Exposure of Lycopersicon Esculentum to Microcystin-LR: Effects in the Leaf Proteome and Toxin Translocation from Water to Leaves and Fruits
Abstract
:1. Introduction
2. Results
2.1. Photosynthetic Efficiency

2.2. Protein Expression in Tomato Leaves
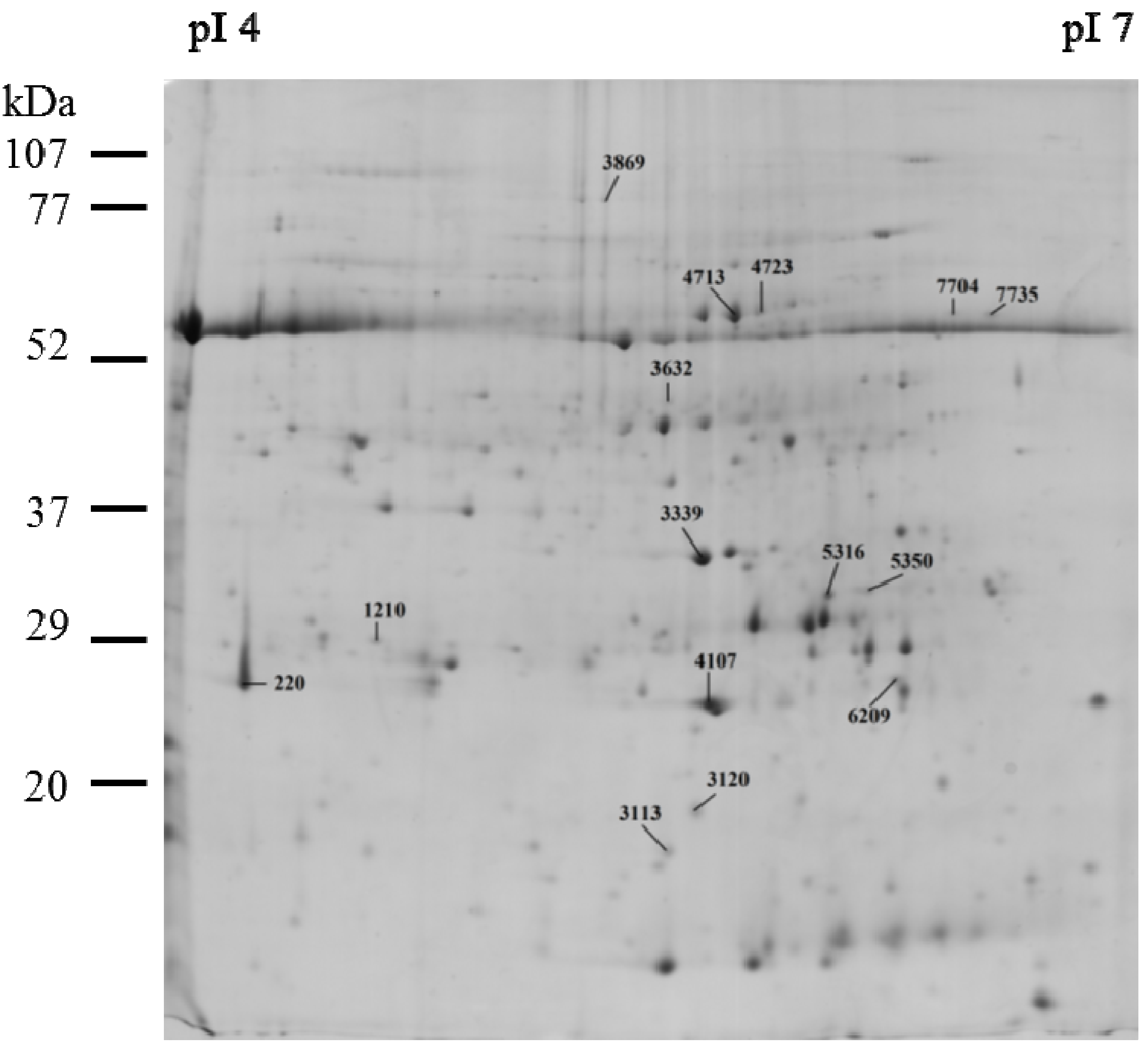
| Biological Function | Protein Name/Accession (NCBI) | SSP | C | MCE | MCP | Stat. | |||
|---|---|---|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | Mean | SD | ||||
| ATP synthesis | ATP synthase CF1 epsilon subunit/gi|89280641 | 3,113 | 2,161.17 | 523.64 | 1,083.10 | 67.11 | 1,614.57 | 289.78 | a↑ |
| ATP synthase CF1 alpha chain/gi|89280620 | 4,713 | 16,331.87 | 415.97 | 9,004.00 | 3,492.91 | 8,123.90 | 4,598.80 | a↓, b↓ | |
| ATP synthase CF1 alpha subunit/gi|28261702 | 4,723 | 1,784.37 | 416.06 | 892.93 | 96.02 | 1,144.53 | 739.62 | a↓ | |
| ATP synthase delta chain, chloroplastic/gi|416681 | 6,209 | 2,544.37 | 344.27 | 1,369.07 | 225.35 | 1,968.50 | 505.96 | a↓ | |
| Carbon fixation | Ribulose-1,5-bisphosphate carboxylase/oxygenase large subunit/gi|77798370 | 7,704 | 265.55 | 197.49 | 503.30 | 0.00 | 650.77 | 158.04 | b↑ |
| Ribulose-1,5-bisphosphate carboxylase/oxygenase large subunit/gi|21634137 | 7,735 | 526.30 | 570.78 | 906.40 | 704.28 | 1331.10 | 293.83 | a↓ | |
| Photosynthesis | Cytochrome b6-f complex iron-sulfur subunit, chloroplastic/gi|68566175 | 3,120 | 2,037.23 | 437.05 | 780.53 | 432.47 | 1,609.97 | 76.46 | b↓ |
| Oxygen-evolving enhancer protein 1, chloroplastic/gi|12644171 | 3,339 | 17,411.43 | 4,066.45 | 21,763.37 | 11,695.21 | 9,877.70 | 688.13 | a↓, b↓ | |
| Oxygen-evolving enhancer protein 2, chloroplastic/gi|350538909 | 4,107 | 29,152.87 | 1,140.43 | 9,704.93 | 4,910.78 | 18,299.77 | 2,863.23 | a↓ | |
| Carbohydrate | Ribose-5-phosphate isomerase/gi|225451299 | 5,316 | 4,453.07 | 361.92 | 2,821.67 | 319.86 | 3,672.17 | 362.55 | a↓ |
| Metabolism | Ribose-5-phosphate isomerase/gi|225451299 | 5,350 | 1,201.00 | 247.53 | 610.03 | 42.48 | 899.83 | 239.29 | b↑ |
| Proteolysis | ATP-dependent Clp protease ATP-binding subunit clpA homolog CD4B, chloroplastic/gi|399213 | 3,869 | 316.30 | 0.00 | 451.53 | 262.70 | 686.13 | 279.57 | a↓, b↓ |
| Embryogenesis and stress response | 24K germin like protein/gi|58244966 | 220 | 18,919.30 | 5,892.38 | 6,637.10 | 948.09 | 7,986.73 | 2,663.81 | b↓ |
| Uncharacterized protein | Heme-binding protein 2/ gi|225470739 | 1210 | 1,193.80 | 332.38 | 711.40 | 223.81 | 576.47 | 187.06 | b↑ |
| Cytoskeleton | Actin/gi|32186906 | 3,632 | 384.07 | 92.56 | 1,043.73 | 1,034.54 | 1,606.87 | 731.04 | a↑ |
2.3. Toxin Quantification in Tomato Tissues
| Tissue | Treatment | MC-LR (μg/kg FW Tissue) | |
|---|---|---|---|
| Week 1 | Week 2 | ||
| Root | C | ND | <LD |
| MCE | ND | 1635.21 ± 941.11 | |
| MCP | ND | <LD | |
| Leaves | C | <LD | <LD |
| MCE | 12,298.18 ± 8962.03 | nd | |
| MCP | ND | <LD | |
| Green tomato | C | <LD | <LD |
| MCE | 5.41 ± 0.49 | <LD | |
| MCP | 5.15 ± 0.93 | <LD | |
| Mature tomato | C | <LD | <LD |
| MCE | 10.52 ± 6.48 | <LD | |
| MCP | 10.83 ± 0.94 | <LD | |
3. Discussion
3.1. Impact of Cyanobacterial Compounds and MC-LR in Plant Metabolism
3.2. Bioaccumulation of MC-LR and Toxin Translocation from Roots to Leaves and Fruits
4. Experimental Section
4.1. Biological Material
4.2. Cyanobacterial Cell Extracts and Quantification of MC-LR
4.3. Experimental Exposure
4.4. MC-LR Extraction, Clean-up and Quantification in Plant Tissues
4.5. Chlorophyll Fluorescence
4.6. Protein Extraction for 2DE
4.7. Two-Dimensional Gel Electrophoresis (2DE)
4.8. Gel Image Acquisition and Protein Expression Analysis
4.9. Protein Identification
4.10. Statistics
5. Conclusions
Supplementary Files
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Prieto, A.I.; Pichardo, S.; Jos, A.; Moreno, I.; Cameán, A.M. Time dependent oxidative stress responses after acute exposure to toxic cyanobacterial cells containing microcystins in tilapia fish (Oreochromis niloticus) under laboratory conditions. Aquat. Toxicol. 2007, 84, 337–345. [Google Scholar] [CrossRef]
- Gurbuz, F.; Metcalf, J.S.; Karahan, A.G.; Codd, G.A. Analysis of dissolved microcystins in surface water samples from Kovada Lake, Turkey. Sci. Total Environ. 2009, 407, 4038–4046. [Google Scholar] [CrossRef]
- Bartram, J.; Carmichael, W.W.; Chorus, I.; Jones, G.; Skulberg, O.S. Introduction. In Toxic cyanobacteria in Water: A Guide to their Public Health Consequences, Monitoring and Management; Chorus, I., Bartram, J., Eds.; World Health Organization; E and FN Spon.: London, UK, 1999; pp. 12–24. [Google Scholar]
- Sivonen, K.; Jones, G. Cyanobacterial toxins. In Toxic Cyanobacteria in Water: A Guide to their Public Health Consequences, Monitoring and Management; Chorus, I., Bartram, J., Eds.; World Health Organization; E and FN Spon.: London, UK, 1999; pp. 41–111. [Google Scholar]
- Wei, Y.; Weng, D.; Li, F.; Zou, X.; Young, D.O.; Ji, J.; Shen, P. Involvement of JNK regulation in oxidative stress-mediated murine liver injury by microcystin-LR. Apoptosis 2008, 13, 1031–1042. [Google Scholar] [CrossRef]
- Puerto, M.; Gutiérrez-Praena, D.; Prieto, A.I.; Pichardo, S.; Jos, A.; Miguel-Carrasco, J.L.; Vázquez, C.M.; Cameán, A.M. Subchronic effects of cyanobacterial cells on the transcription of antioxidant enzyme genes in tilapia (Oreochromis niloticus). Ecotoxicology 2011, 20, 479–490. [Google Scholar] [CrossRef]
- Pichardo, S.; Jos, A.; Zurita, J.L.; Salguero, M.; Cameán, A.M.; Repetto, G. Acute and subacute toxic effects produced by microcystin-YR on the fish cell lines RTG-2 and PLHC-1. Toxicol. In Vitro 2007, 21, 1460–1467. [Google Scholar] [CrossRef]
- Puerto, M.; Pichardo, S.; Jos, A.; Cameán, A.M. Microcystin-LR induces toxic effects in differentiated and undifferentiated Caco-2 cells. Arch. Toxicol. 2010, 84, 405–410. [Google Scholar] [CrossRef]
- International Agency for Research on Cancer (IARC) monographs on the evaluation of carcinogenic risks to humans. Ingested nitrate and nitrite, and cyanobacterial peptide toxins. IARC Monogr. Eval. Carcinog. Risks Hum. 2010, 94, 1–412.
- Atencio, L.; Moreno, I.; Prieto, A.I.; Moyano, R.; Molina, A.M.; Cameán, A.M. Acute effects of Microcystins MC-LR and MC-RR on acid and alkaline phosphatase activities and pathological changes in intraperitoneally exposed tilapia fish (Oreochromis sp.). Toxicol. Pathol. 2008, 36, 449–458. [Google Scholar] [CrossRef]
- Codd, G.; Bell, S.; Kaya, K.; Ward, C.; Beattie, K.; Metcalf, J. Cyanobacterial toxins, exposure routes and human health. Eur. J. Phycol. 1999, 34, 405–415. [Google Scholar] [CrossRef]
- Zurawell, R.W.; Chen, H.R.; Burke, J.M.; Prepas, E.E. patotoxic cyanobacteria: A review of the biological importance of microcystins in freshwater environments. J. Toxicol. Environ. Health B Crit. Rev. 2005, 8, 1–37. [Google Scholar] [CrossRef]
- Xiao, F.G.; Zhao, X.L.; Tang, J.; Gu, X.H.; Zhang, J.P.; Niu, W.M. Necessity of screening water chestnuts for microcystins after cyanobacterial blooms break out. Arch. Environ. Contam. Toxicol. 2009, 57, 256–263. [Google Scholar] [CrossRef]
- Romero-Oliva, C.S.; Contardo-Jara, V.; Block, T.; Pflugmacher, S. Accumulation of microcystin congeners in different aquatic plants and crops—A case study from lake Amatitlán, Guatemala. Ecotoxicol. Environ. Saf. 2014, 102, 121–128. [Google Scholar]
- Mohamed, Z.A.; Al Shehri, A.M. Microcystins in groundwater wells and their accumulation in vegetable plants irrigated with contaminated waters in Saudi Arabia. J. Hazard. Mater. 2009, 172, 310–315. [Google Scholar] [CrossRef]
- Crush, J.R.; Briggs, L.R.; Sprosen, J.M.; Nichols, S.N. Effect of irrigation with lake water containing microcystins on microcystin content and growth of ryegrass, clover, rape, and lettuce. Environ. Toxicol. 2008, 23, 246–252. [Google Scholar] [CrossRef]
- Peuthert, A.; Chakrabarti, S.; Pflugmacher, S. Uptake of microcystins-LR and -LF (cyanobacterial toxins) in seedlings of several important agricultural plant species and the correlation with cellular damage (lipid peroxidation). Environ. Toxicol. 2007, 22, 436–442. [Google Scholar] [CrossRef]
- Kós, P.; Gorzó, G.; Surányi, G.; Borbély, G. Simple and efficient method for isolation and measurement of cyanobacterial hepatotoxins by plant tests (Sinapis alba L.). Anal. Biochem. 1995, 225, 49–53. [Google Scholar] [CrossRef]
- Pereira, S.; Saker, M.L.; Vale, M.; Vasconcelos, V.M. Comparison of sensitivity of grasses (Lolium perenne L. and Festuca rubra L.) and lettuce (Lactuca sativa L.) exposed to water contaminated with microcystins. Bull. Environ. Contam. Toxicol. 2009, 83, 81–84. [Google Scholar]
- El Khalloufi, F.; El Ghazali, I.; Saqrane, S.; Oufdou, K.; Vasconcelos, V.; Oudra, B. Phytotoxic effects of a natural bloom extract containing microcystins on Lycopersicon esculentum. Ecotoxicol. Environ. Saf. 2012, 79, 199–205. [Google Scholar] [CrossRef]
- Chen, J.; Song, L.; Dai, J.; Gan, N.; Liu, Z. Effects of microcystins on the growth and the activity of superoxide dismutase and peroxidase of rape (Brassica napus L.) and rice (Oryza sativa L.). Toxicon 2004, 43, 393–400. [Google Scholar]
- Saqrane, S.; El Ghazali, I.; Oudra, B.; Bouarab, L.; Vasconcelos, V. Effects of cyanobacteria producing microcystins on seed germination and seedling growth of several agricultural plants. J. Environ. Sci. Health B 2008, 43, 443–451. [Google Scholar]
- Máthé, C.; Beyer, D.; Erdodi, F.; Serfozo, Z.; Székvölgyi, L.; Vasas, G.; M-Hamvas, M.; Jámbrik, K.; Gonda, S.; Kiss, A.; et al. Microcystin-LR induces abnormal root development by altering microtubule organization in tissue-cultured common reed (Phragmites australis) plantlets. Aquat. Toxicol. 2009, 92, 122–130. [Google Scholar] [CrossRef]
- El Khalloufi, F.; Oufdou, K.; Lahrouni, M.; El Ghazali, I.; Saqrane, S.; Vasconcelos, V.; Oudra, B. Allelopatic effects of cyanobacteria extracts containing microcystins on Medicago sativa-Rhizobia symbiosis. Ecotoxicol. Environ. Saf. 2011, 74, 431–438. [Google Scholar] [CrossRef]
- Lahrouni, M.; Oufdou, K.; Faghire, M.; Peix, A.; El Khalloufi, F.; Vasconcelos, V.; Oudra, B. Cyanobacterial extracts containing microcystins affect the growth, nodulation process and nitrogen uptake of faba bean (Vicia faba L., Fabaceae). Ecotoxicology 2012, 21, 681–687. [Google Scholar] [CrossRef]
- Smith, R.D.; Walker, J.C. Plant protein phosphatases. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1996, 47, 101–125. [Google Scholar] [CrossRef]
- Pflugmacher, S.; Hofmann, J.; Hübner, B. Effects on growth and physiological parameters in wheat (Triticum aestivum L.) grown in soil and irrigated with cyanobacterial toxin contaminated water. Environ. Toxicol. Chem. 2007, 26, 2710–2716. [Google Scholar] [CrossRef]
- Peuthert, A.; Pflugmacher, S. Influence of the cyanotoxin microcystin-LR on tocopherol in Alfalfa seedlings (Medicago sativa). Toxicon 2010, 56, 411–417. [Google Scholar] [CrossRef]
- Prieto, A.; Campos, A.; Cameán, A.; Vasconcelos, V. Effects on growth and oxidative stress status of rice plants (Oryza sativa) exposed to two extracts of toxin-producing cyanobacteria (Aphanizomenon ovalisporum and Microcystis aeruginosa). Ecotoxicol. Environ. Saf. 2011, 74, 1973–1980. [Google Scholar] [CrossRef]
- Saqrane, S.; Ouahid, Y.; El Ghazali, I.; Oudra, B.; Bouarab, L.; del Campo, F.F. Physiological changes in Triticum durum, Zea mays, Pisum sativum and Lens esculenta cultivars, caused by irrigation with water contaminated with microcystins: A laboratory experimental approach. Toxicon 2009, 53, 786–796. [Google Scholar] [CrossRef]
- Saqrane, S.; Ghazali, I.E.; Ouahid, Y.; Hassni, M.E.; Hadrami, I.E.; Bouarab, L.; del Campo, F.F.; Oudra, B.; Vasconcelos, V. Phytotoxic effects of cyanobacteria extract on the aquatic plant Lemna gibba: Microcystin accumulation, detoxication and oxidative stress induction. Aquat. Toxicol. 2007, 83, 284–294. [Google Scholar] [CrossRef]
- Bibo, L.; Yan, G.; Bangding, X.; Jiantong, L.; Yongding, L. A laboratory study on risk assessment of microcystin-RR in cropland. J. Environ. Manage. 2008, 86, 566–574. [Google Scholar] [CrossRef]
- Chen, J.; Dai, J.; Zhang, H.; Wang, C.; Zhou, G.; Han, Z.; Liu, Z. Bioaccumulation of microcystin and its oxidative stress in the apple (Malus pumila). Ecotoxicology 2010, 19, 796–803. [Google Scholar] [CrossRef]
- Campos, A.; Tedesco, S.; Vasconcelos, V.; Cristobal, S. Proteomic research in bivalves: Towards the identification of molecular markers of aquatic pollution. J. Proteomics 2012, 75, 4346–4359. [Google Scholar] [CrossRef]
- Yin, L.; Huang, J.; Huang, W.; Li, D.; Wang, G.; Liu, Y. Microcystin-RR-induced accumulation of reactive oxygen species and alteration of antioxidant systems in tobacco BY-2 cells. Toxicon 2005, 46, 507–512. [Google Scholar] [CrossRef]
- Peuthert, A.; Lawton, L.; Pflugmacher, S. In vivo influence of cyanobacterial toxins on enzyme activity and gene expression of protein phosphatases in Alfalfa (Medicago sativa). Toxicon 2008, 52, 84–90. [Google Scholar] [CrossRef]
- Chen, J.; Han, F.X.; Wang, F.; Zhang, H.; Shi, Z. Accumulation and phytotoxicity of microcystin-LR in rice (Oryza sativa). Ecotoxicol. Environ. Saf. 2012, 76, 193–199. [Google Scholar] [CrossRef]
- Jámbrik, K.; Máthé, C.; Vasas, G.; Beyer, D.; Molnár, E.; Borbély, G.; M-Hamvas, M. Microcystin-LR induces chromatin alterations and modulates neutral single-strand-preferring nuclease activity in Phragmites australis. J. Plant Physiol. 2011, 168, 678–686. [Google Scholar] [CrossRef]
- Buratti, F.M.; Scardala, S.; Funari, E.; Testai, E. The conjugation of microcystin-RR by human recombinant GSTs and hepatic cytosol. Toxicol. Lett. 2013, 219, 231–238. [Google Scholar] [CrossRef]
- Setlíková, I.; Wiegand, C. Hepatic and branchial glutathione S-transferases of two fish species: Substrate specificity and biotransformation of microcystin-LR. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2009, 149, 515–523. [Google Scholar] [CrossRef]
- Suchy, P.; Berry, J. Detection of total microcystin in fish tissues based on lemieux oxidation, and recovery of 2-methyl-3-methoxy-4-phenylbutanoic acid (MMPB) by solid-phase microextraction gas chromatography-mass spectrometry (SPME-GC/MS). Int. J. Environ. Anal. Chem. 2012, 92, 1443–1456. [Google Scholar] [CrossRef]
- Trapp, S.; Eggen, T. Simulation of the plant uptake of organophosphates and other emerging pollutants for greenhouse experiments and field conditions. Environ. Sci. Pollut. Res. Int. 2013, 20, 4018–4029. [Google Scholar] [CrossRef]
- Václavík, J. Effect of different leaf age on the relationship between the CO2 uptake and water vapour efflux in tobacco plants. Biol. Plant. 1973, 15, 233–236. [Google Scholar] [CrossRef]
- Constable, G.A.; Rawson, H.M. Effect of leaf position, expansion and age on photosynthesis, transpiration and water use efficiency of cotton. Aust. J. Plant Physiol. 1980, 7, 89–100. [Google Scholar] [CrossRef]
- Saker, M.L.; Nogueira, I.C.; Vasconcelos, V.M.; Neilan, B.A.; Eaglesham, G.K.; Pereira, P. First report and toxicological assessment of the cyanobacterium Cylindrospermopsis raciborskii from Portuguese freshwaters. Ecotoxicol. Environ. Saf. 2003, 55, 243–250. [Google Scholar] [CrossRef]
- Vasconcelos, V.M. Uptake and depuration of the heptapeptide toxin microcystin-LR in Mytilus galloprovincialis. Aquat. Toxicol. 1995, 32, 227–237. [Google Scholar] [CrossRef]
- Moreno, I.M.; Pereira, P.; Franca, S.; Cameán, A. Toxic cyanobacteria strains isolated from blooms in the Guadiana river (southwestern Spain). Biol. Res. 2004, 37, 405–417. [Google Scholar]
- Pinheiro, C.; Azevedo, J.; Campos, A.; Loureiro, S.; Vasconcelos, V. Absence of negative allelopathic effects of cylindrospermopsin and microcystin-LR in selected marine and freshwater phytoplankton species. Hydrobiologia 2013, 705, 27–42. [Google Scholar] [CrossRef]
- Maxwell, K.; Johnson, G.N. Chlorophyll fluorescence—A practical guide. J. Exp. Bot. 2000, 51, 659–668. [Google Scholar] [CrossRef]
- Campos, A.; Da Costa, G.; Coelho, A.V.; Fevereiro, P. Identification of bacterial protein markers and enolase as a plant response protein in the infection of Olea europaea subsp. europaea by Pseudomonas savastanoi pv. Savastanoi. Eur. J. Plant Pathol. 2009, 125, 603–616. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Neuhoff, V.; Arold, N.; Taube, D.; Ehrhardt, W. Improved staining of proteins in polyacrylamide gels including isoelectric focusing gels with clear background at nanogram sensitivity using Coomassie Brilliant Blue G-250 and R-250. Electrophoresis 1988, 9, 255–262. [Google Scholar] [CrossRef]
- Puerto, M.; Campos, A.; Prieto, A.; Cameán, A.; de Almeida, A.M.; Coelho, A.V.; Vasconcelos, V. Differential protein expression in two bivalve species; Mytilus galloprovincialis and Corbicula fluminea; exposed to Cylindrospermopsis raciborskii cells. Aquat. Toxicol. 2011, 101, 109–116. [Google Scholar] [CrossRef]
- Pandey, A.; Mann, M. Proteomics to study genes and genomes. Nature 2000, 405, 837–846. [Google Scholar] [CrossRef]
- Gobom, J.; Nordhoff, E.; Mirgorodskaya, E.; Ekman, R.; Roepstorff, P. Sample purification and preparation technique based on nano-scale reversed-phase columns for the sensitive analysis of complex peptide mixtures by matrix-assisted laser desorption/ionization mass spectrometry. J. Mass Spectrom. 1999, 34, 105–116. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Gutiérrez-Praena, D.; Campos, A.; Azevedo, J.; Neves, J.; Freitas, M.; Guzmán-Guillén, R.; Cameán, A.M.; Renaut, J.; Vasconcelos, V. Exposure of Lycopersicon Esculentum to Microcystin-LR: Effects in the Leaf Proteome and Toxin Translocation from Water to Leaves and Fruits. Toxins 2014, 6, 1837-1854. https://doi.org/10.3390/toxins6061837
Gutiérrez-Praena D, Campos A, Azevedo J, Neves J, Freitas M, Guzmán-Guillén R, Cameán AM, Renaut J, Vasconcelos V. Exposure of Lycopersicon Esculentum to Microcystin-LR: Effects in the Leaf Proteome and Toxin Translocation from Water to Leaves and Fruits. Toxins. 2014; 6(6):1837-1854. https://doi.org/10.3390/toxins6061837
Chicago/Turabian StyleGutiérrez-Praena, Daniel, Alexandre Campos, Joana Azevedo, Joana Neves, Marisa Freitas, Remédios Guzmán-Guillén, Ana María Cameán, Jenny Renaut, and Vitor Vasconcelos. 2014. "Exposure of Lycopersicon Esculentum to Microcystin-LR: Effects in the Leaf Proteome and Toxin Translocation from Water to Leaves and Fruits" Toxins 6, no. 6: 1837-1854. https://doi.org/10.3390/toxins6061837
APA StyleGutiérrez-Praena, D., Campos, A., Azevedo, J., Neves, J., Freitas, M., Guzmán-Guillén, R., Cameán, A. M., Renaut, J., & Vasconcelos, V. (2014). Exposure of Lycopersicon Esculentum to Microcystin-LR: Effects in the Leaf Proteome and Toxin Translocation from Water to Leaves and Fruits. Toxins, 6(6), 1837-1854. https://doi.org/10.3390/toxins6061837









