Assessment of Common Cyanotoxins in Cyanobacteria of Biological Loess Crusts
Abstract
:1. Introduction
2. Results
2.1. Chromatographic Analyses
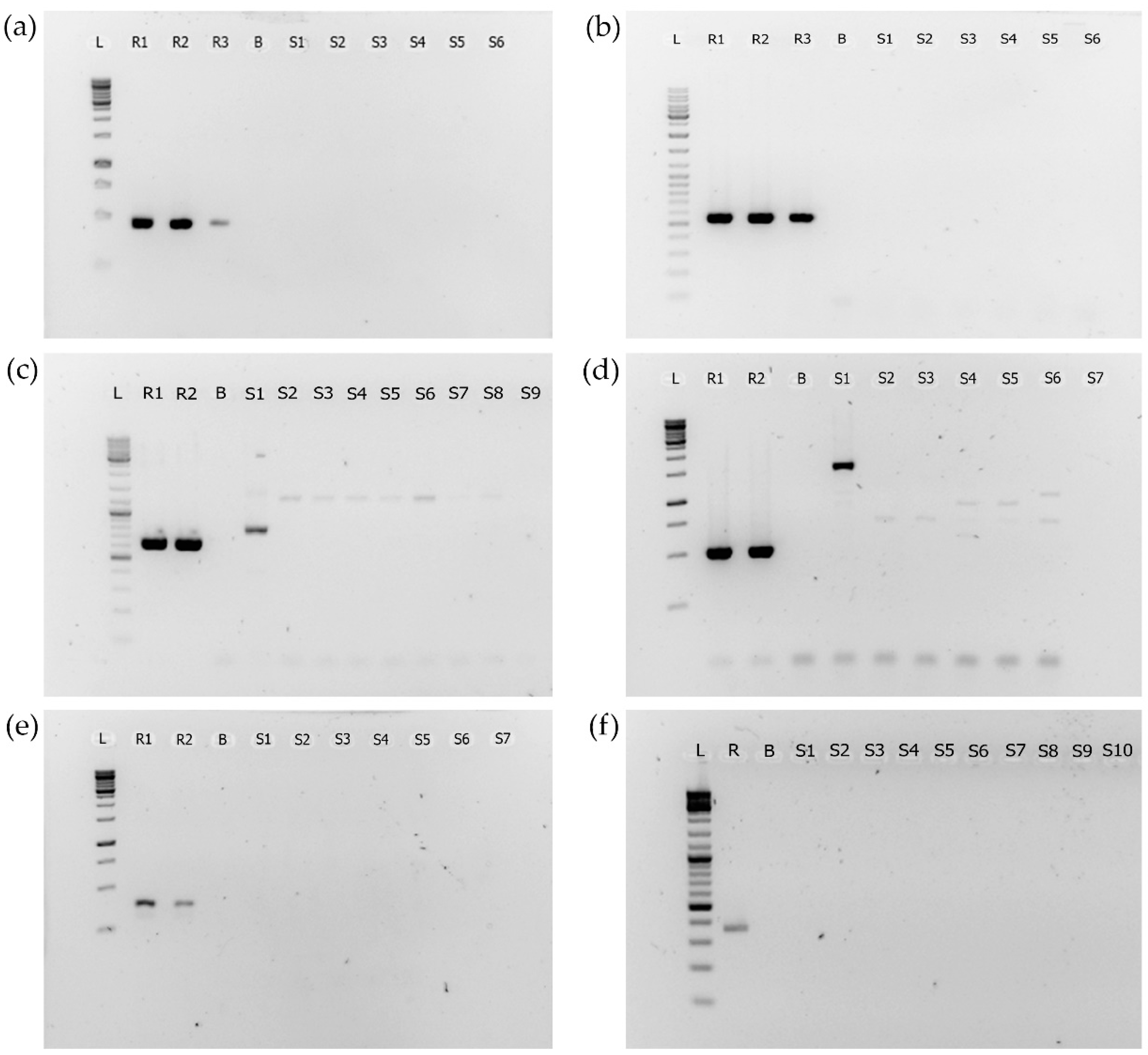
2.2. PCR Analyses
3. Discussion
4. Conclusions
5. Material and Methods
5.1. Biocrust Samples and Cyanobacterial Cultures
5.2. Extraction of Cyanotoxins
5.3. Chromatographic Analyses
5.4. DNA Extraction
5.5. Polymerase Chain Reaction (PCR)
5.6. Reference Strains for PCR Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Codd, G.A.; Meriluoto, J.; Metcalf, J.S. Introduction: Cyanobacteria, cyanotoxins, their human impact, and risk management. In Handbook of Cyanobacterial Monitoring and Cyanotoxin Analysis; Meriluoto, J., Spoof, L., Codd, G.A., Eds.; John Wiley & Sons, Ltd.: Chichester, UK, 2016; pp. 1–8. [Google Scholar] [CrossRef] [Green Version]
- Spoof, L.; Catherine, A. Appendix 3: Tables of microcystins and nodularins. In Handbook of Cyanobacterial Monitoring and Cyanotoxin Analysis; Meriluoto, J., Spoof, L., Codd, G.A., Eds.; John Wiley & Sons, Ltd.: Chichester, UK, 2016; pp. 526–537. [Google Scholar] [CrossRef]
- Svirčev, Z.; Lalić, D.; Savić, G.B.; Tokodi, N.; Backović, D.D.; Chen, L.; Meriluoto, J.; Codd, G.A. Global geographical and historical overview of cyanotoxin distribution and cyanobacterial poisonings. Arch. Toxicol. 2019, 93, 2429–2481. [Google Scholar] [CrossRef] [PubMed]
- Drobac, D.; Tokodi, N.; Simeunović, J.; Baltić, V.; Stanić, D.; Svirčev, Z. Human exposure to cyanotoxins and their effects on health. Arch. Ind. Hyg. Toxicol. 2013, 64, 305–316. [Google Scholar] [CrossRef] [Green Version]
- Prinsep, M.R.; Caplan, F.R.; Moore, R.E.; Patterson, G.M.L.; Honkanen, R.; Boynton, A.L. Microcystin-LA from a blue-green alga belonging to the Stigonematales. Phytochemistry 1992, 31, 1247–1248. [Google Scholar] [CrossRef]
- Honkanen, R.E.; Caplan, F.R.; Baker, K.K.; Baldwin, C.L.; Bobzin, S.C.; Bolis, C.M.; Cabrera, G.M.; Johnson, L.A.; Jung, J.H.; Larsen, L.K.; et al. Protein phosphatase inhibitory activity in extracts of cultured bue-green algae (Cyanophyta). J. Phycol. 1995, 31, 478–486. [Google Scholar] [CrossRef]
- Huang, I.S.; Pietrasiak, N.; Gobler, C.J.; Johansen, J.R.; Burkholder, J.M.; D’Antonio, S.; Zimba, P.V. Diversity of bioactive compound content across 71 genera of marine, freshwater, and terrestrial cyanobacteria. Harmful Algae 2021, 109, 102116. [Google Scholar] [CrossRef] [PubMed]
- Metcalf, J.S.; Richer, R.; Cox, P.A.; Codd, G.A. Cyanotoxins in desert environments may present a risk to human health. Sci. Total Environ. 2012, 421–422, 118–123. [Google Scholar] [CrossRef] [PubMed]
- Chatziefthimiou, A.D.; Banack, S.A.; Cox, P.A. Biocrust-produced cyanotoxins are found vertically in the desert soil profile. Neurotox. Res. 2020, 39, 42–48. [Google Scholar] [CrossRef] [PubMed]
- Metcalf, J.S.; Banack, S.A.; Richer, R.; Cox, P.A. Neurotoxic amino acids and their isomers in desert environments. J. Arid Environ. 2015, 112, 140–144. [Google Scholar] [CrossRef]
- Richer, R.; Banack, S.A.; Metcalf, J.S.; Cox, P.A. The persistence of cyanobacterial toxins in desert soils. J. Arid Environ. 2015, 112, 134–139. [Google Scholar] [CrossRef]
- Faassen, E. Presence of the neurotoxin BMAA in aquatic ecosystems: What do we really know? Toxins 2014, 6, 1109–1138. [Google Scholar] [CrossRef] [Green Version]
- Faassen, E.; Antoniou, M.; Beekman-Lukassen, W.; Blahova, L.; Chernova, E.; Christophoridis, C.; Combes, A.; Edwards, C.; Fastner, J.; Harmsen, J.; et al. A collaborative evaluation of LC-MS/MS based methods for BMAA analysis: Soluble bound BMAA found to be an important fraction. Mar. Drugs 2016, 14, 45. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barger, N.N.; Weber, B.; Garcia-Pichel, F.; Zaady, E.; Belnap, J. Patterns and Controls on Nitrogen Cycling of Biological Soil Crusts. In Biological Soil Crusts: An Organizing Principle in Drylands; Springer International Publishing: Berlin/Heidelberg, Germany, 2016; pp. 257–285. [Google Scholar]
- Belnap, J.; Büdel, B. Biological Soil Crusts as Soil Stabilizers. In Biological Soil Crusts: An Organizing Principle in Drylands; Springer International Publishing: Berlin/Heidelberg, Germany, 2016; pp. 305–320. [Google Scholar]
- Chamizo, S.; Belnap, J.; Eldridge, D.J.; Cantón, Y.; Malam Issa, O. The role of biocrusts in arid land hydrology. In Biological Soil Crusts: An Organizing Principle in Drylands; Springer International Publishing: Berlin/Heidelberg, Germany, 2016; pp. 321–346. [Google Scholar]
- Colesie, C.; Felde, V.J.M.; Büdel, B. Composition and macrostructure of biological soil crusts. In Biological Soil Crusts: An Organizing Principle in Drylands; Weber, B., Büdel, B., Belnap, J., Eds.; Springer International Publishing: Cham, Switzerland, 2016; pp. 159–172. [Google Scholar]
- Sprafke, T.; Obreht, I. Loess: Rock, sediment or soil—What is missing for its definition? Quat. Int. 2016, 399, 198–207. [Google Scholar] [CrossRef]
- Svirčev, Z.; Dulić, T.; Obreht, I.; Codd, G.A.; Lehmkuhl, F.; Marković, S.B.; Hambach, U.; Meriluoto, J. Cyanobacteria and loess—An underestimated interaction. Plant Soil 2019, 439, 293–308. [Google Scholar] [CrossRef] [Green Version]
- Svirčev, Z.; Marković, S.B.; Stevens, T.; Codd, G.A.; Smalley, I.; Simeunović, J.; Obreht, I.; Dulić, T.; Pantelić, D.; Hambach, U. Importance of biological loess crusts for loess formation in semi-arid environments. Quat. Int. 2013, 296, 206–215. [Google Scholar] [CrossRef]
- Obreht, I.; Zeeden, C.; Hambach, U.; Veres, D.; Markovic, S.B.; Lehmkuhl, F. A critical reevaluation of palaeoclimate proxy records from loess in the Carpathian Basin. Earth-Sci. Rev. 2019, 190, 498–520. [Google Scholar] [CrossRef]
- Peterse, F.; Martínez-García, A.; Zhou, B.; Beets, C.J.; Prins, M.A.; Zheng, H.; Eglinton, T.I. Molecular records of continental air temperature and monsoon precipitation variability in East Asia spanning the past 130,000 years. Quat. Sci. Rev. 2014, 83, 76–82. [Google Scholar] [CrossRef]
- Peterse, F.; Prins, M.A.; Beets, C.J.; Troelstra, S.R.; Zheng, H.; Gu, Z.; Schouten, S.; Damsté, J.S.S. Decoupled warming and monsoon precipitation in East Asia over the last deglaciation. Earth Planet. Sci. Lett. 2011, 301, 256–264. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.; Yang, S.; Wang, X.; Hu, J.; Cui, L.; Huang, X.; Jiang, W. Leaf wax n-alkane distributions in Chinese loess since the Last Glacial Maximum and implications for paleoclimate. Quat. Int. 2016, 399, 190–197. [Google Scholar] [CrossRef]
- Shen, J.; Xiao, G.; Wang, Z.; Sun, Q.; Wu, H.; Zhang, C.; Guo, Z. Distribution of n-alkanes in Miocene loess in Qinan, western Chinese Loess Plateau, and its palaeoenvironmental implications. Sci. China Earth Sci. 2017, 60, 921–928. [Google Scholar] [CrossRef]
- Adessi, A.; De Philippis, R.; Rossi, F. Drought-tolerant cyanobacteria and mosses as biotechnological tools to attain land degradation neutrality. Web Ecol. 2021, 21, 65–78. [Google Scholar] [CrossRef]
- Palanački Malešević, T.; Dulić, T.; Obreht, I.; Trivunović, Z.; Marković, R.; Kostić, B.; Važić, T.; Meriluoto, J.; Svirčev, Z. Cyanobacterial potential for restoration of loess surfaces through artificially induced biocrusts. Appl. Sci. 2021, 11, 66. [Google Scholar] [CrossRef]
- Chamizo, S.; Mugnai, G.; Rossi, F.; Certini, G.; De Philippis, R. Cyanobacteria inoculation improves soil stability and fertility on different textured soils: Gaining insights for applicability in soil restoration. Front. Environ. Sci. 2018, 6, 49. [Google Scholar] [CrossRef]
- Lan, S.; Zhang, Q.; Wu, L.; Liu, Y.; Zhang, D.; Hu, C. Artificially accelerating the reversal of desertification: Cyanobacterial inoculation facilitates the succession of vegetation communities. Environ. Sci. Technol. 2014, 48, 307–315. [Google Scholar] [CrossRef] [PubMed]
- Rossi, F.; Li, H.; Liu, Y.; De Philippis, R. Cyanobacterial inoculation (cyanobacterisation): Perspectives for the development of a standardized multifunctional technology for soil fertilization and desertification reversal. Earth-Sci. Rev. 2017, 171, 28–43. [Google Scholar] [CrossRef]
- Dulić, T.; Meriluoto, J.; Malešević, T.P.; Gajić, V.; Važić, T.; Tokodi, N.; Obreht, I.; Kostić, B.; Kosijer, P.; Khormali, F.; et al. Cyanobacterial diversity and toxicity of biocrusts from the Caspian Lowland loess deposits, North Iran. Quat. Int. 2017, 429, 74–85. [Google Scholar] [CrossRef]
- Kurmayer, R.; Sivonen, K.; Wilmotte, A.; Salmaso, N. (Eds.) Molecular Tools for the Detection and Quantification of Toxigenic Cyanobacteria; John Wiley & Sons, Ltd.: Chichester, UK, 2017; p. 440. [Google Scholar]
- Meriluoto, J.; Codd, G.A. Toxic: Cyanobacterial Monitoring and Cyanotoxin Analysis; Åbo Akademi University Press: Turku, Finland, 2005; p. 149. [Google Scholar]
- Meriluoto, J.; Spoof, L.; Codd, G.A. (Eds.) Handbook of Cyanobacterial Monitoring and Cyanotoxin Analysis; John Wiley & Sons, Ltd.: Chichester, UK, 2017; p. 576. [Google Scholar]
- Adamski, M.; Żmudzki, P.; Chrapusta, E.; Bober, B.; Kaminski, A.; Zabaglo, K.; Latkowska, E.; Bialczyk, J. Effect of pH and temperature on the stability of cylindrospermopsin. Characterization of decomposition products. Algal Res. 2016, 15, 129–134. [Google Scholar] [CrossRef]
- Demott, W.R.; Zhang, Q.-X.; Carmichael, W.W. Effects of toxic cyanobacteria and purified toxins on the survival and feeding of a copepod and three species of Daphnia. Limnol. Oceanogr. 1991, 36, 1346–1357. [Google Scholar] [CrossRef] [Green Version]
- Gilbert, J.J. Effect of temperature on the response of planktonic rotifers to a toxic cyanobacterium. Ecology 1996, 77, 1174–1180. [Google Scholar] [CrossRef]
- Jang, M.-H.; Ha, K.; Joo, G.-J.; Takamura, N. Toxin production of cyanobacteria is increased by exposure to zooplankton. Freshw. Biol. 2003, 48, 1540–1550. [Google Scholar] [CrossRef]
- Lampert, W. Inhibitory and toxic effects of blue-green-algae on Daphnia. Int. Rev. Ges. Hydrobiol. 1981, 66, 285–298. [Google Scholar] [CrossRef]
- Lindsay, J.; Metcalf, J.S.; Codd, G.A. Protection against the toxicity of microcystin-LR and cylindrospermopsin in Artemia salina and Daphnia spp. by pre-treatment with cyanobacterial lipopolysaccharide (LPS). Toxicon 2006, 48, 995–1001. [Google Scholar] [CrossRef] [PubMed]
- Maršálek, B.; Bláha, L. Comparison of 17 biotests for detection of cyanobacterial toxicity. Environ. Toxicol. 2004, 19, 310–317. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, J.P.; Cursaro, M.; Froscio, S.M.; Saint, C.P. An examination of the antibiotic effects of cylindrospermopsin on common gram-positive and gram-negative bacteria and the protozoan Naegleria lovaniensis. Environ. Toxicol. 2008, 23, 36–43. [Google Scholar] [CrossRef] [PubMed]
- Figueredo, C.C.; Giani, A.; Bird, D.F. Does allelopathy contribute to Cylindrospermopsis raciborskii (Cyanobacteria) bloom occurence and geographic expansion? J. Phycol. 2007, 43, 256–265. [Google Scholar] [CrossRef]
- Kearns, K.D.; Hunter, M.D. Toxin-producing Anabaena flos-aquae induces settling of Chlamydomonas reinhardtii, a competing motile alga. Microb. Ecol. 2001, 42, 80–86. [Google Scholar] [CrossRef] [PubMed]
- Sedmak, B.; Kosi, G. The role of microcystins in heavy cyanobacterial bloom formation. J. Plankton Res. 1998, 20, 691–708. [Google Scholar] [CrossRef]
- Singh, D.P.; Tyagi, M.B.; Kumar, A.; Thakur, J.K.; Kumar, A. Antialgal activity of a hepatotoxin-producing cyanobacterium, Microcystis aeruginosa. World J. Microbiol. Biotechnol. 2001, 17, 15–22. [Google Scholar] [CrossRef]
- Chia, M.; Kramer, B.; Jankowiak, J.; Bittencourt-Oliveira, M.; Gobler, C. The individual and combined effects of the cyanotoxins, anatoxin-a and microcystin-LR, on the growth, toxin production, and nitrogen fixation of prokaryotic and eukaryotic algae. Toxins 2019, 11, 43. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hu, Z.-Q.; Liu, Y.-D.; Li, D.-H. Physiological and biochemical analyses of microcystin-RR toxicity to the cyanobacterium Synechococcus elongatus. Environ. Toxicol. 2004, 19, 571–577. [Google Scholar] [CrossRef] [PubMed]
- Pflugmacher, S. Promotion of oxidative stress in the aquatic macrophyte Ceratophyllum demersum during biotransformation of the cyanobacterial toxin microcystin-LR. Aquat. Toxicol. 2004, 70, 169–178. [Google Scholar] [CrossRef] [PubMed]
- Romanowska-Duda, Z.A.; Tarczy Ska, M.G. The influence of microcystin-LR and hepatotoxic cyanobacterial extract on the water plant Spirodela oligorrhiza. Environ. Toxicol. 2002, 17, 434–440. [Google Scholar] [CrossRef]
- Saqrane, S.; El Ghazali, I.; Ouahid, Y.; El Hassnib, M.; El Hadrami, I.; Bouarab, L.; del Campo, F.F.; Oudra, B.; Vasconcelos, V. Phytotoxic effects of cyanobacteria extract on the aquatic plant Lemna gibba: Microcystin accumulation, detoxication and oxidative stress induction. Aquat. Toxicol. 2007, 83, 284–294. [Google Scholar] [CrossRef] [PubMed]
- Silva, P.; Vasconcelos, V. Allelopathic effect of Cylindrospermopsis raciborskii extracts on the germination and growth of several plant species. Chem. Ecol. 2010, 26, 263–271. [Google Scholar] [CrossRef]
- Vasas, G.; Gáspár, A.; Surányi, G.; Batta, G.; Gyémánt, G.; M-Hamvas, M.; Máthé, C.; Grigorszky, I.; Molnár, E.; Borbély, G. Capillary electrophoretic assay and purification of cylindrospermopsin, a cyanobacterial toxin from Aphanizomenon ovalisporum, by plant test (Blue-green Sinapis Test). Anal. Biochem. 2002, 302, 95–103. [Google Scholar] [CrossRef] [PubMed]
- Alexova, R.; Fujii, M.; Birch, D.; Cheng, J.; Waite, T.D.; Ferrari, B.C.; Neilan, B.A. Iron uptake and toxin synthesis in the bloom-forming Microcystis aeruginosa under iron limitation. Environ. Microbiol. 2011, 13, 1064–1077. [Google Scholar] [CrossRef]
- Gan, N.; Xiao, Y.; Zhu, L.; Wu, Z.; Liu, J.; Hu, C.; Song, L. The role of microcystins in maintaining colonies of bloom-forming Microcystis spp. Environ. Microbiol. 2012, 14, 730–742. [Google Scholar] [CrossRef]
- Kurmayer, R. The toxic cyanobacterium Nostoc sp. strain 152 produces highest amounts of microcystin and nostophycin under stress conditions. J. Phycol. 2011, 47, 200–207. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oh, H.-M.; Lee, S.J.; Jang, M.-H.; Yoon, B.-D. Microcystin production by Microcystis aeruginosa in a phosphorus-limited chemostat. Appl. Environ. Microbiol. 2000, 66, 176–179. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schatz, D.; Keren, Y.; Vardi, A.; Sukenik, A.; Carmeli, S.; Börner, T.; Dittmann, E.; Kaplan, A. Towards clarification of the biological role of microcystins, a family of cyanobacterial toxins. Environ. Microbiol. 2007, 9, 965–970. [Google Scholar] [CrossRef] [PubMed]
- Berg, K.A.; Lyra, C.; Sivonen, K.; Paulin, L.; Suomalainen, S.; Tuomi, P.; Rapala, J. High diversity of cultivable heterotrophic bacteria in association with cyanobacterial water blooms. ISME J. 2009, 3, 314–325. [Google Scholar] [CrossRef] [PubMed]
- Dziallas, C.; Grossart, H.-P. Increasing oxygen radicals and water temperature select for toxic Microcystis sp. PLoS ONE 2011, 6, e25569. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- El-Shehawy, R.; Gorokhova, E.; Fernández-Piñas, F.; Del Campo, F.F. Global warming and hepatotoxin production by cyanobacteria: What can we learn from experiments? Water Res. 2012, 46, 1420–1429. [Google Scholar] [CrossRef] [PubMed]
- Meissner, S.; Steinhauser, D.; Dittmann, E. Metabolomic analysis indicates a pivotal role of the hepatotoxin microcystin in high light adaptation of Microcystis. Environ. Microbiol. 2015, 17, 1497–1509. [Google Scholar] [CrossRef]
- Pimentel, J.S.M.; Giani, A. Microcystin production and regulation under nutrient stress conditions in toxic Microcystis strains. Appl. Environ. Microbiol. 2014, 80, 5836–5843. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vassilakaki, M.; Pflugmacher, S. Oxidative stress response of Synechocystis sp. (PCC 6803) due to exposure to microcystin-LR and cell-free cyanobacterial crude extract containing microcystin-LR. J. Appl. Phycol. 2008, 20, 219–225. [Google Scholar] [CrossRef]
- Zilliges, Y.; Kehr, J.-C.; Meissner, S.; Ishida, K.; Mikkat, S.; Hagemann, M.; Kaplan, A.; Börner, T.; Dittmann, E. The cyanobacterial hepatotoxin microcystin binds to proteins and increases the fitness of Microcystis under oxidative stress conditions. PLoS ONE 2011, 6, e17615. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rantala, A.; Fewer, D.P.; Hisbergues, M.; Rouhiainen, L.; Vaitomaa, J.; Borner, T.; Sivonen, K. Phylogenetic evidence for the early evolution of microcystin synthesis. Proc. Natl. Acad. Sci. USA 2004, 101, 568–573. [Google Scholar] [CrossRef] [Green Version]
- Murray, S.A.; Mihali, T.K.; Neilan, B.A. Extraordinary conservation, gene loss, and positive selection in the evolution of an ancient neurotoxin. Mol. Biol. Evol. 2011, 28, 1173–1182. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rajeev, L.; Da Rocha, U.N.; Klitgord, N.; Luning, E.G.; Fortney, J.; Axen, S.D.; Shih, P.M.; Bouskill, N.J.; Bowen, B.P.; Kerfeld, C.A.; et al. Dynamic cyanobacterial response to hydration and dehydration in a desert biological soil crust. ISME J. 2013, 7, 2178–2191. [Google Scholar] [CrossRef] [Green Version]
- Van Goethem, M.W.; Osborn, A.R.; Bowen, B.P.; Andeer, P.F.; Swenson, T.L.; Clum, A.; Riley, R.; He, G.; Koriabine, M.; Sandor, L.; et al. Long-read metagenomics of soil communities reveals phylum-specific secondary metabolite dynamics. Commun. Biol. 2021, 4, 1302. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.; Rzymski, P. Programmed cell death-like and accompanying release of microcystin in freshwater bloom-forming cyanobacterium Microcystis: From identification to ecological relevance. Toxins 2019, 11, 706. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Makower, A.K.; Schuurmans, J.M.; Groth, D.; Zilliges, Y.; Matthijs, H.C.P.; Dittmann, E. Transcriptomics-aided dissection of the intracellular and extracellular roles of microcystin in Microcystis aeruginosa PCC 7806. Appl. Environ. Microbiol. 2015, 81, 544–554. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gärtner, G.; Stoyneva-Gärtner, M.; Uzunov, B. Algal toxic compounds and their aeroterrestrial, airborne and other extremophilic producers with attention to soil and plant contamination: A review. Toxins 2021, 13, 322. [Google Scholar] [CrossRef]
- Falfushynska, H.; Horyn, O.; Osypenko, I.; Rzymski, P.; Wejnerowski, L.; Dziuba, M.K.; Sokolova, I.M. Multibiomarker-based assessment of toxicity of central European strains of filamentous cyanobacteria Aphanizomenon gracile and Raphidiopsis raciborskii to zebrafish Danio rerio. Water Res. 2021, 194, 116923. [Google Scholar] [CrossRef]
- Chiswell, R.K.; Shaw, G.R.; Eaglesham, G.; Smith, M.J.; Norris, R.L.; Seawright, A.A.; Moore, M.R. Stability of cylindrospermopsin, the toxin from the cyanobacterium, Cylindrospermopsis raciborskii: Effect of pH, temperature, and sunlight on decomposition. Environ. Toxicol. 1999, 14, 155–161. [Google Scholar] [CrossRef]
- Mazur, H.; Pliński, M. Stability of cyanotoxins, microcystin-LR, microcystin-RR and nodularin in seawater and BG-11 medium of different salinity. Oceanologia 2001, 43, 329–339. [Google Scholar]
- Wörmer, L.; Huerta-Fontela, M.; Cirés, S.; Carrasco, D.; Quesada, A. Natural photodegradation of the cyanobacterial toxins microcystin and cylindrospermopsin. Environ. Sci. Technol. 2010, 44, 3002–3007. [Google Scholar] [CrossRef]
- Efting, A.A.; Snow, D.D.; Fritz, S.C. Cyanobacteria and microcystin in the Nebraska (USA) Sand Hills Lakes before and after modern agriculture. J. Paleolimnol. 2011, 46, 17–27. [Google Scholar] [CrossRef]
- Kaczorowska, A.; Kornijów, R. Palaeoecological evidence for changes over the past 200 years in chironomid communities of a shallow lake exposed to cyanobacterial toxins. Aquat. Ecol. 2012, 46, 465–473. [Google Scholar] [CrossRef] [Green Version]
- Pawlik-Skowrodska, B.; Kornijów, R.; Pirszel, J. Sedimentary imprint of cyanobacterial blooms—A new tool for insight into recent history of lakes. Pol. J. Ecol. 2010, 58, 663–670. [Google Scholar]
- Waters, M.N. A 4700-year history of cyanobacteria toxin production in a shallow subtropical lake. Ecosystems 2016, 19, 426–436. [Google Scholar] [CrossRef]
- Zastepa, A.; Pick, F.R.; Blais, J.M. Distribution and flux of microcystin congeners in lake sediments. Lake Reserv. Manag. 2017, 33, 444–451. [Google Scholar] [CrossRef] [Green Version]
- Zastepa, A.; Taranu, Z.E.; Kimpe, L.E.; Blais, J.M.; Gregory-Eaves, I.; Zurawell, R.W.; Pick, F.R. Reconstructing a long-term record of microcystins from the analysis of lake sediments. Sci. Total Environ. 2017, 579, 893–901. [Google Scholar] [CrossRef] [PubMed]
- Henao, E.; Rzymski, P.; Waters, M. A review on the study of cyanotoxins in paleolimnological research: Current knowledge and future needs. Toxins 2019, 12, 6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rippka, R.; Stanier, R.Y.; Deruelles, J.; Herdman, M.; Waterbury, J.B. Generic assignments, strain histories and properties of pure cultures of cyanobacteria. Microbiology 1979, 111, 1–61. [Google Scholar] [CrossRef] [Green Version]
- Rippka, R. Isolation and Purification of Cyanobacteria; Elsevier: Amsterdam, The Netherlands, 1988; pp. 3–27. [Google Scholar]
- Komárek, J.; Anagnostidis, K. Cyanoprokaryota Teil 1/Part 1: Chroococcales; Springer: Berlin/Heidelberg, Germany, 2008; pp. VI, 548. [Google Scholar]
- Komárek, J.; Anagnostidis, K. Cyanoprokaryota Teil 2/Part 2: Oscillatoriales; Springer: Berlin/Heidelberg, Germany, 2007; pp. IX, 759. [Google Scholar]
- Komárek, J.; Anagnostidis, K. Cyanoprokaryota Teil 3/Part 3: Heterocytous Genera; Springer: Berlin/Heidelberg, Germany, 2013. [Google Scholar]
- Meriluoto, J.; Spoof, L. SOP: Extraction of microcystins in biomass filtered on glass-fibre filters or in freeze-dried cyanobacterial biomass. In Toxic: Cyanobacterial Monitoring and Cyanotoxin Analysis; Meriluoto, J., Codd, G.A., Högnäs, G., Eds.; Acta Academie Aboensis, Series B; Åbo Akademi University Press: Turku, Finland, 2005; pp. 69–71. [Google Scholar]
- Hautala, H.; Lamminmaki, U.; Spoof, L.; Nybom, S.; Meriluoto, J.; Vehniainen, M. Quantitative PCR detection and improved sample preparation of microcystin-producing Anabaena, Microcystis and Planktothrix. Ecotoxicol. Environ. Saf. 2013, 87, 49–56. [Google Scholar] [CrossRef]
- Spoof, L.; Vesterkvist, P.; Lindholm, T.; Meriluoto, J. Screening for cyanobacterial hepatotoxins, microcystins and nodularin in environmental water samples by reversed-phase liquid chromatography–electrospray ionisation mass spectrometry. J. Chromatogr. A 2003, 1020, 105–119. [Google Scholar] [CrossRef]
- Savela, H.; Spoof, L.; Perälä, N.; Preede, M.; Lamminmäki, U.; Nybom, S.; Häggqvist, K.; Meriluoto, J.; Vehniäinen, M. Detection of cyanobacterial sxt genes and paralytic shellfish toxins in freshwater lakes and brackish waters on Åland Islands, Finland. Harmful Algae 2015, 46, 1–10. [Google Scholar] [CrossRef]
- Spoof, L.; Neffling, M.-R.; Meriluoto, J. Fast separation of microcystins and nodularins on narrow-bore reversed-phase columns coupled to a conventional HPLC system. Toxicon 2010, 55, 954–964. [Google Scholar] [CrossRef] [PubMed]
- Gkelis, S.; Rajaniemi, P.; Vardaka, E.; Moustaka-Gouni, M.; Lanaras, T.; Sivonen, K. Limnothrix redekei (Van Goor) Meffert (Cyanobacteria) strains from Lake Kastoria, Greece form a separate phylogenetic group. Microb. Ecol. 2005, 49, 176–182. [Google Scholar] [CrossRef] [PubMed]
- Edwards, U.; Rogall, T.; Blöcker, H.; Emde, M.; Böttger, E.C. Isolation and direct complete nucleotide determination of entire genes. Characterization of a gene coding for 16S ribosomal RNA. Nucleic Acids Res. 1989, 17, 7843–7853. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Taton, A.; Grubisic, S.; Brambilla, E.; Wit, R.D.; Wilmotte, A. Cyanobacterial diversity in natural and artificial microbial mats of Lake Fryxell (McMurdo Dry Valleys, Antarctica): A morphological and molecular approach. Appl. Environ. Microbiol. 2003, 69, 5157–5169. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jungblut, A.-D.; Neilan, B.A. Molecular identification and evolution of the cyclic peptide hepatotoxins, microcystin and nodularin, synthetase genes in three orders of cyanobacteria. Arch. Microbiol. 2006, 185, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Mazmouz, R.; Chapuis-Hugon, F.; Mann, S.; Pichon, V.; Méjean, A.; Ploux, O. Biosynthesis of cylindrospermopsin and 7-epicylindrospermopsin in Oscillatoria sp. strain PCC 6506: Identification of the cyr gene cluster and toxin analysis. Appl. Environ. Microbiol. 2010, 76, 4943–4949. [Google Scholar] [CrossRef] [Green Version]
- Savela, H.; Spoof, L.; Perälä, N.; Vehniäinen, M.; Mankiewicz-Boczek, J.; Jurczak, T.; Kokociński, M.; Meriluoto, J. First report of cyanobacterial paralytic shellfish toxin biosynthesis genes and paralytic shellfish toxin production in Polish freshwater lakes. Adv. Oceanogr. Limnol. 2017, 8, 6319. [Google Scholar] [CrossRef] [Green Version]
- Rantala-Ylinen, A.; Känä, S.; Wang, H.; Rouhiainen, L.; Wahlsten, M.; Rizzi, E.; Berg, K.; Gugger, M.; Sivonen, K. Anatoxin-a synthetase gene cluster of the cyanobacterium Anabaena sp. strain 37 and molecular methods to detect potential producers. Appl. Environ. Microbiol. 2011, 77, 7271–7278. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Possible Biological Functions | References | |
|---|---|---|
| Competitive advantage | Defense mechanism against predators/grazers | [35,36,37,38,39,40,41,42] |
| Competitive interactions with microalgae | [43,44,45,46] | |
| Competitive interactions with cyanobacteria | [43,46,47,48] | |
| Competitive interactions with aquatic and terrestrial plants | [49,50,51,52,53] | |
| Cellular physiology benefits | Tools in the acquisition and retention of nutrients | [44,54,55,56,57,58] |
| Attractants/repellents for heterotrophic microorganisms | [59] | |
| Stress response (infochemicals and radical scavengers) | [58,60,61,62,63,64,65] |
| Toxin | Lowest Cyanotoxin Levels in the Diluted Reference Materials Giving S/N > 3 (pg/µL) * | Corresponding Toxin Levels in Dry Cyanobacterial Material (µg/g) |
|---|---|---|
| dmMC-RR | 3.8 | 0.030 |
| MC-RR | 11.2 | 0.089 |
| MC-YR | 12.4 | 0.097 |
| dmMC-LR | 10.0 | 0.079 |
| MC-LR | 10.7 | 0.085 |
| MC-LY | 11.1 | 0.088 |
| MC-LW | 34.6 | 0.27 |
| MC-LF | 22.0 | 0.18 |
| CYN | 125 | 0.98 |
| STX | 2.4 | 0.002 |
| GTX2/3 ** | 453/171 | 0.36/0.13 |
| Free | SB | Tot | |
|---|---|---|---|
| LOD (µg/g dw) | 0.2 | 0.2 | 2 |
| LOQ (µg/g dw) | 0.5 | 0.6 | 5 |
| Gene | Primer | 5′-3′ Sequence | Annealing t (°C) | Reference |
|---|---|---|---|---|
| 16S RNA | pA 23S30R | AGAGTTTGATCCTGGCTCAG CTTCGCCTCTGTGTGCCTAGGT | 57 | [95,96] |
| mcyE | HEPF HEPR | TTTGGGGTTAACTTTTTTGGGCATAGTC AATTCTTGAGGCTGTAAATCGGGTTT | 61 | [97] |
| cyrJ | cyrJ_F cyrJ_R | TTCTCTCCTTTCCCTATCTCTTTATC GCTACGGTGCTGTACCAAGGGGC | 62 | [98] |
| sxtA | stxA855_F sxtA1480_R | GACTCGGCTTGTTGCTTCCCC GCCAAACTCGCAACAGGAGAAGG | 61 | [92] |
| sxtG | sxtG432_F sxtG928_R | AATGGCAGATCGCAACCGCTAT ACATTCAACCCTGCCCATTCACT | 62 | [92] |
| sxtS | sxtS205_F sxtS566_R | GGAGTATTDGCGGGTGACTATGA GGTGGCTACTTGGTATAACTCGCA | 62 | [99] |
| anaC | anaC-genF anaC-genR | TCTGGTATTCAGTCCCCTCTAT CCCAATAGCCTGTCATCAA | 52 | [100] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dulić, T.; Svirčev, Z.; Palanački Malešević, T.; Faassen, E.J.; Savela, H.; Hao, Q.; Meriluoto, J. Assessment of Common Cyanotoxins in Cyanobacteria of Biological Loess Crusts. Toxins 2022, 14, 215. https://doi.org/10.3390/toxins14030215
Dulić T, Svirčev Z, Palanački Malešević T, Faassen EJ, Savela H, Hao Q, Meriluoto J. Assessment of Common Cyanotoxins in Cyanobacteria of Biological Loess Crusts. Toxins. 2022; 14(3):215. https://doi.org/10.3390/toxins14030215
Chicago/Turabian StyleDulić, Tamara, Zorica Svirčev, Tamara Palanački Malešević, Elisabeth J. Faassen, Henna Savela, Qingzhen Hao, and Jussi Meriluoto. 2022. "Assessment of Common Cyanotoxins in Cyanobacteria of Biological Loess Crusts" Toxins 14, no. 3: 215. https://doi.org/10.3390/toxins14030215
APA StyleDulić, T., Svirčev, Z., Palanački Malešević, T., Faassen, E. J., Savela, H., Hao, Q., & Meriluoto, J. (2022). Assessment of Common Cyanotoxins in Cyanobacteria of Biological Loess Crusts. Toxins, 14(3), 215. https://doi.org/10.3390/toxins14030215






