P.M. Superantigens of Staphylococcus aureus from patients with diabetic foot ulcers. J. Infect. Dis. 2014, 210, 1920–1927. [Google Scholar] [CrossRef] [PubMed]
- Lina, G.; Bohach, G.A.; Nair, S.P.; Hiramatsu, K.; Jouvin-Marche, E.; Mariuzza, R. Standard nomenclature for the superantigens expressed by Staphylococcus. J. Infect. Dis. 2004, 189, 2334–2336. [Google Scholar] [CrossRef] [PubMed]
- Diegelmann, R.F.; Evans, M.C. Wound healing: An overview of acute, fibrotic and delayed healing. Front. Biosci. 2004, 9, 283–289. [Google Scholar] [CrossRef] [PubMed]
- Dinges, M.M.; Orwin, P.M.; Schlievert, P.M. Exotoxins of Staphylococcus aureus. Clin. Microbiol. Rev. 2000, 13, 16–34. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.W.; Kim, S.M.; Kim, J.M.; Oh, B.M.; Kim, J.Y.; Jung, H.J.; Lim, H.J.; Kim, B.S.; Lee, W.J.; Lee, S.J.; et al. Potential immunoinflammatory role of staphylococcal enterotoxin A in atopic dermatitis: Immunohistopathological analysis and in vitro assay. Ann. Dermatol. 2013, 25, 173–180. [Google Scholar] [CrossRef] [PubMed]
- Wilson, G.J.; Seo, K.S.; Cartwright, R.A.; Connelley, T.; Chuang-Smith, O.N.; Merriman, J.A.; Guinane, C.M.; Park, J.Y.; Bohach, G.A.; Schlievert, P.M.; et al. A novel core genome-encoded superantigen contributes to lethality of community-associated MRSA necrotizing pneumonia. PLoS Pathog. 2011, 7, e1002271. [Google Scholar] [CrossRef] [PubMed]
- Boquet, P.; Lemichez, E. Bacterial virulence factors targeting Rho GTPases: Parasitism or symbiosis? Trends Cell Biol. 2003, 13, 238–246. [Google Scholar] [CrossRef]
- Boyer, L.; Doye, A.; Rolando, M.; Flatau, G.; Munro, P.; Gounon, P.; Clément, R.; Pulcini, C.; Popoff, M.R.; Mettouchi, A.; et al. Induction of transient macroapertures in endothelial cells through RhoA inhibition by Staphylococcus aureus factors. J. Cell Biol. 2006, 173, 809–819. [Google Scholar] [CrossRef] [PubMed]
- Lemichez, E.; Lecuit, M.; Nassif, X.; Bourdoulous, S. Breaking the wall: Targeting of the endothelium by pathogenic bacteria. Nat. Rev. Microbiol. 2010, 8, 93–104. [Google Scholar] [CrossRef] [PubMed]
- Rolando, M.; Munro, P.; Stefani, C.; Auberger, P.; Flatau, G.; Lemichez, E. Injection of Staphylococcus aureus EDIN by the Bacillus anthracis protective antigen machinery induces vascular permeability. Infect. Immun. 2009, 77, 3596–3601. [Google Scholar] [CrossRef] [PubMed]
- Munro, P.; Benchetrit, M.; Nahori, M.A.; Stefani, C.; Clément, R.; Michiels, J.F.; Landraud, L.; Dussurget, O.; Lemichez, E. The Staphylococcus aureus epidermal cell differentiation inhibitor toxin promotes formation of infection foci in a mouse model of bacteremia. Infect. Immun. 2010, 78, 3404–3411. [Google Scholar] [CrossRef] [PubMed]
- Inoue, S.; Sugai, M.; Murooka, Y.; Paik, S.Y.; Hong, Y.M.; Ohgai, H.; Suginaka, H. Molecular cloning and sequencing of the epidermal cell differentiation inhibitor gene from Staphylococcus aureus. Biochem. Biophys. Res. Commun. 1991, 174, 459–464. [Google Scholar] [CrossRef]
- Wilde, C.; Aktories, K. The Rho-ADP-ribosylating C3 exoenzyme from Clostridium botulinum and related C3-like transferases. Toxicon 2001, 39, 1647–1660. [Google Scholar] [CrossRef]
- Franke, G.C.; Böckenholt, A.; Sugai, M.; Rohde, H.; Aepfelbacher, M. Epidemiology, variable genetic organization and regulation of the EDIN-B toxin in Staphylococcus aureus from bacteraemic patients. Microbiology 2010, 156, 860–872. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, T.; Hayashi, T.; Takami, H.; Ohnishi, M.; Murata, T.; Nakayama, K.; Asakawa, K.; Ohara, M.; Komatsuzawa, H.; Sugai, M. Complete nucleotide sequence of a Staphylococcus aureus exfoliative toxin B plasmid and identification of a novel ADP-ribosyltransferase, EDIN-C. Infect. Immun. 2001, 69, 7760–7771. [Google Scholar] [CrossRef] [PubMed]
- Czech, A.; Yamaguchi, T.; Bader, L.; Linder, S.; Kaminski, K.; Sugai, M.; Aepfelbacher, M. Prevalence of Rho-inactivating epidermal cell differentiation inhibitor toxins in clinical Staphylococcus aureus isolates. J. Infect. Dis. 2001, 184, 785–788. [Google Scholar] [CrossRef] [PubMed]
- Munro, P.; Clément, R.; Lavigne, J.P.; Pulcini, C.; Lemichez, E.; Landraud, L. High prevalence of edin-C encoding RhoA-targeting toxin in clinical isolates of Staphylococcus aureus. Eur. J. Clin. Microbiol. Infect. Dis. 2011, 30, 965–972. [Google Scholar] [CrossRef] [PubMed]


| Grades | Symptoms |
|---|---|
| Grade 1 | No symptoms, no signs of infection |
| Grade 2 | Lesion only involving the skin (no subcutaneous tissue lesion or systemic disorders) with at least two of the following signs:
|
| Grade 3 |
|
| Grade 4 | Regardless of the local infection, in the presence of systemic signs corresponding to at least two of the following characteristics:
|
| Genes | PEDIS Grades | Total | References | ||
|---|---|---|---|---|---|
| Grade 1 | Grades 2–3 | Grade 4 | |||
| n = 99 | n = 481 | n = 94 | n = 674 | ||
| lukF/luS-PV | 3 (3%) | 15 (3.1%) | 3 (3.2%) | 21 (3.1%) | [48,110,122,145] |
| edin | 6 (6.1%) | 12 (2.5%) | 10 (10.6%) | 28 (4.1%) | [48,110,122,145] |
| tsst | 5 (5.1%) | 40 (8.3%) | 12 (12.8%) | 57 (8.5%) | [48,110,122,145] |
| etA, etB, etD | 4 (4.0%) | 17 (3.5%) | 13 (13.8%) | 34 (5.0%) | [48,110,122,145] |
| sea | 39 (39.4%) | 201 (41.8%) | 39 (41.5%) | 279 (41.4%) | [48,110,122,145] |
| lukDE | 66 (66.7%) | 263 (54.7%) | 49 (52.1%) | 378 (56.1%) | [48,110,122,145] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dunyach-Remy, C.; Ngba Essebe, C.; Sotto, A.; Lavigne, J.-P. Staphylococcus aureus Toxins and Diabetic Foot Ulcers: Role in Pathogenesis and Interest in Diagnosis. Toxins 2016, 8, 209. https://doi.org/10.3390/toxins8070209
Dunyach-Remy C, Ngba Essebe C, Sotto A, Lavigne J-P. Staphylococcus aureus Toxins and Diabetic Foot Ulcers: Role in Pathogenesis and Interest in Diagnosis. Toxins. 2016; 8(7):209. https://doi.org/10.3390/toxins8070209
Chicago/Turabian StyleDunyach-Remy, Catherine, Christelle Ngba Essebe, Albert Sotto, and Jean-Philippe Lavigne. 2016. "Staphylococcus aureus Toxins and Diabetic Foot Ulcers: Role in Pathogenesis and Interest in Diagnosis" Toxins 8, no. 7: 209. https://doi.org/10.3390/toxins8070209
APA StyleDunyach-Remy, C., Ngba Essebe, C., Sotto, A., & Lavigne, J.-P. (2016). Staphylococcus aureus Toxins and Diabetic Foot Ulcers: Role in Pathogenesis and Interest in Diagnosis. Toxins, 8(7), 209. https://doi.org/10.3390/toxins8070209







