First Report of Cylindrospermopsin Production by Two Cyanobacteria (Dolichospermum mendotae and Chrysosporum ovalisporum) in Lake Iznik, Turkey
Abstract
:1. Introduction
2. Results and Discussion
| Parameters | Units | Date | ||
|---|---|---|---|---|
| 22 May 2013 | 22 August 2013 | 28 August 2013 | ||
| Water Temperature | °C | 20.6 | 27.0 | 27.4 |
| pH | - | 8.9 | 8.6 | 9.2 |
| Conductivity | µS·cm−1 | 900 | 1037 | 1056 |
| Secchi depth | m | 7.9 | 1.4 | 1.4 |
| Nitrate + Nitrite | µg·L−1 | 125.6 | 391.4 | 69.6 |
| Soluble Reactive Phosphorus | µg·L−1 | 11.3 | 2.5 | <2 |
| Total Phosphorus | µg·L−1 | 24.8 | 12.7 | 13.4 |
| Total Nitrogen | µg·L−1 | 1889 | 1562 | 1253 |
| Chlorophyll-a | µg·L−1 | 4.2 | 20.9 | 23.1 |
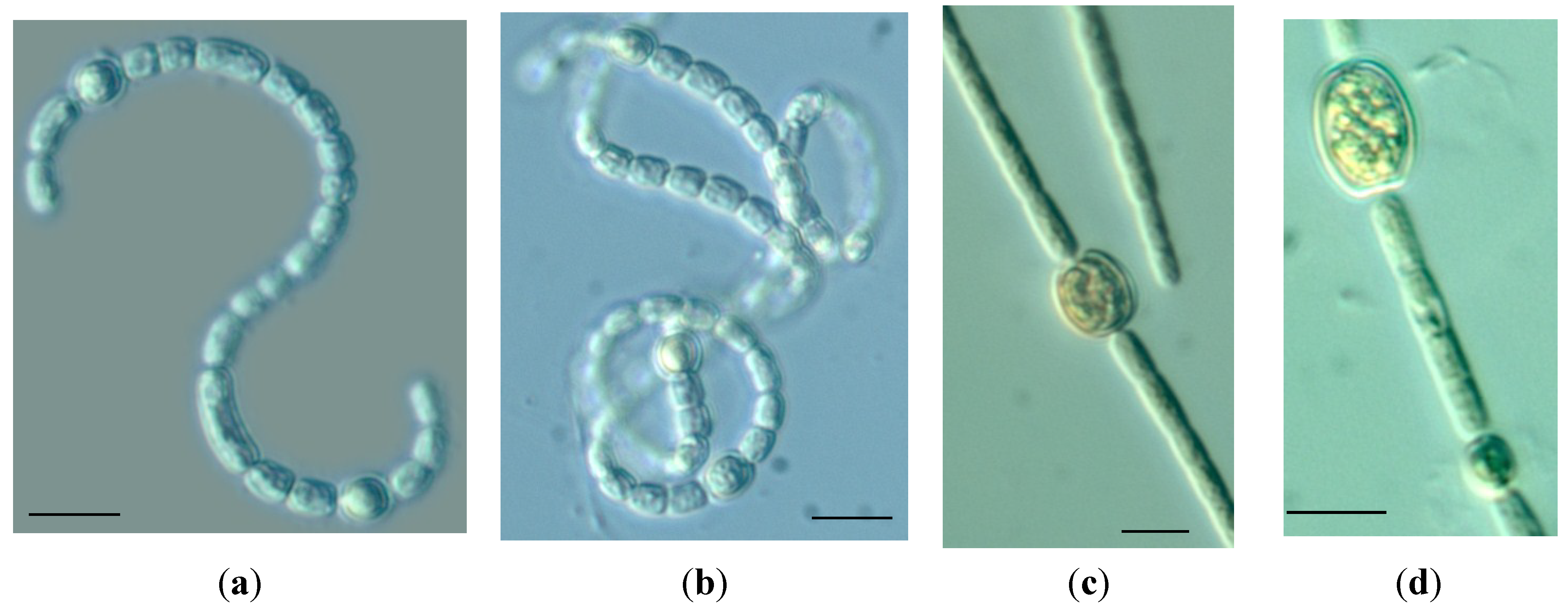
| Species | Morphological characters | Form | Mean ± SD | Min | Max | n |
|---|---|---|---|---|---|---|
| D. mendotae | ||||||
| Cell | ||||||
| length | 4.8 ± 0.7 | 3.4 | 5.8 | 50 | ||
| width | 4.2 ± 0.3 | 3.5 | 5 | |||
| l:w | 1.1 | 0.98 | 1.2 | |||
| Heterocyst | ||||||
| length | 6.5 ± 0.8 | 4.7 | 8.4 | 50 | ||
| width | 5.6 ± 0.5 | 4.1 | 6.6 | |||
| l:w | 1.2 | 1.15 | 1.3 | |||
| Akinete | ||||||
| length | 8.96 ± 1.8 | 7.2 | 11.3 | 5 | ||
| width | 4.8 ± 0.7 | 4.1 | 5.7 | |||
| l:w | 1.9 | 1.8 | 2 | |||
| C. ovalisporum | ||||||
| Cell | ||||||
| length | 6.1 ± 1.8 | 2.9 | 13.4 | 50 | ||
| width | 3.4 ± 0.4 | 2.7 | 4.4 | |||
| l:w | 1.8 | 1.1 | 3 | |||
| Heterocyst | ||||||
| length | 6.2 ± 0.7 | 4.9 | 7.9 | 50 | ||
| width | 4.3 ± 0.7 | 2.7 | 6.5 | |||
| l:w | 1.4 | 1.8 | 1.2 | |||
| Akinete | ||||||
| length | 12.1 ± 2 | 7.9 | 15.5 | 15 | ||
| width | 9.6 ± 1.8 | 6.9 | 14.5 | |||
| l:w | 1.3 | 1.1 | 1.07 |
| Species | Status | Geographical Area | Toxicity | Reference |
|---|---|---|---|---|
| D. mendotae | Environmental | Finland | NA | [21] |
| Bangladesh | NA | [23] | ||
| Brazil | NA | [24] | ||
| Hungary | NA | [22] | ||
| Germany | NA | [25] | ||
| Poland | NA | [26] | ||
| Czech Republic | NA | [20] | ||
| Greece | NA | [27] | ||
| Strain | Finland | Ana | [28] | |
| Japan | NA | [29] | ||
| Denmark | NT | [30] | ||
| Spain | NT* | [31] | ||
| C. ovalisporum | Environmental | Israel | CYN | [10] |
| Greece | MCY | [11] | ||
| Spain | CYN | [12] | ||
| Italy | CYN | [7] | ||
| Strain | Israel | CYN | [10] | |
| Australia | CYN | [13] | ||
| USA | CYN | [32] | ||
| Spain | CYN | [31] |
| Species and toxin | Units | Date | |||
|---|---|---|---|---|---|
| 22 May 2013 | 22 August 2013 | 28 August 2013 | |||
| D. mendotae | Abundance | Cell·L−1 | 4.9 × 107 | - | - |
| Biomass | µg·L−1 | 3471 | - | - | |
| C. ovalisporum | Abundance | Fil·L−1 | - | 2.3 × 107 | 2.0 × 107 |
| Biomass | µg·L−1 | - | 20,960 | 18,413 | |
| Cylindrospermopsin | µg·L−1 | 0.12 | 3.91 | 4.92 | |
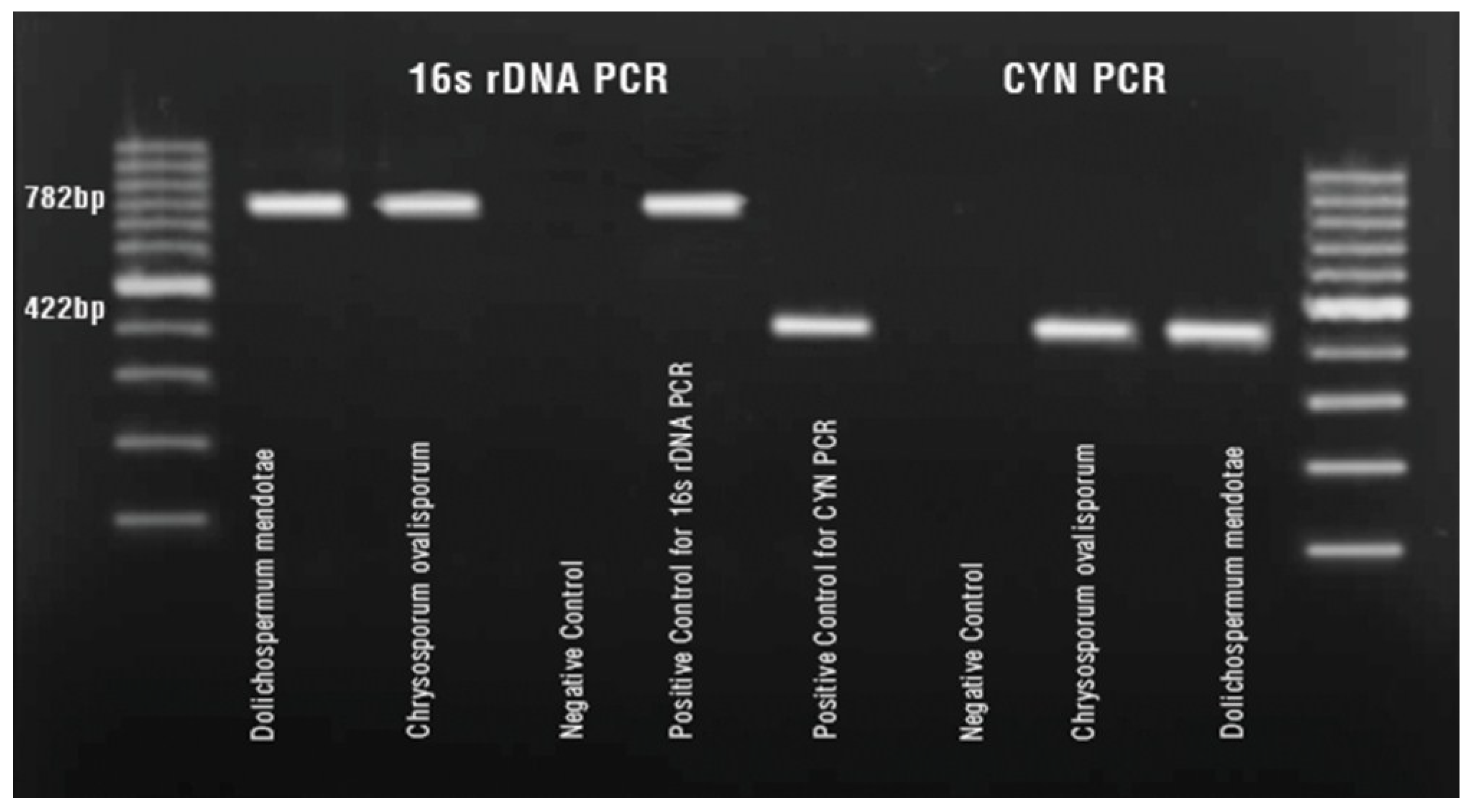
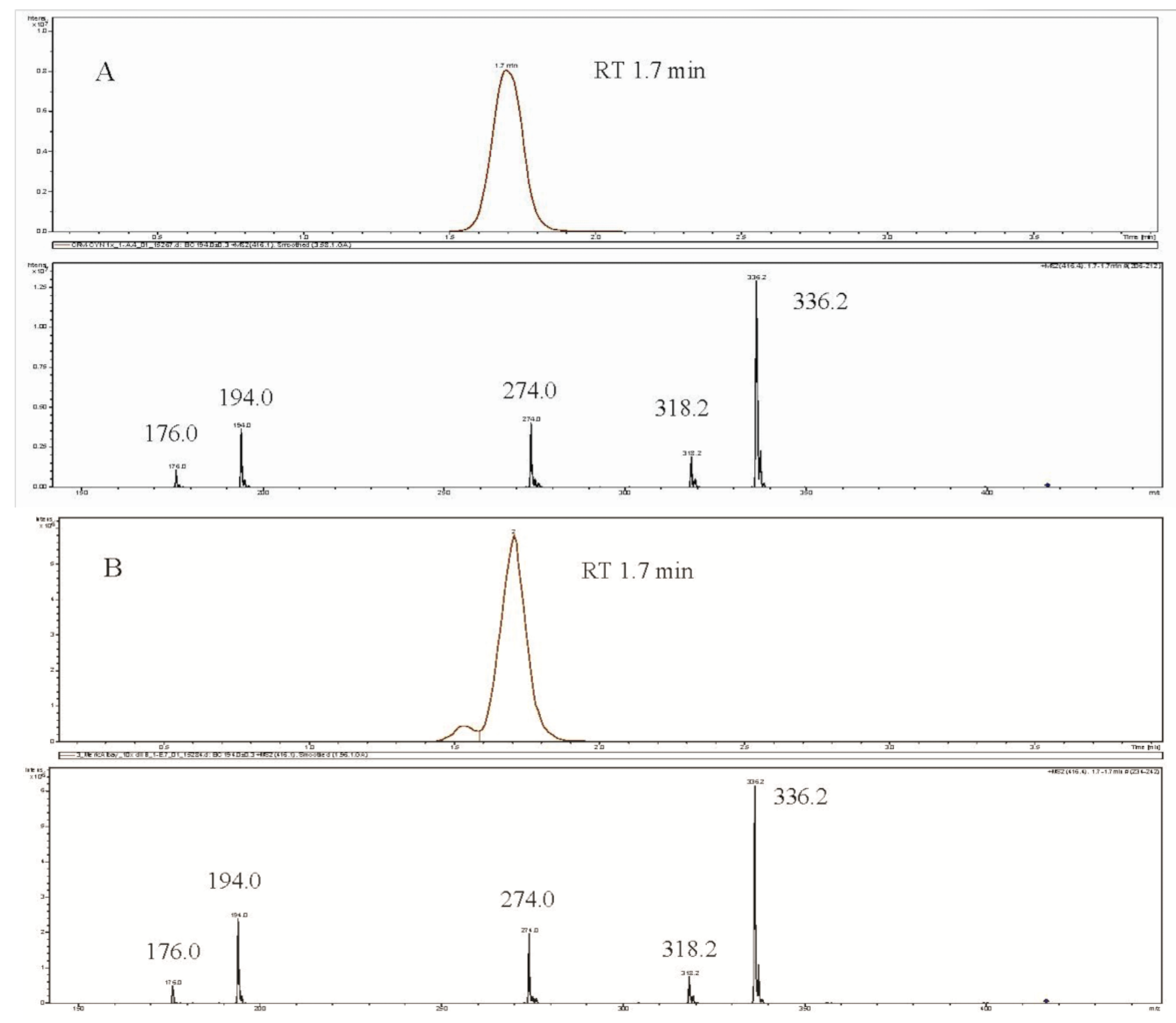
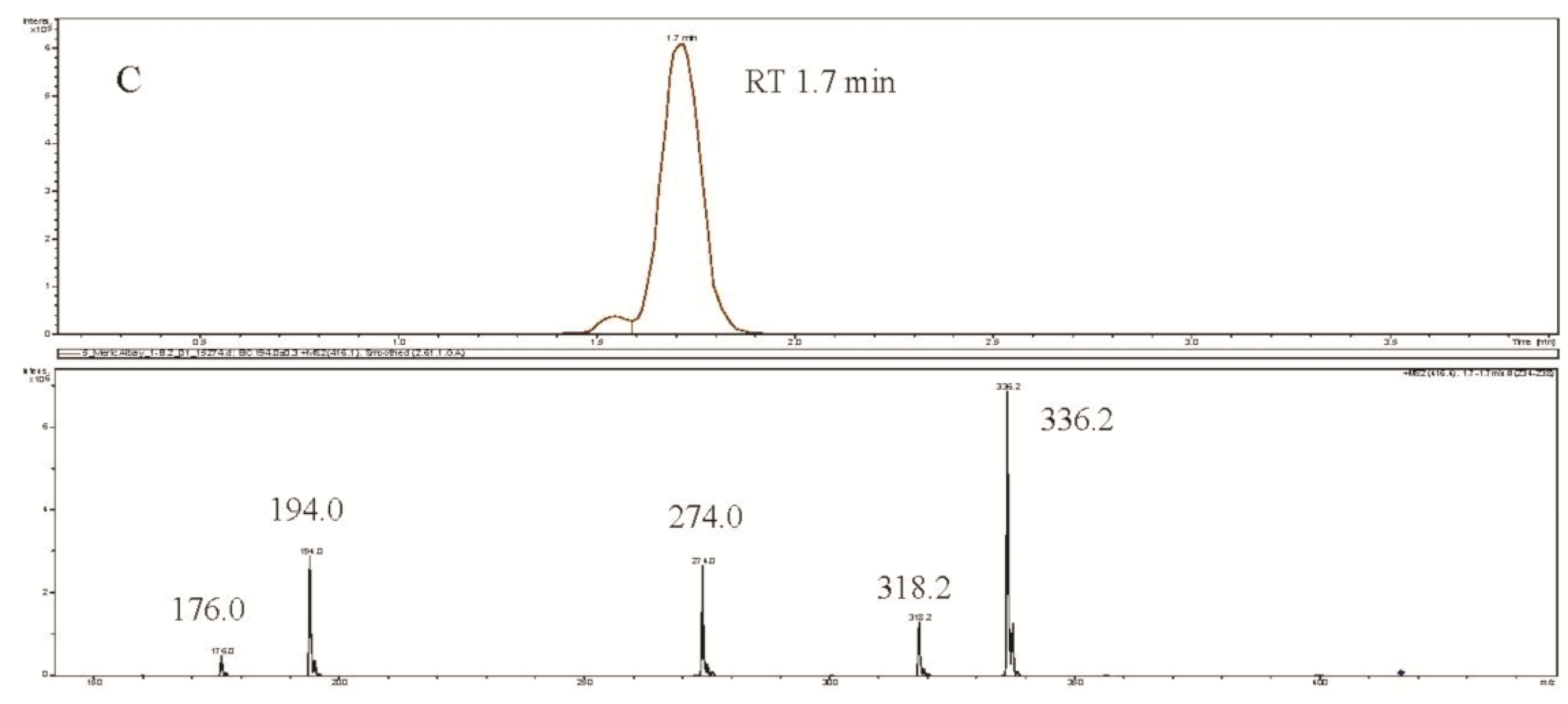
3. Experimental Section
3.1. Study Site
3.2. Sample Collection
3.3. Molecular Analysis
3.4. CYN Extraction
3.5. CYN Analysis Using the LC-MS/MS
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kinnear, S. Cylindrospermopsin: A decade of progress on bioaccumulation research. Mar. Drugs 2010, 8, 542–564. [Google Scholar] [PubMed]
- Griffiths, D.J.; Saker, M.L. The Palm Island mystery disease 20 years on: A review of research on the cyanotoxin cylindrospermopsin. Environ. Toxicol. 2003, 18, 78–93. [Google Scholar] [CrossRef] [PubMed]
- Ohtani, I.; Moore, R.E.; Runnegar, M.T. Cylindrospermopsin: A potent hepatotoxin from the blue-green alga Cylindrospermopsis raciborskii. J. Am. Chem. Soc. 1992, 114, 7941–7942. [Google Scholar] [CrossRef]
- Gutiérrez-Praena, D.; Jos, A.; Pichardo, S.; Cameán, A.M. Oxidative stress responses in tilapia (Oreochromis niloticus) exposed to a single dose of pure cylindrospermopsin under laboratory conditions: Influence of exposure route and time of sacrifice. Aquat. Toxicol. 2011, 105, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Bazin, E.; Mourot, A.; Humpage, A.R.; Fessard, V. Genotoxicity of a freshwater cyanotoxin, cylindrospermopsin, in two human cell lines: Caco-2 and HepaRG. Environ. Mol. Mutagen. 2010, 51, 251–259. [Google Scholar] [PubMed]
- Žegura, B.; Gajski, G.; Štraser, A.; Garaj-Vrhovac, V. Cylindrospermopsin induced DNA damage and alteration in the expression of genes involved in the response to DNA damage, apoptosis and oxidative stress. Toxicon 2011, 58, 471–479. [Google Scholar] [CrossRef] [PubMed]
- Messineo, V.; Melchiorre, S.; di Corcia, A.; Gallo, P.; Bruno, M. Seasonal succession of Cylindrospermopsis raciborskii and Aphanizomenon ovalisporum blooms with cylindrospermopsin occurrence in the volcanic Lake Albano, Central Italy. Environ. Toxicol. 2010, 25, 18–27. [Google Scholar] [PubMed]
- Sukenik, A.; Hadas, O.; Kaplan, A.; Quesada, A. Invasion of Nostocales (cyanobacteria) to subtropical and temperate freshwater lakes—Physiological, regional, and global driving forces. Front. Microbiol. 2012, 3. [Google Scholar] [CrossRef]
- Forti, A. Diagnoses Myxophycearum Novarum, Series IV; Atti e Memorie dell’Accademia di Agricoltura, Scienze e Lettere, Arti e Commercia di Verona: Verona, Italy, 1911; Volume 12, pp. 3–5. [Google Scholar]
- Banker, R.; Carmeli, S.; Hadas, O.; Teltsch, B.; Porat, R.; Sukenik, A. Identification of cylindrospermopsin in Aphanizomenon ovalisporum (cyanophyceae) isolated from Lake Kinneret, Israel. J. Phycol. 1997, 33, 613–616. [Google Scholar] [CrossRef]
- Gkelis, S.; Moustaka-Gouni, M.; Sivonen, K.; Lanaras, T. First report of the cyanobacterium Aphanizomenon ovalisporum Forti in two Greek lakes and cyanotoxin occurrence. J. Plankton Res. 2005, 27, 1295–1300. [Google Scholar] [CrossRef]
- Quesada, A.; Moreno, E.; Carrasco, D.; Paniagua, T.; Wormer, L.; Hoyos, C.D.; Sukenik, A. Toxicity of Aphanizomenon ovalisporum (Cyanobacteria) in a Spanish water reservoir. Eur. J. Phycol. 2006, 41, 39–45. [Google Scholar] [CrossRef]
- Shaw, G.R.; Sukenik, A.; Livne, A.; Chiswell, R.K.; Smith, M.J.; Seawright, A.A.; Norris, R.L.; Eaglesham, G.K.; Moore, M.R. Blooms of the cylindrospermopsin containing cyanobacterium, Aphanizomenon ovalisporum (Forti), in newly constructed lakes, Queensland, Australia. Environ. Toxicol. 1999, 14, 167–177. [Google Scholar] [CrossRef]
- Brient, L.; Lengronne, M.; Bormans, M.; Fastner, J. First occurrence of cylindrospermopsin in freshwater in France. Environ. Toxicol. 2009, 24, 415–420. [Google Scholar] [CrossRef] [PubMed]
- Fastner, J.; Rücker, J.; Stueken, A.; Preussel, K.; Nixdorf, B.; Chorus, I.; Koehler, A.; Wiedner, C. Occurrence of the cyanobacterial toxin cylindrospermopsin in northeast Germany. Environ. Toxicol. 2007, 22, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Wacklin, P.; Hoffmann, L.; Komárek, J. Nomenclatural validation of the genetically revised cyanobacterial genus Dolichospermum (Ralfs ex Bornet et Flahault) comb. nova. Fottea 2009, 9, 59–64. [Google Scholar]
- Zapomělová, E.; Řeháková, K.; Jezberová, J.; Komárková, J. (Polyphasic characterization of eight planktonic Anabaena strains (Cyanobacteria) with reference to the variability of 61 Anabaena populations observed in the field. Hydrobiologia 2010, 639, 99–113. [Google Scholar] [CrossRef]
- Zapomelova, E.; Rehakova, K.; Znachor, P.; Komarkova, J. Morphological diversity of coiled planktonic types of the genus Anabaena (cyanobacteria) in natural populations: Taxonomic consequences. Cryptogam. Algol. 2007, 28, 353–371. [Google Scholar]
- Komárek, J. Cyanoprokaryota: 3rd Part: Heterocytous Genera, 1st ed.; Springer-Verlag: Berlin/Heidelberg, Germany, 2013; p. 1130. [Google Scholar]
- Kopp, R.; Skácelová, O.; Heteša, J.; Marvan, P.; Bešta, T.; Zapomělová, E.; Straková, L.; Bohunıcká, M. A Hundred Years of Phycological Research in the Lednice Pond: The Impact of Environmental Conditions on the Development of Cyanobacteria and Algae; Moravian Museum: Brno, Czech Republic, 2012. [Google Scholar]
- Nõges, P.; Viirret, M. Environmental conditions and the development of Planktonema lauterbornii Schmidle in phytoplankton of Karhijärvi, a lake in SW Finland. Boreal Environ. Res. 2001, 6, 181–190. [Google Scholar]
- Padisák, J.; Hajnal, E.; Krienitz, L.; Lakner, J.; Üveges, V. Rarity, ecological memory, rate of floral change in phytoplankton—And the mystery of the Red Cock. Hydrobiologia 2010, 653, 45–64. [Google Scholar] [CrossRef]
- Affan, A.; Jewel, A.S.; Haque, M.; Khan, S.; Lee, J.B. Seasonal cycle of phytoplankton in aquaculture ponds in Bangladesh. Algae 2005, 20, 43–52. [Google Scholar] [CrossRef]
- Werner, V.R.; Laughinghouse, H.D., IV. Bloom-forming and other planktonic Anabaena (Cyanobacteria) morphospecies with twisted trichomes from Rio Grande do Sul State, Brazil. Nova Hedwig. 2009, 89, 17–47. [Google Scholar] [CrossRef]
- Täuscher, L. Checklisten und Gefährdungsgrade der Algen des Landes Brandenburg. Verh. Bot. Ver. Berl. Brandenbg. 2011, 144, 177–192. [Google Scholar]
- Paturej, E.; Gutkowska, A.; Mierzejewska, J. A review of biological research in the Vistula Lagoon. Oceanol. Hydrobiol. Stud. 2012, 41, 81–88. [Google Scholar] [CrossRef]
- Katsiapi, M.; Moustaka-Gouni, M.; Vardaka, E.; Kormas, K.A. Different phytoplankton descriptors show asynchronous changes in a shallow urban lake (L. Kastoria, Greece) after sewage diversion. Fundam. Appl. Limnol. Arch. Hydrobiol. 2013, 182, 219–230. [Google Scholar] [CrossRef]
- Rantala, J.; Sivonen, K.; Luukkainen, R.; Niemelä, S.I. Anatoxin-a concentration in Anabaena and Aphanizomenon under different environmental conditions and comparison of growth by toxic and non-toxic Anabaena-strains—A laboratory study. J. Appl. Phycol. 1993, 5, 581–591. [Google Scholar] [CrossRef]
- Li, R.; Watanabe, M.; Watanabe, M.M. Taxonomic studies of planktic species of Anabaena based on morphological characteristics in cultured strains. Hydrobiologia 2000, 438, 117–138. [Google Scholar] [CrossRef]
- Gugger, M.; Lyra, C.; Henriksen, P.; Couté, A.; Humbert, J.F.; Sivonen, K. Phylogenetic comparison of the cyanobacterial genera Anabaena and Aphanizomenon. Int. J. Syst. Evol. Microbiol. 2002, 52, 1867–1880. [Google Scholar] [CrossRef] [PubMed]
- Cirés, S.; Wörmer, L.; Ballot, A.; Agha, R.; Wiedner, C.; Velázquez, D.; Casero, M.C.; Quesada, A. Phylogeography of cylindrospermopsin and paralytic shellfish toxin-producing Nostocales cyanobacteria from Mediterranean Europe (Spain). Appl. Environ. Microbiol. 2014, 80, 1359–1370. [Google Scholar] [CrossRef] [PubMed]
- Yılmaz, M.; Phlips, E.J.; Szabo, N.J.; Badylak, S. A comparative study of Florida strains of Cylindrospermopsis and Aphanizomenon for cylindrospermopsin production. Toxicon 2008, 51, 130–139. [Google Scholar] [CrossRef] [PubMed]
- Hadas, O.; Pinkas, R.; Malinsky-Rushansky, N.; Shalev-Alon, G.; Delphine, E.; Berner, T.; Sukenik, A.; Kaplan, A. Physiological variables determined under laboratory conditions may explain the bloom of Aphanizomenon ovalisporum in Lake Kinneret. Eur. J. Phycol. 2012, 37, 259–267. [Google Scholar] [CrossRef]
- Fergusson, K.M.; Saint, C.P. Multiplex PCR assay for Cylindrospermopsis raciborskii and cylindrospermopsin-producing cyanobacteria. Environ. Toxicol. 2003, 18, 120–125. [Google Scholar] [CrossRef] [PubMed]
- Albay, M.; Aykulu, G. Invertebrate grazer-epiphytic algae interactions on submerged macrophytes in a mesotrophic Turkish lake. TrJFAS 2002, 19, 247–258. [Google Scholar]
- Utermöhl, H. Zur vervollkommnung der quantitativen phytoplankton-methodik. Mitt. Int. Ver. Theor. Angew. Limnol. 1958, 9, 1–38. [Google Scholar]
- Hillebrand, H.; Durselen, C.D.; Kirschtel, D.; Pollinger, U.; Zohary, T. Biovolume calculation for pelagic and benthic microalgae. J. Phycol. 1999, 35, 403–424. [Google Scholar] [CrossRef]
- Nusch, E. Comparison of different methods for chlorophyll and phaeopigment determination. Arch. Hydrobiol. Beih. 1980, 14, 14–36. [Google Scholar]
- American Public Health Association (APHA); American Water Works Association (AWWA); Water Environment Federation (WEF). Standart Methods for the Examination of Water and Wastewater, 17th ed.; American Public Health Association: Washington, DC, USA, 1989. [Google Scholar]
- Tillett, D.; Neilan, B.A. Xanthogenate nucleic acid isolation from cultured and environmental cyanobacteria. J. Phycol. 2000, 36, 251–258. [Google Scholar] [CrossRef]
- Saker, M.L.; Jungblut, A.D.; Neilan, B.; Rawn, D.F.K.; Vasconcelos, V.M. Detection of microcystin synthetase genes in health food supplements containing the freshwater cyanobacterium Aphanizomenon flos-aquae. Toxicon 2005, 46, 555–562. [Google Scholar] [CrossRef] [PubMed]
- Chorus, I. Current Approaches to Cyanotoxin Risk Assessment, Risk Management and Regulations in Different Countries; Federal Environment Agency (Umweltbundesamt): Dessau-Rosslau, Germany, 2012. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Akcaalan, R.; Köker, L.; Oğuz, A.; Spoof, L.; Meriluoto, J.; Albay, M. First Report of Cylindrospermopsin Production by Two Cyanobacteria (Dolichospermum mendotae and Chrysosporum ovalisporum) in Lake Iznik, Turkey. Toxins 2014, 6, 3173-3186. https://doi.org/10.3390/toxins6113173
Akcaalan R, Köker L, Oğuz A, Spoof L, Meriluoto J, Albay M. First Report of Cylindrospermopsin Production by Two Cyanobacteria (Dolichospermum mendotae and Chrysosporum ovalisporum) in Lake Iznik, Turkey. Toxins. 2014; 6(11):3173-3186. https://doi.org/10.3390/toxins6113173
Chicago/Turabian StyleAkcaalan, Reyhan, Latife Köker, Ayça Oğuz, Lisa Spoof, Jussi Meriluoto, and Meriç Albay. 2014. "First Report of Cylindrospermopsin Production by Two Cyanobacteria (Dolichospermum mendotae and Chrysosporum ovalisporum) in Lake Iznik, Turkey" Toxins 6, no. 11: 3173-3186. https://doi.org/10.3390/toxins6113173
APA StyleAkcaalan, R., Köker, L., Oğuz, A., Spoof, L., Meriluoto, J., & Albay, M. (2014). First Report of Cylindrospermopsin Production by Two Cyanobacteria (Dolichospermum mendotae and Chrysosporum ovalisporum) in Lake Iznik, Turkey. Toxins, 6(11), 3173-3186. https://doi.org/10.3390/toxins6113173






