Comprehensive Review of Aflatoxin Contamination, Impact on Health and Food Security, and Management Strategies in Pakistan
Abstract
1. Introduction
2. Fungal Producers of Aflatoxins
3. Regulation of Aflatoxins in Foods and Animal Feeds
4. Aflatoxin Contamination of Agricultural Products and Foods in Pakistan
4.1. AF Contamination in Cereals
4.2. AF Contamination in Edible Oilseed Crops
4.3. AF Contamination in Nuts and Dried Fruits
4.4. AF Contamination in Spices
4.5. AF Contamination in Animal Feeds
4.6. AF Contamination of Milk
| Commodity | Area of Collection (Year) | AFs | Analytical Method | Contaminated Samples/Total Samples (Incidence %) | Mean | Range | Over EU MTL (%) * | References |
|---|---|---|---|---|---|---|---|---|
| Raw milk | Punjab, Pakistan (2017–2018) | AFM1 | LC | 134/278 (48.2%) | 70.5 ng/L | – | 17.3 | Waqas et al. [189] |
| Branded milk Non-branded | Lahore, Punjab (2016–2017) | AFM1 AFM1 | ELISA ELISA | 23/40 (58) 40/40 (100) | 225.2 ppt 828.4 ppt | 54.30–577.9 ppt 17.34–2735 ppt | 58 95 | Zahra et al. [227] |
| Raw milk | Dairy farms, Punjab (2015) | AFM1 | ELISA | 200/240 (83.33) | 0.59 µg/L | – | 53* | Akbar et al. [191] |
| Raw milk Processed milk | Peri-urban dairy farms (2016) | AFM1 AFM1 | ELISA ELISA | 372/372 (100) 45/45 (100) | 3164 ng/L 558.1 ng/L | LOD–15994 ng/L 7.3–3935.5 ng/L | 93.3 66.7 | Yunus et al. [184] |
| Raw milk | Quetta | AFM1 | ELISA | 88/100 (88) | 257 ng/L | 236.6–292.9 ng/L | 100 | Fahmid et al. [228] |
| Raw milk Processed milk | Punjab (2013–2015) | AFM1 AFM1 | ELISA ELISA | 294/340 (86.6) 152/230 (66.7) | 0.52 ng/mL 0.13 ng/ml | 0.17–1.63 ng/mL 0.01–0.95 ng/mL | 34.45 * 16.66 * | Tahira et al. [229] |
| Raw milk | Punjab, Pakistan (2015) | AFM1 | ELISA | 844/960 (87.9) | 0.642 µg/L | – | 70* | Akbar et al. [216] |
| Raw milk Raw milk TW (Processed) UHT (Processed) | Lahore, Multan | AFM1 AFM1 AFM1 AFM1 | ELISA ELISA ELISA ELISA | 85/94 (90) 52/56 (92) 30/30 (100) 30/30 (100) | 0.232 µg/L 0.139 µg/L 0.113 µg/L 0.164 µg/L | 0.006–0.521 µg/L 0.015–0.554 µg/L 0.013–0.257 µg/L 0.010–0.345 µg/L | 71 73 56 66 | Ahmad et al. [230] |
| Raw milk Pasteurized milk UHT milk | Islamabad (2016) | AFM1 AFM1 AFM1 | ELISA ELISA ELISA | – – – | 1535 ng/L 939.5 ng/L 254.9 ng/L | 1912–7460 ng/L 32.8–4808 ng/L LOD–1536 ng/L | 90 * 55 * 12.9 * | Yunus et al. [224] |
| Milk product (mithai) | Lahore (2017) | AFM1 | HPLC | 152/200 (76) | – | 0.004–1.49 µg/kg | 80 | Naz et al. [231] |
| Raw milk | Local market, Karachi (2016–2017) | AFM1 | ELISA | 143/156 (91.7) | 346.2 ng/L | 20–3090 ng/L | 80.1 | Asghar et al. [232] |
| Raw milk UHT milk Powdered milk Flavored milk Yogurt Flavored yogurt | Summer, Punjab (2014–2015) | AFM1 AFM1 AFM1 AFM1 AFM1 AFM1 | HPLC HPLC HPLC HPLC HPLC HPLC | 19/32 (59.4) 16/25 (64) 9/32 (28.1) 12/25 (48.0) 11/30 (36.6) 10/25 (40) | 94.9 ng/L 75.2 ng/L 65.1 ng/L 45.3 ng/L 59.6 ng/kg 45.3 ng/kg | LOD–229.6 ng/L LOD–190.8 ng/L LOD–178.5 ng/L LOD–110.1 ng/L LOD–158.2 ng/kg LOD–102.5 ng/kg | 37.5 32.0 12.5 20 20 16 | Iqbal et al. [233] |
| Raw milk UHT milk Powdered milk Flavored milk Yogurt Flavored yogurt | Winter, Punjab (2014–2015) | AFM1 AFM1 AFM1 AFM1 AFM1 AFM1 | HPLC HPLC HPLC HPLC HPLC HPLC | 29/42 (69) 26/35 (74.2) 12/32 (37.5) 15/28 (53.3) 15/36 (41.6) 17/30 (56.6) | 129.6 ng/L 98.5 ng/L 89.7 ng/L 46.4 ng/L 63.6 ng/kg 50.5 ng/kg | LOD–345.8 ng/L LOD–302.9 ng/L LOD–278.4 ng/L LOD–198.3 ng/L LOD–196.3 ng/kg LOD–220.5 ng/kg | 38.1 37.1 15.6 21.4 27.7 40 | Iqbal et al. [233] |
| Raw milk | Punjab (2012–2013) | AFM1 | ELISA | 468/485 (96.5) | – | – | 87.22 * | Aslam et al. [225] |
| Raw milk | Punjab (2013–2014) | AFM1 | ELISA | 483/520 (92.8) | 0.06 µg/L | 0.00–0.26 µg/L | 53 | Ismail et al. [234] |
| Raw milk | Sindh | AFM1 | ELISA | 81/84 (96.43) | 0.38 µg/L | 0.01–0.76 µg/L | 70 * | Jawaid et al. [226] |
| Buffalo milk Cow milk | Faisalabad (2013) | AFM1 AFM1 | HPLC HPLC | – – | 0.081 µg/L 0.0655 µg/L | 0.037–0.114 µg/L 0.0243–0.1 µg/L | 84 72 | Sajid et al. [235] |
| Raw milk | Urban farmhouse, Rural farmhouse, Punjab (2011) | AFM1 AFM1 | HPLC HPLC | 38/59 (64) 25/48 (52) | 0.064 µg/L 0.04 µg/L | LOD–0.98 µg/L LOD–0.71 µg/L | 42 27 | Iqbal et al. [236] |
| Milk Yogurt White cheese Cheese cream Butter | Punjab (2010–2011) | AFM1 AFM1 AFM1 AFM1 AFM1 | HPLC HPLC HPLC HPLC HPLC | 76/107 (71) 59/96 (61) 93/119 (78) 89/150 (59) 33/74 (45) | 150.7 ng/L 90.4 ng/kg 147.8 ng/kg 102.6 ng/kg 69.7 ng/kg | 4–845.4 ng/L 4–615.8 ng/kg 4–595.4 ng/kg 4–456.3 ng/kg 4–413.4 ng/kg | 58 47 15 11 52 | Iqbal et al. [237] |
| Raw milk UHT milk Yogurt Butter Ice cream | Central Punjab during summer (2012) | AFM1 AFM1 AFM1 AFM1 AFM1 | HPLC HPLC HPLC HPLC HPLC | 20/56 (36) 12/39 (31) 13/45 (29) 14/35 (40) 9/37 (24) | 0.028 µg/L 0.021 µg/L 0.019µg/kg 0.015µg/kg 0.012µg/kg | LOD–0.89 µg/L LOD–0.51 µg/L LOD–0.88 µg/kg LOD–0.78 µg/kg LOD–0.34 µg/kg | 13 9 8 7 2 | Iqbal et al. [186] |
| Raw milk UHT milk Yogurt Butter Ice cream | Central Punjab during winter (2012) | AFM1 AFM1 AFM1 AFM1 AFM1 | HPLC HPLC HPLC HPLC HPLC | 19/48 (40) 23/45 (51) 19/51 (37) 21/35 (60) 18/42 (43) | 0.073 µg/L 0.060 µg/L 0.053µg/kg 0.036µg/kg 0.021µg/kg | LOD–0.45 µg/L LOD–0.51 µg/L LOD–0.44 µg/kg LOD–0.57 µg/kg LOD–0.67 µg/kg | 27 24 25 34 17 | Iqbal et al. [186] |
| Buffalo milk Cow milk Goat milk Sheep milk Camel milk | Punjab during summer (2009–2010) | AFM1 AFM1 AFM1 AFM1 AFM1 | HPLC HPLC HPLC HPLC HPLC | – – – – – | 0.042 µg/L 0.022 µg/L 0.018 µg/L 0.024 µg/L 0.010 µg/L | 0.025–0.105 µg/L 0.014–0.095 µg/L 0.009–0.088 µg/L 0.012–0.069 µg/L 0.005–0.081 µg/L | 38 33 36 21 14 | Asi et al. [207] |
| Buffalo milk Cow milk Goat milk Sheep milk Camel milk | Punjab during winter (2009–2010) | AFM1 AFM1 AFM1 AFM1 AFM1 | HPLC HPLC HPLC HPLC HPLC | – – – – – | 0.091 µg/L 0.089 µg/L 0.069 µg/L 0.079 µg/L 0.058 µg/L | 0.050–0.200 µg/L 0.065–0.150 µg/L 0.008–0.090 µg/L 0.010–0.098 µg/L 0.012–0.064 µg/L | 55 56 58 32 27 | Asi et al. [207] |
| Raw milk Raw milk Raw milk Sweets | Local shop milk Household milk Dairy farm milk | AFM1 AFM1 AFM1 AFM1 | ELISA ELISA ELISA ELISA | 137/175 (78) 25/40 (62) 15/17 (88) 134/138 (97) | 0.176 µg/L 0.47 µg/L 0.11 µg/L 0.48 µg/kg | 0.002–1.6 µg/L 0.003–1.9 µg/L 0.002–0.794 µg/L 0.01–1.5 µg/kg | 28.6 45 41 78 | Sadia et al. [238] |
| Buffalo milk Buffalo milk Cow milk Cow milk | Punjab KPK Punjab KPK (2009–2010) | AFM1 AFM1 AFM1 AFM1 | HPLC HPLC HPLC HPLC | 22/48 (45.83) 24/46 (52.17) 20/41 (48.78 22/43 (51.16) | 0.040µg/kg 0.066µg/kg 0.030µg/kg 0.045µg/kg | LOD–0.137 µg/kg LOD–0.350 µg/kg LOD–0.062 µg/kg LOD–0.084 µg/kg | 27 42 30 32 | Iqbal et al. [175] |
| Raw milk | Lahore (2007) | AFM1 | HPLC | 68/84 (81) | 17.38 µg/L | 0.69–100 µg/L | 81 * | Khushi et al. [239] |
| Buffalo milk Cow milk Goat milk Sheep milk Camel milk | Faisalabad (2005) | AFM1 AFM1 AFM1 AFM1 AFM1 | HPLC HPLC HPLC HPLC HPLC | 19/55 (34.5) 15/40 (37.5) 6/30 (20) 4/24 (16.7) 0/20 (0) | 0.013 µg/L 0.014 µg/L 0.002 µg/L 0.002 µg/L 0 | – – – – – | 15.8 20 0 0 0 | Hussain et al. [240] |
| Raw milk | Lahore (2007) | AFM1 | HPLC | 68/84 (81) | 17.38 µg/L | 0.69–100 µg/L | 100 | Khushi et al. [239] |
| Buffalo milk Cow milk | Central areas of Punjab (2007) | AFM1 | HPLC | 153/360 (42) 63/120 (52.5) | 0.027 µg/L 0.044 µg/L | – – | 0.6 * | Hussain et al. [209] |
| Raw milk (Dairy animals) | Punjab (2005) | AFM1 | Immunoaffinity columns and Fluorometer | 168/168 (100) | 0.371µg/L | 0.01–0.70 µg/L | 99.4 3 * | Hussain et al. [10] |
5. Dietary Exposure to Aflatoxins
6. Control Strategies for Aflatoxins Contamination
7. Detoxification of Aflatoxins
7.1. Physical Detoxification
7.2. Chemical Detoxification
7.3. Biological Detoxification
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Winter, G.; Pereg, L.A. Review on the relation between soil and mycotoxins: Effect of aflatoxin on field, food and finance. Eur. J. Soil Sci. 2019, 70, 882–897. [Google Scholar] [CrossRef]
- Diedhiou, P.M.; Bandyopadhyay, R.; Atehnkeng, J.; Ojiambo, P.S. Aspergillus colonization and aflatoxin contamination of maize and sesame kernels in two Agro-ecological zones in Senegal. J. Phytopathol. 2011, 159, 268–275. [Google Scholar] [CrossRef]
- Mahmoud, M.A.; Ali, H.M.; El-Aziz, A.R.M.; Al-Othman, M.R.; Al-Wadai, A.S. Molecular characterization of aflatoxigenic and non-aflatoxigenic Aspergillus flavus isolates collected from corn grains. Genet. Mol. Res. 2014, 13, 9352–9370. [Google Scholar] [CrossRef] [PubMed]
- Richard, J.L. Some major mycotoxins and their mycotoxicoses—An overview. Int. J. Food Microbiol. 2007, 119, 3–10. [Google Scholar] [CrossRef]
- Enyiukwu, D.N.; Awurum, A.N.; Nwaneri, J.A. Mycotoxins in stored agricultural products: Implications to food safety and health and prospects of plant-derived pesticides as novel approach to their management. Greener J. Microbiol. Antimicrob. 2014, 2, 32–48. [Google Scholar] [CrossRef]
- Falade, T.D.O.; Neya, A.; Bonkoungou, S.; Dagno, K.; Basso, A.; Senghor, A.L.; Atehnkeng, J.; Ortega-Beltran, A.; Bandyopadhyay, R. Aflatoxin Contamination of Maize, Groundnut, and Sorghum Grown in Burkina Faso, Mali, and Niger and Aflatoxin Exposure Assessment. Toxins 2022, 14, 700. [Google Scholar] [CrossRef]
- Giray, B.; Girgin, G.; Engin, A.B.; Aydın, S.; Sahin, G. Aflatoxin levels in wheat samples consumed in some regions of Turkey. Food Control 2007, 18, 23–29. [Google Scholar] [CrossRef]
- Kuilman, M.E.; Maas, R.F.; Fink-Gremmels, J. Cytochrome P450-mediated metabolism and cytotoxicity of aflatoxin B1 in bovine hepatocytes. Toxicol. In Vitro 2000, 14, 321–327. [Google Scholar] [CrossRef]
- Benkerroum, N. Aflatoxins: Producing-molds, structure, health issues and incidence in Southeast Asian and Sub-Saharan African Countries. Int. J. Environ. Res. Public Health. 2020, 17, 1215. [Google Scholar] [CrossRef]
- Hussain, I.; Anwar, J. A study on contamination of UHT milk by aflatoxin M1 in raw milk in Punjab province of Pakistan. Food Control. 2008, 19, 393–395. [Google Scholar] [CrossRef]
- Santini, A.; Ritieni, A. Aflatoxins: Risk, Exposure and Remediation. Aflatoxins-Recent Advances and Future Prospects; IntechOpen: London, UK, 2013. Available online: https://www.intechopen.com/chapters/39957 (accessed on 30 October 2022). [CrossRef]
- IARC Working Group on the Evaluation of Carcinogenic Risks to Humans. Chemical Agents and Related Occupations. Lyon (FR): International Agency for Research on Cancer; 2012. (IARC Monographs on the Evaluation of Carcinogenic Risks to Humans, No. 100F.). Available online: https://www.ncbi.nlm.nih.gov/books/NBK304416/ (accessed on 5 July 2022).
- Leong, Y.H.; Latiff, A.A.; Ahmad, N.I.; Rosma, A. Exposure measurement of aflatoxins and aflatoxin metabolites in human body fluids. A short review. Mycotoxin Res. 2012, 28, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Williams, J.H.; Phillips, T.D.; Jolly, P.E.; Stiles, J.K.; Jolly, C.M.; Aggarwal, D. Human aflatoxicosis in developing countries: A review of toxicology, exposure, potential health consequences, and interventions. Am. J. Clin. Nutr. 2004, 80, 1106–1122. [Google Scholar] [CrossRef] [PubMed]
- Shivachandra, S.B.; Sah, R.L.; Singh, S.D.; Kataria, J.M.; Manimaran, K. Immunosuppression in broiler chicks fed aflatoxin and inoculated with fowl adenovirus serotype–4 (FAV–4) associated with hydropericardium syndrome. Vet. Res. Commun. 2003, 27, 39–51. [Google Scholar] [CrossRef]
- Quezada, T.; Cuellar, H.; Jaramillo-Juarez, F.; Valdivia, A.G.; Reyes, J.L. Effects of aflatoxin B1 on the liver and kidney of broiler chickens during development. Comp. Biochem. Physiol. C-Pharmacol. Toxicol. Endocrinol. 2000, 125, 265–272. [Google Scholar] [CrossRef]
- Voth-Gaeddert, L.E.; Stoker, M.; Torres, O.; Oerther, D.B. Association of aflatoxin exposure and height-for-age among young children in Guatemala. Int. J. Environ. Health Res. 2018, 28, 280–292. [Google Scholar] [CrossRef] [PubMed]
- Shuaib, F.M.; Jolly, P.E.; Ehiri, J.E.; Yatich, N.; Jiang, Y.; Funkhouser, E.; Person, S.D.; Wilson, C.; Ellis, W.O.; Wang, J.S.; et al. Association between birth outcomes and aflatoxin B1 biomarker blood levels in pregnant women in Kumasi, Ghana. Trop. Med. Int. Health. 2010, 15, 160–167. [Google Scholar] [CrossRef]
- World Bank. 2022. Available online: https://www.worldbank.org/en/topic/agriculture/brief/food-security-update#:~:text=Globally%2C%20hunger%20levels%20remain%20alarmingly,of%20the%20COVID%2D19%20pandemic (accessed on 18 August 2022).
- Singh, U.; Gupta, S.; Gupta, M.A. Review on study on biological ill effects and health hazards f aflatoxins. Asian J. Adv. Med. 2021, 3, 1–8. [Google Scholar]
- Marr, K.A.; Platt, A.; Tornheim, J.A.; Zhang, S.X.; Datta, K.; Cardozo, C.; Garcia-Vidal, C. Aspergillosis complicating severe coronavirus disease. Emerging Infectious Dis. 2021, 27, 18–25. [Google Scholar] [CrossRef]
- International Agency for Research on Cancer. Globocan 2018. IARC. Available online: https://gco.iarc.fr/today/online-analysis-map?v=2020&mode=population&mode_population=continents&population=900&populations=900&key=asr&sex=0&cancer=11&type=0&statistic=5&prevalence=0&population_groupearth&color_palette=default&map_scale=quantile&map_nb_colors=5&continent=0&rotate=%255B10%252C0%255D (accessed on 18 November 2022).
- Liu, Y.; Wu, F. Global burden of aflatoxin-induced hepatocellular carcinoma: A risk assessment. Environ. Health Perspec. 2010, 118, 818–824. [Google Scholar] [CrossRef]
- Raza, S.; Clifford, G.; Franceschi, S. Worldwide variation in the relative importance of hepatitis B and hepatitis C viruses in hepatocellular carcinoma: A systematic review. Br. J. Cancer. 2007, 96, 1127–1134. [Google Scholar] [CrossRef]
- Wu, F.; Groopman, J.D.; Pestka, J.J. Public health impacts of foodborne mycotoxins. Annu. Rev. Food Sci. Technol. 2014, 5, 351–372. [Google Scholar] [CrossRef] [PubMed]
- Butt, A.S.; Abbas, Z.; Jafri, W. Hepatocellular carcinoma in Pakistan: Where do we stand? Hepat. Mon. 2012, 12, e6023. [Google Scholar] [CrossRef] [PubMed]
- Magnussen, A.; Parsi, M.A. Aflatoxins, hepatocellular carcinoma and public health. World J. gastroenterol. 2013, 19, 1508–1512. [Google Scholar] [CrossRef]
- Arshad, A.; Ashfaq, U.A. Epidemiology of hepatitis C infection in Pakistan: Current estimate and major risk factors. Crit. Rev. Eukaryot. Gene Expr. 2017, 27, 63–77. [Google Scholar] [CrossRef] [PubMed]
- Qazi, J.I.; Fayyaz, Z. Aflatoxin contaminated foods and health risk perspective for Pakistani population. Mycopath. 2006, 4, 27–34. [Google Scholar]
- Nasir, U.; Naeem, I.; Asif, M.; Ismail, A.; Gong, Y.Y.; Routledge, M.N.; Amjad, A.; Fazal, A.; Ismail, Z. Assessment of aflatoxins exposure through urinary biomarker approach and the evaluation of the impacts of aflatoxins exposure on the selected health parameters of the children of Multan city of Pakistan. Food Control 2021, 123, 107863. [Google Scholar] [CrossRef]
- Kumar, P.; Mahato, D.K.; Kamle, M.; Mohanta, T.K.; Kang, S.G. Aflatoxins: A global concern for food safety, human health and their management. Front Microbiol. 2017, 7, 2170. [Google Scholar] [CrossRef] [PubMed]
- Reddy, B.N.; Raghavender, C.R. Outbreaks of Aflatoxicoses in India. African J. Food Agri. Nutr. Develop. 2007, 7, 1–15. [Google Scholar] [CrossRef]
- Umar, S.; Munir, M.T.; Shah, M.A.; Shahzad, M.; Khand, R.A.; Sohoo, M.R.; Khan, A.; Ameen, K.; Rafia-Munire, M.; Saleem, F. Outbreak of aflatoxicosis on a local cattle farm in Pakistan. Veterinaria 2015, 3, 13–17. [Google Scholar]
- CAST. Mycotoxins: Risks in Plant, Animal and Human Systems; Report No. 139; Council for Agricultural Science and Technology: Ames, IA, USA, 2003; Available online: https://www.cast-science.org/publication/mycotoxins-risks-in-plant-animal-and-human-systems/ (accessed on 12 December 2021).
- Wu, F. Global impacts of aflatoxin in maize: Trade and human health. World Mycotoxin J. 2015, 8, 137–142. [Google Scholar] [CrossRef]
- Jallow, A.; Xie, H.; Tang, X.; Qi, Z.; Li, P. Worldwide aflatoxin contamination of agricultural products and foods: From occurrence to control. Compr. Rev. Food Sci. Food Saf. 2021, 20, 2332–2381. [Google Scholar] [CrossRef] [PubMed]
- Battilani, P.; Toscano, P.; Van der Fels-Klerx, H.; Moretti, A.; Leggieri, M.C.; Brera, C.; Rortais, A.; Goumperis, T.; Robinson, T. Aflatoxin B1 contamination in maize in Europe increases due to climate change. Sci. Rep. 2016, 6, 24328. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, N.J.; Bowers, E.; Hurburgh, C.; Wu, F. Potential economic losses to the US corn industry from aflatoxin contamination. Food Addit. Contam. Part A 2016, 33, 540–550. [Google Scholar] [CrossRef] [PubMed]
- Moretti, A.; Pascale, M.; Logrieco, A.F. Mycotoxin risks under a climate change scenario in Europe. Trends Food Sci. Technol. 2019, 84, 38–40. [Google Scholar] [CrossRef]
- Cotty, P.J.; Jaime-Garcia, R. Influences of climate on aflatoxin producing fungi and aflatoxin contamination. Int. J. Food Microbiol. 2017, 119, 109–115. [Google Scholar] [CrossRef]
- Iqbal, J.; Asghar, M.A.; Ahmed, A.; Khan, M.A.; Jamil, K. Aflatoxins contamination in Pakistani brown rice: A comparison of TLC, HPLC, LC–MS/MS and ELISA techniques. Toxicol. Mech. Methods. 2014, 24, 544–551. [Google Scholar] [CrossRef]
- Bennett, J.W.; Machida, M.; Gomi, K. An overview of the genus Aspergillus. In Aspergillus: Molecular Biology and Genomics; Machida, M., Gomi, K., Eds.; Academic Press: Caister, UK, 2010; Volume 1, pp. 1–17. [Google Scholar]
- Abbas, H.K.; Wilkinson, J.R.; Zablotowicz, R.M.; Accinelli, C.; Abel, C.A.; Bruns, H.A.; Weaver, M.A. Ecology of Aspergillus flavus, regulation of aflatoxin production and management strategies to reduce aflatoxin contamination of corn. Toxin Rev. 2009, 28, 142–153. [Google Scholar] [CrossRef]
- Saini, S.S.; Kaur, A. Aflatoxin B1: Toxicity, characteristics and analysis. Global Adv. Res. J. Chem. Material Sci. 2012, 1, 63–70. [Google Scholar]
- Klich, M.A. Aspergillus flavus: The major producer of aflatoxin. Mol. Plant Pathol. 2007, 8, 713–722. [Google Scholar] [CrossRef]
- Scheidegger, K.A.; Payne, G.A. Unlocking the secrets behind secondary metabolism: A review of Aspergillus flavus from pathogenicity to functional genomics. J. Toxicol. 2003, 22, 423–459. [Google Scholar] [CrossRef]
- Horn, B.W.; Moore, G.G.; Carbone, I. Sexual reproduction in Aspergillus flavus. Mycologia 2009, 101, 423–429. [Google Scholar] [CrossRef] [PubMed]
- Leger, R.J.S.; Screen, S.E.; Shams-Pirzadeh, B. Lack of host specialization in Aspergillus flavus. App. Environ. Microbiol. 2000, 66, 320–324. [Google Scholar] [CrossRef] [PubMed]
- Abrar, M.; Anjum, F.; Butt, M.; Pasha, I.; Randhawa, M.; Saeed, F.; Waqas, K. Aflatoxins: Biosynthesis, occurrence, toxicity, and remedies. Crit. Rev. Food Sci. Nutr. 2013, 53, 862–874. [Google Scholar] [CrossRef] [PubMed]
- Negash, D. A review of aflatoxin: Occurrence, prevention, and gaps in both food and feed safety. J. Nutr. Health Food Eng. 2018, 8, 190–197. [Google Scholar] [CrossRef]
- Cotty, P.; Mellon, J. Ecology of aflatoxin producing fungi and biocontrol of aflatoxin contamination. Mycotoxin Res. 2006, 22, 110–117. [Google Scholar] [CrossRef]
- Yu, J. Current understanding on aflatoxin biosynthesis and future perspective in reducing aflatoxin contamination. Toxins 2012, 4, 1024–1057. [Google Scholar] [CrossRef]
- Frisvad, J.C.; Hubka, V.; Ezekiel, C.; Hong, S.-B.; Novakova, A.; Chen, A.; Arzanlou, M.; Larsen, T.O.; Sklenar, F.; Mahakarnchanakul, W.; et al. Taxonomy of Aspergillus section Flavi and their production of aflatoxins, ochratoxins and other mycotoxins. Stud. Mycol. 2019, 93, 1–63. [Google Scholar] [CrossRef]
- Norlia, M.; Jinap, S.; Nor-Khaizura MA, R.; Radu, S.; Chin, C.K.; Samsudin NI, P.; Farawahida, A.H. Molecular characterisation of aflatoxigenic and non-aflatoxigenic strains of Aspergillus section Flavi isolated from imported peanuts along the supply chain in Malaysia. Toxins 2019, 11, 501. [Google Scholar] [CrossRef]
- Varga, J.; Frisvad, J.C.; Samson, R.A. Two new aflatoxin producing species, and an overview of Aspergillus section Flavi. Stud. Mycol. 2011, 69, 57–80. [Google Scholar] [CrossRef]
- Schmidt-Heydt, M.; Rufer, C.E.; Abdel-Hadi, A.; Magan, N.; Geisen, R. The production of aflatoxin B1 or G1 by Aspergillus parasiticus at various combinations of temperature and water activity is related to the ratio of aflS to aflR expression. Mycotoxin Res. 2010, 26, 241–246. [Google Scholar] [CrossRef]
- Yunes, N.; de Oliveira, R.; Reis, T.; Baquiao, A.; Rocha, L.; Correa, B. Effect of temperature on growth, gene expression, and aflatoxin production by Aspergillus nomius isolated from Brazil nuts. Mycotoxin Res. 2020, 36, 173–180. [Google Scholar] [CrossRef] [PubMed]
- Lanier, C.; Garon, D.; Heutte, N.; Kientz, V.; André, V. Comparative Toxigenicity and Associated Mutagenicity of Aspergillus fumigatus and Aspergillus flavus Group Isolates Collected from the Agricultural Environment. Toxins 2020, 12, 458. [Google Scholar] [CrossRef] [PubMed]
- Pildain, M.; Frisvad, J.; Vaamonde, G.; Cabral, D.; Varga, J.; Samson, R. Two novel aflatoxin-producing Aspergillus species from Argentinean peanuts. Int. J. System. Evolution. Microbiol. 2008, 58 Pt 3, 725–735. [Google Scholar] [CrossRef] [PubMed]
- Frisvad, J.C.; Skouboe, P.; Samson, R.A. Taxonomic comparison of three different groups of aflatoxin producers and a new efficient producer of aflatoxin B1, sterigmatocystin and 3-Omethylsterigmatocystin, Aspergillus rambellii sp. nov. Systematic Appl. Microbiol. 2005, 28, 442–453. [Google Scholar] [CrossRef] [PubMed]
- Chang, P.K.; Yu, J.; Ehrlich, K.C.; Boue, S.M.; Montalbano, B.G.; Bhatnagar, D.; Cleveland, T.E. adhA in Aspergillus parasiticus is involved in conversion of 5′-hydroxyaverantin to averufin. Appl. Environ. Microbiol. 2000, 66, 4715–4719. [Google Scholar] [CrossRef]
- Waliyar, F.; Umeh, V.; Traore, A.; Osiru, M.; Ntare, B.R.; Diarra, B.; Kodio, O.; Kumar, K.; Sudini, H. Prevalence and distribution of aflatoxin contamination in groundnut (Arachis hypogaea L.) in Mali, West Africa. Crop Prot. 2015, 70, 1–7. [Google Scholar] [CrossRef]
- Giorni, P.; Leggrieri, M.C.; Magan, N.; Battilani, P. Comparison of temperature and moisture requirements for sporulation of Aspergillus flavus sclerotia on natural and artificial substrates. Fungal Biol. 2012, 116, 637–642. [Google Scholar] [CrossRef]
- Hawkins, L.K.; Windham, G.L.; Williams, W.P. Effect of different postharvest drying temperatures on Aspergillus flavus survival and aflatoxin content in five maize hybrids. J. Food Prot. 2005, 68, 1521–1524. [Google Scholar] [CrossRef]
- Siciliano, I.; Berta, F.; Bosio, P.; Gullino, M.L.; Garibaldi, A. Effect of different temperatures and CO2 levels on Alternaria toxins produced on the cultivated rocket, cabbage, and cauliflower. World Mycotoxin J. 2017, 10, 63–71. [Google Scholar] [CrossRef]
- Matumba, L.; Sulyok, M.; Njoroge, S.M.; Ediage, E.N.; Van Poucke, C.; De Saeger, S.; Krska, R. Uncommon occurrence ratios of aflatoxin B1, B2, G1, and G2 in maize and groundnuts from Malawi. Mycotoxin Res. 2015, 31, 57–62. [Google Scholar] [CrossRef]
- Yoshinari, T.; Noda, Y.; Yoda, K.; Sezaki, H.; Nagasawa, H.; Sakuda, S. Inhibitory activity of blasticidin a, a strong aflatoxin production inhibitor, on protein synthesis of yeast: Selective inhibition of aflatoxin production by protein synthesis inhibitors. J. Antibiot. 2010, 63, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Eshelli, M.; Harvey, L.; Edrada-Ebel, R.; McNeil, B. Metabolomics of the bio-degradation process of aflatoxin B1 by actinomycetes at an initial pH of 6.0. Toxins 2015, 7, 439–456. [Google Scholar] [CrossRef] [PubMed]
- Dalie, D.K.D.; Deschamps, A.M.; Richard-Forget, F. Lactic acid bacteria potential for control of mold growth and mycotoxins: A review. Food Control 2010, 21, 370–380. [Google Scholar] [CrossRef]
- Al-Shikli, R.A.; Abdul-rasool, A.A.; Al-Hiti, M.M. Effect of some storage condition upon the survival of some fungal spores. Iraqi J. Pharm. Sci. 2010, 19, 1–10. [Google Scholar] [CrossRef]
- Rushing, B.R.; Selim, M.I. Aflatoxin B1: A review on metabolism, toxicity, occurrence in food, occupational exposure, and detoxification methods. Food Chem. Toxicol. 2019, 124, 81–100. [Google Scholar] [CrossRef] [PubMed]
- Mahbobinejhad, Z.; Aminian, H.; Ebrahimi, L.; Vahdati, K. Reduction of aflatoxin production by exposing Aspergillus flavus to CO2. J. Crop Prot. 2019, 8, 441–448. [Google Scholar]
- Liu, J.; Sun, L.; Zhang, N.; Zhang, J.; Guo, J.; Li, C.; Qi, D. Effects of nutrients in substrates of different grains on aflatoxin B1 production by Aspergillus flavus. BioMed Res. Int. 2016, 2016, 7232858. [Google Scholar] [CrossRef]
- Wang, B.; Han, X.; Bai, Y.; Lin, Z.; Qiu, M.; Nie, X.; Wang, S. Effects of nitrogen metabolism on growth and aflatoxin biosynthesis in Aspergillus flavus. J. Hazard. Mat. 2017, 324 Pt B, 691–700. [Google Scholar] [CrossRef]
- Bolu, S.; Elelu, N.; Risikat, A.; Sola-Ojo, F.; Daramola, K.; Omotosho, V.; Atitebi, A.; Afeni, M. Effect of Vitamins, Amino Acids and Phyto-Active Biomolecules on Aspergillus flavus in Poultry Production. In Effect of Vitamins, Amino Acids and Phyto-Active Biomolecules on Aspergillus flavus in Poultry Production; Intech: London, UK, 2020. [Google Scholar]
- Kinyungu, S.W. Efficacy of Pre-Harvest Aspergillus flavus Biocontrol Treatment on Reducing Aflatoxin Accumulation during Drying. Ph.D. Thesis, Purdue University Graduate School, West Lafayette, IN, USA, 2019. [Google Scholar]
- Manjunath, K.; Mohana, D.C. Assessment of contamination levels and characterization of Aflatoxin B1 producing Aspergillus flavus strain from food and feed samples from local markets of South India. Int. J. Life Sci. 2018, 6, 326–334. [Google Scholar]
- Zhang, L.; Dou, X.W.; Zhang, C.; Logrieco, A.; Yang, M.-H. A review of current methods for analysis of mycotoxins in herbal medicines. Toxins 2018, 10, 65. [Google Scholar] [CrossRef]
- Van Egmond, H.P.; Schothorst, R.C.; Jonker, M.A. Regulations relating to mycotoxins in food: Perspectives in a global and European context. Anal. Bioanalyt Chem. 2007, 389, 147–157. [Google Scholar] [CrossRef] [PubMed]
- Bui-Klimke, T.R.; Guclu, H.; Kensler, T.W.; Yuan, J.-M.; Wu, F. Aflatoxin Regulations and Global Pistachio Trade: Insights from Social Network Analysis. PLoS ONE 2014, 9, e92149. [Google Scholar] [CrossRef] [PubMed]
- Payne, G.A.; Yu, J. Ecology, development and gene regulation in Aspergillus flavus. In Aspergillus: Molecular Biology and Genomics; Machida, M., Gomi, K., Eds.; Caister Acad. Press: Norwich, UK, 2010; pp. 157–171. [Google Scholar]
- Australia Government New Zealand Food Standards Code–Schedule 19–Maximum Levels of Contaminants and Natural Toxicants. Available online: https://www.legislation.gov.au/Details/F2017C00333 (accessed on 15 November 2021).
- Ministry of Health. National Health Surveillance Agency. Resolution No. 7, of February 18, 2011. Available online: https://bvsms.saude.gov.br/bvs/saudelegis/anvisa/2011/res0007_18_02_2011_rep.html (accessed on 15 November 2021).
- Government of Canada. List of Contaminants and Other Adulterating Substances in Foods. 2021. Available online: https://www.canada.ca/en/health-canada/services/food-nutrition/food-safety/chemical-contaminants/contaminants-adulteratingsubstancesfoods.html (accessed on 15 November 2021).
- USDA, United States Department of Agriculture. China Releases Standard for Maximum Levels of Mycotoxins in Foods. 2021. Available online: https://gain.fas.usda.gov/Recent%20GAIN%20Publications/China%20Releases%20Standard%\0for%20Maximum%20Levels%20of%20Mycotoxins%20in%20Foods%20_Beijing_China%20%20Peoples%20Republic%20of_5–9–2018.pdf (accessed on 15 November 2021).
- Srianujata, S. Regulatory update and control measures for prevention and reduction of mycotoxins contamination in foods and feeds. In Proceedings of the FFTCeKU Conference, International Seminar on Risk Assessment and Risk Management of Mycotoxins for Food Safety in Asia, Pattaya, Thailand, 5–9 September 2011; Kasetsart University: Bangkok, Thailand, 2011. [Google Scholar]
- Yoshizawa, T.A. Current situation of mycotoxin management in Asia in relation to recent actions in Japan. In Proceedings of the FFTCeKU 2011 Conference, International Seminar on Risk Ssessment and Risk Management of Mycotoxins for Food Safety in Asia, Pattaya, Thailand, 5–9 September 2011; Kasetsart University: Bangkok, Thailand, 2011. [Google Scholar]
- Lalah, J.O.; Omwoma, S.; Orony, D.A. Aflatoxin B1: Chemistry, environmental and diet sources and potential exposure in human in Kenya. In Xi-Dai Long Aflatoxin B1 Occurrence, Detection and Toxicological Effects; IntechOpen: London, UK, 2019. [Google Scholar]
- Suparmo, F.M.C.; Setyabudi, S.; Hidayat, C. Integrated control management of aflatoxins in Indonesia. In Proceedings of the FFTCeKU 2011 Conference, International Seminar on Risk Assessment and Risk Management of Mycotoxins for Food Safety in Asia, Pattaya, Thailand, 5–9 September 2011; Kasetsart University: Bangkok, Thailand, 2011. [Google Scholar]
- Kawamura, O.; Kiyama, I.; Soontornjanagit, M.; Hojo, E.; Matsumoto, T. Development and application of immunological methods for mycotoxins and toxicology. In Proceedings of the FFTCeKU 2011 Conference, International Seminar on Risk Assessment and Risk Management of Mycotoxins for Food Safety in Asia, Pattaya, Thailand, 5–9 September 2011; Kasetsart University: Bangkok, Thailand, 2011. [Google Scholar]
- Okello, D.K.; Kaaya, A.N.; Bisikwa, J.; Were, M.; Oloka, H.K. Management of Aflatoxins in Groundnuts: A Manual for Farmers, Processors, Traders and Consumers in Uganda; National Agricultural Research Organisation: Entebbe, Uganda, 2010; pp. 1–38. [Google Scholar]
- Chun, H.S. Risk assessment of mycotoxin contamination in Korean foods. In Proceedings of the FFTCeKU 2011 Conference, International Seminar on Risk Assessment and Risk Management of Mycotoxins for Food Safety in Asia, Pattaya, Thailand, 5–9 September 2011; Kasetsart University: Bangkok, Thailand, 2011. [Google Scholar]
- Atherstone, C.; Delia, G.; Johanna, L.; Francesca, N.; Erastus, K.; Timothy, W.; Victor, M. Aflatoxin Standards for Feed. Building an Aflatoxin Safe East African Community—Technical Policy Paper 7; International Institute of Tropical Agriculture: Ibadan, Nigeria, 2015. [Google Scholar] [CrossRef]
- Rustia, A.S.; Sales, J.M.; Tejada, A.W. Occurrence of aflatoxin contamination in foods: Present status in the Philippines. In Proceedings of the FFTCeKU Conference, International Seminar on Risk Assessment and Risk Management of Mycotoxins for Food Safety in Asia, Pattaya, Thailand, 5–9 September 2011; Kasetsart University: Bangkok, Thailand, 2011. [Google Scholar]
- Ncube, J.; Maphosa, M. Current State of Knowledge on Groundnut Aflatoxins and Their Management from a Plant Breeding Perspective: Lessons for Africa. Sci. Afr. 2020, 7, e00264. [Google Scholar] [CrossRef]
- Chen, M.-T.; Hsu, Y.-H.; Wang, T.-S.; Chien, S.-W. Mycotoxin monitoring for commercial foodstuffs in Taiwan. J. Food Drug Anal. 2016, 24, 147–156. [Google Scholar] [CrossRef] [PubMed]
- U.S. Food and Drug Administration, Center for Food Safety and Applied Nutrition (USFDA). Guidance for Industry: Action Levels for Poisonous or Deleterious Substances in Human Food and Animal Feed; U.S. Food and Drug Administration, Center for Food Safety and Applied Nutrition: Washington, DC, USA, 2000. [Google Scholar]
- European Commission. Directive 178/2006/EC of the European Parliament and of the Council of 23 February 2005 on the Maximum Levels of Pesticides in or on Food and Feed Plant and Animal Origin Official Journal of the European Union; European Commission: Geneva, Switzerland, 2016. [Google Scholar]
- Ilyas, F. Samples of Vegetable, Fruit Found Highly Contaminated. Dawn.com. 2012. Available online: http://www.dawn.com/news/768407/samples-of-vegetable-fruit-found-highly-contaminated (accessed on 29 September 2014).
- Khatoon, S.; Hanif, N.; Tahira, I.; Sultana, N.; Sultana, K.; Ayub, N. Natural occurrence of aflatoxins, zearalenone and trichothecenes in maize grown in Pakistan. Pak. J. Bot. 2012, 44, 231–236. [Google Scholar]
- Tania, A.; Akram, A.; Hanif, N.Q.; Ajmal, M.; Seerat, W.; Nijabat, A.; Mehak, A. Proximate composition, fungal isolation and contamination of aflatoxin B1 in chickpea seeds from the Punjab, Pakistan. Nat. Prod. Res. 2022, 2022, 2065674. [Google Scholar] [CrossRef]
- Hussain, A.; Sohail, M.; Ullah, S. Aflatoxin contamination of spices sold in different markets of Peshawar. J. Chem. Soc. Pak. 2012, 34, 1052–1055. [Google Scholar]
- Mobeen, A.K.; Aftab, A.; Asif, A.; Zuzzer, A.S. Aflatoxins B1 and B2 contamination of peanut and peanut products and subsequent microwave detoxification. J. Pharm. Nutr. Sci. 2011, 1, 1–3. [Google Scholar] [CrossRef]
- Food and Agriculture Organization (FAO). World Agriculture: Towards 2015/2030: Summary Report; FAO: Rome, Italy, 2002. [Google Scholar]
- Awika, J. Major cereal grains production and use around the world. advances in cereal science: Implications to Food Processing and Health Promotion. In Advances in Cereal Science: Implications to Food Processing and Health Promotion; American Chemical Society: New York, NY, USA, 2011; Volume 1089, pp. 1–13. [Google Scholar] [CrossRef]
- Andrade, P.; Caldas, E. Aflatoxins in cereals: Worldwide occurrence and dietary risk assessment. World Mycotoxin J. 2015, 8, 415–431. [Google Scholar] [CrossRef]
- Food and Agriculture Organization Statistical Databases (FAOSTAT). 2021. Available online: http://faostat.fao.org (accessed on 18 October 2022).
- Hussain, A.; Ali, J.; Ullah, S. Studies on contamination level of aflatoxins in Pakistani rice. Chem. Soc. Pak. 2011, 33, 481–484. [Google Scholar]
- Food and Agriculture Organization Statistical Databases (FAOSTAT). 2019. Available online: http://faostat.fao.org (accessed on 10 July 2020).
- G.O.P. Ministry of Finance. Economic Survery of Pakistan; G.O.P. Ministry of Finance: Islamabad, Pakistan, 2016–2017. [Google Scholar]
- Tariq, M.; Iqbal, H. Maize in Pakistan—An overview. Kasetsart J. 2010, 44, 757–763. [Google Scholar]
- Reddy, K.R.N.; Reddy, C.S.; Muralidharan, K. Potential of botanical and biocontrol agents on growth and aflatoxin production by Aspergillus flavus infecting rice grains. Food Control 2009, 20, 173–178. [Google Scholar] [CrossRef]
- Hussaini, A.M.; Timothy, A.G.; Olufunmilayo, H.A.; Ezekiel, A.S.; Godwin, H.O. Fungi and some mycotoxins found in mouldy sorghum in Niger State, Nigeria. World J. Agri. Sci. 2009, 5, 5–17. [Google Scholar]
- Lutfullah, G.; Hussain, A. Studies on contamination level of aflatoxins in some cereals and beans of Pakistan. Food Control 2012, 23, 32–36. [Google Scholar] [CrossRef]
- Iqbal, S.; Asi, M.R.; Arino, A.; Akram, N.; Zuber, M. Aflatoxin contamination in different fractions of rice from Pakistan and estimation of dietary intakes. Mycotoxin Res. 2012, 28, 175–180. [Google Scholar] [CrossRef]
- Xia, L.; Routledge, M.N.; Rasheed, H.; Ismail, A.; Dong, Y.; Jiang, T.; Gong, Y.Y. Biomonitoring of Aflatoxin B1 and Deoxynivalenol in a Rural Pakistan Population Using Ultra-Sensitive LC-MS/MS Method. Toxins 2020, 12, 591. [Google Scholar] [CrossRef]
- Zahra, N.; Idrees, A.; Aslam, M.; Noreen, Z.; Masood, S.; Saeed, M.K.; Kalim, I.; Nisa, A.; Hina, S.; Ahmad, I.; et al. Effect of moisture content on aflatoxin B1 production in wheat flour samples collected from Lahore, Pakistan. Pak. J. Anal. Environ. Chem. 2019, 20, 184–189. [Google Scholar] [CrossRef]
- Alim, M.; Iqbal, S.Z.; Mehmood, Z.; Asi, M.R.; Zikar, H.; Chanda, H.; Malik, N. Survey of mycotoxins in retail market cereals, derived products and evaluation of their dietary intake. Food Control 2018, 84, 471–477. [Google Scholar] [CrossRef]
- Asghar, M.A.; Ahmed, A.; Iqbal, J.; Zahir, E.; Nauman, H. Fungal flora and aflatoxin contamination in Pakistani wheat kernels (Triticum aestivum L.) and their attribution in seed germination. J. Food Drug Analysis 2016, 24, 635–643. [Google Scholar] [CrossRef]
- Iqbal, S.; Asi, M.R.; Jinap, S.; Rashid, U. Detection of aflatoxins and zearalenone contamination in wheat derived products. Food Control 2014, 35, 223–226. [Google Scholar] [CrossRef]
- Tahir, N.I.; Hussain, S.; Asghar, A.; Syed, S.K.; Idrees, M.A.; Ahmad, M.A. Aflatoxin concentrations in various rice varieties of Punjab, Pakistan. RADS J. Biol. Res. App. Sci. 2021, 12, 39–53. [Google Scholar] [CrossRef]
- Nazir, A.; Kalim, I.; Imran, M.; Bilal, M.A.; Zahra, N.; Ahmad, A.; Iqbal, M.; Fazal, U.; Ehtisham-ul-Haque, S. Incidences and bio-detoxification of aflatoxins in rice and cattle feed crops under different Agro-ecological zones. Polish J. Environ. Stud. 2021, 30, 1949–1954. [Google Scholar] [CrossRef]
- Majeed, S.; Boevre, M.D.; Saeger, S.D.; Rauf, W.; Tawab, A.; Habib, F.; Rahman, M.; Iqbal, M. Multiple mycotoxins in rice: Occurrence and health risk assessment in children and adults of Punjab, Pakistan. Toxins 2018, 10, 77. [Google Scholar] [CrossRef] [PubMed]
- Sultana, N.; Tahira, I.; Kausar, M.; Hassan, S.M.; Hanif, N.Q. Dietary exposure and natural occurrence of total aflatoxins in basmati rice of Pakistan. J. Food Prot. 2017, 80, 331–337. [Google Scholar] [CrossRef] [PubMed]
- Mukhtar, H.; Farooq, Z.; Manzoor, M. Determination of aflatoxins in super kernel rice types consumed in different regions of Punjab, Pakistan. J. Anim. Plant Sci. 2016, 26, 542–548. [Google Scholar]
- Nisa, A.; Zahra, N.; Yasha, N.B. Comparative study of aflatoxins in brown rice samples of local and import quality. Int. Food Res. J. 2016, 23, 243–247. [Google Scholar]
- Iqbal, S.Z.; Asi, M.R.; Hanif, U.; Zuber, M.; Jinap, S. The presence of aflatoxins and ochratoxin A in rice and rice products; and evaluation of dietary intake. Food Chem. 2016, 210, 135–140. [Google Scholar] [CrossRef]
- Firdous, S.; Ashfaq, A.; Khan, S.J.; Khan, N. Aflatoxins in corn and rice sold in Lahore, Pakistan. Food Addit. Contam. Part B 2014, 7, 95–98. [Google Scholar] [CrossRef]
- Asghar, M.A.; Iqbal, J.; Ahmed, A.; Khan, M.A. Occurrence of aflatoxins contamination in brown rice from Pakistan. Iran. J. Public Health 2014, 43, 291–299. [Google Scholar]
- Asghar, M.A.; Iqbal, J.; Ahmed, A.; Shamsuddin, Z.A.; Khan, M.A. Incidence of aflatoxins in export quality basmati rice collected from different areas of Pakistan. Sci. Technol. Develop. 2013, 32, 110–119. [Google Scholar]
- Majeed, S.; Iqbal, M.; Asi, M.R.; Iqbal, S.Z. Aflatoxins and ochratoxin A contamination in rice, corn and corn products from Punjab, Pakistan. J. Cereal Sci. 2013, 58, 446–450. [Google Scholar] [CrossRef]
- Firdous, S.; Ejaz, N.; Aman, T.; Khan, N. Occurrence of aflatoxins in export–quality Pakistani rice. Food Addit. Contam. Part B 2012, 5, 121–125. [Google Scholar] [CrossRef] [PubMed]
- Murad, M.; Ahmad, K.S.; Iram, S.; Hanif, N.Q.; Gul, M.M.; Elnaggar, A.Y.; El-Bahy, Z.M. Mycotoxins in Zea mays, their quantification and HPLC analysis of physico-biological detoxification. Nat. Prod. Res. 2021, 21, 1–5. [Google Scholar] [CrossRef]
- Hussain, M.; Shoaib, A.; Nisa, A.-U.; Aftab, M.; Ali, R. Aflatoxins levels in branded and non-branded corn from Lahore, Pakistan. Mycopath 2020, 18, 7–10. [Google Scholar]
- Hassan, S.W.; Sadef, Y.; Hussain, S.; Asi, M.R.; Ashraf, M.Y.; Anwar, S.; Malik, A. Unusual pattern of aflatoxins and ochratoxin in commercially grown maize varieties of Pakistan. Toxicon 2020, 182, 66–71. [Google Scholar] [CrossRef]
- Manzoor, M.; Farooq, Z.; Iqbal, S.; Mukhtar, H.; Nawaz, M. Quantification of aflatoxins in maize samples collected from various parts of the Punjab, Pakistan. J. Anim. Plant Sci. 2018, 28, 1656–1661. [Google Scholar] [CrossRef]
- Iram, W.; Anjum, T.; Abbas, M.; Khan, A.M. Aflatoxins and ochratoxin A in maize of Punjab, Pakistan. Food Addit. Contam. Part B 2014, 7, 57–62. [Google Scholar] [CrossRef]
- Shah, H.; Simpson, T.J.; Alam, S.; Khattak, K.F.; Perveen, S. Mould incidence and mycotoxin contamination in maize kernels from Swat valley, Northwest Frontier Province of Pakistan. Food Chem. Toxicol. 2010, 48, 1111–1116. [Google Scholar] [CrossRef]
- Ahsan, S.; Bhatti, I.A.; Asi, M.R.; Bhatti, H.N.; Sheikh, M.A. Occurrence of aflatoxins in maize grains from central areas of Punjab, Pakistan. Int. J. Agric. Biol. 2010, 12, 571–575. [Google Scholar]
- Din, N.U.; Mahmood, A.; Khattak, G.S.S.; Saeed, I.; Hassan, M.F. High yielding groundnut (Arachis hypogaea L.) variety Golden. Pak. J. Bot. 2009, 41, 2217–2222. [Google Scholar]
- Iqbal, N.; Hussain, S.; Ahmed, Z.; Yang, F.; Wang, X.; Liu, W.; Liu, J. Comparative analysis of maize–soybean strip intercropping systems: A review. Plant Prod. Sci. 2019, 22, 131–142. [Google Scholar] [CrossRef]
- Zorzete, P.; Reis, T.; Felicio, J.; Baquiao, A.; Makimoto, P.; Correa, B. Fungi, mycotoxins and phytoalexin in peanut varieties, during plant growth in the field. Food Chem. 2011, 129, 957–964. [Google Scholar] [CrossRef] [PubMed]
- Abbas, M.; Khan, A.M.; Asi, M.R.; Akhtar, J. Pervasiveness of aflatoxin in peanuts growing in the area of Pothohar, Pakistan. Int. J. Nutr. Food Eng. 2012, 6, 738–741. [Google Scholar] [CrossRef]
- Waqas, M.; Iqbal, S.Z.; Abdull Razis, A.F.; Pervaiz, W.; Ahmad, T.; Usman, S.; Ali, N.B.; Asi, M.R. Occurrence of Aflatoxins in Edible Vegetable Seeds and Oil Samples Available in Pakistani Retail Markets and Estimation of Dietary Intake in Consumers. Int. J. Environ. Res. Public Health 2021, 18, 8015. [Google Scholar] [CrossRef]
- Hussain, A.; Rahman, Z.; Khan, M. Detection of aflatoxins in peanut oils marketed in Peshawar, Pakistan using thin layer chromatography. J. Food Qual. Hazards Control 2021, 8, 87–91. [Google Scholar] [CrossRef]
- Iqbal, S.Z.; Asi, M.R.; Zuber, M.; Akrama, N.; Batool, N. Aflatoxin’s contamination in peanut and peanut products commercially available in retail markets of Punjab, Pakistan. Food Control 2013, 32, 83–86. [Google Scholar] [CrossRef]
- Mushtaq, M.; Sultana, B.; Anwar, F.; Khan, M.Z.; Ashrafuzzaman, M. Occurrence of Aflatoxins in Selected Processed Foods from Pakistan. Int. J. Mol. Sci. 2012, 13, 8324–8337. [Google Scholar] [CrossRef]
- Lutfullah, G.; Hussain, A. Studies on contamination level of aflatoxins in some dried fruits and nuts of Pakistan. Food Control 2011, 22, 426–429. [Google Scholar] [CrossRef]
- Iqbal, S.Z.; Waqas, M.; Razis, A.F.A.; Usman, S.; Ali, N.B.; Asi, M.R. Variation of aflatoxin levels in stored edible seed and oil samples and risk assessment in the local population. Toxins 2022, 14, 642. [Google Scholar] [CrossRef]
- Ajmal, M.; Akram, A.; Hanif, N.Q.; Mukhtar, T.; Arshad, M. Mycobiota isolation and aflatoxin B1 contamination in fresh and stored sesame seeds from rainfed and irrigated zones of Punjab, Pakistan. J. Food Protec. 2021, 84, 1673–1682. [Google Scholar] [CrossRef]
- Hathout, A.S.; Aly, S.E. Biological detoxification of mycotoxins: A review. Annal. Microbiol. 2014, 64, 905–919. [Google Scholar] [CrossRef]
- Ajmal, M.; Alshannaq, A.F.; Moon, H.; Choi, D.; Akram, A.; Nayyar, B.G.; Gibbons, J.G.; Yu, J.-H. Characterization of 260 isolates of Aspergillus Section Flavi obtained from sesame seeds in Punjab, Pakistan. Toxins 2022, 14, 117. [Google Scholar] [CrossRef] [PubMed]
- Food and Agriculture Organization Statistical Databases. (FAOSTAT). 2020. Available online: http://faostat.fao.org (accessed on 15 March 2021).
- Fatima, G.; Khan, I.A.; Buerkert, A. Socio-economic characterisation of date palm (Phoenix dactylifera L.) growers and date value chains in Pakistan. SpringerPlus 2016, 5, 1222. [Google Scholar] [CrossRef] [PubMed]
- Ali, S.; Ali, A.; Sartaj, A.; Ali, M.; Ali, A. Natural occurrence of aflatoxin B1 in dry fruits of Gilgit-Baltistan, Pakistan. Fresenius Environ. Bull. 2020, 29, 2018–2022. [Google Scholar]
- Razis, A.F.A.; Shehzad, M.M.; Usman, S.; Ali, N.B.; Iqbal, S.Z.; Naheed, N.; Asi, M.R. Seasonal variation in aflatoxin levels in edible seeds, estimation of its dietary intake and vitamin e levels in southern areas of Punjab, Pakistan. Int. J. Environ. Res. Public Health 2020, 17, 8964. [Google Scholar] [CrossRef] [PubMed]
- Alghalibi, S.M.S.; Shater, A.M. Mycoflora and mycotoxin contamination of some dried fruits in Yemen Republic. Assiut Univ. Bull. Environ. Res. 2004, 7, 19–27. [Google Scholar] [CrossRef]
- Asghar, M.A.; Ahmed, A.; Zahir, E.; Asghar, M.A.; Iqbal, J.; Walker, G. Incidence of aflatoxins contamination in dry fruits and edible nuts collected from Pakistan. Food Control 2017, 78, 169–175. [Google Scholar] [CrossRef]
- Iqbal, S.Z.; Asi, M.R.; Mehmood, Z.; Shahid, M.; Sehar, M.; Malik, N. Co-occurrence of aflatoxins and ochratoxin A in nuts, dry fruits, and nuty products. J. Food Saf. 2018, 38, e12462. [Google Scholar] [CrossRef]
- Masood, M.; Iqbal, S.Z.; Asi, M.R.; Malik, N. Natural occurrence of aflatoxins in dry fruits and edible nuts. Food Control 2015, 55, 62–65. [Google Scholar] [CrossRef]
- Arifeen, M. Chilli: The most valuable cash crop. The Financial Daily International. 2009. Available online: http://thefinancialdaily.com/NewsDetail/86342.aspx/26/5/09 (accessed on 30 December 2020).
- Nordin, S.; Samsudin, N.A.; Esah, E.M.; Zakaria, L.; Selamat, J.; Rahman, M.A.H.; Mahror, N. Prevalence, Identification and Mycotoxigenic Potential of Fungi in Common Spices Used in Local Malaysian Cuisines. Foods 2022, 11, 2548. [Google Scholar] [CrossRef]
- Elshafie, A.E.; Al-Rashdi, T.A.; Al-Bahry, S.N.; Bakheit, C.S. Fungi and aflatoxins associated with spices in the Sultanate of Oman. Mycopathologia 2002, 155, 155–160. [Google Scholar] [CrossRef] [PubMed]
- Mandeel, Q.A. Fungal contamination of some imported spices. Mycopathologia 2005, 159, 291–298. [Google Scholar] [CrossRef] [PubMed]
- Hashem, M.; Alamri, S. Contamination of common spices in Saudi Arabia markets with potential mycotoxin-producing fungi. Saudi J. Biol. Sci. 2010, 17, 167–175. [Google Scholar] [CrossRef] [PubMed]
- Sahar, N.; Arif, S.; Iqbal, S.; Riaz, S.; Fatima, T.; Ara, J.; Banks, J. Effects of drying surfaces and physical attributes on the development of Aflatoxins (AFs) in red chilies. J. food Process. Preservat. 2017, 46, e16173. [Google Scholar] [CrossRef]
- Abrar, M.; Anjum, F.M.; Zahoor, T.; Nawaz, H. Effect of storage period and irradiation doses on red chilies. Pak. J. Nutr. 2009, 8, 1287–1291. [Google Scholar] [CrossRef]
- Iqbal, S.Z.; Mumtaz, A.; Mahmood, Z.; Waqas, M.; Ghaffar, A.; Ismail, A.; Pervaiz, W. Assessment of aflatoxins and ochratoxin a in chili sauce samples and estimation of dietary intake. Food Control 2021, 121, 107621. [Google Scholar] [CrossRef]
- Akhund, S.; Akram, A.; Hanif, N.Q.; Qureshi, R.; Naz, F.; Nayyar, B.G. Pre-harvest aflatoxins and Aspergillus flavus contamination in variable germplasms of red chillies from Kunri, Pakistan. Mycotoxin Res. 2017, 33, 147–155. [Google Scholar] [CrossRef]
- Naz, N.; Kashif, A.; Kanwal, K.; Khan, A.M.; Abbas, M. Quantitative scrutinization of aflatoxins in different spices from Pakistan. Int. J. Anal. Chem. 2016, 16, 4907425. [Google Scholar] [CrossRef] [PubMed]
- Asghar, M.A.; Zahir, E.; Rantilal, S.; Ahmed, A.; Iqbal, J. Aflatoxins in composite spices collected from local markets of Karachi, Pakistan. Food Addit Contam. Part B 2016, 9, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.A.; Asghar, M.A.; Iqbal, J.; Ahmed, A.; Shamsuddin, Z.A. Aflatoxins contamination and prevention in red chillies (Capsicum annuum L.) in Pakistan. Food Addit. Contam. Part B Surveill. 2014, 7, 1–6. [Google Scholar] [CrossRef]
- Iqbal, S.Z.; Paterson, R.R.M.; Bhatti, I.A.; Asi, M.R.; Sheikh, M.A.; Bhatti, H.N. Aflatoxin B1 in chilies from the Punjab region, Pakistan. Mycotoxin Resou. 2010, 26, 205–209. [Google Scholar] [CrossRef] [PubMed]
- Paterson, R.R.M. Aflatoxins contamination in chili samples from Pakistan. Food Control 2007, 18, 817–820. [Google Scholar] [CrossRef]
- Iqbal, S.Z.; Asi, M.R.; Arino, A. Aflatoxin M1 contamination in cow and buffalo milk samples from the Northwest Frontier Province (NWFP) and Punjab provinces of Pakistan. Food Addit Contam. Part B 2011, 4, 282–288. [Google Scholar] [CrossRef]
- Schweiggert, U.; Kammerer, D.R.; Carle, R.; Schieber, A. Characterization of carotenoids and carotenoid esters in red pepper pods (Capsicum Annuum L.) by high-performance liquid chromatography/atmospheric pressure chemical ionization mass spectrometry. Rapid Commun. Mass Spectrom. 2005, 19, 2617–2628. [Google Scholar] [CrossRef]
- Zafar, F.; Yasmin, N.; Hasan, R.; Naim, T.; Qureshi, A. A study on the analysis of ochratoxin-A in different poultry feed ingredients. Pak. J. Pharm. Sci. 2001, 14, 5–7. [Google Scholar]
- Bhatti, B.M.; Talat, T.; Sardar, R. Estimation of aflatoxin B1 in feed ingredients and compound poultry feeds. Pak. Vet. J. 2001, 21, 57–60. [Google Scholar]
- Rashid, N.; Bajwa, M.; Rafeeq, M.; Khan, M.; Ahmad, Z.; Tariq, M.; Wadood, A.; Abbas, F. Prevalence of aflatoxin B1 in finished commercial broiler feed from West Central Pakistan. J. Anim. Plant Sci. 2012, 22, 6–10. [Google Scholar]
- Anjum, M.A.; Sahota, A.W.; Akram, M.; Ali, I. Prevalence of mycotoxins in poultry feeds and feed ingredients in Punjab (Pakistan). J. Animal Plant Sci. 2011, 21, 117–120. [Google Scholar]
- Alam, S.; Shah, H.U.; Khan, H.; Magan, N. The effect of substrate, season, and agroecological zone on mycoflora and aflatoxin contamination of poultry feed from Khyber Pakhtunkhwa, Pakistan. Mycopathologia 2012, 174, 341–349. [Google Scholar] [CrossRef]
- Khan, S.H.; Hasan, S.; Sardar, R.; Anjum, M.A. Occurrence of aflatoxin B1 in poultry feed and feed ingredients in Pakistan. IJAVMS 2011, 5, 30–42. [Google Scholar] [CrossRef]
- Anjum, M.; Khan, S.; Sahota, A.; Sardar, R. Assessment of aflatoxin B1 in commercial poultry feed and feed ingredients. J. Animal Plant Sci. 2012, 22, 268–272. [Google Scholar]
- Yunus, A.W.; Ullah, A.; Lindahl, J.F.; Anwar, Z.; Ullah, A.; Saif, S.; Ali, M.; Bin Zahur, A.; Irshad, H.; Javaid, S.; et al. Aflatoxin contamination of milk produced in peri-urban farms of pakistan: Prevalence and contributory factors. Front Microbiol. 2020, 11, 159. [Google Scholar] [CrossRef]
- Summia, K.; Yasmeen, R.; Nisa, A.; Djeffa, A.; Jabeen, T. Estimation of Aflatoxins in Cattle Feed Used in and Around Lahore District. Biologia 2020, 66, 185–191. [Google Scholar]
- Sarwat, A.; Rauf, W.; Majeed, S.; De Boevre, M.; De Saeger, S.; Iqbal, M. LC-MS/MS based appraisal of multi-mycotoxin co-occurrence in poultry feeds from different regions of Punjab, Pakistan. Food Addit. Contam. Part B 2022, 15, 106–122. [Google Scholar] [CrossRef] [PubMed]
- Naveed, M.; Haleem, K.S.; Ghazanfar, S.; Tauseef, I.; Bano, N.; Adetunji, C.O.; Saleem, M.H.; Alshaya, H.; Paray, B.A. Quantitative Estimation of Aflatoxin Level in Poultry Feed in Selected Poultry Farms. Hindawi BioMed Res. Inter. 2022, 2022, 5397561. [Google Scholar] [CrossRef]
- Majeed, F.; Akbar, A.; Shafee, M.; Sadia, H.; Iqbal, S.; Phadungchob, B.; Khan, G.; Khan, S. Detection of Mycotoxigenic Fungi and Mycotoxins in Poultry Feed of Balochistan Pakistan. J. Hellenic Vet. Med. Soci. 2021, 72, 2741–2746. [Google Scholar] [CrossRef]
- Waqas, M.; Pervaiz, W.; Zia, K.M.; Iqbal, S.Z. Assessment of aflatoxin B1 in animal feed and aflatoxin M1 in raw milk samples of different species of milking animals from Punjab, Pakistan. J. Food Saf. 2021, 41, e12893. [Google Scholar] [CrossRef]
- Shar, Z.H.; Pirkash, O.; Shar, H.H.; Sherazi, S.T.H.; Mahesar, S.A. Aflatoxins in cotton seeds and cotton seed cake from Pakistan. Food Addit. Contam. Part B 2020, 13, 72–76. [Google Scholar] [CrossRef]
- Akbar, N.; Nasir, M.; Naeem, N.; Ahmad, M.D.; Saeed, F.; Anjum, F.; Iqbal, S.; Imran, M.; Tufail, T.; Shah, F.; et al. Assessment of aflatoxin in milk and feed samples and impact of seasonal variations in the Punjab, Pakistan. Food Sci. Nutr. 2020, 8, 2699–2709. [Google Scholar] [CrossRef] [PubMed]
- Iram, S.; Fareed, S.; Chaudhary, M.; Iqbal, M.U.N.; Ghani, R.; Khan, T.A.; Abbas, T. Identification of Aspergillus flavus and aflatoxin in home mix layer poultry feed in relation to seasons in Karachi, Pakistan. Trop. Anim Health Prod. 2019, 51, 1321–1327. [Google Scholar] [CrossRef] [PubMed]
- Ismail, A.; Riaz, M.; Akhtar, S.; Yoo, S.H.; Park, S.; Abid, M.; Aziz, M.; Ahmad, Z. Seasonal variation of aflatoxin B1 content in dairy feed. J. Animal Feed Sci. 2017, 26, 33–37. [Google Scholar] [CrossRef]
- Iqbal, S.Z.; Asi, M.R.; Nisar, S.; Zia, K.M.; Jinap, S.; Malik, N. A limited survey of aflatoxins and zearalenone in feed and feed ingredients from Pakistan. J. Food Protec. 2016, 79, 1798–1801. [Google Scholar] [CrossRef] [PubMed]
- Chohan, K.A.; Awan, F.; Ali, M.M.; Iqbal, U.; Ijaz, M. Assessment of aflatoxin in dairy concentrate feeds, total mixed rations, silage and various feed ingredients in Pakistan. Pak. J. Zoo. 2016, 48, 277–280. [Google Scholar]
- Yunus, A.; Nasir, M.; Aziz, T.; Bohm, J. Prevalence of poultry diseases in district Chakwal and their interaction with mycotoxicosis: Effects of season and feed. J. Anim. Plant Sci. 2009, 19, 1–5. [Google Scholar]
- Hanif, N. Mycotoxin contamination in cattle feed and feed ingredients. Pak. Vet. J. 2009, 29, 211–213. [Google Scholar]
- Hanif, N.Q.; Muhammad, G.; Siddique, M.; Khanum, A.; Ahmed, T.; Gadahai, J.A.; Kaukab, G. Clinico-pathomorphological, serum biochemical and histological studies in broilers fed ochratoxin A and a toxin deactivator (Mycofix® Plus). Br. Poult. Sci. 2008, 49, 632–642. [Google Scholar] [CrossRef]
- Tangendjaja, B.; Rachmawati, S.; Wina, E. Origins and factors associated with mycotoxins level in corn used as animal feed in Indonesia. Indones. J. Agric. Sci. 2008, 9, 68–76. [Google Scholar] [CrossRef]
- Chauhan, N.M.; Washe, A.P.; Minota, T. Fungal infection and aflatoxin contamination in maize collected from Gedeo zone, Ethiopia. Springer Plus 2016, 5, 753. [Google Scholar] [CrossRef]
- Sanchis, V.; Magan, N. Environmental conditions affecting mycotoxins. In Mycotoxins in Food: Detection and Control; CRC Press: Boca Raton, FL, USA, 2004; pp. 174–189. Available online: https://www.researchgate.net/profile/Naresh_Magan/publication/235334797_Mycotoxins_in_Food_Detection_and_Control/links/02e7e5326d7082f043000000.p (accessed on 23 November 2021).
- Kamkar, A.; Karim, G.; Aliabadi, F.S.; Khaksar, R. Fate of aflatoxin M1 in Iranian white cheese processing. Food Chem. Toxicol. 2008, 46, 2236–2238. [Google Scholar] [CrossRef]
- Mwalwayo, D.S.; Thole, B. Prevalence of aflatoxin and fumonisins (B1 + B2) in maize consumed in rural Malawi. Toxicol. Rep. 2016, 3, 173–179. [Google Scholar] [CrossRef]
- Azziz-Baumgartner, E.; Lindblade, K.; Gieseker, K.; Rogers, H.S.; Kieszak, S.; Njapau, H.; Schleicher, R.; McCoy, L.F.; Misore, A.; DeCock, K.; et al. Case-control study of an acute aflatoxicosis outbreak, Kenya, 2004. Environ. Health Perspect. 2005, 113, 1779–1783. [Google Scholar] [CrossRef] [PubMed]
- Rastogi, S.; Dwivedi, P.; Khanna, S.K.; Das, M. Detection of Aflatoxin M1 contamination in milk and infant milk products from Indian markets by ELISA. Food Control 2004, 15, 287–290. [Google Scholar] [CrossRef]
- Reddy, K.R.N.; Salleh, B. Co-occurrence of moulds and mycotoxins in corn grains used for animal feeds in Malaysia. J. Anim. Vet. Adv. 2011, 10, 668–673. [Google Scholar] [CrossRef]
- Asi, M.R.; Iqbal, S.Z.; Arino, A.; Hussain, A. Effect of seasonal variations and lactation times on aflatoxin M1 contamination in milk of different species from Punjab, Pakistan. Food Control 2012, 25, 34–38. [Google Scholar] [CrossRef]
- Pei, S.C.; Zhang, Y.Y.; Eremin, S.A.; Lee, W.J. Detection of aflatoxin M1 in milk products from China by ELISA using monoclonal antibodies. Food Control 2009, 20, 1080–1085. [Google Scholar] [CrossRef]
- Hussain, I.; Anwar, J.; Munawar, M.; Asi, M. Variation of levels of aflatoxin M1 in raw milk from different localities in the central areas of Punjab, Pakistan. Food Control 2008, 19, 1126–1129. [Google Scholar] [CrossRef]
- Iqbal, S.Z.; Asi, M.R.; Jinap, S. Variation of aflatoxin M1 contamination in milk and milk products collected during winter and summer seasons. Food Control 2013, 34, 714–718. [Google Scholar] [CrossRef]
- Ghajarbeygi, P.; Palizban, M.; Mahmoudi, R.; Jahed Khaniki, G.; Pakbin, B. Aflatoxin M1 contamination of cow’s raw milk in different seasons from Qazvin Province, Iran. J. Biol. Today World 2016, 5, 173–176. [Google Scholar] [CrossRef]
- Shokri, H.; Torabi, S. The effect of milk composition, yeast-mould numbers and seasons on aflatoxin M1 amounts in camel milk. J. Food Saf. 2017, 37, e12300. [Google Scholar] [CrossRef]
- Mahmood, N.; Khalid, M.; Kouser, S. The role of agricultural credit in the growth of livestock sector: A case study of Faisalabad. Pak. Vet. J. 2009, 29, 81–84. [Google Scholar]
- Economic Survey of Pakistan. 2019–2020. Available online: https://www.finance.gov.pk/survey/chapter_20/02_Agriculture.pdf (accessed on 15 January 2021).
- Ali, A.; Khan, M. Livestock ownership in ensuring rural household food security in Pakistan. J. Anim. Plant Sci. 2013, 23, 313–318. [Google Scholar]
- Akbar, N.; Nasir, M.; Naeem, N.; Ahmad, M.; Iqbal, S.; Rashid, A.; Imran, M.; Gondal, T.A.; Atif, M.; Salehi, B.; et al. Occurrence and seasonal variations of aflatoxin M1 in milk from Punjab, Pakistan. Toxins 2019, 11, 574. [Google Scholar] [CrossRef]
- Mahmoudi, R.; Norian, R. Aflatoxin B1 and M1 contamination in cow feeds and milk from Iran. Food Agri. Immunol. 2015, 26, 131–137. [Google Scholar] [CrossRef]
- Maqbool, U.; Anwar-Ul-Haq; Ahmad, M. ELISA determination of Aflatoxin M1 in milk and dairy products in Pakistan. Toxicol. Environ. Chem. 2009, 91, 241–249. [Google Scholar] [CrossRef]
- Akande, K.; Abubakar, M.; Adegbola, T.; Bogoro, S. Nutritional and health implications of mycotoxins in animal feeds: A review. Pak. J. Nutr. 2006, 5, 398–403. [Google Scholar] [CrossRef]
- Rahmani, J.; Alipour, S.; Miri, A.; Fakhri, Y.; Riahi, S.M.; Keramati, H.; Moradi, M.; Amanidaz, N.; Pouya, R.H.; Bahmani, Z.; et al. The prevalence of aflatoxin M1 in milk of middle east region: A systematic review, meta-analysis and probabilistic health risk assessment. Food Chem. Toxicol. 2018, 118, 653–666. [Google Scholar] [CrossRef]
- Razzaghi-Abyaneh, M.; Chang, P.-K.; Shams-Ghahfarokhi, M.; Rai, M. Global health issues of aflatoxins in food and agriculture: Challenges and opportunities. Front. Microbiol. 2014, 5, 420. [Google Scholar] [CrossRef] [PubMed]
- Duarte, S.C.; Almeida, A.M.; Teixeira, A.S.; Pereira, A.L.; Falcao, A.C.; Pena, A.; Lino, C.M. Aflatoxin M1 in marketed milk in portugal: Assessment of human and animal exposure. Food Control 2013, 30, 411–417. [Google Scholar] [CrossRef]
- Younas, M.; Yaqoob, M. Feed Resources of Livestock in the Punjab, Pakistan. Livest. Res. Rural. Dev. 2005, 17, 2005. Available online: http://www.lrrd.org/lrrd17/2/youn17018.htm (accessed on 27 August 2022).
- Yunus, A.W.; Imtiaz, N.; Khan, H.; Nawaz, M.; Ibrahim, M.; Zafar, Y. Aflatoxin Contamination of Milk Marketed in Pakistan: A Longitudinal Study. Toxins 2019, 11, 110. [Google Scholar] [CrossRef] [PubMed]
- Aslam, N.; Tipu, M.Y.; Ishaq, M.; Cowling, A.; McGill, D.; Warriach, H.M.; Wynn, P. Higher levels of aflatoxin M1 contamination and poorer composition of milk supplied by informal milk marketing chains in Pakistan. Toxins 2016, 8, 347. [Google Scholar] [CrossRef]
- Jawaid, S.; Talpur, F.N.; Nizamani, S.M.; Afridi, H.I. Contamination profile of aflatoxin M1 residues in milk supply chain of Sindh, Pakistan. Toxicol. Rep. 2015, 2, 1418–1422. [Google Scholar] [CrossRef] [PubMed]
- Zahra, N.; Jamil, N.; Ahmad, S.R.; Kalim, I.; Saeed, M.K.; Ahmed, I.; Syed, Q.; Munir, S. Assessment of milk quality by analyzing aflatoxin M1 in branded and non-branded milk samples collected from different shops/areas in Lahore, Pakistan. Pak J. Zoo. 2020, 52, 373–375. [Google Scholar] [CrossRef]
- Fahmid, S.; Sajjad, A.; Ali, J.; Khan, M. Incidence of aflatoxin M1 in fresh milk marketed in Quetta, Pakistan. Indo-Am. J. Pharm. Sci. 2019, 6, 1460–1464. [Google Scholar]
- Tahira, I.; Sultana, N.; Munir, A.; Hasan, S.M.; Hanif, N.Q. Report: Occurrence of Aflatoxin M1 in raw and processed milk consumed in Pakistan. Pak. J. Pharm. Sci. 2019, 32, 1097–1101. [Google Scholar]
- Ahmad, M.; Awais, M.; Alia, S.W.; Khana, H.A.A.; Riazb, M.; Sultana, A.; Bashira, M.S.; Chaudhry, A.I. Occurrence of Aflatoxin M1 in raw and processed milk and assessment of daily intake in Lahore, Multan cities of Pakistan. Food Addit. Contam. Part B 2019, 12, 18–23. [Google Scholar] [CrossRef]
- Naz, N.; Abbas, M.; Rubab, A.; Kanwal, K. Occurrence of Aflatoxin M1 in Milk-based Mithae samples from Pakistan. Open Chem. 2019, 17, 1140–1145. [Google Scholar] [CrossRef]
- Asghar, M.A.; Ahmed, A.; Asghar, M.A. Aflatoxin M1 in fresh milk collected from local markets of Karachi, Pakistan. Food Addit. Contam. Part B 2018, 11, 167–174. [Google Scholar] [CrossRef]
- Iqbal, S.Z.; Asi, M.R.; Malik, N. The seasonal variation of aflatoxin M1 in milk and dairy products and assessment of dietary intake in Punjab, Pakistan. Food Control 2017, 79, 292–296. [Google Scholar] [CrossRef]
- Ismail, A.; Riaz, M.; Levin, R.E.; Akhtar, S.; Gong, Y.Y.; Hameed, A. Seasonal prevalence level of aflatoxin M1 and its estimated daily intake in Pakistan. Food Control 2016, 60, 461–465. [Google Scholar] [CrossRef]
- Sajid, M.; Randhawa, M.; Zahoor, T.; Sultan, J. Quantitative assessment of aflatoxin (AFM1) in milk collected from dairy farms in Faisalabad, Pakistan. J. Chem. Society Pak. 2015, 37, 594–598. [Google Scholar]
- Iqbal, S.; Asi, M.R.; Selamat, J. Aflatoxin M1 in milk from urban and rural farmhouses of Punjab, Pakistan. Food Addit. Contam. Part B Surveill. 2014, 7, 17–20. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, S.Z.; Asi, M.R. Assessment of aflatoxin M1 in milk and milk products from Punjab. Pakistan. Food Control 2013, 30, 235–239. [Google Scholar] [CrossRef]
- Sadia, A.; Jabbar, M.A.; Deng, Y.; Hussain, E.A.; Riffat, S.; Naveed, S.; Arif, M. A survey of aflatoxin M1 in milk and sweets of Punjab, Pakistan. Food Control 2012, 26, 235–240. [Google Scholar] [CrossRef]
- Khushi, M.Z.; Tipu, Y.; Abbas, M.; Khan, A.M.; Anjum, A.A. Monitoring of aflatoxin M–1 in market raw milk in Lahore City, Pakistan. Pak. J. Zoo. 2010, 42, 697–700. [Google Scholar]
- Hussain, I.; Anwar, J.; Asi, M.; Munawar, M.; Kashif, M. AFM1 contamination in milk from five dairy species in Pakistan. Food Control 2010, 21, 122–124. [Google Scholar] [CrossRef]
- Udovicki, B.; Djekic, L.; Kljusuric, J.G.; Papageorgiou, M.; Skendi, A.; Djugum, J.; Rajkovic, A. Exposure assessment and risk characterization of aflatoxins intake through consumption of maize products in the adult populations of Serbia, Croatia and Greece. Food Addit. Contam. Part A 2019, 36, 940–951. [Google Scholar] [CrossRef] [PubMed]
- Li, K.; Chen, W.; Qiu, F.; Liang, Z.; Yang, M.; Wang, Z. Dietary exposure assessment to aflatoxins in nine kinds of foodstuff by Mont Carlo non-parametric probability approach in Shenzhen City. Wei Sheng Yan Jiu = J. Hygiene Res. 2018, 47, 827–832. [Google Scholar]
- European Food Safety Authority (EFSA). Opinion of the scientific committee on a request from EFSA related to a harmonised approach for risk assessment of substances which are both Genotoxic and Carcinogenic. EFSA J. 2005, 3, 282. [Google Scholar] [CrossRef]
- Al-Jaal, B.A.; Jaganjac, M.; Barcaru, A.; Horvatovich, P.L.; Lati, A. Aflatoxin, fumonisin, ochratoxin, zearalenone and deoxynivalenol biomarkers in human biological fluids: A systematic literature review, 2001–2018. Food Chem. Toxicol. 2019, 129, 211–228. [Google Scholar] [CrossRef]
- Routledge, M.N.; Gong, Y.Y. Molecular epidemiology of aflatoxin exposure and child health. Mutagenesis 2012, 27, 797. [Google Scholar]
- Turna, N.S.; Wu, F. Estimation of tolerable daily intake (tdi) for immunological effects of aflatoxin. Risk Anal. 2022, 42, 431–438. [Google Scholar] [CrossRef] [PubMed]
- Al-Jaal, B.; Salama, S.; Al-Qasmi, N.; Jaganjac, M. Mycotoxin contamination of food and feed in the Gulf Cooperation Council countries and its detection. Toxicon Off. J. Int. Soc. Toxinol. 2019, 171, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Abdullah, A.B.; Ito, S.; Adhana, K. Estimate of Rice Consumption in Asian Countries and the world Towards 2050. 2005. Available online: http://worldfood.apionet.or.jp/alias.pdf (accessed on 16 July 2021).
- Joint FAO/WHO Expert Committee on Food Additives. Meeting. 73th & World Health Organization. 2011. Safety Evaluation of Certain Food Additives and Contaminants: Prepared by the Seventy-Third Meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA). World Health Organization. Available online: https://apps.who.int/iris/handle/10665/44521 (accessed on 13 March 2022).
- Raad, F.; Nasreddine, L.; Hilan, C.; Bartosik, M.; Parent-Massin, D. Dietary exposure to aflatoxins, ochratoxin A and deoxynivalenol from a total diet study in an adult urban Lebanese population. Food Chem. Toxicol. 2014, 73, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Xia, L.; Rasheed, H.; Routledge, M.N.; Wu, H.; Gong, Y.Y. Super-Sensitive LC-MS analyses of exposure biomarkers for multiple mycotoxins in a rural Pakistan population. Toxins 2022, 14, 193. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.; Ismail, A.; Gong, Y.Y.; Akhtar, S.; Hussain, M. Concentration of aflatoxin M1 and selected heavy metals in mother milk samples from Pakistan. Food Control 2018, 91, 344–348. [Google Scholar] [CrossRef]
- Bhatti, A.B.H.; Dar, F.S.; Waheed, A.; Shafique, K.; Sultan, F.; Shah, N.H. Hepatocellular carcinoma in Pakistan: National trends and global perspective. Gastroenterol. Res. Pract. 2016, 2016, 5942306. [Google Scholar] [CrossRef]
- Murphy, P.; Hendrich, S.; Landgren, C.; Bryant, C. Food Mycotoxins: An Update. J. Food Sci. 2006, 71, 51–65. [Google Scholar] [CrossRef]
- Kabak, B.; Dobson, A.; Var, I. Strategies to Prevent Mycotoxin Contamination of Food and Animal Feed: A Review. Cri. Rev. Food Sci. Nutr. 2006, 46, 593–619. [Google Scholar] [CrossRef]
- Codex Alimentarius Commission. CAC/RCP 51. Prevention and Reduction of 33 Mycotoxin Contamination in Cereals, including Annexes on Ochratoxin A, Zearalenone, Fumonisins and Tricothecenes. 2003. Available online: http://www.codexalimentarius.net/web/standard_list.jsp (accessed on 20 May 2022).
- Dorner, J.; Cole, R. Effect of application of nontoxigenic strains of Aspergillus flavus and A. parasiticus on subsequent aflatoxin contamination of peanuts in storage. J. Stored Prod. Res. 2002, 38, 329–339. [Google Scholar] [CrossRef]
- Lewis, M.H.; Carbone, I.; Luis, J.M.; Payne, G.A.; Bowen, K.L.; Hagan, A.K.; Kemerait, R.; Heiniger, R.; Ojiambo, P.S. Biocontrol strains differentially shift the genetic structure of indigenous soil populations of Aspergillus flavus. Front. Microbiol. 2019, 10, 1738. [Google Scholar] [CrossRef]
- Cotty, P.J. Development of Aspergillus Flavus AF36. 2005. Available online: https://biopesticide.ucr.edu/abstracts/assets/Cotty_abstract.pdf (accessed on 25 May 2022).
- Dorner, J.; Lamb, M.C. Development and commercial use of afla-Guard®, an aflatoxin biocontrol agent. Mycotoxin Res. 2006, 22, 33–38. [Google Scholar] [CrossRef] [PubMed]
- Mauro, A.; Garcia-Cela, E.; Pietri, A.; Cotty, P.J.; Battilani, P. Biological Control Products for Aflatoxin Prevention in Italy: Commercial Field Evaluation of Atoxigenic Aspergillus flavus Active Ingredients. Toxins 2018, 10, 30. [Google Scholar] [CrossRef] [PubMed]
- Senghor, A.; Ortega-Beltran, A.; Atehnkeng, J.; Jarju, P.; Cotty, P.; Bandyopadhyay, R. Aflasafe SN01 is the first biocontrol product approved for aflatoxin mitigation in two nations, Senegal and The Gambia. Plant Dis. 2021, 105, 1461–1473. [Google Scholar] [CrossRef]
- Agbetiameh, D.; Ortega-Beltran, A.; Awuah, R.T.; Atehnkeng, J.; Elzein, A.; Cotty, P.J.; Bandyopadhyay, R. Field efficacy of two atoxigenic biocontrol products for mitigation of aflatoxin contamination in maize and groundnut in Ghana. Biol. Control 2020, 150, 104351. [Google Scholar] [CrossRef] [PubMed]
- Bandyopadhyay, R.; Ortega-Beltran, A.; Akande, A.; Mutegi, C.; Atehnkeng, J.; Kaptoge, L.; Senghor, A.; Adhikari, B.; Cotty, P.J. Biological control of aflatoxins in Africa: Current status and potential challenges in the face of climate change. World Mycotoxin J. 2016, 9, 771–789. [Google Scholar] [CrossRef]
- Konlambigue, M.; Ortega-Beltran, A.; Bandyopadhyay, R.; Shanks, T.; Landreth, E.; Jacob, O. Lessons Learned on Scaling Aflasafe® through Commercialization in Sub-Saharan Africa; International Food Policy Research Institute (IFPRI): Washington, DC, USA, 2020. [Google Scholar] [CrossRef]
- Mahuku, G.S. AR-NAFAKA Project Aflatoxin Management: 2016–2017 Progress. In Proceedings of the Africa RISING—NAFAKA Scaling Project End-of-Project Phase Review Meeting, Dar es Salaam, Tanzania, 3–4 July 2017; International Institute of Tropical Agriculture: Ibadan, Nigeria, 2017. Available online: https://cgspace.cgiar.org/bitstream/handle/10568/89001/ar_mahuku_jul2017.pdf?sequence=1&isAllowed=y (accessed on 15 June 2021).
- Grubisha, L.C.; Cotty, P. Genetic analysis of the Aspergillus flavus vegetative compatibility group to which a biological control agent that limits aflatoxin contamination in US crops belongs. Appl. Environ. Microbiol. 2015, 81, 5889–5899. [Google Scholar] [CrossRef]
- Ren, X.; Zhang, Q.; Zhang, W.; Mao, J.; Li, P. Control of aflatoxigenic molds by antagonistic microorganisms: Inhibitory behaviors, bioactive compounds, related mechanisms, and influencing factors. Toxins 2020, 12, 24. [Google Scholar] [CrossRef]
- Mostafa, A.A.; Al-Rahmah, A.N.; Abdel-Megeed, A.; Sayed, S.R.; Hatamleh, A.A. Antagonistic activities of some fungal strains against the toxigenic Aspergillus flavus isolate and its aflatoxins productivity. J. Pure App. Microbiol. 2013, 7, 169–178. [Google Scholar]
- Bernaldez, V.; Rodriguez, A.; Martin, A.; Lozano, D.; Cordoba, J.J. Development of a multiplex qPCR method for simultaneous quantification in dry-cured ham of an antifungal-peptide Penicillium chrysogenum strain used as protective culture and aflatoxin producing moulds. Food Control 2014, 36, 257–265. [Google Scholar] [CrossRef]
- Kong, Q.; Chi, C.; Yu, J.; Shan, S.; Li, Q.; Li, Q.; Guan, B.; Nierman, W.C.; Bennett, J.W. The inhibitory effect of Bacillus megaterium on aflatoxin and cyclopiazonic acid biosynthetic pathway gene expression in Aspergillus flavus. App. Microbiol. Biotechnol. 2014, 98, 5161–5172. [Google Scholar] [CrossRef]
- Siahmoshteh, F.; Hamidi-Esfahani, Z.; Spadaro, D.; Shams-Ghahfarokhi, M.; Razzaghi-Abyaneh, M. Unraveling the mode of antifungal action of Bacillus subtilis and Bacillus amyloliquefaciens as potential biocontrol agents against aflatoxigenic Aspergillus parasiticus. Food Control 2018, 89, 300–307. [Google Scholar] [CrossRef]
- Yang, X.; Zhang, Q.; Chen, Z.-Y.; Liu, H.; Li, P. Investigation of Pseudomonas fluorescens strain 3JW1 on preventing and reducing aflatoxin contaminations in peanuts. PLoS ONE 2017, 12, e0178810. [Google Scholar] [CrossRef] [PubMed]
- Ghanbari, R.; Molaee Aghaee, E.; Rezaie, S.; Jahed Khaniki, G.; Alimohammadi, M.; Soleimani, M.; Noorbakhsh, F. The inhibitory effect of lactic acid bacteria on aflatoxin production and expression of aflR gene in Aspergillus parasiticus. J. Food Saf. 2018, 38, e12413. [Google Scholar] [CrossRef]
- Bradford, K.J.; Dahal, P.; Van Asbrouck, J.; Kunusoth, K.; Bello, P.; Thompson, J.; Wu, F. The dry chain: Reducing postharvest losses and improving food safety in humid climates. Trends Food Sci. Technol. 2018, 71, 84–93. [Google Scholar] [CrossRef]
- Bakhtavar, M.A.; Afzal, I.; Basra1, S.M.A.; Wahid, A. Implementing the ‘dry chain’ during storage reduces losses and maintains quality of maize grain. Food Sec. 2019, 11, 345–357. [Google Scholar] [CrossRef]
- Shi, H. Investigation of Methods for Reducing Aflatoxin Contamination in Distillers’ Grains. Ph.D. Thesis, Purdue University, West Lafayette, IN, USA, 2016. Available online: https://docs.lib.purdue.edu/open_access_dissertations/1000 (accessed on 3 February 2021).
- Xu, Y.A.; Doel, A.; Watson, S.; Routledge, M.N.; Elliott, C.T.; Moore, S.E.; Gong, Y.Y. Study of an Educational Hand Sorting Intervention for Reducing Aflatoxin B1 in Groundnuts in Rural Gambia. J. Food Prot. 2017, 80, 44–49. [Google Scholar] [CrossRef]
- Yin, Y.-N.; Yan, L.-Y.; Jiang, J.-H.; Ma, Z.H. Biological control of aflatoxin contamination of crops. J. Zhejiang Uni. Sci. B 2008, 9, 787–792. [Google Scholar] [CrossRef]
- Zhang, H.; Dong, M.; Yang, Q.; Apaliya, M.T.; Li, J.; Zhang, X. Biodegradation of zearalenone by Saccharomyces cerevisiae: Possible involvement of ZEN responsive proteins of the yeast. J. Proteom. 2016, 143, 416–423. [Google Scholar] [CrossRef]
- Sipos, P.; Peles, F.; Brasso, D.L.; Beri, B.; Pusztahelyi, T.; Pocsi, I.; Gyori, Z. Physical and chemical methods for reduction in aflatoxin content of feed and food. Toxins 2021, 13, 204. [Google Scholar] [CrossRef]
- Huwig, A.; Freimund, S.; Kappeli, O.; Dutler, H. Mycotoxin detoxication of animal feed by different adsorbents. Toxicol. Lett. 2001, 122, 179–188. [Google Scholar] [CrossRef]
- Khan, M.A.; Asghar, M.A.; Ahmed, A.; Iqbal, J.; Shamsuddin, Z.A. Reduction of aflatoxins in dundi-cut whole red chillies (Capsicum Indicum) by manual sorting technique. Sci. Technol. Develop. 2013, 32, 16–23. [Google Scholar]
- Khan, F.A.; Zahoor, M. In vivo detoxification of aflatoxin B1 by magnetic carbon nanostructures prepared from bagasse. BMC Vet. Res. 2014, 10, 255. [Google Scholar] [CrossRef] [PubMed]
- Saleemi, M.K.; Ashraf, K.; Gul, S.T.; Naseem, M.N.; Sajid, M.S.; Mohsin, M.; He, C.; Zubair, M.; Khan, A. Toxicpathological effects of feeding aflatoxins B1 in broilers and its ameliosration with indigenous mycotoxin binder. Ecotoxicol. Environ. Saf. 2020, 187, 109712. [Google Scholar] [CrossRef] [PubMed]
- Summia, K.; Yasmeen, R.; Zahra, N. Detection of Aflatoxins B1 from Layer and Broiler Feed Samples Collected from Different Cities of Punjab, Pakistan. J. Anim. Health Prod. 2021, 9, 435–442. [Google Scholar] [CrossRef]
- Awan, H.S.; Ahmad, K.S.; Iram, S.; Hanif, N.Q.; Gul, M.M. Analysis and quantification of naturally occurring aflatoxin B1 in dry fruits with subsequent physical and biological detoxification. Nat. Prod. Res. 2021, 36, 3100–3104. [Google Scholar] [CrossRef]
- Gillani, S.W.H.S.; Sadef, Y.; Imran, M.; Raza, H.M.F.; Ghani, A.; Anwar, S.; Ashraf, M.Y.; Hussain, S. Determination and detoxification of aflatoxin and ochratoxin in maize from different regions of Pakistan. Environ. Monit. Assess. 2022, 194, 613. [Google Scholar] [CrossRef]
- Anjum, T.; Iran, W.; Iqbal, M.; Ghaffar, A.; Abbas, M. Identification of degradation products of aflatoxin B1 and B2 resulting after their biodetoxification by aqueous extracts of Acacia nilotica. World Mycotoxin J. 2020, 13, 499–514. [Google Scholar] [CrossRef]
- Rychlik, M.; Humpf, H.-U.; Marko, D.; Danicke, S.; Mally, A.; Berthiller, F.; Klaffke, H.; Lorenz, N. Proposal of a comprehensive definition of modified and other forms of mycotoxins including “masked” mycotoxins. Mycotoxin Res. 2014, 30, 197–205. [Google Scholar] [CrossRef]
- Suman, M.; Generotti, S. Transformation of mycotoxins upon food processing: Masking, binding and degradation phenomena. In Masked Mycotoxins in Food: Formation, Occurrence and Toxicological Relevance; Dall’Asta, C., Berthiller, F., Eds.; RSC Publishing: Cambridge, MA, USA, 2015; pp. 73–89. [Google Scholar]
- Tian, F.; Chun, H.S. Natural products for preventing and controlling aflatoxin contamination of food. In Aflatoxin: Control, Analysis, Detection and Health Risks; Abdulra’uf, L.B., Ed.; IntechOpen: London, UK, 2017. [Google Scholar] [CrossRef]
- Ciegler, A.; Lillehoj, E.B.; Peterson, R.E.; Hall, H.H. Microbial detoxification of aflatoxin. Applied Microbiol. 1966, 14, 934–939. [Google Scholar] [CrossRef]
- El-Nezami, H.; Polychronaki, N.; Yuan, K.L.; Haskard, C.; Juvonen, R.; Salminen, S.; Mykkänen, H. Chemical moieties and interactions involved in the binding of zearalenone to the surface of Lactobacillus rhamnosus strains GG. J. Agri. Food Chem. 2004, 52, 4577–4581. [Google Scholar] [CrossRef]
- Azeem, N.; Nawaz, M.; Anjum, A.A.; Saeed, S.; Sana, S.; Mustafa, A.; Yousuf, M.R. Activity and anti-aflatoxigenic effect of indigenously characterized probiotic lactobacilli against Aspergillus flavus-a common poultry feed contaminant. Animals 2019, 9, 166. [Google Scholar] [CrossRef] [PubMed]
- Madubuonu, N.; Aisida, S.O.; Ahmad, I.; Botha, S.; Zhao, T.K.; Maaza, M.; Ezema, F.I. Bio-inspired iron oxide nanoparticles using Psidium guajava aqueous extract for antibacterial activity. Appl. Phy. A. 2020, 126, 1–8. [Google Scholar] [CrossRef]
- Aisida, S.O.; Akpa, P.A.; Ahmad, I.; Zhao, T.-k.; Maaza, M.; Ezema, F.I. Bio-inspired encapsulation and functionalization of iron oxide nanoparticles for biomedical applications. Eur. Polym. J. 2020, 122, 109371. [Google Scholar] [CrossRef]
- Asghar, M.A.; Zahir, E.; Asghar, M.A.; Iqbal, J.; Rehman, A.A. Facile, one-pot biosynthesis and characterization of iron, copper and silver nanoparticles using Syzygium cumini leaf extract: As an effective antimicrobial and aflatoxin B1 adsorption agents. PLoS ONE 2020, 15, e0234964. [Google Scholar] [CrossRef] [PubMed]
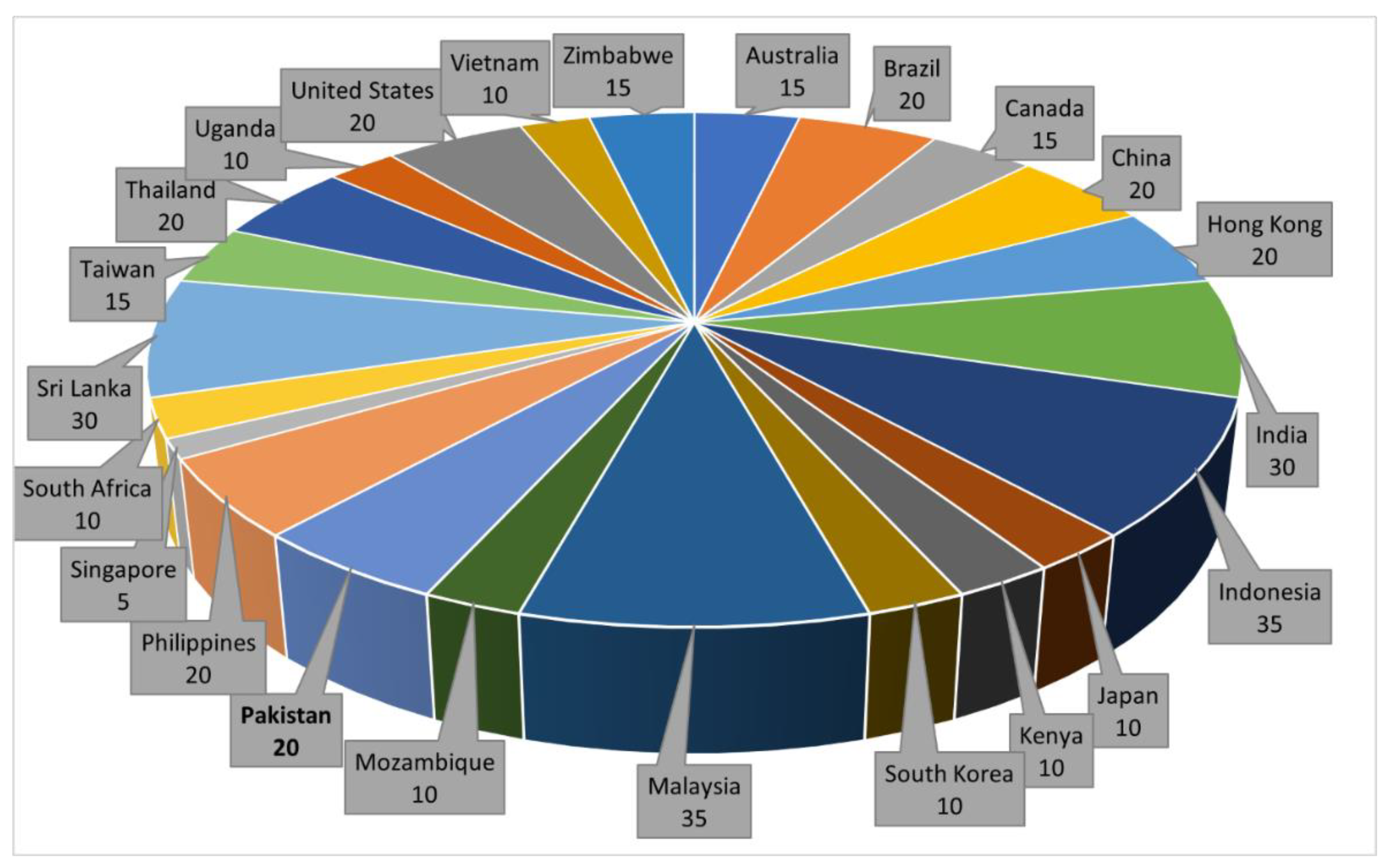
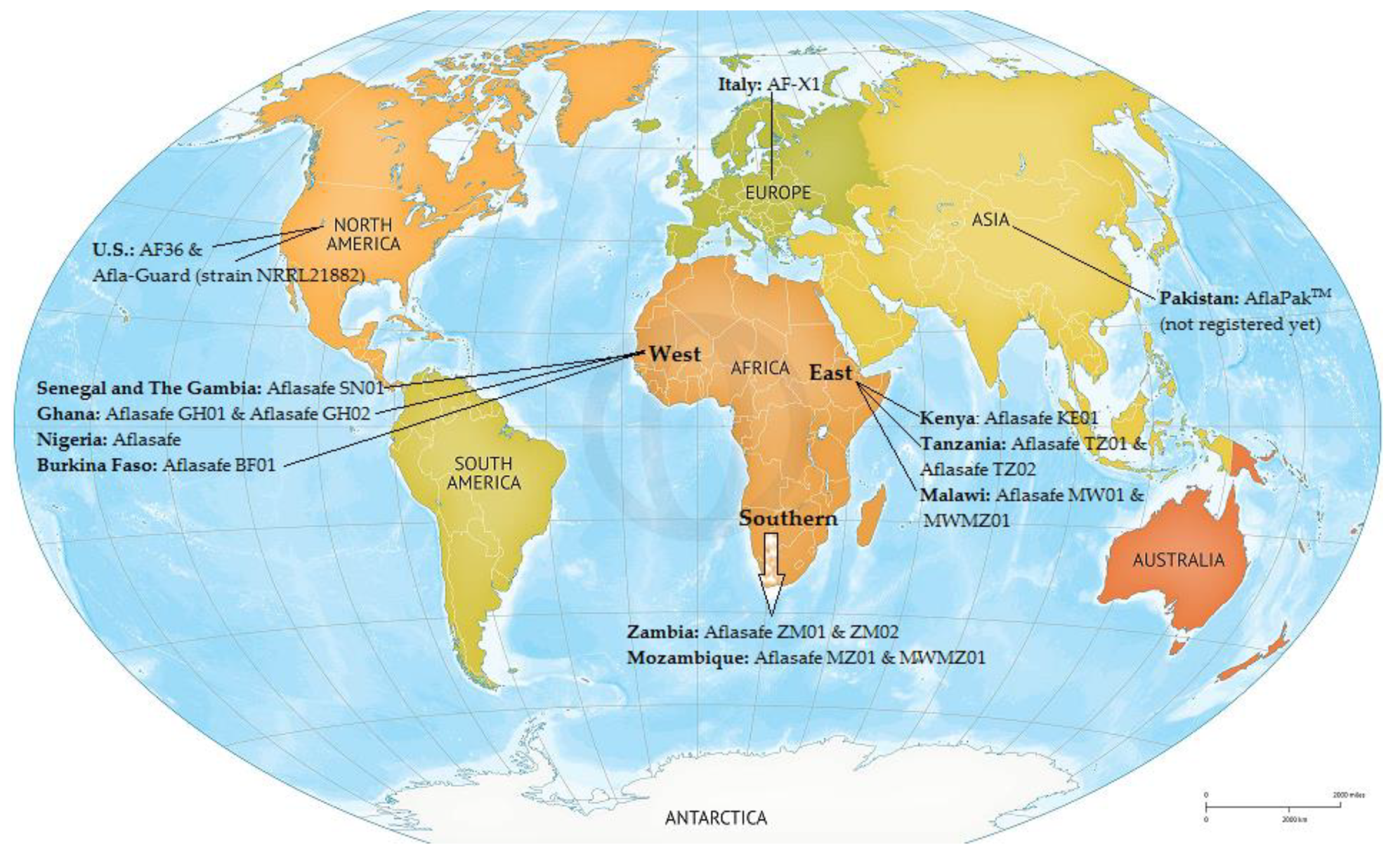
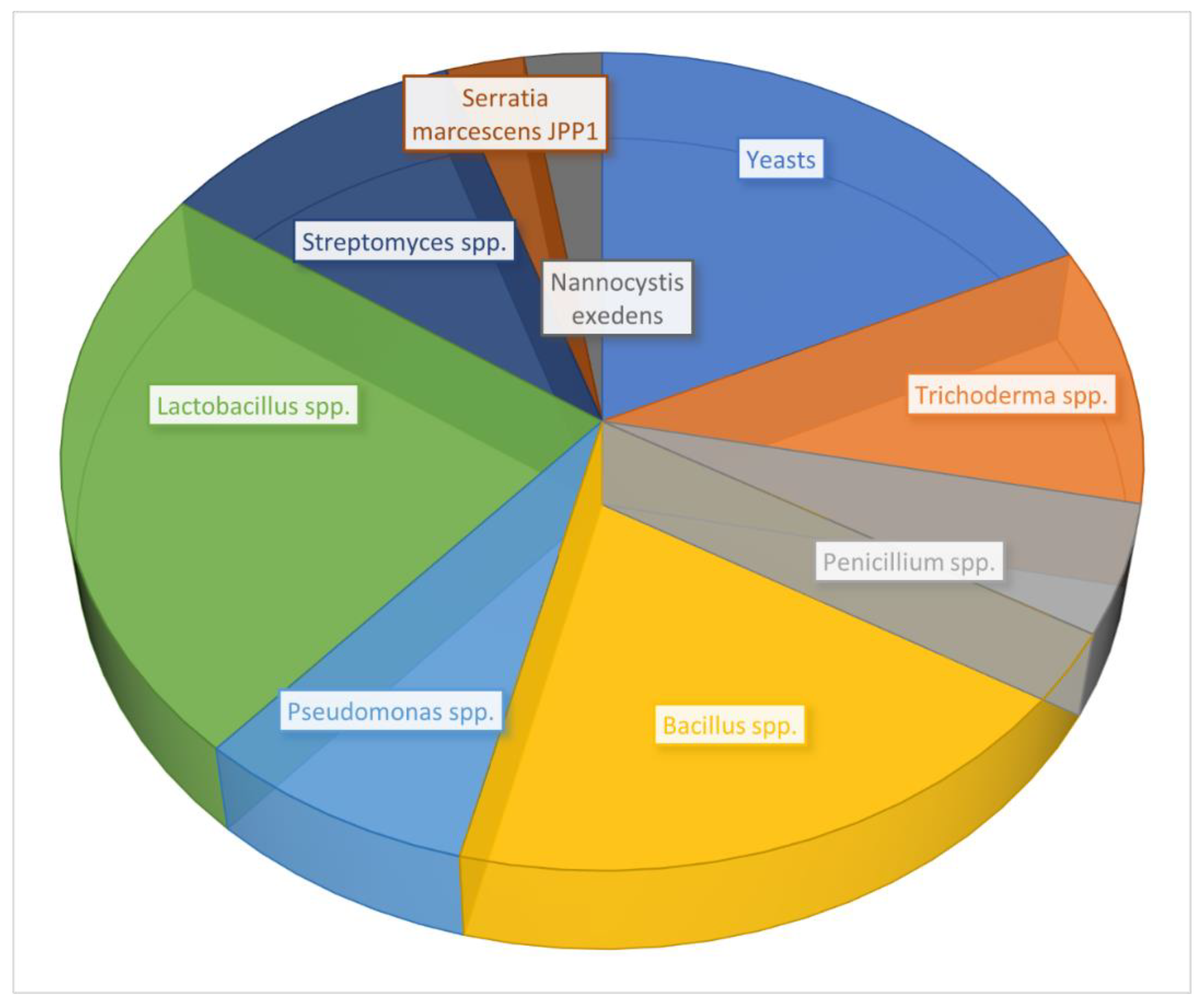
| Commodity | Area of Collection (Year) | AFs | Analytical Method | Contaminated Samples/Total Samples (Incidence %) | Mean (ppb) | Range (ppb) | Over EU MTL (%) * | References |
|---|---|---|---|---|---|---|---|---|
| Wheat | Punjab (2014) | AFB1 | LCMS/MS | 3/195 (2) | 0.04 | LOD–1.59 | – | Xia et al. [116] |
| Wheat flour | Lahore (2018) | AFB1 | TLC | 10/30 (33.33) | 2.197 | 1.83–3.01 | 30 | Zahra et al. [117] |
| Cereal products | Local market, Punjab (2016) | AFB1 AFTs | HPLC HPLC | 121/229 (53) 121/229 (53) | 1.87 2.99 | 1.70–8.20 LOD–9.95 | 43 22 | Alim et al. [118] |
| Wheat grains | Pakistan (2014) | AFB1 AFB2 AFTs | HPLC HPLC HPLC | 48/185 (26) 13/185 (7) 48/185 (26) | 0.51 0.02 0.53 | 0.05–4.78 0.02–0.48 0.02–5.26 | 45.8 0 10.4 | Asghar et al. [119] |
| Wheat based products | Local market, Punjab | AFTs | HPLC | 44/147 (30) | 8.076 | LOD–61.6 | 27.2 | Iqbal et al. [120] |
| Non-branded rice | Punjab (2019) | AFTs | TLC, ELISA | 65/100 (65) | 3.51 | 0.75–8.92 | 27.6 | Tahir et al. [121] |
| Unpacked rice | Punjab | AFB1 | TLC, ELISA | 12/50 (24) | 6.98 | LOD–20.1 | 58 | Nazir et al. [122] |
| Rice | Punjab (2014) | AFB1 | LCMS/MS | 41/62 (66) | 5.04 | LOD–71.56 | – | Xia et al. [116] |
| Rice | Local market, Punjab (2015) | AFB1 AFB2 | LCMS/MS LCMS/MS | 101/180 (56) 87/180 (48) | 5.84 1.91 | 1.5–40 1.5–9.15 | – – | Majeed et al. [123] |
| Brown rice Brown rice White rice White rice | Punjab (2010–2015) | AFTs AFB1 AFTs AFB1 | TLC TLC TLC TLC | 154/1081 (14.24) 854/1081 (79) 216/1170 (18.46) 896/1170 (76.58) | 3.1 8.98 3.27 5.83 | 0.11–49.50 0.95–45 1.37–69.3 0.1–64.05 | – – – – | Sultana et al. [124] |
| Super kernel (SK) basmati rice | Local markets, Punjab | AFTs AFB1 AFB2 | HPLC HPLC HPLC | 28/48 (58) 27/48 (56) 16/48 (33) | 13.92 – – | 2–26 0–27 – | 93 100 – | Mukhtar et al. [125] |
| Brown rice | Local market | AFTs | TLC | 23/25 (92) | – | – | 60 | Nisa et al. [126] |
| Rice and rice products | Local markets, Central Punjab (2012—2013) | AFB1 AFTs | HPLC HPLC | 73/208 (35) 73/208 (35) | 2.40 6.36 | LOD–21.3 LOD–32.2 | 19 24 | Iqbal et al. [127] |
| SK basmati rice SK basmati rice Basmati rice Parboiled rice Broken rice | City area, Lahore | AFB1 AFB2 AFB1 AFB1 AFB1 | TLC, HPLC TLC, HPLC TLC, HPLC TLC, HPLC TLC, HPLC | 48/361 (13.3) 6/361 (1.9) 107/585 (18.3) 30/70 (42.9) 4/11 (36.4) | – – – – – | 1.1–32.9 1.0–8.1 1.0–15.4 1.1–9.2 2.1–25.3 | 6.4 0.3 6.0 10 36.4 | Firdous et al. [128] |
| Brown rice | Karachi (2013) | AFB1 AFTs AFB1 AFTs | TLC HPLC LC-MS/MS ELISA | 100/120 (83) 100/120 (83) 104/120 (87) 104/120 (87) | 3.56 3.79 3.73 3.89 | 0.21–10.54 0.21–11.89 0.10–10.88 0.10–12.39 | – | Iqbal et al. [41] |
| Brown rice | Vendors (2006—2011) | AFB1 AFB2 AFTs | TLC TLC TLC | 250/262 (95.4) 20/262 (7.6) 250/262 (95.4) | 3.80 0.09 3.89 | 1.07–24.65 0.52–2.62 1.07–27.27 | 23.68 0 23.68 | Asghar et al. [129] |
| Export quality basmati rice | Vendors (2006—2011) | AFB1 | TLC | 1500/2047 (73.3) | 1.15 | 1.17–6.91 | 2.74 | Asghar et al. [130] |
| Rice | Retail markets, Punjab (2011—2012) | AFB1 | HPLC | 38/68 (56) | 8.23 | – | 18 | Majeed et al. [131] |
| Brown, white and parboiled rice | Export areas (2010) | AFB1 AFB2 | HPLC HPLC | 254/519 (48.9) 24/519 (4.6) | 1.18 0.61 | LOD–16.7 0.5–2.64 | 5.58 – | Firdous et al. [132] |
| White rice Brown rice Broken rice Paddy rice Parboiled rice | Retail markets, Fields, Punjab (2010) | AFTs AFTs AFTs AFTs AFTs | HPLC HPLC HPLC HPLC HPLC | 39/93 (42) 28/84 (33) 55/109 (50) 37/58 (64) 26/69 (37) | 7.10 9.85 8.50 16.35 14.20 | LOD–58.2 LOD–28.7 LOD–38.5 LOD–68.3 LOD–39.4 | 48 43 90 40 73 | Iqbal et al. [115] |
| Rice | (2008—2009) | AFB1 AFTs | TLC TLC | 28/40 (70) 28/40 (70) | 3.7 4.9 | – – | 50 – | Hussain et al. [108] |
| Maize | Northern areas, KPK | AFB1 AFB2 | HP–TLC HP–TLC | 9/10 (90) 5/10 (50) | 27.87 1.35 | 9.09–48.48 0.05–3.75 | – – | Murad et al. [133] |
| Branded corn Non-branded corn Non-branded corn Non-branded corn | Local market, Lahore (2016) | AFB1 AFB1 AFB2 AFG1 | TLC TLC TLC TLC | 75/75 (100) – 9/75 (12) 6/75 (8) | 7.07 67.37 8.95 16.46 | 1.02–7.07 1.25–67.73 0–8.95 0–16.46 | 23 64 12 8 | Hussain et al. [134] |
| Maize | (2016—2019) | AFB1 AFB2 AFG1 AFG2 AFTs | HPLC HPLC HPLC HPLC HPLC | 267/324 (82.4) 20/324 (6.2) 13/324 (4.0) 7/324 (2.2) 267/324 (82.4) | 17.76 0.50 0.20 0.07 18.53 | 0.69–315.32 1.35–25.65 0.98–11.41 0.94–4.50 0.69–356.72 | 66.6 0 0 0 56.2 | Asghar et al. [88] |
| Maize | Faisalabad | AFTs | HPLC | 41/46 (89) | 29.8 | 27.4–362.8 | 100 | Hassan et al. [135] |
| Yellow maize | Punjab | AFB1 AFB2 AFG2 | HPLC HPLC HPLC | 28/36 (77.7) 22/36 (61.1) 14/36 (38.8) | – – – | 11–17 9–13 5–9 | 100 100 100 | Manzoor et al. [136] |
| AFB1 AFB2 AFG2 | HPLC HPLC HPLC | 20/36 (55.5) 13/36 (36.1) 7/36 (19.4) | – – – | 7–13 2–6 1–6 | 100 – – | |||
| Corn based product | City area, Lahore | AFB1 AFB2 | HPLC HPLC | 52/100 (52) 25/100 (25) | – – | 2.0 –1405.3 1.0—55.2 | 52 – | Firdous et al. [128] |
| Maize | Punjab | AFB1 AFB2 | HPLC HPLC | 73/75 (97.3) 58/75 (78.9) | 54.54 1.46 | LOD–409.3 LOD–8.03 | 77.3 28 | Iram et al. [137] |
| Corn Corn Corn products Corn products | Retail markets, local industries, Punjab (2011–2012) | AFB1 AFTs AFB1 AFTs | HPLC HPLC HPLC HPLC | 37/105 (35.23) 37/105 (35.23) 43/102 (42.15) 43/102 (42.15) | 7.90 12.08 5.47 7.85 | – – – – | 14 28 14 20 | Majeed et al. [130] |
| Maize | Pakistan (2006–2007) | AFB1 AFB2 AFG1 | HPTLC HPLC HPLC | 18/65 (27.69) 12/65 (18.46) 2/65 (1.3) | 192 40 9 | 5–850 3–187 8–11 | 27.69 18.46 | Khatoon et al. [100] |
| Maize | Upper Swat, Lower Swat, KPK (2007) | AFB1 AFB1 | HPLC HPLC | 14/18 (77.78) 16/18 (88.89) | 14.94 16.22 | 0–30.96 0–27.28 | 50 * 37 * | Shah et al. [138] |
| Maize | Urban areas Semi-urban Rural areas | AFTs AFTs AFTs | HPLC HPLC HPLC | (80) (87) (90) | 45 54 62 | – – – | – – – | Ahsan et al. [139] |
| Commodity | Area of Collection (Year) | AFs | Analytical Method | Contaminated Samples/Total Samples (Incidence %) | Mean (ppb) | Range (ppb) | Over EU MTL (%) * | References |
|---|---|---|---|---|---|---|---|---|
| Local edible seeds Local edible seeds Imported edible seeds Imported edible seeds | Local market, Punjab (2019) | AFB1 AFTs AFB1 AFTs | HPLC HPLC HPLC HPLC | 108/189 (57) 108/189 (57) 92/162 (56.7) 92/162 (56.7) | 13.551 21.17 9.21 15.59 | – – – – | 100 100 100 100 | Waqas et al. [144] |
| Local edible oil Local edible oil Imported edible oil Imported edible oil | Local market, Punjab (2019) | AFB1 AFTs AFB1 AFTs | HPLC HPLC HPLC HPLC | 103/213 (48.3) 103/213 (48.3) 78/180 (43.3) 78/180 (43.3) | 8.83 14.52 5.04 9.34 | – – – – | – – – – | Waqas et al. [144] |
| Peanut oil | Local market, Peshawar (2020—2021) | AFB1 AFB2 AFG1 AFTs | TLC TLC TLC TLC | 42/60 (70) 31/60 (51.7) 2/60 (3.3) 42/60 (70) | 6.20 3.40 4.90 8.59 | 0.12–25 0.2–22 2.4–7.4 0.12–55 | 41.7 – – 35 | Hussain et al. [145] |
| Raw peanuts in shells Peanuts without shells Roasted peanuts R. P. without shells Peanuts butter Peanuts cookies Packed peanuts (nimko) | Local market, Punjab | AFTs AFTs AFTs AFTs AFTs AFTs AFTs | HPLC HPLC HPLC HPLC HPLC HPLC HPLC | 13/22 (59) 16/29 (55) 19/31 (61) 13/19 (68) 16/32 (50) 10/24 (42) 8/41 (20) | 6.4 9.6 10.4 12.3 2.4 4.6 3.4 | LOD–59.8 LOD–82.1 LOD–71.3 LOD–119 LOD–32.2 LOD–31.8 LOD–11.4 | 32 17 29 36 25 13 – | Iqbal et al. [146] |
| Roasted peanut Salty peanuts Unripe peanuts | Pothohar plateau, Punjab | AFB1 AFB1 AFB1 | HPLC HPLC HPLC | 19/24 (79.25) 14/24 (58.5) 15/24 (62.5) | 45.03 38.45 42.67 | 14–98 14–85 15–85 | 100 100 100 | Abbas et al. [143] |
| Peanut (nimko) | Lahore and Faisalabad | AFTs | HPLC | 3/5 (60) | – | 0.21–2.08 | – | Mushtaq et al. [147] |
| Peanuts with shell Peanut without shell | KPK and Northern areas (2009) | AFTs AFTs | LC LC | 4/10 (40) 5/10 (50) | 5.10 5.20 | 1.5–14.5 0.7–12.8 | 10 20 | Lutfullah et al. [148] |
| Peanut and peanut products | Local market | AFB1 AFB2 | TLC TLC | – – | 66.48 7.71 | 5.0–183.2 7–46.7 | – – | Mobeen et al. [103] |
| Sesame seeds | Punjab (2021) | AFB1 AFTs | HPLC HPLC | 14/45 (31.1) 14/45 (31.1) | 9.96 11.7 | LOD–65.5 LOD–65.5 | Iqbal et al. [149] | |
| Sesame seeds | Rainfed (fresh) Rainfed (stored) Irrigated areas (fresh) Irrigated (stored) (2018) | AFB1 AFB1 AFB1 AFB1 | TLC TLC TLC TLC | 44/50 (88) 50/50 (100) 48/50 (96) 49/50 (98) | 15.74 33.8 20.5 27.56 | 1.2–60 23–50 12–48 15–60 | 100 100 100 100 | Ajmal et al. [150] |
| Commodity | Area of Collection (Year) | AFs | Analytical Method | Contaminated Samples/Total Samples (Incidence %) | Mean (ppb) | Range (ppb) | Over EU MTL (%) * | Reference |
|---|---|---|---|---|---|---|---|---|
| Nuts (winter) Nuts (summer) | Southern Punjab (2019) | AFTs AFTs | HPLC HPLC | 180/414 (43.4) 122/365 (33.4) | 15.43 12.71 | 0.05–50.5 0.05–45.6 | 11.5 * 12.2 * | Razis et al. [156] |
| Dry fruits Apricot kernel Apricot kernel Apricot kernel | Gilgit-Baltistan, Ghizer, Skardu | AFB1 AFB1 AFB1 AFB1 | TLC TLC TLC TLC | 75/300 (25) 7/50 (14) 10/50 (20) 13/50 (26) | 5 2 3 3.5 | 0.5–15 0.5–4 1–6 0.5–5 | 40 28.57 30 30.76 | Ali et al. [155] |
| Walnut Walnut Walnut | AFB1 AFB1 AFB1 | TLC TLC TLC | 11/50 (22) 15/50 (30) 19/50 (38) | 4.5 6 8 | 0.6–10 0.6–13 0.6–15 | 45.5 40 47.37 | ||
| Nuts | Punjab and KPK (2016– 2017) | AFTs | HPLC | 128/320 (40) | 4.8 | LOD–20.70 | 34.6 | Iqbal et al. [146] |
| Apricot kernels Almonds with shell Almonds without shell Walnuts with shell Walnuts without shell | Local market, KPK, and northern areas (2009) | AFTs AFTs AFTs AFTs AFTs | LC LC LC LC LC | 4/15 (26.67) 0/10 (0) 3/10 (30) 4/10 (40) 7/10 (70) | 2.65 – 2.13 6.45 3.43 | 0.7–5.6 – 1.2–3.4 1.5–13.5 0.8–10.8 | 25 – – 50 42.85 | Lutfullah et al. [148] |
| Pistachios with shell Without shell Pine nuts with shell | Local market, KPK, and northern areas (2009) | AFTs AFTs AFTs | LC LC LC | 2/10 (20) 5/10 (50) 2/10 (20) | 2.10 6.34 3.25 | 1.2–3.0 2.0–14.0 2.6–3.9 | – 40 – | Lutfullah et al. [148] |
| Commodity | Area of Collection (Year) | AFs | Analytical Method | Contaminated Samples/Total Samples (Incidence %) | Mean (ppb) | Range (ppb) | Over EU MTL (%) * | Reference |
|---|---|---|---|---|---|---|---|---|
| Red chili sauce Red chili sauce Green chili sauce Green chili sauce Red chili garlic sauce Red chili garlic sauce | Dhaba, local restaurants, and fast-food outlets, Punjab (2018) | AFB1 AFTs AFB1 AFTs AFB1 AFTs | LC LC LC LC LC LC | 65/87 (74.71) 65/87 (74.71) 60/80 (75) 60/80 (75) 64/85 (75) 65/87 (74.71) | 9.7 23.86 4.43 15.76 4.36 18.86 | LOD–115.5 LOD–120 LOD–71.20 LOD–78 LOD–85.5 LOD–90 | – – – – – – | Iqbal et al. [168] |
| Dried red chilies | Kunri, Sindh (2012) | AFB1 AFB2 AFG1 AFTs | TLC, HPLC TLC, HPLC TLC, HPLC TLC, HPLC | 46/69 (67) 46/69 (67) 1/69 (1.44) 46/69 (67) | 131.7 108.98 0.4 243.21 | 1.2–600 – – – | – – – – | Akhund et al. [169] |
| Packed spices Unpacked spices | Local market (2014) | AFTs AFTs | HPLC HPLC | 49/100 (49) 74/100 (74) | 4.789 26.34 | – – | – – | Naz et al. [170] |
| Unpacked composite spices | Local market, Karachi, (2015) | AFTs | HPLC | 58/75 (77) | 4.63 | 0.68–25.74 | 3 * | Asghar et al. [171] |
| Whole red chilies Powder Crushed | Warehouses, supermarkets, and shops (2006–2011) | AFTs AFTs AFTs | TLC TLC TLC | 223/226 (98.7) 64/69 (92.8) 33/36 (94.4) | 11.7 27.8 31.2 | 1.3–91.4 5.1–77.9 1.6–93.7 | 9.7 * 52.8 * 52.8 * | Khan et al. [172] |
| Spices | Local market, Peshawar | AFTs | TLC | 12/18 (66.67) | 2.86 | 1.86–6.54 | 58 | Hussain et al. [102] |
| Whole chilies Powdered chilies | Faisalabad (2008) | AFB1 AFB1 | HPLC HPLC | 16/22 (73) 19/22 (86.4) | 32.20 26.64 | 0.00–89.56 0.00–96.3 | – – | Iqbal et al. [173] |
| Dried chilies | Local market | AFTs | HPLC | 13/13 (100) | 33.43 | 0.1–96.2 | 76.92 | Paterson [174]. |
| Commodity | Area of Collection (Year) | Afs | Analytical Method | Contaminated Samples/Total Samples (Incidence %) | Mean (ppb) | Range (ppb) | Over EU MTL (%) * | References |
|---|---|---|---|---|---|---|---|---|
| Poultry feed | Punjab (2019) | AFB1 | LC-MS | 150/150 (100) | 39.9 | – | 73 | Sarwat et al. [186] |
| Poultry feed samples | Northern Pakistan (2018) | AFB1 | ELISA | 37/40 (92.5) | 54.56 | – | – | Naveed et al. [187] |
| Poultry feed | Baluchistan | AFB1 | ELISA | 77/100 (77) | – | – | – | Majeed et al. [188] |
| Cattle feed | Punjab | AFB1 | TLC and ELISA | 25/60 (41.66) | 15.59 | 1.9–28.5 | 12 | Nazir et al. [122] |
| Animal feed | Punjab, Pakistan (2017–2018) | AFB1 | LC | 126/193 (65.3%) | 25.0 | – | 45.6 | Waqas et al. [189] |
| Cotton seeds Cotton seeds cake | Karachi and Hyderabad | AFB1 | ELISA and HPLC | 88/110 (80) 97/110 (88) | 69 89 | – – | 64 71 | Shar et al. [190] |
| Bovine feed | Lahore | AFB1 AFB2 AFTs | TLC TLC TLC | 30/50 (60) 10/50 (20) 30/50 (60) | 214.9 46.4 229.7 | 24.1–361.4 4.5–81.3 28.6–394.3 | – – – | Summia et al. [185] |
| Animal feed | Dairy farms, Punjab (2015) | AFB1 | ELISA | (53) | 43.15 | 8–119 | 95 * | Akbar et al. [191] |
| Mixed layer poultry feed | Karachi (2015–2016) | AFB1 | ELISA | 132/132 (100) | 25.51 | 4.23–72.27 | 31 | Iram et al. [192] |
| Commercial feed Fresh fodder Leftover bread | Punjab (2014–2015) | AFB1 AFB1 AFB1 | ELISA ELISA ELISA | 22/72 (30.5) 2/72 (2.8) 62/72 (88.9) | 4.92 3.04 6.72 | 1.04–9.76 0.64–4.61 3.96–11.34 | 30.5 2.8 88.9 | Ismail et al. [193] |
| Animal feed | Farmers, industry, and general stores, Punjab (2012–2013) | AFB1 AFTs | HPLC HPLC | 69/105 (66) 69/105 (66) | 4.71 6.84 | 0.09–145.7 LOQ–165.5 | – – | Iqbal et al. [194] |
| Feed ingredients Dairy feed | Lahore (2013–2014) | AFB1 AFB1 | ELISA ELISA | 54/125 (43) 41/90 (45.56) | 36.53 16.54 | 1.02–210.07 0.56–55.17 | – – | Chohan et al. [195] |
| Poultry feed ingredient Poultry feeds | Various parts of country (2009–2010) | AFB1 AFB1 | TLC TLC | 46/77 (60) (44.39) | 37.62 23.75 | LOD–56 LOD–78 | – – | Anjum et al. [183] |
| Poultry feed | Broiler farms (2009–2010) | AFB1 | TLC | 88/96 (91.66) | 47.64 | 10–166 | 82.30 * | Rashid et al. [179] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ajmal, M.; Bedale, W.; Akram, A.; Yu, J.-H. Comprehensive Review of Aflatoxin Contamination, Impact on Health and Food Security, and Management Strategies in Pakistan. Toxins 2022, 14, 845. https://doi.org/10.3390/toxins14120845
Ajmal M, Bedale W, Akram A, Yu J-H. Comprehensive Review of Aflatoxin Contamination, Impact on Health and Food Security, and Management Strategies in Pakistan. Toxins. 2022; 14(12):845. https://doi.org/10.3390/toxins14120845
Chicago/Turabian StyleAjmal, Maryam, Wendy Bedale, Abida Akram, and Jae-Hyuk Yu. 2022. "Comprehensive Review of Aflatoxin Contamination, Impact on Health and Food Security, and Management Strategies in Pakistan" Toxins 14, no. 12: 845. https://doi.org/10.3390/toxins14120845
APA StyleAjmal, M., Bedale, W., Akram, A., & Yu, J.-H. (2022). Comprehensive Review of Aflatoxin Contamination, Impact on Health and Food Security, and Management Strategies in Pakistan. Toxins, 14(12), 845. https://doi.org/10.3390/toxins14120845






