Quantitative Proteomic Analysis of Zearalenone-Induced Intestinal Damage in Weaned Piglets
Abstract
1. Introduction
2. Results
2.1. Serum ZEN, α-ZOL, and β-ZOL
2.2. Apparent Nutrient Digestibility
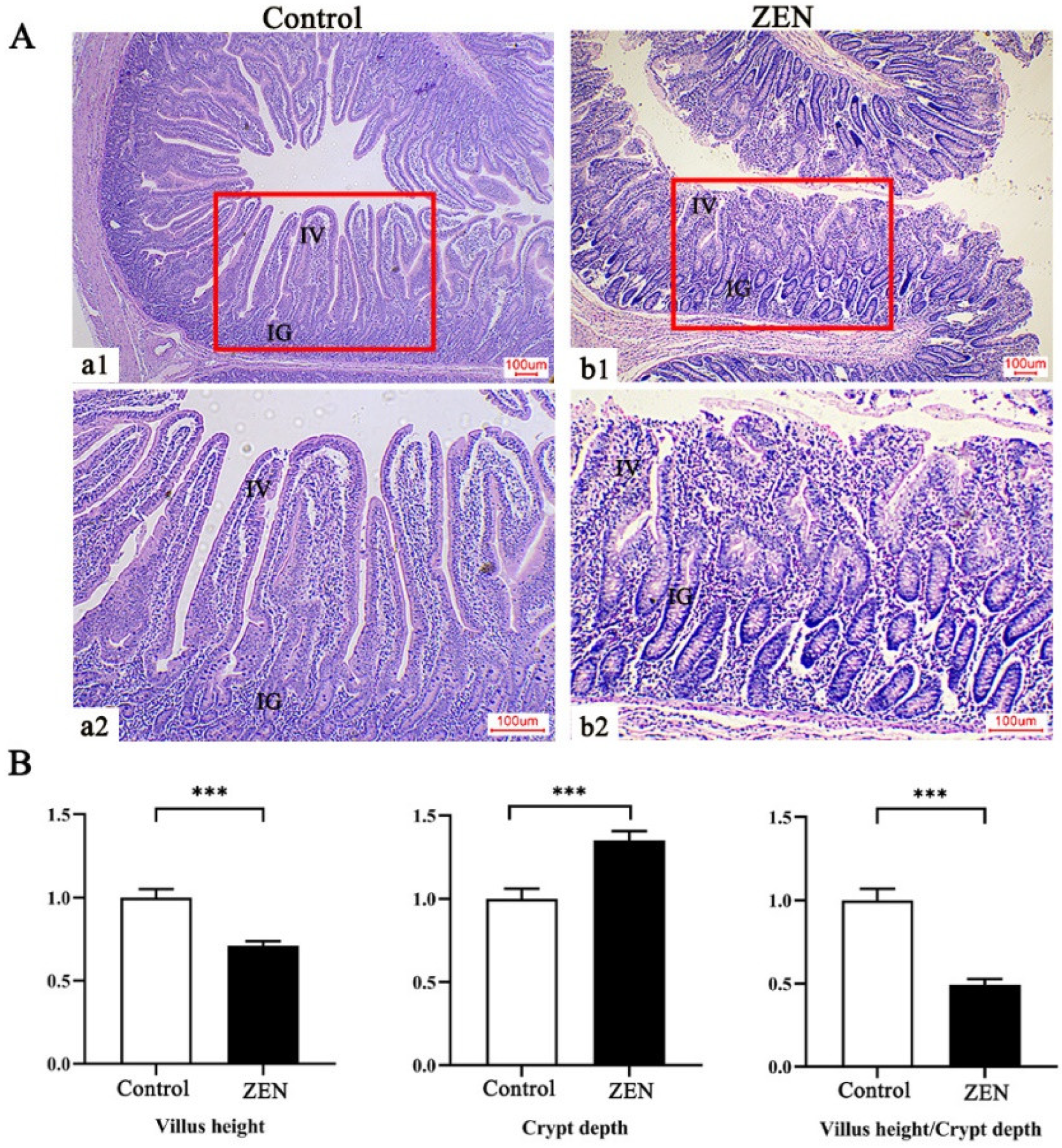
2.3. Histopathological Examination
2.4. Intestinal Permeability of Weaned Piglets
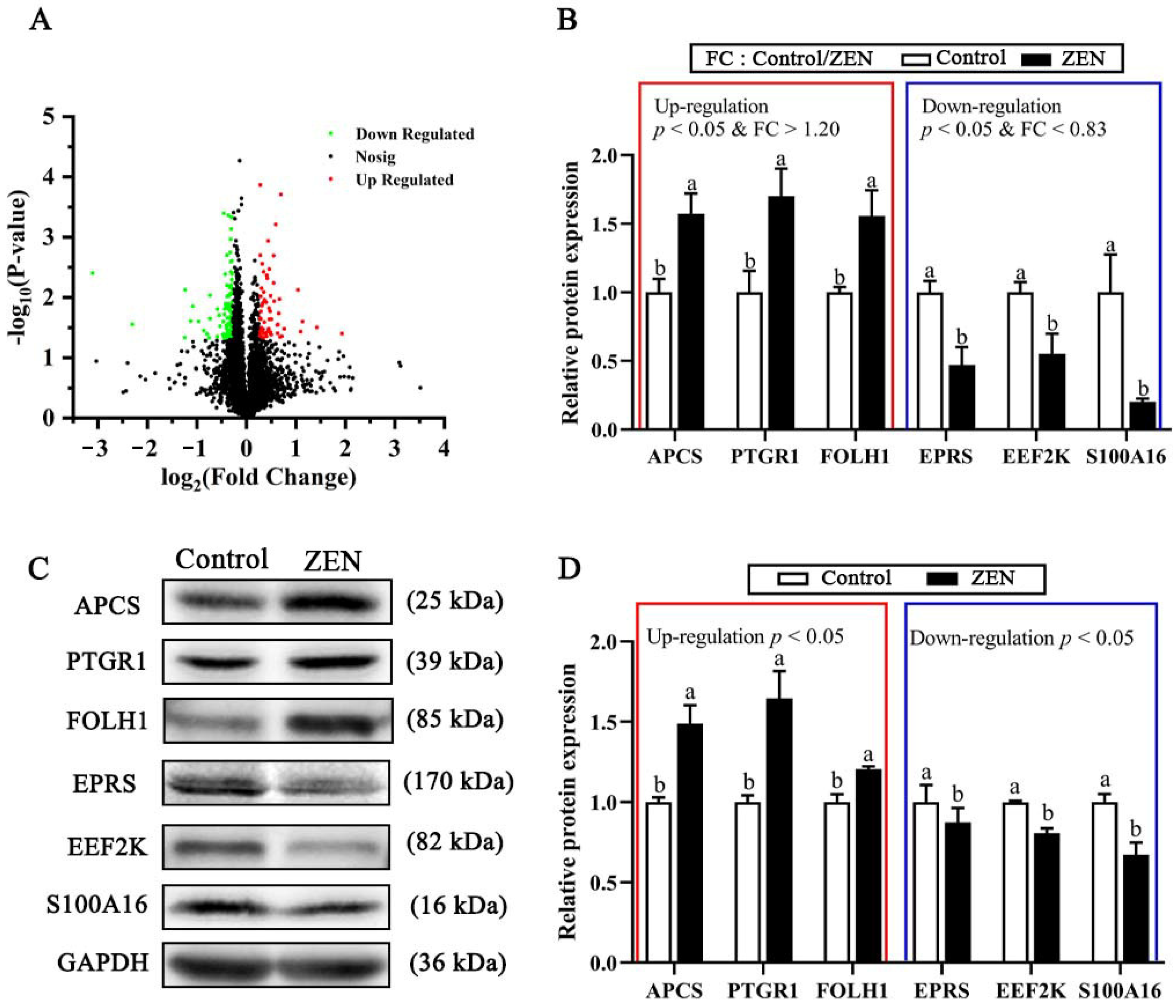
2.5. Quantitative Mapping of Jejunum Proteome in the Control and ZEN Treatment
2.6. Top Ten Up-Regulated or Down-Regulated DEPs
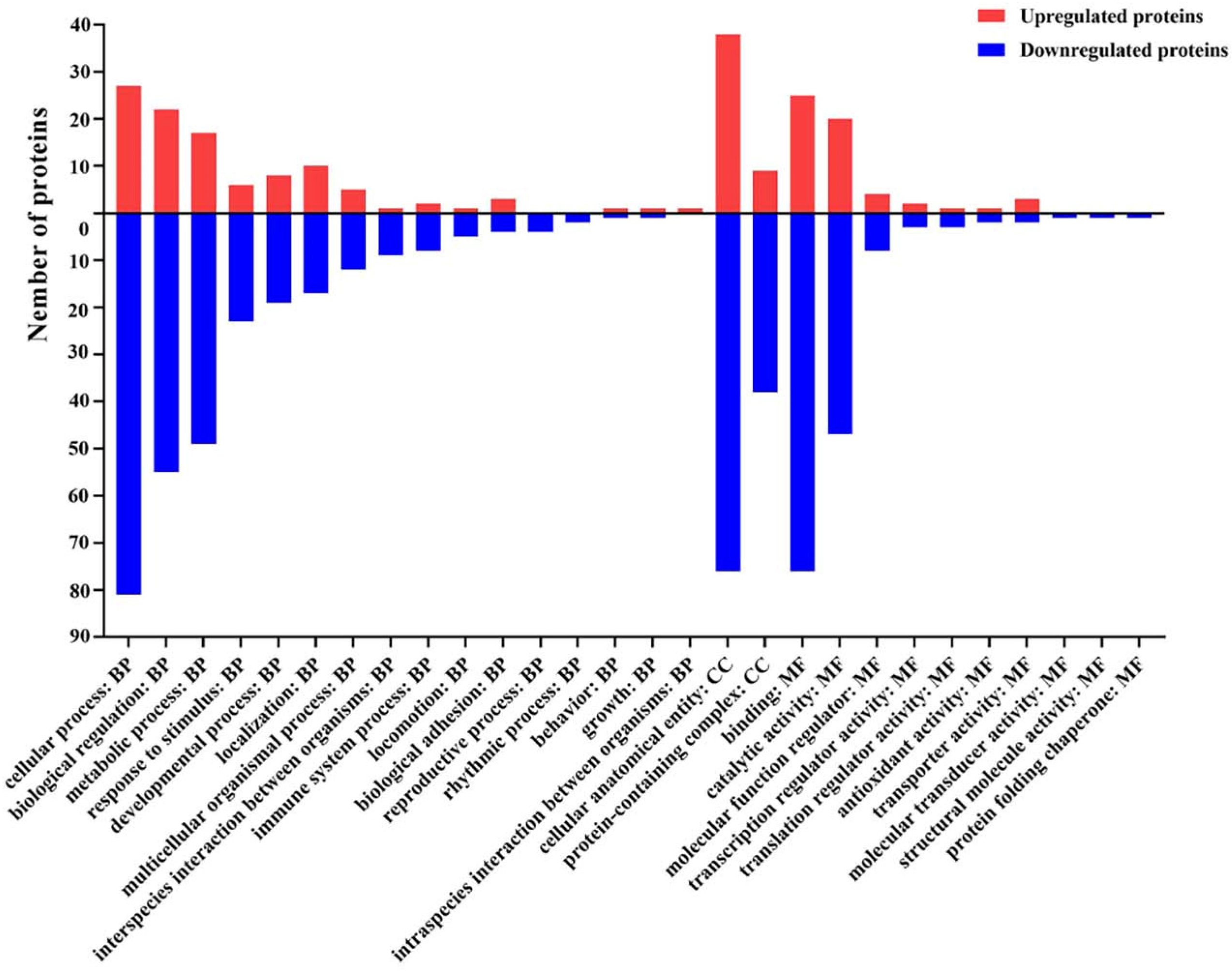
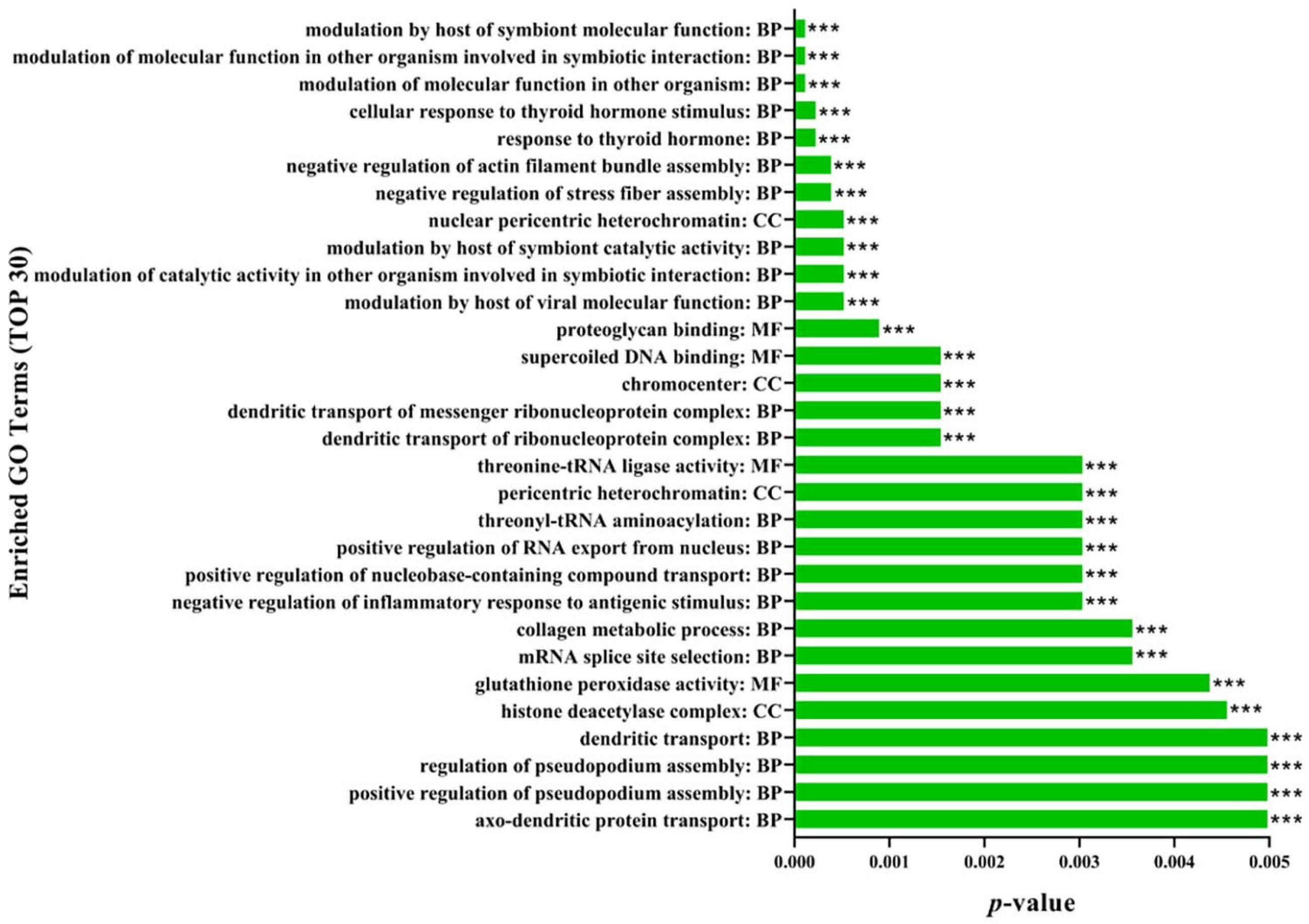
2.7. Gene Ontology (GO) Functional Annotation and Enrichment Analysis of DEPs
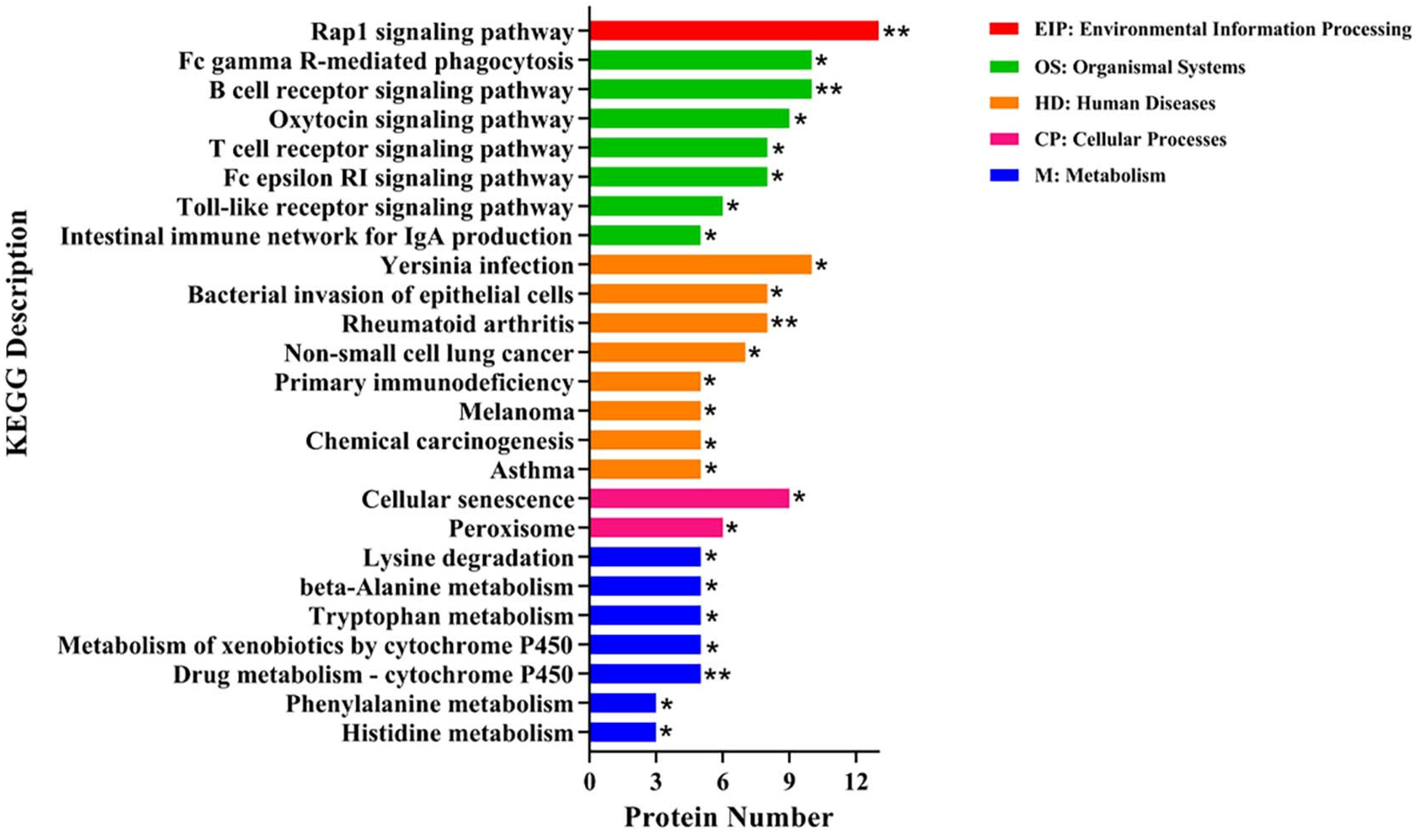
2.8. Kyoto Encyclopedia of Gene and Genomes (KEGG) Enrichment Analysis of DEPs
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Animals, Treatments, and Feeding Management
5.2. Sample Collection
5.3. Serum ZEN, α-ZOL, and β-ZOL
5.4. Apparent Nutrient Digestibility
5.5. Histopathological Examination of Jejunum
5.6. Intestinal Permeability of Weaned Piglets
5.7. Sample Preparation and TMT Labeling
5.8. High pH Reversed-Phase Liquid Chromatography (HPLC) Fractionation and Liquid Chromatography–Tandem Mass Spectrometry (LC−MS/MS) Analysis
5.9. Sequence Database Searching and Protein Identification
5.10. Bioinformatics Analysis
5.11. Western Blot
5.12. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gajecka, M.; Waskiewicz, A.; Zielonka, L.; Golinski, P.; Rykaczewska, A.; Lisieska-Zolnierczyk, S.; Gajecki, M.T. Mycotoxin levels in the digestive tissues of immature gilts exposed to zearalenone and deoxynivalenol. Toxicon 2018, 153, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Kowalska, K.; Habrowska-Gorczynska, D.E.; Piastowska-Ciesielska, A.W. Zearalenone as an endocrine disruptor in humans. Environ. Toxicol Pharm. 2016, 48, 141–149. [Google Scholar] [CrossRef] [PubMed]
- Rogowska, A.; Pomastowski, P.; Sagandykova, G.; Buszewski, B. Zearalenone and its metabolites: Effect on human health, metabolism and neutralisation methods. Toxicon 2019, 162, 46–56. [Google Scholar] [CrossRef] [PubMed]
- Przybylska-Gornowicz, B.; Tarasiuk, M.; Lewczuk, B.; Prusik, M.; Ziolkowska, N.; Zielonka, L.; Gajecki, M.; Gajecka, M. The effects of low doses of two Fusarium toxins, zearalenone and deoxynivalenol, on the pig jejunum. A light and electron microscopic study. Toxins 2015, 7, 4684–4705. [Google Scholar] [CrossRef]
- Cheng, Q.; Jiang, S.Z.; Huang, L.B.; Yang, W.R.; Yang, Z.B. Zearalenone regulates key factors of the Kelch-like erythroid cell-derived protein with CNC homology-associated protein 1-nuclear factor erythroid 2-related factor 2 signaling pathway in duodenum of post-weaning gilts. Anim. Biosci. 2021, 34, 1403–1414. [Google Scholar] [CrossRef]
- Chen, X.X.; Yang, C.W.; Huang, L.B.; Niu, Q.S.; Jiang, S.Z.; Chi, F. Zearalenone Altered the Serum Hormones, Morphologic and Apoptotic Measurements of Genital Organs in Post-weaning Gilts. Asian Australas. J. Anim. Sci. 2015, 28, 171–179. [Google Scholar] [CrossRef]
- Oswald, I.P. Role of intestinal epithelial cells in the innate immune defence of the pig intestine. Vet. Res. 2006, 37, 359–368. [Google Scholar] [CrossRef]
- Van Le Thanh, B.; Lessard, M.; Chorfi, Y.; Guay, F. The efficacy of anti-mycotoxin feed additives in preventing the adverse effects of wheat naturally contaminated with Fusarium mycotoxins on performance, intestinal barrier function and nutrient digestibility and retention in weanling pigs. Can. J. Anim. Sci. 2015, 95, 197–209. [Google Scholar] [CrossRef]
- Zhang, W.; Zhang, S.; Wang, J.; Shan, A.; Xu, L. Changes in intestinal barrier functions and gut microbiota in rats exposed to zearalenone. Ecotoxicol. Environ. Saf. 2020, 204, 111072. [Google Scholar] [CrossRef]
- Campbell, J.M.; Crenshaw, J.D.; Polo, J. The biological stress of early weaned piglets. J. Anim. Sci. Biotechnol. 2013, 4, 124–127. [Google Scholar] [CrossRef]
- Fink-Gremmels, J.; Malekinejad, H. Clinical effects and biochemical mechanisms associated with exposure to the mycoestrogen zearalenone. Anim. Feed. Sci. Technol. 2007, 137, 326–341. [Google Scholar] [CrossRef]
- Schoevers, E.J.; Santos, R.R.; Colenbrander, B.; Fink-Gremmels, J.; Roelen, B.A. Transgenerational toxicity of Zearalenone in pigs. Reprod. Toxicol. 2012, 34, 110–119. [Google Scholar] [CrossRef] [PubMed]
- Saenz, J.S.; Kurz, A.; Ruczizka, U.; Bunger, M.; Dippel, M.; Nagl, V.; Grenier, B.; Ladinig, A.; Seifert, J.; Selberherr, E. Metaproteomics Reveals Alteration of the Gut Microbiome in Weaned Piglets Due to the Ingestion of the Mycotoxins Deoxynivalenol and Zearalenone. Toxins 2021, 13, 583. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Xu, C.; Yang, Z.; Yang, W.; Huang, L.; Wang, S.; Liu, F.; Liu, M.; Wang, Y.; Jiang, S. Effects of Dietary Zearalenone Exposure on the Growth Performance, Small Intestine Disaccharidase, and Antioxidant Activities of Weaned Gilts. Animals 2020, 10, 2157. [Google Scholar] [CrossRef]
- Zhang, Q.; Huang, L.; Leng, B.; Li, Y.; Jiao, N.; Jiang, S.; Yang, W.; Yuan, X. Zearalenone Affect the Intestinal Villi Associated with the Distribution and the Expression of Ghrelin and Proliferating Cell Nuclear Antigen in Weaned Gilts. Toxins 2021, 13, 736. [Google Scholar] [CrossRef]
- Zhang, P.; Jing, C.; Liang, M.; Jiang, S.; Huang, L.; Jiao, N.; Li, Y.; Yang, W. Zearalenone Exposure Triggered Cecal Physical Barrier Injury through the TGF-beta1/Smads Signaling Pathway in Weaned Piglets. Toxins 2021, 13, 902. [Google Scholar] [CrossRef]
- Lahjouji, T.; Bertaccini, A.; Neves, M.; Puel, S.; Oswald, I.P.; Soler, L. Acute Exposure to Zearalenone Disturbs Intestinal Homeostasis by Modulating the Wnt/β-Catenin Signaling Pathway. Toxins 2020, 12, 113. [Google Scholar] [CrossRef]
- Manzoni, C.; Kia, D.A.; Vandrovcova, J.; Hardy, J.; Wood, N.W.; Lewis, P.A.; Ferrari, R. Genome, transcriptome and proteome: The rise of omics data and their integration in biomedical sciences. Briefings Bioinform. 2018, 19, 286–302. [Google Scholar] [CrossRef]
- Soler, L.; Stella, A.; Seva, J.; Pallares, F.J.; Lahjouji, T.; Burlet-Schiltz, O.; Oswald, I.P. Proteome changes induced by a short, non-cytotoxic exposure to the mycoestrogen zearalenone in the pig intestine. J. Proteom. 2020, 224, 103842. [Google Scholar] [CrossRef]
- Aslam, B.; Basit, M.; Nisar, M.A.; Khurshid, M.; Rasool, M.H. Proteomics: Technologies and Their Applications. J. Chromatogr. Sci. 2017, 55, 182–196. [Google Scholar] [CrossRef]
- Zhou, X.; Zhou, L.; Ge, X.; Guo, X.; Han, J.; Zhang, Y.; Yang, H. Quantitative Proteomic Analysis of Porcine Intestinal Epithelial Cells Infected with Porcine Deltacoronavirus Using iTRAQ-Coupled LC-MS/MS. J. Proteome Res. 2020, 19, 4470–4485. [Google Scholar] [CrossRef] [PubMed]
- Zinedine, A.; Soriano, J.M.; Molto, J.C.; Manes, J. Review on the toxicity, occurrence, metabolism, detoxification, regulations and intake of zearalenone: An oestrogenic mycotoxin. Food Chem. Toxicol. 2007, 45, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Dänicke, S.; Winkler, J. Invited review: Diagnosis of zearalenone (ZEN) exposure of farm animals and transfer of its residues into edible tissues (carry over). Food Chem. Toxicol. 2015, 84, 225–249. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zheng, Y.; Tao, H.; Liu, J.; Zhao, P.; Yang, F.; Lv, Z.; Wang, J. Effects of Bacillus subtilis ZJ-2019-1 on Zearalenone Toxicosis in Female Gilts. Toxins 2021, 13, 788. [Google Scholar] [CrossRef] [PubMed]
- Wan, B.; Huang, L.; Jing, C.; Li, Y.; Jiao, N.; Liang, M.; Jiang, S.; Yang, W. Zearalenone promotes follicle development through activating the SIRT1/PGC-1alpha signaling pathway in the ovaries of weaned gilts. J. Anim. Sci. 2022, 100, skac058. [Google Scholar] [CrossRef]
- Fleck, S.C.; Churchwell, M.I.; Doerge, D.R. Metabolism and pharmacokinetics of zearalenone following oral and intravenous administration in juvenile female pigs. Food Chem. Toxicol. 2017, 106, 193–201. [Google Scholar] [CrossRef]
- Bouhet, S.; Oswald, I.P. The effects of mycotoxins, fungal food contaminants, on the intestinal epithelial cell-derived innate immune response. Vet. Immunol. Immunopathol. 2005, 108, 199–209. [Google Scholar] [CrossRef]
- Jiang, S.Z.; Yang, Z.B.; Yang, W.R.; Yao, B.Q.; Zhao, H.; Liu, F.X.; Chen, C.C.; Chi, F. Effects of Feeding Purified Zearalenone Contaminated Diets with or without Clay Enterosorbent on Growth, Nutrient Availability, and Genital Organs in Post-weaning Female Pigs. Asian Australas. J. Anim. Sci. 2010, 23, 74–81. [Google Scholar] [CrossRef]
- Wang, J.P.; Chi, F.; Kim, I.H. Effects of montmorillonite clay on growth performance, nutrient digestibility, vulva size, faecal microflora, and oxidative stress in weaning gilts challenged with zearalenone. Anim. Feed. Sci. Technol. 2012, 178, 158–166. [Google Scholar] [CrossRef]
- Pluske, J.R.; Thompson, M.J.; Atwood, C.S.; Bird, P.H.; Williams, I.H.; Hartmann, P.E. Maintenance of villus height and crypt depth, and enhancement of disaccharide digestion and monosaccharide absorption, in piglets fed on cows’ whole milk after weaning. Br. J. Nutr. 1996, 76, 409–422. [Google Scholar] [CrossRef]
- Xu, Z.R.; Hu, C.H.; Xia, M.S.; Zhan, X.A.; Wang, M.Q. Effects of dietary fructooligosaccharide on digestive enzyme activities, intestinal microflora and morphology of male broilers. Poult. Sci. 2003, 82, 1030–1036. [Google Scholar] [CrossRef]
- Liu, M.; Gao, R.; Meng, Q.; Zhang, Y.; Bi, C.; Shan, A. Toxic effects of maternal zearalenone exposure on intestinal oxidative stress, barrier function, immunological and morphological changes in rats. PLoS ONE 2014, 9, e106412. [Google Scholar] [CrossRef] [PubMed]
- Lundin, J.I.; Checkoway, H. Endotoxin and cancer. Environ. Health Perspect. 2009, 117, 1344–1350. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Yu, B.; Chen, D.; Huang, Z.; Mao, X.; Zheng, P.; Yu, J.; Luo, J.; He, J. Chlorogenic acid improves intestinal barrier functions by suppressing mucosa inflammation and improving antioxidant capacity in weaned pigs. J. Nutr. Biochem. 2018, 59, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.L.; Li, F.C.; Yang, W.R.; Jiang, S.Z.; Li, Y. Supplementation with Exogenous Catalase from Penicillium notatum in the Diet Ameliorates Lipopolysaccharide-Induced Intestinal Oxidative Damage through Affecting Intestinal Antioxidant Capacity and Microbiota in Weaned Pigs. Microbiol. Spectr. 2021, 9, e00654-21. [Google Scholar] [CrossRef]
- Chen, J.L.; Zheng, P.; Zhang, C.; Yu, B.; He, J.; Yu, J.; Luo, J.Q.; Mao, X.B.; Huang, Z.Q.; Chen, D.W. Benzoic acid beneficially affects growth performance of weaned pigs which was associated with changes in gut bacterial populations, morphology indices and growth factor gene expression. J. Anim. Physiol. Anim. Nutr. 2017, 101, 1137–1146. [Google Scholar] [CrossRef]
- Geng, H.; Tan, X.; Zhao, M.; Ma, Y.; Li, Y. Proteomic analysis of zearalenone toxicity on mouse thymic epithelial cells. J. Appl. Toxicol. 2022, 42, 660–670. [Google Scholar] [CrossRef]
- Gajecka, M.; Tarasiuk, M.; Zielonka, L.; Dabrowski, M.; Nicpon, J.; Baranowski, M.; Gajecki, M.T. Changes in the metabolic profile and body weight of pre-pubertal gilts during prolonged monotonic exposure to low doses of zearalenone and deoxynivalenol. Toxicon 2017, 125, 32–43. [Google Scholar] [CrossRef]
- Wang, X.; Yu, H.; Shan, A.; Jin, Y.; Fang, H.; Zhao, Y.; Shen, J.; Zhou, C.; Zhou, Y.; Fu, Y.; et al. Toxic effects of Zearalenone on intestinal microflora and intestinal mucosal immunity in mice. Food Agric. Immunol. 2018, 29, 1002–1011. [Google Scholar] [CrossRef]
- Lorenowicz, M.J.; van Gils, J.; de Boer, M.; Hordijk, P.L.; Fernandez-Borja, M. Epac1-Rap1 signaling regulates monocyte adhesion and chemotaxis. J. Leukoc. Biol. 2006, 80, 1542–1552. [Google Scholar] [CrossRef]
- Cai, Y.; Sukhova, G.K.; Wong, H.K.; Xu, A.; Tergaonkar, V.; Vanhoutte, P.M.; Tang, E.H. Rap1 induces cytokine production in pro-inflammatory macrophages through NFkappaB signaling and is highly expressed in human atherosclerotic lesions. Cell Cycle 2015, 14, 3580–3592. [Google Scholar] [CrossRef] [PubMed]
- Seda, V.; Mraz, M. B-cell receptor signalling and its crosstalk with other pathways in normal and malignant cells. Eur. J. Haematol. 2015, 94, 193–205. [Google Scholar] [CrossRef]
- Casola, S.; Otipoby, K.L.; Alimzhanov, M.; Humme, S.; Uyttersprot, N.; Kutok, J.L.; Carroll, M.C.; Rajewsky, K. B cell receptor signal strength determines B cell fate. Nat. Immunol. 2004, 5, 317–327. [Google Scholar] [CrossRef] [PubMed]
- Khan, W.N. B cell receptor and BAFF receptor signaling regulation of B cell homeostasis. J. Immunol. 2009, 183, 3561–3567. [Google Scholar] [CrossRef] [PubMed]
- Tanigaki, K.; Chambliss, K.L.; Yuhanna, I.S.; Sacharidou, A.; Ahmed, M.; Atochin, D.N.; Huang, P.L.; Shaul, P.W.; Mineo, C. Endothelial Fcγ Receptor IIB Activation Blunts Insulin Delivery to Skeletal Muscle to Cause Insulin Resistance in Mice. Diabetes 2016, 65, 1996–2005. [Google Scholar] [CrossRef] [PubMed]
- Baerenwaldt, A.; Nimmerjahn, F. Immune regulation: FcgammaRIIB—regulating the balance between protective and autoreactive immune responses. Immunol. Cell Biol. 2008, 86, 482–484. [Google Scholar] [CrossRef] [PubMed]
- Aricescu, A.R.; Jones, E.Y. Immunoglobulin superfamily cell adhesion molecules: Zippers and signals. Curr. Opin. Cell Biol. 2007, 19, 543–550. [Google Scholar] [CrossRef]
- Bucciarelli, L.G.; Wendt, T.; Rong, L.; Lalla, E.; Hofmann, M.A.; Goova, M.T.; Taguchi, A.; Yan, S.F.; Yan, S.D.; Stern, D.M.; et al. RAGE is a multiligand receptor of the immunoglobulin superfamily: Implications for homeostasis and chronic disease. Cell Mol. Life Sci. 2002, 59, 1117–1128. [Google Scholar] [CrossRef]
- Stavnezer, J.; Schrader, C.E. IgH chain class switch recombination: Mechanism and regulation. J. Immunol. 2014, 193, 5370–5378. [Google Scholar] [CrossRef]
- Jiang, H.; Liu, J.; Wu, T.; Liu, M.; Sun, Q.; Wang, J.; Zhu, R.; Qu, G.; Li, S.; Liu, H.; et al. Proteomics analysis of important molecules in serum from meningitic piglets caused by Streptococcus suis serotype 2. J. Infect. Dev. Ctries. 2020, 14, 502–510. [Google Scholar] [CrossRef]
- Liu, Y.; Liu, H.; Cao, H.; Song, B.; Zhang, W.; Zhang, W. PBK/TOPK mediates promyelocyte proliferation via Nrf2-regulated cell cycle progression and apoptosis. Oncol. Rep. 2015, 34, 3288–3296. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.H.; Jeong, Y.J.; Won, J.Y.; Sim, H.I.; Park, Y.; Jin, H.S. PBK/TOPK Is a Favorable Prognostic Biomarker Correlated with Antitumor Immunity in Colon Cancers. Biomedicines 2022, 10, 299. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Q.; Yao, S.; Dong, Y.; Liu, D.; Wang, H.; Jiang, P.; Dai, C.; Lv, H.; Cao, C.; Zhou, Z.; et al. Down-regulation of PBK inhibits proliferation of human endometrial stromal cells in thin endometrium. Reprod. Biol. Endocrinol. 2022, 20, 25. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Fu, D.; Shao, H.; Kong, X.; Wang, W.; Yang, X.; Nyachoti, C.M.; Yin, Y. Impacts of birth weight on plasma, liver and skeletal muscle neutral amino acid profiles and intestinal amino acid transporters in suckling Huanjiang mini-piglets. PLoS ONE 2012, 7, e50921. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.J.; Song, Q.L.; Xie, C.Y.; Chu, L.C.; Thacker, P.A.; Htoo, J.K.; Qiao, S.Y. Estimation of the ideal standardized ileal digestible tryptophan to lysine ratio for growing pigs fed low crude protein diets supplemented with crystalline amino acids. Livest. Sci. 2012, 149, 260–266. [Google Scholar] [CrossRef]
- Hasan, M.S.; Crenshaw, M.A.; Liao, S.F. Dietary lysine affects amino acid metabolism and growth performance, which may not involve the GH/IGF-1 axis, in young growing pigs1. J. Anim. Sci. 2020, 98, skaa004. [Google Scholar] [CrossRef]
- Benevenga, N.J.; Blemings, K.P. Unique aspects of lysine nutrition and metabolism. J. Nutr. 2007, 137, 1610S–1615S. [Google Scholar] [CrossRef]
- Han, H.; Yin, J.; Wang, B.; Huang, X.; Yao, J.; Zheng, J.; Fan, W.; Li, T.; Yin, Y. Effects of dietary lysine restriction on inflammatory responses in piglets. Sci. Rep. 2018, 8, 2451. [Google Scholar] [CrossRef]
- Le Floc’h, N.; Seve, B. Biological roles of tryptophan and its metabolism: Potential implications for pig feeding. Livest. Sci. 2007, 112, 23–32. [Google Scholar] [CrossRef]
- Bai, M.; Liu, H.; Xu, K.; Oso, A.O.; Wu, X.; Liu, G.; Tossou, M.C.; Al-Dhabi, N.A.; Duraipandiyan, V.; Xi, Q.; et al. A review of the immunomodulatory role of dietary tryptophan in livestock and poultry. Amino Acids 2017, 49, 67–74. [Google Scholar] [CrossRef]
- Jo, H.; Kong, C.; Song, M.; Kim, B.G. Effects of dietary deoxynivalenol and zearalenone on apparent ileal digestibility of amino acids in growing pigs. Anim. Feed. Sci. Technol. 2016, 219, 77–82. [Google Scholar] [CrossRef]
- Song, C.W.; Lee, J.; Ko, Y.S.; Lee, S.Y. Metabolic engineering of Escherichia coli for the production of 3-aminopropionic acid. Metab. Engl. 2015, 30, 121–129. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Lu, X.; Chen, K.; Yang, J.; Zhang, A.; Wang, X.; Ouyang, P. β-alanine production using whole-cell biocatalysts in recombinant Escherichia coli. Mol. Catal. 2018, 449, 93–98. [Google Scholar] [CrossRef]
- Könst, P.M.; Franssen, M.C.R.; Scott, E.L.; Sanders, J.P.M. A study on the applicability of l-aspartate α-decarboxylase in the biobased production of nitrogen containing chemicals. Green Chem. 2009, 11, 1646–1652. [Google Scholar] [CrossRef]
- Castellvi, A.; Pequerul, R.; Barracco, V.; Juanhuix, J.; Pares, X.; Farres, J. Structural and biochemical evidence that ATP inhibits the cancer biomarker human aldehyde dehydrogenase 1A3. Commun. Biol. 2022, 5, 354. [Google Scholar] [CrossRef]
- Chaudhry, K.K.; Samak, G.; Shukla, P.K.; Mir, H.; Gangwar, R.; Manda, B.; Isse, T.; Kawamoto, T.; Salaspuro, M.; Kaihovaara, P.; et al. ALDH2 Deficiency Promotes Ethanol-Induced Gut Barrier Dysfunction and Fatty Liver in Mice. Alcohol. Clin. Exp. Res. 2015, 39, 1465–1475. [Google Scholar] [CrossRef] [PubMed]
- Zhou, M.; Yang, L.J.; Yang, W.R.; Huang, L.B.; Zhou, X.M.; Jiang, S.Z.; Yang, Z.B. Effects of zearalenone on the localization and expression of the growth hormone receptor gene in the uteri of post-weaning piglets. Asian Australas. J. Anim. Sci. 2018, 31, 32–39. [Google Scholar] [CrossRef] [PubMed]
- AOAC. Official Methods of Analysis, 17th ed.; AOAC: Washington, DC, USA, 2000. [Google Scholar]
- Chen, J.; Li, F.; Yang, W.; Jiang, S.; Li, Y. Comparison of Gut Microbiota and Metabolic Status of Sows With Different Litter Sizes During Pregnancy. Front. Vet. Sci. 2021, 8, 793174. [Google Scholar] [CrossRef]
- Gao, M.; Jiang, Y.; Xiao, X.; Peng, Y.; Xiao, X.; Yang, M. Protective effect of pioglitazone on sepsis-induced intestinal injury in a rodent model. J. Surg. Res. 2015, 195, 550–558. [Google Scholar] [CrossRef]
| Items | Control | ZEN | p-Value |
|---|---|---|---|
| ZEN, ng/mL | 0.00 ± 0.00 | 0.20 ± 0.01 | <0.001 |
| α-Zearalenol, ng/mL | 0.00 ± 0.00 | 1.18 ± 0.04 | <0.001 |
| β-Zearalenol, ng/mL | 0.00 ± 0.00 | 1.17 ± 0.05 | <0.001 |
| Items | Control | ZEN | p-Value |
|---|---|---|---|
| Dry matter, % | 89.16 ± 0.57 | 87.65 ± 1.62 | 0.398 |
| Organics matter, % | 89.55 ± 0.59 | 86.54 ± 1.98 | 0.167 |
| Ether extract, % | 88.85 ± 1.39 | 84.24 ± 0.74 | 0.011 |
| Crude protein, % | 86.04 ± 0.72 | 83.10 ± 0.83 | 0.018 |
| Items | Control | ZEN | p-Value |
|---|---|---|---|
| Endotoxin, U/mL | 0.13 ± 0.00 | 0.67 ± 0.02 | <0.001 |
| Diamine oxidase, U/mL | 20.28 ± 0.59 | 24.84 ± 0.72 | <0.001 |
| D-lactate, μg/mL | 4.98 ± 0.06 | 5.65 ± 0.09 | <0.001 |
| Accession | Protein Name | FC | p-Value | Main Function |
|---|---|---|---|---|
| F1SCC9 | SERPIN domain-containing protein (LOC106504545) | 3.8176 | 0.0398 | serine-type endopeptidase inhibitor activity |
| A0A480Q1D2 | Beta-parvin isoform X3 | 2.6862 | 0.0315 | actin binding |
| A0A4 X 1SK13 | Ig-like domain-containing protein | 2.1993 | 0.0250 | N/A |
| A0A480THA1 | Glutathione S-transferase kappa 1 isoform a (Fragment) | 2.1353 | 0.0368 | glutathione peroxidase activity |
| A0A4X1UQN8 | Uncharacterized protein (FRA10AC1) | 2.0615 | 0.0075 | N/A |
| A0A287B7R3 | 15-oxoprostaglandin 13-reductase (PTGR1) | 1.7014 | 0.0330 | prostaglandin reductase activity |
| A0A480F5A7 | Threonyl-tRNA synthetase | 1.6397 | 0.0441 | threonine-tRNA ligase activity |
| A0A4X1UWY7 | Uncharacterized protein | 1.6200 | 0.0002 | serine-type endopeptidase inhibitor activity |
| A0A481C6Y8 | Nuclear autoantigenic sperm protein | 1.6020 | 0.0452 | N/A |
| A0A4X1W295 | Apolipoprotein C-II (APOC2) | 1.5911 | 0.0107 | enzyme activator activity |
| Accession | Protein Name | FC | p-Value | Main Function |
|---|---|---|---|---|
| A0A480L9T0 | Glutathione S-transferase kappa 1 isoform a (Fragment) | 0.1161 | 0.0040 | glutathione peroxidase activity |
| A0A4X1W0M9 | EF-hand domain-containing protein (S100A16) | 0.2025 | 0.0279 | calcium ion binding |
| A0A4X1SNR4 | Ig-like domain-containing protein | 0.4225 | 0.0460 | N/A |
| P12068 | Lysozyme C-2 | 0.4242 | 0.0075 | immunization and immune enhancement |
| A0A480VWY5 | Enoyl-CoA hydratase | 0.4587 | 0.0245 | enoyl-CoA hydratase activity |
| A0A480T775 | Bifunctional glutamate/proline-tRNA ligase | 0.4717 | 0.0140 | aminoacyl-tRNA synthetase |
| A0A480XP77 | Translation initiation factor eIF-2B subunit delta isoform 2 | 0.5118 | 0.0249 | translation initiation factor activity |
| A0A0B8RZL6 | Eukaryotic elongation factor 2 kinase (EEF2K) | 0.5509 | 0.0355 | calmodulin binding |
| A0A4X1TGG9 | Threonyl-tRNA synthetase (TARS1) | 0.5778 | 0.0404 | threonine-tRNA ligase activity |
| A0A4X1ULA3 | HIT domain-containing protein | 0.5920 | 0.0467 | catalytic activity |
| Items | Value |
|---|---|
| Proteome Discoverer version: | 2.4 |
| Protein Database | uniprot-taxonomy-9823.unique.fasta |
| Cys alkylation | Iodoacetamide |
| Dynamic Modification | Oxidation (M), Acetyl (Protein N-Terminus), Met-loss (Protein N-Terminus), Met-loss+ Acetyl (Protein N-Terminus) |
| Static Modification | Carbamidomethyl (C), TMT 6plex (K), TMT 6plex (N-Terminus) |
| Enzyme Name | Trypsin (Full) |
| Max. Missed Cleavage Sites | 2 |
| Precursor Mass Tolerance | 20 ppm |
| Fragment Mass Tolerance | 0.02 Da |
| Validation based on | q-value |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ma, L.; Jiang, Y.; Lu, F.; Wang, S.; Liu, M.; Liu, F.; Huang, L.; Li, Y.; Jiao, N.; Jiang, S.; et al. Quantitative Proteomic Analysis of Zearalenone-Induced Intestinal Damage in Weaned Piglets. Toxins 2022, 14, 702. https://doi.org/10.3390/toxins14100702
Ma L, Jiang Y, Lu F, Wang S, Liu M, Liu F, Huang L, Li Y, Jiao N, Jiang S, et al. Quantitative Proteomic Analysis of Zearalenone-Induced Intestinal Damage in Weaned Piglets. Toxins. 2022; 14(10):702. https://doi.org/10.3390/toxins14100702
Chicago/Turabian StyleMa, Lulu, Yanping Jiang, Fuguang Lu, Shujing Wang, Mei Liu, Faxiao Liu, Libo Huang, Yang Li, Ning Jiao, Shuzhen Jiang, and et al. 2022. "Quantitative Proteomic Analysis of Zearalenone-Induced Intestinal Damage in Weaned Piglets" Toxins 14, no. 10: 702. https://doi.org/10.3390/toxins14100702
APA StyleMa, L., Jiang, Y., Lu, F., Wang, S., Liu, M., Liu, F., Huang, L., Li, Y., Jiao, N., Jiang, S., Yuan, X., & Yang, W. (2022). Quantitative Proteomic Analysis of Zearalenone-Induced Intestinal Damage in Weaned Piglets. Toxins, 14(10), 702. https://doi.org/10.3390/toxins14100702





