Fluorescence Localization and Comparative Ultrastructural Study of Periplocoside NW from Periploca sepium Bunge in the Midgut of the Oriental Amyworm, Mythimna separata Walker (Lepidoptera: Noctuidae)
Abstract
:1. Introduction
2. Results
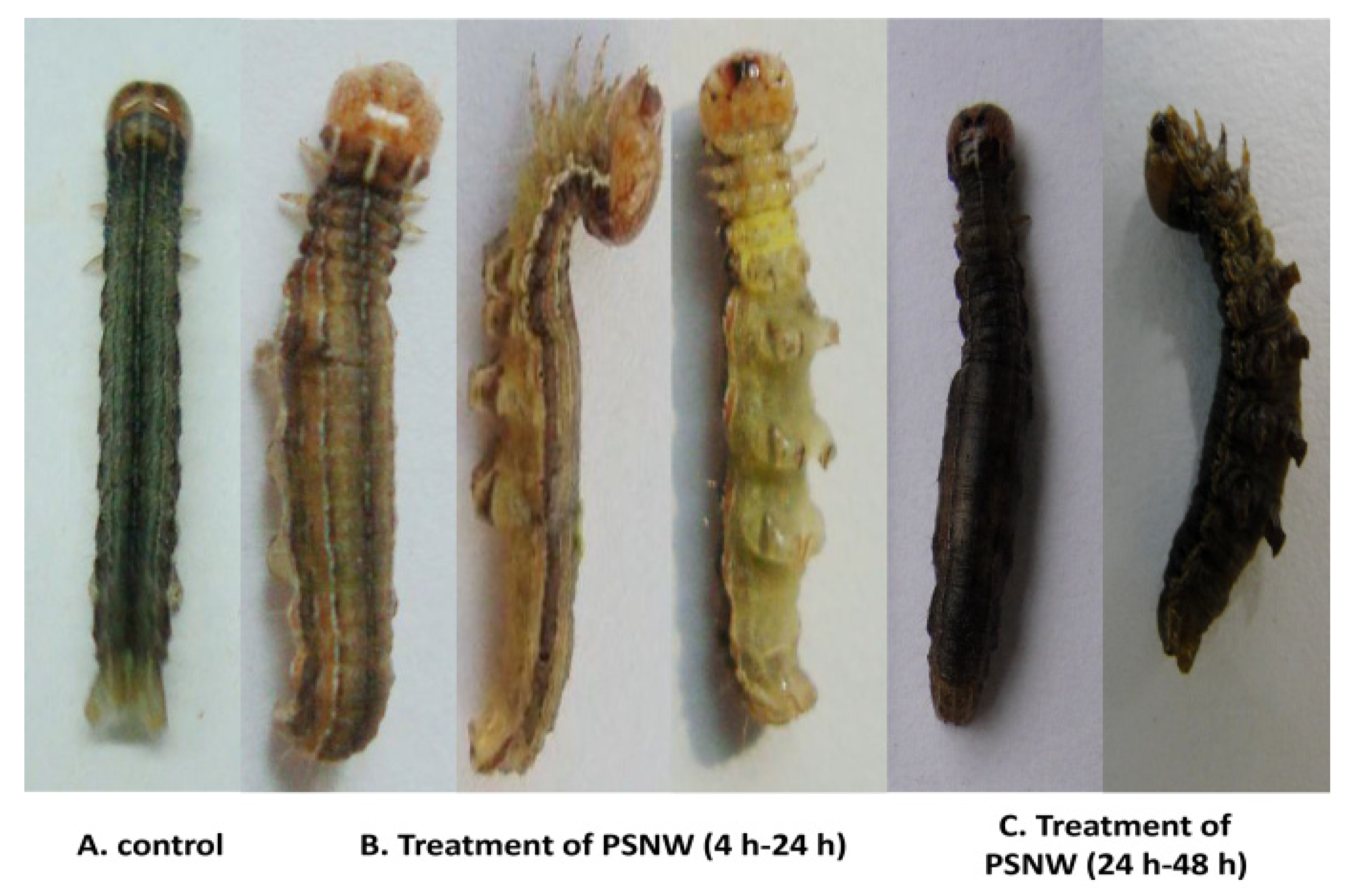
2.1. Symptom Observations
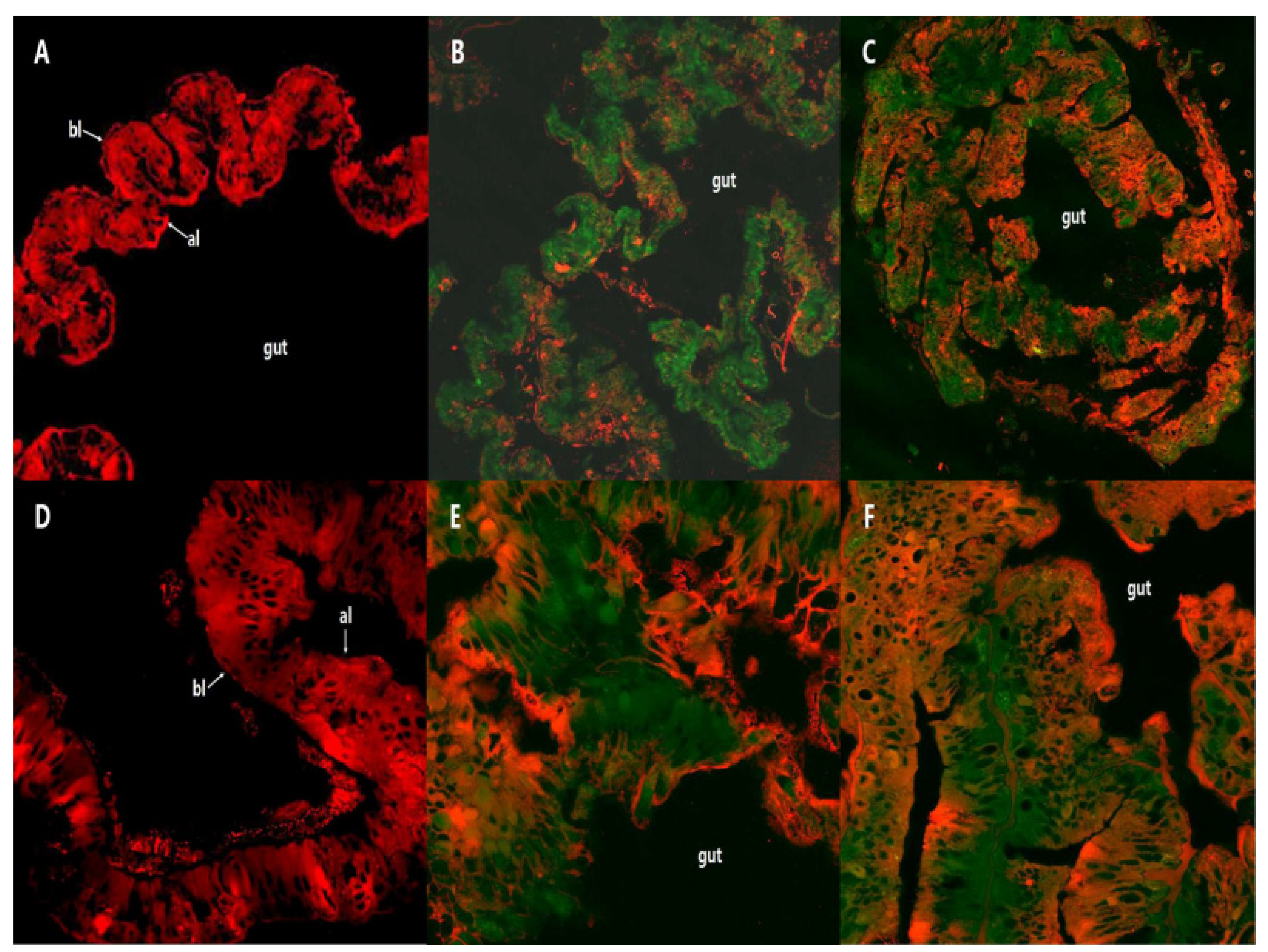
2.2. Fluorescence Localization of PSNW in the Midgut of M. separata Larvae
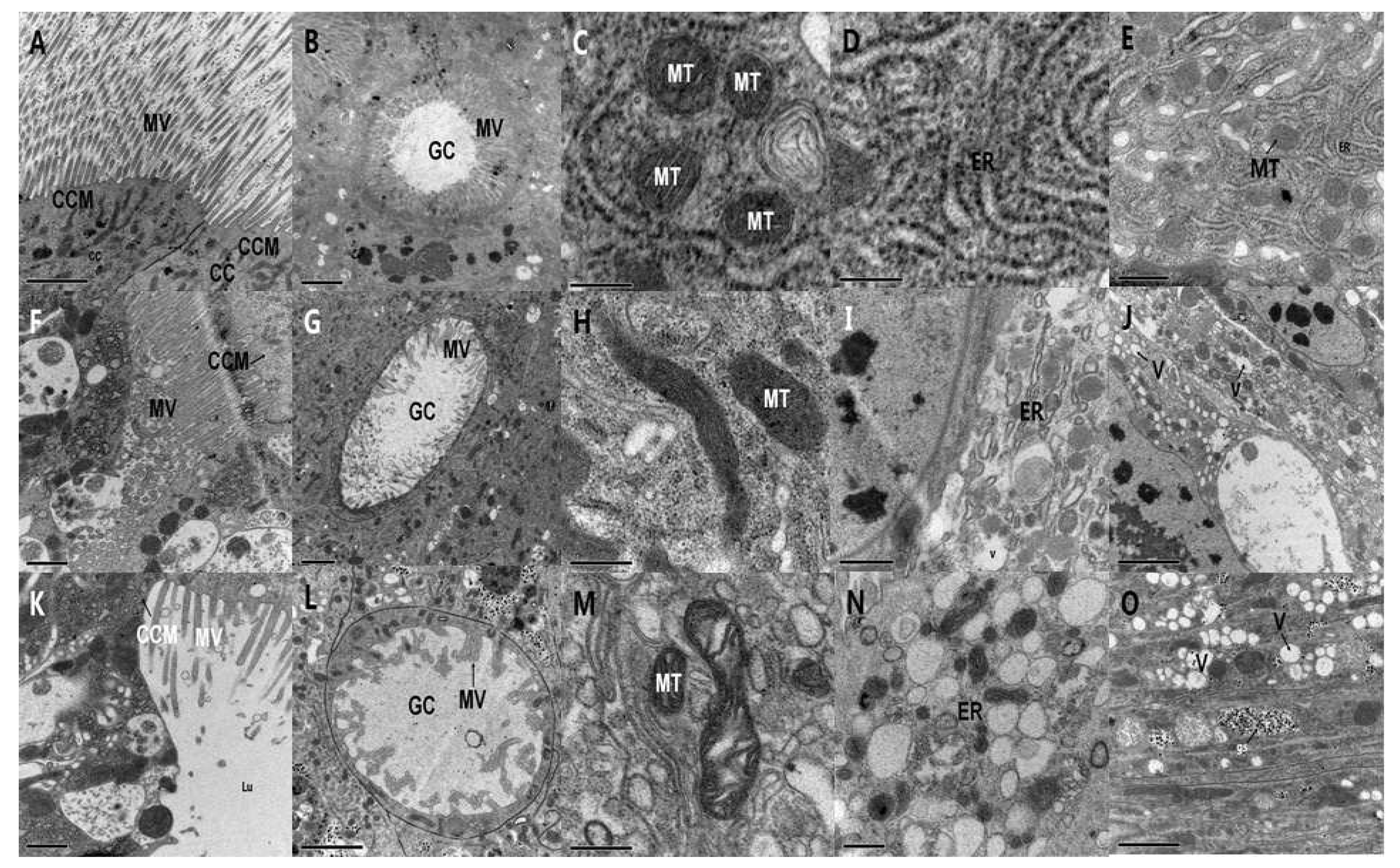
2.3. Comparative Ultrastructural Study of the Effect of PSNW on the Midgut of M. separata Larvae and A. ispilon Larvae
2.3.1. Ultrastructure Effects of PSNW on Midgut Cells of M. separata Larvae
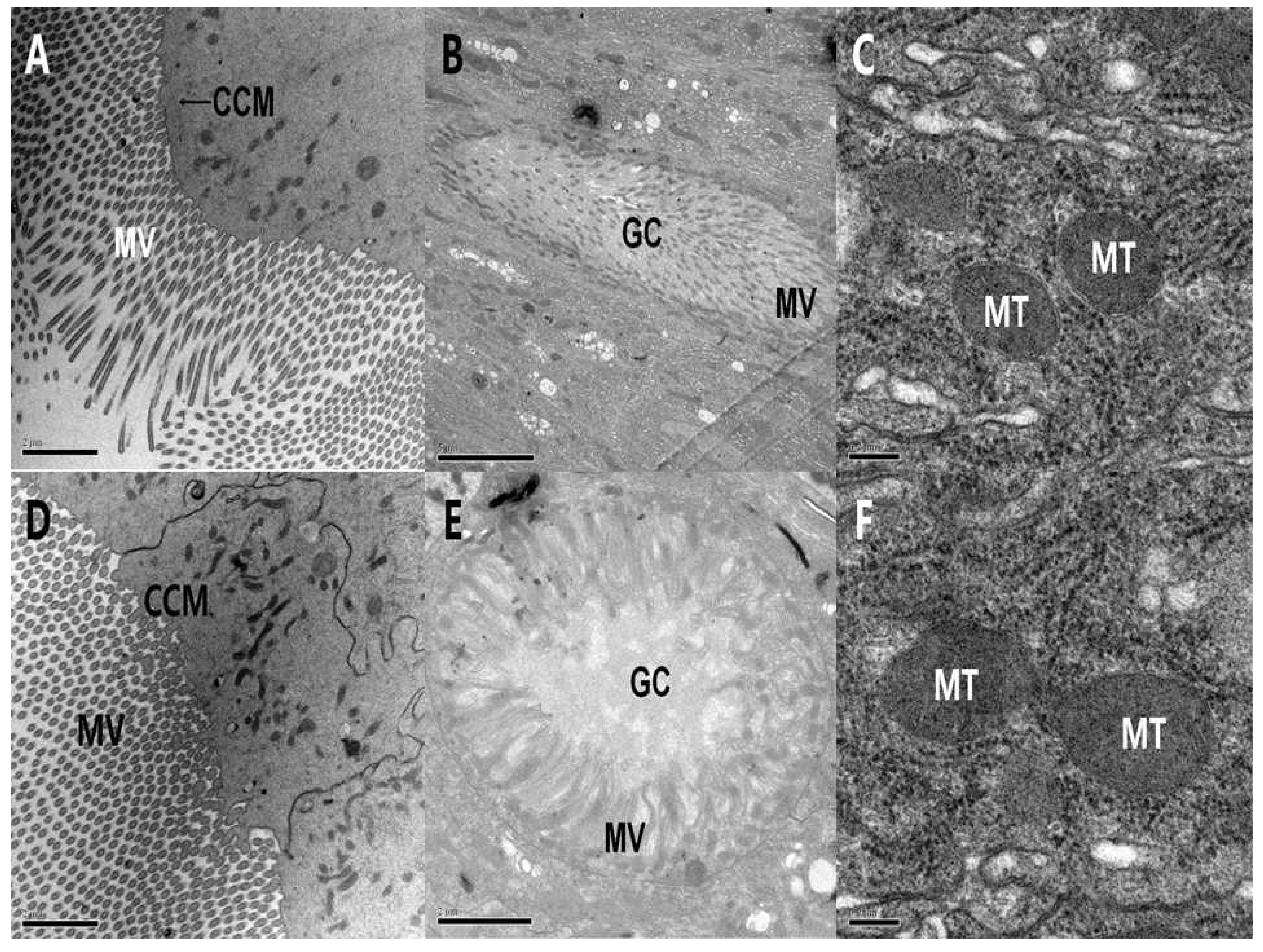
2.3.2. Ultrastructure Effects of PSNW on the Midgut Cells of A. ispilon Larvae
3. Discussion
4. Materials and Methods
4.1. Compounds
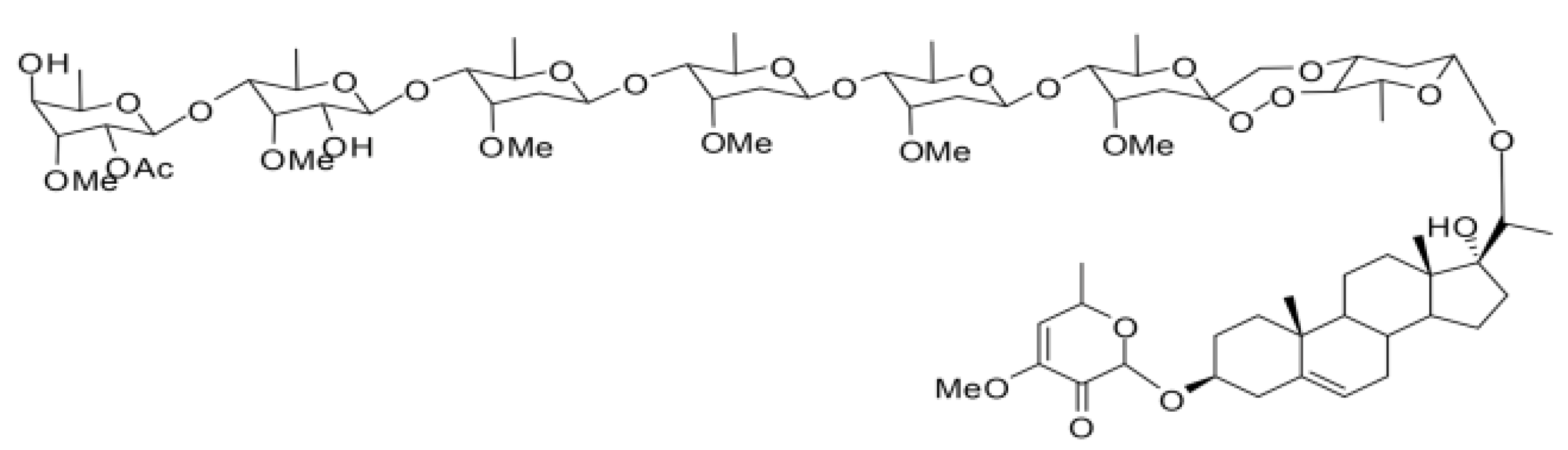
4.2. Insects
4.3. Insect Treatment
4.4. Symptoms Observation
4.5. Fluorescence Localization Studies
4.5.1. Fluorescence Marker of PSNW (FNW)
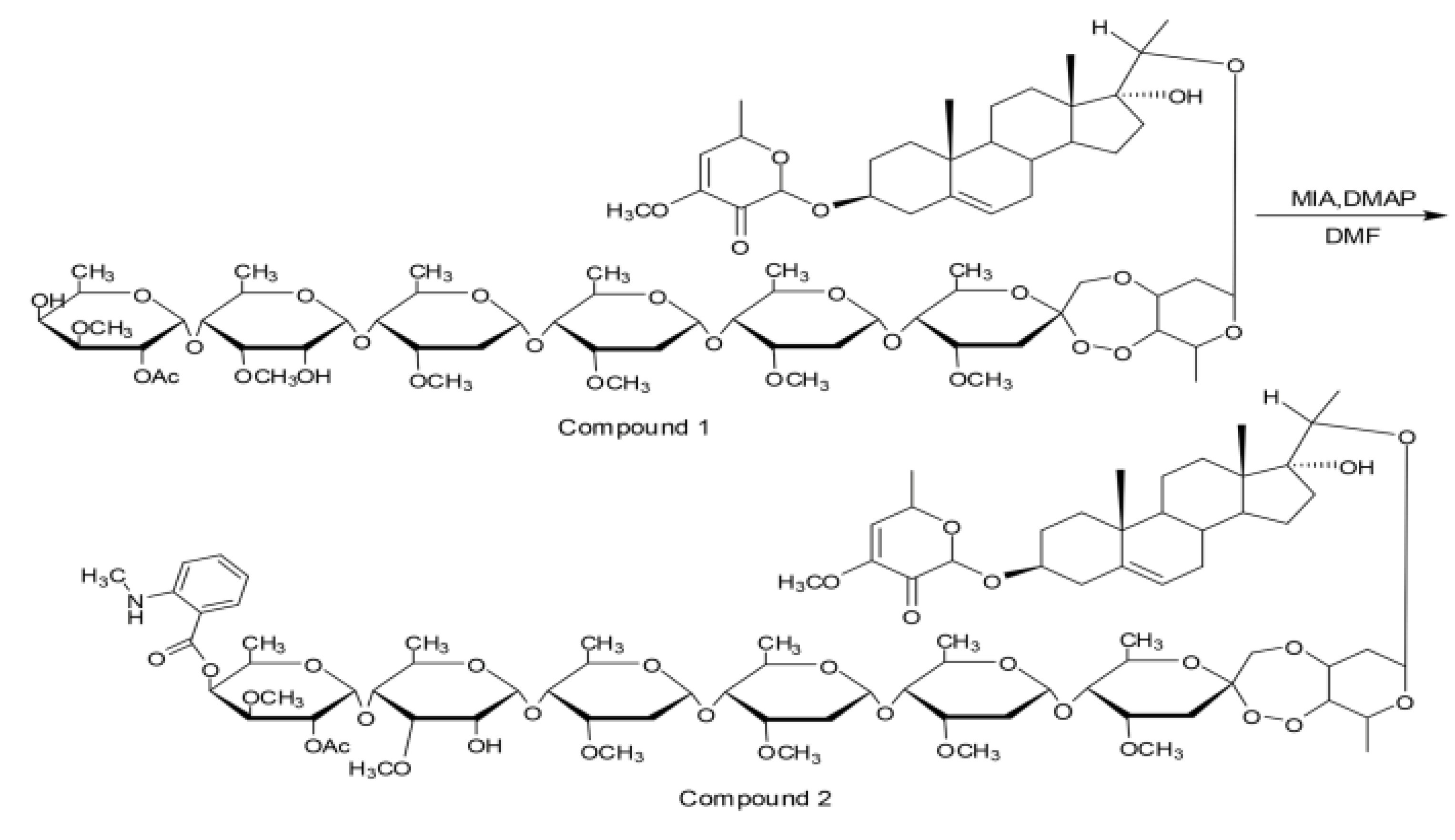
4.5.2. Incubation
4.5.3. Fixing and Staining
4.5.4. CLSM
4.6. Transmission Electron Microscopy (TEM)
5. Conclusions
Abbreviations
| MV | microvilli |
| GC | goblet cell |
| CC | columnar cell |
| CCM | columnar cell membrane |
| MT | mitochondria |
| ER | endoplasmic reticulum |
| V | vacuole |
| Lu | midgut lumen |
| bl | basal lamina |
| al | apical lamina |
| gs | â-type glycogenosome |
Acknowledgements
Author Contributions
Conflicts of Interest
References
- Isman, M.B. Botanical insecticides, deterrents, and repellents in modern agriculture and an increasingly regulated world. Annu. Rev. Entomol. 2006, 51, 45–66. [Google Scholar] [CrossRef]
- Duke, S.O.; Cantrell, C.L.; Meepagala, K.M.; Wedge, D.E.; Tabanca, N.; Schrader, K.K. Natural toxins for use in pest management. Toxins 2010, 2, 1943–1962. [Google Scholar] [CrossRef]
- Rosell, G.; Quero, C.; Coll, J.; Guerrero, A. Biorational insecticides in pest management. J. Pesti. Sci. 2008, 33, 103–121. [Google Scholar] [CrossRef]
- Lü, M.; Wu, W.J.; Liu, H.X. Effects of fraxinellone on the midgut ultrastructural changes of Mythimna separata Walker. Pestic. Biochem. Physiol. 2010, 98, 263–268. [Google Scholar] [CrossRef]
- Tokiwa, T.; Harada, K.; Matsumura, T.; Tukiyama, T. Oriental medicinal herb, Periploca sepium, extract inhibits growth and IL-6 production of human synovial fibroblast-like cells. Biol. Pharm. Bull. 2004, 27, 1691–1693. [Google Scholar] [CrossRef]
- Chu, S.S.; Jiang, G.H.; Liu, W.L.; Liu, Z.L. Insecticidal activity of the root bark essential oil of Periploca sepium Bunge and its main component. Nat. Prod. Res. 2012, 26, 926–932. [Google Scholar] [CrossRef]
- Hu, Z.N.; Lian, X.H.; Liu, J.; Wu, W.J. Effects of periplocoside NW from Periploca. sepium on muscle cell [Ca2+]i of adults of Mythimna separata. Highlights Sciencepaper Online 2012, 5, 1858–1863. [Google Scholar]
- Zhao, Y.C.; Shi, B.J.; Hu, Z.N. The insecticidal activity of periplocoside NW. Chin. Bull. Entomol. 2008, 6, 022. [Google Scholar]
- Singh, P. A general purpose laboratory diet mixture for rearing insects. Int. J. Trop. Insect Sci. 1983, 4, 357–362. [Google Scholar] [CrossRef]
- Fuxe, K. Cellular localization of monoamines in the median eminence and the infundibular stem of some mammals. Z. Zellforsch. Mikrosk. Anatomie 1964, 61, 710–724. [Google Scholar] [CrossRef]
- Rocha, L.L.V.; Neves, C.A.; Zanuncio, J.C.; Serrão, J.É. Digestive cells in the midgut of Triatoma vitticeps (Stal, 1859) in different starvation periods. Comptes. Rendus. Biol. 2010, 333, 405–415. [Google Scholar] [CrossRef]
- Gross, E.M.; Brune, A.; Walenciak, O. Gut pH, redox conditions and oxygen levels in an aquatic caterpillar: Potential effects on the fate of ingested tannins. J. Insect Physiol. 2008, 54, 462–471. [Google Scholar] [CrossRef]
- Brown, S.E.; Cao, A.T.; Dobson, P.; Hines, E.R. Txp40, A ubiquitous insecticidal toxin protein from Xenorhabdus and Photorhabdus bacteria. Appl. Environ. Microbiol. 2006, 72, 1653–1662. [Google Scholar] [CrossRef]
- Scudeler, E.L.; Santos, D.C. Effects of neem oil (Azadirachta indica A. Juss) on midgut cells of predatory larvae Ceraeochrysa claveri (Navás, 1911) (Neuroptera: Chrysopidae). Micron. 2013, 44, 125–132. [Google Scholar] [CrossRef]
- Qi, Z.J.; Xue, X.P.; Wu, W.J.; Zhang, J.W.; Yang, R.Y. Preparation of monoclonal antibody against celangulin V and immunolocalization of receptor in the oriental armyworm, Mythimna separata walker (Lepidoptera: Noctuidae). J. Agric. Food Chem. 2006, 54, 7600–7605. [Google Scholar] [CrossRef]
- Qi, Z.J.; Shi, B.J.; Hu, Z.N.; Zhang, Y.X. Ultrastructural effects of Celangulin V on midgut cells of the oriental armyworm, Mythimna separata walker (Lepidoptera: Noctuidae). Ecotoxicol. Environ. Saf. 2011, 74, 439–444. [Google Scholar] [CrossRef]
- Gill, S.S.; Cowles, E.A.; Pietrantonio, P.V. The mode of action of Bacillus thuringiensis endotoxins. Annu. Rev. Entomol. 1992, 37, 615–634. [Google Scholar] [CrossRef]
- Aronson, A.I.; Shai, Y. Why Bacillus thuringiensis insecticidal toxins are so effective: unique features of their mode of action. FEMS Microbiol. Lett. 2001, 195, 1–8. [Google Scholar] [CrossRef]
- Li, Y.F.; Liu, Y.L.; Song, Z.Q. Research advances on the insecticidal plant Celastrus angulatus. Pestic. Shenyang 2006, 45, 148. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Feng, M.; Zhao, J.; Zhang, J.; Hu, Z.; Wu, W. Fluorescence Localization and Comparative Ultrastructural Study of Periplocoside NW from Periploca sepium Bunge in the Midgut of the Oriental Amyworm, Mythimna separata Walker (Lepidoptera: Noctuidae). Toxins 2014, 6, 1575-1585. https://doi.org/10.3390/toxins6051575
Feng M, Zhao J, Zhang J, Hu Z, Wu W. Fluorescence Localization and Comparative Ultrastructural Study of Periplocoside NW from Periploca sepium Bunge in the Midgut of the Oriental Amyworm, Mythimna separata Walker (Lepidoptera: Noctuidae). Toxins. 2014; 6(5):1575-1585. https://doi.org/10.3390/toxins6051575
Chicago/Turabian StyleFeng, Mingxing, Juan Zhao, Jiwen Zhang, Zhaonong Hu, and Wenjun Wu. 2014. "Fluorescence Localization and Comparative Ultrastructural Study of Periplocoside NW from Periploca sepium Bunge in the Midgut of the Oriental Amyworm, Mythimna separata Walker (Lepidoptera: Noctuidae)" Toxins 6, no. 5: 1575-1585. https://doi.org/10.3390/toxins6051575
APA StyleFeng, M., Zhao, J., Zhang, J., Hu, Z., & Wu, W. (2014). Fluorescence Localization and Comparative Ultrastructural Study of Periplocoside NW from Periploca sepium Bunge in the Midgut of the Oriental Amyworm, Mythimna separata Walker (Lepidoptera: Noctuidae). Toxins, 6(5), 1575-1585. https://doi.org/10.3390/toxins6051575



