Yeast Reporter Assay to Identify Cellular Components of Ricin Toxin A Chain Trafficking
Abstract
:1. Introduction
2. Results and Discussion
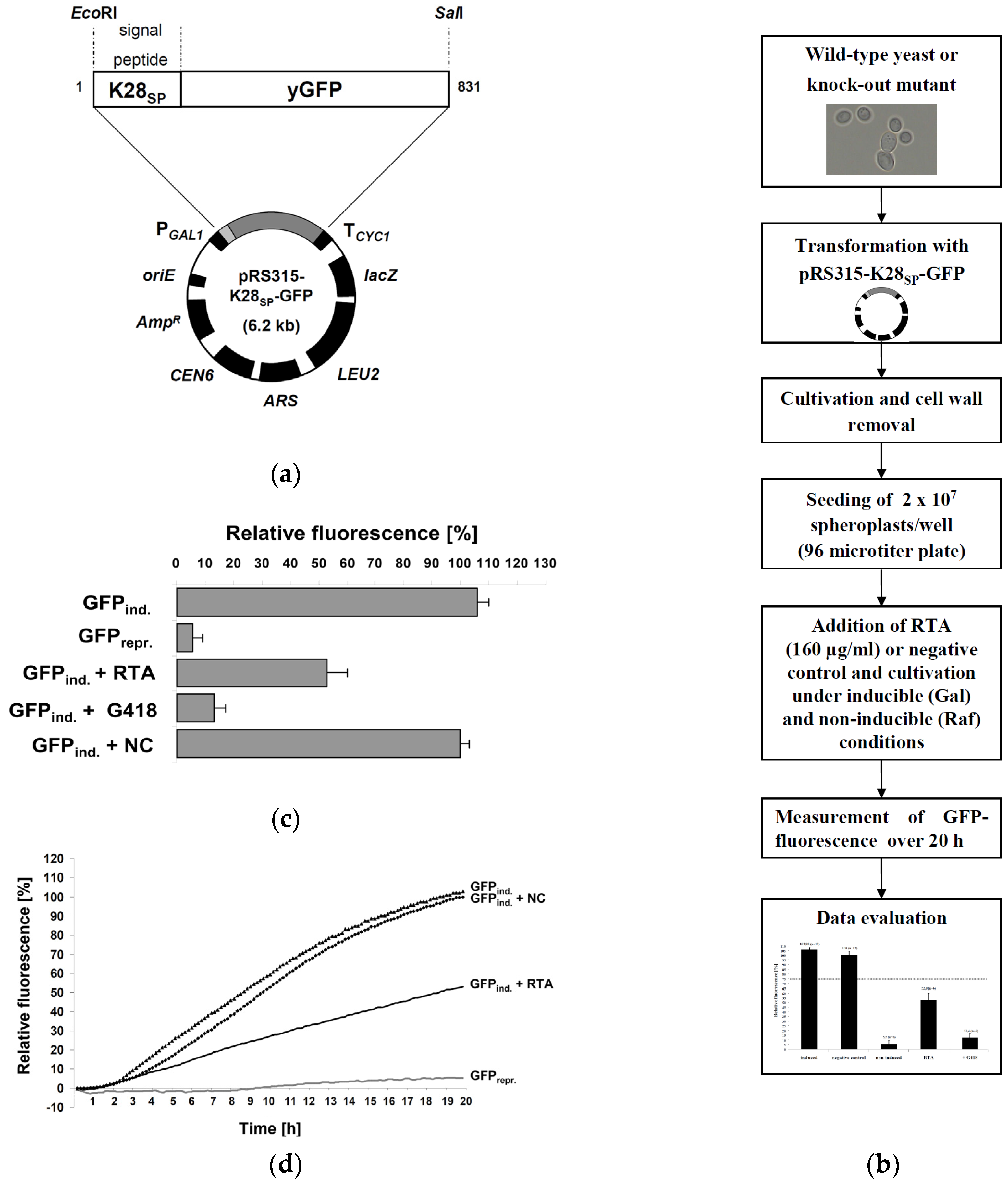
2.1. Fluorescence-Based Reporter Assay for RTA Toxicity in Yeast
2.2. Assay Validation
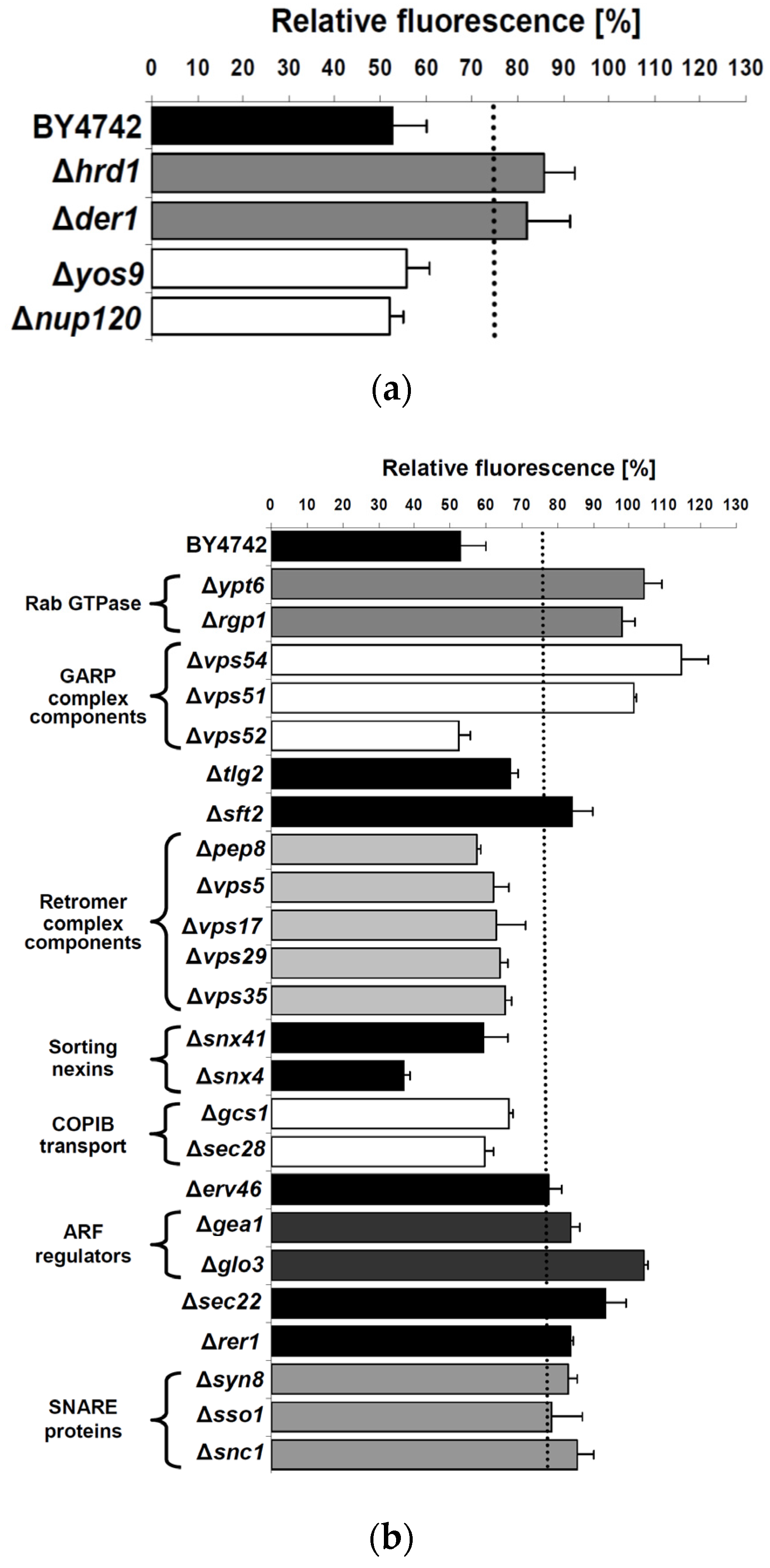
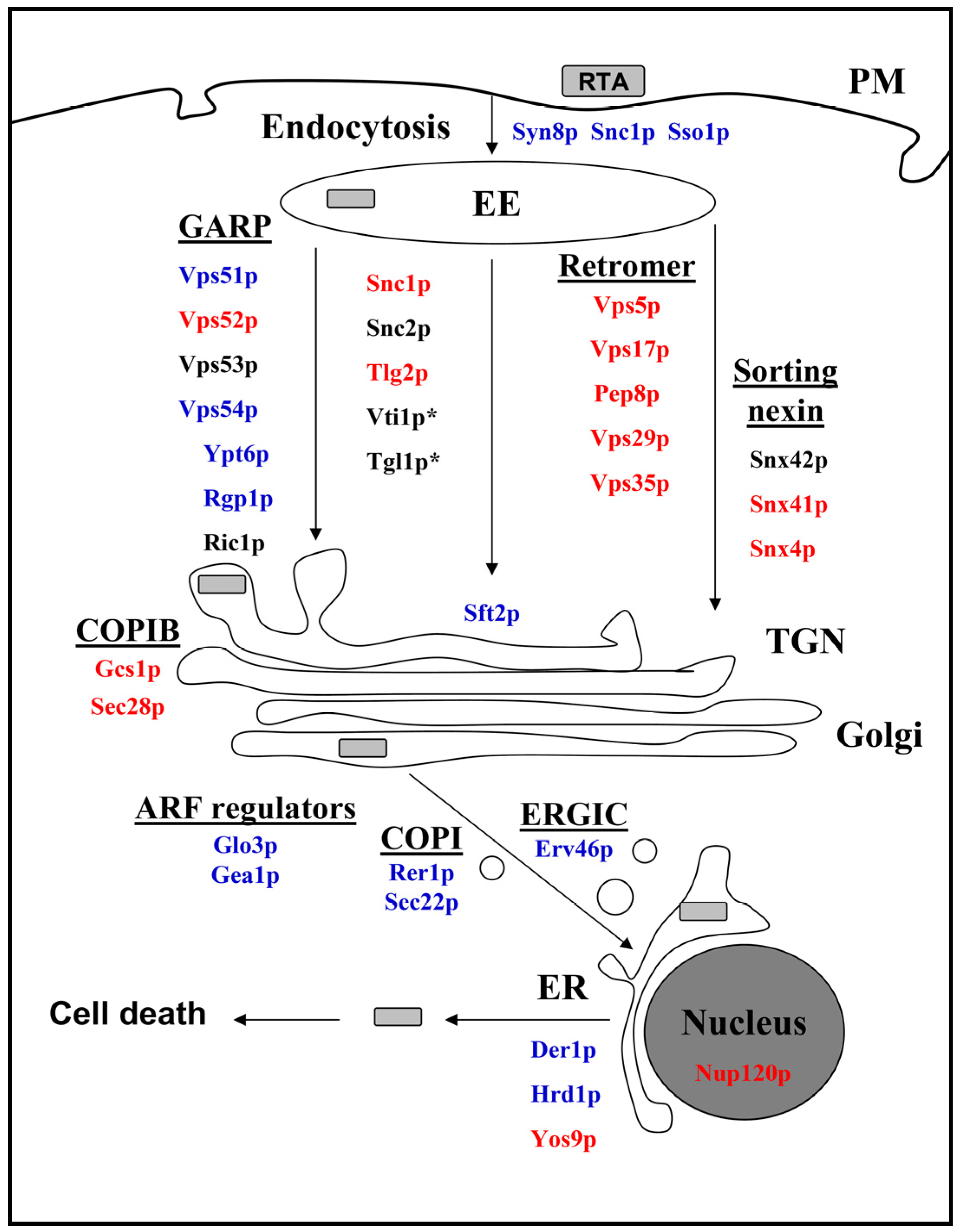
2.3. Cellular Pathways Involved in Endosome-to-Golgi Transport of RTA
2.4. Impact of Golgi-to-ER Transport and/or Endocytosis Components on RTA Toxicity
3. Materials and Methods
3.1. Escherichia coli Strains, Plasmids, Culture Media and Genetic Techniques
3.2. Affinity Purification of RTA
3.3. Yeast Strains, Transformation and Culture Media
3.4. GFP-Fluorescence Assay
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wei, G.Q.; Liu, R.S.; Wang, Q.; Liu, W.Y. Toxicity of two type II ribosome-inactivating proteins (cinnamomin and ricin) to domestic silkworm larvae. Arch. Insect Biochem. Physiol. 2004, 57, 160–165. [Google Scholar] [CrossRef] [PubMed]
- Hartley, M.R.; Lord, J.M. Cytotoxic ribosome-inactivating lectins from plants. Biochim. Biophys. Acta 2004, 1701, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Holmberg, L.; Nygard, O. Depurination of A4256 in 28 S rRNA by the ribosome-inactivating proteins from barley and ricin results in different ribosome conformations. J. Mol. Biol. 1996, 259, 81–94. [Google Scholar] [CrossRef] [PubMed]
- Kvalvaag, A.S.; Pust, S.; Sandvig, K. Vps11, a subunit of the tethering complexes HOPS and CORVET, is involved in regulation of glycolipid degradation and retrograde toxin transport. Commun. Integr. Biol. 2014, 7. [Google Scholar] [CrossRef] [PubMed]
- Moreau, D.; Kumar, P.; Wang, S.C.; Chaumet, A.; Chew, S.Y.; Chevalley, H.; Bard, F. Genome-wide RNAi screens identify genes required for Ricin and PE intoxications. Dev. Cell 2011, 21, 231–244. [Google Scholar] [CrossRef] [PubMed]
- Stechmann, B.; Bai, S.K.; Gobbo, E.; Lopez, R.; Merer, G.; Pinchard, S.; Panigai, L.; Tenza, D.; Raposo, G.; Beaumelle, B.; et al. Inhibition of retrograde transport protects mice from lethal ricin challenge. Cell 2010, 141, 231–242. [Google Scholar] [CrossRef] [PubMed]
- Utskarpen, A.; Slagsvold, H.H.; Iversen, T.G.; Walchli, S.; Sandvig, K. Transport of ricin from endosomes to the Golgi apparatus is regulated by rab6a and rab6a′. Traffic 2006, 7, 663–672. [Google Scholar] [CrossRef] [PubMed]
- Lord, M.J.; Jolliffe, N.A.; Marsden, C.J.; Pateman, C.S.; Smith, D.C.; Spooner, R.A.; Watson, P.D.; Roberts, L.M. Ricin. Mechanisms of cytotoxicity. Toxicol. Rev. 2003, 22, 53–64. [Google Scholar] [CrossRef] [PubMed]
- Spooner, R.A.; Lord, J.M. Ricin trafficking in cells. Toxins 2015, 7, 49–65. [Google Scholar] [CrossRef] [PubMed]
- Moya, M.; Dautry-Varsat, A.; Goud, B.; Louvard, D.; Boquet, P. Inhibition of coated pit formation in Hep2 cells blocks the cytotoxicity of diphtheria toxin but not that of ricin toxin. J. Cell Biol. 1985, 101, 548–559. [Google Scholar] [CrossRef] [PubMed]
- Iversen, T.G.; Skretting, G.; Llorente, A.; Nicoziani, P.; van Deurs, B.; Sandvig, K. Endosome to Golgi transport of ricin is independent of clathrin and of the Rab9- and Rab11-GTPases. Mol. Biol. Cell 2001, 12, 2099–2107. [Google Scholar] [CrossRef] [PubMed]
- Sandvig, K.; Olsnes, S. Entry of the toxic proteins abrin, modeccin, ricin, and diphtheria toxin into cells. II. Effect of pH, metabolic inhibitors, and ionophores and evidence for toxin penetration from endocytotic vesicles. J. Biol. Chem. 1982, 257, 7504–7513. [Google Scholar] [PubMed]
- Moisenovich, M.; Tonevitsky, A.; Maljuchenko, N.; Kozlovskaya, N.; Agapov, I.; Volknandt, W.; Bereiter-Hahn, J. Endosomal ricin transport: Involvement of Rab4- and Rab5-positive compartments. Histochem. Cell Biol. 2004, 121, 429–439. [Google Scholar] [CrossRef] [PubMed]
- Sandvig, K.; Bergan, J.; Kavaliauskiene, S.; Skotland, T. Lipid requirements for entry of protein toxins into cells. Prog. Lipid Res. 2014, 54, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Simpson, J.C.; Roberts, L.M.; Lord, J.M. Free ricin a chain reaches an early compartment of the secretory pathway before it enters the cytosol. Exp. Cell Res. 1996, 229, 447–451. [Google Scholar] [CrossRef] [PubMed]
- Wales, R.; Roberts, L.M.; Lord, J.M. Addition of an endoplasmic reticulum retrieval sequence to ricin A chain significantly increases its cytotoxicity to mammalian cells. J. Biol. Chem. 1993, 268, 23986–23990. [Google Scholar] [PubMed]
- Becker, B.; Schmitt, M.J. Adapting yeast as model to study ricin toxin a uptake and trafficking. Toxins 2011, 3, 834–847. [Google Scholar] [CrossRef] [PubMed]
- Van Deurs, B.; Sandvig, K.; Petersen, O.W.; Olsnes, S.; Simons, K.; Griffiths, G. Estimation of the amount of internalized ricin that reaches the trans-Golgi network. J. Cell Biol. 1988, 106, 253–267. [Google Scholar] [CrossRef] [PubMed]
- Lord, J.M.; Roberts, L.M.; Robertus, J.D. Ricin: Structure, mode of action, and some current applications. FASEB J. 1994, 8, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Day, P.J.; Owens, S.R.; Wesche, J.; Olsnes, S.; Roberts, L.M.; Lord, J.M. An interaction between ricin and calreticulin that may have implications for toxin trafficking. J. Biol. Chem. 2001, 276, 7202–7208. [Google Scholar] [CrossRef] [PubMed]
- Slominska-Wojewodzka, M.; Gregers, T.F.; Walchli, S.; Sandvig, K. EDEM is involved in retrotranslocation of ricin from the endoplasmic reticulum to the cytosol. Mol. Biol. Cell 2006, 17, 1664–1675. [Google Scholar] [CrossRef] [PubMed]
- Slominska-Wojewodzka, M.; Pawlik, A.; Sokolowska, I.; Antoniewicz, J.; Wegrzyn, G.; Sandvig, K. The role of EDEM2 compared with EDEM1 in ricin transport from the endoplasmic reticulum to the cytosol. Biochem. J. 2014, 457, 485–496. [Google Scholar] [CrossRef] [PubMed]
- Simpson, J.C.; Roberts, L.M.; Romisch, K.; Davey, J.; Wolf, D.H.; Lord, J.M. Ricin a chain utilises the endoplasmic reticulum-associated protein degradation pathway to enter the cytosol of yeast. FEBS Lett. 1999, 459, 80–84. [Google Scholar] [CrossRef]
- Herrera, C.; Klokk, T.I.; Cole, R.; Sandvig, K.; Mantis, N.J. A bispecific antibody promotes aggregation of ricin toxin on cell surfaces and alters dynamics of toxin internalization and trafficking. PLoS ONE 2016, 11. [Google Scholar] [CrossRef] [PubMed]
- Yermakova, A.; Klokk, T.I.; O’Hara, J.M.; Cole, R.; Sandvig, K.; Mantis, N.J. Neutralizing monoclonal antibodies against disparate epitopes on ricin toxin’s enzymatic subunit interfere with intracellular toxin transport. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef] [PubMed]
- Allen, S.C.; Moore, K.A.; Marsden, C.J.; Fulop, V.; Moffat, K.G.; Lord, J.M.; Ladds, G.; Roberts, L.M. The isolation and characterization of temperature-dependent ricin A chain molecules in Saccharomyces cerevisiae. FEBS J. 2007, 274, 5586–5599. [Google Scholar] [CrossRef] [PubMed]
- Yan, Q.; Li, X.P.; Tumer, N.E. Wild type RTA and less toxic variants have distinct requirements for Png1 for their depurination activity and toxicity in Saccharomyces cerevisiae. PLoS ONE 2015, 9, e113719. [Google Scholar] [CrossRef] [PubMed]
- Bonifacino, J.S.; Rojas, R. Retrograde transport from endosomes to the trans-Golgi network. Nat. Rev. Mol. Cell Biol. 2006, 7, 568–579. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.C.; Miller, E.A.; Goldberg, J.; Orci, L.; Schekman, R. Bi-directional protein transport between the ER and Golgi. Annu. Rev. Cell Dev. Biol. 2004, 20, 87–123. [Google Scholar] [CrossRef] [PubMed]
- Duden, R. ER-to-Golgi transport: Cop I and Cop II function (review). Mol. Membr. Biol. 2003, 20, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Seaman, M.N. Endosome protein sorting: Motifs and machinery. Cell. Mol. Life Sci. 2008, 65, 2842–2858. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, M.J. Cloning and expression of a cDNA copy of the viral K28 killer toxin gene in yeast. Mol. Gen. Genet. 1995, 246, 236–246. [Google Scholar] [CrossRef] [PubMed]
- Endo, Y.; Tsurugi, K. RNA N-glycosidase activity of ricin A-chain. Mechanism of action of the toxic lectin ricin on eukaryotic ribosomes. J. Biol. Chem. 1987, 262, 8128–8130. [Google Scholar] [PubMed]
- Eiden-Plach, A.; Zagorc, T.; Heintel, T.; Carius, Y.; Breinig, F.; Schmitt, M.J. Viral preprotoxin signal sequence allows efficient secretion of green fluorescent protein by Candida glabrata, Pichia pastoris, Saccharomyces cerevisiae, and Schizosaccharomyces pombe. Appl. Environ. Microbiol. 2004, 70, 961–966. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Spooner, R.A.; Allen, S.C.; Guise, C.P.; Ladds, G.; Schnoder, T.; Schmitt, M.J.; Lord, J.M.; Roberts, L.M. Folding-competent and folding-defective forms of ricin A chain have different fates after retrotranslocation from the endoplasmic reticulum. Mol. Biol. Cell 2010, 21, 2543–2554. [Google Scholar] [CrossRef] [PubMed]
- Luo, Z.; Gallwitz, D. Biochemical and genetic evidence for the involvement of yeast Ypt6-GTPase in protein retrieval to different Golgi compartments. J. Biol. Chem. 2003, 278, 791–799. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Warner, J.R. Mutation of the rab6 homologue of Saccharomyces cerevisiae, Ypt6, inhibits both early Golgi function and ribosome biosynthesis. J. Biol. Chem. 1996, 271, 16813–16819. [Google Scholar] [PubMed]
- Siniossoglou, S.; Peak-Chew, S.Y.; Pelham, H.R. Ric1p and Rgp1p form a complex that catalyses nucleotide exchange on Ypt6p. EMBO J. 2000, 19, 4885–4894. [Google Scholar] [CrossRef] [PubMed]
- Perez-Victoria, F.J.; Bonifacino, J.S. Dual roles of the mammalian GARP complex in tethering and SNARE complex assembly at the trans-Golgi network. Mol. Cell. Biol. 2009, 29, 5251–5263. [Google Scholar] [CrossRef] [PubMed]
- Conibear, E.; Stevens, T.H. Vps52p, Vps53p, and Vps54p form a novel multisubunit complex required for protein sorting at the yeast late Golgi. Mol. Biol. Cell 2000, 11, 305–323. [Google Scholar] [CrossRef] [PubMed]
- Amessou, M.; Fradagrada, A.; Falguieres, T.; Lord, J.M.; Smith, D.C.; Roberts, L.M.; Lamaze, C.; Johannes, L. Syntaxin 16 and syntaxin 5 are required for efficient retrograde transport of several exogenous and endogenous cargo proteins. J. Cell Sci. 2007, 120, 1457–1468. [Google Scholar] [CrossRef] [PubMed]
- Conchon, S.; Cao, X.; Barlowe, C.; Pelham, H.R. Got1p and Sft2p: Membrane proteins involved in traffic to the Golgi complex. EMBO J. 1999, 18, 3934–3946. [Google Scholar] [CrossRef] [PubMed]
- Johannes, L.; Popoff, V. Tracing the retrograde route in protein trafficking. Cell 2008, 135, 1175–1187. [Google Scholar] [CrossRef] [PubMed]
- Seaman, M.N.; McCaffery, J.M.; Emr, S.D. A membrane coat complex essential for endosome-to-Golgi retrograde transport in yeast. J. Cell Biol. 1998, 142, 665–681. [Google Scholar] [CrossRef] [PubMed]
- Hettema, E.H.; Lewis, M.J.; Black, M.W.; Pelham, H.R. Retromer and the sorting nexins Snx4/41/42 mediate distinct retrieval pathways from yeast endosomes. EMBO J. 2003, 22, 548–557. [Google Scholar] [CrossRef] [PubMed]
- Day, K.J.; Staehelin, L.A.; Glick, B.S. A three-stage model of Golgi structure and function. Histochem. Cell Biol. 2013, 140, 239–249. [Google Scholar] [CrossRef] [PubMed]
- Robinson, M.; Poon, P.P.; Schindler, C.; Murray, L.E.; Kama, R.; Gabriely, G.; Singer, R.A.; Spang, A.; Johnston, G.C.; Gerst, J.E. The Gcs1 Arf-GAP mediates Snc1,2 v-SNARE retrieval to the Golgi in yeast. Mol. Biol. Cell 2006, 17, 1845–1858. [Google Scholar] [CrossRef] [PubMed]
- Eugster, A.; Frigerio, G.; Dale, M.; Duden, R. COP I domains required for coatomer integrity, and novel interactions with ARF and ARF-GAP. EMBO J. 2000, 19, 3905–3917. [Google Scholar] [CrossRef] [PubMed]
- Otte, S.; Belden, W.J.; Heidtman, M.; Liu, J.; Jensen, O.N.; Barlowe, C. Erv41p and Erv46p: New components of COPII vesicles involved in transport between the ER and Golgi complex. J. Cell Biol. 2001, 152, 503–518. [Google Scholar] [CrossRef] [PubMed]
- Orci, L.; Ravazzola, M.; Mack, G.J.; Barlowe, C.; Otte, S. Mammalian Erv46 localizes to the endoplasmic reticulum-Golgi intermediate compartment and to cis-Golgi cisternae. Proc. Natl. Acad. Sci. USA 2003, 100, 4586–4591. [Google Scholar] [CrossRef] [PubMed]
- Shibuya, A.; Margulis, N.; Christiano, R.; Walther, T.C.; Barlowe, C. The Erv41-Erv46 complex serves as a retrograde receptor to retrieve escaped ER proteins. J. Cell Biol. 2015, 208, 197–209. [Google Scholar] [CrossRef] [PubMed]
- Lewis, S.M.; Poon, P.P.; Singer, R.A.; Johnston, G.C.; Spang, A. The ArfGAP Glo3 is required for the generation of COPI vesicles. Mol. Biol. Cell 2004, 15, 4064–4072. [Google Scholar] [CrossRef] [PubMed]
- Spang, A.; Herrmann, J.M.; Hamamoto, S.; Schekman, R. The ADP ribosylation factor-nucleotide exchange factors Gea1p and Gea2p have overlapping, but not redundant functions in retrograde transport from the Golgi to the endoplasmic reticulum. Mol. Biol. Cell 2001, 12, 1035–1045. [Google Scholar] [CrossRef] [PubMed]
- Peyroche, A.; Paris, S.; Jackson, C.L. Nucleotide exchange on ARF mediated by yeast Gea1 protein. Nature 1996, 384, 479–481. [Google Scholar] [CrossRef] [PubMed]
- Sato, K.; Sato, M.; Nakano, A. Rer1p, a retrieval receptor for endoplasmic reticulum membrane proteins, is dynamically localized to the Golgi apparatus by coatomer. J. Cell Biol. 2001, 152, 935–944. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Flanagan, J.J.; Barlowe, C. Sec22p export from the endoplasmic reticulum is independent of snare pairing. J. Biol. Chem. 2004, 279, 27225–27232. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Barlowe, C. Analysis of Sec22p in endoplasmic reticulum/Golgi transport reveals cellular redundancy in SNARE protein function. Mol. Biol. Cell 2002, 13, 3314–3324. [Google Scholar] [CrossRef] [PubMed]
- Sandvig, K.; Torgersen, M.L.; Engedal, N.; Skotland, T.; Iversen, T.G. Protein toxins from plants and bacteria: Probes for intracellular transport and tools in medicine. FEBS Lett. 2010, 584, 2626–2634. [Google Scholar] [CrossRef] [PubMed]
- Lewis, M.J.; Nichols, B.J.; Prescianotto-Baschong, C.; Riezman, H.; Pelham, H.R. Specific retrieval of the exocytic snare Snc1p from early yeast endosomes. Mol. Biol. Cell 2000, 11, 23–38. [Google Scholar] [CrossRef] [PubMed]
- Lewis, M.J.; Pelham, H.R. A new yeast endosomal SNARE related to mammalian syntaxin 8. Traffic 2002, 3, 922–929. [Google Scholar] [CrossRef] [PubMed]
- Aalto, M.K.; Ronne, H.; Keranen, S. Yeast syntaxins Sso1p and Sso2p belong to a family of related membrane proteins that function in vesicular transport. EMBO J. 1993, 12, 4095–4104. [Google Scholar] [PubMed]
- Gurunathan, S.; Chapman-Shimshoni, D.; Trajkovic, S.; Gerst, J.E. Yeast exocytic v-SNAREs confer endocytosis. Mol. Biol. Cell 2000, 11, 3629–3643. [Google Scholar] [CrossRef] [PubMed]
- Protopopov, V.; Govindan, B.; Novick, P.; Gerst, J.E. Homologs of the synaptobrevin/VAMP family of synaptic vesicle proteins function on the late secretory pathway in S. cerevisiae. Cell 1993, 74, 855–861. [Google Scholar] [CrossRef]
- David, D.; Sundarababu, S.; Gerst, J.E. Involvement of long chain fatty acid elongation in the trafficking of secretory vesicles in yeast. J. Cell Biol. 1998, 143, 1167–1182. [Google Scholar] [CrossRef] [PubMed]
- Rossi, G.; Salminen, A.; Rice, L.M.; Brunger, A.T.; Brennwald, P. Analysis of a yeast snare complex reveals remarkable similarity to the neuronal SNARE complex and a novel function for the C terminus of the SNAP-25 homolog, Sec9. J. Biol. Chem. 1997, 272, 16610–16617. [Google Scholar] [CrossRef] [PubMed]
- Bennett, M.K.; Garcia-Arraras, J.E.; Elferink, L.A.; Peterson, K.; Fleming, A.M.; Hazuka, C.D.; Scheller, R.H. The syntaxin family of vesicular transport receptors. Cell 1993, 74, 863–873. [Google Scholar] [CrossRef]
- Gerst, J.E.; Rodgers, L.; Riggs, M.; Wigler, M. SNC1, a yeast homolog of the synaptic vesicle-associated membrane protein/synaptobrevin gene family: Genetic interactions with the RAS and CAP genes. Proc. Natl. Acad. Sci. USA 1992, 89, 4338–4342. [Google Scholar] [CrossRef] [PubMed]
- Sambrook, J.; Maniatis, T.; Fritsch, E.F. Molecular Cloning: A Laboratory Manual, 2nd ed.; Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY, USA, 1989. [Google Scholar]
- Cormack, B.P.; Bertram, G.; Egerton, M.; Gow, N.A.; Falkow, S.; Brown, A.J. Yeast-enhanced green fluorescent protein (yEGFP)a reporter of gene expression in Candida albicans. Microbiology 1997, 143 Pt 2, 303–311. [Google Scholar] [CrossRef] [PubMed]
- Horton, R.M.; Hunt, H.D.; Ho, S.N.; Pullen, J.K.; Pease, L.R. Engineering hybrid genes without the use of restriction enzymes: Gene splicing by overlap extension. Gene 1989, 77, 61–68. [Google Scholar] [CrossRef]
- Sikorski, R.S.; Hieter, P. A system of shuttle vectors and yeast host strains designed for efficient manipulation of DNA in Saccharomyces cerevisiae. Genetics 1989, 122, 19–27. [Google Scholar] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Becker, B.; Schnöder, T.; Schmitt, M.J. Yeast Reporter Assay to Identify Cellular Components of Ricin Toxin A Chain Trafficking. Toxins 2016, 8, 366. https://doi.org/10.3390/toxins8120366
Becker B, Schnöder T, Schmitt MJ. Yeast Reporter Assay to Identify Cellular Components of Ricin Toxin A Chain Trafficking. Toxins. 2016; 8(12):366. https://doi.org/10.3390/toxins8120366
Chicago/Turabian StyleBecker, Björn, Tina Schnöder, and Manfred J. Schmitt. 2016. "Yeast Reporter Assay to Identify Cellular Components of Ricin Toxin A Chain Trafficking" Toxins 8, no. 12: 366. https://doi.org/10.3390/toxins8120366
APA StyleBecker, B., Schnöder, T., & Schmitt, M. J. (2016). Yeast Reporter Assay to Identify Cellular Components of Ricin Toxin A Chain Trafficking. Toxins, 8(12), 366. https://doi.org/10.3390/toxins8120366





