Combined Effects of Bee Venom Acupuncture and Morphine on Oxaliplatin-Induced Neuropathic Pain in Mice
Abstract
:1. Introduction
2. Results
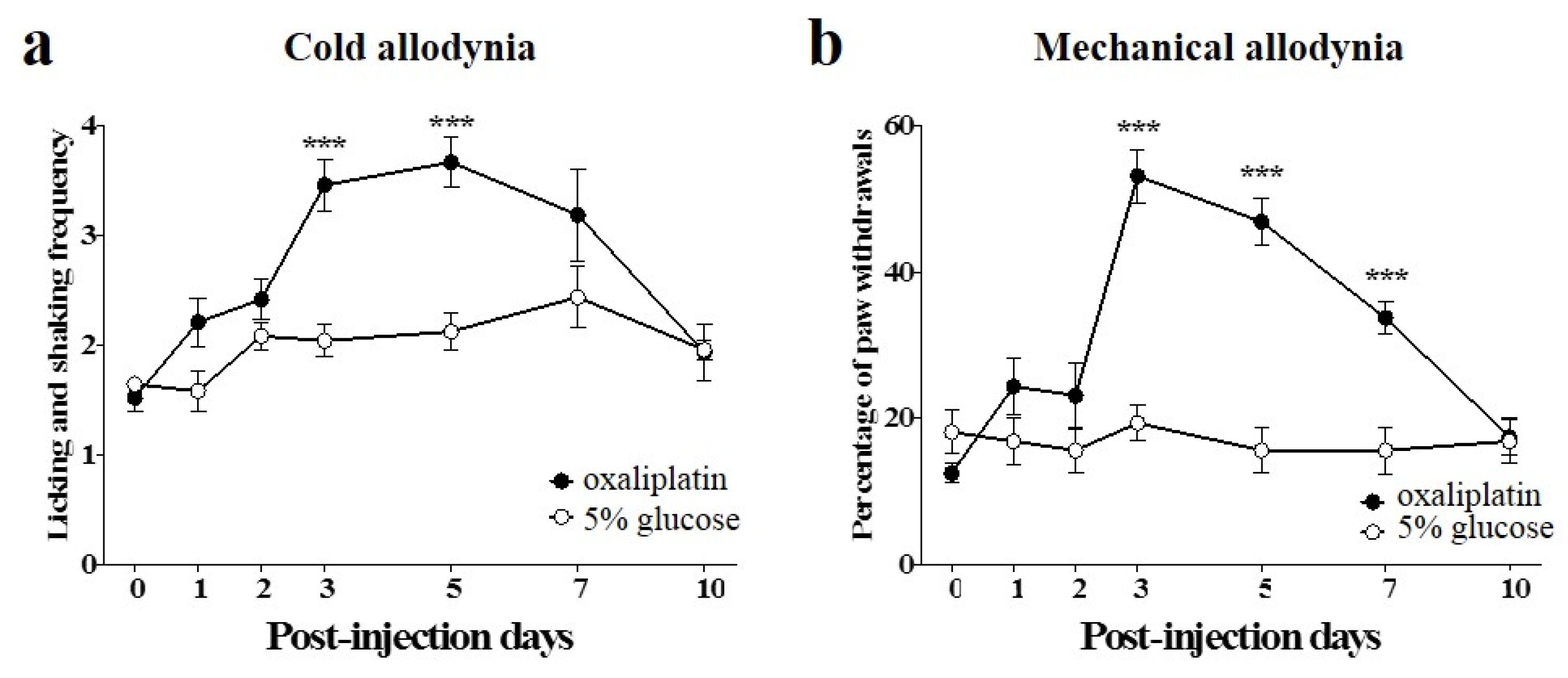
2.1. Induction of Cold and Mechanical Allodynia by a Single Intraperitoneal Administration of Oxaliplatin in Mice
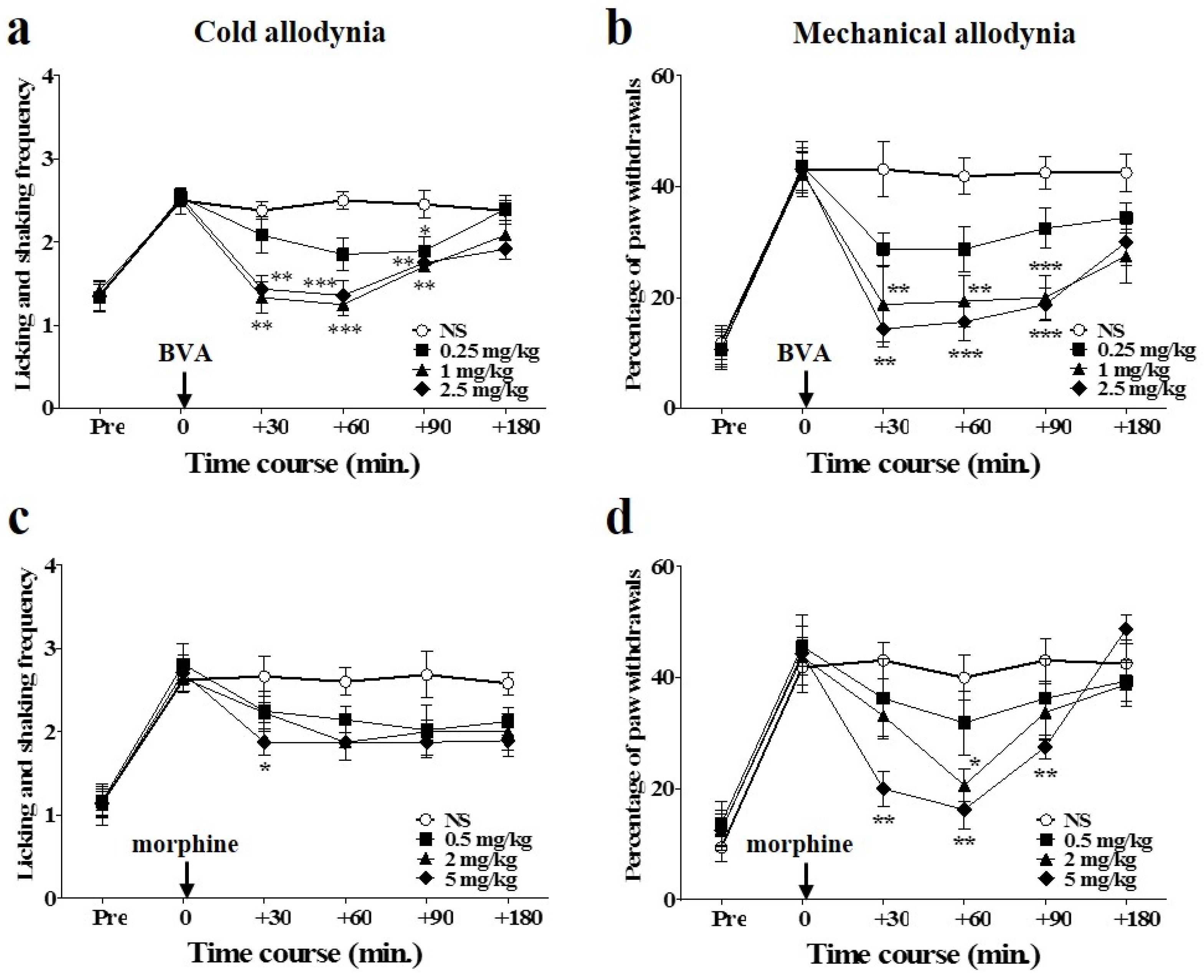
2.2. Time Course of Dose-Dependent Effects of BVA or Morphine on Oxaliplatin-Induced Cold and Mechanical Allodynia in Mice
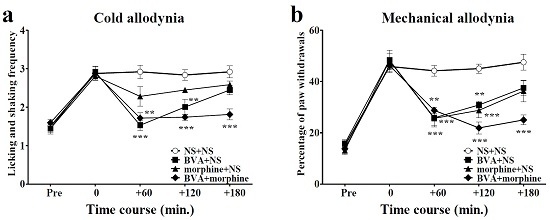
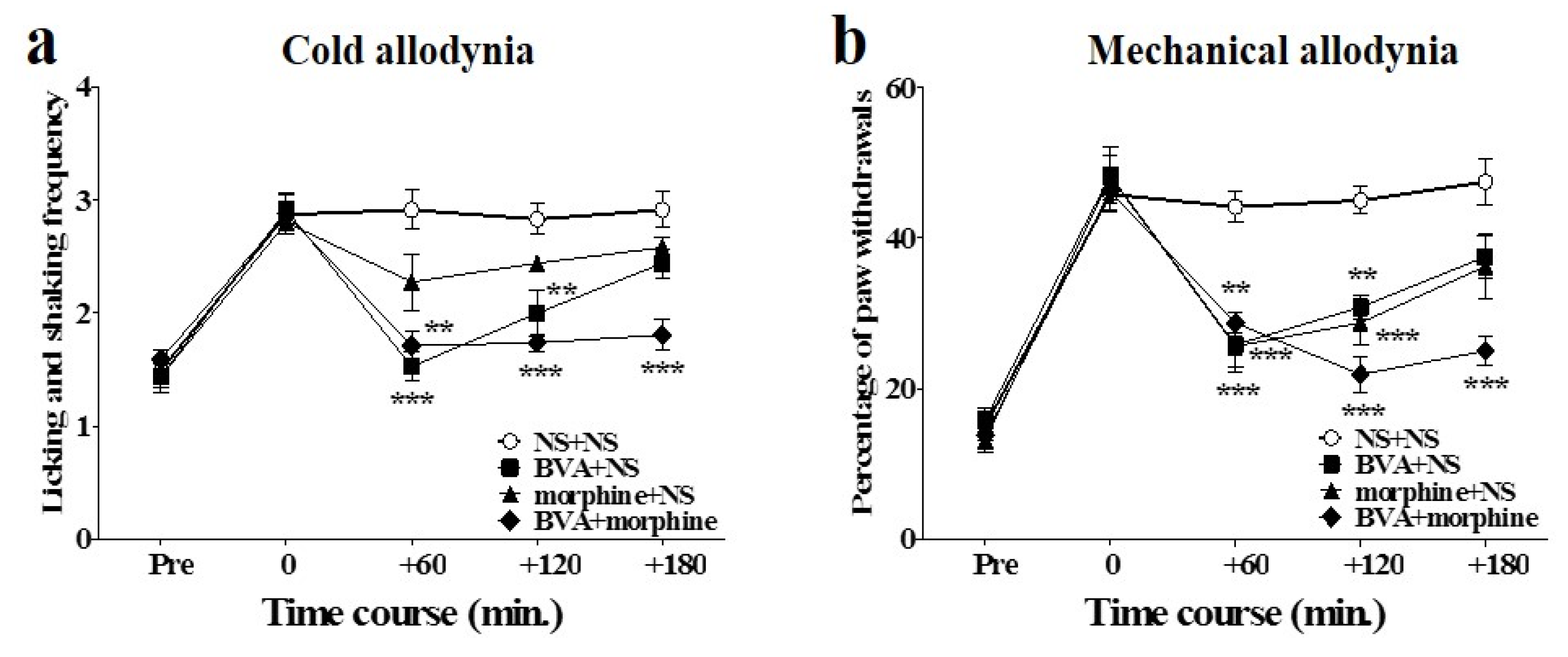
2.3. Time Course of Combined Effects of BVA and Morphine on Oxaliplatin-Induced Cold and Mechanical Allodynia in Mice
2.4. Involvement of Spinal Opioidergic and 5-HT3 Receptors but not of α2-Adrenergic Receptors in the Combination Effect of BVA and Morphine on Oxaliplatin-Induced Allodynia in Mice

3. Discussion
4. Experimental Section
4.1. Animals
4.2. Oxaliplatin Administration
4.3. Behavioral Test
4.4. BVA and Morphine Treatment
4.5. Antagonist Treatment
4.6. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Screnci, D.; McKeage, M.; Galettis, P.; Hambley, T.; Palmer, B.; Baguley, B. Relationships between hydrophobicity, reactivity, accumulation and peripheral nerve toxicity of a series of platinum drugs. Br. J. Cancer 2000, 82, 966–972. [Google Scholar] [CrossRef] [PubMed]
- Ferlay, J.; Soerjomataram, I.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in globocan 2012. Int. J. Cancer 2015, 136, E359–E386. [Google Scholar] [CrossRef] [PubMed]
- Arany, I.; Safirstein, R.L. Cisplatin nephrotoxicity. Semin. Nephrol. 2003, 23, 460–464. [Google Scholar] [CrossRef]
- Ling, B.; Coudoré-Civiale, M.-A.; Balayssac, D.; Eschalier, A.; Coudoré, F.; Authier, N. Behavioral and immunohistological assessment of painful neuropathy induced by a single oxaliplatin injection in the rat. Toxicology 2007, 234, 176–184. [Google Scholar] [CrossRef] [PubMed]
- Gamelin, E.; Gamelin, L.; Bossi, L.; Quasthoff, S. Clinical aspects and molecular basis of oxaliplatin neurotoxicity: Current management and development of preventive measures. Semin. Oncol. 2002, 29, 21–33. [Google Scholar] [CrossRef] [PubMed]
- Pasetto, L.M.; D’Andrea, M.R.; Rossi, E.; Monfardini, S. Oxaliplatin-related neurotoxicity: How and why? Crit. Rev. Oncol./Hematol. 2006, 59, 159–168. [Google Scholar] [CrossRef] [PubMed]
- Lehky, T.J.; Leonard, G.D.; Wilson, R.H.; Grem, J.L.; Floeter, M.K. Oxaliplatin-induced neurotoxicity: Acute hyperexcitability and chronic neuropathy. Muscle Nerve 2004, 29, 387–392. [Google Scholar] [CrossRef] [PubMed]
- Wolf, S.; Barton, D.; Kottschade, L.; Grothey, A.; Loprinzi, C. Chemotherapy-induced peripheral neuropathy: Prevention and treatment strategies. Eur. J. Cancer 2008, 44, 1507–1515. [Google Scholar] [CrossRef] [PubMed]
- Argyriou, A.A.; Polychronopoulos, P.; Iconomou, G.; Chroni, E.; Kalofonos, H.P. A review on oxaliplatin-induced peripheral nerve damage. Cancer Treat. Rev. 2008, 34, 368–377. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.; Lee, B.; Seo, J. A case of the reduction of cervical disc protrusion on the ct scanning after 5 months by oriental medical treatments added by bee venom acupuncture. J. Kor. AM Meridian Pointol. Soc. 2001, 18, 141–148. [Google Scholar]
- Kim, Y.-H.; Yook, T.-H.; Song, B.-Y.; Lee, D.-H. Clinical report of oriental medicine treatment with bee venom therapy of progressive muscle atrophy 1 patient. J. Pharmacopunct. 2000, 3, 119–140. [Google Scholar]
- Cho, S.-Y.; Shim, S.-R.; Rhee, H.Y.; Park, H.-J.; Jung, W.-S.; Moon, S.-K.; Park, J.-M.; Ko, C.-N.; Cho, K.-H.; Park, S.-U. Effectiveness of acupuncture and bee venom acupuncture in idiopathic parkinson’s disease. Parkinsonism Relat. Disord. 2012, 18, 948–952. [Google Scholar] [CrossRef] [PubMed]
- Son, D.J.; Lee, J.W.; Lee, Y.H.; Song, H.S.; Lee, C.K.; Hong, J.T. Therapeutic application of anti-arthritis, pain-releasing, and anti-cancer effects of bee venom and its constituent compounds. Pharmacol. Ther. 2007, 115, 246–270. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-D.; Park, H.-J.; Chae, Y.; Lim, S. An overview of bee venom acupuncture in the treatment of arthritis. Evid. Based Complement. Altern. Med. 2005, 2, 79–84. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.S.; Pittler, M.H.; Shin, B.-C.; Kong, J.C.; Ernst, E. Bee venom acupuncture for musculoskeletal pain: A review. J. Pain 2008, 9, 289–297. [Google Scholar] [CrossRef] [PubMed]
- Yoon, J.; Jeon, J.-H.; Lee, Y.-W.; Cho, C.-K.; Kwon, K.-R.; Shin, J.-E.; Sagar, S.; Wong, R.; Yoo, H.-S. Sweet bee venom pharmacopuncture for chemotherapy-induced peripheral neuropathy. J. Acupunct. Meridian Stud. 2012, 5, 156–165. [Google Scholar] [CrossRef] [PubMed]
- Lim, B.-S.; Moon, H.J.; Li, D.X.; Gil, M.; Min, J.K.; Lee, G.; Bae, H.; Kim, S.K.; Min, B.-I. Effect of bee venom acupuncture on oxaliplatin-induced cold allodynia in rats. Evid. Based Complement. Altern. Med. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Li, D.X.; Yoon, H.; Go, D.; Quan, F.S.; Min, B.I.; Kim, S.K. Serotonergic mechanism of the relieving effect of bee venom acupuncture on oxaliplatin-induced neuropathic cold allodynia in rats. BMC Complement. Altern. Med. 2014, 14, 471. [Google Scholar] [CrossRef] [PubMed]
- Han, J.S. Acupuncture: Neuropeptide release produced by electrical stimulation of different frequencies. Trends Neurosci. 2003, 26, 17–22. [Google Scholar] [CrossRef]
- Kim, W.; Kim, S.K.; Min, B.-I. Mechanisms of electroacupuncture-induced analgesia on neuropathic pain in animal model. Evid. Based Complement. Altern. Med. 2013. [Google Scholar] [CrossRef] [PubMed]
- Baek, Y.H.; Huh, J.E.; Lee, J.D.; Park, D.S. Antinociceptive effect and the mechanism of bee venom acupuncture (apipuncture) on inflammatory pain in the rat model of collagen-induced arthritis: Mediation by α2-adrenoceptors. Brain Res. 2006, 1073–1074, 305–310. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.W.; Kwon, Y.B.; Han, H.J.; Yang, I.S.; Beitz, A.J.; Lee, J.H. Antinociceptive mechanisms associated with diluted bee venom acupuncture (apipuncture) in the rat formalin test: Involvement of descending adrenergic and serotonergic pathways. Pharmacol. Res. 2005, 51, 183–188. [Google Scholar] [CrossRef] [PubMed]
- Yekkirala, A.S.; Kalyuzhny, A.E.; Portoghese, P.S. Standard opioid agonists activate heteromeric opioid receptors: Evidence for morphine and [d-ala2-mephe4-glyol5] enkephalin as selective μ–δ agonists. ACS Chem. Neurosci. 2009, 1, 146–154. [Google Scholar] [CrossRef] [PubMed]
- Arner, S.; Arner, B. Differential effects of epidural morphine in the treatment of cancer-related pain. Acta Anaesthesiol. Scand. 1985, 29, 32–36. [Google Scholar] [CrossRef] [PubMed]
- Cherny, N.; Thaler, H.; Friedlander-Klar, H.; Lapin, J.; Foley, K.; Houde, R.; Portenoy, R. Opioid responsiveness of cancer pain syndromes caused by neuropathic or nociceptive mechanisms a combined analysis of controlled, single-dose studies. Neurology 1994, 44, 857. [Google Scholar] [CrossRef] [PubMed]
- Portenoy, R.K.; Foley, K.M.; Inturrisi, C.E. The nature of opioid responsiveness and its implications for neuropathic pain: New hypotheses derived from studies of opioid infusions. Pain 1990, 43, 273–286. [Google Scholar] [CrossRef]
- Hershman, D.L.; Lacchetti, C.; Dworkin, R.H.; Smith, E.M.L.; Bleeker, J.; Cavaletti, G.; Chauhan, C.; Gavin, P.; Lavino, A.; Lustberg, M.B. Prevention and management of chemotherapy-induced peripheral neuropathy in survivors of adult cancers: American society of clinical oncology clinical practice guideline. J. Clin. Oncol. 2014. [Google Scholar] [CrossRef] [PubMed]
- Gilron, I.; Bailey, J.M.; Tu, D.; Holden, R.R.; Weaver, D.F.; Houlden, R.L. Morphine, gabapentin, or their combination for neuropathic pain. N. Engl. J. Med. 2005, 352, 1324–1334. [Google Scholar] [CrossRef] [PubMed]
- Moon, H.J.; Lim, B.-S.; Lee, D.-I.; Ye, M.S.; Lee, G.; Min, B.-I.; Bae, H.; Na, H.S.; Kim, S.K. Effects of electroacupuncture on oxaliplatin-induced neuropathic cold hypersensitivity in rats. J. Physiol. Sci. 2014, 64, 151–156. [Google Scholar] [CrossRef] [PubMed]
- Ling, B.; Coudoré, F.; Decalonne, L.; Eschalier, A.; Authier, N. Comparative antiallodynic activity of morphine, pregabalin and lidocaine in a rat model of neuropathic pain produced by one oxaliplatin injection. Neuropharmacology 2008, 55, 724–728. [Google Scholar] [CrossRef] [PubMed]
- Ormseth, M.J.; Scholz, B.A.; Boomershine, C.S. Duloxetine in the management of diabetic peripheral neuropathic pain. Patient Preference Adherence 2011, 5, 343–356. [Google Scholar] [PubMed]
- Serpell, M.; Group, N.P.S. Gabapentin in neuropathic pain syndromes: A randomised, double-blind, placebo-controlled trial. Pain 2002, 99, 557–566. [Google Scholar] [CrossRef]
- Kang, S.-Y.; Roh, D.-H.; Choi, J.-W.; Ryu, Y.; Lee, J.-H. Repetitive treatment with diluted bee venom attenuates the induction of below-level neuropathic pain behaviors in a rat spinal cord injury model. Toxins 2015, 7, 2571–2585. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.-W.; Chaplan, S.R.; Yaksh, T.L. Systemic and supraspinal, but not spinal, opiates suppress allodynia in a rat neuropathic pain model. Neurosci. Lett. 1995, 199, 111–114. [Google Scholar] [CrossRef]
- Lemberg, K.; Kontinen, V.K.; Viljakka, K.; Kylänlahti, I.; Yli-Kauhaluoma, J.; Kalso, E. Morphine, oxycodone, methadone and its enantiomers in different models of nociception in the rat. Anesthesia Analg. 2006, 102, 1768–1774. [Google Scholar] [CrossRef] [PubMed]
- Roh, D.-H.; Kwon, Y.-B.; Kim, H.-W.; Ham, T.-W.; Yoon, S.-Y.; Kang, S.-Y.; Han, H.-J.; Lee, H.-J.; Beitz, A.J.; Lee, J.-H. Acupoint stimulation with diluted bee venom (apipuncture) alleviates thermal hyperalgesia in a rodent neuropathic pain model: Involvement of spinal alpha 2-adrenoceptors. J. Pain 2004, 5, 297–303. [Google Scholar] [CrossRef] [PubMed]
- Kwon, Y.-B.; Kang, M.-S.; Han, H.-J.; Beitz, A.J.; Lee, J.-H. Visceral antinociception produced by bee venom stimulation of the zhongwan acupuncture point in mice: Role of α 2 adrenoceptors. Neurosci. Lett. 2001, 308, 133–137. [Google Scholar] [CrossRef]
- Kwon, Y.B.; Ham, T.W.; Kim, H.W.; Roh, D.H.; Yoon, S.Y.; Han, H.J.; Yang, I.S.; Kim, K.W.; Beitz, A.J.; Lee, J.H. Water soluble fraction (<10 kda) from bee venom reduces visceral pain behavior through spinal α 2-adrenergic activity in mice. Pharmacol. Biochem. Behav. 2005, 80, 181–187. [Google Scholar] [PubMed]
- Kwon, Y.B.; Yoon, S.Y.; Kim, H.W.; Roh, D.H.; Kang, S.Y.; Ryu, Y.H.; Choi, S.M.; Han, H.J.; Lee, H.J.; Kim, K.W. Substantial role of locus coeruleus-noradrenergic activation and capsaicin-insensitive primary afferent fibers in bee venom’s anti-inflammatory effect. Neurosci. Res. 2006, 55, 197–203. [Google Scholar] [CrossRef] [PubMed]
- Tsuchiya, M.; Yamazaki, H.; Hori, Y. Enkephalinergic neurons express 5-HT3 receptors in the spinal cord dorsal horn: Single cell RT-PCR analysis. Neuroreport 1999, 10, 2749–2753. [Google Scholar] [CrossRef] [PubMed]
- Fukushima, T.; Tsuda, M.; Kofuji, T.; Hori, Y. Physiological properties of enkephalin-containing neurons in the spinal dorsal horn visualized by expression of green fluorescent protein in bac transgenic mice. BMC Neurosci. 2011, 12, 36. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.W.; Zhang, Z.H.; Wang, R.; Xie, Y.F.; Qiao, J.T.; Dafny, N. Norepinephrine and serotonin-induced antinociception are blocked by naloxone with different dosages. Brain Res. Bull. 1994, 35, 113–117. [Google Scholar] [PubMed]
- Giordano, J. Analgesic profile of centrally administered 2-methylserotonin against acute pain in rats. Eur. J. Pharmacol. 1991, 199, 233–236. [Google Scholar] [CrossRef]
- Yaksh, T.; Tyce, G. Microinjection of morphine into the periaqueductal gray evokes the release of serotonin from spinal cord. Brain res. 1979, 171, 176–181. [Google Scholar] [CrossRef]
- Li, D.; Lee, Y.; Kim, W.; Lee, K.; Bae, H.; Kim, S.K. Analgesic effects of bee venom derived phospholipase a2 in a mouse model of oxaliplatin-induced neuropathic pain. Toxins 2015, 7, 2422–2434. [Google Scholar] [CrossRef] [PubMed]
- Flatters, S.J.; Bennett, G.J. Ethosuximide reverses paclitaxel-and vincristine-induced painful peripheral neuropathy. Pain 2004, 109, 150–161. [Google Scholar] [CrossRef] [PubMed]
- Joseph, E.K.; Levine, J.D. Comparison of oxaliplatin-and cisplatin-induced painful peripheral neuropathy in the rat. J. Pain 2009, 10, 534–541. [Google Scholar] [CrossRef] [PubMed]
- Shibata, K.; Sugawara, T.; Fujishita, K.; Shinozaki, Y.; Matsukawa, T.; Suzuki, T.; Koizumi, S. The astrocyte-targeted therapy by bushi for the neuropathic pain in mice. PLoS ONE 2011, 6, e23510. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.-W.; Kwon, Y.-B.; Ham, T.-W.; Roh, D.-H.; Yoon, S.-Y.; Lee, H.-J.; Han, H.-J.; Yang, I.-S.; Beitz, A.J.; Lee, J.-H. Acupoint stimulation using bee venom attenuates formalin-induced pain behavior and spinal cord fos expression in rats. J. Vet. Med. Sci. 2003, 65, 349–355. [Google Scholar] [CrossRef] [PubMed]
- Yoon, S.-Y.; Roh, D.-H.; Kwon, Y.-B.; Kim, H.-W.; Seo, H.-S.; Han, H.-J.; Lee, H.-J.; Beitz, A.J.; Lee, J.-H. Acupoint stimulation with diluted bee venom (apipuncture) potentiates the analgesic effect of intrathecal clonidine in the rodent formalin test and in a neuropathic pain model. J. Pain 2009, 10, 253–263. [Google Scholar] [CrossRef] [PubMed]
- Omiya, Y.; Suzuki, Y.; Yuzurihara, M.; Murata, M.; Aburada, M.; Kase, Y.; Takeda, S. Antinociceptive effect of shakuyakukanzoto, a kampo medicine, in diabetic mice. J. Pharmacol. Sci. 2005, 99, 373–380. [Google Scholar] [CrossRef] [PubMed]
- Roh, D.-H.; Kim, H.-W.; Yoon, S.-Y.; Kang, S.-Y.; Kwon, Y.-B.; Cho, K.-H.; Han, H.-J.; Ryu, Y.-H.; Choi, S.-M.; Lee, H.-J. Bee venom injection significantly reduces nociceptive behavior in the mouse formalin test via capsaicin-insensitive afferents. J. Pain 2006, 7, 500–512. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, W.; Kim, M.J.; Go, D.; Min, B.-I.; Na, H.S.; Kim, S.K. Combined Effects of Bee Venom Acupuncture and Morphine on Oxaliplatin-Induced Neuropathic Pain in Mice. Toxins 2016, 8, 33. https://doi.org/10.3390/toxins8020033
Kim W, Kim MJ, Go D, Min B-I, Na HS, Kim SK. Combined Effects of Bee Venom Acupuncture and Morphine on Oxaliplatin-Induced Neuropathic Pain in Mice. Toxins. 2016; 8(2):33. https://doi.org/10.3390/toxins8020033
Chicago/Turabian StyleKim, Woojin, Min Joon Kim, Donghyun Go, Byung-Il Min, Heung Sik Na, and Sun Kwang Kim. 2016. "Combined Effects of Bee Venom Acupuncture and Morphine on Oxaliplatin-Induced Neuropathic Pain in Mice" Toxins 8, no. 2: 33. https://doi.org/10.3390/toxins8020033
APA StyleKim, W., Kim, M. J., Go, D., Min, B.-I., Na, H. S., & Kim, S. K. (2016). Combined Effects of Bee Venom Acupuncture and Morphine on Oxaliplatin-Induced Neuropathic Pain in Mice. Toxins, 8(2), 33. https://doi.org/10.3390/toxins8020033