Effects of Harmful Algal Blooms on Fish and Shellfish Species: A Case Study of New Zealand in a Changing Environment
Abstract
1. Introduction
New Zealand Aquaculture and HABs
2. Effects of Bloom Forming HAB Species on Species of Relevance to the NZ Aquaculture Industry
2.1. Alexandrium and Gymnodinium spp.
2.2. Karenia spp.
2.3. Dinophysis spp.
2.4. Pseudo-nitzschia spp.
2.5. The Raphidophytes: Heterosigma and Fibrocapsa
2.6. Other Bloom Forming HAB Species in New Zealand
3. Future Issues
3.1. Ichthyotoxic HAB Species Present in New Zealand
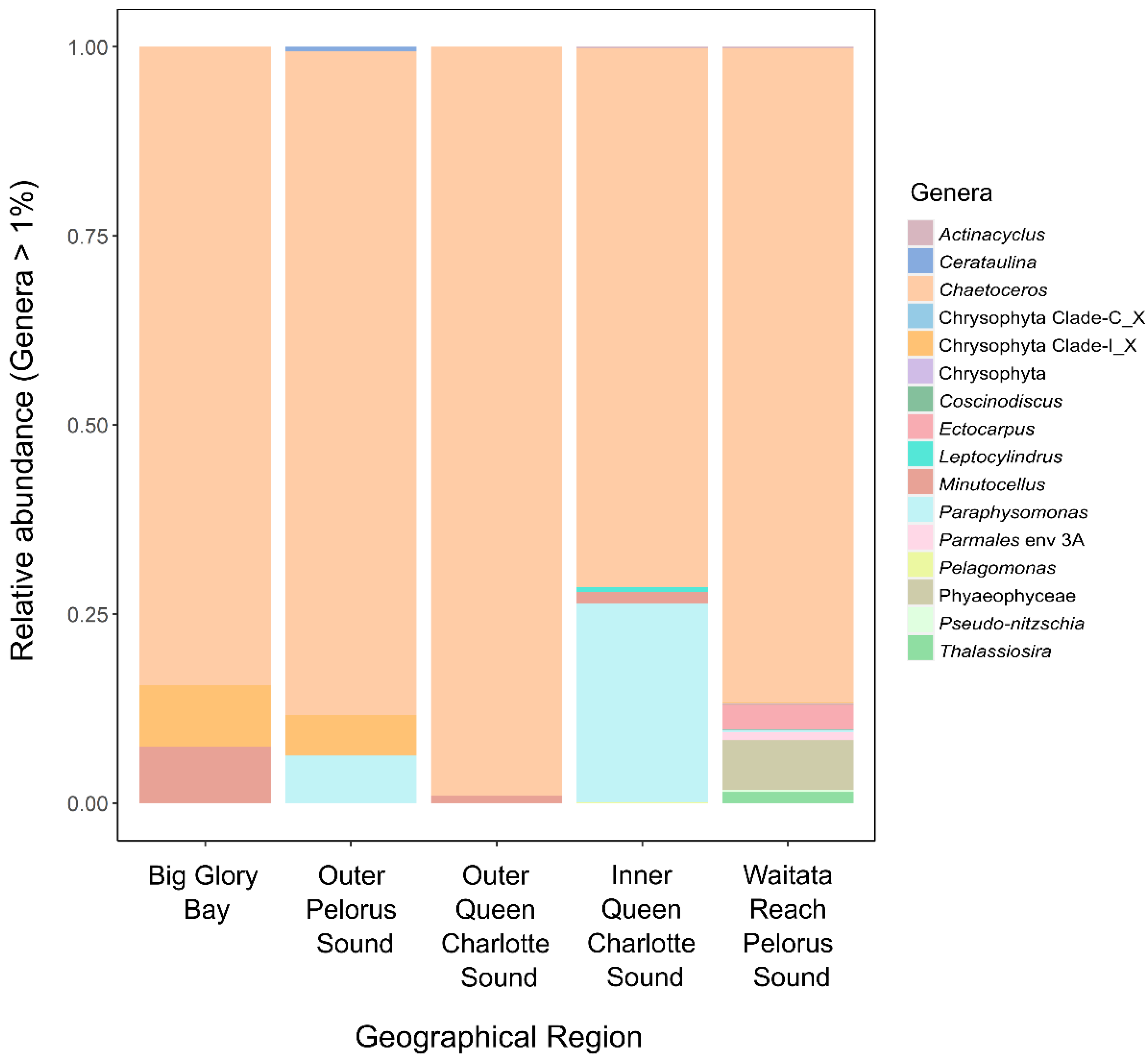
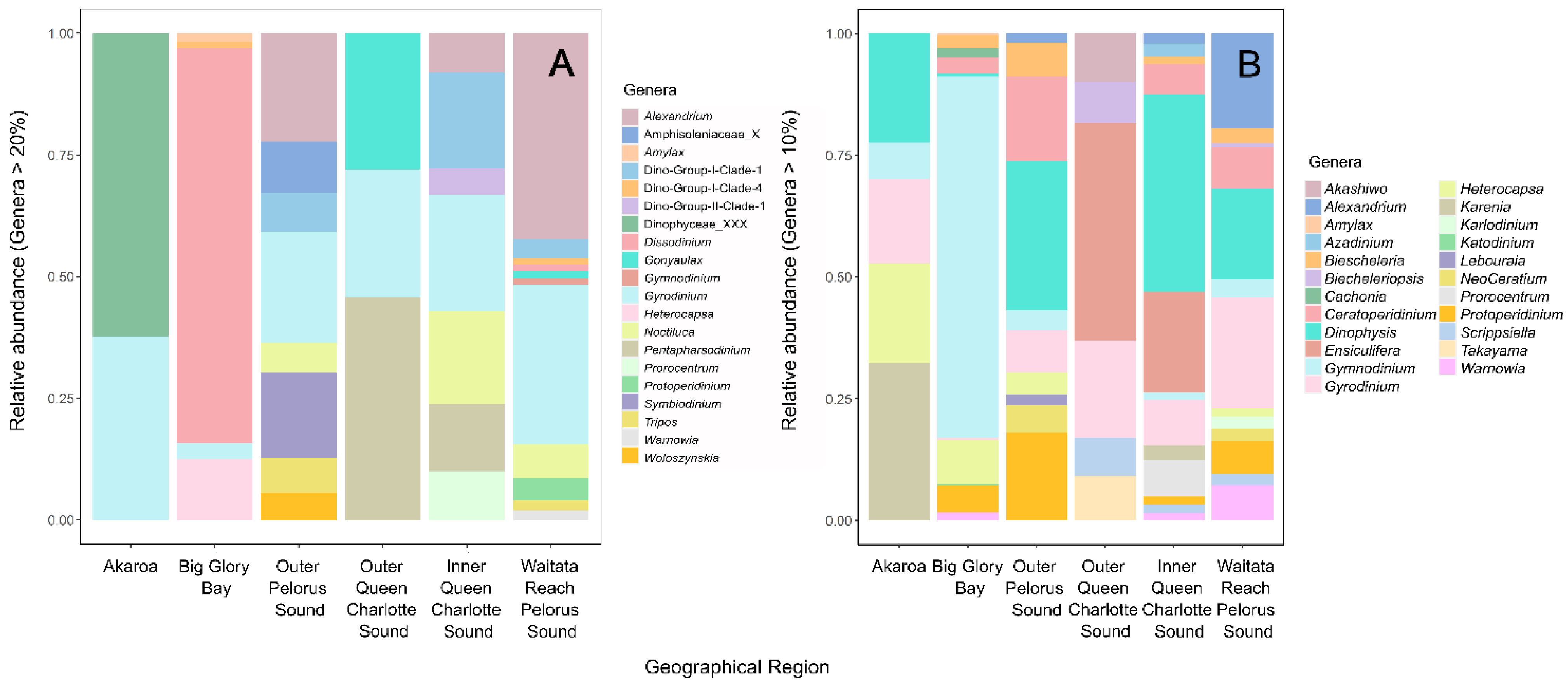
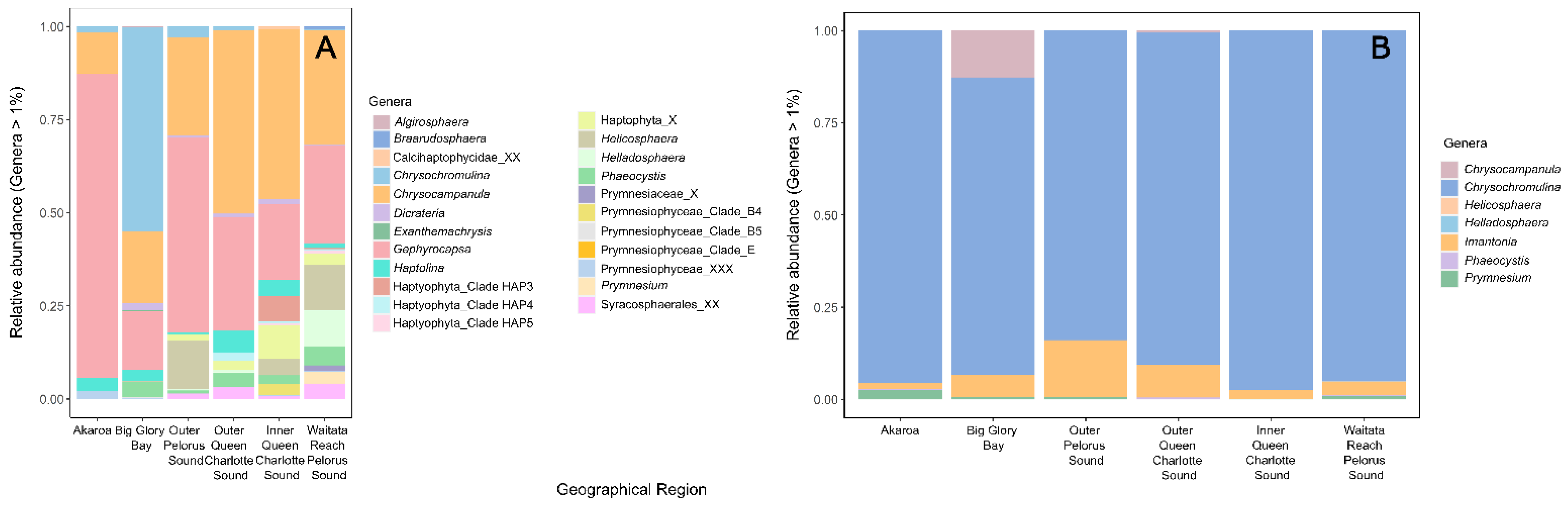
3.1.1. Potentially Harmful Taxa Detected Using High-Throughput Sequencing Metabarcoding
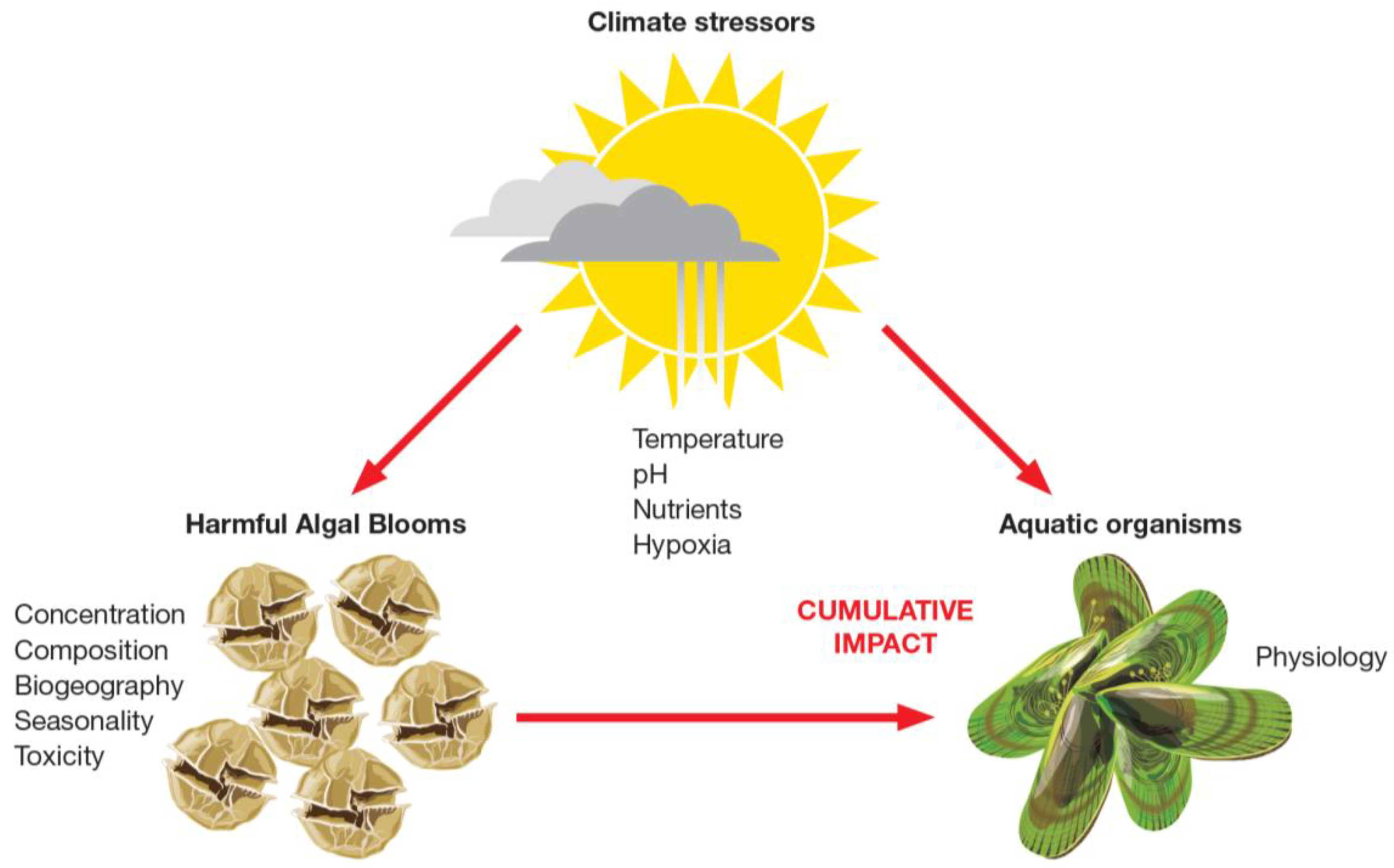
3.2. HABs in a Changing Environment
4. Conclusions and Future Directions
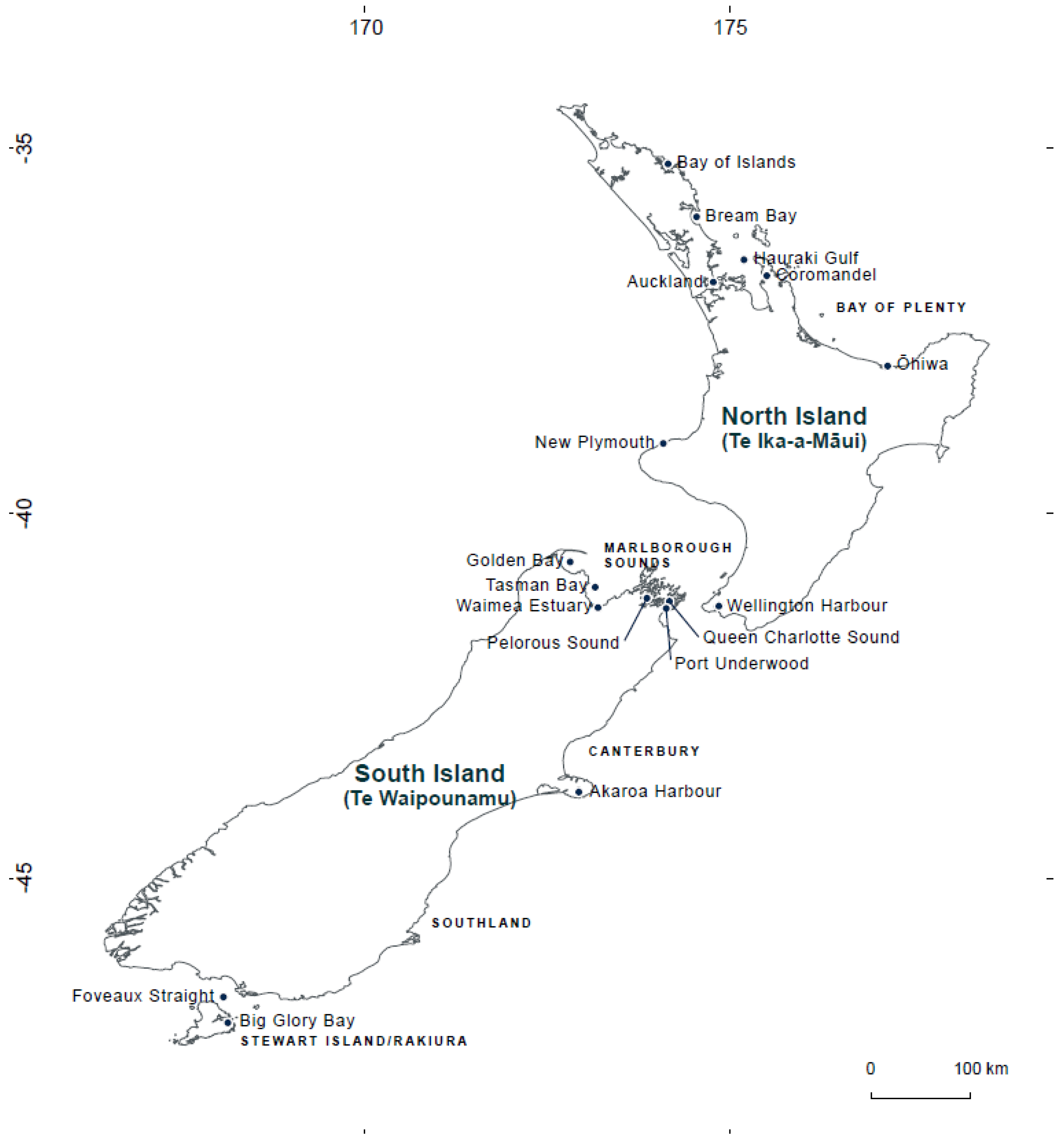
5. Materials and Methods
5.1. Sampling, PCR Conditions and High-Throughput Sequencing
5.2. Amplicon Sequence Variant Inference and Taxonomic Assignments
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Anderson, D.M.; Cembella, A.D.; Hallegraeff, G.M. Progress in understanding harmful algal blooms: Paradigm shifts and new technologies for research, monitoring, and management. Annu. Rev. Mar. Sci. 2012, 4, 143–176. [Google Scholar] [CrossRef] [PubMed]
- Landsberg, J.H. The effects of harmful algal blooms on aquatic organisms. Rev. Fish. Sci. 2002, 10, 113–390. [Google Scholar] [CrossRef]
- Shumway, S.E. A review of the effects of algal blooms on shellfish and aquaculture. J. World Aquac. Soc. 1990, 21, 65–104. [Google Scholar] [CrossRef]
- Van Dolah, F.M. Marine algal toxins: Origins, health effects, and their increased occurrence. Environ. Health Perspect. 2000, 108, 133–141. [Google Scholar] [CrossRef]
- Hallegraeff, G.M. Ocean climate change, phytoplankton community responses, and harmful algal blooms: A formidable predictive challenge. J. Phycol. 2010, 46, 220–235. [Google Scholar] [CrossRef]
- Brown, A.R.; Lilley, M.; Shutler, J.; Lowe, C.; Artioli, Y.; Torres, R.; Berdalet, E.; Tyler, C.R. Assessing risks and mitigating impacts of harmful algal blooms on mariculture and marine fisheries. Rev. Aquac. 2020, 12, 1663–1688. [Google Scholar] [CrossRef]
- Hoagland, P.; Scatasta, S. The economic effects of harmful algal blooms. In Ecology of Harmful Algae; Granéli, E., Turner, J.T., Eds.; Springer: Berlin/Heidelberg, Germany, 2006; pp. 391–402. [Google Scholar]
- Martino, S.; Gianella, F.; Davidson, D. An approach for evaluating the economic impacts of harmful algal blooms: The effects of blooms of toxic Dinophysis spp. on the productivity of Scottish shellfish farms. Harmful Algae 2020, 99, 101912. [Google Scholar] [CrossRef]
- Trainer, V.L. Global HAB: Evaluating, Reducing and Mitigating the Cost of Harmful Algal Blooms: A Compendium of Case Studies; North Pacific Marine Science Organization: Sidney, BC, Canada, 2020; 107p. [Google Scholar]
- Flewelling, L.; Naar, J.; Abbott, J.; Baden, D.; Barros, N.; Bossart, G.; Dechraoui Bottein, M.-Y.; Hammond, D.; Haubold, E.; Heil, C.; et al. Brevetoxicosis: Red tides and marine mammal mortalities. Nature 2005, 435, 755–756. [Google Scholar] [CrossRef]
- Leverone, J.R.; Blake, N.J.; Pierce, R.H.; Shumway, S.E. Effects of the dinoflagellate Karenia brevis on larval development in three species of bivalve mollusc from Florida. Toxicon 2006, 48, 75–84. [Google Scholar] [CrossRef]
- Rolton, A.; Vignier, J.; Soudant, P.; Shumway, S.E.; Bricelj, V.M.; Volety, A.K. Effects of the red tide dinoflagellate, Karenia brevis, on early development of the eastern oyster Crassostrea virginica and northern quahog Mercenaria mercenaria. Aquat. Toxicol. 2014, 155, 199–206. [Google Scholar] [CrossRef]
- Rolton, A.; Vignier, J.; Volety, A.K.; Pierce, R.H.; Henry, M.; Shumway, S.E.; Bricelj, V.M.; Hégaret, H.; Soudant, P. Effects of field and laboratory exposure to the toxic dinoflagellate Karenia brevis on the reproduction of the eastern oyster, Crassostrea virginica, and subsequent development of offspring. Harmful Algae 2016, 57, 13–26. [Google Scholar] [CrossRef] [PubMed]
- Rolton, A.; Vignier, J.; Volety, A.K.; Shumway, S.E.; Bricelj, V.M.; Soudant, P. Impacts of exposure to the toxic dinoflagellate Karenia brevis on reproduction of the northern quahog, Mercenaria mercenaria. Aquat. Toxicol. 2018, 202, 153–162. [Google Scholar] [CrossRef] [PubMed]
- Lenzen, M.; Li, M.; Murray, S.A. Impacts of harmful algal blooms on marine aquaculture in a low-carbon future. Harmful Algae 2021, 110, 102143. [Google Scholar] [CrossRef] [PubMed]
- Castrec, J.; Hégaret, H.; Huber, M.; Le Grand, J.; Huvet, A.; Tallec, K.; Boulais, M.; Soudant, P.; Fabioux, C. The toxic dinoflagellate Alexandrium minutum impairs the performance of oyster embryos and larvae. Harmful Algae 2020, 92, 101744. [Google Scholar] [CrossRef] [PubMed]
- Castrec, J.; Fabioux, C.; Le Goïc, N.; Boulais, M.; Soudant, P.; Hégaret, H. The toxic dinoflagellate Alexandrium minutum affects oyster gamete health and fertilization potential. Mar. Environ. Res. 2021, 169, 105401. [Google Scholar] [CrossRef]
- Corriere, M.; Soliño, L.; Costa, P.R. Effects of the marine biotoxins okadaic acid and dinophysistoxins on fish. J. Mar. Sci. Eng. 2021, 9, 293. [Google Scholar] [CrossRef]
- Costa, P.R. Impact and effects of paralytic shellfish poisoning toxins derived from harmful algal blooms to marine fish. Fish Fish. 2016, 17, 226–248. [Google Scholar] [CrossRef]
- Gosselin, L.A.; Qian, P.Y. Juvenile mortality in benthic marine invertebrates. Mar. Ecol. Prog. Ser. 1997, 146, 265–282. [Google Scholar] [CrossRef]
- Pechenik, J.A. Environmental influences on larval survival and development. In Reproduction of Marine Invertebrates; Giese, A.C., Pearse, J.S., Pearse, V.B., Eds.; Blackwell Scientific Publications: Palo Alto, CA, USA, 1987; p. 551e608. [Google Scholar]
- Rolton, A.; Soudant, P.; Vignier, J.; Pierce, R.; Henry, M.; Shumway, S.E.; Bricelj, V.M.; Volety, A.K. Susceptibility of gametes and embryos of the eastern oyster, Crassostrea virginica, to Karenia brevis and its toxins. Toxicon 2015, 99, 6–15. [Google Scholar] [CrossRef]
- Matsuyama, Y.; Oda, T. Toxic effects of harmful algal blooms on finfish and shellfish. In Handbook of Algal Science, Technology and Medicine (Part IX: Algal Toxicology, Chapter 34th); Konur, O., Ed.; Academic Press: London, UK, 2020; pp. 543–560. [Google Scholar]
- Davidson, K.; Jardine, S.L.; Martino, S.; Myre, G.B.; Peck, L.E.; Raymond, R.N.; West, J.J. The Economic Impacts of Harmful Algal Blooms on Salmon Cage Aquaculture. In GlobalHAB. Evaluating, Reducing and Mitigating the Cost of Harmful Algal Blooms: A Compendium of Case Studies; Trainer, V.L., Ed.; North Pacific Marine Science Organization: Sidney, BC, Canada, 2020; 107p. [Google Scholar]
- Hallegraeff, G.M.; Schweibold, L.; Jaffrezic, E.; Rhodes, L.; MacKenzie, L.; Hay, B.; Farrell, H. Overview of Australian and New Zealand harmful algal species occurrences and their societal impacts in the period 1985 to 2018, including a compilation of historic records. Harmful Algae 2021, 102, 101848. [Google Scholar] [CrossRef]
- King, T.L.; Nguyen, N.; Doucette, G.J.; Wang, Z.; Bill, B.D.; Peacock, M.B.; Madera, S.L.; Elston, R.A.; Trainer, V.L. Hiding in plain sight: Shellfish-killing phytoplankton in Washington State. Harmful Algae 2021, 105, 102032. [Google Scholar] [CrossRef] [PubMed]
- Samdal, I.A.; Edvardsen, B. Massive salmon mortalities during a Chrysochromulina leadbeateri bloom in Northern Norway. Harmful Algae News 2020, 64, 4–5. [Google Scholar]
- Rhodes, L.; Edwards, A.R.; Bojo, O.; Chang, F.H. Phylum Haptophyta. In New Zealand Inventory of Biodiversity Volume 3: Kingdoms, Bacteria, Protozoa, Chromista, Plantae and Fungi; Gordon, D.P., Ed.; Canterbury University Press: Christchurch, New Zealand, 2011; Volume 3, pp. 312–321. [Google Scholar]
- Basti, L.; Endo, M.; Segawa, S. Physiological, pathological, and defense alterations in Manila clams (short-neck clams), Ruditapes philippinarum, induced by Heterocapsa circularisquama. J. Shellfish. Res. 2011, 30, 829–844. [Google Scholar] [CrossRef]
- Galimany, E.; Sunila, I.; Hégaret, H.; Ramón, M.; Wikfors, G.H. Experimental exposure of the blue mussel (Mytilus edulis, L.) to the toxic dinoflagellate Alexandrium fundyense: Histopathology, immune responses, and recovery. Harmful Algae 2008, 7, 702–711. [Google Scholar] [CrossRef]
- Galimany, E.; Sunila, I.; Hégaret, H.; Ramón, M.; Wikfors, G.H. Pathology and immune response of the blue mussel (Mytilus edulis L.) after an exposure to the harmful dinoflagellate Prorocentrum minimum. Harmful Algae 2008, 7, 630–638. [Google Scholar] [CrossRef]
- Hégaret, H.; Wikfors, G.H.; Shumway, S.E. Diverse feeding responses of five species of bivalve mollusc when exposed to three species of Harmful Algae. J. Shellfish. Res. 2007, 26, 549–559. [Google Scholar] [CrossRef]
- Hégaret, H.; Da Silva, P.M.; Wikfors, G.H.; Haberkorn, H.; Shumway, S.E.; Soudant, P. In vitro interactions between several species of harmful algae and haemocytes of bivalve molluscs. Cell Biol. Toxicol. 2011, 27, 249–266. [Google Scholar] [CrossRef]
- Shumway, S.E.; Cucci, T.L. The effects of the toxic dinoflagellate Protogonyaulax tamarensis on the feeding and behavior of bivalve molluscs. Aquat. Toxicol. 1987, 10, 9–27. [Google Scholar] [CrossRef]
- Wikfors, G.H.; Smolowitz, R.M. Experimental and Histological Studies of Four Life-History Stages of the Eastern Oyster, Crassostrea virginica, exposed to a Cultured Strain of the Dinoflagellate Prorocentrum minimum. Biol Bull. 1995, 188, 313–328. [Google Scholar] [CrossRef]
- Aquaculture New Zealand. Aquaculture New Zealand, New Zealand Aquaculture: A Sector Overview with Key Facts and Statistics; Aquaculture New Zealand Industry—Aquaculture New Zealand: Nelson, New Zealand, 2020; pp. 1–26. [Google Scholar]
- FAO. Fishery and Aquaculture Statistics. Global aquaculture production 1950–2019 (FishstatJ). In FAO Fisheries Division; FAO: Rome, Italy, 2021; Available online: https://www.fao.org/fishery/statistics/software/fishstatj/en (accessed on 10 March 2022).
- Atalah, J.; Forrest, B.M. Forecasting mussel settlement using historical data and boosted regression trees. Aquac. Environ. Interact. 2019, 11, 625–638. [Google Scholar] [CrossRef]
- How Climate Change Affects New Zealand. Ministry for the Environment. Available online: https://environment.govt.nz/facts-and-science/climate-change/how-climate-change-affects-new-zealand/ (accessed on 14 April 2022).
- Oldham, K.; (Marine Farming Association, Blenheim, New Zealand). Personal communication, 2021.
- Chang, F.H.; Anderson, C.; Boustead, N.C. First record of a Heterosigma (Raphidophyceae) bloom with associated mortality of cage-reared salmon in Big Glory Bay, New Zealand. N. Z. J. Mar. Freshw. Res. 1990, 24, 461–469. [Google Scholar] [CrossRef]
- MacKenzie, L.; Truman, P.; Mountfort, D.; White, D. Dinophysis acuta blooms and associated shellfish toxicity in New Zealand. In Proceedings of the VIII International Conference on Harmful Algae-Abstracts and Posters Classification, Vigo, Spain, 25–29 June 1997. [Google Scholar]
- MacKenzie, L.; Smith, K.F.; Rhodes, L.L.; Brown, A.; Langi, V.; Edgar, M.; Lovell, G.; Preece, M. Mortalities of sea-cage salmon (Oncorhynchus tshawytscha) due to a bloom of Pseudochattonella verruculosa (Dictyophyceae) in Queen Charlotte Sound, New Zealand. Harmful Algae 2011, 11, 45–53. [Google Scholar] [CrossRef]
- MacKenzie, L.; Harwood, T.; Boundy, M.; Smith, K.; Knight, B.; Jiang, W.; McNabb, P.; Selwood, A.; van Ginkel, R.; Langi, V.; et al. An Alexandrium catenella bloom and associated saxitoxin contamination of shellfish, Queen Charlotte Sound, March–June 2011. In A Report for MAF Food Safety; Cawthron Institute: Nelson, New Zealand, 2011; 38p. [Google Scholar]
- MacKenzie, L. The risk to New Zealand shellfish aquaculture from paralytic shellfish poisoning (PSP) toxins. N. Z. J. Mar. Freshw. Res. 2014, 48, 430–465. [Google Scholar] [CrossRef]
- Liu, H.; Kelly, M.S.; Campbell, D.A.; Dong, S.L.; Zhu, J.X.; Wang, S.F. Exposure to domoic acid affects larval development of king scallop Pecten maximus (Linnaeus, 1758). Aquat. Toxicol. 2007, 81, 152–158. [Google Scholar] [CrossRef]
- Sauvey, A.; Denis, F.; Hégaret, H.; Le Roy, B.; Lelong, C.; Jolly, O.; Pavie, M.; Fauchot, J. Interactions between Filter-Feeding Bivalves and Toxic Diatoms: Influence on the Feeding Behavior of Crassostrea gigas and Pecten maximus and on Toxin Production by Pseudo-nitzschia. Toxins 2021, 13, 577. [Google Scholar] [CrossRef]
- Mafra, L.L.; Bricelj, V.M.; Ouellette, C.; Leger, C.; Bates, S.S. Mechanisms contributing to low domoic acid uptake by oysters feeding on Pseudo-nitzschia cells. I. Filtration and pseudofeces production. Aquat. Biol. 2009, 6, 201–212. [Google Scholar] [CrossRef]
- Mafra, L.L.; Bricelj, V.M.; Ward, J.E. Mechanisms contributing to low domoic acid uptake by oysters feeding on Pseudo-nitzschia cells. II. Selective rejection. Aquat. Biol. 2009, 6, 213–226. [Google Scholar] [CrossRef]
- Thessen, A.E.; Soniat, T.M.; Dortch, Q.; Doucette, G.J. Crassostrea virginica grazing on toxic and non-toxic diatoms. Toxicon 2010, 55, 570–579. [Google Scholar] [CrossRef]
- Mafra, L.L.; Bricelj, V.M.; Ouellette, C.; Bates, S.S. Feeding mechanics as the basis for differential uptake of the neurotoxin domoic acid by oysters, Crassostrea virginica, and mussels, Mytilus edulis. Aquat. Toxicol. 2010, 97, 160–171. [Google Scholar] [CrossRef]
- De Rijcke, M.; Vandegehuchte, M.B.; Bussche, J.V.; Nevejan, N.; Vanhaecke, L.; De Schamphelaere, K.A.C.; Janssen, C.R. Common European harmful algal blooms affect the viability and innate immune responses of Mytilus edulis larvae. Fish Shellfish. Immunol. 2015, 47, 175–181. [Google Scholar] [CrossRef]
- Jones, T.O.; Whyte, J.N.; Ginther, N.G.; Townsend, L.D.; Iwama, G.K. Haemocyte changes in the Pacific oyster, Crassostrea gigas, caused by exposure to domoic acid in the diatom Pseudonitzschia pungens f. multiseries. Toxicon 1995, 33, 347–353. [Google Scholar] [CrossRef]
- Delegrange, A.; Vincent, D.; Courcot, L.; Amara, R. Testing the vulnerability of juvenile sea bass (Dicentrarchus labrax) exposed to the harmful algal bloom (HAB) species Pseudo-nitzschia delicatissima. Aquaculture 2015, 437, 167–174. [Google Scholar] [CrossRef]
- Marsden, I.D.; Contreras, A.M.; MacKenzie, L.; Munro, M.H.G. A comparison of the physiological responses, behaviour and biotransformation of paralytic shellfish poisoning toxins in a surf-clam (Paphies donacina) and the green-lipped mussel (Perna canaliculus). Mar. Freshw. Res. 2016, 67, 1163–1174. [Google Scholar] [CrossRef]
- Marsden, I.D.; Shumway, S.E. Effects of the toxic dinoflagellate Alexandrium tamarense on the greenshell mussel Perna canaliculus. N. Z. J. Mar. Freshw. Res. 1992, 26, 371–378. [Google Scholar] [CrossRef]
- Rolton, A.; Cawthron Institute, New Zealand. Unpublished data. 2021.
- Contreras, A.M.; Marsden, I.D.; Munro, M.H.G. Effects of short-term exposure to paralytic shellfish toxins on clearance rates and toxin uptake in five species of New Zealand bivalve. Mar. Freshw. Res. 2012, 63, 166–174. [Google Scholar] [CrossRef]
- Contreras, A.M.; Marsden, I.D.; Munro, M.H.G. Physiological effects and biotransformation of PSP toxins in the New Zealand scallop, Pecten novaezelandiae. J. Shellfish. Res. 2012, 31, 1151–1159. [Google Scholar] [CrossRef]
- Gainey, L.F., Jr.; Shumway, S.E. Physiological effects of Protogonyaulax tamarensis on cardiac activity in bivalve molluscs. Comp. Biochem. Physiol. Part C Comp. Pharmacol. 1988, 91, 159–164. [Google Scholar] [CrossRef]
- Lesser, M.P.; Shumway, S.E. Effects of toxic dinoflagellates on clearance rates and survival in juvenile bivalve molluscs. J. Shellfish Res. 1993, 12, 377–381. [Google Scholar]
- Shumway, S.E.; Pierce, F.C.; Knowlton, K. The effects of Protogonyaulax tamarensis on byssus production in Mytilus edulis L., Modiolus modiolus Linnaeus, 1758 and Geukensia demissa Dillwyn. Comp. Biochem. Physiol. A. 1987, 87, 1021–1023. [Google Scholar] [CrossRef]
- Lassudrie, M.; Soudant, P.; Nicolas, J.L.; Miner, P.; Le Grand, J.; Lambert, C.; Le Goïc, N.; Hégaret, H.; Fabioux, C. Exposure to the toxic dinoflagellate Alexandrium catenella modulates juvenile oyster Crassostrea gigas hemocyte variables subjected to different biotic conditions. Fish Shellfish Immunol. 2016, 51, 104–115. [Google Scholar] [CrossRef]
- Yan, T.; Zhou, M.; Fu, M.; Wang, Y.; Yu, R.; Li, J. Inhibition of egg hatching success and larvae survival of the scallop, Chlamys farreri, associated with exposure to cells and cell fragments of the dinoflagellate Alexandrium tamarense. Toxicon 2001, 39, 1239–1244. [Google Scholar] [CrossRef]
- Yan, T.; Zhou, M.; Fu, M.; Yu, R.; Wang, Y.; Li, J. Effects of the dinoflagellate Alexandrium tamarense on early development of the scallop Argopecten irradians concentricus. Aquaculture 2003, 217, 167–178. [Google Scholar] [CrossRef]
- Castrec, J.; Soudant, P.; Payton, L.; Tran, D.; Miner, P.; Lambert, C.; Le Goïc, N.; Huvet, A.; Quillien, V.; Boullot, F.; et al. Bioactive extracellular compounds produced by the dinoflagellate Alexandrium minutum are highly detrimental for oysters. Aquat. Toxicol. 2018, 199, 188–198. [Google Scholar] [CrossRef] [PubMed]
- Castrec, J.; Hégaret, H.; Alunno-Bruscia, M.; Picard, M.; Soudant, P.; Petton, B.; Boulais, M.; Suquet, M.; Quéau, I.; Ratiskol, D.; et al. The dinoflagellate Alexandrium minutum affects development of the oyster Crassostrea gigas, through parental or direct exposure. Environ. Pollut. 2019, 246, 827–836. [Google Scholar] [CrossRef] [PubMed]
- Haberkorn, H.; Lambert, C.; Le Goïc, N.; Moal, J.; Suquet, M.; Guéguen, M.; Sunila, I.; Soudant, P. Effects of Alexandrium minutum exposure on nutrition-related processes and reproductive output in oysters Crassostrea gigas. Harmful Algae 2010, 9, 427–439. [Google Scholar] [CrossRef]
- Haberkorn, H.; Lambert, C.; Le Goïc, N.; Quéré, C.; Bruneau, A.; Riso, R.; Auffret, M.; Soudant, P. Cellular and biochemical responses of the oyster Crassostrea gigas to controlled exposures to metals and Alexandrium minutum. Aquat. Toxicol. 2014, 147, 158–167. [Google Scholar] [CrossRef]
- Fabioux, C.; Sulistiyani, Y.; Haberkorn, H.; Hégaret, H.; Soudant, P. Exposure to toxic Alexandrium minutum activates the antioxidant and detoxifying systems of the oyster Crassostrea Gigas. Harmful Algae 2015, 48, 55–62. [Google Scholar] [CrossRef]
- Lassus, P.; Bardouil, M.; Beliaeff, B.; Masselin, P.; Naviner, M.; Truquet, P. Effect of a continuous supply of the toxic dinoflagellate Alexandrium minutum Halim on the feeding behavior of the Pacific oyster (Crassostrea gigas Thunberg). J. Shellfish. Res. 1999, 18, 211–216. [Google Scholar]
- Mat, A.M.; Haberkorn, H.; Bourdineaud, J.P.; Massabuau, J.C.; Tran, D. Genetic and genotoxic impacts in the oyster Crassostrea gigas exposed to the harmful alga Alexandrium minutum. Aquat. Toxicol. 2013, 140, 458–465. [Google Scholar] [CrossRef]
- Tran, D.; Ciutat, A.; Mat, A.; Massabuau, J.C.; Hégaret, H.; Lambert, C.; Le Goïc, N.; Soudant, P. The toxic dinoflagellate Alexandrium minutum disrupts daily rhythmic activities at gene transcription, physiological and behavioral levels in the oyster Crassostrea gigas. Aquat. Toxicol. 2015, 158, 41–49. [Google Scholar] [CrossRef]
- Le Goïc, N.; Hégaret, H.; Boulais, M.; Béguel, J.P.; Lambert, C.; Fabioux, C.; Soudant, P. Flow cytometric assessment of morphology, viability, and production of reactive oxygen species of Crassostrea gigas oocytes. Application to Toxic dinoflagellate (Alexandrium minutum) exposure. Cytom. Part A 2014, 85, 1049–1056. [Google Scholar] [CrossRef] [PubMed]
- Borcier, E.; Morvezen, R.; Boudry, P.; Miner, P.; Charrier, G.; Laroche, J.; Hégaret, H. Effects of bioactive extracellular compounds and paralytic shellfish toxins produced by Alexandrium minutum on growth and behaviour of juvenile great scallops Pecten maximus. Aquat. Toxicol. 2017, 184, 142–154. [Google Scholar] [CrossRef] [PubMed]
- Taylor, F.J.; Taylor, N.J.; Walsby, J.R. A Bloom of the planktonic diatom, Cerataulina pelagica, off the coast of northeastern New Zealand in 1983, and its contribution to an associated mortality of fish and benthic fauna. Int. Rev. Hydrobiol. 1985, 70, 773–795. [Google Scholar] [CrossRef]
- MacKenzie, L.; Sims, I.; Beuzenberg, V.; Gillespie, P. Mass accumulation of mucilage caused by dinoflagellate polysaccharide exudates in Tasman Bay, New Zealand. Harmful Algae 2002, 1, 69–83. [Google Scholar] [CrossRef]
- Mello, D.F.; Proença, L.A.D.O.; Barracco, M.A. Comparative study of various immune parameters in three bivalve species during a natural bloom of Dinophysis acuminata in Santa Catarina Island, Brazil. Toxins 2010, 2, 1166–1178. [Google Scholar] [CrossRef] [PubMed]
- Simões, E.; Vieira, R.C.; Schramm, M.A.; Mello, D.F.; Pontinha, V.D.A.; Da Silva, P.M.; Barracco, M.A. Impact of harmful algal blooms (Dinophysis acuminata) on the immune system of oysters and mussels from Santa Catarina, Brazil. J. Mar. Biol. Assoc. UK 2015, 95, 773–781. [Google Scholar] [CrossRef]
- Gaillard, S.; Le Goïc, N.; Malo, F.; Boulais, M.; Fabioux, C.; Zaccagnini, L.; Carpentier, L.; Sibat, M.; Réveillon, D.; Séchet, V.; et al. Cultures of Dinophysis sacculus, D. acuminata and pectenotoxin 2 affect gametes and fertilization success of the Pacific oyster, Crassostrea gigas. Environ. Pollut. 2020, 265, 114840. [Google Scholar] [CrossRef]
- Glibert, P.M.; Landsberg, J.H.; Evans, J.J.; Al-Sarawi, M.A.; Faraj, M.; Al-Jarallah, M.A.; Haywood, A.; Ibrahem, S.; Klesius, P.; Powell, C.; et al. A fish kill of massive proportion in Kuwait Bay, Arabian Gulf, 2001: The roles of bacterial disease, harmful algae, and eutrophication. Harmful Algae 2002, 1, 215–231. [Google Scholar] [CrossRef]
- Mee, L.D.; Espinosa, M.; Diaz, G. Paralytic shellfish poisoning with a Gymnodinium catenatum red tide on the Pacific coast of Mexico. Mar. Environ. Res. 1986, 19, 77–92. [Google Scholar] [CrossRef]
- García-Lagunas, N.; Romero-Geraldo, R.; Hernández-Saavedra, N.Y. Genomics study of the exposure effect of Gymnodinium catenatum, a paralyzing toxin producer, on Crassostrea gigas’ defense system and detoxification genes. PLoS ONE 2013, 8, e72323. [Google Scholar] [CrossRef]
- García-Lagunas, N.; de Jesus Romero-Geraldo, R.; Hernández-Saavedra, N.Y. Changes in gene expression and histological injuries as a result of exposure of Crassostrea gigas to the toxic dinoflagellate Gymnodinium catenatum. J. Molluscan Stud. 2016, 82, 193–200. [Google Scholar]
- Matsuyama, Y.; Usuki, H.; Uchida, T.; Kotani, Y. Effects of harmful algae on the early planktonic larvae of the oyster, Crassostrea gigas. In Harmful Algal Blooms 2000; Hallegraeff, G.M., Blackburn, S.I., Bolch, C.J., Lewis, R.J., Eds.; UNESCO: Paris, France, 2001; pp. 411–414. [Google Scholar]
- Estrada, N.; Ascencio, F.; Shoshani, L.; Contreras, R.G. Apoptosis of hemocytes from lions-paw scallop Nodipecten subnodosus induced with paralyzing shellfish poison from Gymnodinium catenatum. Immunobiology 2014, 219, 964–974. [Google Scholar] [CrossRef] [PubMed]
- Estrada, N.; Rodríguez-Jaramillo, C.; Contreras, G.; Ascencio, F. Effects of induced paralysis on hemocytes and tissues of the giant lions-paw scallop by paralyzing shellfish poison. Mar. Biol. 2010, 157, 1401–1415. [Google Scholar] [CrossRef]
- Escobedo-Lozano, A.Y.; Estrada, N.; Ascencio, F.; Contreras, G.; Alonso-Rodriguez, R. Accumulation, biotransformation, histopathology and paralysis in the Pacific calico scallop Argopecten ventricosus by the paralyzing toxins of the dinoflagellate Gymnodinium catenatum. Mar. Drugs 2012, 10, 1044–1065. [Google Scholar] [CrossRef] [PubMed]
- Chang, F.H. Gymnodinium brevisulcatum sp. Nov. (Gymnodiniales, Dinophyceae), a new species isolated from the 1998 summer toxic bloom in Wellington Harbour, New Zealand. Phycologia 1999, 38, 377–384. [Google Scholar] [CrossRef]
- Holland, P.T.; Shi, F.; Satake, M.; Hamamoto, Y.; Ito, E.; Beuzenberg, V.; McNabb, P.; Munday, R.; Briggs, L.; Truman, P.; et al. Novel toxins produced by the dinoflagellate Karenia brevisulcata. Harmful Algae 2012, 13, 47–57. [Google Scholar] [CrossRef]
- Shi, F.; McNabb, P.; Rhodes, L.; Holland, P.; Webb, S.; Adamson, J.; Immers, A.; Gooneratne, R.; Holland, J. The toxic effects of three dinoflagellate species from the genus Karenia on invertebrate larvae and finfish. N. Z. J. Mar. Freshw. Res. 2012, 46, 149–165. [Google Scholar] [CrossRef]
- Neely, T.; Campbell, L. A modified assay to determine hemolytic toxin variability among Karenia clones isolated from the Gulf of Mexico. Harmful Algae 2006, 5, 592–598. [Google Scholar] [CrossRef]
- Li, X.; Yan, T.; Yu, R.; Zhou, M. A review of Karenia mikimotoi: Bloom events, physiology, toxicity and toxic mechanism. Harmful Algae 2019, 90, 101702. [Google Scholar] [CrossRef]
- Sakamoto, S.; Lim, W.A.; Lu, D.; Dai, X.; Orlova, T.; Iwataki, M. Harmful algal blooms and associated fisheries damage in East Asia: Current status and trends in China, Japan, Korea and Russia. Harmful Algae 2021, 102, 101787. [Google Scholar] [CrossRef]
- PIRSA. Fish Kill Investigation: Coffin Bay Harmful Algal (Karenia mikimotoi) Bloom February 2014; Primary Industries and Aquaculture Division, Aquatic Animal Health Unit: Adelaide, Australia, 2014.
- Chang, F.H. A review of knowledge of a group of closely related, economically important toxic Gymnodinium/ Gyrodinium (Dinophyceae) species in New Zealand. J. R. Soc. N. Z. 1996, 26, 381–394. [Google Scholar] [CrossRef]
- Chang, F.H.; Ryan, K.G. Karenia concordia sp. nov.(Gymnodiniales, Dinophyceae), a new nonthecate dinoflagellate isolated from the New Zealand northeast coast during the 2002 harmful algal bloom events. Phycologia 2004, 43, 552–562. [Google Scholar] [CrossRef]
- Smith, K.; Rhodes, L.; Selwood, A.; Marfell, M.; Zeewoldt, C. Massive Karenia mikimotoi bloom in Northland, New Zealand: Use of traditional and molecular techniques for rapid identification of HAB species. Harmful Algae News 2007, 34, 1–3. [Google Scholar]
- Matsuyama, Y.; Koizumi, Y.; Uchida, T. Effect of harmful phytoplankton on the survival of the abalones. Bull. Nansei Natl. Fish. Res. Inst. 1998, 31, 19–24. [Google Scholar]
- Laabir, M.; Gentien, P. Survival of toxic dinoflagellates after gut passage in the Pacific oyster Crassostrea gigas Thunburg. J. Shellfish Res. 1999, 18, 217–222. [Google Scholar]
- Matsuyama, Y.; Uchida, T.; Honjo, T. Effects of harmful dinoflagellates, gymnodinium mikimotoi and heterocapsa circularisquama, red-tide on filtering rate of bivalve molluscs. Fish. Sci. 1999, 65, 248–253. [Google Scholar] [CrossRef]
- Widdows, J.; Moore, M.N.; Lowe, D.M.; Salkeld, P.N. Some effects of a dinoflagellate bloom (Gyrodinium aureolum) on the mussel, Mytilus edulis. J. Mar. Biol. Assoc. UK 1979, 59, 522–524. [Google Scholar] [CrossRef]
- Hégaret, H.; da Silva, P.M.; Wikfors, G.H.; Lambert, C.; De Bettignies, T.; Shumway, S.E.; Soudant, P. Hemocyte responses of Manila clams, Ruditapes philippinarum, with varying parasite, Perkinsus olseni, severity to toxic-algal exposures. Aquat. Toxicol. 2007, 84, 469–479. [Google Scholar] [CrossRef]
- Lin, J.N.; Yan, T.; Zhang, Q.C.; Wang, Y.F.; Liu, Q.; Zhou, M.J. Effects of Karenia mikimotoi blooms on antioxidant enzymes in gastropod abalone, Haliotis discus hannai. Mar. Sci. 2016, 40, 17–22. [Google Scholar]
- Basti, L.; Nagai, S.; Go, J.; Okano, S.; Nagai, K.; Watanabe, R.; Suzuki, T.; Tanaka, Y. Differential inimical effects of Alexandrium spp. and Karenia spp. on cleavage, hatching, and two larval stages of Japanese pearl oyster Pinctada fucata martensii. Harmful Algae 2015, 43, 1–12. [Google Scholar] [CrossRef]
- Basti, L.; Go, J.; Okano, S.; Higuchi, K.; Nagai, S.; Nagai, K. Sublethal and antioxidant effects of six ichthyotoxic algae on early-life stages of the Japanese pearl oyster. Harmful Algae 2021, 103, 102013. [Google Scholar] [CrossRef] [PubMed]
- Botes, L.; Sym, S.D.; Pitcher, G.C. Karenia cristata sp. nov. and Karenia bicuneiformis sp. nov.(Gymnodiniales, Dinophyceae): Two new Karenia species from the South African coast. Phycologia 2003, 42, 563–571. [Google Scholar] [CrossRef]
- Helm, M.M.; Hepper, B.T.; Spencer, B.E.; Walne, P.R. Lugworm mortalities and a bloom of Gyrodinium aureolum Hulburt in the eastern Irish Sea, autumn 1971. J. Mar. Biol. Assoc. UK 1974, 54, 857–869. [Google Scholar] [CrossRef]
- Go, J.; Nagai, K.; Segawa, S.; Honjo, T. Mortality and impact on life activity of Akoya pearl oysters caused by red tides of dinoflagellate Karenia mikimotoi. Nippon. Suisan Gakkaishi 2016, 82, 737–742. [Google Scholar] [CrossRef]
- MacKenzie LHaywood, A.; Adamson, J.; Truman, P.; Till, D.; Seki, T.; Satake, M.; Yasusmoto, T. Gymnodimine contamination of shellfish in New Zealand. In Harmful and Toxic Algal Blooms; Yasumoto, T., Oshima, Y., Fukuyo, Y., Eds.; Inter-Governmental Oceanographic Commission of UNESCO: Tokyo, Japan, 1996; pp. 97–100. [Google Scholar]
- de Salas, M.F.; Rhodes, L.L.; Mackenzie, L.A.; Adamson, J.E.; Ponikla, K. Gymnodinoid genera Karenia and Takayama (Dinophyceae) in New Zealand coastal waters. N. Z. J. Mar. Freshw. Res. 2005, 39, 135–139. [Google Scholar] [CrossRef]
- Mardones, J.I.; Norambuena, L.; Paredes, J.; Fuenzalida, G.; Dorantes-Aranda, J.J.; Chang, K.J.L.; Guzmán, L.; Krock, B.; Hallegraeff, G. Unraveling the Karenia selliformis complex with the description of a non-gymnodimine producing Patagonian phylotype. Harmful Algae 2020, 98, 101892. [Google Scholar] [CrossRef]
- Kuroda, H.; Azumaya, T.; Setou, T.; Hasegawa, N. Unprecedented Outbreak of Harmful Algae in Pacific Coastal Waters off Southeast Hokkaido, Japan, during Late Summer 2021 after Record-Breaking Marine Heatwaves. J. Mar. Sci. Eng. 2021, 9, 1335. [Google Scholar] [CrossRef]
- MacKenzie, L.; Cawthron Institute, New Zealand. Unpublished data. 2021.
- da Silva, P.M.; Hégaret, H.; Lambert, C.; Wikfors, G.H.; Le Goïc, N.; Shumway, S.E.; Soudant, P. Immunological responses of the Manila clam (Ruditapes philippinarum) with varying parasite (Perkinsus olseni) burden, during a long-term exposure to the harmful alga, Karenia selliformis, and possible interactions. Toxicon 2008, 51, 563–573. [Google Scholar] [CrossRef]
- de Salas, M.F.; Bolch, C.J.; Hallegraeff, G.M. Karenia umbella sp. nov. (Gymnodiniales, Dinophyceae), a new potentially ichthyotoxic dinoflagellate species from Tasmania, Australia. Phycologia 2004, 43, 166–175. [Google Scholar] [CrossRef]
- Rhodes, L.; Munday, R.; Briggs, L. Ostreopsis siamensis and palytoxin-related compounds in New Zealand: A risk to human health? In Proceedings of the 12th International Conference on Harmful Algae, Copenhagen, Denmark, 4–8 September 2006; International Society for the Study of Harmful Algae and Intergovernmental Oceanographic Commission of UNESCO: Copenhagen, Denmark, 2008; pp. 326–329. [Google Scholar]
- Shears, N.T.; Ross, P.M. Blooms of benthic dinoflagellates of the genus Ostreopsis; an increasing and ecologically important phenomenon on temperate reefs in New Zealand and worldwide. Harmful Algae 2009, 8, 916–925. [Google Scholar] [CrossRef]
- Chang, F.H.; Ryan, K.G. Prymnesium calathiferum sp. nov. (Prymnesiophyceae), a new species isolated from Northland, New Zealand. Phycologia 1985, 24, 191–198. [Google Scholar] [CrossRef]
- MacKenzie, L. Toxic and noxious phytoplankton in Big Glory Bay, Stewart Island, New Zealand. J. Appl. Phycol. 1991, 3, 19–34. [Google Scholar] [CrossRef]
- Black, E.A.; Whyth, J.N.C.; Bagshaw, J.W.; Ginther, N.G. The effects of Heterosigma akashiwo on juvenile Oncorhynchus tshawytscha and its implications for fish culture. J. Appl. Ichthyol. 1991, 7, 168–175. [Google Scholar] [CrossRef]
- Gaines, G.; Taylor, F.J.R. A mariculturist’s guide to potentially harmful marine phytoplankton of the Pacific coast North America. Marine Resources Section, Fisheries Branch, B.C. Ministry of the Environment. Inf. Rep. 1986, 10. [Google Scholar]
- Khan, S.; Arakawa, O.; Onoue, Y. Neurotoxins in a toxic red tide of Heterosigma akashiwo (Raphidophyceae) in Kagoshima Bay, Japan. Aquac. Res. 1997, 28, 9–14. [Google Scholar] [CrossRef]
- Banno, K.; Oda, T.; Nagai, K.; Nagai, S.; Tanaka, Y.; Basti, L. Deleterious effects of harmful dinoflagellates and raphidophytes on egg viability and spermatozoa swimming velocity in the Japanese pearl oyster Pinctada fucata martensii. J. Shellfish Res. 2018, 37, 41–48. [Google Scholar] [CrossRef]
- Basti, L.; Nagai, K.; Go, J.; Okano, S.; Oda, T.; Tanaka, Y.; Nagai, S. Lethal effects of ichthyotoxic raphidophytes, Chattonella marina, C. antiqua, and Heterosigma akashiwo, on post-embryonic stages of the Japanese pearl oyster, Pinctada fucata martensii. Harmful Algae 2016, 59, 112–122. [Google Scholar] [CrossRef]
- Wang, L.P.; Yan, T.; Zhou, M.J. Impacts of HAB species Heterosigma akashiwo on early development of the scallop Argopecten irradians Lamarck. Aquaculture 2006, 255, 374–383. [Google Scholar] [CrossRef]
- Keppler, C.J.; Hoguet, J.; Smith, K.; Ringwood, A.H.; Lewitus, A.J. Sublethal effects of the toxic alga Heterosigma akashiwo on the southeastern oyster (Crassostrea virginica). Harmful Algae 2005, 4, 275–285. [Google Scholar] [CrossRef]
- Keppler, C.J.; Lewitus, A.J.; Ringwood, A.H.; Hoguet, J.; Staton, T. Sublethal cellular effects of short-term raphidophyte and brevetoxin exposures on the eastern oyster Crassostrea virginica. Mar. Ecol. Prog. Ser. 2006, 312, 141–147. [Google Scholar] [CrossRef]
- de Boer, M.K.; Boeree, C.; Sjollema, S.B.; de Vries, T.; Rijnsdorp, A.D.; Buma, A.G.J. The toxic effect of the marine raphidophyte Fibrocapsa japonica on larvae of the common flatfish sole (Solea solea). Harmful Algae 2012, 17, 92–101. [Google Scholar] [CrossRef]
- Guiry, M.D.; Guiry, M.D.; Guiry, G.M. AlgaeBase; World-Wide Electronic Publication, National University of Ireland: Galway, Ireland, 2021; Available online: http://www.algaebase.org (accessed on 28 April 2021).
- John, U.; Litaker, R.W.; Montresor, M.; Murray, S.; Brosnahan, M.L.; Anderson, D.M. Formal revision of the Alexandrium tamarense species complex (Dinophyceae) taxonomy: The introduction of five species with emphasis on molecular-based (rDNA) classification. Protist 2014, 165, 779–804. [Google Scholar] [CrossRef] [PubMed]
- Ogata, T.; Kodama, M. Ichthyotoxicity found in cultured media of Protogonyaulax spp. Mar. Biol. 1986, 92, 31–34. [Google Scholar] [CrossRef]
- Katsuo, D.; Yamaguchi, K.; Matsuyama, Y.; Oda, T. A new simple screening method for the detection of cytotoxic substances produced by harmful red tide phytoplankton. Harmful Algae 2007, 6, 790–798. [Google Scholar] [CrossRef]
- Shumway, S.E.; Cucci, T.L.; Gainey, L.; Yentsch, C.M. A preliminary study of the behavioral and physiological effects of Gonyaulax tamarensis on bivalve molluscs. In Toxic Dinoflagellates; Anderson, D.M., White, A.W., Baden, D.G., Eds.; Elsevier: New York, NY, USA, 1985; pp. 389–394. [Google Scholar]
- MacKenzie, L.; (Cawthron Institute, Nelson, New Zealand). Personal communication, 2022.
- Chang, F.H.; Mackenzie, L.; Till, D.; Hannah, D.; Rhodes, L. The first toxic shellfish outbreaks and the associated phytoplankton blooms in early 1993 in New Zealand. In Harmful Marine Algal Bloom; Lassus, P., Arzul, G., Gentien, P., Marcaillou, C., Eds.; Technique et Documentation, Lavoisier Publishing: Paris, France, 1995; pp. 145–150. [Google Scholar]
- Chang, F.H.; Anderson, D.M.; Kulis, D.M.; Till, D.G. Toxin production of Alexandrium minutum (Dinophyceae) from the bay of Plenty, New Zealand. Toxicon 1997, 35, 393–409. [Google Scholar] [CrossRef]
- MacKenzie, L.; Rhodes, L.L.; Till, D.; Chang, F.H.; Kaspar, H.; Haywood, A.; Kapa, J.; Walker, B. A Gymnodinium sp. bloom and the contamination of shellfish with lipid soluble toxins in New Zealand, January–April 1993. In Harmful Marine Algal Blooms; Lassus, P., Arzul, G., Gentien, P., Marcaillou, C., Eds.; Technique et Documentation, Lavoisier Publishing: Paris, France, 1995; pp. 795–800. [Google Scholar]
- MacKenzie, L.; Haywood, A.; Adamson, J.; Truman, P.; Till, D.; Satake, M.; Yasumoto, T. Gymnodimine contamination of shellfish in New Zealand 1994. In Proceedings of the Seventh International Conference on Toxic Phytoplankton, Sendai, Japan, 11–16 July 1994. [Google Scholar]
- Lush, G.J.; Negri, A.; Hallegraeff, G.M. Exotoxins produced by the toxic dinoflagellate Alexandrium minutum: Characterisation by radioreceptor and neuroblastoma assays during the growth cycle. In Proceedings of the 9th International Conference-Harmful Algal Blooms 2000, Hobart, Australia, 7–11 February 2000; Hallegraeff, G.M., Blackburn, S.I., Bolch, C.J., Lewis, R.J., Eds.; Intergovernmental Oceanographic Commission of UNESCO: Paris, France, 2001; pp. 268–271. [Google Scholar]
- MacKenzie, L.; Beauchamp, T. Gymnodinium catenatum in New Zealand: A new problem for public health and the shellfish industry. In Cawthron Report; Cawthron Institute: Nelson, New Zealand, 2001; 10p. [Google Scholar]
- Haywood, A.J.; Steidinger, K.; Truby, E.W.; Bergquist, P.; Bergquist, P.L.; Adamson, J.; MacKenzie, L. Comparative morphology and molecular phylogenetic analysis of three new species of the genus Karenia (Dinophyceae) from New Zealand. J. Phycol. 2004, 40, 165–179. [Google Scholar] [CrossRef]
- Rhodes, L.L.; Haywood, A.J.; Ballantine, W.J.; MacKenzie, A.L. Algal blooms and climate anomalies in north-east New Zealand, August-December 1992. N. Z. J. Mar. Freshw. Res. 1993, 27, 419–430. [Google Scholar] [CrossRef]
- Ericson, K. Making Space for Red Tide: Discolored Water and the Early Twentieth Century Bayscape of Japanese Pearl Cultivation. J. Hist. Biol. 2017, 50, 393–423. [Google Scholar] [CrossRef]
- Okaichi, T. Red Tides; Terra Scientific Publishing Company: Tokyo, Japan, 2004; p. 439. [Google Scholar]
- Todd, K. Report for NZ Marine Biotoxin Technical Committee; Cawthron Institute: Nelson, New Zealand, 2002; 30p. [Google Scholar]
- Jasperse, J.A. Marine toxins and New Zealand shellfish. In Proceedings of the Workshop on Research Issues, Wellington, New Zealand, 10–11 June 1993; The Royal Society of New Zealand, Miscellaneous Series 24. 1993. 68p. [Google Scholar]
- Iwataki, M.; Lum, W.M.; Kuwata, K.; Takahashi, K.; Arima, D.; Kuribayashi, T.; Kosaka, Y.; Hasegawa, N.; Watanabe, T.; Shikata, T.; et al. Morphological variation and phylogeny of Karenia selliformis (Gymnodiniales, Dinophyceae) in an intensive cold-water algal bloom in eastern Hokkaido, Japan. Harmful Algae 2022, 114, 102204. [Google Scholar] [CrossRef]
- Mountfort, D.; Beuzenberg, V.; MacKenzie, L.; Rhodes, L. Enhancement of growth and gymnodimine production by the marine dinoflagellate, Karenia Selliformis. Harmful Algae 2006, 5, 658–664. [Google Scholar] [CrossRef]
- Seki, T.; Satake, M.; Mackenzie, L.; Kaspar, H.F.; Yasumoto, T. Gymnodimine, a new marine toxin of unprecedented structure isolated from New Zealand oysters and the dinoflagellate, Gymnodinium sp. Tetrahedron Lett. 1995, 36, 7093–7096. [Google Scholar] [CrossRef]
- MacKenzie, L.; Beuzenberg, V.; Holland, P.; McNabb, P.; Suzuki, T.; Selwood, A. Pectenotoxin and okadaic acid-based toxin profiles in Dinophysis acuta and Dinophysis acuminata from New Zealand. Harmful Algae 2005, 4, 75–85. [Google Scholar] [CrossRef]
- Mackenzie, L. A long-term time series of Dinophysis acuminata blooms and associated shellfish toxin contamination in Port Underwood, Marlborough Sounds, New Zealand. Toxins 2019, 11, 74. [Google Scholar] [CrossRef]
- Basti, L.; Uchida, H.; Kanamori, M.; Matsushima, R.; Suzuki, T.; Nagai, S. Mortality and pathology of Japanese scallop, Patinopecten (Mizuhopecten) yessoensis, and noble scallop, Mimachlamys nobilis, fed monoclonal culture of PTX-producer, Dinophysis caudata. In Marine and Freshwater Harmful Algae, Proceedings of the 16th International Conference on Harmful Algae, Wellington, New Zealand, 27–30 October 2014; MacKenzie, A.L., Ed.; Cawthron Institute, Nelson, New Zealand and International Society for the Study of Harmful Algae: Wellington, New Zealand, 2014; pp. 105–108. [Google Scholar]
- Rountos, K.J.; Kim, J.J.; Hattenrath-Lehmann, T.K.; Gobler, C.J. Effects of the harmful algae, Alexandrium catenella and Dinophysis acuminata, on the survival, growth, and swimming activity of early life stages of forage fish. Mar. Environ. Res. 2019, 148, 46–56. [Google Scholar] [CrossRef]
- Rhodes, L.; Jiang, W.; Knight, B.; Adamson, J.; Smith, K.; Langi, V.; Edgar, M. The genus Pseudo-nitzschia (Bacillariophyceae) in New Zealand: Analysis of the last decade’s monitoring data. N. Z. J. Mar. Freshw. Res. 2013, 47, 490–503. [Google Scholar] [CrossRef]
- Nishimura, T.; Murray, J.S.; Boundy, M.J.; Balci, M.; Bowers, H.A.; Smith, K.F.; Harwood, D.T.; Rhodes, L.L. Update of the Planktonic Diatom Genus Pseudo-nitzschia in Aotearoa New Zealand Coastal Waters: Genetic Diversity and Toxin Production. Toxins 2021, 13, 637. [Google Scholar] [CrossRef]
- Holland, P.T.; McNabb, P.; Rhodes, L.L.; Selwood, A.I.; Neil, T. Amnesic shellfish poisoning toxins in New Zealand shellfish: Detection of an unusual domoic acid isomer using a newly validated LC–MS/MS method. In Molluscan Shellfish Safety; Xanta de Galicia & IOC of UNESCO: Santiago de Compostela, Spain, 2003; pp. 29–42. [Google Scholar]
- Rhodes, L.L.; Holland, P.T.; Adamson, J.E.; McNabb, P.; Selwood, A.I. Production of a new isomer of domoic acid by New Zealand isolates of the diatom Pseudo-nitzschia australis. In Molluscan Shellfish Safety; Xanta de Galicia & IOC of UNESCO: Santiago de Compostela, Spain, 2003; pp. 43–48. [Google Scholar]
- Liefer, J.D.; Robertson, A.; MacIntyre, H.L.; Smith, W.L.; Dorsey, C.P. Characterization of a toxic Pseudo-nitzschia spp. bloom in the Northern Gulf of Mexico associated with domoic acid accumulation in fish. Harmful Algae 2013, 26, 20–32. [Google Scholar] [CrossRef]
- Scholin, C.A.; Gulland, F.; Doucette, G.J.; Benson, S.; Busman, M.; Chavez, F.P.; Cordaro, J.; DeLong, R.; De Vogelaere, A.; Harvey, J.; et al. Mortality of sea lions along the central California coast linked to a toxic diatom bloom. Nature 2000, 403, 80–84. [Google Scholar] [CrossRef]
- Lefebvre, K.A.; Frame, E.R.; Kendrick, P.S. Domoic acid and fish behavior: A review. Harmful Algae 2012, 13, 126–130. [Google Scholar] [CrossRef]
- Rhodes, L.L.; Mackenzie, A.L.; Kaspar, H.F.; Todd, K.E. Harmful algae and mariculture in New Zealand. ICES J. Mar. Sci. 2001, 58, 398–403. [Google Scholar] [CrossRef]
- Cho, E.S.; Rhodes, L.L.; Kim, H.K. The comparison of two strains of Fibrocapsa japonica (Raphidophyceae) in New Zealand and Japan. J. Fish. Sci. Technol. 1999, 2, 58–65. [Google Scholar]
- Nguyen-Ngoc, L.; Doan-Nhu, H.; Larsen, J.; Phan-Tan, L.; Nguyen, X.V.; Lundholm, N.; Van Chu, T.; Huynh-Thi, D.N. Morphological and genetic analyses of Ostreopsis (Dinophyceae, Gonyaulacales, Ostreopsidaceae) species from Vietnamese waters with a re-description of the type species, O. siamensis. J. Phycol. 2021, 57, 1059–1083. [Google Scholar] [CrossRef] [PubMed]
- Rhodes, L.L.; Towers, N.; Briggs, L.; Munday, R.; Adamson, J. Uptake of palytoxin-like compounds by feeding shellfish with Ostreopsis siamensis (Dinophyceae). N. Z. J. Mar. Freshw. Res. 2002, 36, 631–636. [Google Scholar] [CrossRef]
- Rhodes, L. World-wide occurrence of the toxic dinoflagellate genus Ostreopsis Schmidt. Toxicon 2011, 57, 400–407. [Google Scholar] [CrossRef]
- Rhodes, L.; (Cawthron Institute, Nelson, New Zealand). Personal communication, 2022.
- Shilo, M. The toxic principles of Prymnesium parvum. In The Water Environment, Algal Toxins and Health; Carmichael, W.W., Ed.; Plenum Press: London, UK, 1981; pp. 37–47. [Google Scholar]
- Wagstaff, B.A.; Hems, E.S.; Rejzek, M.; Pratscher, J.; Brooks, E.; Kuhaudomlarp, S.; O’Neill, E.C.; Donaldson, M.I.; Lane, S.; Currie, J.; et al. Insights into toxic Prymnesium parvum blooms: The role of sugars and algal viruses. Biochem. Soc. Trans. 2018, 46, 413–421. [Google Scholar] [CrossRef]
- Jones, J.B.; Rhodes, L.L. Suffocation of pilchards (Sardinops sagax) by a green microalgal bloom, Wellington Harbour, New Zealand, December 1993, N. Z. J. Mar. Freshw. Res. 1994, 28, 379–384. [Google Scholar] [CrossRef]
- Albright, L.J.; Yang, C.Z.; Johnson, S. Sub-lethal concentrations of the harmful diatoms, Chaetoceros concavicornis and C. convolutus, increase mortality rates of penned Pacific salmon. Aquaculture 1993, 117, 215–225. [Google Scholar] [CrossRef]
- López-Cortés, D.J.; Núñez-Vázquez, E.J.; Band-Schmidt, C.J.; Gárate-Lizárraga, I.; Hernández-Sandoval, F.E.; Bustillos-Guzmán, J.J. Mass fish die-off during a diatom bloom in the Bahía de La Paz, Gulf of California. Hidrobiológica 2015, 25, 39–48. [Google Scholar]
- Lassudrie, M.; Soudant, P.; Richard, G.; Henry, N.; Medhioub, W.; da Silva, P.M.; Donval, A.; Bunel, M.; LeGoïc, N.; Lambert, C.; et al. Physiological responses of Manila clams Venerupis (=Ruditapes) philippinarum with varying parasite Perkinsus olseni burden to toxic algal Alexandrium ostenfeldii exposure. Aquat. Toxicol. 2014, 154, 27–38. [Google Scholar] [CrossRef] [PubMed]
- Medhioub, W.; Lassus, P.; Truquet, P.; Bardouil, M.; Amzil, Z.; Sechet, V.; Sibat, M.; Soudant, P. Spirolide uptake and detoxification by Crassostrea gigas exposed to the toxic dinoflagellate Alexandrium ostenfeldii. Aquaculture 2012, 358, 108–115. [Google Scholar] [CrossRef]
- Kim, C.H.; Cho, H.J.; Shin, J.B.; Moon, C.H.; Matsouka, K. Regeneration from hyaline cysts of Cochlodinium polykrikoides (Gymnodiniales, Dinophyceae), a red tide organism along the Korean coast. Phycologia 2002, 41, 667–669. [Google Scholar] [CrossRef]
- Griffith, A.W.; Shumway, S.E.; Gobler, C.J. Differential mortality of North Atlantic bivalve molluscs during harmful algal blooms caused by the dinoflagellate, Cochlodinium (aka Margalefidinium) polykrikoides. Estuaries Coasts 2019, 42, 190–203. [Google Scholar] [CrossRef]
- Tang, Y.Z.; Gobler, C.J. Cochlodinium polykrikoides blooms and clonal isolates from the northwest Atlantic coast cause rapid mortality in larvae of multiple bivalve species. Mar. Biol. 2009, 156, 2601–2611. [Google Scholar] [CrossRef]
- Matsuyama, Y. Harmful Effect of Dinoflagellate Heterocapsa circularisquama on Shellfish Aquaculture in Japan. Jpn. Agric. Res. Q. 1999, 33, 283–293. [Google Scholar]
- Matsuyama, Y. Physiological and ecological studies on harmful dinoflagellate Heterocapsa circularisquama: II. Clarification on toxicity of H. circularisquama and its mechanisms causing shellfish kills. Bull. Fish. Res. Agency. 2003, 9, 13–117. [Google Scholar]
- Basti, L.; Go, J.; Higuchi, K.; Nagai, K.; Segawa, S. Effects of the toxic dinoflagellate Heterocapsa circularisquama on larvae of the pearl oyster Pinctada fucata martensii (Dunker, 1873). J. Shellfish. Res. 2011, 30, 177–186. [Google Scholar] [CrossRef]
- Balci, M.; Nishimura, T.; Smith, K.; Rhodes, L.; Murray, S.; MacKenzie, L. First report of Heterocapsa minima from Aotearoa/New Zealand. Harmful Algae News 2020, 64, 8–9. [Google Scholar]
- Adolf, J.E.; Bachvaroff, T.R.; Deeds, J.R.; Place, A.R. Ichthyotoxic Karlodinium veneficum (Ballantine) J Larsen in the Upper Swan River Estuary (Western Australia): Ecological conditions leading to a fish kill. Harmful Algae 2015, 48, 83–93. [Google Scholar] [CrossRef]
- Deeds, J.R.; Terlizzi, D.E.; Adolf, J.E.; Stoecker, D.K.; Place, A.R. Toxic activity from cultures of Karlodinium micrum (=Gyrodinium galatheanum) (Dinophyceae)—A dinoflagellate associated with fish mortalities in an estuarine aquaculture facility. Harmful Algae 2002, 1, 169–189. [Google Scholar] [CrossRef]
- Goshorn, D.; Deeds, J.R.; Tango, P.; Poukish, C.; Place, A.R.; McGinty, M.; Butler, W.; Luckett, C.; Magnien, R. Occurrence of Karlodinium micrum and its association with fish kills in Maryland estuaries. In Harmful Algae 2002, Florida Fish and Wildlife Conservation Commission; Steidinger, K.A., Landsberg, J.H., Tomas, C.R., Vargo, G.A., Eds.; Florida Institute of Oceanography, and Intergovernmental Oceanographic Commission of UNESCO: St. Petersburg, FL, USA; pp. 361–363.
- Brownlee, E.F.; Sellner, S.G.; Sellner, K.G.; Nonogaki, H.; Adolf, J.E.; Bachvaroff, T.R.; Place, A.R. Responses of Crassostrea virginica (Gmelin) and C. ariakensis (Fujita) to bloom-forming phytoplankton including ichthyotoxic Karlodinium veneficum (Ballantine). J. Shellfish Res. 2008, 27, 581–591. [Google Scholar] [CrossRef]
- Glibert, P.M.; Alexander, J.; Merritt, D.W.; North, E.W.; Stoecker, D.K. Harmful algae pose additional challenges for oyster restoration: Impacts of the harmful algae Karlodinium veneficum and Prorocentrum minimum on early life stages of the oysters Crassostrea virginica and Crassostrea ariakensis. J. Shellfish Res. 2007, 26, 919–925. [Google Scholar] [CrossRef]
- Pease, S.K.; Reece, K.S.; O’Brien, J.; Hobbs, P.L.; Smith, J.L. Oyster hatchery breakthrough of two HABs and potential effects on larval eastern oysters (Crassostrea virginica). Harmful Algae 2021, 101, 101965. [Google Scholar] [CrossRef] [PubMed]
- Place, A.R.; Brownlee, E.F.; Sellner, S.G.; Sellner, K.G.; Nonogaki, H.; Adolf, J.A.; Bachvaroff, T.R. Bivalve responses to ichthyotoxic Karlodinium veneficum (Ballantine). In Proceedings of the 12th International Conference on Harmful Algae, Copenhagen, Denmark, 4–8 September 2006; pp. 5–8. [Google Scholar]
- Stoecker, D.; Adolf, J.E.; Place, A.R.; Glibert, P.; Meritt, D. Effects of the dinoflagellates Karlodinium veneficum and Prorocentrum minimum on early life history stages of the eastern oyster (Crassostrea virginica). Mar. Biol. 2008, 154, 81–90. [Google Scholar] [CrossRef]
- Galimany, E.; Place, A.R.; Ramón, M.; Jutson, M.; Pipe, R.K. The effects of feeding Karlodinium veneficum (PLY# 103; Gymnodinium veneficum Ballantine) to the blue mussel Mytilus edulis. Harmful Algae 2008, 7, 91–98. [Google Scholar]
- Burkholder, J.M.; Glasgow, H.B., Jr.; Deamer-Melia, N. Overview and present status of the toxic Pfiesteria complex (Dinophyceae). Phycologia 2001, 40, 186–214. [Google Scholar] [CrossRef]
- Kane, A.S.; Oldach, D.; Reimschuessel, R. Fish lesions in the Chesapeake Bay: Pfiesteria-like dinoflagellates and other etiologies. Md. Med. J. 1998, 47, 106–112. [Google Scholar]
- Springer, J.J.; Shumway, S.E.; Burkholder, J.M.; Glasgow, H.B. Interactions between the toxic estuarine dinoflagellate Pfiesteria piscicida and two species of bivalve mollusc. Mar. Ecol. Prog. Ser. 2002, 245, 1–10. [Google Scholar] [CrossRef][Green Version]
- Park, T.G.; Bolch, C.J.S.; Hallegraeff, G.M. Larval Crassostrea bivalve and Artemia brine shrimp bioassays to assess toxicity and micropredation by the heterotrophic dinoflagellates Cryptoperidiniopsis brodyi and Pfiesteria piscicida from Australian waters. J. Plankton Res. 2007, 29, 791–801. [Google Scholar] [CrossRef]
- Rhodes, L.L.; Burkholder, J.M.; Glasgow, H.B.; Rublee, P.A.; Allen, C.; Adamson, J.E. Pfiesteria shumwayae (Pfiesteriaceae) in New Zealand. N. Z. J. Mar. Freshw. Res. 2002, 36, 621–630. [Google Scholar] [CrossRef]
- Shumway, S.E.; Burkholder, J.M.; Springer, J. Effects of the estuarine dinoflagellate Pfiesteria shumwayae (Dinophyceae) on survival and grazing activity of several shellfish species. Harmful Algae 2006, 5, 442–458. [Google Scholar] [CrossRef]
- Pearce, I.; Handlinger, J.H.; Hallegraeff, G.M. Histopathology in Pacific oyster (Crassostrea gigas) spat caused by the dinoflagellate Prorocentrum rhathymum. Harmful Algae 2005, 4, 61–74. [Google Scholar] [CrossRef]
- Johnsen, G.; Dalløkken, R.; Eikrem, W.; Legrand, C.; Aure, J.; Skjoldal, H.R. Eco-physiology, bio-optics and toxicity of the ichthyotoxic Chrysochromulina leadbeateri (Prymnesiophyceae). J. Phycol. 1999, 35, 1465–1476. [Google Scholar] [CrossRef]
- Okaichi, T. Red Tides in the Seto Inland Sea. In Sustainable Development in the Seto Inland Sea, Japan from the Viewpoint of Fisheries; Okaichi, T., Yanagi, T., Eds.; TERRAPUB: Tokyo, Japan, 1997; pp. 251–304. [Google Scholar]
- Hallegraeff, G.M.; Munday, B.L.; Baden, D.G.; Whitney, P.L. Chatonnella marina Raphidophyte Bloom Associated with Mortality of Cultured Bluefin Tuna (Thunnus maccoyii) in South Australia; Reguera, B., Blanco, J., Ferandz, M.L., Wyatt, T., Eds.; Xunta de Galacia and IOC: Santiago de Compostela, Spain, 1998; pp. 93–96. [Google Scholar]
- Marshall, J.A.; Hallegraeff, G.M. Comparative ecophysiology of the harmful alga Chattonella marina (Raphidophyceae) from South Australian and Japanese waters. J. Plankton Res. 1999, 21, 1809–1822. [Google Scholar] [CrossRef]
- Rhodes, L.; Smith, K.; MacKenzie, L.; Moisan, C. Checklist of the planktonic marine dinoflagellates of New Zealand. N. Z. J. Mar. Freshw. Res. 2019, 54, 86–101. [Google Scholar] [CrossRef]
- Sheng, J.; Malkiel, E.; Katz, J.; Adolf, J.E.; Place, A.R. A dinoflagellate exploits toxins to immobilise prey prior to ingestion. Proc. Natl. Acad. Sci. USA 2010, 107, 2082–2087. [Google Scholar] [CrossRef]
- Yang, Z.B.; Hodgkiss, I.J. Hong Kong’s worst “red tide”—Causative factors reflected in a phytoplankton study at Port Shelter station 1998. Harmful Algae 2004, 3, 149–161. [Google Scholar] [CrossRef]
- de Salas, M.F.; Laza-Martínez, A.; Hallegraeff, G.M. Five new species of Karlodinium and one new Takayama (Kareniaceae, Dinophyceae) from open Southern Ocean waters. J. Phycol. 2008, 44, 241–257. [Google Scholar] [CrossRef]
- Rhodes, L.; Smith, K. A checklist of the benthic and epiphytic marine dinoflagellates of New Zealand, including Rangitāhua/Kermadec Islands. N. Z. J. Mar. Freshw. Res. 2018, 53, 258–277. [Google Scholar] [CrossRef]
- Sutton, P.J.H.; Bowen, M. Ocean temperature change around New Zealand over the last 36 years. N. Z. J. Mar. Freshw. Res. 2019, 53, 305–326. [Google Scholar] [CrossRef]
- Salinger, M.J.; Renwick, J.; Behrens, E.; Mullan, A.B.; Diamond, H.J.; Sirguey, P.; Smith, R.; Trought, M.C.T.; Alexander, V.L.; Cullen, N.J.; et al. The unprecedented coupled ocean-atmosphere summer heatwave in the New Zealand region 2017/18: Drivers, mechanisms and impacts. Environ. Res. Lett. 2019, 14, 044023. [Google Scholar] [CrossRef]
- Burkholder, J.M.; Moeller, P.D.R.; Gordon, A.S.; Lewitus, A.J.; Ramsdell, J.S.; Glasgow, H.B.; Marshall, H.G.; Morton, S.L. Status of Pfiesteria science, including tests of Pfiesteria shumwayae strain CCMP2089 for ichthyotoxicity and toxin. In Harmful Algae 2002—Proceedings of the Xth International Conference on Harmful Algae; Steidinger, K.A., Landsberg, J.H., Tomas, C.R., Vargo, G.A., Eds.; Florida Fish and Wildlife Conservation Commission, Florida Inst. Oceanography, and IOC, UNESCO: St. Petersburg, FL, USA, 2004; pp. 50–52. [Google Scholar]
- Rhodes, L.L.; Adamson, J.E.; Rublee, P.; Schaeffer, E. Geographic distribution of Pfiesteria piscicida and P. shumwayae (Pfiesteriaceae) in Tasman Bay and Canterbury, New Zealand (2002–2003). N. Z. J. Mar. Freshw. Res. 2006, 40, 211–220. [Google Scholar] [CrossRef][Green Version]
- Rublee, P.; Allen, C.; Schaefer, E.; Rhodes, L.; Adamson, J.; Lapworth, C.; Burkholder, J.; Glasgow, H. Global distribution of toxic Pfiesteria complex species detected by PCR assay. In Harmful Algae 2002—Proceedings of the Xth International Conference on Harmful Algae, St. Pete Beach, FL, USA, 21–25 October 2002; Steidinger, K.A., Landsberg, J.H., Tomas, C.R., Vargo, G.A., Eds.; Florida Fish and Wildlife Conservation Commission, Florida Institute of Oceanography, and IOC of UNESCO: St. Petersburg, FL, USA, 2004; pp. 320–322. [Google Scholar]
- Dorantes-Aranda, J.J.; Seger, A.; Mardones, J.I.; Nichols, P.D.; Hallegraeff, G.M. Progress in understanding algal bloom-mediated fish kills: The role of superoxide radicals, phycotoxins and fatty acids. PLoS ONE 2015, 10, 0133549. [Google Scholar] [CrossRef]
- Ahumada-Fierro, N.; Garcia-Mendoza, E.; Sandoval-Gil, J.M.; Band-Schimdt, C. Photosynthesis and photoprotection characteristics related ROS production in three Chattonella (Raphidophyceae) species. J. Phycol. 2021, 57, 941–954. [Google Scholar] [CrossRef] [PubMed]
- McNabb, P.; Rhodes, L.; Adamson, J.; Holland, P. Brevetoxin—An elusive toxin in New Zealand waters. Afr. J. Mar. Sci. 2006, 28, 375–377. [Google Scholar] [CrossRef]
- Smith, K.F.; Kohli, G.S.; Murray, S.A.; Rhodes, L.L. Assessment of the metabarcoding approach for community analysis of benthic-epiphytic dinoflagellates using mock communities. N. Z. J. Mar. Freshw. Res. 2017, 51, 555–576. [Google Scholar] [CrossRef]
- Rhodes, L.L.; Kelly, C.; Hall, J.A. Comparison of growth characteristics of New Zealand isolates of the prymnesiophytes Chrysochromulina quadrikonta and C. camellawith those of the ichthyotoxic species C. polylepis. J. Plankton Res. 1994, 16, 69–82. [Google Scholar]
- Broekhuizen, N.; Plew, D.R.; Pinkerton, M.H.; Gall, M.G. Sea temperature rise over the period 2002–2020 in Pelorus Sound, New Zealand–with possible implications for the aquaculture industry. N. Z. J. Mar. Freshw. Res. 2021, 55, 46–64. [Google Scholar] [CrossRef]
- Farrell, H.; Seebacher, F.; O’Connor, W.; Zammit, A.; Harwood, D.T.; Murray, S. Warm temperature acclimation impacts metabolism of paralytic shellfish toxins from Alexandrium minutum in commercial oysters. Glob. Chang. Biol. 2015, 21, 3402–3413. [Google Scholar] [CrossRef]
- Hawkins, A.J.S. Effects of temperature change on ectotherm metabolism and evolution: Metabolic and physiological interrelations underlying the superiority of multi-locus heterozygotes in heterogeneous environments. J. Therm. Biol. 1995, 20, 23–33. [Google Scholar] [CrossRef]
- Peck, L.S.; Webb, K.E.; Bailey, D.M. Extreme sensitivity of biological function to temperature in Antarctic marine species. Funct. Ecol. 2004, 18, 625–630. [Google Scholar] [CrossRef]
- Hégaret, H.; Smolowitz, R.M.; Sunila, I.; Shumway, S.E.; Alix, J.; Dixon, M.; Wikfors, G.H. Combined effects of a parasite, QPX, and the harmful-alga, Prorocentrum minimum on northern quahogs, Mercenaria mercenaria. Mar. Environ. Res. 2010, 69, 337–344. [Google Scholar] [CrossRef] [PubMed]
- Lassudrie, M.; Soudant, P.; Nicolas, J.L.; Fabioux, C.; Lambert, C.; Miner, P.; Le Grand, J.; Petton, B.; Hegaret, H. Interaction between toxic dinoflagellate Alexandrium catenella exposure and disease associated with herpesvirus OsHV-1 μVar in Pacific oyster spat Crassostrea gigas. Harmful Algae 2015, 45, 53–61. [Google Scholar] [CrossRef]
- Griffith, A.W.; Gobler, C.J. Harmful algal blooms: A climate change co-stressor in marine and freshwater ecosystems. Harmful Algae 2020, 91, 101590. [Google Scholar] [CrossRef]
- Karlson, B.; Andersen, P.; Arneborg, L.; Cembella, A.; Eikrem, W.; John, U.; West, J.J.; Klemm, K.; Kobos, J.; Lehtinen, S.; et al. Harmful algal blooms and their effects in coastal seas of Northern Europe. Harmful Algae 2021, 102, 101989. [Google Scholar] [CrossRef]
- Ford, S.E.; Bricelj, V.M.; Lambert, C.; Paillard, C. Deleterious effects of a non PST bioactive compound (s) from Alexandrium tamarense on bivalve hemocytes. Mar. Biol. 2008, 154, 241–253. [Google Scholar] [CrossRef]
- Tillmann, U.; Alpermann, T.L.; da Purificação, R.C.; Krock, B.; Cembella, A. Intra-population clonal variability in allelochemical potency of the toxigenic dinoflagellate Alexandrium tamarense. Harmful Algae 2009, 8, 759–769. [Google Scholar] [CrossRef]
- Beesoo, R.L.; Auzoux Bordenave, S.; Réveillon, D.; Hervé, D.; Hogeveen, K.; Fessard, V.; Lassudrie, M. Ichthyotoxicity and underlying mechanisms of French Karlodinium veneficum strains using in vitro fish gill cell-based bioassays. In Proceedings of the ICHA 19th International Conference on Harmful Algae, La Paz, Mexico, 10–15 October 2021; p. 162. [Google Scholar]
- Rhodes, L.L.; Smith, K.F.; Murray, J.S.; Nishimura, T.; Finch, S.C. Ciguatera fish poisoning: The risk from an Aotearoa/New Zealand perspective. Toxins 2020, 12, 50. [Google Scholar] [CrossRef]
- Condie, S.A.; Oliver, E.C.; Hallegraeff, G.M. Environmental drivers of unprecedented Alexandrium catenella dinoflagellate blooms off eastern Tasmania, 2012–2018. Harmful Algae 2019, 87, 101628. [Google Scholar] [CrossRef]
- Álvarez, G.; Díaz, P.A.; Godoy, M.; Araya, M.; Ganuza, I.; Pino, R.; Álvarez, F.; Rengal, J.; Hernández, C.; Uribe, E.; et al. Paralytic shellfish toxins in surf clams Mesodesma donacium during a large bloom of Alexandrium catenella dinoflagellates associated to an intense shellfish mass mortality. Toxins 2019, 11, 188. [Google Scholar] [CrossRef] [PubMed]
- Bricelj, V.M.; Connell, L.; Konoki, K.; MacQuarrie, S.P.; Scheuer, T.; Catterall, W.A.; Trainer, V.L. Sodium channel mutation leading to saxitoxin resistance in clams increases risk of PSP. Nature 2005, 434, 763–767. [Google Scholar] [CrossRef] [PubMed]
- Starr, M.; Lair, S.; Michaud, S.; Scarratt, M.; Quilliam, M.; Lefaivre, D.; Robert, M.; Wotherspoon, A.; Michaud, R.; Ménard, N.; et al. Multispecies mass mortality of marine fauna linked to a toxic dinoflagellate bloom. PLoS ONE 2017, 12, e01s76299. [Google Scholar] [CrossRef] [PubMed]
- MacKenzie, A.L.; Webber, S.; Watts, A.M.; Tonks, A. Historical deposits of Alexandrium catenella resting cysts in the sediments of Queen Charlotte Sound, New Zealand. N. Z. J. Mar. Fresh. Res. 2016, 50, 195–208. [Google Scholar] [CrossRef]
- Sellner, K.G.; Rensel, J.E. Prevention, control, and mitigation of harmful algal bloom impacts on fish, shellfish, and human consumers. In Harmful Algal Blooms: A compendium Desk Reference; Shumway, S.E., Burkholder, J.A.M., Morton, S.L., Eds.; John Wiley & Sons Ltd.: Hoboken, NJ, USA, 2018; pp. 435–492. [Google Scholar]
- Cruz, R.C.; Reis Costa, P.; Vinga, S.; Krippahl, L.; Lopes, M.B. A Review of recent machine learning advances for forecasting harmful algal blooms and shellfish contamination. J. Mar. Sci. Eng. 2021, 9, 283. [Google Scholar] [CrossRef]
- Mayer, L. Floating Closed Containment. Aquaculture North America. 2019. Available online: https://www.aquaculturenorthamerica.com/floating-closed-containment-2362/ (accessed on 13 April 2022).
- Kozich, J.J.; Westcott, S.L.; Baxter, N.T.; Highlander, S.K.; Schloss, P.D. Development of a dual-index sequencing strategy and curation pipeline for analyzing amplicon sequence data on the MiSeq Illumina sequencing platform. Appl. Environ. Microbiol. 2013, 79, 5112–5120. [Google Scholar] [CrossRef]
- Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet. J. 2011, 17, 10–12. [Google Scholar] [CrossRef]
- Callahan, B.J.; McMurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J.A.; Holmes, S.P. DADA2: High-resolution sample inference from Illumina amplicon data. Nat. Methods 2016, 13, 581. [Google Scholar] [CrossRef]
- Guillou, L.; Bachar, D.; Audic, S.; Bass, D.; Berney, C.; Bittner, L.; Boutte, C.; Burgaud, G.; de Vargas, C.; Decelle, J.; et al. The Protist Ribosomal Reference database (PR2): A catalog of unicellular eukaryote small sub-unit rRNA sequences with curated taxonomy. Nucleic Acids Res. 2012, 41, 597–604. [Google Scholar] [CrossRef]
- Smith, K.F.; Biessy, L.; Argyle, P.A.; Trnski, T.; Halafihi, T.; Rhodes, L.L. Molecular identification of Gambierdiscus and Fukuyoa (Dinophyceae) from environmental samples. Mar. Drugs 2017, 15, 243. [Google Scholar] [CrossRef]
- Bittner, L.; Gobet, A.; Audic, S.; Romac, S.; Egge, E.S.; Santini, S.; Ogata, H.; Probert, I.; Edvardsen, B.; De Vargas, C. Diversity patterns of uncultured Haptophytes unravelled by pyrosequencing in Naples Bay. Mol. Ecol. 2013, 22, 87–101. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Garrity, G.M.; Tiedje, J.M.; Cole, J.R. Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl. Environ. Microbiol. 2007, 73, 5261–5267. [Google Scholar] [CrossRef] [PubMed]
- McMurdie, P.J.; Holmes, S. Phyloseq: An R package for reproducible interactive analysis and graphics of microbiome census data. PLoS ONE 2013, 8, e61217. [Google Scholar] [CrossRef] [PubMed]
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis; Springer: Berlin, Germany, 2016. [Google Scholar]
- Zhan, A.; Hulák, M.; Sylvester, F.; Huang, X.; Adebayo, A.A.; Abbott, C.L.; Adamowicz, S.J.; Heath, D.D.; Cristescu, M.E.; MacIsaac, H.J.; et al. High sensitivity of 454 pyrosequencing for detection of rare species in aquatic communities. Methods Ecol. Evol. 2013, 4, 558–565. [Google Scholar]
- Egge, E.; Bittner, L.; Andersen, T.; Audic, S.; de Vargas, C.; Edvardsen, B. 454 pyrosequencing to describe microbial eukaryotic community composition, diversity and relative abundance: A test for marine haptophytes. PLoS ONE 2013, 8, e74371. [Google Scholar]
- Scholin, C.A.; Herzog, M.; Sogin, M.; Anderson, D.M. Identification of group-specific and strain-specific genetic-markers for globally distributed Alexandrium (Dinophyceae). II. Sequence analysis of a fragment of the LSU rRNA gene. J. Phycol. 1994, 30, 999–1011. [Google Scholar]
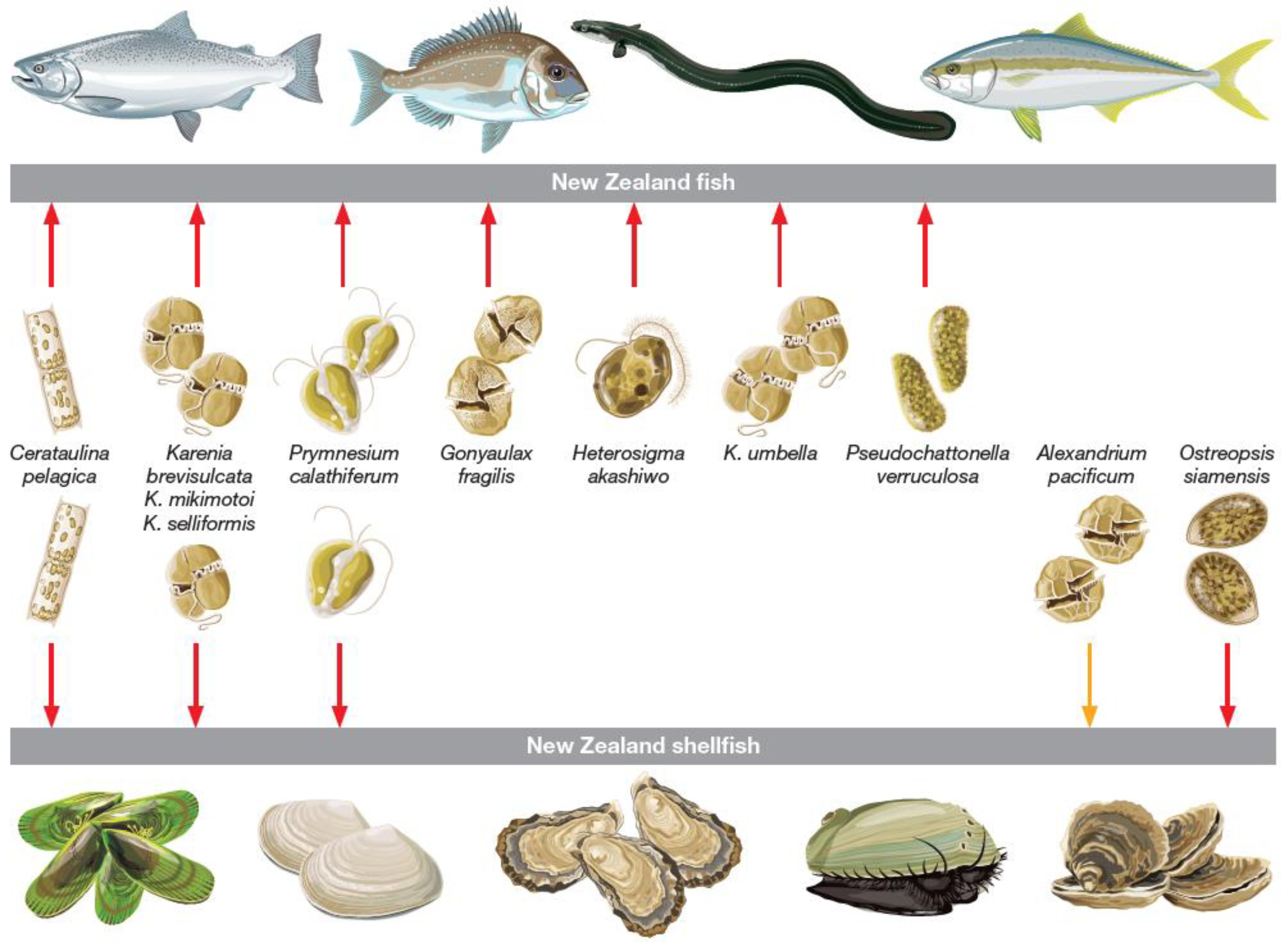

| Class | Genus | Species # | Effects on Shellfish and Fish | Exposure to | Key References |
|---|---|---|---|---|---|
| Bacillariophyceae | Pseudo-nitzschia | Larvae of Pecten maxiumus: growth, development and survival reduced | Domoic Acid (DA) | [46] | |
| P. australis (DA-producer) P. fraudulenta (DA-producer) | Juvenile P. maximus & Crassostrea gigas: Preferential filtration of non-toxic algae C.g.: Reduction in clearance rate (P. australis only) | Whole cell culture | [47] | ||
| P. multiseries (DA-producer) | Juvenile & adult C. virginica: increased pseudofaeces production, lower filtration rates | Whole cell culture | [48,49,50] | ||
| C.v. & Mytilus edulis: selective rejection of toxic cells | Whole cell culture | [51] | |||
| M.e.: no effect on larval survival & development but, increased phenoloxidase production | Whole cell culture & DA | [52] | |||
| Adult C. gigas: Reduction in the number and phagocytic activity of hemocytes | Whole cell culture | [53] | |||
| P. delicatissima (non-toxic) | Adult C.v.: lower filtration rate | Whole cell culture | [50] | ||
| Juvenile sea bass (Dicentrarchus labrax): mucus over production in gills | Whole cell culture | [54] | |||
| Dictyophyceae | Pseudochattonella | P. verruculosa (Hara & Chihara) | Mortality of sea-cage salmon, Oncorhynchus tshawytscha | Field bloom | [43] |
| Dinophyceae | Alexandrium | Adult and larvae of various fish spp.: mortalities and impairment of sensory-motor function | Field blooms, whole cell culture, paralytic shellfish toxins & saxitoxins | [19] (and references there in) | |
| Alexandrium pacificum | Adult Perna canaliculus: Reduction in byssus production, reversable temporary increase in oxygen uptake | Whole cell culture | [55,56] (as A. tamarense) | ||
| P.c. spat (2 mm): Reduction in byssal pad production and growth | Whole cell culture | [57] | |||
| Adult Pecten novaezelandiae: clearance rate reduced and altered in other bivalve species. | Whole cell culture | [58,59] (as A. tamarense) | |||
| Adult Argopecten irradians, Geukensia demissa, Mercenaria mercenaria, Mya arenaria, Mytilus edulis, Ostrea edulis, Paphies donacina, Placopecten magellanicus and Spisula solidissima: various effects including shell valve closure, changes in clearance rate, production of mucus, inhibition of byssus production, violent swimming & erratic siphon activity | Whole cell culture | [34,55,60,61,62] (as Gonyaulax tamarensis, Protogonyaulax tamarensis and A. tamarense) | |||
| Juvenile C. gigas: changes in hemocyte parameters | Whole cell culture | [63] (as A. catenella) | |||
| A. irradians larvae: increased mortalities; reduced activity, and growth of larvae; reduced attachment and climbing ability of juveniles | Whole cell culture & saxitoxin | [64,65] (as A. tamarense, strain ATHK) | |||
| Alexandrium minutum | Adult C. gigas: reduced clearance and filtration rate, increased shell micro-closures, inflammatory response in digestive gland, increased circulating hemocyte concentration and phagocytic ability, genotoxic effects and increased detoxification/antioxidant gene expression. Reduced motility and ATP content of sperm, smaller larval size and increased mortalities at settlement | Whole cell culture | [66,67,68,69,70,71,72,73] | ||
| Gametes and larvae of C.g.: increased ROS in oocytes, decrease in sperm viability & fertilization. Reduced larval hatching, swimming, feeding, growth, settlement and survival | Whole cell culture | [16,17,74] | |||
| Adult P. maximus: delayed shell growth, alteration of sensing processes, less efficient escape response, muscular damage | Whole cell culture | [75] | |||
| Cerataulina pelagica | C. pelagica | Mortality of Fin fish & shellfish | Field bloom (lack of oxygen) | [76] | |
| Gonyaulax | Gonyaulax fragilis | Mortality of marine fauna including fin-fish. | Field bloom (slime) | [77] | |
| Dinophysis | Lethal and sub-lethal effects on various fin fish species, including: behavioural changes, poor co-ordination, inactivity, oxidative stress and histological changes in adults, and: reduced hatching, swimming activity, growth & feeding of larvae | Okadaic Acid & dinophysis toxins | [18] (and references there in) | ||
| Dinophysis acuminata | Adult C. gigas, Perna perna & Anomalocardia brasiliana: Changes in the hemocyte immunological parameters, especially in P. perna. Hemocyte infiltration in the digestive gland of C. gigas. | Field bloom | [78,79] | ||
| Gametes of C.g.: increased oocyte mortality & reduced fertilization success | Whole cell culture | [80] | |||
| Gymnodinium | Gymnodinium catenatum | Mortality of finfish | Field blooms | [81,82] | |
| C. gigas spat (3 mm): reduced clearance rate, increased valve closure & pseudofaeces production. Inflammation of the gill and digestive gland | Whole cell culture | [83,84] | |||
| C.g. larvae: no observable effects | Whole cell culture | [85] | |||
| Juvenile Argopecten ventricosus & Nodipecten subnodosus | Whole cell culture | [86,87,88] | |||
| Karenia | K. brevisulcatum (Chang) Gert Hansen & Moestrup | Mortalities of fish and shellfish | Field blooms | [89,90] | |
| Mortality of various larval & juvenile fish and shellfish species (Oncorhynchus tschawytscha, Chrysophrys auratus, P. canaliculus, Evechinus chloroticus and Haliotis iris, Pleurobranchia maculata, C. gigas) | Whole cell culture, cell free culture, SPE extract, purified brevisulcatic acids | [91] | |||
| K. mikimotoi (Miyake & Kominami ex Oda) Gert Hansen & Moestrup | Fish (Sciaenops ocellatus) erythrocytes: hemolytic activity | Crude algal extract | [92] | ||
| Lethal and sublethal effects on finfish and shellfish | Field blooms, whole cell culture | [23,93,94,95] (and references there in) | |||
| Fin fish, eel and abalone mortalities | Field blooms | [96,97,98] | |||
| Various adult shellfish species: reduced clearance rates, changes in immune functions, reduced escape locomotion and paralysis | Field blooms, whole cell culture | [33,61,93,99,100,101,102,103,104] (as Gyrodinium aureolum) | |||
| Various larval shellfish species: embryo, larval and spat mortalities, reduction in activity rate | Whole cell culture, SPE extract, bloom water, filtered bloom water | [85,91,105,106,107,108,109] | |||
| K. sellifomis Haywood, Steidinger & MacKenzie | Mortality of various fin fish & shellfish, especially Haliotis iris, Paphies subtriangulata & P. canaliculus | Field bloom | [110,111] | ||
| Mortality of various fin fish & shellfish | Field bloom | [112,113] (references there in) | |||
| C. gigas larvae: mortalities | Whole cell culture | [114] | |||
| Adult Ruditapes philippinarum: hemocyte variables changed | Whole cell culture | [103,115] | |||
| K. umbella de Salas, Bolch & Hallegraeff | Mortalities of Oncorhynchus mykiss & Salmo salar | Field bloom | [116] | ||
| Ostreopsis | O. cf. siamensis | Mortalities of sea urchins, Evechinus chloroticus | Field bloom | [117,118] | |
| Haptophyceae | Prymnesium | P. calathiferum Chang & Ryan | Fish and shellfish mortalities | Field bloom | [119] |
| Raphidophyceae | Heterosigma | H. akashiwo (Hada) Sournia | Mortality of O. tschawytscha | Field bloom | [41,120] |
| Mortalities of various juvenile and adult fin fish | Field blooms, toxins from blooms | [94,121,122,123] | |||
| Pinctada fucata martensii & Argopecten irradians gametes & larvae: Reduced sperm swimming velocity, increased mortalities & abnormalities, reduced activity of trocophore & D-larvae | Cell free culture, whole cell culture | [124,125,126] | |||
| Adult C. virginica: shell closure, reduction in filtration and increased hepatopancreas lysosomal destabilization | Whole cell culture | [32,127] | |||
| C. virginica and M. mercenaria hemocytes (in vitro): mortality | Whole cell culture, culture filtrate | [33] | |||
| Fibrocapsa | F. japonica | Adult C. virginica: increased lysosomal destabilization in digestive gland | Bloom water | [128] | |
| Solea solea larvae: mortality | Whole cell culture, culture extracts | [129] |
| Class | Genus | Species # | Effects on Shellfish and Fish | Exposure to | Key References |
|---|---|---|---|---|---|
| Dinophyceae | Alexandrium | A. ostenfeldii | Adult Ruditapes philippinarum & C. gigas: tissue inflammatory response, changes in hemocyte morphology, oxidative stress response in the gills | Whole cell culture | [173,174] |
| Cochlodinium (=Margalefidinium) | C. polykrikoides | Mortalities of finfish | Field blooms | [94,175] | |
| Adult Argopecten irradians, C. gigas, M. mercenaria: mortalities and reduced growth | Field blooms | [176] (and references there in) | |||
| Juvenile A. irradians and Cyprinodon variegates: mortalities | Whole cell culture, cell free culture medium | [177] | |||
| Heterocapsa | H. cf. circularisquama Horiguchi | Pinctada fucata, C. gigas, M. galloprovincialis, Venerupis philippinarum, Suculus diversicolor: Adults: Mortalities, reduced filtration rate. Larvae: activity rate, development rate and survival reduced | Field blooms, bloom water, whole cell culture | [29,85,178,179,180] | |
| H. illdefina (Herman & Sweeney) Morrill & Loeblich III | None known | [114,181] | |||
| Karlodinium | K. veneficum (Ballentine) Larsen | Fin fish mortalities | Field bloom, whole cell culture, cell lysate | [182,183,184] | |
| C. virginica & C. ariakensis larvae, spat & juveniles: increased mortalities & abnormalities, reduction in swimming and activity, reduced growth rates. | Whole cell culture | [185,186,187,188,189] | |||
| Juvenile & adult Mytilus edulis & Mercenaria mercenaria: increased hemocyte phagocytosis and ROS production, reduced growth rates | Whole cell culture | [188,190] | |||
| Pfiesteria | Pfiesteria spp. | Fin fish mortalities | Field blooms | [191,192] | |
| P. piscicida Steidinger & Burkholder | A. irradians, C. gigas & C. virginica larvae: mortalities | Whole cells | [193,194] | ||
| P. shumwayae Glasgow & Burkholder (syn. Pseudopfiesteria shumwayae (Glasgow & Burkholder)) | Ichthyotoxic in vitro | Whole cell culture | [195] | ||
| A. irradians, C. virginica, M. mercenaria, Perna viridis: mortalities of larvae & adults | Whole cell culture | [196] | |||
| Prorocentrum | P. rathymum Loeblich, Shirley & Schmidt | C. gigas spat: mortality | Methanol extracts | [197] | |
| Haptophyceae | Chrysochromulina | C. leadbeateri Estep, Davis, Hargreaves & Sieburth | Mortalities of Salmo salar | Field bloom | [27,198] |
| Pavlomulina | P. ranunculiformis Sym, Pienaar & Kawachi | Attaching to C. gigas larvae | whole cell culture | [28] | |
| Raphidophyceae | Chattonella | C. antiqua (Hada) Ono (syn. C. marina var. antiqua (Hada) Demura & Kawachi) | Fin fish mortalities | Field bloom | [94,199] |
| Mortalities Thunnus maccoyii | Field bloom | [200,201] | |||
| Pinctada fucata martensii: reduced sperm swimming velocity, increased larval mortalities & abnormalities, reduced activity | Whole cell culture | [124,125] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rolton, A.; Rhodes, L.; Hutson, K.S.; Biessy, L.; Bui, T.; MacKenzie, L.; Symonds, J.E.; Smith, K.F. Effects of Harmful Algal Blooms on Fish and Shellfish Species: A Case Study of New Zealand in a Changing Environment. Toxins 2022, 14, 341. https://doi.org/10.3390/toxins14050341
Rolton A, Rhodes L, Hutson KS, Biessy L, Bui T, MacKenzie L, Symonds JE, Smith KF. Effects of Harmful Algal Blooms on Fish and Shellfish Species: A Case Study of New Zealand in a Changing Environment. Toxins. 2022; 14(5):341. https://doi.org/10.3390/toxins14050341
Chicago/Turabian StyleRolton, Anne, Lesley Rhodes, Kate S. Hutson, Laura Biessy, Tony Bui, Lincoln MacKenzie, Jane E. Symonds, and Kirsty F. Smith. 2022. "Effects of Harmful Algal Blooms on Fish and Shellfish Species: A Case Study of New Zealand in a Changing Environment" Toxins 14, no. 5: 341. https://doi.org/10.3390/toxins14050341
APA StyleRolton, A., Rhodes, L., Hutson, K. S., Biessy, L., Bui, T., MacKenzie, L., Symonds, J. E., & Smith, K. F. (2022). Effects of Harmful Algal Blooms on Fish and Shellfish Species: A Case Study of New Zealand in a Changing Environment. Toxins, 14(5), 341. https://doi.org/10.3390/toxins14050341





