Toxins for Transgenic Resistance to Hemipteran Pests
Abstract
1. Hemiptera of Agricultural Importance
1.1. Hemiptera as Primary Pests
1.2. Specialization for Ingestion of Plant Sap
1.3. Management of Hemipteran Pests
2. Insecticidal Toxins Derived from Bacillus thuringiensis
2.1. Hemipteran-Active Bt Toxins
| Toxin | Toxicity | Specificity | Reference |
|---|---|---|---|
| Cry2, Cry3A, Cry4 | Some | Potato aphid, Macrosiphum euphorbiae | [48] |
| Cry4Aa Cry11Aa Cry3A, | LC50: 70-100 μg/mL 100% mortality at 500 μg/mL 60% mortality at 500 μg/mL | Pea aphid, Acyrthosiphon pisum | [50] |
| Vip1Ae-Vip2Ae | LC50: 0.576 μg/mL | Cotton aphid, Aphis gossypii | [53] |
| Patent | Toxin | Specificity | Investigators |
|---|---|---|---|
| US 2009/0,068,159 | TIC809, ET37, TIC810, TIC 812 | Lygus bugs and coleopteran pests | [54] |
| US 2010/0,064,394 | TIC853 | Lygus bugs | [55] |
| US 1993/5,262,159 | Bt isolates | Aphids | [47] |
2.2. Bases for Low Toxicity of Bt Toxins against Sap-Sucking Insects
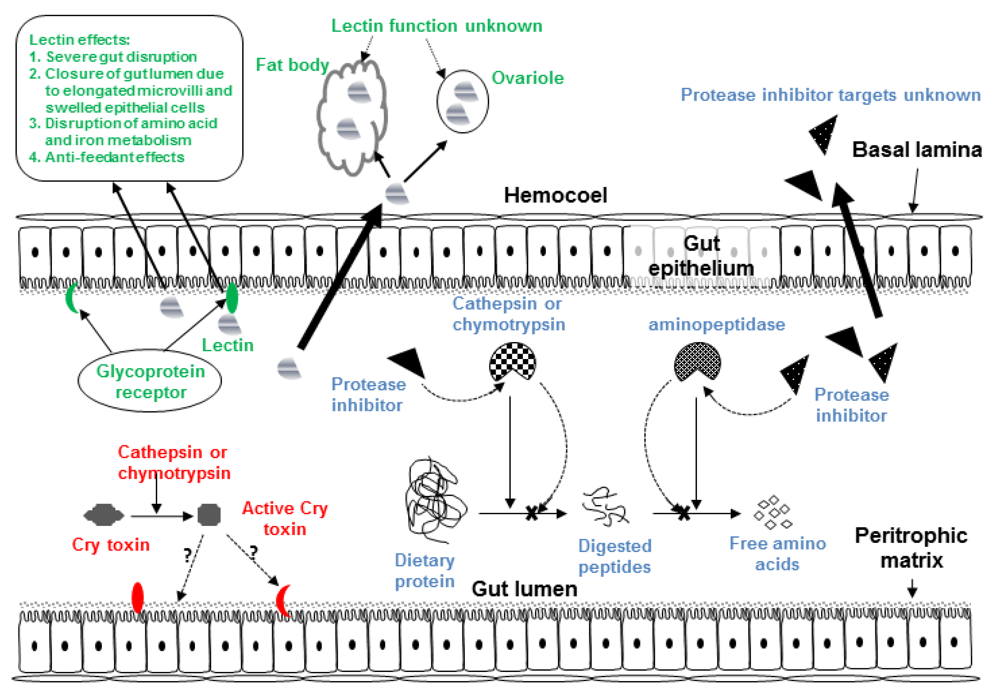
2.3. Cry Toxin Modification for Enhanced Hemipteran Toxicity
3. Plant Defense Proteins: Lectins
3.1. Impact of Lectins on Herbivorous Insects
3.2. Hemipteran-Active Lectins
| Lectin (carbohydrate specificity) | Target insect toxicity (feeding assay/transgenic plant resistance) | References |
|---|---|---|
| GNA: Galanthus nivalis | Aphids (−/+) | [95,96] |
| agglutinin (Mannose) | Brown planthopper (+/+) | [100,111,112] |
| Green peach aphid (+/−) | [85] | |
| Pea aphid (−/+) | [113] | |
| Mustard aphid (−/+) | [110] | |
| ASA: Allium sativum agglutinin | Green peach aphid (+/−) | [85] |
| (Mannose) | Brown planthopper (−/+) | [104,105] |
| Mustard aphid(−/+) | [110] | |
| Chickpea aphid (+/+) Tobacco aphid (−/+) | [106] [114] | |
| ConA: Concanavalin A (Mannose, Galactose) | Green peach aphid (+/+) | [85,115] |
| Pea aphid (+/−) | [116] | |
| SNA-1: Sambucus nigra agglutinin(NeuAc(a-2,6)Gal/GalNAc) | Pea aphid (+/−) | [117] |
| WGA: Wheat germ agglutinin | Mustard aphid (+/−) | [118] |
| (N-Acetylglucosoamine) | Brown planthopper (+/+) | [111] |
| NPA: Narcissus pseudonarcissus agglutinin (Mannose) | Green peach aphid (+/−) | [85] |
| ATL: Arum maculatum tuber lectin (Mannose) | Chickpea aphid (+/−) | [106] |
| ACA onion: Allium cepa agglutinin (Galactose) | Mustard aphid (−/+) | [110] |
| Pea aphid (+/+) | [116,119] | |
| DEA: Dieffenbachia sequina agglutinin (thyroglobulin and asialofetuin [120]; mannose and complex sugar moities) | Chickpea aphid (+/+) | [106] |
| CEA: Colocasia esculenta agglutinin (thyroglobulin and asialofetuin [120]; mannose and complex sugar moities) | Chickpea aphid | [106] |
| PHA Phaseolus vulgaris agglutinin (Galactose, N-Acetylgalactosoamine) | Western tarnished plant bug; | [121] |
| PTA: Pinellia ternata agglutinin (Mannose) | Pea aphid; planthopper | [107,108,109] |
4. Antimetabolic Plant Protease Inhibitors
4.1. The Digestive and Salivary Proteases of the Hemiptera
| Hemipteran pest | Proteolytic activity | Plant Protease inhibitors | References |
|---|---|---|---|
| Pea aphid | Cysteine and aminopeptidase | OC-I, Barley cystatin, Bowman-Birk, Serpin | [49,132,133,134,137,138,139,140] |
| Green peach aphid | Cysteine and aminopeptidase | OC-I | [116,138] |
| Cotton aphid | Cathepsin-L like | OC-I | [57,138] |
| Cereal aphid | Cysteine and chymotrypsin-like | [135,141] | |
| Plant bug, Lygus hesperus | Aspartic and serine | [62] | |
| Plant bug, Lygus lineolaris | Serine and cysteine trypsin-like and chymotrypsin-like | [26] | |
| Planthopper | Trypsin and cathepsin B-like | [142] |
4.2. Insecticidal Effects of PIs against Hemiptera
5. Other Approaches
6. Conclusions and Future Outlook
Conflict of Interest
References
- Lu, Y.; Wu, K.; Jiang, Y.; Xia, B.; Li, P.; Feng, H.; Wyckhuys, K.A.G.; Guo, Y. Mirid bug outbreaks in multiple crops correlated with wide-scale adoption of Bt cotton in China. Science 2010, 328, 1151–1154. [Google Scholar]
- Blackman, R.L. Aphids on the Worlds Crops. An Identification and Information Guide; John Wiley & Sons: New York, NY, USA, 2000. [Google Scholar]
- van Emden, H.; Harrington, R. Aphids as Crop Pests; CABI Publishing: London, UK, 2007; p. 717. [Google Scholar]
- Faria, C.A.; Wackers, F.L.; Pritchard, J.; Barrett, D.A.; Turlings, T.C. High susceptibility of Bt maize to aphids enhances the performance of parasitoids of lepidopteran pests. PLoS One 2007, 2, e600. [Google Scholar]
- Powell, G.; Tosh, C.R.; Hardie, J. Host plant selection by aphids: Behavioral, evolutionary, and applied perspectives. Annu. Rev. Entomol. 2006, 51, 309–330. [Google Scholar] [CrossRef]
- Miles, P.W. Specific Responses and Damage Caused by Aphidoidea. In Aphids. Their biology, Natural Enemies and Control; Minks, A.K., Harrewijn, P., Eds.; Elsevier: Amsterdam, The Netherlands, 1989; Volume C, pp. 23–47. [Google Scholar]
- Sylvester, E.S. Viruses Transmitted by Aphids. In Aphids. Their Biology, Natural Enemies and Control; Minks, A.K., Harrewijn, P., Eds.; Elsevier: Amsterdam, The Netherlands, 1989; Volume C, pp. 65–88. [Google Scholar]
- Esquivel, J.F.; Mowery, S.V. Host plants of the tarnished plant bug (Heteroptera : Miridae) in Central Texas. Environ. Entomol. 2007, 36, 725–730. [Google Scholar] [CrossRef]
- Wheeler, A.G., Jr. Biology of the Plant Bugs (Hemiptera: Miridae): Pests, Predators, Opportunists; Comstock Publishing Associates: Ithaca, NY, USA, 2001; pp. i–xv, 1–507. [Google Scholar]
- Scott, D.R. An annotated listing of host plants of Lygus hesperus Knight. Bull. Entomol. Soc. Am. 1977, 23, 19–22. [Google Scholar]
- Young, O.P. Host plants of the tarnished plant bug, Lygus lineolaris (Heteroptera, Miridae). Ann. Entomol. Soc. Am. 1986, 79, 747–762. [Google Scholar]
- Brandt, S.L.; Coudron, T.A.; Habibi, J.; Brown, G.R.; Ooagan, O.M.; Wagner, R.M.; Wright, M.K.; Backus, E.A.; Huesing, J.E. Interaction of two Bacillus thuringiensis δ-endotoxins with the digestive system of Lygus hesperus. Curr. Microbiol. 2004, 48, 1–9. [Google Scholar] [CrossRef]
- Strong, F.E. Physiology of injury caused by Lygus hesperus. J. Econ. Entomol. 1970, 63, 808–814. [Google Scholar]
- McPherson, J.E.; McPherson, R.M. Stink Bugs of Economic Importance in America North of Mexico; CRC Press: Boca Raton, FL, USA, 2000. [Google Scholar]
- Reay-Jones, F.P.F. Spatial and temporal patterns of stink bugs (Hemiptera: Pentatomidae) in wheat. Environ. Entomol. 2010, 39, 944–955. [Google Scholar] [CrossRef]
- Young, W.R.; Teetes, G.L. Sorghum entomology. Annu. Rev. Entomol. 1977, 22, 193–218. [Google Scholar] [CrossRef]
- Michelbacher, A.E.; Middlekauff, W.W.; Bacon, O.G. Stink bug injury to tomatoes in California. J. Econ. Entomol. 1952, 45, 126–126. [Google Scholar]
- Viator, H.P.; Pantoja, A.; Smith, C.M. Damage to wheat seed quality and yield by the rice stink bug and Southern green stink bug (Hemiptera, Pentatomidae). J. Econ. Entomol. 1983, 76, 1410–1413. [Google Scholar]
- Medrano, E.G.; Bell, A.A. Role of Pantoea agglomerans in opportunistic bacterial seed and boll rot of cotton (Gossypium hirsutum) grown in the field. J. Appl. Microbiol. 2007, 102, 134–143. [Google Scholar] [CrossRef]
- Oliveira, M.R.V.; Henneberry, T.J.; Anderson, P. History, current status, and collaborative research projects for Bemisia tabaci. Crop Prot. 2001, 20, 709–723. [Google Scholar] [CrossRef]
- Faria, M.; Wraight, S.P. Biological control of Bemisia tabaci with fungi. Crop Prot. 2001, 20, 767–778. [Google Scholar] [CrossRef]
- Henneberry, T.J.; Jech, L.F.; Hendrix, D.L.; Steele, T. Bemisia argentifolii (Homoptera : Aleyrodidae) honeydew and honeydew sugar relationships to sticky cotton. Southwest. Entomol. 2000, 25, 1–14. [Google Scholar]
- Chen, J.; McAuslane, H.J.; Carle, R.B.; Webb, S.E. Impact of Bemisia argentifolii (Homoptera : Auchenorrhyncha : Aleyrodidae) infestation and squash silverleaf disorder on Zucchini yield and quality. J. Econ. Entomol. 2004, 97, 2083–2094. [Google Scholar] [CrossRef]
- Brown, J.K.; Czosnek, H. Whitefly transmission of plant viruses. Adv. Bot. Res. 2002, 36, 65–100. [Google Scholar] [CrossRef]
- Colvin, J.; Omongo, C.A.; Govindappa, M.R.; Stevenson, P.C.; Maruthi, M.N.; Gibson, G.; Seal, S.E.; Muniyappa, V. Host-plant viral infection effects on arthropod-vector population growth, development and behaviour: Management and epidemiological implications. Plant Virus Epidemiol. 2006, 67, 419–452. [Google Scholar] [CrossRef]
- Zhu, Y.C.; Zeng, F.R.; Oppert, B. Molecular cloning of trypsin-like cDNAs and comparison of proteinase activities in the salivary glands and gut of the tarnished plant bug Lygus lineolaris (Heteroptera : Miridae). Insect Biochem. Mol. Biol. 2003, 33, 889–899. [Google Scholar] [CrossRef]
- Lindsay, K.L.; Marshall, A.T. Ultrastructure of the filter chamber complex in the alimentary canal of Eurymela distincta Signoret (Homoptera, Eurymelidae). Int. J. Insect Morphol. Embryol. 1980, 9, 179–198. [Google Scholar] [CrossRef]
- Cheung, W.W.K.; Purcell, A.H. Ultrastructure of the digestive system of the leafhopper Euscelidius variegatus Kirshbaum (Homoptera, Cicadellidae), with and without congenital bacterial infections. Int. J. Insect Morphol. Embryol. 1993, 22, 49–61. [Google Scholar] [CrossRef]
- Martoja, R.; Ballan-Dufrancais, C. The Ultrastructure of Digestive and Excretory Organs. In Insect Ultrastructure; King, R.C., Akai, H., Eds.; Plenum Press: New York, NY, USA, 1984; Volume 2, pp. 199–268. [Google Scholar]
- Cristofoletti, P.T.; de Sousa, F.A.; Rahbe, Y.; Terra, W.R. Characterization of a membrane-bound aminopeptidase purified from Acyrthosiphon pisum midgut cells. A major binding site for toxic mannose lectins. FEBS J. 2006, 273, 5574–5588. [Google Scholar] [CrossRef]
- Karley, A.J.; Ashford, D.A.; Minto, L.M.; Pritchard, J.; Douglas, A.E. The significance of gut sucrase activity for osmoregulation in the pea aphid, Acyrthosiphon pisum. J. Insect Physiol. 2005, 51, 1313–1319. [Google Scholar] [CrossRef]
- Ragsdale, D.W.; Landis, D.A.; Brodeur, J.; Heimpel, G.E.; Desneux, N. Ecology and management of the soybean aphid in north America. Annu. Rev. Entomol. 2011, 56, 375–399. [Google Scholar] [CrossRef]
- Bhatia, V.; Uniyal, P.L.; Bhattacharya, R. Aphid resistance in Brassica crops: Challenges, biotechnological progress and emerging possibilities. Biotechnol. Adv. 2011, 29, 879–888. [Google Scholar] [CrossRef]
- Devonshire, A.L. Resistance of Aphids to Insecticides. In Aphids, Their Biology, Natural Enemies and Control; Minks, A.K., Harrewijn, P., Eds.; Elsevier: Amsterdam, The Netherlands, 1989; Volume C, pp. 123–139. [Google Scholar]
- Foster, S.P.; Harrington, R.; Dewar, A.M.; Denholm, I.; Devonshire, A.L. Temporal and spatial dynamics of insecticide resistance in Myzus persicae (Hemiptera: Aphididae). Pest Manag. Sci. 2002, 58, 895–907. [Google Scholar] [CrossRef]
- Diaz-Montano, J.; Reese, J.C.; Schapaugh, W.T.; Campbell, L.R. Characterization of antibiosis and antixenosis to the soybean aphid (Hemiptera : Aphididae) in several soybean genotypes. J. Econ. Entomol. 2006, 99, 1884–1889. [Google Scholar] [CrossRef]
- Hesler, L.S.; Dashiell, K.E.; Lundgren, J.G. Characterization of resistance to Aphis glycines in soybean accessions. Euphytica 2007, 154, 91–99. [Google Scholar] [CrossRef]
- Hill, C.B.; Li, Y.; Hartman, G.L. Resistance to the soybean aphid in soybean germplasm. Crop Sci. 2004, 44, 98–106. [Google Scholar] [CrossRef]
- Li, Y.; Hill, C.B.; Carlson, S.R.; Diers, B.W.; Hartman, G.L. Soybean aphid resistance genes in the soybean cultivars Dowling and Jackson map to linkage group M. Mol. Breed. 2007, 19, 25–34. [Google Scholar]
- Pal, N.; Boyapalle, S.; Beckett, R.; Miller, W.A.; Bonning, B.C. A baculovirus-expressed dicistrovirus that is infectious to aphids. J. Virol. 2007, 81, 9339–9345. [Google Scholar] [Green Version]
- Grenier, A.M.; Duport, G.; Pages, S.; Condemine, G.; Rahbe, Y. The phytopathogen Dickeya dadantii (Erwinia chrysanthemi 3937) is a pathogen of the pea aphid. Appl. Environ. Microbiol. 2006, 72, 1956–1965. [Google Scholar]
- Schnepf, E.; Crickmore, N.; van Rie, J.; Lereclus, D.; Baum, J.; Feitelson, J.; Zeigler, D.R.; Dean, D.H. Bacillus thuringiensis and its pesticidal crystal proteins. Microbiol. Mol. Biol. Rev. 1998, 62, 775–806. [Google Scholar]
- Knowles, B.H.; Ellar, D.J. Colloid-osmotic lysis is a general feature of the mechanism of action of Bacillus thuringiensis delta-endotoxins with different insect specificity. Biochim. Biophys. Acta 1987, 924, 509–518. [Google Scholar] [CrossRef]
- Grochulski, P.; Masson, L.; Borisova, S.; Pusztaicarey, M.; Schwartz, J.L.; Brousseau, R.; Cygler, M. Bacillus thuringiensis Cry1A(a) insecticidal toxin—crystal structure and channel formation. J. Mol. Biol. 1995, 254, 447–464. [Google Scholar] [CrossRef]
- Bravo, A.; Gomez, I.; Conde, J.; Munoz-Garay, C.; Sanchez, J.; Miranda, R.; Zhuang, M.; Gill, S.S.; Soberon, M. Oligomerization triggers binding of a Bacillus thuringiensis Cry1Ab pore-forming toxin to aminopeptidase N receptor leading to insertion into membrane microdomains. Biochim. Biophys. Acta-Biomembr. 2004, 1667, 38–46. [Google Scholar] [CrossRef]
- Zhang, X.; Candas, M.; Griko, N.B.; Taussig, R.; Bulla, L.A., Jr. A mechanism of cell death involving an adenylyl cyclase/PKA signaling pathway is induced by the Cry1Ab toxin of Bacillus thuringiensis. Proc. Natl. Acad. Sci. USA 2006, 103, 9897–9902. [Google Scholar]
- Payne, J.R.; Cannon, R.J.C. Use of Bacillus thuringiensis Isolates for Controlling Pests in the Family Aphididae. US Patent 5,262,159, 16 November 1993. [Google Scholar]
- Walters, F.S.; English, L.H. Toxicity of Bacillus thuringiensis δ-endotoxins toward the potato aphid in an artificial diet bioassay. Entomol. Exp. Appl. 1995, 77, 211–216. [Google Scholar] [CrossRef]
- Cristofoletti, P.T.; Ribeiro, A.F.; Deraison, C.; Rahbe, Y.; Terra, W.R. Midgut adaptation and digestive enzyme distribution in a phloem feeding insect, the pea aphid Acyrthosiphon pisum. J. Insect Physiol. 2003, 49, 11–24. [Google Scholar] [CrossRef]
- Porcar, M.; Grenier, A.M.; Federici, B.; Rahbe, Y. Effects of Bacillus thuringiensis δ-endotoxins on the pea aphid (Acyrthosiphon pisum). Appl. Environ. Microbiol. 2009, 75, 4897–4900. [Google Scholar] [CrossRef]
- Ali, M.I.; Luttrell, R.G. Response estimates for assessing heliothine susceptibility to Bt toxins. J. Econ. Entomol. 2009, 102, 1935–1947. [Google Scholar] [CrossRef]
- Park, Y.; Abdullah, M.A.F.; Taylor, M.D.; Rahman, K.; Adang, M.J. Enhancement of Bacillus thuringiensis Cry3Aa and Cry3Bb toxicities to coleopteran larvae by a toxin-binding fragment of an insect cadherin. Appl. Environ. Microbiol. 2009, 75, 3086–3092. [Google Scholar] [CrossRef]
- Sattar, S.; Maiti, M.K. Molecular characterization of a novel vegetative insecticidal protein from Bacillus thuringiensis effective against sap-sucking insect pest. J. Microbiol. Biotechnol. 2011, 21, 937–946. [Google Scholar] [CrossRef]
- Baum, J.A.; Roberts, J.K.; Zhang, B.; Anderson, H.; Chay, C.A. Insecticidal compositions and methods for makeing insect-resistant transgenic plants. US 2009/0,068,159 A1, 12 March 2009. [Google Scholar]
- Baum, J.; Flasinski, S.; Heck, G.R.; Penn, S.R.; Sukuru, U.R.; Shi, X. Novel hemipteran and coleopteran active toxin proteins from Bacillus thuringiensis. US 2010/0,064,394, 11 March 2010. [Google Scholar]
- Li, H.R.; Chougule, N.P.; Bonning, B.C. Interaction of the Bacillus thuringiensis delta endotoxins Cry1Ac and Cry3Aa with the gut of the pea aphid, Acyrthosiphon pisum (Harris). J. Invertebr. Pathol. 2011, 107, 69–78. [Google Scholar] [CrossRef]
- Deraison, C.; Darboux, I.; Duportets, L.; Gorojankina, T.; Rahbe, Y.; Jouanin, L. Cloning and characterization of a gut-specific cathepsin L from the aphid Aphis gossypii. Insect Mol. Biol. 2004, 13, 165–177. [Google Scholar] [CrossRef]
- Carroll, J.; Convents, D.; van Damme, J.; Boets, A.; van Rie, J.; Ellar, D.J. Intramolecular proteolytic cleavage of Bacillus thuringiensis Cry3A delta-endotoxin may facilitate its coleopteran toxicity. J. Invertebr. Pathol. 1997, 70, 41–49. [Google Scholar] [CrossRef]
- Yamagiwa, M.; Esaki, M.; Otake, K.; Inagaki, M.; Komano, T.; Amachi, T.; Sakai, H. Activation process of dipteran-specific insecticidal protein produced by Bacillus thuringiensis subsp. israelensis. Appl. Environ. Microbiol. 1999, 65, 3464–3469. [Google Scholar]
- Garczynski, S.F.; Crim, J.W.; Adang, M.J. Identification of putative insect brush border membrane-binding molecules specific to Bacillus thuringiensis delta-endotoxin by protein blot analysis. Appl. Environ. Microbiol. 1991, 57, 2816–2820. [Google Scholar]
- Aranda, E.; Sanchez, J.; Peferoen, M.; Guereca, L.; Bravo, A. Interactions of Bacillus thuringiensis crystal proteins with the midgut epithelial cells of Spodoptera frugiperda (Lepidoptera: Noctuidae). J. Invertebr. Pathol. 1996, 68, 203–212. [Google Scholar] [CrossRef]
- Wright, M.K.; Brandt, S.L.; Coudron, T.A.; Wagner, R.M.; Habibi, J.; Backus, E.A.; Huesing, J.E. Characterization of digestive proteolytic activity in Lygus hesperus Knight (Hemiptera : Miridae). J. Insect Physiol. 2006, 52, 717–728. [Google Scholar] [CrossRef]
- Habibi, J.; Brandt, S.L.; Coudron, T.A.; Wagner, R.M.; Wright, M.K.; Backus, E.A.; Huesing, J.E. Uptake, flow, and digestion of casein and green fluorescent protein in the digestive system of Lygus hesperus Knight. Arch. Insect Biochem. Physiol. 2002, 50, 62–74. [Google Scholar] [CrossRef]
- Walters, F.S.; Stacy, C.M.; Lee, M.K.; Palekar, N.; Chen, J.S. An engineered chymotrypsin/cathepsing site in domain I renders Bacillus thuringiensis Cry3A active against western corn rootworm larvae. Appl. Environ. Microbiol. 2008, 74, 367–374. [Google Scholar] [CrossRef]
- Mehlo, L.; Gahakwa, D.; Nghia, P.T.; Loc, N.T.; Capell, T.; Gatehouse, J.A.; Gatehouse, A.M.R.; Christou, P. An alternative strategy for sustainable pest resistance in genetically enhanced crops. Proc. Natl. Acad. Sci. USA 2005, 102, 7812–7816. [Google Scholar]
- Glynn, L.E. Lectins N. Sharon and H. Lis. Chapman and Hall, London and New York, 127 pages, £19.50 (1989). Cell Biochem. Funct. 1990, 78, 190. [Google Scholar] [CrossRef]
- Kilpatrick, D.C. Mannan-binding lectin: Clinical significance and applications. Biochim. Biophys. Acta-Gen. Subj. 2002, 1572, 401–413. [Google Scholar] [CrossRef]
- van Damme, E.J.M.; Lannoo, N.; Peumans, W.J. Plant lectins. Adv. Bot. Res. 2008, 48, 107–209. [Google Scholar] [CrossRef]
- Peumans, W.J.; Vandamme, E.J.M. Lectins as plant defense proteins. Plant Physiol. 1995, 109, 347–352. [Google Scholar]
- Howard, I.K.; Horton, C.B.; Sage, H.J. Studies on appearance and location of hemagglutinins from a common lentil during the life cycle of a plant. Arch. Biochem. Biophys. 1972, 149, 323–326. [Google Scholar] [CrossRef]
- Brill, L.M.; Evans, C.J.; Hirsch, A.M. Expression of MsLEC1- and MsLEC2-antisense genes in alfalfa plant lines causes severe embryogenic, developmental and reproductive abnormalities. Plant J. 2001, 25, 453–461. [Google Scholar] [CrossRef]
- Huang, G.L.; Huang, H.L.; Zhang, H.C.; Wang, P.G. Structure-function relations of carbohydrates by neoglycolipid arrays. Appl. Biochem. Biotechnol. 2006, 133, 211–215. [Google Scholar] [CrossRef]
- Rodrigues Macedo, M.L.; Damico, D.C.; Machado Freire, M.G.; Toyama, M.H.; Marangoni, S.; Novello, J.C. Purification and characterization of an N-acetylglucosamine-binding lectin from Koelreuteria paniculata seeds and its effect on the larval development of Callosobruchus maculatus (Coleoptera: Bruchidae) and Anagasta kuehniella (Lepidoptera: Pyralidae). J. Agric. Food Chem. 2003, 51, 2980–2986. [Google Scholar]
- Luo, Y.; Xu, X.; Liu, J.; Li, J.; Sun, Y.; Liu, Z.; Liu, J.; van Damme, E.; Balzarini, J.; Bao, J. A novel mannose-binding tuber lectin from Typhonium divaricatum (L.) Decne (family Araceae) with antiviral activity against HSV-II and anti-proliferative effect on human cancer cell lines. J. Biochem. Mol. Biol. 2007, 40, 358–367. [Google Scholar] [CrossRef]
- Poulsen, M.; Schroder, M.; Wilcks, A.; Kroghsbo, S.; Lindecrona, R.H.; Miller, A.; Frenzel, T.; Danier, J.; Rychlik, M.; Shu, Q.; et al. Safety testing of GM-rice expressing PHA-E lectin using a new animal test design. Food Chem. Toxicol. 2007, 45, 364–377. [Google Scholar] [CrossRef]
- Peumans, W.J.; Damme, E.J.M.V. Prevalence, biological activity and genetic manipulation of lectins in foods. Trends Food Sci. Technol. 1996, 7, 132–138. [Google Scholar] [CrossRef]
- Pusztai, A.; Ewen, S.W.B.; Grant, G.; Peumans, W.J.; Damme, E.J.M.V.; Coates, M.E.; Bardocz, S. Lectins and also bacteria modify the glycosylation of gut surface receptors in the rat. Glycoconjugate 1995, 12, 22–35. [Google Scholar] [CrossRef]
- Pusztai, A.; Koninkx, J.; Hendriks, H.; Kok, W.; Hulscher, S.; Damme, E.J.M.V.; Peumans, W.J.; Grant, G.; Bardocz, S. Effect of the insecticidal Galanthus nivalis agglutinin on metabolism and the activities of brush boarder enzymes in the rat small intestine. Nutr. Biochem. 1996, 7, 677–682. [Google Scholar] [CrossRef]
- Vandenborre, G.; Smagghe, G.; van Damme, E.J. Plant lectins as defense proteins against phytophagous insects. Phytochemistry 2011, 72, 1538–1550. [Google Scholar] [CrossRef]
- Rahbe, Y.; Febvay, G. Protein toxicity to aphids—an in vitro test on Acythosiphon pisum. Entomol. Exp. Appl. 1993, 67, 149–160. [Google Scholar] [CrossRef]
- Du, J.P.; Foissac, X.; Carss, A.; Gatehouse, A.M.R.; Gatehouse, J.A. Ferritin acts as the most abundant binding protein for snowdrop lectin in the midgut of rice brown planthoppers (Nilaparvata lugens). Insect Biochem. Mol. Biol. 2000, 30, 297–305. [Google Scholar] [CrossRef]
- Sadeghi, A.; Smagghe, G.; Proost, P.; van Damme, E.J.M. Ferritin acts as a target site for the snowdrop lectin (GNA) in the midgut of the cotton leafworm Spodoptera littoralis. Insect Sci. 2008, 15, 513–519. [Google Scholar] [CrossRef]
- Fitches, E.; Wiles, D.; Douglas, A.E.; Hinchliffe, G.; Audsley, N.; Gatehouse, J.A. The insecticidal activity of recombinant garlic lectins towards aphids. Insect Biochem. Mol. Biol. 2008, 38, 905–915. [Google Scholar] [CrossRef]
- Majumder, P.; Mondal, H.A.; Das, S. Insecticidal activity of Arum maculatum tuber lectin and its binding to the glycosylated insect gut receptors. J. Agric. Food Chem. 2005, 53, 6725–6729. [Google Scholar] [CrossRef]
- Sauvion, N.; Rahbe, Y.; Peumans, W.J.; VanDamme, E.J. M.; Gatehouse, J.A.; Gatehouse, A.M.R. Effects of GNA and other mannose binding lectins on development and fecundity of the peach-potato aphid Myzus persicae. Entomol. Exp. Appl. 1996, 79, 285–293. [Google Scholar] [CrossRef]
- Sauvion, N.; Nardon, C.; Febvay, G.; Gatehouse, A.M.R.; Rahbe, Y. Binding of the insecticidal lectin Concanavalin A in pea aphid, Acyrthosiphon pisum (Harris) and induced effects on the structure of midgut epithelial cells. J. Insect Physiol. 2004, 50, 1137–1150. [Google Scholar] [CrossRef]
- Sauvion, N.; Charles, H.; Febvay, G.; Rahbe, Y. Effects of jackbean lectin (ConA) on the feeding behaviour and kinetics of intoxication of the pea aphid, Acyrthosiphon pisum. Entomol. Exp. Appl. 2004, 110, 31–44. [Google Scholar] [CrossRef]
- Fitches, E.; Woodhouse, S.D.; Edwards, J.P.; Gatehouse, J.A. In vitro and in vivo binding of snowdrop (Galanthus nivalis agglutinin; GNA) and jackbean (Canavalia ensiformis; Con A) lectins within tomato moth (Lacanobia oleracea) larvae; mechanisms of insecticidal action. J. Insect Physiol. 2001, 47, 777–787. [Google Scholar] [CrossRef]
- Powell, K.S.; Spence, J.; Bharathi, M.; Gatehouse, J.A.; Gatehouse, A.M.R. Immunohistochemical and developmental studies to elucidate the mechanism of action of the snowdrop lectin on the rice brown planthopper, Nilaparvata lugens (Stal). J. Insect Physiol. 1998, 44, 529–539. [Google Scholar] [CrossRef]
- Gatehouse, A.M.R.; Dewey, F.M.; Dove, J.; Fenton, K.A.; Pusztai, A. Effect of seed lectins from Phaseolus vulgaris on the development of larvae of Callosobruchis maculatus—Mechanism of toxicity. J. Sci. Food Agric. 1984, 35, 373–380. [Google Scholar] [CrossRef]
- Czapla, T.H.; Lang, B.A. Effect of plant lectins on the larval development of European corn borer (Lepidoptera, Pyralidae) and Southern corn rootworm (Coleoptera, Chysomelidae). J. Econ. Entomol. 1990, 83, 2480–2485. [Google Scholar]
- Murdock, L.L.; Huesing, J.E.; Nielsen, S.S.; Pratt, R.C.; Shade, R.E. Biological effects of plant lectins on the cowpea weevil. Phytochemistry 1990, 29, 85–89. [Google Scholar]
- Janzen, D.H.; Juster, H.B.; Liener, I.E. Insecticidal action of phytohemagglutinin in black beans on a bruchid beetle. Science 1976, 192, 795–796. [Google Scholar]
- Harper, S.M.; Crenshaw, R.W.; Mullins, M.A.; Privalle, L.S. Lectin binding to insect brush border membranes. J. Econ. Entomol. 1995, 88, 1197–1202. [Google Scholar]
- Shi, Y.; Wang, M.B.; Powell, K.S.; Vandamme, E.; Hilder, V.A.; Gatehouse, A.M.R.; Boulter, D.; Gatehouse, J.A. Use of the rice sucrose synthase-1 promoter to direct phloem-specific expression of beta-glucuronidase and snowdrop lectin genes in transgenic tobacco plants. J. Exp. Bot. 1994, 45, 623–631. [Google Scholar] [CrossRef]
- Hilder, V.A.; Powell, K.S.; Gatehouse, A.M.R.; Gatehouse, J.A.; Gatehouse, L.N.; Shi, Y.; Hamilton, W.D.O.; Merryweather, A.; Newell, C.A.; Timans, J.C. Expression of snowdrop lectin in transgenic tobacco plants results in added protection against aphids. Transgenic Res. 1995, 4, 18–25. [Google Scholar] [CrossRef]
- Gatehouse, A.M.R.; Down, R.E.; Powell, K.S.; Sauvion, N.; Rahbe, Y.; Newell, C.A.; Merryweather, A.; Hamilton, W.D.O.; Gatehouse, J.A. Transgenic potato plants with enhanced resistance to the peach-potato aphid Myzus persicae. Entomol. Exp. Appl. 1996, 79, 295–307. [Google Scholar] [CrossRef]
- Rao, K.V.; Rathore, K.S.; Hodges, T.K.; Fu, X.; Stoger, E.; Sudhakar, D.; Williams, S.; Christou, P.; Bharathi, M.; Bown, D.P.; et al. Expression of snowdrop lectin (GNA) in transgenic rice plants confers resistance to rice brown planthopper. Plant J. 1998, 15, 469–477. [Google Scholar] [CrossRef]
- Stoger, E.; Williams, S.; Christou, P.; Down, R.E.; Gatehouse, J.A. Expression of the insecticidal lectin from snowdrop (Galanthus nivalis agglutinin; GNA) in transgenic wheat plants: Effects on predation by the grain aphid Sitobion avenae. Mol. Breed. 1999, 5, 65–73. [Google Scholar] [CrossRef]
- Foissac, X.; Loc, N.T.; Christou, P.; Gatehouse, A.M.R.; Gatehouse, J.A. Resistance to green leafhopper (Nephotettix virescen ) and brown planthopper (Nilaparvata lugens) in transgenic rice expressing snowdrop lectin (Galanthus nivalis agglutinin; GNA). J. Insect Physiol. 2000, 46, 573–583. [Google Scholar] [CrossRef]
- Bandyopadhyay, S.; Roy, A.; Das, S. Binding of garlic (Allium sativum) leaf lectin to the gut receptors of homopteran pests is correlated to its insecticidal activity. Plant Sci. 2001, 161, 1025–1033. [Google Scholar] [CrossRef]
- Dutta, I.; Majumder, P.; Saha, P.; Ray, K.; Das, S. Constitutive and phloem specific expression of Allium sativum leaf agglutinin (ASAL) to engineer aphid (Lipaphis erysimi) resistance in transgenic Indian mustard (Brassica juncea). Plant Sci. 2005, 169, 996–1007. [Google Scholar] [CrossRef]
- Dutta, I.; Saha, P.; Majumder, P.; Sarkar, A.; Chakraborti, D.; Banerjee, S.; Das, S. The efficacy of a novel insecticidal protein, Allium sativum leaf lectin (ASAL), against homopteran insects monitored in transgenic tobacco. Plant Biotechnol. J. 2005, 3, 601–611. [Google Scholar] [CrossRef]
- Saha, P.; Dasgupta, I.; Das, S. A novel approach for developing resistance in rice against phloem limited viruses by antagonizing the phloem feeding hemipteran vectors. Plant Mol. Biol. 2006, 62, 735–752. [Google Scholar] [CrossRef]
- Saha, P.; Majumder, P.; Dutta, I.; Ray, T.; Roy, S.C.; Das, S. Transgenic rice expressing Allium sativum leaf lectin with enhanced resistance against sap-sucking insect pests. Planta 2006, 223, 1329–1343. [Google Scholar] [CrossRef]
- Chakraborti, D.; Sarkar, A.; Mondal, H.A.; Das, S. Tissue specific expression of potent insecticidal, Allium sativum leaf agglutinin (ASAL) in important pulse crop, chickpea (Cicer arietinum L.) to resist the phloem feeding Aphis craccivora. Transgenic Res. 2009, 18, 529–544. [Google Scholar] [CrossRef]
- Yao, J.H.; Pang, Y.Z.; Qi, H.X.; Wan, B.L.; Zhao, X.Y.; Kong, W.W.; Sun, X.F.; Tang, K.X. Transgenic tobacco expressing Pinellia ternata agglutinin confers enhanced resistance to aphids. Transgenic Res. 2003, 12, 715–722. [Google Scholar] [CrossRef]
- Yao, J.H.; Zhao, X.Y.; Liao, Z.H.; Lin, J.; Chen, Z.H.; Chen, F.; Song, J.; Sun, X.F.; Tang, K.X. Cloning and molecular characterization of a novel lectin gene from Pinellia ternata. Cell Res. 2003, 13, 301–308. [Google Scholar] [CrossRef]
- Zhang, H.-Y.; Wu, X.-J.; Tang, K.-X.; Wang, X.-D.; Sun, X.-F.; Zhou, K.-D. A primary study of transferring the Pinellia tenata agglutinin (pta) gene into rice and expression. Acta Genet. Sinica 2003, 30, 1013–1019. [Google Scholar]
- Hossain, M.A.; Maiti, M.K.; Basu, A.; Sen, S.; Ghosh, A.K.; Sen, S.K. Transgenic expression of onion leaf lectin gene in Indian mustard offers protection against aphid colonization. Crop Sci. 2006, 46, 2022–2032. [Google Scholar] [CrossRef]
- Powell, K.S.; Gatehouse, A.M.R.; Hilder, V.A.; Vandamme, E.J. M.; Peumans, W.J.; Boonjawat, J.; Horsham, K.; Gatehouse, J.A. Different antimetabolic effects of related lectins towards nymphal stages of Nilaparvata lugens. Entomol. Exp. Appl. 1995, 75, 61–65. [Google Scholar] [CrossRef]
- Nagadhara, D.; Ramesh, S.; Pasalu, I.C.; Rao, Y.K.; Sarma, N.P.; Reddy, V.D.; Rao, K.V. Transgenic rice plants expressing the snowdrop lectin gene (gna) exhibit high-level resistance to the whitebacked planthopper (Sogatella furcifera). Theor. Appl. Genet. 2004, 109, 1399–1405. [Google Scholar] [CrossRef]
- Wang, Z.Y.; Zhang, K.W.; Sun, X.F.; Tang, K.X.; Zhang, J.R. Enhancement of resistance to aphids by introducing the snowdrop lectin gene gna into maize plants. J. Biosci. 2005, 30, 627–638. [Google Scholar] [CrossRef]
- Sadeghi, A.; Smagghe, G.; Broeders, S.; Hernalsteens, J.P.; De Greve, H.; Peumans, W.J.; van Damme, E.J. Ectopically expressed leaf and bulb lectins from garlic (Allium sativum L.) protect transgenic tobacco plants against cotton leafworm (Spodoptera littoralis). Transgenic Res. 2008, 17, 9–18. [Google Scholar] [CrossRef]
- Gatehouse, A.M.R.; Davison, G.M.; Stewart, J.N.; Galehouse, L.N.; Kumar, A.; Geoghegan, I.E.; Birch, A.N.E.; Gatehouse, J.A. Concanavalin A inhibits development of tomato moth (Lacanobia oleracea) and peach-potato aphid (Myzus persicae) when expressed in transgenic potato plants. Mol. Breed. 1999, 5, 153–165. [Google Scholar] [CrossRef]
- Rahbe, Y.; Sauvion, N.; Febvay, G.; Peumans, W.J.; Gatehouse, A.M.R. Toxicity of lectins and processing of ingested proteins in the pea aphid Acyrthosiphon pisum. Entomol. Exp. Appl. 1995, 76, 143–155. [Google Scholar] [CrossRef]
- Shahidi-Noghabi, S.; van Damme, E.J.; Mahdian, K.; Smagghe, G. Entomotoxic action of Sambucus nigra agglutinin I in Acyrthosiphon pisum aphids and Spodoptera exigua caterpillars through caspase-3-like-dependent apoptosis. Arch. Insect Biochem. Physiol. 2010, 75, 207–220. [Google Scholar] [CrossRef]
- Kanrar, S.; Venkateswari, J.; Kirti, P.B.; Chopra, V.L. Transgenic Indian mustard (Brassica juncea) with resistance to the mustard aphid (Lipaphis erysimi Kalt.). Plant Cell Rep. 2002, 20, 976–981. [Google Scholar] [CrossRef]
- Wu, J.; Luo, X.; Guo, H.; Xiao, J.; Tian, Y. Transgenic cotton, expressing Amaranthus caudatus agglutinin, confers enhanced resistance to aphids. Plant Breed. 2006, 125, 390–394. [Google Scholar] [CrossRef]
- van Damme, E.J.M.; Nakamura-Tsuruta, S.; Smith, D.F.; Ongenaert, M.; Winter, H.C.; Rouge, P.; Goldstein, I.J.; Mo, H.; Kominami, J.; Culerrier, R.; et al. Phylogenetic and specificity studies of two-domain GNA-related lectins: Generation of multispecificity through domain duplication and divergent evolution. Biochem. J. 2007, 404, 51–61. [Google Scholar] [CrossRef]
- Habibi, J.; Backus, E.A.; Huesing, J.E. Effects of phytohemagglutinin (PHA) on the structure of midgut epithelial cells and localization of its binding sites in western tarnished plant bug, Lygus hesperus Knight. J. Insect Physiol. 2000, 46, 611–619. [Google Scholar] [CrossRef]
- Hamshou, M.; Smagghe, G.; Shahidi-Noghabi, S.; de Geyter, E.; Lannoo, N.; van Damme, E.J.M. Insecticidal properties of Sclerotinia sclerotiorum agglutinin and its interaction with insect tissues and cells. Insect Biochem. Mol. Biol. 2010, 40, 883–890. [Google Scholar] [CrossRef]
- Qi, G.; Lan, N.; Ma, X.; Yu, Z.; Zhao, X. Controlling Myzus persicae with recombinant endophytic fungi Chaetomium globosum expressing Pinellia ternata agglutinin. J. Appl. Microbiol. 2011, 110, 1314–1322. [Google Scholar] [CrossRef]
- Ryan, C.A. Protease inhibitors in plants—genes for improving defenses against insects and pathogens. Annu. Rev. Phytopathol. 1990, 28, 425–449. [Google Scholar] [CrossRef]
- Jongsma, M.A.; Beekwilder, J. Co-evolution of insect proteases and plant protease inhibitors. Curr. Protein Pept. Sci. 2011, 12, 437–447. [Google Scholar] [CrossRef]
- Broadway, R.M.; Duffey, S.S. The effect of dietary protein on the growth and digestive physiology of larval Heliothis zea and Spodoptera exigua. J. Insect Physiol. 1986, 32, 673–680. [Google Scholar] [CrossRef]
- Tamhane, V.A.; Chougule, N.P.; Giri, A.P.; Dixit, A.R.; Sainani, M.N.; Gupta, V.S. In vivo and in vitro effect of Capsicum annum proteinase inhibitors on Helicoverpa armigera gut proteinases. Biochim. Biophys. Acta-Gen. Subj. 2005, 1722, 156–167. [Google Scholar] [CrossRef]
- Broadway, R.M. Are insects resistant to plant proteainase inhibitors? J. Insect Physiol. 1995, 41, 107–116. [Google Scholar] [CrossRef]
- Jongsma, M.A.; Bakker, P.L.; Peters, J.; Bosch, D.; Stiekema, W.J. Adaptation of Spodoptera exigua larvae to plant proteinase inhibitors by induction of gut proteinase activity insensitive to inhibition. Proc. Natl. Acad. Sci. USA 1995, 92, 8041–8045. [Google Scholar]
- Bown, D.P.; Wilkinson, H.S.; Gatehouse, J.A. Differentially regulated inhibitor-sensitive and insensitive protease genes from the phytophagous insect pest, Helicoverpa armigara, are members of complex multigene families. Insect Biochem. Mol. Biol. 1997, 27, 625–638. [Google Scholar] [CrossRef]
- Chougule, N.P.; Giri, A.P.; Sainani, M.N.; Gupta, V.S. Gene expression patterns of Helicoverpa armigera gut proteases. Insect Biochem. Mol. Biol. 2005, 35, 355–367. [Google Scholar] [CrossRef]
- Rispe, C.; Kutsukake, M.; Doublet, V.; Hudaverdian, S.; Legeai, F.; Simon, J.C.; Tagu, D.; Fukatsu, T. Large gene family expansion and variable selective pressures for cathepsin B in aphids. Mol. Biol. Evol. 2008, 25, 5–17. [Google Scholar]
- Carrillo, L.; Martinez, M.; Alvarez-Alfageme, F.; Castanera, P.; Smagghe, G.; Diaz, I.; Ortego, F. A barley cysteine-proteinase inhibitor reduces the performance of two aphid species in artificial diets and transgenic Arabidopsis plants. Transgenic Res. 2011, 20, 305–319. [Google Scholar] [CrossRef]
- Kutsukake, M.; Shibao, H.; Nikoh, N.; Morioka, M.; Tamura, T.; Hoshino, T.; Ohgiya, S.; Fukatsu, T. Venomous protease of aphid soldier for colony defense. Proc. Natl. Acad. Sci. USA 2004, 101, 11338–11343. [Google Scholar]
- Pyati, P.; Bandani, A.R.; Fitches, E.; Gatehouse, J.A. Protein digestion in cereal aphids (Sitobion avenae) as a target for plant defence by endogenous proteinase inhibitors. J. Insect Physiol. 2011, 57, 881–891. [Google Scholar] [CrossRef]
- The International Aphid Genomics. Genome sequence of the pea aphid Acyrthosiphon pisum. PLoS Biol. 2010, 8, e1000313. [CrossRef]
- Alvarez-Alfageme, F.; Maharramov, J.; Carrillo, L.; Vandenabeele, S.; Vercammen, D.; van Breusegem, F.; Smagghe, G. Potential use of a serpin from arabidopsis for pest control. PLoS One 2011, 6, e20278. [Google Scholar]
- Rahbe, Y.; Deraison, C.; Bonade-Bottino, M.; Girard, C.; Nardon, C.; Jouanin, L. Effects of the cysteine protease inhibitor oryzacystatin (OC-I) on different aphids and reduced performance of Myzus persicae on OC-I expressing transgenic oilseed rape. Plant Sci. 2003, 164, 441–450. [Google Scholar] [CrossRef]
- Rahbe, Y.; Ferrasson, E.; Rabesona, H.; Quillien, L. Toxicity to the pea aphid Acyrthosiphon pisum of anti-chymotrypsin isoforms and fragments of Bowman-Birk protease inhibitors from pea seeds. Insect Biochem. Mol. Biol. 2003, 33, 299–306. [Google Scholar] [CrossRef]
- Consortium, I.A.G. Genome sequence of the pea aphid Acyrthosiphon pisum. PLoS Biol. 2010, 8, e1000313. [Google Scholar] [CrossRef]
- Tran, P.; Cheesbrough, T.M.; Keickhefer, R.W. Plant proteinase inhibitors are potential anticereal aphid compounds. J. Econ. Entomol. 1997, 90, 1672–1677. [Google Scholar]
- Foissac, X.; Edwards, M.G.; Du, J.P.; Gatehouse, A.M.; Gatehouse, J.A. Putative protein digestion in a sap-sucking homopteran plant pest (rice brown plant hopper; Nilaparvata lugens: Delphacidae)—identification of trypsin-like and cathepsin B-like proteases. Insect Biochem. Mol. Biol. 2002, 32, 967–978. [Google Scholar] [CrossRef]
- Wu, K.; Li, W.; Feng, H.; Guo, Y. Seasonal abundance of the mirids, Lygus lucorum and Adelphocoris spp. (Hemiptera : Miridae) on Bt cotton in northern China. Crop Prot. 2002, 21, 997–1002. [Google Scholar] [CrossRef]
- Zeng, F.R.; Zhu, Y.C.; Cohen, A.C. Molecular cloning and partial characterization of a trypsin-like protein in salivary glands of Lygus hesperus (Hemiptera : Miridae). Insect Biochem. Mol. Biol. 2002, 32, 455–464. [Google Scholar] [CrossRef]
- Zeng, F.; Zhu, Y.; Cohen, A. Partial characterization of trypsin-like protease and molecular cloning of a trypsin-like precursor cDNA in salivary glands of Lygus lineolaris. Comp. Biochem. Physiol. B-Biochem. Mol. Biol. 2002, 131, 453–463. [Google Scholar] [CrossRef]
- Cohen, A.C. Organization of digestion and preliminary characterization of salivary trypsin-like enzymes in a predaceous heteropteran, Zelus renardii. J. Insect Physiol. 1993, 39, 823–829. [Google Scholar] [CrossRef]
- Cohen, A.C. Solid-to-liquid feeding: The inside(s) story of extra-oral digestion in predaceous arthropoda. Am. Entomol. 1998, 44, 103–115. [Google Scholar]
- Salvucci, M.E.; Rosell, R.C.; Brown, J.K. Uptake and metabolism of leaf proteins by the silverleaf whitefly. Arch. Insect Biochem. Physiol. 1998, 39, 155–165. [Google Scholar] [CrossRef]
- Gattolin, S.; Newbury, H.J.; Bale, J.S.; Tseng, H.-M.; Barrett, D.A.; Pritchard, J. A diurnal component to the variation in sieve tube amino acid content in wheat. Plant Physiol. 2008, 147, 912–921. [Google Scholar] [CrossRef]
- Gatehouse, J.A. Prospects for Using Proteinase Inhibitors to Protect Transgenic Plants Against Attack by Herbivorous Insects. Curr. Protein Pept. Sci. 2011, 12, 409–416. [Google Scholar] [CrossRef]
- Bown, D.P.; Wilkinson, H.S.; Gatehouse, J.A. Regulation of expression of genes encoding digestive proteases in the gut of a polyphagous lepidopteran larva in response to dietary protease inhibitors. Physiol. Entomol. 2004, 29, 278–290. [Google Scholar] [CrossRef]
- Cowgill, S.E.; Atkinson, H.J. A sequential approach to risk assessment of transgenic plants expressing protease inhibitors: Effects on nontarget herbivorous insects. Transgenic Res. 2003, 12, 439–449. [Google Scholar] [CrossRef]
- Sadeghi, A.; van Damme, E.J.; Smagghe, G. Evaluation of the susceptibility of the pea aphid, Acyrthosiphon pisum, to a selection of novel biorational insecticides using an artificial diet. J. Insect Sci. 2009, 9, 1–8. [Google Scholar]
- Martin, P.A.; Hirose, E.; Aldrich, J.R. Toxicity of Chromobacterium subtsugae to southern green stink bug (Heteroptera: Pentatomidae) and corn rootworm (Coleoptera: Chrysomelidae). J. Econ. Entomol. 2007, 100, 680–684. [Google Scholar] [CrossRef]
- Staniscuaski, F.; Ferreira-Dasilva, C.T.; Mulinari, F.; Pires-Alves, M.; Carlini, C.R. Insecticidal effects of canatoxin on the cotton stainer bug Dysdercus peruvianus (Hemiptera: Pyrrhocoridae). Toxicon 2005, 45, 753–760. [Google Scholar] [CrossRef]
- Down, R.E.; Fitches, E.C.; Wiles, D.P.; Corti, P.; Bell, H.A.; Gatehouse, J.A.; Edwards, J.P. Insecticidal spider venom toxin fused to snowdrop lectin is toxic to the peach-potato aphid, Myzus persicae (Hemiptera: Aphididae) and the rice brown planthopper, Nilaparvata lugens (Hemiptera: Delphacidae). Pest Manag. Sci. 2006, 62, 77–85. [Google Scholar] [CrossRef]
- Hinchliffe, G.; Bown, D.P.; Gatehouse, J.A.; Fitches, E. Insecticidal activity of recombinant avidin produced in yeast. J. Insect Physiol. 2010, 56, 629–639. [Google Scholar] [CrossRef]
- Gressent, F.; Da Silva, P.; Eyraud, V.; Karaki, L.; Royer, C. Pea Albumin 1 subunit b (PA1b), a promising bioinsecticide of plant origin. Toxins 2011, 3, 1502–1517. [Google Scholar] [CrossRef]
- Gressent, F.; Duport, G.; Rahioui, I.; Pauchet, Y.; Bolland, P.; Specty, O.; Rahbe, Y. Biological activity and binding site characteristics of the PA1b Entomotoxin on insects from different orders. J. Insect Sci. 2007, 7, 1–10. [Google Scholar]
- Down, R.E.; Matthews, H.J.; Audsley, N. Oral activity of FMRFamide-related peptides on the pea aphid Acyrthosiphon pisum (Hemiptera: Aphididae) and degradation by enzymes from the aphid gut. Regul. Pept. 2011, 171, 11–18. [Google Scholar] [CrossRef]
- Nachman, R.J.; Hamshou, M.; Kaczmarek, K.; Zabrocki, J.; Smagghe, G. Biostable and PEG polymer-conjugated insect pyrokinin analogs demonstrate antifeedant activity and induce high mortality in the pea aphid Acyrthosiphon pisum (Hemiptera: Aphidae). Peptides 2012, 34, 266–273. [Google Scholar] [CrossRef]
- Nachman, R.J.; Mahdian, K.; Nassel, D.R.; Isaac, R.E.; Pryor, N.; Smagghe, G. Biostable multi-Aib analogs of tachykinin-related peptides demonstrate potent oral aphicidal activity in the pea aphid Acyrthosiphon pisum (Hemiptera: Aphidae). Peptides 2011, 32, 587–594. [Google Scholar] [CrossRef]
- de Geyter, E.; Smagghe, G.; Rahbe, Y.; Geelen, D. Triterpene saponins of Quillaja saponaria show strong aphicidal and deterrent activity against the pea aphid Acyrthosiphon pisum. Pest Manag. Sci. 2012, 68, 164–169. [Google Scholar] [CrossRef]
- Huvenne, H.; Smagghe, G. Mechanisms of dsRNA uptake in insects and potential of RNAi for pest control: A review. J Insect Physiol. 2010, 56, 227–235. [Google Scholar] [CrossRef]
- Price, D.R.; Gatehouse, J.A. RNAi-mediated crop protection against insects. Trends Biotechnol. 2008, 26, 393–400. [Google Scholar] [CrossRef]
- Baum, J.A.; Bogaert, T.; Clinton, W.; Heck, G.R.; Feldmann, P.; Ilagan, O.; Johnson, S.; Plaetinck, G.; Munyikwa, T.; Pleau, M.; et al. Control of coleopteran insect pests through RNA interference. Nat. Biotechnol. 2007, 25, 1322–1326. [Google Scholar]
- Terenius, O.; Papanicolaou, A.; Garbutt, J.S.; Eleftherianos, I.; Huvenne, H.; Kanginakudru, S.; Albrechtsen, M.; An, C.; Aymeric, J.L.; Barthel, A.; et al. RNA interference in Lepidoptera: An overview of successful and unsuccessful studies and implications for experimental design. J. Insect Physiol. 2011, 57, 231–245. [Google Scholar] [CrossRef]
- Pitino, M.; Coleman, A.D.; Maffei, M.E.; Ridout, C.J.; Hogenhout, S.A. Silencing of aphid genes by dsRNA feeding from plants. PLoS One 2011, 6, e25709. [Google Scholar]
- Jaubert-Possamai, S.; Le Trionnaire, G.; Bonhomme, J.; Christophides, G.K.; Rispe, C.; Tagu, D. Gene knockdown by RNAi in the pea aphid Acyrthosiphon pisum. BMC Biotechnol. 2007, 7, 63. [Google Scholar] [CrossRef]
- Zha, W.; Peng, X.; Chen, R.; Du, B.; Zhu, L.; He, G. Knockdown of Midgut Genes by dsRNA-Transgenic Plant-Mediated RNA Interference in the Hemipteran Insect Nilaparvata lugens. PLoS One 2011, 6, e20504. [Google Scholar]
- Upadhyay, S.K.; Chandrashekar, K.; Thakur, N.; Verma, P.C.; Borgio, J.F.; Singh, P.K.; Tuli, R. RNA interference for the control of whiteflies (Bemisia tabaci) by oral route. J. Biosci. 2011, 36, 153–161. [Google Scholar] [CrossRef]
- Mutti, N.S.; Louis, J.; Pappan, L.K.; Pappan, K.; Begum, K.; Chen, M.-S.; Park, Y.; Dittmer, N.; Marshall, J.; Reese, J.C.; Reeck, G.R. A protein from the salivary glands of the pea aphid, Acyrthosiphon pisum, is essential in feeding on a host plant. Proc. Natl. Acad. Sci. USA 2008, 105, 9965–9969. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Chougule, N.P.; Bonning, B.C. Toxins for Transgenic Resistance to Hemipteran Pests. Toxins 2012, 4, 405-429. https://doi.org/10.3390/toxins4060405
Chougule NP, Bonning BC. Toxins for Transgenic Resistance to Hemipteran Pests. Toxins. 2012; 4(6):405-429. https://doi.org/10.3390/toxins4060405
Chicago/Turabian StyleChougule, Nanasaheb P., and Bryony C. Bonning. 2012. "Toxins for Transgenic Resistance to Hemipteran Pests" Toxins 4, no. 6: 405-429. https://doi.org/10.3390/toxins4060405
APA StyleChougule, N. P., & Bonning, B. C. (2012). Toxins for Transgenic Resistance to Hemipteran Pests. Toxins, 4(6), 405-429. https://doi.org/10.3390/toxins4060405





