Measurement of Microcystin Activity in Human Plasma Using Immunocapture and Protein Phosphatase Inhibition Assay
Abstract
1. Introduction
2. Results
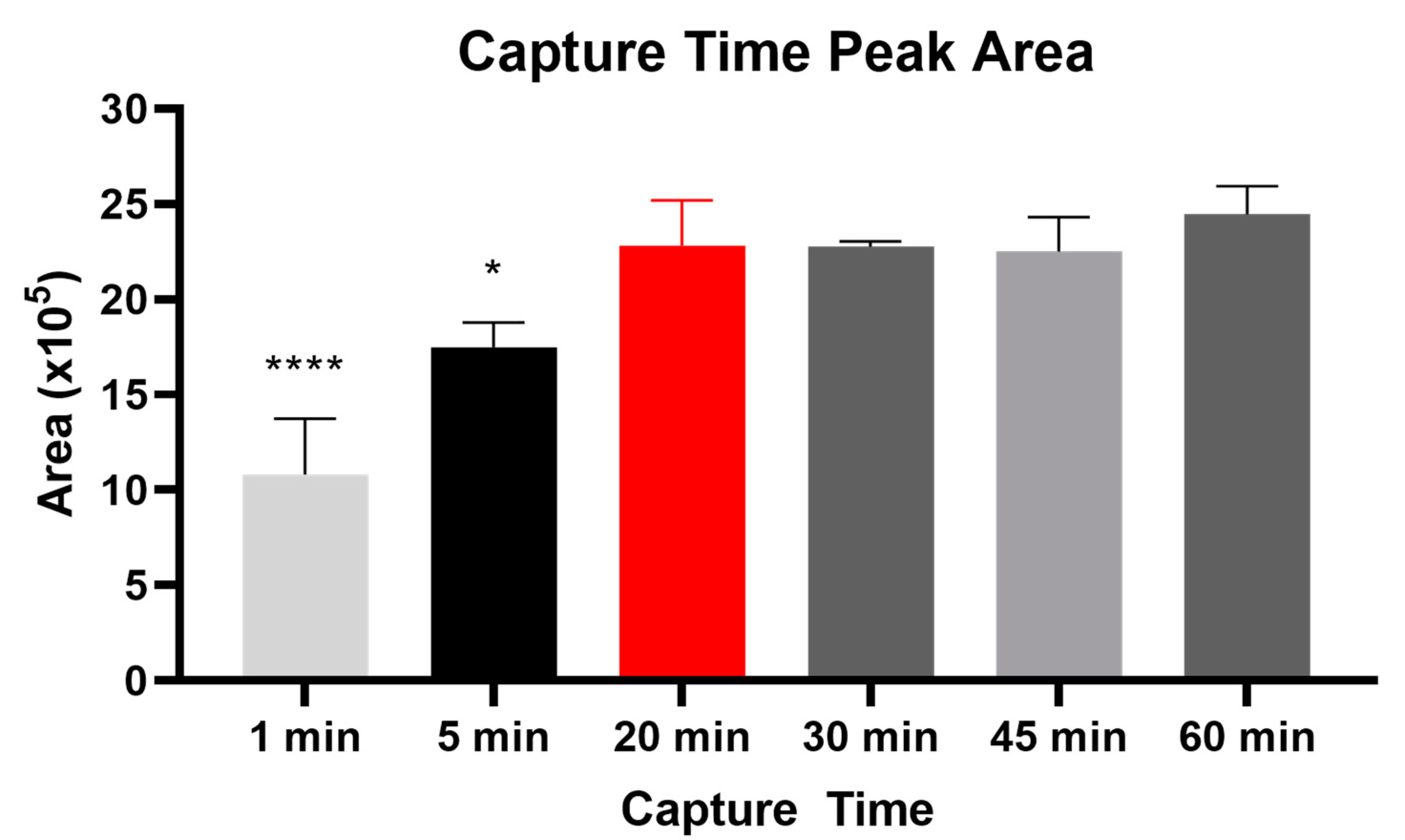
2.1. Method Optimization
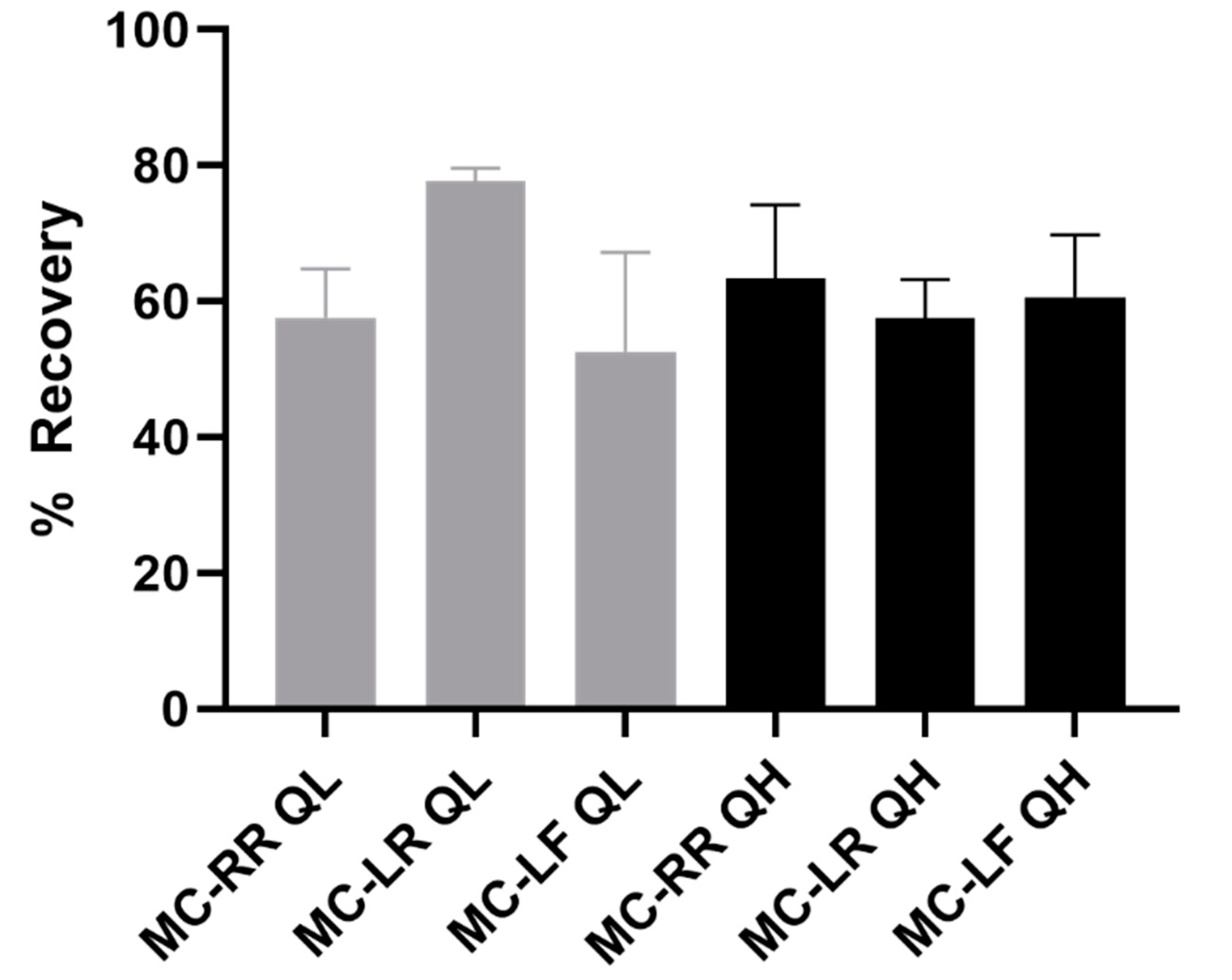
2.2. MC Immunocapture Recovery
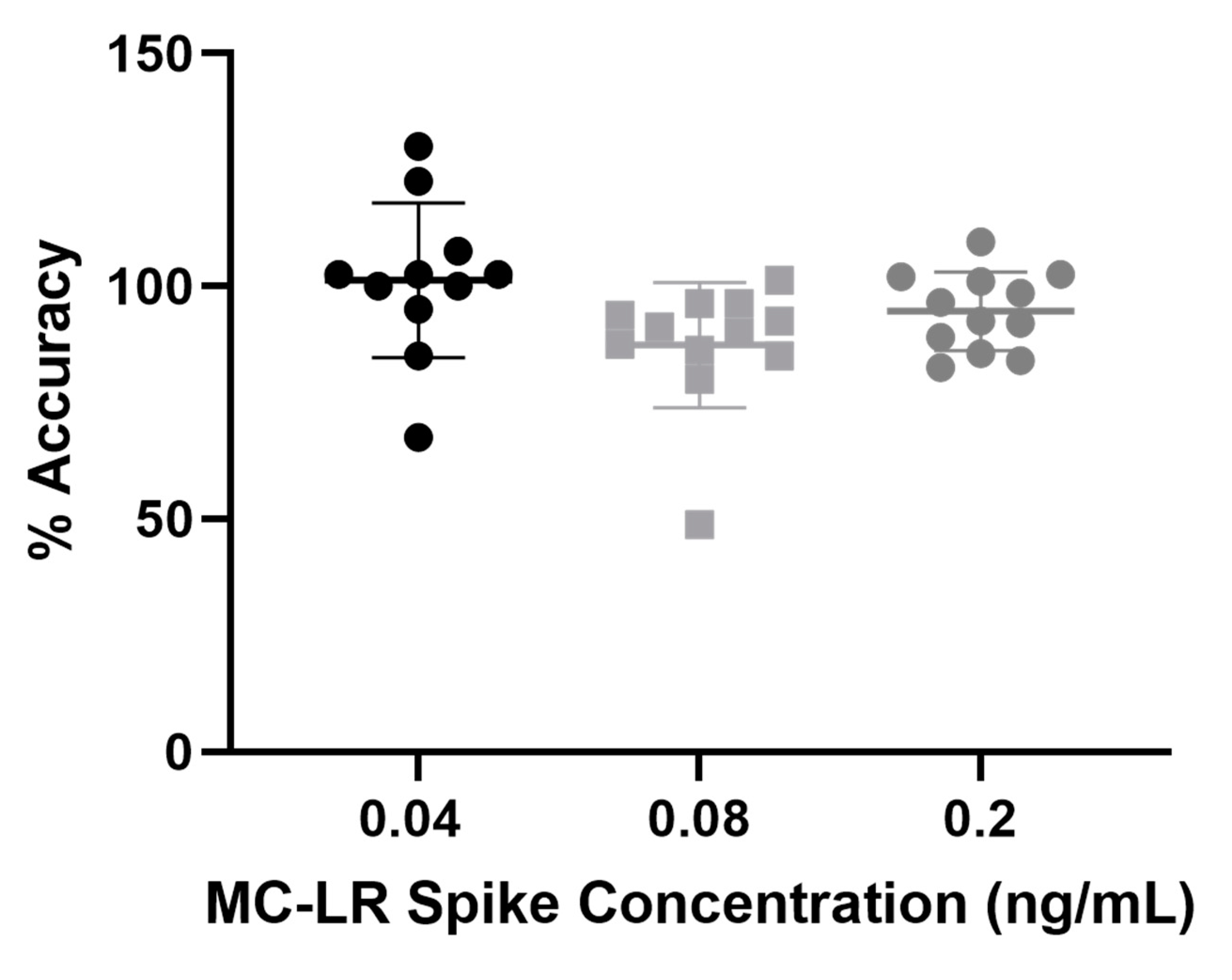
2.3. Validation of MC-LR Detection Using PPIA in Human Plasma
2.4. Detection of Conjugated MCs
2.5. Analysis of Plasma Samples from Florida Residents
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Chemicals and Materials
5.2. Human Plasma
5.3. Preparation of Calibrators and Quality Control Samples
5.4. MC Immunocapture and Protein Phosphatase Inhibition Assay
5.5. MC Capture Time Optimization
5.6. Preparation of Samples for Immunocapture Recovery
5.7. Conjugated MC Synthesis, Confirmation, and Specificity
5.8. Assay Automation
5.9. Data Analysis and Software
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Disclaimer
References
- Codd, G.A. Cyanobacterial Toxins: Occurrence, Properties and Biological Significance. Water Sci. Technol. 1995, 32, 149–156. [Google Scholar] [CrossRef]
- Carmichael, W.W.; Beasley, V.; Bunner, D.L.; Eloff, J.N.; Falconer, I.; Gorham, P.; Harada, K.I.; Krishnamurthy, T.; Min-Juan, Y.; Moore, R.E.; et al. Naming of Cyclic Heptapeptide Toxins of Cyanobacteria (Blue-Green Algae). Toxicon 1988, 26, 971–973. [Google Scholar] [CrossRef]
- Duy, T.N.; Lam, P.K.S.; Shaw, G.R.; Connell, D.W. Toxicology and Risk Assessment of Freshwater Cyanobacterial (Blue-Green Algal) Toxins in Water. In Reviews of Environmental Contamination and Toxicology: Continuation of Residue Reviews; Ware, G.W., Ed.; Springer: New York, NY, USA, 2000; pp. 113–185. ISBN 978-1-4757-6429-1. [Google Scholar]
- Carmichael, W.W. A Status Report on Planktonic Cyanobacteria (Blue-Green Algae) and Their Toxins; U.S. Environmental Protection Agency, Research and Development, Environmental Monitoring Systems Laboratory: Cincinnati, OH, USA, 1992.
- Kubickova, B.; Babica, P.; Hilscherová, K.; Šindlerová, L. Effects of Cyanobacterial Toxins on the Human Gastrointestinal Tract and the Mucosal Innate Immune System. Environ. Sci. Eur. 2019, 31, 31. [Google Scholar] [CrossRef]
- Fonseca, V.B.; da Sopezki, M.S.; Yunes, J.S.; Zanette, J. Effect of a Toxic Microcystis Aeruginosa Lysate on the MRNA Expression of Proto-Oncogenes and Tumor Suppressor Genes in Zebrafish. Ecotoxicol. Environ. Saf. 2018, 161, 729–734. [Google Scholar] [CrossRef] [PubMed]
- Lone, Y.; Koiri, R.K.; Bhide, M. An Overview of the Toxic Effect of Potential Human Carcinogen Microcystin-LR on Testis. Toxicol. Rep. 2015, 2, 289–296. [Google Scholar] [CrossRef]
- Ueno, Y.; Nagata, S.; Tsutsumi, T.; Hasegawa, A.; Watanabe, M.F.; Park, H.D.; Chen, G.C.; Chen, G.; Yu, S.Z. Detection of Microcystins, a Blue-Green Algal Hepatotoxin, in Drinking Water Sampled in Haimen and Fusui, Endemic Areas of Primary Liver Cancer in China, by Highly Sensitive Immunoassay. Carcinogenesis 1996, 17, 1317–1321. [Google Scholar] [CrossRef]
- Falconer, I.R. Tumor Promotion and Liver Injury Caused by Oral Consumption of Cyanobacteria. Environ. Toxicol. Water Qual. 1991, 6, 177–184. [Google Scholar] [CrossRef]
- Backer, L.C.; McNeel, S.V.; Barber, T.; Kirkpatrick, B.; Williams, C.; Irvin, M.; Zhou, Y.; Johnson, T.B.; Nierenberg, K.; Aubel, M.; et al. Recreational Exposure to Microcystins during Algal Blooms in Two California Lakes. Toxicon 2010, 55, 909–921. [Google Scholar] [CrossRef]
- D’Anglada, L.V.; Donohue, J.M.; Strong, J.; Hawkins, B.; Armstrong, A.Q.; Fleming, C.; Carlson-Lynch, H.; Blain, R. Health Effects Support Document for the Cyanobacterial Toxin Microcystins; United States Environmental Protection Agency: Washington, DC, USA, 2015.
- Williams, C.D.; Aubel, M.T.; Chapman, A.D.; D’Aiuto, P.E. Identification of Cyanobacterial Toxins in Florida’s Freshwater Systems. Lake Reserv. Manag. 2007, 23, 144–152. [Google Scholar] [CrossRef]
- Svirčev, Z.; Drobac, D.; Tokodi, N.; Mijović, B.; Codd, G.A.; Meriluoto, J. Toxicology of Microcystins with Reference to Cases of Human Intoxications and Epidemiological Investigations of Exposures to Cyanobacteria and Cyanotoxins. Arch. Toxicol. 2017, 91, 621–650. [Google Scholar] [CrossRef]
- Roberts, V.A.; Vigar, M.; Backer, L.; Veytsel, G.E.; Hilborn, E.D.; Hamelin, E.I.; Vanden Esschert, K.L.; Lively, J.Y.; Cope, J.R.; Hlavsa, M.C.; et al. Surveillance for Harmful Algal Bloom Events and Associated Human and Animal Illnesses—One Health Harmful Algal Bloom System, United States, 2016–2018. MMWR Morb. Mortal. Wkly. Rep. 2020, 69, 1889–1894. [Google Scholar] [CrossRef] [PubMed]
- Svirčev, Z.; Lalić, D.; Bojadžija Savić, G.; Tokodi, N.; Drobac Backović, D.; Chen, L.; Meriluoto, J.; Codd, G.A. Global Geographical and Historical Overview of Cyanotoxin Distribution and Cyanobacterial Poisonings; Springer: Berlin/Heidelberg, Germany, 2019; Volume 93, ISBN 0123456789. [Google Scholar]
- Chen, L.; Giesy, J.P.; Adamovsky, O.; Svir, Z.; Meriluoto, J.; Codd, G.A.; Mijovic, B.; Shi, T.; Tuo, X.; Li, S.; et al. Challenges of Using Blooms of Microcystis spp. in Animal Feeds: A Comprehensive Review of Nutritional, Toxicological and Microbial Health Evaluation. Sci. Total Environ. 2021, 764, 142319. [Google Scholar] [CrossRef] [PubMed]
- Jochimsen, E.M.; Carmichael, W.W.; An, J.; Cardo, D.M.; Cookson, S.T.; Holmes, C.E.M.; Antunes, M.B.; de Filho, D.A.M.; Lyra, T.M.; Barreto, V.S.T.; et al. Liver Failure and Death after Exposure to Microcystins at a Hemodialysis Center in Brazil. N. Engl. J. Med. 1998, 338, 873–878. [Google Scholar] [CrossRef] [PubMed]
- Gupta, N.; Pant, S.C.; Vijayaraghavan, R.; Rao, P.V.L. Comparative Toxicity Evaluation of Cyanobacterial Cyclic Peptide Toxin Microcystin Variants (LR, RR, YR) in Mice. Toxicology 2003, 188, 285–296. [Google Scholar] [CrossRef]
- Stoner, R.D.; Adams, W.H.; Slatkin, D.N.; Siegelman, H.W. The Effects of Single L-Amino Acid Substitutions on the Lethal Potencies of the Microcystins. Toxicon 1989, 27, 825–828. [Google Scholar] [CrossRef]
- WHO. Guidelines for Drinking-Water Quality; WHO: Geneva, Switzerland, 1998; Volume 2.
- Drinking Water Health Advisory for the Cyanobacterial Microcystin Toxins; United States Environmental Protection Agency: Washington, DC, USA, 2015.
- Bouaïcha, N.; Miles, C.O.; Beach, D.G.; Labidi, Z.; Djabri, A.; Benayache, N.Y.; Nguyen-Quang, T. Structural Diversity, Characterization and Toxicology of Microcystins. Toxins 2019, 11, 714. [Google Scholar] [CrossRef]
- Botes, D.P.; Wessels, P.L.; Kruger, H.; Runnegar, M.T.C.; Santikarn, S.; Smith, R.J.; Barna, J.C.J.; Williams, D.H. Structural Studies on Cyanoginosins-LR, -YR, -YA, and -YM, Peptide Toxins from Microcystis Aeruginosa. J. Chem. Soc. Perkin Trans. 1 1985, 1985, 2747–2748. [Google Scholar] [CrossRef]
- Sivonen, K. Chapter 3 Cyanobacterial Toxins. In Toxic Cyanobacteria. Water, a Guide to Their Health Consequences, Monitoring and Management; CRC Press: Boca Raton, FL, USA, 1999. [Google Scholar]
- Stotts, R.R.; Na, M.; Haschek, W.M.; Riivehart, K.L.; Careticxael, W.W.; Dahlem, A.M.; Beasley, V.A.L.R.; Em, A.M.D.; Structural, V.R.B. Structural Modifications Imparting Reduced Toxicity in Microcystins from Microcystis spp. Toxicon 1993, 31, 783–789. [Google Scholar] [CrossRef]
- Gulledge, B.M.; Aggen, J.B.; Eng, H.; Sweimeh, K.; Chamberlin, A.R. Microcystin Analogues Comprised Only of Adda and a Single Additional Amino Acid Retain Moderate Activity as PP1/PP2A Inhibitors. Bioorg. Med. Chem. Lett. 2003, 13, 2907–2911. [Google Scholar] [CrossRef]
- Namikoshi, M.; Rinehart, K.L.; Sakai, R.; Stotts, R.R.; Dahlem, A.M.; Beasley, V.R.; Carmichael, W.W.; Evans, W.R. Identification of 12 Hepatotoxins from a Homer Lake Bloom of the Cyanobacteria Microcystis Aeruginosa, Microcystis Viridis, and Microcystis Wesenbergii: Nine New Microcystins. J. Org. Chem. 1992, 57, 866–872. [Google Scholar] [CrossRef]
- Monks, N.R.; Liu, S.; Xu, Y.; Yu, H.; Bendelow, A.S.; Moscow, J.A. Potent Cytotoxicity of the Phosphatase Inhibitor Microcystin LR and Microcystin Analogues in OATP1B1- and OATP1B3-Expressing HeLa Cells. Mol. Cancer Ther. 2007, 6, 587–598. [Google Scholar] [CrossRef] [PubMed]
- Komatsu, M.; Furukawa, T.; Ikeda, R.; Takumi, S.; Nong, Q.; Aoyama, K.; Akiyama, S.I.; Keppler, D.; Takeuchi, T. Involvement of Mitogen-Activated Protein Kinase Signaling Pathways in Microcystin-LR-Induced Apoptosis after Its Selective Uptake Mediated by OATP1B1 and OATP1B3. Toxicol. Sci. 2007, 97, 407–416. [Google Scholar] [CrossRef] [PubMed]
- MacKintosh, R.W.; Dalby, K.N.; Campbell, D.G.; Cohen, P.T.W.; Cohen, P.; MacKintosh, C. The Cyanobacterial Toxin Microcystin Binds Covalently to Cysteine-273 on Protein Phosphatase 1. FEBS Lett. 1995, 371, 236–240. [Google Scholar] [CrossRef] [PubMed]
- Honkanen, R.E.; Zwiller, J.; Moore, R.E.; Daily, S.L.; Khatra, B.S.; Dukelow, M.; Boynton, A.L. Characterization of Microcystin-LR, a Potent Inhibitor of Type 1 and Type 2A Protein Phosphatases. J. Biol. Chem. 1990, 265, 19401–19404. [Google Scholar] [CrossRef]
- Fontanillo, M.; Köhn, M. Microcystins: Synthesis and Structure–Activity Relationship Studies toward PP1 and PP2A. Bioorg. Med. Chem. 2018, 26, 1118–1126. [Google Scholar] [CrossRef]
- Xing, Y.; Xu, Y.; Chen, Y.; Jeffrey, P.D.; Chao, Y.; Lin, Z.; Li, Z.; Strack, S.; Stock, J.B.; Shi, Y. Structure of Protein Phosphatase 2A Core Enzyme Bound to Tumor-Inducing Toxins. Cell 2006, 127, 341–353. [Google Scholar] [CrossRef]
- Eriksson, J.E.; Toivola, D.; Meriluoto, J.A.O.; Karaki, H.; Han, Y.G.; Hartshorne, D. Hepatocyte Deformation Induced by Cyanobacterial Toxins Reflects Inhibition of Protein Phosphatases. Biochem. Biophys. Res. Commun. 1990, 173, 1347–1353. [Google Scholar] [CrossRef]
- Wharton, R.E.; Cunningham, B.R.; Schaefer, A.M.; Guldberg, S.M.; Hamelin, E.I.; Johnson, R.C. Measurement of Microcystin and Nodularin Activity in Human Urine by Immunocapture-Protein Phosphatase 2a Assay. Toxins 2019, 11, 729. [Google Scholar] [CrossRef]
- Ikehara, T.; Imamura, S.; Oshiro, N.; Ikehara, S.; Shinjo, F.; Yasumoto, T. A Protein Phosphatase 2A (PP2A) Inhibition Assay Using a Recombinant Enzyme for Rapid Detection of Microcystins. Toxicon 2008, 51, 1368–1373. [Google Scholar] [CrossRef]
- Hilborn, E.D.; Carmichael, W.W.; Soares, R.M.; Yuan, M.; Servaites, J.C.; Barton, H.A.; Azevedo, S. Serologic Evaluation of Human Microcystin Exposure. Environ. Toxicol. Int. J. 2007, 22, 459–463. [Google Scholar] [CrossRef]
- Hilborn, E.D.; Carmichael, W.W.; Yuan, M.; Azevedo, S.M.F.O. A Simple Colorimetric Method to Detect Biological Evidence of Human Exposure to Microcystins. Toxicon 2005, 46, 218–221. [Google Scholar] [CrossRef] [PubMed]
- Yuan, M.; Carmichael, W.W.; Hilborn, E.D. Microcystin Analysis in Human Sera and Liver from Human Fatalities in Caruaru, Brazil 1996. Toxicon 2006, 48, 627–640. [Google Scholar] [CrossRef] [PubMed]
- Wharton, R.E.; Ojeda-Torres, G.; Cunningham, B.; Feyereisen, M.C.; Hill, K.L.; Abbott, N.L.; Seymour, C.; Hill, D.; Lang, J.; Hamelin, E.I.; et al. Quantification of Microcystin-LR in Human Urine by Immunocapture Liquid Chromatography Tandem Mass Spectrometry. Chem. Res. Toxicol. 2018, 31, 898–903. [Google Scholar] [CrossRef] [PubMed]
- Palagama, D.S.W.; Baliu-Rodriguez, D.; Lad, A.; Levison, B.S.; Kennedy, D.J.; Haller, S.T.; Westrick, J.; Hensley, K.; Isailovic, D. Development and Applications of Solid-Phase Extraction and Liquid Chromatography-Mass Spectrometry Methods for Quantification of Microcystins in Urine, Plasma, and Serum. J. Chromatogr. A 2018, 1573, 66–77. [Google Scholar] [CrossRef]
- Zhang, H.; Gonzales, G.B.; Beloglazova, N.V.; De Saeger, S.; Shen, J.; Zhang, S.; Yang, S.; Wang, Z. Development of a Validated Direct Injection-Liquid Chromatographic Tandem Mass Spectrometric Method under Negative Electrospray Ionization for Quantitation of Nine Microcystins and Nodularin-R in Lake Water. J. Chromatogr. A 2020, 1609, 460432. [Google Scholar] [CrossRef]
- Harada, K.-I.; Kondo, F.; Lawton, L. Laboratory Analysis of Cyanotoxins. In Toxic Cyanobacteria in Water; WHO: Geneva, Switzerland, 1999; pp. 369–405. [Google Scholar]
- U.S. Department of Health and Human Services. Bioanalytical Method Validation: Guidance for Industry; In U.S. Department of Health and Human Services, Food and Drug Administration, Center for Drug Evalution and Research (CDER), Center for Veterinary Medicine (CVM); Food and Drug Administration: Rockville, MD, USA, 2018; p. 25. ISBN 1984-8250.
- Chen, J.; Xie, P.; Li, L.; Xu, J.; Al, C.E.T. First Identification of the Hepatotoxic Microcystins in the Serum of a Chronically Exposed Human Population Together with Indication of Hepatocellular Damage. Toxicol. Sci. 2009, 108, 81–89. [Google Scholar] [CrossRef]
- Robinson, N.A.; Pace, J.G.; Matson, C.F.; Miura, G.A.; Lawrence, W.B. Tissue Distribution, Excretion, and Hepatic Biotransformation of Microcystin-LR in Mice; Army Medical Research Institute of Infectious Diseases: Frederick, MD, USA, 1990. [Google Scholar]
- Armbruster, D.A.; Pry, T. Limit of Blank, Limit of Detection and Limit of Quantitation. Clin. Biochem. Rev. 2008, 29 (Suppl. S1), S49–S52. [Google Scholar]
- NCCLS. Protocols for Determination of Limits of Detection and Limits of Quantitation; Approved Guideline; NCCLS: Wayne, PA, USA, 2008; Volume 24. [Google Scholar]
- Kondo, F.; Matsumoto, H.; Yamada, S.; Ishikawa, N.; Ito, E.; Nagata, S.; Ueno, Y.; Suzuki, M.; Harada, K.I. Detection and Identification of Metabolites of Microcystins Formed in Vivo in Mouse and Rat Livers. Chem. Res. Toxicol. 1996, 9, 1355–1359. [Google Scholar] [CrossRef]
- Greer, B.; Meneely, J.P.; Elliott, C.T. Uptake and Accumulation of Microcystin-LR Based on Exposure through Drinking Water: An Animal Model Assessing the Human Health Risk. Sci. Rep. 2018, 8, 4913. [Google Scholar] [CrossRef]
- Zhang, W.; Liang, G.; Wu, L.; Tuo, X.; Wang, W.; Chen, J.; Xie, P. Why Mammals More Susceptible to the Hepatotoxic Microcystins than Fish: Evidences from Plasma and Albumin Protein Binding through Equilibrium Dialysis. Ecotoxicology 2013, 22, 1012–1019. [Google Scholar] [CrossRef]
- Baliu-Rodriguez, D.; Kucheriavaia, D.; Palagama, D.S.W.; Lad, A.; O’Neill, G.M.; Birbeck, J.A.; Kennedy, D.J.; Haller, S.T.; Westrick, J.A.; Isailovic, D. Development and Application of Extraction Methods for LC-MS Quantification of Microcystins in Liver Tissue. Toxins 2020, 12, 263. [Google Scholar] [CrossRef] [PubMed]
- Miles, C.; Stirling, D. Toxin Mass List COM V16.0 (Microcystin and Nodularin Lists and Mass Calculators for Mass Spectrometry of Microcystins, Nodularins, Saxitoxins and Anatoxins). 2019. Available online: https://www.researchgate.net/publication/337258461_Toxin_mass_list_COM_v160_microcystin_and_nodularin_lists_and_mass_calculators_for_mass_spectrometry_of_microcystins_nodularins_saxitoxins_and_anatoxins/stats (accessed on 22 April 2022).
- Altaner, S.; Puddick, J.; Fessard, V.; Feurstein, D.; Zemskov, I.; Wittmann, V.; Dietrich, D.R. Simultaneous Detection of 14 Microcystin Congeners from Tissue Samples Using UPLC-ESI-MS/MS and Two Different Deuterated Synthetic Microcystins as Internal Standards. Toxins 2019, 11, 388. [Google Scholar] [CrossRef] [PubMed]
- Heussner, A.H.; Altaner, S.; Kamp, L.; Rubio, F.; Dietrich, D.R. Pitfalls in Microcystin Extraction and Recovery from Human Blood Serum. Chem. Biol. Interact. 2014, 223, 87–94. [Google Scholar] [CrossRef] [PubMed]
- Kondo, F.; Ikai, Y.; Oka, H.; Okumura, M.; Ishikawa, N.; Harada, K.I.; Matsuura, K.; Murata, H.; Suzuki, M. Formation, Characterization, and Toxicity of the Glutathione and Cysteine Conjugates of Toxic Heptapeptide Microcystins. Chem. Res. Toxicol. 1992, 5, 591–596. [Google Scholar] [CrossRef]

| MC-LR (ng/mL) | Average (ng/mL) | Percent Accuracy | Percent RSD | |
|---|---|---|---|---|
| Interday | 0.50 | 0.521 | 104 | 10.7 |
| 0.30 | 0.333 | 111 | 10.9 | |
| 0.20 | 0.228 | 114 | 7.18 | |
| 0.10 | 0.097 | 97 | 3.96 | |
| 0.05 | 0.0495 | 99 | 15.8 | |
| 0.04 | 0.0447 | 112 | 12.6 | |
| 0.03 | 0.032 | 107 | 22.0 | |
| Intraday | 0.20 | 0.203 | 102 | 18.6 |
| 0.05 | 0.0575 | 115 | 8.7 |
| Published Method | Analytically Validated? | Matrix | Limit of Detection | Lowest Reportable Limit | MC-LR Recovery |
|---|---|---|---|---|---|
| Measurement of Microcystin Activity in Human Plasma Using Immunocapture and Protein Phosphatase Inhibition Assay * | Yes | Human Plasma | 0.0183 ng/mL | 0.0300 ng/mL | 77.8% |
| Measurement of microcystin and nodularin activity in human urine by immunocapture-protein phosphatase 2A assay [35] | Yes | Human Urine | 0.0283 ng/mL | 0.0500 ng/mL | 77.6% |
| A simple colorimetric method to detect biological evidence of human exposure to microcystins [38] | No | Human Serum | 0.147 ng/mL | 0.500 ng/mL | N/A |
| Development and applications of solid-phase extraction and liquid chromatography-mass spectrometry methods for quantification of microcystins in urine, plasma, and serum [41] | No | Human Serum | Signal to noise of ~3 | Signal to noise of ~10 | 98.4% (avg.) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cunningham, B.R.; Wharton, R.E.; Lee, C.; Mojica, M.A.; Krajewski, L.C.; Gordon, S.C.; Schaefer, A.M.; Johnson, R.C.; Hamelin, E.I. Measurement of Microcystin Activity in Human Plasma Using Immunocapture and Protein Phosphatase Inhibition Assay. Toxins 2022, 14, 813. https://doi.org/10.3390/toxins14110813
Cunningham BR, Wharton RE, Lee C, Mojica MA, Krajewski LC, Gordon SC, Schaefer AM, Johnson RC, Hamelin EI. Measurement of Microcystin Activity in Human Plasma Using Immunocapture and Protein Phosphatase Inhibition Assay. Toxins. 2022; 14(11):813. https://doi.org/10.3390/toxins14110813
Chicago/Turabian StyleCunningham, Brady R., Rebekah E. Wharton, Christine Lee, Mike A. Mojica, Logan C. Krajewski, Shirley C. Gordon, Adam M. Schaefer, Rudolph C. Johnson, and Elizabeth I. Hamelin. 2022. "Measurement of Microcystin Activity in Human Plasma Using Immunocapture and Protein Phosphatase Inhibition Assay" Toxins 14, no. 11: 813. https://doi.org/10.3390/toxins14110813
APA StyleCunningham, B. R., Wharton, R. E., Lee, C., Mojica, M. A., Krajewski, L. C., Gordon, S. C., Schaefer, A. M., Johnson, R. C., & Hamelin, E. I. (2022). Measurement of Microcystin Activity in Human Plasma Using Immunocapture and Protein Phosphatase Inhibition Assay. Toxins, 14(11), 813. https://doi.org/10.3390/toxins14110813





