Non-Linear Relationships between Aflatoxin B1 Levels and the Biological Response of Monkey Kidney Vero Cells
Abstract
:1. Introduction
- The incidence of hepatocellular carcinoma (HCC) is significantly elevated in the Hispanic community in Bexar County, Texas [27]. The AFB1-lysine adduct level detected in 20.6% of evaluated individual serums ranged from 1.01 to 16.57 pg/mg. Aflatoxin M1 (AFM1), a metabolite of AFB1, detected in 11.7% of the urine samples ranged from 1.89 to 935.5 pg/mg creatinine. AFM1 levels were associated with increased consumption of corn tortillas, nuts and rice.
- On the basis of a review of the literature, Matsuda et al. [28] suggest that mycotoxins represent risk biomarkers for nonviral hepatocellular carcinoma, a common malignant disease with poor prognosis.
- Ghasemi-Kebria et al. [29] found a positive relationship between the aflatoxin content of 100 wheat flour samples in areas of Iran and a high risk of esophageal cancer.
- Hamid et al. [30] report that about 4.5 billion of the world’s population is exposed to aflatoxin-contaminated food, especially in low-income countries, and that dietary exposure to aflatoxins is a major HCC risk factor.
- Sun et al. [31] reported that on the basis of the measured content of AFB1 and fumonisin B1 of 209 food samples in three areas of China, co-exposure to the two mycotoxins in rural China seems to contribute to the etiology of human chronic diseases in high-risk areas.
- Asim et al. [32] found that, in comparison with high-aflatoxin-exposure countries, such as China and Taiwan, the aflatoxin level in India, as well as the hepatocellular carcinoma rate is relatively low and that high hepatitis B virus (HBV)-DNA serum levels increased the risk of liver cancer.
- In another study, Asai et al. [33] reported that red chili peppers from Bolivia and Peru contaminated with aflatoxins at concentrations above maximum levels in spices proposed by the European Commission are consumed by populations that have a high incidence of gallbladder cancer.
- Dairy animal feed contamination by AFB1 near the European Union tolerance of 5 μg/kg results in a concentration of AFM1 in milk higher than the European Commission maximum tolerance level, suggesting that AFM1 in milk may originate from dairy cows [34].
- On the basis of a study that showed that AFB1 in cows is carried over to AFM1 in milk, the authors suggest that the maximum AFB1 level in feed should not exceed 1.4 μg/kg, a value 3.6-times lower than the maximum residue level currently applied in Israel [35].
- A new stable isotope dilution assay was used to detect the AFMI content of condensed milk, milk-based infant formula and table cream imported into the United States [36].
- An outbreak of aflatoxin poisoning in 65 dogs was associated with two corn meals containing 1640 ppb and 1770 ppb of AFB1, respectively [37].
- Duck mortality increased with increasing concentrations of AFB1 in the diets [38].
- AFB1 produced dose-related DNA damage in fetal livers of both chicken and turkey ova, with turkey embryos showing slightly more susceptibility to AFB1 damage than chicken embryos [39].
- Hepatic cytochrome P450 1A5 is the dominant enzyme for AFB1 bioactivation and metabolism of environmentally-relevant AFB1 concentrations in turkey liver [5].
- Bacillus subtilis bacteria ameliorated damage of liver and kidney tissue in laying hens exposed to AFB1 [40].
2. Results
2.1. Low Doses of AFB1 Stimulate Cell Growth and Activity
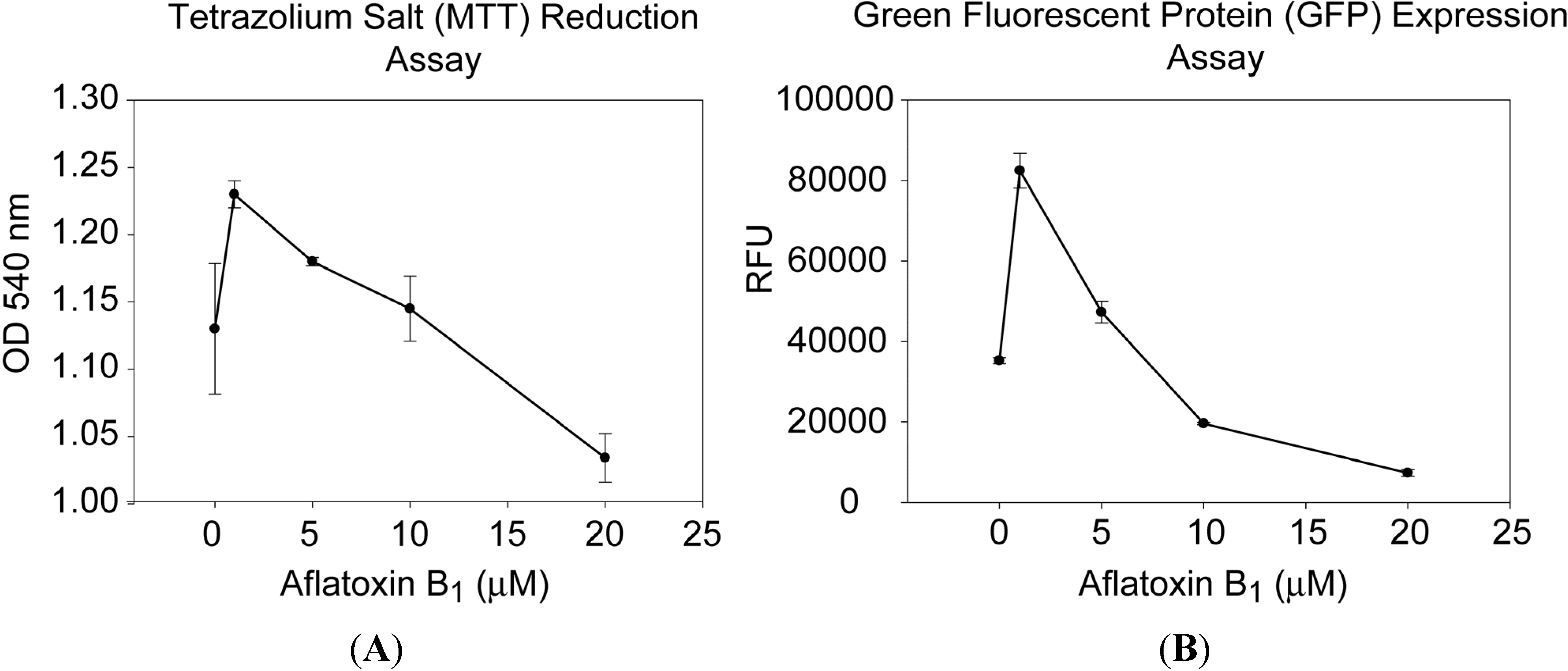
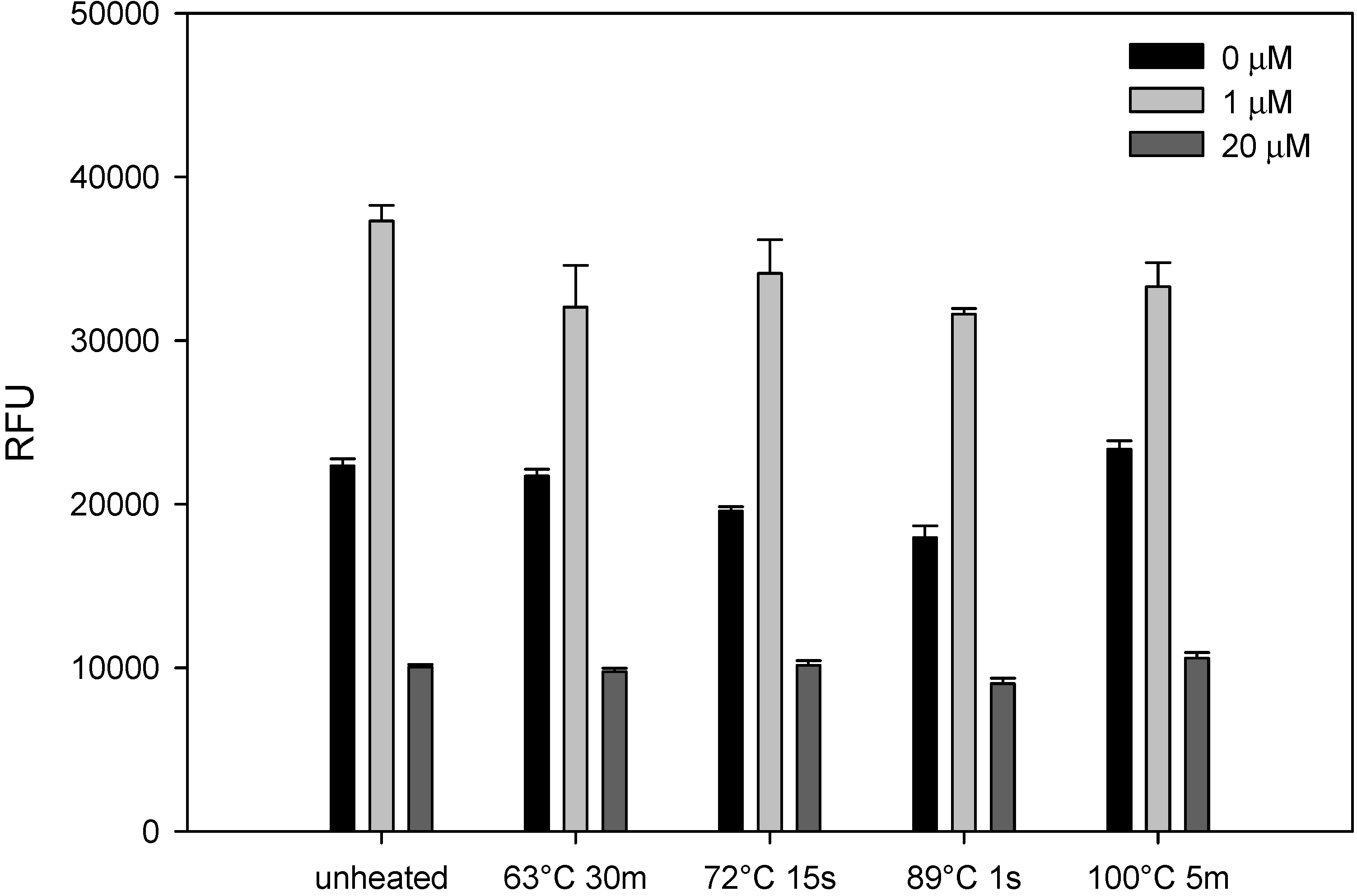
2.2. Heat Treatment Did Not Affect AFB1 Stability in Milk
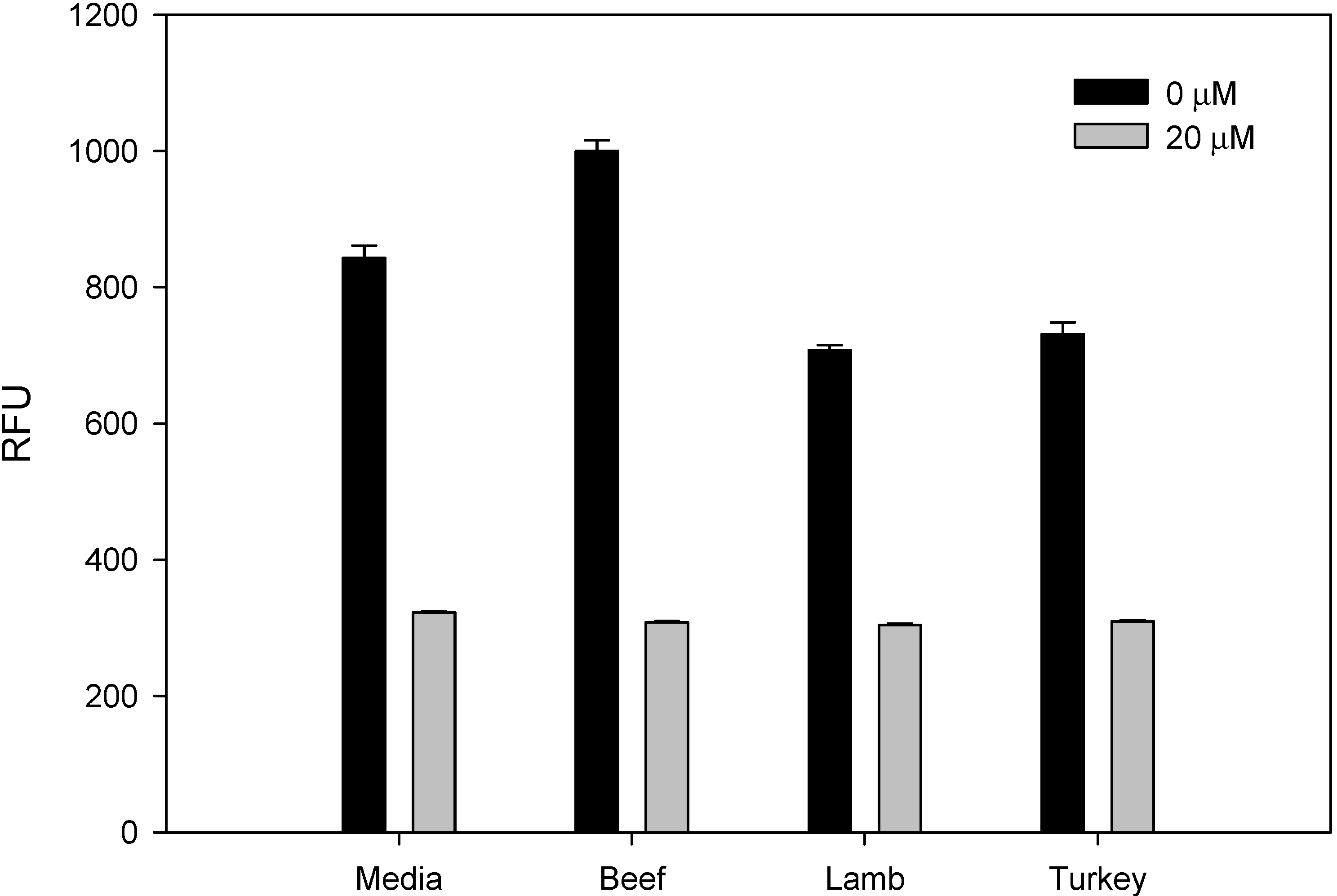
2.3. Detection of AFB1 in Beef, Lamb and Turkey Meat
3. Discussion
- On the basis of a review of more than 500 National Toxicology Program Technical Reports, Waddell [50] concluded that the mere presence of DNA adducts of AFB1 does not necessarily lead to tumor formation, that all of the carcinogenicity studies show a linear response when the dose is on a logarithmic scale, that the concept of hormesis, defined as a dose-response effect characterized by low-dose stimulation and high-dose inhibition [51], probably applies to carcinogenesis and that a cumulative dose may be a better indicator than a daily dose.
- On the basis of simulation studies, Lutz et al. [52] concluded that non-linear dose-response curves are often observed in tests of carcinogenicity in rodents and that linear extrapolation of a human risk could be justified, even if animal bioassays show non-linearity.
- During a long-term study, Jossé et al. [49] found that AFB1 induced a dose-dependent cumulative cytotoxicity in human HepRG hepatocytes, suggesting that these cells represent an in vitro liver cell model for measuring acute and chronic toxicity and genotoxicity of AFB1 in human livers.
- Williams et al. [53,54] observed nonlinearities and thresholds in rat liver carcinogenesis caused by 2-acetylamianaofluorene and diethyl nitrosamine. The authors interpret the results to reflect thresholds for the initiation of liver cancer by these carcinogens and an exaggerated response at high exposures, due to toxicity and compensatory hepatocyte proliferation. The possibility, therefore, exists of defining safe low exposure levels (SEL) to carcinogens.
- Using the MTT assay, Ruiz et al. [55] investigated the cytotoxicity in Vero cells induced by individual and combinations of three Fusarium toxins (beauvericin, deoxynivalenol and T-2 toxin). All combinations exhibited antagonistic effects. The highest antagonistic results were obtained with the binary mixture of deoxynivalenol and the T-2 toxin, suggesting the need to evaluate the potential toxicities of combinations of mycotoxins that may be present in some food. By contrast, Bouaziz et al. [56] found that mixtures of the fusarial toxins, zearalenone and T-2, induced higher cytotoxicity in Vero cells, suggesting that each combination of mycotoxins needs to be evaluated for toxicity.
- Using membrane-engineered Vero cells, Larou et al. [57] developed a rapid 3-min biosensor assay for AFM1, suggesting the need to determine the applicability of the assay to mycotoxin-containing food.
4. Materials and Methods
4.1. Materials
4.2. Effect of Heat on AFB1 Bioactivity in Milk
4.3. Cell Culture
4.4. Plaque Assays for the Purification and Titration of the Adenovirus
4.5. Quantifying Recombinant Adenovirus Expressing Vectors that Encode the GFP Gene
4.6. MTT Assay for the Metabolic Activity and Viability of Cells
4.7. Statistical Analysis
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Magnussen, A.; Parsi, M.A. Aflatoxins, hepatocellular carcinoma and public health. World J. Gastroenterol. 2013, 19, 1508–1512. [Google Scholar] [CrossRef]
- Lewis, L.; Onsongo, M.; Njapau, H.; Schurz-Rogers, H.; Luber, G.; Kieszak, S.; Nyamongo, J.; Backer, L.; Dahiye, A.M.; Misore, A.; et al. Aflatoxin contamination of commercial maize products during an outbreak of acute aflatoxicosis in eastern and central Kenya. Environ. Health Perspect. 2005, 113, 1763–1767. [Google Scholar] [CrossRef]
- Krishnamachari, K.A.V.R.; Bhat, R.V.; Nagarajan, V.; Tilak, T.B.G. Hepatitis due to aflatoxicosis. An outbreak in western India. Lancet 1975, 1, 1061–1063. [Google Scholar]
- Cousin, M.A.; Riley, R.T.; Pestka, J.J. Foodborne Mycotoxins: Chemistry, Biology, Ecology, and Toxicology. In Foodborne Pathogens-Microbiology and Molecular Biology; Fratamico, P.M., Bhunia, A.K., Smith, J.L., Eds.; Caister Academic Press: Norfolk, UK, 2005; pp. 163–226. [Google Scholar]
- Rawal, S.; Coulombe, R.A. Metabolism of aflatoxin B1 in turkey liver microsomes: The relative roles of cytochromes P450 1A5 and 3A37. Toxicol. Appl. Pharmacol. 2011, 254, 349–354. [Google Scholar] [CrossRef]
- Friedman, M.; Rasooly, R. Review of the inhibition of biological activities of food-related selected toxins by natural compounds. Toxins 2013, 5, 743–775. [Google Scholar] [CrossRef]
- Liao, C.D.; Wong, J.W.; Zhang, K.; Hayward, D.G.; Lee, N.S.; Trucksess, M.W. Multi-mycotoxin analysis of finished grain and nut products using high-performance liquid chromatography-triple-quadrupole mass spectrometry. J. Agric. Food Chem. 2013, 61, 4771–4782. [Google Scholar] [CrossRef]
- Yu, F.Y.; Gribas, A.V.; Vdovenko, M.M.; Sakharov, I.Y. Development of ultrasensitive direct chemiluminescent enzyme immunoassay for determination of aflatoxin B1 in food products. Talanta 2013, 107, 25–29. [Google Scholar] [CrossRef]
- Luna, A.S.; Luiz, R.A.; Lima, I.C.A.; Março, P.H.; Valderrama, P.; Boqué, R.; Ferré, J. Simultaneous determination of aflatoxins B2 and G2 in peanuts using spectrofluorescence coupled with parallel factor analysis. Anal. Chim. Acta 2013, 778, 9–14. [Google Scholar] [CrossRef]
- Li, X.; Li, P.; Zhang, Q.; Li, R.; Zhang, W.; Zhang, Z.; Ding, X.; Tang, X. Multi-component immunochromatographic assay for simultaneous detection of aflatoxin B1, ochratoxin A and zearalenone in agro-food. Biosens. Bioelectron. 2013, 49, 426–432. [Google Scholar] [CrossRef]
- Shimshoni, J.A.; Cuneah, O.; Sulyok, M.; Krska, R.; Galon, N.; Sharir, B.; Shlosberg, A. Mycotoxins in corn and wheat silage in Israel. Food Addit. Contam. Part A 2013. [Google Scholar] [CrossRef]
- Kana, J.R.; Gnonlonfin, B.G.J.; Harvey, J.; Wainaina, J.; Wanjuki, I.; Skilton, R.A.; Teguia, A. Assessment of aflatoxin contamination of maize, peanut meal and poultry feed mixtures from different agroecological zones in Cameroon. Toxins 2013, 5, 884–894. [Google Scholar] [CrossRef]
- Spanjer, M.C.; Rensen, P.M.; Scholten, J.M. LC-MS/MS multi-method for mycotoxins after single extraction, with validation data for peanut, pistachio, wheat, maize, cornflakes, raisins and figs. Food Addit. Contam. Part A 2008, 25, 472–489. [Google Scholar] [CrossRef]
- Tosun, H.; Arslan, R. Determination of aflatoxin B1 levels in organic spices and herbs. Sci. World J. 2013, 2013. [Google Scholar] [CrossRef]
- Guo, Y.; Yuan, Y.; Yue, T. Aflatoxin M1 in milk products in China and dietary risk assessment. J. Food Prot. 2013, 76, 849–853. [Google Scholar] [CrossRef]
- Tchana, A.N.; Moundipa, P.F.; Tchouanguep, F.M. Aflatoxin contamination in food and body fluids in relation to malnutrition and cancer status in Cameroon. Int. J. Environ. Res. Public Health 2010, 7, 178–188. [Google Scholar] [CrossRef]
- Herzallah, S.M. Determination of aflatoxins in eggs, milk, meat and meat products using HPLC fluorescent and UV detectors. Food Chem. 2009, 114, 1141–1146. [Google Scholar] [CrossRef]
- Chen, D.; Cao, X.; Tao, Y.; Wu, Q.; Pan, Y.; Huang, L.; Wang, X.; Wang, Y.; Peng, D.; Liu, Z.; et al. Development of a sensitive and robust liquid chromatography coupled with tandem mass spectrometry and a pressurized liquid extraction for the determination of aflatoxins and ochratoxin A in animal derived foods. J. Chromatogr. A 2012, 1253, 110–119. [Google Scholar] [CrossRef]
- Yard, E.E.; Daniel, J.H.; Lewis, L.S.; Rybak, M.E.; Paliakov, E.M.; Kim, A.A.; Montgomery, J.M.; Bunnell, R.; Abudo, M.U.; Akhwale, W.; et al. Human aflatoxin exposure in Kenya, 2007: A cross-sectional study. Food Addit. Contam. Part A 2013, 30, 1322–1331. [Google Scholar] [CrossRef]
- Almeida, M.I.; Almeida, N.G.; Carvalho, K.L.; Gonçalves, G.A.; Silva, C.N.; Santos, E.A.; Garcia, J.C.; Vargas, E.A. Co-occurrence of aflatoxins B1, B2, G1 and G2, ochratoxin A, zearalenone, deoxynivalenol, and citreoviridin in rice in Brazil. Food Addit. Contam. Part A 2012, 29, 694–703. [Google Scholar] [CrossRef]
- Jager, A.V.; Tedesco, M.P.; Souto, P.C.M.C.; Oliveira, C.A.F. Assessment of aflatoxin intake in São Paulo, Brazil. Food Control 2013, 33, 87–92. [Google Scholar] [CrossRef]
- Bansal, J.; Pantazopoulos, P.; Tam, J.; Cavlovic, P.; Kwong, K.; Turcotte, A.M.; Lau, B.P.Y.; Scott, P.M. Surveys of rice sold in Canada for aflatoxins, ochratoxin a and fumonisins. Food Addit. Contam. Part A 2011, 28, 767–774. [Google Scholar] [CrossRef]
- Reddy, K.R.; Farhana, N.I.; Salleh, B. Occurrence of Aspergillus spp. and aflatoxin B1 in Malaysian foods used for human consumption. J. Food Sci. 2011, 76, T99–T104. [Google Scholar] [CrossRef]
- Toteja, G.S.; Mukherjee, A.; Diwakar, S.; Singh, P.; Saxena, B.N.; Sinha, K.K.; Sinha, A.K.; Kumar, N.; Nagaraja, K.V.; Bai, G.; et al. Aflatoxin B1 contamination in wheat grain samples collected from different geographical regions of India: A multicenter study. J. Food Prot. 2006, 69, 1463–1467. [Google Scholar]
- Chen, S.; Zhang, H. Development of a microwave-assisted-extraction-based method for the determination of aflatoxins B1, G1, B2, and G2 in grains and grain products. Anal. Bioanal. Chem. 2013, 405, 1623–1630. [Google Scholar] [CrossRef]
- Rasooly, R.; Hernlem, B.; Friedman, M. Low levels of aflatoxin B1, ricin, and milk enhance recombinant protein production in mammalian cells. PLoS One 2013, 8, e71682. [Google Scholar] [CrossRef]
- Johnson, N.M.; Qian, G.; Xu, L.; Tietze, D.; Marroquin-Cardona, A.; Robinson, A.; Rodriguez, M.; Kaufman, L.; Cunningham, K.; Wittmer, J.; et al. Aflatoxin and PAH exposure biomarkers in a U.S. population with a high incidence of hepatocellular carcinoma. Sci. Total Environ. 2010, 408, 6027–6031. [Google Scholar] [CrossRef]
- Matsuda, Y.; Wakai, T.; Kubota, M.; Osawa, M.; Sanpei, A.; Fujimaki, S. Mycotoxins are conventional and novel risk biomarkers for hepatocellular carcinoma. World J. Gastroenterol. 2013, 19, 2587–2590. [Google Scholar] [CrossRef]
- Ghasemi-Kebria, F.; Joshaghani, H.; Taheri, N.S.; Semnani, S.; Aarabi, M.; Salamat, F.; Roshandel, G. Aflatoxin contamination of wheat flour and the risk of esophageal cancer in a high risk area in Iran. Cancer Epidemiol. 2013, 37, 290–293. [Google Scholar] [CrossRef]
- Hamid, A.S.; Tesfamariam, S.G.; Zhang, Y.; Zhang, Z.G. Aflatoxin B1-induced hepatocellular carcinoma in developing countries: Geographical distribution, mechanism of action and prevention (Review). Oncol. Lett. 2013, 5, 1087–1092. [Google Scholar]
- Sun, G.; Wang, S.; Hu, X.; Su, J.; Zhang, Y.; Xie, Y.; Zhang, H.; Tang, L.; Wang, J.S. Co-contamination of aflatoxin B1 and fumonisin B1 in food and human dietary exposure in three areas of China. Food Addit. Contam. Part A 2011, 28, 461–470. [Google Scholar] [CrossRef]
- Asim, M.; Sarma, M.P.; Kar, P. Etiological and molecular profile of hepatocellular cancer from India. Int. J. Cancer 2013, 133, 437–445. [Google Scholar] [CrossRef]
- Asai, T.; Tsuchiya, Y.; Okano, K.; Piscoya, A.; Nishi, C.Y.; Ikoma, T.; Oyama, T.; Ikegami, K.; Yamamoto, M. Aflatoxin contamination of red chili pepper from Bolivia and Peru, countries with high gallbladder cancer incidence rates. Asian Pac. J. Cancer Prev. 2012, 13, 5167–5170. [Google Scholar] [CrossRef]
- Battacone, G.; Nudda, A.; Palomba, M.; Mazzette, A.; Pulina, G. The transfer of aflatoxin M1 in milk of ewes fed diet naturally contaminated by aflatoxins and effect of inclusion of dried yeast culture in the diet. J. Dairy Sci. 2009, 92, 4997–5004. [Google Scholar] [CrossRef]
- Britzi, M.; Friedman, S.; Miron, J.; Solomon, R.; Cuneah, O.; Shimshoni, J.A.; Soback, S.; Ashkenazi, R.; Armer, S.; Shlosberg, A. Carry-over of aflatoxin B1 to aflatoxin M1 in high yielding Israeli cows in mid- and late-lactation. Toxins 2013, 5, 173–183. [Google Scholar] [CrossRef]
- Zhang, K.; Wong, J.W.; Hayward, D.G.; Vaclavikova, M.; Liao, C.D.; Trucksess, M.W. Determination of mycotoxins in milk-based products and infant formula using stable isotope dilution assay and liquid chromatography tandem mass spectrometry. J. Agric. Food Chem. 2013, 61, 6265–6273. [Google Scholar] [CrossRef]
- Wouters, A.T.B.; Casagrande, R.A.; Wouters, F.; Watanabe, T.T.N.; Boabaid, F.M.; Cruz, C.E.F.; Driemeier, D. An outbreak of aflatoxin poisoning in dogs associated with aflatoxin B1-contaminated maize products. J. Vet. Diagn. Invest. 2013, 25, 282–287. [Google Scholar] [CrossRef]
- Wan, X.L.; Yang, Z.B.; Yang, W.R.; Jiang, S.Z.; Zhang, G.G.; Johnston, S.L.; Chi, F. Toxicity of increasing aflatoxin B1 concentrations from contaminated corn with or without clay adsorbent supplementation in ducklings. Poult. Sci. 2013, 92, 1244–1253. [Google Scholar] [CrossRef]
- Williams, J.G.; Deschl, U.; Williams, G.M. DNA damage in fetal liver cells of turkey and chicken eggs dosed with aflatoxin B1. Arch. Toxicol. 2011, 85, 1167–1172. [Google Scholar] [CrossRef]
- Ma, Q.G.; Gao, X.; Zhou, T.; Zhao, L.H.; Fan, Y.; Li, X.Y.; Lei, Y.P.; Ji, C.; Zhang, J.Y. Protective effect of Bacillus subtilis ANSB060 on egg quality, biochemical and histopathological changes in layers exposed to aflatoxin B1. Poult. Sci. 2012, 91, 2852–2857. [Google Scholar] [CrossRef]
- Robens, J.F.; Richard, J.L. Aflatoxins in animal and human health. Rev. Environ. Contam. Toxicol. 1992, 127, 69–94. [Google Scholar] [CrossRef]
- Wu, F.; Miller, J.D.; Casman, E.A. The economic impact of Bt corn resulting from mycotoxin reduction. J. Toxicol. Toxin Rev. 2004, 23, 397–424. [Google Scholar] [CrossRef]
- FDA. Kansas Aflatoxin Relief Response; Letter to Kansas Department of Agriculture, Oct 5, 2012. U.S. Food and Drug Administration (FDA)—Kansas Department of Agriculture: Document Center. Available online: http://www.ksda.gov/includes/document_center/mainportal/Policy/Kansas%20Aflatoxin%20Relief%20Reponse%202012%20.pdf (accessed on 22 July 2013).
- FAO and IFIF. Health hazards associated with animal feed (Section I). FAO Animal Production and Health Manual No. 9. Good Practices for the Feed Industry-Implementing the Codex Alimentarius Code of Practice on Good Animal Feeding; Rome, 2010. Available online: http://www.fao.org/docrep/012/i1379e/i1379e01.pdf (accessed on 22 July 2013).
- Berridge, M.V.; Herst, P.M.; Tan, A.S. Tetrazolium dyes as tools in cell biology: New insights into their cellular reduction. Biotechnol. Annu. Rev. 2005, 11, 127–152. [Google Scholar] [CrossRef]
- El Golli-Bennour, E.; Kouidhi, B.; Bouslimi, A.; Abid-Essefi, S.; Hassen, W.; Bacha, H. Cytotoxicity and genotoxicity induced by aflatoxin B1, ochratoxin A, and their combination in cultured Vero cells. J. Biochem. Mol. Toxicol. 2010, 24, 42–50. [Google Scholar] [CrossRef]
- Gawade, S.P. Photodynamic studies on aflatoxin B1 using UV radiation in the presence of methylene blue. Indian J. Pharm. Educ. Res. 2010, 44, 142–147. [Google Scholar]
- Peña, S.D.; Durán-de-Bazúa, M.C. Efecto toxico de las aflatoxinas en la dieta. Cienc. Desarro. 1990, 16, 61–70. [Google Scholar]
- Jossé, R.; Aninat, C.; Glaise, D.; Dumont, J.; Fessard, V.; Morel, F.; Poul, J.-M.; Guguen-Guillouzo, C.; Guillouzo, A. Long-term functional stability of human HepaRG hepatocytes and use for chronic toxicity and genotoxicity studies. Drug Metab. Dispos. 2008, 36, 1111–1118. [Google Scholar] [CrossRef]
- Waddell, W.J. Critique of dose response in carcinogenesis. Hum. Exp. Toxicol. 2006, 25, 413–436. [Google Scholar] [CrossRef]
- Díaz, G.J.; Calabrese, E.; Blain, R. Aflatoxicosis in chickens (Gallus gallus): An example of hormesis? Poult. Sci. 2008, 87, 727–732. [Google Scholar] [CrossRef]
- Lutz, W.K.; Gaylor, D.W.; Conolly, R.B.; Lutz, R.W. Nonlinearity and thresholds in dose-response relationships for carcinogenicity due to sampling variation, logarithmic dose scaling, or small differences in individual susceptibility. Toxicol. Appl. Pharmacol. 2005, 207, S565–S569. [Google Scholar] [CrossRef]
- Williams, G.M.; Iatropoulos, M.J.; Jeffrey, A.M.; Luo, F.Q.; Wang, C.X.; Pittman, B. Diethylnitrosamine exposure-responses for DNA ethylation, hepatocellular proliferation, and initiation of carcinogenesis in rat liver display non-linearities and thresholds. Arch. Toxicol. 1999, 73, 394–402. [Google Scholar] [CrossRef]
- Williams, G.M.; Iatropoulos, M.J.; Jeffrey, A.M. Mechanistic basis for nonlinearities and thresholds in rat liver carcinogenesis by the DNA-reactive carcinogens 2-acetylaminofluorene and diethylnitrosamine. Toxicol. Pathol. 2000, 28, 388–395. [Google Scholar] [CrossRef]
- Ruiz, M.-J.; Macáková, P.; Juan-García, A.; Font, G. Cytotoxic effects of mycotoxin combinations in mammalian kidney cells. Food Chem. Toxicol. 2011, 49, 2718–2724. [Google Scholar] [CrossRef]
- Bouaziz, C.; Bouslimi, A.; Kadri, R.; Zaied, C.; Bacha, H.; Abid-Essefi, S. The in vitro effects of zearalenone and T-2 toxins on Vero cells. Exp. Toxicol. Pathol. 2013, 65, 497–501. [Google Scholar] [CrossRef]
- Larou, E.; Yiakoumettis, I.; Kaltsas, G.; Petropoulos, A.; Skandamis, P.; Kintzios, S. High throughput cellular biosensor for the ultra-sensitive, ultra-rapid detection of aflatoxin M1. Food Control 2013, 29, 208–212. [Google Scholar] [CrossRef]
- Friedman, M.; Henika, P.R.; Levin, C.E. Bactericidal activities of health-promoting, food-derived powders against the foodborne pathogens Escherichia coli, Listeria monocytogenes, Salmonella enterica, and Staphylococcus aureus. J. Food Sci. 2013, 78, M270–M275. [Google Scholar] [CrossRef]
- Friedman, M.; Rasooly, R.; Do, P.M.; Henika, P.R. The olive compound hydroxytyrosol inactivates Staphylococcus aureus bacteria and Staphylococcal enterotoxin A (SEA). J. Food Sci. 2011, 76, M558–M563. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Rasooly, R.; Hernlem, B.; He, X.; Friedman, M. Non-Linear Relationships between Aflatoxin B1 Levels and the Biological Response of Monkey Kidney Vero Cells. Toxins 2013, 5, 1447-1461. https://doi.org/10.3390/toxins5081447
Rasooly R, Hernlem B, He X, Friedman M. Non-Linear Relationships between Aflatoxin B1 Levels and the Biological Response of Monkey Kidney Vero Cells. Toxins. 2013; 5(8):1447-1461. https://doi.org/10.3390/toxins5081447
Chicago/Turabian StyleRasooly, Reuven, Bradley Hernlem, Xiaohua He, and Mendel Friedman. 2013. "Non-Linear Relationships between Aflatoxin B1 Levels and the Biological Response of Monkey Kidney Vero Cells" Toxins 5, no. 8: 1447-1461. https://doi.org/10.3390/toxins5081447
APA StyleRasooly, R., Hernlem, B., He, X., & Friedman, M. (2013). Non-Linear Relationships between Aflatoxin B1 Levels and the Biological Response of Monkey Kidney Vero Cells. Toxins, 5(8), 1447-1461. https://doi.org/10.3390/toxins5081447





