Preventive Effects of Bee Venom Derived Phospholipase A2 on Oxaliplatin-Induced Neuropathic Pain in Mice
Abstract
:1. Introduction
2. Results
2.1. Preventive Effects of bvPLA2 on Oxaliplatin-Induced Cold and Mechanical Allodynia

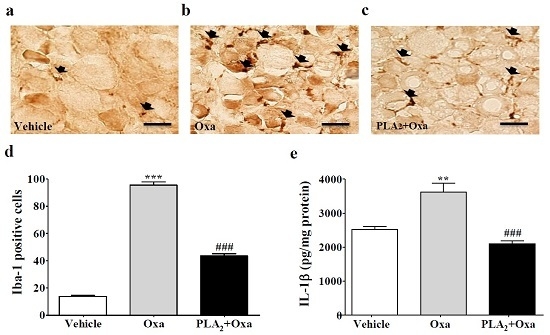
2.2. Inhibition of Macrophages and Pro-Inflammatory Cytokines in the Lumbar DRG by bvPLA2 Pre-Treatment
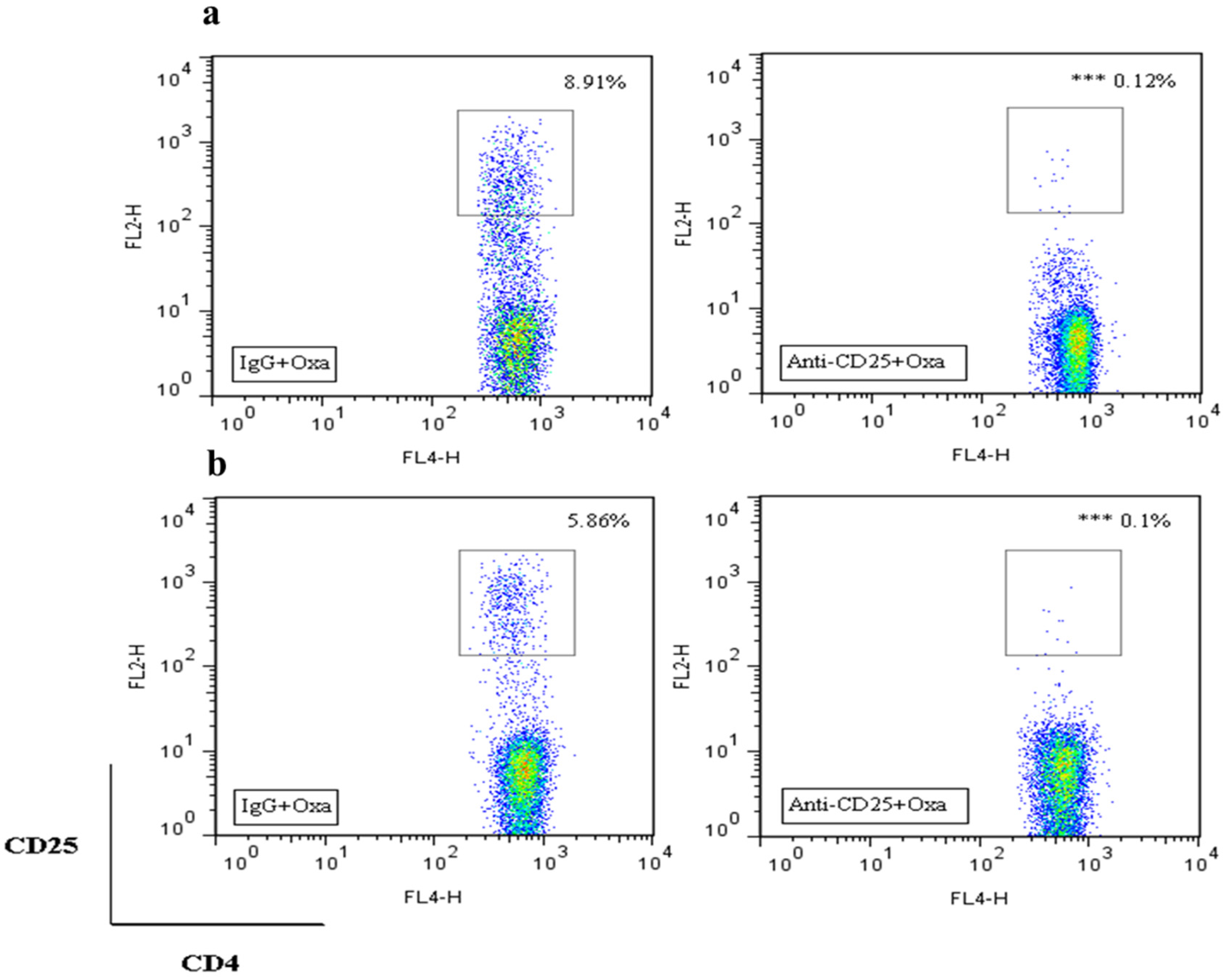
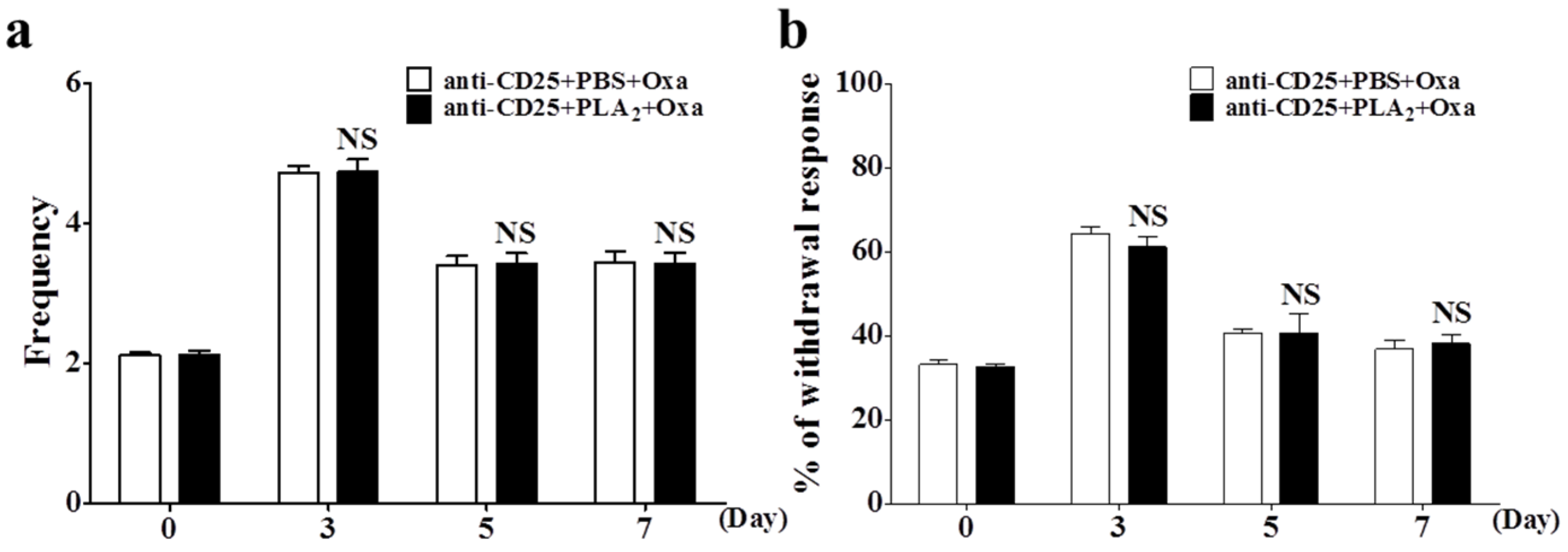
2.3. Effects of bvPLA2 Pre-Treatment on Oxaliplatin-Induced Neuropathic Pain in Treg Depleted Mice
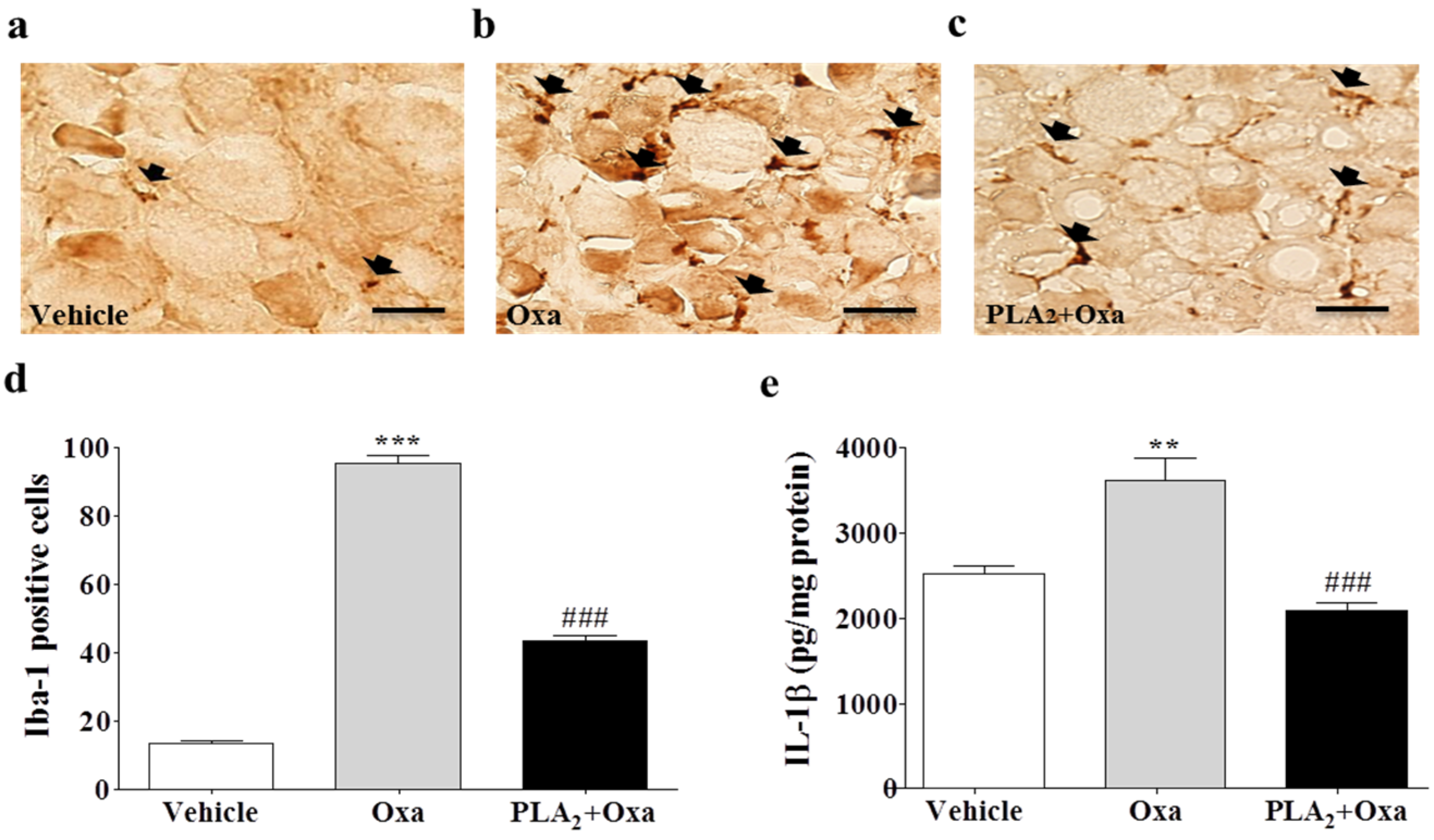
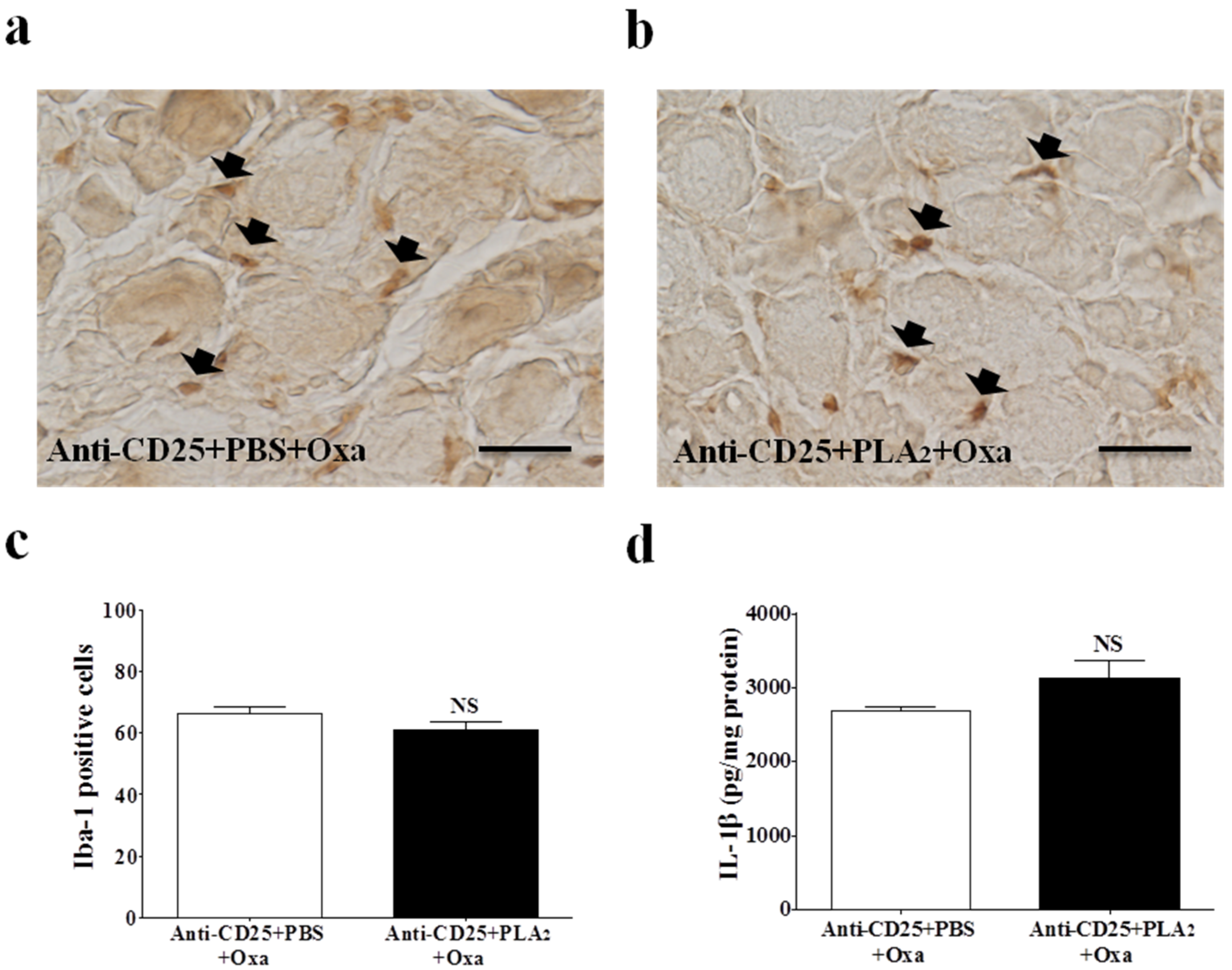
2.4. Effects of bvPLA2 Pre-Treatment on Macrophages and Pro-Inflammatory Cytokines in the DRG of Treg Depleted Mice
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Behavioral Tests
4.3. Oxaliplatin Administration and bvPLA2 Treatment
4.4. Depletion of Tregs
4.5. Immunohistochemistry
4.6. Assessment of Cytokines in the DRG by Enzyme-Linked Immunosorbent Assay (ELISA)
4.7. Statistical Analysis
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Baker, D.E. Oxaliplatin: A new drug for the treatment of metastatic carcinoma of the colon or rectum. Rev. Gastroenterol. Disord. 2003, 3, 31–38. [Google Scholar] [PubMed]
- Screnci, D.; McKeage, M.J.; Galettis, P.; Hambley, T.W.; Palmer, B.D.; Baguley, B.C. Relationships between hydrophobicity, reactivity, accumulation and peripheral nerve toxicity of a series of platinum drugs. Br. J. Cancer 2000, 82, 966–972. [Google Scholar] [CrossRef] [PubMed]
- Muggia, F.M. Recent updates in the clinical use of platinum compounds for the treatment of gynecologic cancers. Semin. Oncol. 2004, 31, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Petit, T.; Benider, A.; Yovine, A.; Bougnoux, P.; Spaeth, D.; Maindrault-Goebel, F.; Serin, D.; Tigaud, J.D.; Eymard, J.C.; Simon, H.; et al. Phase II study of an oxaliplatin/vinorelbine combination in patients with anthracycline- and taxane-pre-treated metastatic breast cancer. Anti-cancer Drugs 2006, 17, 337–343. [Google Scholar] [CrossRef] [PubMed]
- Desoize, B.; Madoulet, C. Particular aspects of platinum compounds used at present in cancer treatment. Crit. Rev. Oncol. Hematol. 2002, 42, 317–325. [Google Scholar] [CrossRef]
- Extra, J.-M.; Marty, M.; Brienza, S.; Misset, J.-L. Pharmacokinetics and safety profile of oxaliplatin. Semin. Oncol. 1998, 25, 13–22. [Google Scholar] [PubMed]
- Cassidy, J.; Misset, J.-L. Oxaliplatin-related side effects: Characteristics and management. Semin. Oncol. 2002, 29, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Quasthoff, S.; Hartung, H.P. Chemotherapy-induced peripheral neuropathy. J. Neurol. 2002, 249, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, C.; Cota, G. Calcium block of Na+ channels and its effect on closing rate. Proc. Natl. Acad. Sci. USA 1999, 96, 4154–4157. [Google Scholar] [CrossRef] [PubMed]
- Kottschade, L.A.; Sloan, J.A.; Mazurczak, M.A.; Johnson, D.B.; Murphy, B.P.; Rowland, K.M.; Smith, D.A.; Berg, A.R.; Stella, P.J.; Loprinzi, C.L. The use of vitamin e for the prevention of chemotherapy-induced peripheral neuropathy: Results of a randomized phase III clinical trial. Support. Care Cancer 2011, 19, 1769–1777. [Google Scholar] [CrossRef] [PubMed]
- Amara, S. Oral glutamine for the prevention of chemotherapy-induced peripheral neuropathy. Ann. Pharmacother. 2008, 42, 1481–1485. [Google Scholar] [CrossRef] [PubMed]
- Wolf, S.; Barton, D.; Kottschade, L.; Grothey, A.; Loprinzi, C. Chemotherapy-induced peripheral neuropathy: Prevention and treatment strategies. Eur. J. Cancer 2008, 44, 1507–1515. [Google Scholar] [CrossRef] [PubMed]
- Pachman, D.; Barton, D.; Watson, J.; Loprinzi, C. Chemotherapy-induced peripheral neuropathy: Prevention and treatment. Clin. Pharmacol. Ther. 2011, 90, 377–387. [Google Scholar] [CrossRef] [PubMed]
- Tofaris, G.K.; Patterson, P.H.; Jessen, K.R.; Mirsky, R. Denervated schwann cells attract macrophages by secretion of leukemia inhibitory factor (lif) and monocyte chemoattractant protein-1 in a process regulated by interleukin-6 and lif. J. Neurosci. 2002, 22, 6696–6703. [Google Scholar] [PubMed]
- Hu, P.; McLachlan, E. Macrophage and lymphocyte invasion of dorsal root ganglia after peripheral nerve lesions in the rat. Neuroscience 2002, 112, 23–38. [Google Scholar] [CrossRef]
- Abbadie, C.; Lindia, J.A.; Cumiskey, A.M.; Peterson, L.B.; Mudgett, J.S.; Bayne, E.K.; DeMartino, J.A.; MacIntyre, D.E.; Forrest, M.J. Impaired neuropathic pain responses in mice lacking the chemokine receptor CCR2. Proc. Natl. Acad. Sci. USA 2003, 100, 7947–7952. [Google Scholar] [CrossRef] [PubMed]
- Scholz, J.; Woolf, C.J. The neuropathic pain triad: Neurons, immune cells and glia. Nat. Neurosci. 2007, 10, 1361–1368. [Google Scholar] [CrossRef] [PubMed]
- Miller, R.J.; Jung, H.; Bhangoo, S.K.; White, F.A. Cytokine and chemokine regulation of sensory neuron function. Sens. Nerv. 2009, 194, 417–449. [Google Scholar]
- Myers, R.R.; Heckman, H.M.; Rodriguez, M. Reduced hyperalgesia in nerve-injured wld mice: Relationship to nerve fiber phagocytosis, axonal degeneration, and regeneration in normal mice. Exp. Neurol. 1996, 141, 94–101. [Google Scholar] [CrossRef] [PubMed]
- Peters, C.M.; Jimenez-Andrade, J.M.; Jonas, B.M.; Sevcik, M.A.; Koewler, N.J.; Ghilardi, J.R.; Wong, G.Y.; Mantyh, P.W. Intravenous paclitaxel administration in the rat induces a peripheral sensory neuropathy characterized by macrophage infiltration and injury to sensory neurons and their supporting cells. Exp. Neurol. 2007, 203, 42–54. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.Z.; Li, D.; Liu, C.C.; Cui, Y.; Zhu, H.Q.; Zhang, W.W.; Li, Y.Y.; Xin, W.J. CX3CL1-mediated macrophage activation contributed to paclitaxel-induced DRG neuronal apoptosis and painful peripheral neuropathy. Brain Behav. Immun. 2014, 40, 155–165. [Google Scholar] [CrossRef] [PubMed]
- Ledeboer, A.; Jekich, B.M.; Sloane, E.M.; Mahoney, J.H.; Langer, S.J.; Milligan, E.D.; Martin, D.; Maier, S.F.; Johnson, K.W.; Leinwand, L.A.; et al. Intrathecal interleukin-10 gene therapy attenuates paclitaxel-induced mechanical allodynia and proinflammatory cytokine expression in dorsal root ganglia in rats. Brain Behav. Immun. 2007, 21, 686–698. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.M.; Lehky, T.J.; Brell, J.M.; Dorsey, S.G. Discovering cytokines as targets for chemotherapy-induced painful peripheral neuropathy. Cytokine 2012, 59, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Bluestone, J.A.; Abbas, A.K. Natural versus adaptive regulatory T cells. Nat. Rev. Immunol. 2003, 3, 253–257. [Google Scholar] [CrossRef] [PubMed]
- Sakaguchi, S.; Ono, M.; Setoguchi, R.; Yagi, H.; Hori, S.; Fehervari, Z.; Shimizu, J.; Takahashi, T.; Nomura, T. Foxp3+CD25+CD4+ natural regulatory T cells in dominant self-tolerance and autoimmune disease. Immunol. Rev. 2006, 212, 8–27. [Google Scholar] [CrossRef] [PubMed]
- Austin, P.J.; Kim, C.F.; Perera, C.J.; Moalem-Taylor, G. Regulatory T cells attenuate neuropathic pain following peripheral nerve injury and experimental autoimmune neuritis. Pain 2012, 153, 1916–1931. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Lee, G.; Park, S.; Chung, H.S.; Lee, H.; Kim, J.Y.; Nam, S.; Kim, S.K.; Bae, H. Bee venom mitigates cisplatin-induced nephrotoxicity by regulating CD4+CD25+Foxp3+ regulatory T cells in mice. Evid. Based Complement. Altern. Med. 2013, 2013, 221–229. [Google Scholar] [CrossRef] [PubMed]
- Monti, M.C.; Casapullo, A.; Santomauro, C.; D’Auria, M.V.; Riccio, R.; Gomez-Paloma, L. The molecular mechanism of bee venom phospholipase A2 inactivation by bolinaquinone. Chembiochem 2006, 7, 971–980. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Kinnunen, P.K. Modulation of the activity of secretory phospholipase A2 by antimicrobial peptides. Antimicrob. Agents Chemother. 2003, 47, 965–971. [Google Scholar] [CrossRef] [PubMed]
- Murakami, M.; Sato, H.; Miki, Y.; Yamamoto, K.; Taketomi, Y. A new era of secreted phospholipase A2. J. Lipid Res. 2015, 56, 1248–1261. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Keum, D.J.; won Kwak, J.; Chung, H.-S.; Bae, H. Bee venom phospholipase A2 protects against acetaminophen-induced acute liver injury by modulating regulatory T cells and IL-10 in mice. PLoS ONE 2014, 9, e114726. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Lee, H.; Lee, G.; Jang, H.; Kim, S.S.; Yoon, H.; Kang, G.H.; Hwang, D.S.; Kim, S.K.; Chung, H.S.; et al. Phospholipase A2 inhibits cisplatin-induced acute kidney injury by modulating regulatory T cells by the CD206 mannose receptor. Kidney Int. 2015, 88, 550–559. [Google Scholar] [CrossRef] [PubMed]
- Andre, T.; Boni, C.; Mounedji-Boudiaf, L.; Navarro, M.; Tabernero, J.; Hickish, T.; Topham, C.; Zaninelli, M.; Clingan, P.; Bridgewater, J.; et al. Oxaliplatin, fluorouracil, and leucovorin as adjuvant treatment for colon cancer. N. Engl. J. Med. 2004, 350, 2343–2351. [Google Scholar] [CrossRef] [PubMed]
- Schmoll, H.J.; Cartwright, T.; Tabernero, J.; Nowacki, M.P.; Figer, A.; Maroun, J.; Price, T.; Lim, R.; van Cutsem, E.; Park, Y.S.; et al. Phase III trial of capecitabine plus oxaliplatin as adjuvant therapy for stage III colon cancer: A planned safety analysis in 1864 patients. J. Clin. Oncol. 2007, 25, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Raymond, E.; Faivre, S.; Woynarowski, J.M.; Chaney, S.G. Oxaliplatin: Mechanism of action and antineoplastic activity. Semin. Oncol. 1998, 25, 4–12. [Google Scholar] [PubMed]
- Alcindor, T.; Beauger, N. Oxaliplatin: A review in the era of molecularly targeted therapy. Curr. Oncol. 2011, 18, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, J.T.; Lipp, H.P. Toxicity of platinum compounds. Expert Opin. Pharmacother. 2003, 4, 889–901. [Google Scholar] [CrossRef] [PubMed]
- Austin, P.J.; Moalem-Taylor, G. The neuro-immune balance in neuropathic pain: Involvement of inflammatory immune cells, immune-like glial cells and cytokines. J. Neuroimmunol. 2010, 229, 26–50. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.F.; Moalem-Taylor, G. Detailed characterization of neuro-immune responses following neuropathic injury in mice. Brain Res. 2011, 1405, 95–108. [Google Scholar] [CrossRef] [PubMed]
- Morin, N.; Owolabi, S.A.; Harty, M.W.; Papa, E.F.; Tracy, T.F., Jr.; Shaw, S.K.; Kim, M.; Saab, C.Y. Neutrophils invade lumbar dorsal root ganglia after chronic constriction injury of the sciatic nerve. J. Neuroimmunol. 2007, 184, 164–171. [Google Scholar] [CrossRef] [PubMed]
- Barrette, B.; Hebert, M.A.; Filali, M.; Lafortune, K.; Vallieres, N.; Gowing, G.; Julien, J.P.; Lacroix, S. Requirement of myeloid cells for axon regeneration. J. Neurosci. 2008, 28, 9363–9376. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.M.; An, J. Cytokines, inflammation, and pain. Int. Anesthesiol. Clin. 2007, 45, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Watkins, L.R.; Wiertelak, E.P.; Goehler, L.E.; Smith, K.P.; Martin, D.; Maier, S.F. Characterization of cytokine-induced hyperalgesia. Brain Res. 1994, 654, 15–26. [Google Scholar] [CrossRef]
- Perkins, M.N.; Kelly, D. Interleukin-1β induced-desArg9bradykinin-mediated thermal hyperalgesia in the rat. Neuropharmacology 1994, 33, 657–660. [Google Scholar] [CrossRef]
- Ramer, M.S.; Murphy, P.G.; Richardson, P.M.; Bisby, M.A. Spinal nerve lesion-induced mechanoallodynia and adrenergic sprouting in sensory ganglia are attenuated in interleukin-6 knockout mice. Pain 1998, 78, 115–121. [Google Scholar] [CrossRef]
- Cunha, F.Q.; Poole, S.; Lorenzetti, B.B.; Ferreira, S.H. The pivotal role of tumour necrosis factor alpha in the development of inflammatory hyperalgesia. Br. J. Pharmacol. 1992, 107, 660–664. [Google Scholar] [CrossRef] [PubMed]
- Sorkin, L.S.; Xiao, W.H.; Wagner, R.; Myers, R.R. Tumour necrosis factor-α induces ectopic activity in nociceptive primary afferent fibres. Neuroscience 1997, 81, 255–262. [Google Scholar] [CrossRef]
- Zhang, J.M.; Li, H.; Liu, B.; Brull, S.J. Acute topical application of tumor necrosis factor α evokes protein kinase A-dependent responses in rat sensory neurons. J. Neurophysiol. 2002, 88, 1387–1392. [Google Scholar] [PubMed]
- Xie, W.R.; Deng, H.; Li, H.; Bowen, T.L.; Strong, J.A.; Zhang, J.M. Robust increase of cutaneous sensitivity, cytokine production and sympathetic sprouting in rats with localized inflammatory irritation of the spinal ganglia. Neuroscience 2006, 142, 809–822. [Google Scholar] [CrossRef] [PubMed]
- Copray, J.C.; Mantingh, I.; Brouwer, N.; Biber, K.; Kust, B.M.; Liem, R.S.; Huitinga, I.; Tilders, F.J.; van Dam, A.M.; Boddeke, H.W. Expression of interleukin-1β in rat dorsal root ganglia. J. Neuroimmunol. 2001, 118, 203–211. [Google Scholar] [CrossRef]
- O’Connor, R.A.; Anderton, S.M. Foxp3+ regulatory T cells in the control of experimental CNS autoimmune disease. J. Neuroimmunol. 2008, 193, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Flatters, S.J.; Bennett, G.J. Ethosuximide reverses paclitaxel- and vincristine-induced painful peripheral neuropathy. Pain 2004, 109, 150–161. [Google Scholar] [CrossRef] [PubMed]
- Joseph, E.K.; Levine, J.D. Comparison of oxaliplatin- and cisplatin-induced painful peripheral neuropathy in the rat. J. Pain 2009, 10, 534–541. [Google Scholar] [CrossRef] [PubMed]
- Shibata, K.; Sugawara, T.; Fujishita, K.; Shinozaki, Y.; Matsukawa, T.; Suzuki, T.; Koizumi, S. The astrocyte-targeted therapy by bushi for the neuropathic pain in mice. PLoS ONE 2011, 6, e23510. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, D.; Kim, W.; Shin, D.; Jung, Y.; Bae, H.; Kim, S.K. Preventive Effects of Bee Venom Derived Phospholipase A2 on Oxaliplatin-Induced Neuropathic Pain in Mice. Toxins 2016, 8, 27. https://doi.org/10.3390/toxins8010027
Li D, Kim W, Shin D, Jung Y, Bae H, Kim SK. Preventive Effects of Bee Venom Derived Phospholipase A2 on Oxaliplatin-Induced Neuropathic Pain in Mice. Toxins. 2016; 8(1):27. https://doi.org/10.3390/toxins8010027
Chicago/Turabian StyleLi, Dongxing, Woojin Kim, Dasom Shin, Yongjae Jung, Hyunsu Bae, and Sun Kwang Kim. 2016. "Preventive Effects of Bee Venom Derived Phospholipase A2 on Oxaliplatin-Induced Neuropathic Pain in Mice" Toxins 8, no. 1: 27. https://doi.org/10.3390/toxins8010027
APA StyleLi, D., Kim, W., Shin, D., Jung, Y., Bae, H., & Kim, S. K. (2016). Preventive Effects of Bee Venom Derived Phospholipase A2 on Oxaliplatin-Induced Neuropathic Pain in Mice. Toxins, 8(1), 27. https://doi.org/10.3390/toxins8010027