Rapid Detection of Saxitoxin Using a Nucleic Acid Aptamer Biosensor Based on Graphene Oxide as a Fluorescence Quencher
Abstract
1. Introduction
2. Results and Discussion
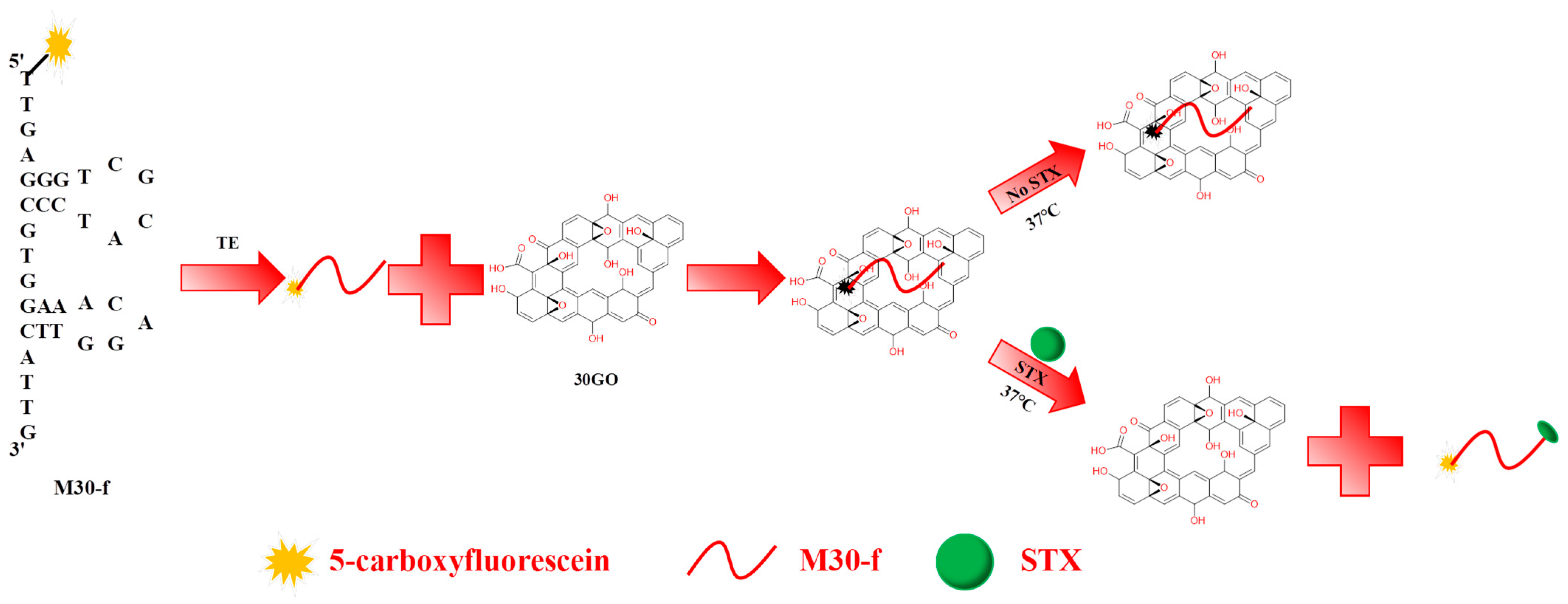
2.1. Experimental Principle
2.2. 30GO Characterisation
2.2.1. SEM Analysis
2.2.2. FT-IR and UV Analysis
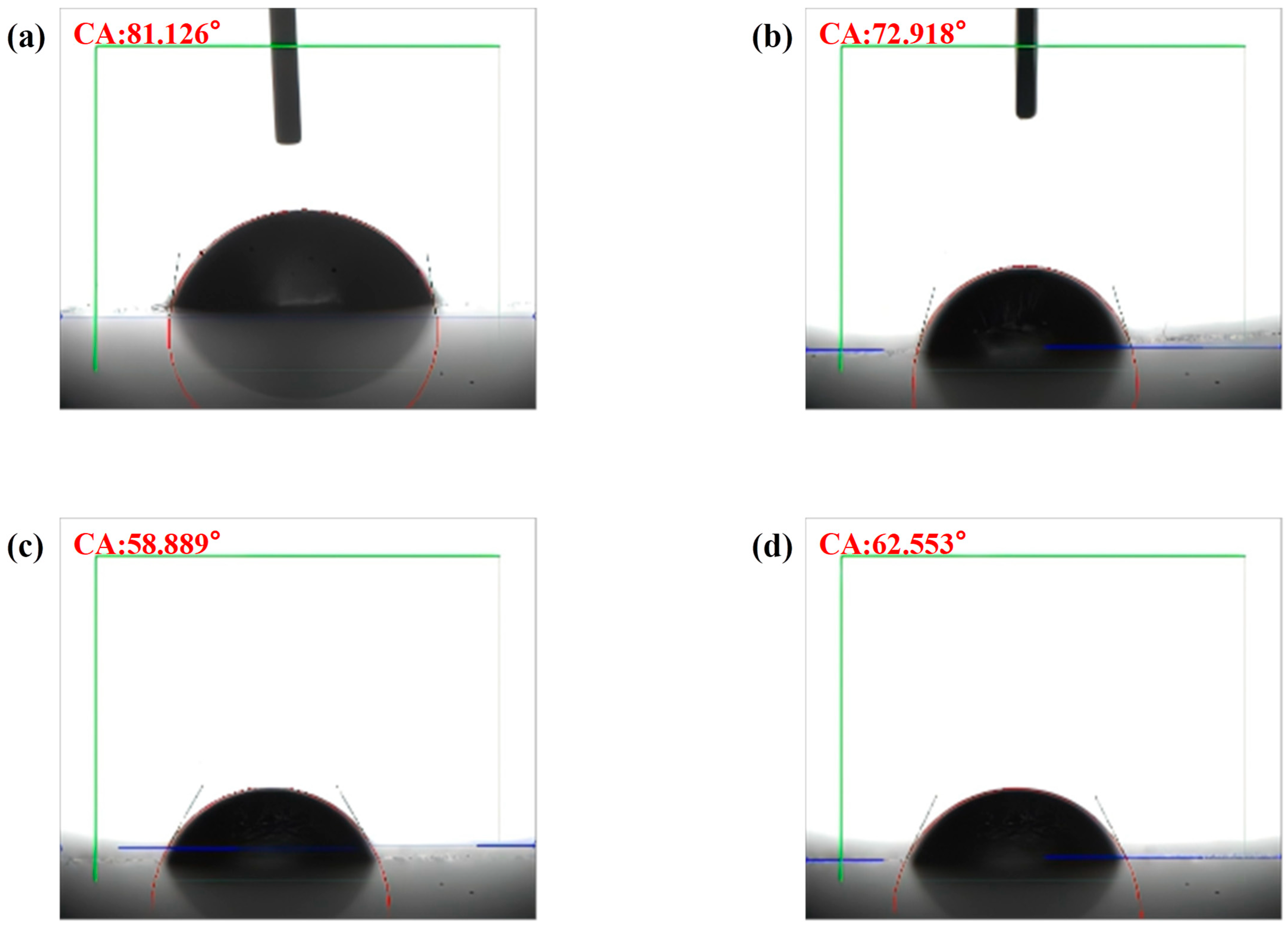
2.2.3. Contact Angle Analysis
2.2.4. XRD and AFM Analysis
2.2.5. XPS Analysis
2.3. STX Standard Solution Assay
2.3.1. Feasibility Confirmation Studies
2.3.2. Optimization of Experimental Parameters
2.3.3. Detection of STX Using the Developed Aptamer Sensor
2.3.4. Selectivity Test
2.4. Actual Sample Test
3. Conclusions
4. Materials and Methods
4.1. Materials
4.2. Ultrasonic Dispersion of GO
4.3. Material Characterization
4.4. STX Standard Solution Test
4.5. Specificity Test
4.6. Pre-Treatment of Clam and Razor Clam Samples
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kellmann, R.; Mihali Troco, K.; Jeon Young, J.; Pickford, R.; Pomati, F.; Neilan Brett, A. Biosynthetic Intermediate Analysis and Functional Homology Reveal a Saxitoxin Gene Cluster in Cyanobacteria. Appl. Environ. Microbiol. 2008, 74, 4044–4053. [Google Scholar] [CrossRef] [PubMed]
- Akbar, M.A.; Mohd Yusof, N.Y.; Tahir, N.I.; Ahmad, A.; Usup, G.; Sahrani, F.K.; Bunawan, H. Biosynthesis of Saxitoxin in Marine Dinoflagellates: An Omics Perspective. Mar. Drugs 2020, 18, 103. [Google Scholar] [CrossRef] [PubMed]
- Oshima, Y.; Hasegawa, M.; Yasumoto, T.; Hallegraeff, G.; Blackburn, S. Dinoflagellate Gymnodinium catenatum as the source of paralytic shellfish toxins in Tasmanian shellfish. Toxicon 1987, 25, 1105–1111. [Google Scholar] [CrossRef] [PubMed]
- Chekan, J.R.; Fallon, T.R.; Moore, B.S. Biosynthesis of marine toxins. Curr. Opin. Chem. Biol. 2020, 59, 119–129. [Google Scholar] [CrossRef]
- Cabado, A.G.; Lago, J.; González, V.; Blanco, L.; Paz, B.; Diogène, J.; Ferreres, L.; Rambla-Alegre, M. Detoxification of paralytic shellfish poisoning toxins in naturally contaminated mussels, clams and scallops by an industrial procedure. Food Chem. Toxicol. 2020, 141, 111386. [Google Scholar] [CrossRef]
- Pedrosa, C.; Souza, L.R.Q.; Gomes, T.A.; de Lima, C.V.F.; Ledur, P.F.; Karmirian, K.; Barbeito-Andres, J.; Costa, M.D.N.; Higa, L.M.; Rossi, Á.D.; et al. The cyanobacterial saxitoxin exacerbates neural cell death and brain malformations induced by Zika virus. PLoS Negl. Trop. Dis. 2020, 14, e0008060. [Google Scholar] [CrossRef]
- Farabegoli, F.; Blanco, L.; Rodríguez, L.P.; Vieites, J.M.; Cabado, A.G. Phycotoxins in Marine Shellfish: Origin, Occurrence and Effects on Humans. Mar. Drugs 2018, 16, 188. [Google Scholar] [CrossRef]
- Guillotin, S.; Delcourt, N. Marine Neurotoxins’ Effects on Environmental and Human Health: An OMICS Overview. Mar. Drugs 2022, 20, 18. [Google Scholar] [CrossRef]
- Watanabe, R.; Takayanagi, Y.; Chiba, O.; Itoda, S.; Ishizuka, H.; Odagi, M.; Ozawa, M.; Uchida, H.; Matsushima, R.; Konoki, K.; et al. Nontoxic Enantiomeric Reference Materials for Saxitoxins. Anal. Chem. 2022, 94, 11144–11150. [Google Scholar] [CrossRef]
- Deng, H.; Shang, X.; Zhu, H.; Huang, N.; Wang, L.; Sun, M. Saxitoxin: A Comprehensive Review of Its History, Structure, Toxicology, Biosynthesis, Detection, and Preventive Implications. Mar. Drugs 2025, 23, 277. [Google Scholar] [CrossRef]
- Cheng, J.; Pi, S.; Ye, S.; Gao, H.; Yao, L.; Jiang, Z.; Song, Y.; Xi, L. A new simple screening method for the detection of paralytic shellfish poisoning toxins. Chin. J. Oceanol. Limnol. 2012, 30, 786–790. [Google Scholar] [CrossRef]
- Podduturi, R.; Schlüter, L.; Liu, T.; Osti, J.A.S.; Moraes, M.d.A.B.; Jørgensen, N.O.G. Monitoring of saxitoxin production in lakes in Denmark by molecular, chromatographic and microscopic approaches. Harmful Algae 2021, 101, 101966. [Google Scholar] [CrossRef]
- Coleman, R.; Lemire, S.W.; Bragg, W.; Garrett, A.; Ojeda-Torres, G.; Wharton, R.; Hamelin, E.; Thomas, J.; Johnson, R.C. Development and validation of a high-throughput online solid-phase extraction–liquid chromatography–tandem mass spectrometry method for the detection of gonyautoxins 1&4 and gonyautoxins 2&3 in human urine. Biomed. Chromatogr. 2017, 31, e3954. [Google Scholar] [CrossRef]
- Noureen, B.; Ullah, N.; Tian, Y.; Du, L.; Chen, W.; Wu, C.; Wang, P. An electrochemical PAH-modified aptasensor for the label-free and highly-sensitive detection of saxitoxin. Talanta 2022, 240, 123185. [Google Scholar] [CrossRef]
- Bragg, W.A.; Lemire, S.W.; Coleman, R.M.; Hamelin, E.I.; Johnson, R.C. Detection of human exposure to saxitoxin and neosaxitoxin in urine by online-solid phase extraction-liquid chromatography–tandem mass spectrometry. Toxicon 2015, 99, 118–124. [Google Scholar] [CrossRef] [PubMed]
- Ling, S.; Xiao, S.; Xie, C.; Wang, R.; Zeng, L.; Wang, K.; Zhang, D.; Li, X.; Wang, S. Preparation of Monoclonal Antibody for Brevetoxin 1 and Development of Ic-ELISA and Colloidal Gold Strip to Detect Brevetoxin 1. Toxins 2018, 10, 75. [Google Scholar] [CrossRef] [PubMed]
- Eangoor, P.; Indapurkar, A.S.; Vakkalanka, M.D.; Knaack, J.S. Multiplexed ELISA screening assay for nine paralytic shellfish toxins in human plasma. Analyst 2019, 144, 4702–4707. [Google Scholar] [CrossRef]
- Tuerk, C.; Gold, L. Systematic evolution of ligands by exponential enrichment: RNA ligands to bacteriophage T4 DNA polymerase. Science 1990, 249, 505–510. [Google Scholar] [CrossRef]
- Xing, H.; Tang, L.; Yang, X.; Hwang, K.; Wang, W.; Yin, Q.; Wong, N.Y.; Dobrucki, L.W.; Yasui, N.; Katzenellenbogen, J.A.; et al. Selective Delivery of an Anticancer Drug with Aptamer-Functionalized Liposomes to Breast Cancer Cells In Vitro and In Vivo. J. Mater. Chem. B 2013, 1, 5288–5297. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.-X.; Ye, Z.-Z.; Si, C.-Y.; Ying, Y.-B. Application of Aptamer Based Biosensors for Detection of Pathogenic Microorganisms. Chin. J. Anal. Chem. 2012, 40, 634–642. [Google Scholar] [CrossRef]
- Nutiu, R.; Li, Y. In vitro selection of structure-switching signaling aptamers. Angew. Chem. Int. Ed. Engl. 2005, 44, 1061–1065. [Google Scholar] [CrossRef]
- Tolle, F.; Brändle, G.M.; Matzner, D.; Mayer, G. A Versatile Approach Towards Nucleobase-Modified Aptamers. Angew. Chem. Int. Ed. 2015, 54, 10971–10974. [Google Scholar] [CrossRef]
- Zhao, L.; Huang, Y.; Dong, Y.; Han, X.; Wang, S.; Liang, X. Aptamers and Aptasensors for Highly Specific Recognition and Sensitive Detection of Marine Biotoxins: Recent Advances and Perspectives. Toxins 2018, 10, 427. [Google Scholar] [CrossRef]
- Ma, C.; Wu, K.; Zhao, H.; Liu, H.; Wang, K.; Xia, K. Fluorometric aptamer-based determination of ochratoxin A based on the use of graphene oxide and RNase H-aided amplification. Mikrochim. Acta 2018, 185, 347. [Google Scholar] [CrossRef]
- Song, S.-H.; Gao, Z.-F.; Guo, X.; Chen, G.-H. Aptamer-Based Detection Methodology Studies in Food Safety. Food Anal. Methods 2019, 12, 966–990. [Google Scholar] [CrossRef]
- Dikin, D.A.; Stankovich, S.; Zimney, E.J.; Piner, R.D.; Dommett, G.H.; Evmenenko, G.; Nguyen, S.T.; Ruoff, R.S. Preparation and characterization of graphene oxide paper. Nature 2007, 448, 457–460. [Google Scholar] [CrossRef] [PubMed]
- Yugender Goud, K.; Hayat, A.; Satyanarayana, M.; Sunil Kumar, V.; Catanante, G.; Vengatajalabathy Gobi, K.; Marty, J.L. Aptamer-based zearalenone assay based on the use of a fluorescein label and a functional graphene oxide as a quencher. Microchim. Acta 2017, 184, 4401–4408. [Google Scholar] [CrossRef]
- Li, M.; Zhou, X.; Ding, W.; Guo, S.; Wu, N. Fluorescent aptamer-functionalized graphene oxide biosensor for label-free detection of mercury(II). Biosens. Bioelectron. 2013, 41, 889–893. [Google Scholar] [CrossRef]
- Mungse, H.P.; Khatri, O.P. Chemically Functionalized Reduced Graphene Oxide as a Novel Material for Reduction of Friction and Wear. J. Phys. Chem. C 2014, 118, 14394–14402. [Google Scholar] [CrossRef]
- Dobrota, A.S.; Pašti, I.A.; Mentus, S.V.; Skorodumova, N.V. A general view on the reactivity of the oxygen-functionalized graphene basal plane. Phys. Chem. Chem. Phys. 2016, 18, 6580–6586. [Google Scholar] [CrossRef]
- Zhou, K.; Zhu, Y.; Yang, X.; Jiang, X.; Li, C. Preparation of graphene–TiO2 composites with enhanced photocatalytic activity. New J. Chem. 2011, 35, 353–359. [Google Scholar] [CrossRef]
- Zhang, Y.; Pan, C. TiO2/graphene composite from thermal reaction of graphene oxide and its photocatalytic activity in visible light. J. Mater. Sci. 2011, 46, 2622–2626. [Google Scholar] [CrossRef]
- Shen, J.; Shi, M.; Yan, B.; Ma, H.; Li, N.; Ye, M. Ionic liquid-assisted one-step hydrothermal synthesis of TiO2-reduced graphene oxide composites. Nano Res. 2011, 4, 795–806. [Google Scholar] [CrossRef]
- Sun, N.; Meng, Z.; Zhen, L.; Qin, Q.; Liu, W.; Xue, J.; Sun, Y.; Tong, P. Effect of GO on oxytetracycline and Cu2+ co-adsorption in a yellow–brown soil. Appl. Surf. Sci. 2025, 702, 163345. [Google Scholar] [CrossRef]
- Li, D.; Müller, M.B.; Gilje, S.; Kaner, R.B.; Wallace, G.G. Processable aqueous dispersions of graphene nanosheets. Nat. Nanotechnol. 2008, 3, 101–105. [Google Scholar] [CrossRef]
- Habte, A.T.; Ayele, D.W. Synthesis and Characterization of Reduced Graphene Oxide (rGO) Started from Graphene Oxide (GO) Using the Tour Method with Different Parameters. Adv. Mater. Sci. Eng. 2019, 2019, 5058163. [Google Scholar] [CrossRef]
- He, P.; Zhao, Z.; Tan, Y.; E, H.; Zuo, M.; Wang, J.; Yang, J.; Cui, S.; Yang, X. Photocatalytic Degradation of Deoxynivalenol Using Cerium Doped Titanium Dioxide under Ultraviolet Light Irradiation. Toxins 2021, 13, 481. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Campos, D.; Reyes-Valderrama, M.I.; Mendoza-Anaya, D.; Escobar-Alarcón, L.; Rodríguez-Lugo, V. Enhanced methodology for graphene oxide transformation: Unraveling the effects of ultrasonication on morphological and structural characteristics. MRS Adv. 2024, 9, 743–748. [Google Scholar] [CrossRef]
- Han, Z.; Sun, L.; Chu, Y.; Wang, J.; Wei, C.; Liu, Y.; Jiang, Q.; Han, C.; Yan, H.; Song, X. Ultrasonication-Tailored Graphene Oxide of Varying Sizes in Multiple-Equilibrium-Route-Enhanced Adsorption for Aqueous Removal of Acridine Orange. Molecules 2023, 28, 4179. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Chen, L.; Meng, Y. Fabrication and characterization of reduced graphene oxide by atmospheric pressure plasma jet. AIP Adv. 2020, 10, 015216. [Google Scholar] [CrossRef]
- Surekha, G.; Krishnaiah, K.V.; Ravi, N.; Padma Suvarna, R. FTIR, Raman and XRD analysis of graphene oxide films prepared by modified Hummers method. J. Phys. Conf. Ser. 2020, 1495, 012012. [Google Scholar] [CrossRef]
- Khan, H.; Yerramilli, A.S.; D’Oliveira, A.; Alford, T.L.; Boffito, D.C.; Patience, G.S. Experimental methods in chemical engineering: X-ray diffraction spectroscopy—XRD. Can. J. Chem. Eng. 2020, 98, 1255–1266. [Google Scholar] [CrossRef]
- Ganguly, A.; Sharma, S.; Papakonstantinou, P.; Hamilton, J. Probing the Thermal Deoxygenation of Graphene Oxide Using High-Resolution In Situ X-ray-Based Spectroscopies. J. Phys. Chem. C 2011, 115, 17009–17019. [Google Scholar] [CrossRef]
- Yamada, Y.; Yasuda, H.; Murota, K.; Nakamura, M.; Sodesawa, T.; Sato, S. Analysis of heat-treated graphite oxide by X-ray photoelectron spectroscopy. J. Mater. Sci. 2013, 48, 8171–8198. [Google Scholar] [CrossRef]
- Ren, P.-G.; Yan, D.-X.; Ji, X.; Chen, T.; Li, Z.-M. Temperature dependence of graphene oxide reduced by hydrazine hydrate. Nanotechnology 2011, 22, 055705. [Google Scholar] [CrossRef] [PubMed]
- Krishnamoorthy, K.; Veerapandian, M.; Yun, K.; Kim, S.J. The chemical and structural analysis of graphene oxide with different degrees of oxidation. Carbon 2013, 53, 38–49. [Google Scholar] [CrossRef]
- Veerapandian, M.; Lee, M.-H.; Krishnamoorthy, K.; Yun, K. Synthesis, characterization and electrochemical properties of functionalized graphene oxide. Carbon 2012, 50, 4228–4238. [Google Scholar] [CrossRef]
- Serrano, P.C.; Nunes, G.E.; Avila, L.B.; Reis, C.P.S.; Gomes, A.M.C.; Reis, F.T.; Sartorelli, M.L.; Melegari, S.P.; Matias, W.G.; Bechtold, I.H. Electrochemical impedance biosensor for detection of saxitoxin in aqueous solution. Anal. Bioanal. Chem. 2021, 413, 6393–6399. [Google Scholar] [CrossRef]
- Zheng, W.; Liu, X.; Li, Q.; Shu, Z.; Li, Z.; Zhang, L. A simple electrochemical aptasensor for saxitoxin detection. RSC Adv. 2022, 12, 23801–23807. [Google Scholar] [CrossRef]
- Dou, X.; Xu, S.; Jiang, Y.; Ding, Z.; Xie, J. Aptamers-functionalized nanoscale MOFs for saxitoxin and tetrodotoxin sensing in sea foods through FRET. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2023, 284, 121827. [Google Scholar] [CrossRef]
- Zhu, L.; Zeng, W.; Li, Y.; Han, Y.; Wei, J.; Wu, L. Development of magnetic fluorescence aptasensor for sensitive detection of saxitoxin based on Fe3O4@Au-Pt nanozymes. Sci. Total Environ. 2024, 921, 171236. [Google Scholar] [CrossRef]
- Cheng, S.; Zheng, B.; Yao, D.; Wang, Y.; Tian, J.; Liu, L.; Liang, H.; Ding, Y. Determination of Saxitoxin by Aptamer-Based Surface-Enhanced Raman Scattering. Anal. Lett. 2019, 52, 902–918. [Google Scholar] [CrossRef]
- Ha, S.-J.; Park, J.-H.; Lee, B.; Kim, M.-G. Label-Free Direct Detection of Saxitoxin Based on a Localized Surface Plasmon Resonance Aptasensor. Toxins 2019, 11, 274. [Google Scholar] [CrossRef] [PubMed]
- Armbruster, D.A.; Pry, T. Limit of blank, limit of detection and limit of quantitation. Clin. Biochem. Rev. 2008, 29 (Suppl. 1), S49–S52. [Google Scholar] [PubMed]




| Aptasensors | Linear Detection Range | LOD | Literature Resources | ||
|---|---|---|---|---|---|
| Reported Range | Converted Range (nM) | Reported LOD | Converted LOD (nM) | ||
| Electrochemical aptasensor | 0.9–30 nM | 0.9–30 | 0.38 nM | 0.38 | [48] |
| Electrochemical aptasensor | 1–1000 nM | 1–1000 | 1 nM | 1 | [49] |
| Fluorescent aptasensor | 0–24 ng/mL | 0–64.48 | 1.8 ng/mL | 4.84 | [50] |
| Fluorescence aptasensor | 1–5000 nM | 1–5000 | 0.6 nM | 0.6 | [51] |
| SERS sensor | 10–200 nM | 10–200 | 11.7 nM | 11.7 | [52] |
| LSPR-based aptasensor | 5–10,000 μg/L | 16.72–33,440 | 2.46 μg/L | 8.23 | [53] |
| Fluorescent aptasensor | 10–100,000 ng/L | 2.7 × 10−2–268.7 | 0.098 μg/L | 0.26 | This work |
| Specimen | Addition Level (μg/kg) | Recovery (%, Mean) | RSD (%) |
|---|---|---|---|
| Manila clam | 1 | 89.12 | 2.83 |
| 10 | 97.72 | 2.13 | |
| 100 | 104.71 | 1.65 | |
| Razor clam | 1 | 109.65 | 1.40 |
| 10 | 102.33 | 2.59 | |
| 100 | 91.20 | 2.91 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiao, Y.; Yang, L.; Hao, J.; Wen, Y.; Wang, J.; E, H.; Zhao, Z.; Chen, Y.; Yang, X. Rapid Detection of Saxitoxin Using a Nucleic Acid Aptamer Biosensor Based on Graphene Oxide as a Fluorescence Quencher. Toxins 2025, 17, 430. https://doi.org/10.3390/toxins17090430
Jiao Y, Yang L, Hao J, Wen Y, Wang J, E H, Zhao Z, Chen Y, Yang X. Rapid Detection of Saxitoxin Using a Nucleic Acid Aptamer Biosensor Based on Graphene Oxide as a Fluorescence Quencher. Toxins. 2025; 17(9):430. https://doi.org/10.3390/toxins17090430
Chicago/Turabian StyleJiao, Yi, Liqing Yang, Junping Hao, Yuhang Wen, Jianhua Wang, Hengchao E, Zhiyong Zhao, Yufeng Chen, and Xianli Yang. 2025. "Rapid Detection of Saxitoxin Using a Nucleic Acid Aptamer Biosensor Based on Graphene Oxide as a Fluorescence Quencher" Toxins 17, no. 9: 430. https://doi.org/10.3390/toxins17090430
APA StyleJiao, Y., Yang, L., Hao, J., Wen, Y., Wang, J., E, H., Zhao, Z., Chen, Y., & Yang, X. (2025). Rapid Detection of Saxitoxin Using a Nucleic Acid Aptamer Biosensor Based on Graphene Oxide as a Fluorescence Quencher. Toxins, 17(9), 430. https://doi.org/10.3390/toxins17090430






