Cure and Curse: E. coli Heat-Stable Enterotoxin and Its Receptor Guanylyl Cyclase C
Abstract
:1. Introduction
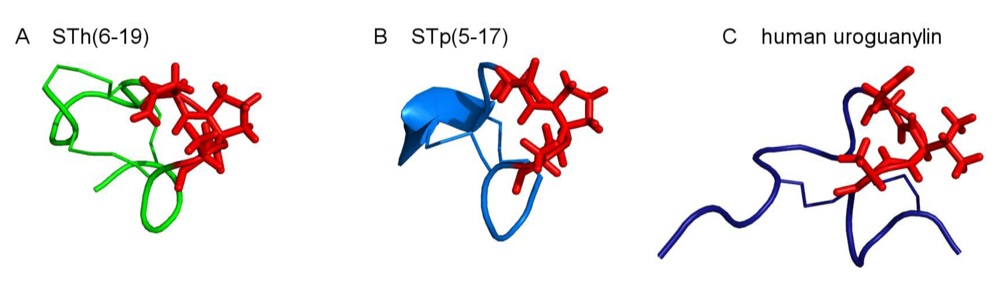
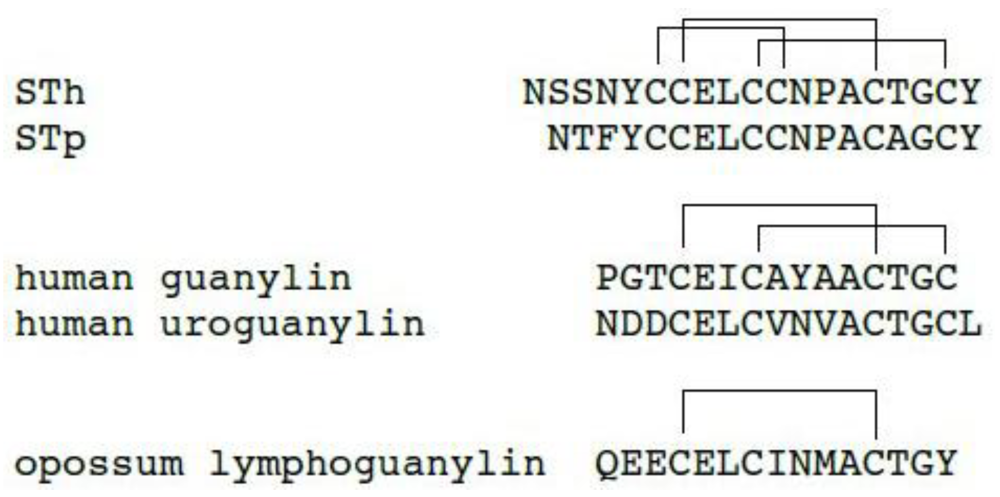
2. Structure of ST Peptides
3. The Heat-Stable Enterotoxin Receptor: Intestinal GC-C
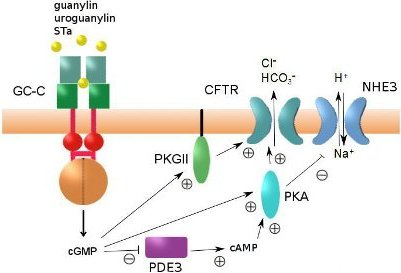
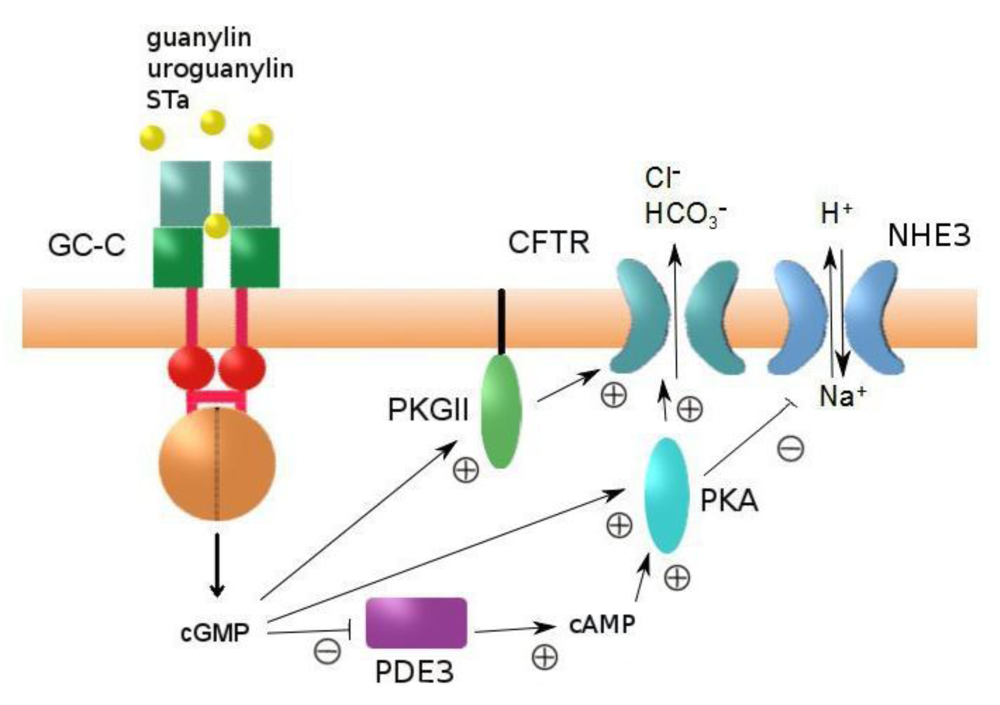
4. Regulation of Intestinal Fluid Secretion and STa-Induced Diarrhea
5. Involvement of GC-C in Colorectal Cancer
6. Outlook
Acknowledgments
References
- World Health Organization. Future Directions for Research on Enterotoxigenic Escherichia coli Vaccines for Developing Countries. Wkly. Epidemiol. Rec. 2006, 81, 97–104. [Google Scholar] [PubMed]
- Turner, S.M.; Chaudhuri, R.R.; Jiang, Z.D.; DuPont, H.; Gyles, C.; Penn, C.W.; Pallen, M.J.; Henderson, I.R. Phylogenetic Comparisons Reveal Multiple Acquisitions of the Toxin Genes by Enterotoxigenic Escherichia coli Strains of Different Evolutionary Lineages. J. Clin. Microbiol. 2006, 44, 4528–4536. [Google Scholar]
- Sack, D.A.; McLaughlin, J.C.; Sack, R.B.; Orskov, F.; Orskov, I. Enterotoxigenic Escherichia coli Isolated from Patients at a Hospital in Dacca. J. Infect. Dis. 1977, 135, 275–280. [Google Scholar]
- Spangler, B.D. Structure and Function of Cholera Toxin and the Related Escherichia coli Heat-Labile Enterotoxin. Microbiol. Rev. 1992, 56, 622–647. [Google Scholar]
- Sack, R.B. Human Diarrheal Disease Caused by Enterotoxigenic Escherichia coli. Annu. Rev. Microbiol. 1975, 29, 333–353. [Google Scholar]
- Dreyfus, L.A.; Harville, B.; Howard, D.E.; Shaban, R.; Beatty, D.M.; Morris, S.J. Calcium Influx Mediated by the Escherichia coli Heat-Stable Enterotoxin B (STB). Proc. Natl. Acad. Sci. USA 1993, 90, 3202–3206. [Google Scholar]
- Moseley, S.L.; Samadpour-Motalebi, M.; Falkow, S. Plasmid Association and Nucleotide Sequence Relationships of Two Genes Encoding Heat-Stable Enterotoxin Production in Escherichia coli H-10407. J. Bacteriol. 1983, 156, 441–443. [Google Scholar]
- So, M.; McCarthy, B.J. Nucleotide Sequence of the Bacterial Transposon Tn1681 Encoding a Heat-Stable (ST) Toxin and Its Identification in Enterotoxigenic Escherichia coli Strains. Proc. Natl. Acad. Sci. USA 1980, 77, 4011–4015. [Google Scholar]
- Dallas, W.S. The Heat-Stable Toxin I Gene from Escherichia coli 18D. J. Bacteriol. 1990, 172, 5490–5493. [Google Scholar]
- Klipstein, F.A.; Engert, R.F.; Houghten, R.A. Immunological Properties of Purified Klebsiella pneumoniae Heat-Stable Enterotoxin. Infect. Immun. 1983, 42, 838–841. [Google Scholar]
- Takao, T.; Tominaga, N.; Shimonishi, Y.; Hara, S.; Inoue, T.; Miyama, A. Primary Structure of Heat-Stable Enterotoxin Produced by Yersinia Enterocolitica. Biochem. Biophys. Res. Commun. 1984, 125, 845–851. [Google Scholar]
- Guarino, A.; Capano, G.; Malamisura, B.; Alessio, M.; Guandalini, S.; Rubino, A. Production of Escherichia coli STa-Like Heat-Stable Enterotoxin by Citrobacter freundii Isolated from Humans. J. Clin. Microbiol. 1987, 25, 110–114. [Google Scholar]
- Takeda, T.; Peina, Y.; Ogawa, A.; Dohi, S.; Abe, H.; Nair, G.B.; Pal, S.C. Detection of Heat-Stable Enterotoxin in a Cholera Toxin Gene-Positive Strain of Vibrio cholerae O1. FEMS Microbiol. Lett. 1991, 64, 23–27. [Google Scholar]
- Takao, T.; Shimonishi, Y.; Kobayashi, M.; Nishimura, O.; Arita, M.; Takeda, T.; Honda, T.; Miwatani, T. Amino Acid Sequence of Heat-Stable Enterotoxin Produced by Vibrio cholerae Non-01. FEBS Lett. 1985, 193, 250–254. [Google Scholar]
- Arita, M.; Takeda, T.; Honda, T.; Miwatani, T. Purification and Characterization of Vibrio cholerae Non-O1 Heat-Stable Enterotoxin. Infect. Immun. 1986, 52, 45–49. [Google Scholar]
- Huang, X.; Yoshino, K.; Nakao, H.; Takeda, T. Nucleotide Sequence of a Gene Encoding the Novel Yersinia Enterocolitica Heat-Stable Enterotoxin that Includes a Pro-Region-Like Sequence in Its Mature Toxin Molecule. Microb. Pathog. 1997, 22, 89–97. [Google Scholar]
- Savarino, S.J.; Fasano, A.; Robertson, D.C.; Levine, M.M. Enteroaggregative Escherichia coli Elaborate a Heat-Stable Enterotoxin Demonstrable in an in Vitro Rabbit Intestinal Model. J. Clin. Invest. 1991, 87, 1450–1455. [Google Scholar]
- Savarino, S.J.; Fasano, A.; Watson, J.; Martin, B.M.; Levine, M.M.; Guandalini, S.; Guerry, P. Enteroaggregative Escherichia coli Heat-Stable Enterotoxin 1 Represents another Subfamily of E. coli Heat-Stable Toxin. Proc. Natl. Acad. Sci. USA 1993, 90, 3093–3097. [Google Scholar]
- Paton, J.C.; Paton, A.W. Pathogenesis and Diagnosis of Shiga Toxin-Producing Escherichia coli Infections. Clin. Microbiol. Rev. 1998, 11, 450–479. [Google Scholar]
- Yoshimura, S.; Ikemura, H.; Watanabe, H.; Aimoto, S.; Shimonishi, Y.; Hara, S.; Takeda, T.; Miwatani, T.; Takeda, Y. Essential Structure for Full Enterotoxigenic Activity of Heat-Stable Enterotoxin Produced by Enterotoxigenic Escherichia coli. FEBS Lett. 1985, 181, 138–142. [Google Scholar]
- Currie, M.G.; Fok, K.F.; Kato, J.; Moore, R.J.; Hamra, F.K.; Duffin, K.L.; Smith, C.E. Guanylin: An Endogenous Activator of Intestinal Guanylate Cyclase. Proc. Natl. Acad. Sci. USA 1992, 89, 947–951. [Google Scholar]
- Hamra, F.K.; Forte, L.R.; Eber, S.L.; Pidhorodeckyj, N.V.; Krause, W.J.; Freeman, R.H.; Chin, D.T.; Tompkins, J.A.; Fok, K.F.; Smith, C.E. Uroguanylin: Structure and Activity of a Second Endogenous Peptide that Stimulates Intestinal Guanylate Cyclase. Proc. Natl. Acad. Sci. USA 1993, 90, 10464–10468. [Google Scholar]
- Forte, L.R.; Eber, S.L.; Fan, X.; London, R.M.; Wang, Y.; Rowland, L.M.; Chin, D.T.; Freeman, R.H.; Krause, W.J. Lymphoguanylin: Cloning and Characterization of a Unique Member of the Guanylin Peptide Family. Endocrinology 1999, 140, 1800–1806. [Google Scholar]
- Shimonishi, Y.; Hidaka, Y.; Koizumi, M.; Hane, M.; Aimoto, S.; Takeda, T.; Miwatani, T.; Takeda, Y. Mode of Disulfide Bond Formation of a Heat-Stable Enterotoxin (STh) Produced by a Human Strain of Enterotoxigenic Escherichia coli. FEBS Lett. 1987, 215, 165–170. [Google Scholar]
- Gariepy, J.; Judd, A.K.; Schoolnik, G.K. Importance of Disulfide Bridges in the Structure and Activity of Escherichia coli Enterotoxin ST1b. Proc. Natl. Acad. Sci. USA 1987, 84, 8907–8911. [Google Scholar]
- Yamasaki, S.; Hidaka, Y.; Hideaki, I.; Takeda, Y.; Shimonishi, Y. Structural Requirements for the Spatial Structure and Toxicity of Heat-Stable Enterotoxin (STh) of Enterotoxigenic Escherichia Coli. Bull. Chem. Soc. Jpn. 1988, 61, 1701–1706. [Google Scholar]
- Skelton, N.J.; Garcia, K.C.; Goeddel, D.V.; Quan, C.; Burnier, J.P. Determination of the Solution Structure of the Peptide Hormone Guanylin: Observation of a Novel Form of Topological Stereoisomerism. Biochemistry 1994, 33, 13581–13592. [Google Scholar]
- Marx, U.C.; Klodt, J.; Meyer, M.; Gerlach, H.; Rosch, P.; Forssmann, W.G.; Adermann, K. One Peptide, Two Topologies: Structure and Interconversion Dynamics of Human Uroguanylin Isomers. J. Pept. Res. 1998, 52, 229–240. [Google Scholar]
- Klodt, J.; Kuhn, M.; Marx, U.C.; Martin, S.; Rosch, P.; Forssmann, W.G.; Adermann, K. Synthesis, Biological Activity and Isomerism of Guanylate Cyclase C-Activating Peptides Guanylin and Uroguanylin. J. Pept. Res. 1997, 50, 222–230. [Google Scholar]
- Schulz, A.; Escher, S.; Marx, U.C.; Meyer, M.; Rosch, P.; Forssmann, W.G.; Adermann, K. Carboxy-Terminal Extension Stabilizes the Topological Stereoisomers of Guanylin. J. Pept. Res. 1998, 52, 518–525. [Google Scholar]
- Schulz, A.; Marx, U.C.; Tidten, N.; Lauber, T.; Hidaka, Y.; Adermann, K. Side Chain Contributions to the Interconversion of the Topological Isomers of Guanylin-Like Peptides. J. Pept. Sci. 2005, 11, 319–330. [Google Scholar]
- Okamoto, K.; Takahara, M. Synthesis of Escherichia coli Heat-Stable Enterotoxin STp as a Pre-Pro form and Role of the Pro Sequence in Secretion. J. Bacteriol. 1990, 172, 5260–5265. [Google Scholar]
- Rasheed, J.K.; Guzman-Verduzco, L.M.; Kupersztoch, Y.M. Two Precursors of the Heat-Stable Enterotoxin of Escherichia coli: Evidence of Extracellular Processing. Mol. Microbiol. 1990, 4, 265–273. [Google Scholar]
- Pugsley, A.P. The Complete General Secretory Pathway in Gram-Negative Bacteria. Microbiol. Rev. 1993, 57, 50–108. [Google Scholar]
- Yamanaka, H.; Fuke, Y.; Hitotsubashi, S.; Fujii, Y.; Okamoto, K. Functional Properties of Pro Region of Escherichia coli Heat-Stable Enterotoxin. Microbiol. Immunol. 1993, 37, 195–205. [Google Scholar]
- Yamanaka, H.; Kameyama, M.; Baba, T.; Fujii, Y.; Okamoto, K. Maturation Pathway of Escherichia coli Heat-Stable Enterotoxin I: Requirement of DsbA for Disulfide Bond Formation. J. Bacteriol. 1994, 176, 2906–2913. [Google Scholar]
- Yamanaka, H.; Nomura, T.; Fujii, Y.; Okamoto, K. Extracellular Secretion of Escherichia coli Heat-Stable Enterotoxin I across the Outer Membrane. J. Bacteriol. 1997, 179, 3383–3390. [Google Scholar]
- Yang, Y.; Gao, Z.; Guzman-Verduzco, L.M.; Tachias, K.; Kupersztoch, Y.M. Secretion of the STA3 Heat-Stable Enterotoxin of Escherichia coli: Extracellular Delivery of Pro-STA Is Accomplished by either Pro or STA. Mol. Microbiol. 1992, 6, 3521–3529. [Google Scholar]
- Batisson, I.; der Vartanian, M. Extracellular DsbA-Insensitive Folding of Escherichia coli Heat-Stable Enterotoxin STa in Vitro. J. Biol. Chem. 2000, 275, 10582–10589. [Google Scholar]
- Batisson, I.; der Vartanian, M.; Gaillard-Martinie, B.; Contrepois, M. Full Capacity of Recombinant Escherichia coli Heat-Stable Enterotoxin Fusion Proteins for Extracellular Secretion, Antigenicity, Disulfide Bond Formation, and Activity. Infect. Immun. 2000, 68, 4064–4074. [Google Scholar]
- Matecko, I.; Burmann, B.M.; Schweimer, K.; Kalbacher, H.; Einsiedel, J.; Gmeiner, P.; Rösch, P. Structural Characterization of the E. Coli Heat Stable Enterotoxin STh. Open Spectrosc. J. 2008, 2, 34–39. [Google Scholar]
- Ozaki, H.; Sato, T.; Kubota, H.; Hata, Y.; Katsube, Y.; Shimonishi, Y. Molecular Structure of the Toxin Domain of Heat-Stable Enterotoxin Produced by a Pathogenic Strain of Escherichia coli. A Putative Binding Site for a Binding Protein on Rat Intestinal Epithelial Cell Membranes. J. Biol. Chem. 1991, 266, 5934–5941. [Google Scholar] [PubMed]
- Sato, T.; Ozaki, H.; Hata, Y.; Kitagawa, Y.; Katsube, Y.; Shimonishi, Y. Structural Characteristics for Biological Activity of Heat-Stable Enterotoxin Produced by Enterotoxigenic Escherichia coli: X-ray Crystallography of Weakly Toxic and Nontoxic Analogs. Biochemistry 1994, 33, 8641–8650. [Google Scholar]
- Sato, T.; Shimonishi, Y. Structural Features of Escherichia coli Heat-Stable Enterotoxin that Activates Membrane-Associated Guanylyl Cyclase. J. Pept. Res. 2004, 63, 200–206. [Google Scholar]
- Richardson, J.S. The Anatomy and Taxonomy of Protein Structure. Adv. Protein Chem. 1981, 34, 167–339. [Google Scholar]
- Carpick, B.W.; Gariepy, J. Structural Characterization of Functionally Important Regions of the Escherichia coli Heat-Stable Enterotoxin STIb. Biochemistry 1991, 30, 4803–4809. [Google Scholar]
- Waldman, S.A.; O'Hanley, P. Influence of a Glycine or Proline Substitution on the Functional Properties of a 14-Amino-Acid Analog of Escherichia coli Heat-Stable Enterotoxin. Infect. Immun. 1989, 57, 2420–2424. [Google Scholar]
- Wolfe, H.R.; Waldman, S.A. A Comparative Molecular Field Analysis (COMFA) of the Structural Determinants of Heat-Stable Enterotoxins Mediating Activation of Guanylyl Cyclase C. J. Med. Chem. 2002, 45, 1731–1734. [Google Scholar]
- Gariepy, J.; Lane, A.; Frayman, F.; Wilbur, D.; Robien, W.; Schoolnik, G.K.; Jardetzky, O. Structure of the Toxic Domain of the Escherichia coli Heat-Stable Enterotoxin ST I. Biochemistry 1986, 25, 7854–7866. [Google Scholar]
- Schulz, S.; Green, C.K.; Yuen, P.S.; Garbers, D.L. Guanylyl Cyclase Is a Heat-Stable Enterotoxin Receptor. Cell 1990, 63, 941–948. [Google Scholar]
- Vaandrager, A.B.; Schulz, S.; de Jonge, H.R.; Garbers, D.L. Guanylyl Cyclase C Is an N-Linked Glycoprotein Receptor that Accounts for Multiple Heat-Stable Enterotoxin-Binding Proteins in the Intestine. J. Biol. Chem. 1993, 268, 2174–2179. [Google Scholar]
- de Jonge, H.R. Properties of Guanylate Cyclase and Levels of Cyclic GMP in Rat Small Intestinal Villous and Crypt Cells. FEBS Lett. 1975, 55, 143–152. [Google Scholar]
- Basu, N.; Arshad, N.; Visweswariah, S.S. Receptor Guanylyl Cyclase C (GC-C): Regulation and Signal Transduction. Mol. Cell. Biochem. 2010, 334, 67–80. [Google Scholar]
- Di Guglielmo, M.D.; Park, J.; Schulz, S.; Waldman, S.A. Nucleotide Requirements for CDX2 Binding to the Cis Promoter Element Mediating Intestine-Specific Expression of Guanylyl Cyclase C. FEBS Lett. 2001, 507, 128–132. [Google Scholar]
- Swenson, E.S.; Mann, E.A.; Jump, M.L.; Giannella, R.A. Hepatocyte Nuclear Factor-4 Regulates Intestinal Expression of the guanylin/heat-Stable Toxin Receptor. Am. J. Physiol. 1999, 276, G728–G736. [Google Scholar]
- Li, P.; Lin, J.E.; Chervoneva, I.; Schulz, S.; Waldman, S.A.; Pitari, G.M. Homeostatic Control of the Crypt-Villus Axis by the Bacterial Enterotoxin Receptor Guanylyl Cyclase C Restricts the Proliferating Compartment in Intestine. Am. J. Pathol. 2007, 171, 1847–1858. [Google Scholar]
- de Sauvage, F.J.; Camerato, T.R.; Goeddel, D.V. Primary Structure and Functional Expression of the Human Receptor for Escherichia coli Heat-Stable Enterotoxin. J. Biol. Chem. 1991, 266, 17912–17918. [Google Scholar]
- Vaandrager, A.B. Structure and Function of the Heat-Stable Enterotoxin receptor/guanylyl Cyclase C. Mol. Cell. Biochem. 2002, 230, 73–83. [Google Scholar]
- Sharma, R.K. Membrane Guanylate Cyclase Is a Beautiful Signal Transduction Machine: Overview. Mol. Cell. Biochem. 2010, 334, 3–36. [Google Scholar]
- van den Akker, F.; Zhang, X.; Miyagi, M.; Huo, X.; Misono, K.S.; Yee, V.C. Structure of the Dimerized Hormone-Binding Domain of a Guanylyl-Cyclase-Coupled Receptor. Nature 2000, 406, 101–104. [Google Scholar]
- Hasegawa, M.; Matsumoto-Ishikawa, Y.; Hijikata, A.; Hidaka, Y.; Go, M.; Shimonishi, Y. Disulfide Linkages and a Three-Dimensional Structure Model of the Extracellular Ligand-Binding Domain of Guanylyl Cyclase C. Protein J. 2005, 24, 315–325. [Google Scholar]
- Lauber, T.; Tidten, N.; Matecko, I.; Zeeb, M.; Rosch, P.; Marx, U.C. Design and Characterization of a Soluble Fragment of the Extracellular Ligand-Binding Domain of the Peptide Hormone Receptor Guanylyl Cyclase-C. Protein Eng. Des. Sel. 2009, 22, 1–7. [Google Scholar]
- Hasegawa, M.; Hidaka, Y.; Matsumoto, Y.; Sanni, T.; Shimonishi, Y. Determination of the Binding Site on the Extracellular Domain of Guanylyl Cyclase C to Heat-Stable Enterotoxin. J. Biol. Chem. 1999, 274, 31713–31718. [Google Scholar]
- Hasegawa, M.; Shimonishi, Y. Recognition and Signal Transduction Mechanism of Escherichia coli Heat-Stable Enterotoxin and Its Receptor, Guanylate Cyclase C. J. Pept. Res. 2005, 65, 261–271. [Google Scholar]
- Biswas, K.H.; Shenoy, A.R.; Dutta, A.; Visweswariah, S.S. The Evolution of Guanylyl Cyclases as Multidomain Proteins: Conserved Features of Kinase-Cyclase Domain Fusions. J. Mol. Evol. 2009, 68, 587–602. [Google Scholar]
- Koller, K.J.; de Sauvage, F.J.; Lowe, D.G.; Goeddel, D.V. Conservation of the Kinaselike Regulatory Domain Is Essential for Activation of the Natriuretic Peptide Receptor Guanylyl Cyclases. Mol. Cell. Biol. 1992, 12, 2581–2590. [Google Scholar]
- Hanks, S.K.; Hunter, T. Protein Kinases 6. The Eukaryotic Protein Kinase Superfamily: Kinase (Catalytic) Domain Structure and Classification. FASEB J. 1995, 9, 576–596. [Google Scholar] [PubMed]
- Hirayama, T.; Wada, A.; Iwata, N.; Takasaki, S.; Shimonishi, Y.; Takeda, Y. Glycoprotein Receptors for a Heat-Stable Enterotoxin (STh) Produced by Enterotoxigenic Escherichia coli. Infect. Immun. 1992, 60, 4213–4220. [Google Scholar]
- Vaandrager, A.B.; Schulz, S.; de Jonge, H.R.; Garbers, D.L. Guanylyl Cyclase C Is an N-Linked Glycoprotein Receptor that Accounts for Multiple Heat-Stable Enterotoxin-Binding Proteins in the Intestine. J. Biol. Chem. 1993, 268, 2174–2179. [Google Scholar]
- Ghanekar, Y.; Chandrashaker, A.; Tatu, U.; Visweswariah, S.S. Glycosylation of the Receptor Guanylate Cyclase C: Role in Ligand Binding and Catalytic Activity. Biochem. J. 2004, 379, 653–663. [Google Scholar]
- Hasegawa, M.; Kawano, Y.; Matsumoto, Y.; Hidaka, Y.; Fujii, J.; Taniguchi, N.; Wada, A.; Hirayama, T.; Shimonishi, Y. Expression and Characterization of the Extracellular Domain of Guanylyl Cyclase C from a Baculovirus and Sf21 Insect Cells. Protein Expr. Purif. 1999, 15, 271–281. [Google Scholar]
- Hasegawa, M.; Hidaka, Y.; Wada, A.; Hirayama, T.; Shimonishi, Y. The Relevance of N-Linked Glycosylation to the Binding of a Ligand to Guanylate Cyclase C. Eur. J. Biochem. 1999, 263, 338–346. [Google Scholar]
- Vaandrager, A.B.; van der Wiel, E.; Hom, M.L.; Luthjens, L.H.; de Jonge, H.R. Heat-Stable Enterotoxin Receptor/Guanylyl Cyclase C Is an Oligomer Consisting of Functionally Distinct Subunits, which Are Non-Covalently Linked in the Intestine. J. Biol. Chem. 1994, 269, 16409–16415. [Google Scholar]
- Vijayachandra, K.; Guruprasad, M.; Bhandari, R.; Manjunath, U.H.; Somesh, B.P.; Srinivasan, N.; Suguna, K.; Visweswariah, S.S. Biochemical Characterization of the Intracellular Domain of the Human Guanylyl Cyclase C Receptor Provides Evidence for a Catalytically Active Homotrimer. Biochemistry 2000, 39, 16075–16083. [Google Scholar]
- He, X.; Chow, D.; Martick, M.M.; Garcia, K.C. Allosteric Activation of a Spring-Loaded Natriuretic Peptide Receptor Dimer by Hormone. Science 2001, 293, 1657–1662. [Google Scholar]
- Ostedgaard, L.S.; Baldursson, O.; Welsh, M.J. Regulation of the Cystic Fibrosis Transmembrane Conductance Regulator Cl− Channel by Its R Domain. J. Biol. Chem. 2001, 276, 7689–7692. [Google Scholar]
- Vaandrager, A.B.; Tilly, B.C.; Smolenski, A.; Schneider-Rasp, S.; Bot, A.G.; Edixhoven, M.; Scholte, B.J.; Jarchau, T.; Walter, U.; Lohmann, S.M.; Poller, W.C.; de Jonge, H.R. CGMP Stimulation of Cystic Fibrosis Transmembrane Conductance Regulator Cl− Channels Co-Expressed with cGMP-Dependent Protein Kinase Type II but Not Type Ibeta. J. Biol. Chem. 1997, 272, 4195–4200. [Google Scholar]
- Vaandrager, A.B.; Smolenski, A.; Tilly, B.C.; Houtsmuller, A.B.; Ehlert, E.M.; Bot, A.G.; Edixhoven, M.; Boomaars, W.E.; Lohmann, S.M.; de Jonge, H.R. Membrane Targeting of cGMP-Dependent Protein Kinase Is Required for Cystic Fibrosis Transmembrane Conductance Regulator Cl− Channel Activation. Proc. Natl. Acad. Sci. USA 1998, 95, 1466–1471. [Google Scholar]
- Chao, A.C.; de Sauvage, F.J.; Dong, Y.J.; Wagner, J.A.; Goeddel, D.V.; Gardner, P. Activation of Intestinal CFTR Cl− Channel by Heat-Stable Enterotoxin and Guanylin via cAMP-Dependent Protein Kinase. EMBO J. 1994, 13, 1065–1072. [Google Scholar]
- Tousson, A.; Fuller, C.M.; Benos, D.J. Apical Recruitment of CFTR in T-84 Cells Is Dependent on cAMP and Microtubules but Not Ca2+ or Microfilaments. J. Cell Sci. 1996, 109, 1325–1334. [Google Scholar]
- Kleizen, B.; Braakman, I.; de Jonge, H.R. Regulated Trafficking of the CFTR Chloride Channel. Eur. J. Cell Biol. 2000, 79, 544–556. [Google Scholar]
- Golin-Bisello, F.; Bradbury, N.; Ameen, N. STa and cGMP Stimulate CFTR Translocation to the Surface of Villus Enterocytes in Rat Jejunum and Is Regulated by Protein Kinase G. Am. J. Physiol. Cell Physiol. 2005, 289, C708–C716. [Google Scholar]
- He, P.; Yun, C.C. Mechanisms of the Regulation of the Intestinal Na+/H+ Exchanger NHE3. J. Biomed. Biotechnol. 2010. [Google Scholar]
- Lucas, M.L.; Thom, M.M.; Bradley, J.M.; O'Reilly, N.F.; McIlvenny, T.J.; Nelson, Y.B. Escherichia coli Heat Stable (STa) Enterotoxin and the Upper Small Intestine: Lack of Evidence in Vivo for Net Fluid Secretion. J. Membr. Biol. 2005, 206, 29–42. [Google Scholar]
- Lucas, M.L. A Reconsideration of the Evidence for Escherichia coli STa (Heat Stable) Enterotoxin-Driven Fluid Secretion: A New View of STa Action and a New Paradigm for Fluid Absorption. J. Appl. Microbiol. 2001, 90, 7–26. [Google Scholar]
- Bryant, A.P.; Busby, R.W.; Bartolini, W.P.; Cordero, E.A.; Hannig, G.; Kessler, M.M.; Pierce, C.M.; Solinga, R.M.; Tobin, J.V.; Mahajan-Miklos, S.; et al. Linaclotide Is a Potent and Selective Guanylate Cyclase C Agonist that Elicits Pharmacological Effects Locally in the Gastrointestinal Tract. Life Sci. 2010, 86, 760–765. [Google Scholar]
- Eutamene, H.; Bradesi, S.; Larauche, M.; Theodorou, V.; Beaufrand, C.; Ohning, G.; Fioramonti, J.; Cohen, M.; Bryant, A.P.; Kurtz, C.; et al. Guanylate Cyclase C-Mediated Antinociceptive Effects of Linaclotide in Rodent Models of Visceral Pain. Neurogastroenterol. Motil. 2010, 22, 312–e84. [Google Scholar]
- Sellers, Z.M.; Childs, D.; Chow, J.Y.; Smith, A.J.; Hogan, D.L.; Isenberg, J.I.; Dong, H.; Barrett, K.E.; Pratha, V.S. Heat-Stable Enterotoxin of Escherichia coli Stimulates a Non-CFTR-Mediated Duodenal Bicarbonate Secretory Pathway. Am. J. Physiol. Gastrointest. Liver Physiol. 2005, 288, G654–G663. [Google Scholar]
- Sellers, Z.M.; Mann, E.; Smith, A.; Ko, K.H.; Giannella, R.; Cohen, M.B.; Barrett, K.E.; Dong, H. Heat-Stable Enterotoxin of Escherichia coli (STa) Can Stimulate Duodenal HCO3(-) Secretion via a Novel GC-C- and CFTR-Independent Pathway. FASEB J. 2008, 22, 1306–1316. [Google Scholar]
- Hugues, M.; Crane, M.; Hakki, S.; O'Hanley, P.; Waldman, S.A. Identification and Characterization of a New Family of High-Affinity Receptors for Escherichia coli Heat-Stable Enterotoxin in Rat Intestinal Membranes. Biochemistry 1991, 30, 10738–10745. [Google Scholar]
- Hakki, S.; Robertson, D.C.; Waldman, S.A. A 56 kDa Binding Protein for Escherichia coli Heat-Stable Enterotoxin Isolated from the Cytoskeleton of Rat Intestinal Membrane Does Not Possess Guanylate Cyclase Activity. Biochim. Biophys. Acta 1993, 1152, 1–8. [Google Scholar]
- Mann, E.A.; Jump, M.L.; Wu, J.; Yee, E.; Giannella, R.A. Mice Lacking the Guanylyl Cyclase C Receptor Are Resistant to STa-Induced Intestinal Secretion. Biochem. Biophys. Res. Commun. 1997, 239, 463–466. [Google Scholar]
- Schulz, S.; Lopez, M.J.; Kuhn, M.; Garbers, D.L. Disruption of the Guanylyl Cyclase-C Gene Leads to a Paradoxical Phenotype of Viable but Heat-Stable Enterotoxin-Resistant Mice. J. Clin. Invest. 1997, 100, 1590–1595. [Google Scholar]
- Sindice, A.; Basoglu, C.; Cerci, A.; Hirsch, J.R.; Potthast, R.; Kuhn, M.; Ghanekar, Y.; Visweswariah, S.S.; Schlatter, E. Guanylin, Uroguanylin, and Heat-Stable Euterotoxin Activate Guanylate Cyclase C and/or a Pertussis Toxin-Sensitive G Protein in Human Proximal Tubule Cells. J. Biol. Chem. 2002, 277, 17758–17764. [Google Scholar]
- Carrithers, S.L.; Ott, C.E.; Hill, M.J.; Johnson, B.R.; Cai, W.; Chang, J.J.; Shah, R.G.; Sun, C.; Mann, E.A.; Fonteles, M.C.; et al. Guanylin and Uroguanylin Induce Natriuresis in Mice Lacking Guanylyl Cyclase-C Receptor. Kidney Int. 2004, 65, 40–53. [Google Scholar]
- Steinbrecher, K.A.; Tuohy, T.M.; Goss, K.H.; Scott, M.C.; Witte, D.P.; Groden, J.; Cohen, M.B. Expression of Guanylin Is Downregulated in Mouse and Human Intestinal Adenomas. Biochem. Biophys. Res. Commun. 2000, 273, 225–230. [Google Scholar]
- Carrithers, S.L.; Parkinson, S.J.; Goldstein, S.; Park, P.; Robertson, D.C.; Waldman, S.A. Escherichia coli Heat-Stable Toxin Receptors in Human Colonic Tumors. Gastroenterology 1994, 107, 1653–1661. [Google Scholar]
- Carrithers, S.L.; Barber, M.T.; Biswas, S.; Parkinson, S.J.; Park, P.K.; Goldstein, S.D.; Waldman, S.A. Guanylyl Cyclase C Is a Selective Marker for Metastatic Colorectal Tumors in Human Extraintestinal Tissues. Proc. Natl. Acad. Sci. USA 1996, 93, 14827–14832. [Google Scholar]
- Schulz, S.; Hyslop, T.; Haaf, J.; Bonaccorso, C.; Nielsen, K.; Witek, M.E.; Birbe, R.; Palazzo, J.; Weinberg, D.; Waldman, S.A. A Validated Quantitative Assay to Detect Occult Micrometastases by Reverse Transcriptase-Polymerase Chain Reaction of Guanylyl Cyclase C in Patients with Colorectal Cancer. Clin. Cancer Res. 2006, 12, 4545–4552. [Google Scholar]
- Waldman, S.A.; Hyslop, T.; Schulz, S.; Barkun, A.; Nielsen, K.; Haaf, J.; Bonaccorso, C.; Li, Y.; Weinberg, D.S. Association of GUCY2C Expression in Lymph Nodes with Time to Recurrence and Disease-Free Survival in pN0 Colorectal Cancer. JAMA 2009, 301, 745–752. [Google Scholar]
- Snook, A.E.; Stafford, B.J.; Li, P.; Tan, G.; Huang, L.; Birbe, R.; Schulz, S.; Schnell, M.J.; Thakur, M.; Rothstein, J.L.; et al. Guanylyl Cyclase C-Induced Immunotherapeutic Responses Opposing Tumor Metastases without Autoimmunity. J. Natl. Cancer Inst. 2008, 100, 950–961. [Google Scholar]
- Wolfe, H.R.; Mendizabal, M.; Lleong, E.; Cuthbertson, A.; Desai, V.; Pullan, S.; Fujii, D.K.; Morrison, M.; Pither, R.; Waldman, S.A. In Vivo Imaging of Human Colon Cancer Xenografts in Immunodeficient Mice using a Guanylyl Cyclase C—Specific Ligand. J. Nucl. Med. 2002, 43, 392–399. [Google Scholar]
- Giblin, M.F.; Gali, H.; Sieckman, G.L.; Owen, N.K.; Hoffman, T.J.; Forte, L.R.; Volkert, W.A. In Vitro and in Vivo Comparison of Human Escherichia coli Heat-Stable Peptide Analogues Incorporating the 111In-DOTA Group and Distinct Linker Moieties. Bioconjug. Chem. 2004, 15, 872–880. [Google Scholar]
- Giblin, M.F.; Sieckman, G.L.; Watkinson, L.D.; Daibes-Figueroa, S.; Hoffman, T.J.; Forte, L.R.; Volkert, W.A. Selective Targeting of E. coli Heat-Stable Enterotoxin Analogs to Human Colon Cancer Cells. Anticancer Res. 2006, 26, 3243–3251. [Google Scholar] [PubMed]
- Giblin, M.F.; Sieckman, G.L.; Shelton, T.D.; Hoffman, T.J.; Forte, L.R.; Volkert, W.A. In Vitro and in Vivo Evaluation of 177Lu- and 90Y-Labeled E. coli Heat-Stable Enterotoxin for Specific Targeting of Uroguanylin Receptors on Human Colon Cancers. Nucl. Med. Biol. 2006, 33, 481–488. [Google Scholar] [CrossRef] [PubMed]
- Giblin, M.F.; Gali, H.; Sieckman, G.L.; Owen, N.K.; Hoffman, T.J.; Volkert, W.A.; Forte, L.R. In Vitro and in Vivo Evaluation of 111In-Labeled E. coli Heat-Stable Enterotoxin Analogs for Specific Targeting of Human Breast Cancers. Breast Cancer Res. Treat. 2006, 98, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Tian, X.; Michal, A.M.; Li, P.; Wolfe, H.R.; Waldman, S.A.; Wickstrom, E. STa Peptide Analogs for Probing Guanylyl Cyclase C. Biopolymers 2008, 90, 713–723. [Google Scholar]
- Liu, D.; Overbey, D.; Watkinson, L.D.; Daibes-Figueroa, S.; Hoffman, T.J.; Forte, L.R.; Volkert, W.A.; Giblin, M.F. In Vivo Imaging of Human Colorectal Cancer using Radiolabeled Analogs of the Uroguanylin Peptide Hormone. Anticancer Res. 2009, 29, 3777–3783. [Google Scholar]
- Urbanski, R.; Carrithers, S.L.; Waldman, S.A. Internalization of E. coli ST Mediated by Guanylyl Cyclase C in T84 Human Colon Carcinoma Cells. Biochim. Biophys. Acta 1995, 1245, 29–36. [Google Scholar]
- Pitari, G.M.; Zingman, L.V.; Hodgson, D.M.; Alekseev, A.E.; Kazerounian, S.; Bienengraeber, M.; Hajnoczky, G.; Terzic, A.; Waldman, S.A. Bacterial Enterotoxins Are Associated with Resistance to Colon Cancer. Proc. Natl. Acad. Sci. USA 2003, 100, 2695–2699. [Google Scholar]
- Parkin, D.M.; Bray, F.; Ferlay, J.; Pisani, P. Global Cancer Statistics, 2002. CA Cancer J. Clin. 2005, 55, 74–108. [Google Scholar]
- Pitari, G.M.; Di Guglielmo, M.D.; Park, J.; Schulz, S.; Waldman, S.A. Guanylyl Cyclase C Agonists Regulate Progression through the Cell Cycle of Human Colon Carcinoma Cells. Proc. Natl. Acad. Sci. USA 2001, 98, 7846–7851. [Google Scholar]
- Pitari, G.M.; Lin, J.E.; Shah, F.J.; Lubbe, W.J.; Zuzga, D.S.; Li, P.; Schulz, S.; Waldman, S.A. Enterotoxin Preconditioning Restores Calcium-Sensing Receptor-Mediated Cytostasis in Colon Cancer Cells. Carcinogenesis 2008, 29, 1601–1607. [Google Scholar]
- Li, P.; Lin, J.E.; Snook, A.E.; Gibbons, A.V.; Zuzga, D.S.; Schulz, S.; Pitari, G.M.; Waldman, S.A. Colorectal Cancer Is a Paracrine Deficiency Syndrome Amenable to Oral Hormone Replacement Therapy. Clin. Transl. Sci. 2008, 1, 163–167. [Google Scholar]
- Birbe, R.; Palazzo, J.P.; Walters, R.; Weinberg, D.; Schulz, S.; Waldman, S.A. Guanylyl Cyclase C Is a Marker of Intestinal Metaplasia, Dysplasia, and Adenocarcinoma of the Gastrointestinal Tract. Hum. Pathol. 2005, 36, 170–179. [Google Scholar]
- Witek, M.E.; Nielsen, K.; Walters, R.; Hyslop, T.; Palazzo, J.; Schulz, S.; Waldman, S.A. The Putative Tumor Suppressor Cdx2 Is Overexpressed by Human Colorectal Adenocarcinomas. Clin. Cancer Res. 2005, 11, 8549–8556. [Google Scholar]
- Li, P.; Waldman, S.A. Corruption of Homeostatic Mechanisms in the Guanylyl Cyclase C Signaling Pathway Underlying Colorectal Tumorigenesis. Cancer Biol. Ther. 2010, 10, 211–218. [Google Scholar]
- Lin, J.E.; Li, P.; Snook, A.E.; Schulz, S.; Dasgupta, A.; Hyslop, T.M.; Gibbons, A.V.; Marszlowicz, G.; Pitari, G.M.; Waldman, S.A. The Hormone Receptor GUCY2C Suppresses Intestinal Tumor Formation by Inhibiting AKT Signaling. Gastroenterology 2010, 138, 241–254. [Google Scholar]
- Li, P.; Lin, J.E.; Marszlowicz, G.P.; Valentino, M.A.; Chang, C.; Schulz, S.; Pitari, G.M.; Waldman, S.A. GCC Signaling in Colorectal Cancer: Is Colorectal Cancer a Paracrine Deficiency Syndrome? Drug News Perspect. 2009, 22, 313–318. [Google Scholar]
- Shailubhai, K.; Yu, H.H.; Karunanandaa, K.; Wang, J.Y.; Eber, S.L.; Wang, Y.; Joo, N.S.; Kim, H.D.; Miedema, B.W.; Abbas, S.Z.; Boddupalli, S.S.; Currie, M.G.; Forte, L.R. Uroguanylin Treatment Suppresses Polyp Formation in the Apc(Min/+) Mouse and Induces Apoptosis in Human Colon Adenocarcinoma Cells via Cyclic GMP. Cancer Res. 2000, 60, 5151–5157. [Google Scholar]
- Pitari, G.M.; Baksh, R.I.; Harris, D.M.; Li, P.; Kazerounian, S.; Waldman, S.A. Interruption of Homologous Desensitization in Cyclic Guanosine 3',5'-Monophosphate Signaling Restores Colon Cancer Cytostasis by Bacterial Enterotoxins. Cancer Res. 2005, 65, 11129–11135. [Google Scholar]
© 2010 by the authors; licensee MDPI, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Weiglmeier, P.R.; Rösch, P.; Berkner, H. Cure and Curse: E. coli Heat-Stable Enterotoxin and Its Receptor Guanylyl Cyclase C. Toxins 2010, 2, 2213-2229. https://doi.org/10.3390/toxins2092213
Weiglmeier PR, Rösch P, Berkner H. Cure and Curse: E. coli Heat-Stable Enterotoxin and Its Receptor Guanylyl Cyclase C. Toxins. 2010; 2(9):2213-2229. https://doi.org/10.3390/toxins2092213
Chicago/Turabian StyleWeiglmeier, Philipp R., Paul Rösch, and Hanna Berkner. 2010. "Cure and Curse: E. coli Heat-Stable Enterotoxin and Its Receptor Guanylyl Cyclase C" Toxins 2, no. 9: 2213-2229. https://doi.org/10.3390/toxins2092213
APA StyleWeiglmeier, P. R., Rösch, P., & Berkner, H. (2010). Cure and Curse: E. coli Heat-Stable Enterotoxin and Its Receptor Guanylyl Cyclase C. Toxins, 2(9), 2213-2229. https://doi.org/10.3390/toxins2092213