BoNT/A in the Urinary Bladder—More to the Story than Silencing of Cholinergic Nerves
Abstract
1. Botulinum Neurotoxins
2. Effects of BoNT/A in Bladder Disorders
3. Bladder Physiology
4. Use of BoNT/A to Treat Bladder Disorders
5. Effect of BoNT/A on Sensory Nerves—Direct Action
6. Effect of BoNT/A on Sensory Nerves—Changes in Neuropeptide Release
7. Effect of BoNT/A on Sensory Neurotransmission—Changes to Urothelial Function
8. SV2 and SNAP-25 Expression in the Urinary Bladder
| Reference | Species | Where Was SNAP-25 Detected? | Methods | Findings |
|---|---|---|---|---|
| [41] | Rat and human | Bladder mucosa (urothelium and lamina propria) and primary urothelial cells. | Detected SNAP-25 and SNAP-23 in rat mucosa tissue by performing gel electrophoresis of RNA. Protein expression in human and rat mucosa and primary cells shown by immunoblot and immunocytochemistry. | Human and rat urothelium expresses SNAP-25 and SNAP-23. BoNT/A incubation in rat urothelial cells led to significantly decreased SNAP-25 protein levels, suggesting cleavage. |
| [101] | Human | Nerve fibers in suburothelium and detrusor, not in urothelial or muscle cells. | Human bladders from organ donors were used for immunofluorescence staining of SNAP-25. | Dense SNAP-25 immunoreactivity in the suburothelium and detrusor layer, no labelling within urothelium or muscle cells. |
| [103] | Human | Urothelial cells and suburothelial tissues. | Human bladders of OAB patients received Lipotoxin (liposome encapsulated BoNT/A). IHC performed at baseline and 3 months post treatment. | SNAP-25 was expressed in the urothelium and suburothelial nerve fibers, expression decreased post-treatment in patients responding to treatment. |
| [110] | Human | Bladder mucosa (including the urothelium, lamina propria and a few discontinuous muscularis mucosa). | Human bladders of OAB patients received Lipotoxin and BoNT/A injection. IHC and immunoblotting performed at baseline and 3 months post-treatment. | SNAP-25 expressed in the bladder mucosa, expression was significantly reduced in patients who received BoNT/A injection, suggesting cleavage, but not in patients who received Lipotoxin. |
| [111] | Human | Intradetrusor nerve fibres. | Human bladders of NDO patients received BoNT/A injection and samples were taken for IHC. However, they did not mention the composition of the samples (urothelium, lamina propria or detrusor). | SNAP-25 expression was shown by IF in the neuronal fibers within the detrusor smooth muscle in untreated and treated patients. |
| [112] | Rat | Urothelium, suburothelium and muscle (images not clear as to whether expression is in the muscle cells or nerve fibers). | Treated rat bladders with BoNT/A using different protocols, conducted IHC for SNAP-25 on samples taken post-treatment. | SNAP-25 expression was shown by IHC in the bladder; however, it is unclear which cell types specifically. BoNT/A injection significantly reduced SNAP-25 expression, suggesting cleavage. |
| [102] | Rat | Suburothelial nerve fibers. | Treated rat bladders with Lipotoxin or BoNT/A instillation, conducted IF and western blotting for SNAP-25. | Liposome only and BoNT/A instilled bladders showed staining for SNAP-25 in the subuthelial nerve fibers, however Lipotoxin treated bladders showed significantly reduced SNAP-25 staining, suggesting cleavage. |
| [113] | Rat | Bladder (does not specify where in the bladder expression was found). | Treated rat bladders with BoNT/A after inducing CYP model of interstitial cystitis. Conducted IHC or immunoblotting for SNAP-25. | SNAP-25 expression was significantly reduced after BoNT treatment, suggesting cleavage. |
| [99] | Rat | No expression in the urothelium. | Investigated SNARE complexes in urothelium, conducted IF, electron microscopy and western blotting. | Mouse urothelium expressed SNAP-23, SNAP-25 was not found in the urothelium, through IF or Western blot, however was present in choroid plexus (brain stem) tissue that was used as control. |
| [98] | Mouse | No expression in the urothelium. | Investigated SNARE complexes in the urothelium and bladder, conducted immunoblotting for t-SNAREs and v-SNAREs. | Mouse urothelium expresses SNAP-23 but not SNAP-25. |
| [114] | Rat | Intradetrusor fibres and cultured DRG. | Injected rat bladders with BoNT/A, performed IHC Cultured DRG cells were treated with BoNT/A and performed ICC. | SNAP-25 expression found in intradetrusor fibers and cultured DRGs. |
| Reference | Species | Where Was Cleaved SNAP-25 Detected? | Methods | Findings |
|---|---|---|---|---|
| [104] | Guinea pig | Suburothelial nerve fibers only. | Guinea pig bladders were treated with BoNT/A, through intravesical instillation and intramural injection, conducted IHC on bladders for cleaved SNAP-25. | Cleaved SNAP-25 immunoreactive fibers in the mucosa and muscular layer. BoNT/A instillation did not cleave SNAP-25. |
| [111] | Human | Intradetrusor nerve fibers. | Patients with neurogenic detrusor overactivity (NDO) received intradetrusor BoNT/A. Western blot and immunofluorescence for cleaved SNAP-25 expression. | Detected cleaved SNAP-25 in bladder samples using Western blotting. |
| [115] | Mouse | Suburothelial nerve fibers. | Injected 0.5U of Botox or Dysport into dome of mouse bladders, three days later conducted IHC of cleaved SNAP-25. | Injection of both forms of BoNT/A led to cleavage of SNAP-25 in nerve fibers of the lamina propria. However, images show the whole bladder, and zoom in only on the lamina propria. Appears to be some staining in the urothelium of the Botox-treated bladder. |
| [110] | Human | Bladder mucosa (including the urothelium, lamina propria and a few discontinuous muscularis mucosa). | Human bladders of OAB patients received Lipotoxin and BoNT/A injection. IHC and immunoblotting performed at baseline and 3 months post-treatment. | Cleaved SNAP-25 was found in bladder mucosa of patients who received BoNT/A injection, not Lipotoxin. |
| [114] | Rat | Intradetrusor fibres and cultured DRGs. | Injected rat bladders with BoNT/A, performed IHC Cultured DRG cells were treated with BoNT/A and performed ICC. | Cleaved SNAP-25 was found in intradetrusor fibers and cultured DRGs. |
| Reference | Species | Where Was SV2 Detected? | Methods | Findings |
|---|---|---|---|---|
| [117] | Human | Dorsal root ganglion neurons and nerve fibers within the bladder. | Collected bladder tissue from patients with IDO, PBS and controls, cultured human DRG neurons, Conducted IHC on bladder tissue and calcium imaging on DRG neurons. | SV2 was expressed in DRG neurons, immunoreactivity significantly increased in injured neurons. SV2 expressed in nerve fibers within the urothelium, suburothelium, and detrusor. |
| [116] | Human | Urothelial cells. | Cultured human urothelial cell lines and conducted PCR. | Urothelial cells express SV2-A and SV2-B. |
| [118] | Human | Parasympathetic nerves innervating detrusor. | Biopsies of detrusor muscle from patients with sensory urgency and control patients, conducted IHC for SV2 and P2X receptors. | Used SV2 as a neuronal marker for parasympathetic nerves. There was no staining in detrusor muscle itself. |
| [103] | Human | Urothelium and suburothelial fibers. | Instilled 200U Lipotoxin (liposome encapsulated BoNT/A) into bladders of OAB patients, IHC for SV2 expression before treatment and 3 months after. | IHC and western blotting shows SV2 expression in the urothelium (apical cells and suburothelium). |
| [101] | Human | Nerve fibers in the suburothelium and detrusor. | Human bladders from organ donors were used for immunofluorescence staining of SV2. | Dense SV2 immunoreactivity in the suburothelium and detrusor layer colocalized with VAChT and CGRP positive fibers. They found no labelling within urothelium or muscle cells. |
| [41] | Rat and human | Bladder mucosa. | IHC and gel electrophoresis of rat and human bladder mucosa (urothelium and lamina propria) and cultured urothelial cells. | SV2 expression found in human and rat mucosa, and rat cultured urothelial cells. Expression was not found in human urothelial cells. |
| [110] | Human | Bladder mucosa (including the urothelium, lamina propria and a few discontinuous muscularis mucosa). | Human bladders of OAB patients received Lipotoxin and BoNT/A injection. IHC and immunoblotting performed at baseline and 3 months post treatment. | SV2 expression found in mucosa of OAB patients at baseline and one month after BoNT/A injection and Lipotoxin treatment. |
| [123] | Guinea pig | Suburothelial nerve fibers. | IHC staining for SV2 conducted on fixed guinea pig bladders. | SV2 expression found in suburothelial nerve fibers, authors used SV2 as a marker for efferent nerves. |
9. Evidence in Support of a Classical Mechanism for BoNT/A in the Bladder
10. Use of Animal Models in Urology Research
11. BoNT Research and Future Prospects
12. Conclusions
13. Limitations
14. Methods
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lacy, D.B.; Stevens, R.C. Sequence homology and structural analysis of the clostridial neurotoxins. J. Mol. Biol. 1999, 291, 1091–1104. [Google Scholar] [CrossRef]
- FDA. Highlights of Prescribing Information These highlights do not Include all the Information Needed to Use BOTOX® Safely and Effectively. See Full Prescribing Information for BOTOX. BOTOX (Onabotulinumtoxina) for Injection, for Intramuscular. Available online: www.fda.gov/medwatch (accessed on 24 June 2021).
- Dong, M.; Yeh, F.; Tepp, W.H.; Dean, C.; Johnson, E.A.; Janz, R.; Chapman, E.R. SV2 is the protein receptor for botulinum neurotoxin A. Science 2006, 312, 592–596. [Google Scholar] [CrossRef]
- Koriazova, L.K.; Montal, M. Translocation of botulinum neurotoxin light chain protease through the heavy chain channel. Nat. Struct. Biol. 2003, 10, 13–18. [Google Scholar] [CrossRef]
- Schiavo, G.; Rossetto, O.; Catsicas, S.; de Laureto, P.P.; DasGupta, B.R.; Benfenati, F.; Montecucco, C. Identification of the nerve terminal targets of botulinum neurotoxin serotypes A, D, and E. J. Biol. Chem. 1993, 268, 23784–23787. [Google Scholar] [CrossRef]
- Burstein, R.; Zhang, X.; Levy, D.; Aoki, K.R.; Brin, M.F. Selective inhibition of meningeal nociceptors by botulinum neurotoxin type A: Therapeutic implications for migraine and other pains. Cephalalgia 2014, 34, 853–869. [Google Scholar] [CrossRef] [PubMed]
- Collins, V.M.; Daly, D.M.; Liaskos, M.; McKay, N.G.; Sellers, D.; Chapple, C.; Grundy, D. Onabotulinumtoxin A significantly attenuates bladder afferent nerve firing and inhibits ATP release from the urothelium. BJU Int. 2013, 112, 1018–1026. [Google Scholar]
- Chapple, C.; Sievert, K.-D.; MacDiarmid, S.; Khullar, V.; Radziszewski, P.; Nardo, C.; Thompson, C.; Zhou, J.; Haag-Molkenteller, C. OnabotulinumtoxinA 100 U significantly improves all idiopathic overactive bladder symptoms and quality of life in patients with overactive bladder and urinary incontinence: A randomised, double-blind, placebo-controlled trial. Eur. Urol. 2013, 64, 249–256. [Google Scholar] [CrossRef]
- Kuo, H.; Jiang, Y.; Tsai, Y.; Kuo, Y. Intravesical botulinum toxin-A injections reduce bladder pain of interstitial cystitis/bladder pain syndrome refractory to conventional treatment–A prospective, multicenter, randomized, double-blind, placebo-controlled clinical trial. Neurourol. Urodyn. 2016, 35, 609–614. [Google Scholar] [CrossRef]
- Smith, C.P.; Radziszewski, P.; Borkowski, A.; Somogyi, G.T.; Boone, T.B.; Chancellor, M.B. Botulinum toxin a has antinociceptive effects in treating interstitial cystitis. Urology 2004, 64, 871–875. [Google Scholar] [CrossRef] [PubMed]
- Yoshimura, N.; Chancellor, M.B. Neurophysiology of Lower Urinary Tract Function and Dysfunction. Rev. Urol. 2003, 5 (Suppl. 8), S3. Available online: /pmc/articles/PMC1502389/ (accessed on 20 December 2021). [PubMed]
- Silva, C.; Silva, J.; Castro, H.; Reis, F.; Dinis, P.; Avelino, A.; Cruz, F. Bladder sensory desensitization decreases urinary urgency. BMC Urol. 2007, 7, 9. [Google Scholar] [CrossRef] [PubMed]
- Silva, C.; Rio, M.E.; Cruz, F. Desensitization of bladder sensory fibers by intravesical resiniferatoxin, a capsaicin analog: Long-term results for the treatment of detrusor hyperreflexia. Eur. Urol. 2000, 38, 444–452. Available online: https://www.karger.com/Article/FullText/20322 (accessed on 8 June 2021). [CrossRef]
- Ikeda, Y.; Zabbarova, I.V.; Birder, L.A.; de Groat, W.C.; McCarthy, C.J.; Hanna-Mitchell, A.T.; Kanai, A.J. Botulinum Neurotoxin Serotype A Suppresses Neurotransmitter Release from Afferent as Well as Efferent Nerves in the Urinary Bladder. Eur. Urol. 2012, 62, 1157–1164. [Google Scholar] [CrossRef]
- Abrams, P.; Cardozo, L.; Fall, M.; Griffiths, D.; Rosier, P.; Ulmsten, U.; van Kerrebroeck, P.; Victor, A.; Wein, A. The standardisation of terminology in lower urinary tract function: Report from the standardisation sub-committee of the International Continence Society. Urology 2003, 61, 37–49. [Google Scholar] [CrossRef]
- Koziol, J.A.; Clark, D.C.; Gittes, R.F.; Tan, E.M. The natural history of interstitial cystitis: A survey of 374 patients. J. Urol. 1993, 149, 465–469. [Google Scholar] [CrossRef]
- Hanno, P.; Dmochowski, R. Status of international consensus on interstitial cystitis/bladder pain syndrome/painful bladder syndrome: 2008 snapshot. Neurourol. Urodyn. Off. J. Int. Cont. Soc. 2009, 28, 274–286. [Google Scholar] [CrossRef] [PubMed]
- Kelleher, C.; Hakimi, Z.; Zur, R.; Siddiqui, E.; Maman, K.; Aballéa, S.; Nazir, J.; Chapple, C. Efficacy and tolerability of mirabegron compared with antimuscarinic monotherapy or combination therapies for overactive bladder: A systematic review and network meta-analysis. Eur. Urol. 2018, 74, 324–333. [Google Scholar] [CrossRef]
- Urinary Incontinence and Pelvic Organ Prolapse in Women: Management NICE Guideline. 2019. Available online: www.nice.org.uk/guidance/ng123 (accessed on 27 August 2021).
- Zagorodnyuk, V.P.; Costa, M.; Brookes, S.J.H. Major classes of sensory neurons to the urinary bladder. Auton. Neurosci.-Basic Clin. 2006, 126, 390–397. [Google Scholar] [CrossRef]
- Igawa, Y.; Yamazaki, Y.; Takeda, H.; Hayakawa, K.; Akahane, M.; Ajisawa, Y.; Yoneyama, T.; Nishizawa, O.; Andersson, K. Functional and molecular biological evidence for a possible β3-adrenoceptor in the human detrusor muscle. Br. J. Pharmacol. 1999, 126, 819–825. [Google Scholar] [CrossRef]
- Kunisawa, Y.; Kawabe, K.; Niijima, T.; Honda, K.; Takenaka, O. A pharmacological study of alpha adrenergic receptor subtypes in smooth muscle of human urinary bladder base and prostatic urethra. J. Urol. 1985, 134, 396–398. [Google Scholar] [CrossRef]
- Persson, K.; Igawa, Y.; Mattiasson, A.; Andersson, K. Effects of inhibition of the L-arginine/nitric oxide pathway in the rat lower urinary tract in vivo and in vitro. Br. J. Pharmacol. 1992, 107, 178–184. [Google Scholar] [CrossRef] [PubMed]
- Matsui, M.; Motomura, D.; Fujikawa, T.; Jiang, J.; Takahashi, S.; Manabe, T.; Taketo, M.M. Mice lacking M2 and M3 muscarinic acetylcholine receptors are devoid of cholinergic smooth muscle contractions but still viable. J. Neurosci. Off. J. Soc. Neurosci. 2002, 22, 10627–10632. [Google Scholar] [CrossRef]
- Sibley, G.N. A comparison of spontaneous and nerve-mediated activity in bladder muscle from man, pig and rabbit. J. Physiol. 1984, 354, 431–443. Available online: https://physoc.onlinelibrary.wiley.com/doi/full/10.1113/jphysiol.1984.sp015386 (accessed on 8 June 2021). [CrossRef]
- Yoshida, M.; Inadome, A.; Maeda, Y.; Satoji, Y.; Masunaga, K.; Sugiyama, Y.; Murakami, S. Non-neuronal cholinergic system in human bladder urothelium. Urology 2006, 67, 425–430. [Google Scholar] [CrossRef]
- Moro, C.; Uchiyama, J.; Chess-Williams, R. Urothelial/Lamina Propria Spontaneous Activity and the Role of M3 Muscarinic Receptors in Mediating Rate Responses to Stretch and Carbachol. Urology 2011, 78, 1442.e9–1442.e15. Available online: https://linkinghub.elsevier.com/retrieve/pii/S0090429511023065 (accessed on 6 June 2021). [CrossRef] [PubMed]
- Birder, L.A.; Apodaca, G.; De Groat, W.C.; Kanai, A.J. Adrenergic- and capsaicin-evoked nitric oxide release from urothelium and afferent nerves in urinary bladder. Am. J. Physiol.-Renal Physiol. 1998, 275, F226–F229. [Google Scholar] [CrossRef]
- Butler, D.S.C.; Ambite, I.; Nagy, K.; Cafaro, C.; Ahmed, A.; Nadeem, A.; Filenko, N.; Tran, T.H.; Andersson, K.; Wullt, B.; et al. Neuroepithelial control of mucosal inflammation in acute cystitis. Sci. Rep. 2018, 8, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, D.R.; Kennedy, I.; Burton, T.J. ATP is released from rabbit urinary bladder epithelial cells by hydrostatic pressure changes–possible sensory mechanism? J. Physiol. 1997, 505, 503–511. [Google Scholar] [CrossRef]
- Burnstock, G.; Dumsday, B.; Smythe, A. Atropine resistant excitation of the urinary bladder: The possibility of transmission via nerves releasing a purine nucleotide. Br. J. Pharmacol. 1972, 44, 451–461. [Google Scholar] [CrossRef]
- Llewellyn-Smith, I.J.; Burnstock, G. Ultrastructural localization of P2X3receptors in rat sensory neurons. Neuroreport 1998, 9, 2545–2550. [Google Scholar] [CrossRef] [PubMed]
- Nakagomi, H.; Yoshiyama, M.; Mochizuki, T.; Miyamoto, T.; Komatsu, R.; Imura, Y.; Morizawa, Y.; Hiasa, M.; Miyaji, T.; Kira, S. Urothelial ATP Exocytosis: Regulation of Bladder Compliance in the Urine Storage Phase. Sci. Rep. 2016, 6, 1–14. Available online: https://pubmed.ncbi.nlm.nih.gov/27412485/ (accessed on 14 June 2021). [CrossRef]
- Silberfeld, A.; Chavez, B.; Obidike, C.; Daugherty, S.; De Groat, W.C.; Beckel, J.M. LPS-mediated release of ATP from urothelial cells occurs by lysosomal exocytosis. Neurourol. Urodyn. 2020, 39, 1321–1329. [Google Scholar] [CrossRef]
- Wang, E.C.Y.; Lee, J.; Ruiz, W.G.; Balestreire, E.M.; Von Bodungen, M.; Barrick, S.; Cockayne, D.A.; Birder, L.A.; Apodaca, G. ATP and purinergic receptor–dependent membrane traffic in bladder umbrella cells. J. Clin. Investig. 2005, 115, 2412. [Google Scholar] [CrossRef] [PubMed]
- Vlaskovska, M.; Kasakov, L.; Rong, W.; Bodin, P.; Bardini, M.; Cockayne, D.A.; Ford, A.P.; Burnstock, G. P2X3 knock-out mice reveal a major sensory role for urothelially released ATP. J. Neurosci. Off. J. Soc. Neurosci. 2001, 21, 5670–5677. [Google Scholar] [CrossRef]
- Rong, W.; Spyer, K.M.; Burnstock, G. Activation and sensitisation of low and high threshold afferent fibres mediated by P2X receptors in the mouse urinary bladder. J. Physiol. 2002, 541, 591–600. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; De Groat, W.C. Nitric oxide modulates bladder afferent nerve activity in the in vitro urinary bladder–pelvic nerve preparation from rats with cyclophosphamide induced cystitis. Brain Res. 2013, 1490, 83. [Google Scholar] [CrossRef]
- A Munoz, A.; Somogyi, G.T.; Boone, T.B.; Smith, C.P. Central Inhibitory Effect of Intravesically Applied Botulinum Toxin A in Chronic Spinal Cord Injury. Neurourol. Urodyn. 2011, 30, 1376–1381. Available online: https://onlinelibrary.wiley.com/doi/full/10.1002/nau.21068 (accessed on 18 June 2021). [CrossRef] [PubMed]
- Smith, C.P.; Gangitano, D.A.; Munoz, A.; Salas, N.A.; Boone, T.B.; Aoki, K.R.; Francis, J.; Somogyi, G.T. Botulinum toxin type A normalizes alterations in urothelial ATP and NO release induced by chronic spinal cord injury. Neurochem. Int. 2008, 52, 1068–1075. [Google Scholar] [CrossRef]
- Hanna-Mitchell, A.T.; Wolf-Johnston, A.S.; Barrick, S.R.; Kanai, A.J.; Chancellor, M.B.; de Groat, W.C.; Birder, L.A. Effect of Botulinum Toxin A on Urothelial-Release of ATP and Expression of SNARE Targets within the Urothelium. Neurourol. Urodyn. 2015, 34, 79–84. [Google Scholar] [CrossRef] [PubMed]
- Khera, M.; Somogyi, G.T.; Kiss, S.; Boone, T.B.; Smith, C.P. Botulinum toxin A inhibits ATP release from bladder urothelium after chronic spinal cord injury. Neurochem. Int. 2004, 45, 987–993. [Google Scholar] [CrossRef]
- Smith, C.P.; Vemulakonda, V.M.; Kiss, S.; Boone, T.B.; Somogyi, G.T. Enhanced ATP release from rat bladder urothelium during chronic bladder inflammation: Effect of botulinum toxin A. Neurochem. Int. 2005, 47, 291–297. [Google Scholar] [CrossRef] [PubMed]
- Dykstra, D.D.; Sidi, A.A.; Scott, A.B.; Pagel, J.M.; Goldish, G.G. Effects of botulinum A toxin on detrusor-sphincter dyssynergia in spinal cord injury patients. J. Urol. 1988, 139, 919–922. [Google Scholar] [CrossRef]
- Schurch, B.; Stöhrer, M.; Kramer, G.; Schmid, D.M.; Gaul, G.; Hauri, D. Botulinum-A toxin for treating detrusor hyperreflexia in spinal cord injured patients: A new alternative to anticholinergic drugs? Preliminary results. J. Urol. 2000, 164, 692–697. [Google Scholar] [CrossRef]
- Denys, P.; le Normand, L.; Ghout, I.; Costa, P.; Chartier-Kastler, E.; Grise, P.; Hermieu, J.F.; Amarenco, G.; Karsenty, G.; Saussine, C.; et al. Efficacy and safety of low doses of onabotulinumtoxina for the treatment of refractory idiopathic overactive bladder: A multicentre, double-blind, randomised, placebo-controlled dose-ranging study. Eur. Urol. 2012, 61, 520–529. [Google Scholar] [CrossRef]
- Schurch, B.; de Sèze, M.; Denys, P.; Chartier-Kastler, E.; Haab, F.; Everaert, K.; Plante, P.; Perrouin-Verbe, B.; Kumar, C.; Fraczek, S.; et al. Botox Detrusor Hyperreflexia Study Team. Botulinum toxin type A is a safe and effective treatment for neurogenic urinary incontinence: Results of a single treatment, randomized, placebo controlled 6-month study. J. Urol. 2005, 174, 196–200. [Google Scholar] [CrossRef]
- Dmochowski, R.; Chapple, C.; Nitti, V.W.; Chancellor, M.; Everaert, K.; Thompson, C.; Daniell, G.; Zhou, J.; Haag-Molkenteller, C. Efficacy and safety of onabotulinumtoxinA for idiopathic overactive bladder: A double-blind, placebo controlled, randomized, dose ranging trial. J. Urol. 2010, 184, 2416–2422. [Google Scholar] [CrossRef]
- Ginsberg, D.; Gousse, A.; Keppenne, V.; Sievert, K.D.; Thompson, C.; Lam, W.; Brin, M.F.; Jenkins, B.; Haag-Molkenteller, C. Phase 3 Efficacy and Tolerability Study of Onabotulinumtoxina for Urinary Incontinence from Neurogenic Detrusor Overactivity. J. Urol. 2012, 187, 2131–2139. Available online: https://pubmed.ncbi.nlm.nih.gov/22503020/ (accessed on 14 June 2021). [CrossRef]
- Kuo, H.; Chancellor, M.B. Comparison of Intravesical Botulinum Toxin Type A Injections Plus Hydrodistention with Hydrodistention Alone for the Treatment of Refractory Interstitial Cystitis/Painful Bladder Syndrome. BJU Int. 2009, 104, 657–661. [Google Scholar] [CrossRef] [PubMed]
- Pinto, R.; Lopes, T.; Frias, B.; Silva, A.; Silva, J.A.; Silva, C.M.; Cruz, C.; Cruz, F.; Dinis, P. Trigonal injection of botulinum toxin A in patients with refractory bladder pain syndrome/interstitial cystitis. Eur. Urol. 2010, 58, 360–365. [Google Scholar] [CrossRef] [PubMed]
- Giannantoni, A.; Costantini, E.; Stasi SM di Tascini, M.C.; Bini, V.; Porena, M. Botulinum A toxin intravesical injections in the treatment of painful bladder syndrome: A pilot study. Eur. Urol. 2006, 49, 704–709. [Google Scholar] [CrossRef]
- Cruz, F.; Herschorn, S.; Aliotta, P.; Brin, M.; Thompson, C.; Lam, W.; Daniell, G.; Heesakkers, J.; Haag-Molkenteller, C. Efficacy and safety of onabotulinumtoxinA in patients with urinary incontinence due to neurogenic detrusor overactivity: A randomised, double-blind, placebo-controlled trial. Eur. Urol. 2011, 60, 742–750. [Google Scholar] [CrossRef] [PubMed]
- Chung, M.D.S.-D.; Kuo, M.D.Y.-C.; Kuo, M.D.H.-C. Intravesical onabotulinumtoxinA injections for refractory painful bladder syndrome. Pain Physician 2012, 15, 197–202. [Google Scholar] [PubMed]
- Kalsi, V.; Apostolidis, A.; Popat, R.; Gonzales, G.; Fowler, C.J.; Dasgupta, P. Quality of life changes in patients with neurogenic versus idiopathic detrusor overactivity after intradetrusor injections of botulinum neurotoxin type A and correlations with lower urinary tract symptoms and urodynamic changes. Eur. Urol. 2006, 49, 528–535. [Google Scholar] [CrossRef] [PubMed]
- Blasi, J.; Chapman, E.R.; Link, E.; Binz, T.; Yamasaki, S.; de Camilli, P.; Südhof, T.C.; Niemann, H.; Jahn, R. Botulinum neurotoxin A selectively cleaves the synaptic protein SNAP-25. Nature 1993, 365, 160–163. [Google Scholar] [CrossRef] [PubMed]
- de Groat, W.C.; Nadelhaft, I.; Milne, R.J.; Booth, A.M.; Morgan, C.; Thor, K. Organization of the sacral parasympathetic reflex pathways to the urinary bladder and large intestine. J. Auton. Nerv. Syst. 1981, 3, 135–160. [Google Scholar] [CrossRef]
- Häbler, H.J.; Jänig, W.; Koltzenburg, M. Activation of unmyelinated afferent fibres by mechanical stimuli and inflammation of the urinary bladder in the cat. J. Physiol. 1990, 425, 545–562. [Google Scholar] [CrossRef]
- Apostolidis, A.; Popat, R.; Yiangou, Y.; Cockayne, D.; Ford, A.P.D.W.; Davis, J.B.; Dasgupta, P.; Fowler, C.J.; Anand, P. Decreased Sensory Receptors P2X3 and TRPV1 in Suburothelial Nerve Fibers Following Intradetrusor Injections of Botulinum Toxin for Human Detrusor Overactivity. J. Urol. 2005, 174, 977–983. Available online: https://pubmed.ncbi.nlm.nih.gov/16094018/ (accessed on 6 June 2021). [CrossRef]
- Shimizu, T.; Shibata, M.; Toriumi, H.; Iwashita, T.; Funakubo, M.; Sato, H.; Kuroi, T.; Ebine, T.; Koizumi, K.; Suzuki, N. Reduction of TRPV1 expression in the trigeminal system by botulinum neurotoxin type-A. Neurobiol. Dis. 2012, 48, 367–378. [Google Scholar] [CrossRef] [PubMed]
- Shin, M.C.; Wakita, M.; Xie, D.J.; Yamaga, T.; Iwata, S.; Torii, Y.; Harakawa, T.; Ginnaga, A.; Kozaki, S.; Akaike, N. Inhibition of Membrane Na+ Channels by A Type Botulinum Toxin at Femtomolar Concentrations in Central and Peripheral Neurons. J. Pharmacol. Sci. 2012, 118, 33–42. [Google Scholar] [CrossRef]
- Erman, A.; Veranič, P.; Pšeničnik, M.; Jezernik, K. Superficial cell differentiation during embryonic and postnatal development of mouse urothelium. Tissue Cell 2006, 38, 293–301. [Google Scholar] [CrossRef]
- Jost, S.P.; Gosling, J.A.; Dixon, J.S. The morphology of normal human bladder urothelium. J. Anat. 1989, 167, 103–115. Available online: http://www.ncbi.nlm.nih.gov/pubmed/2630525 (accessed on 9 June 2021).
- Krhut, J.; Zvara, P. Intravesical Instillation of Botulinum Toxin A: An in vivo Murine Study and Pilot Clinical Trial. Int. Urol. Nephrol. 2011, 43, 337–343. Available online: https://pubmed.ncbi.nlm.nih.gov/20563845/ (accessed on 10 June 2021). [CrossRef] [PubMed]
- Chuang, Y.-C.; Yoshimura, N.; Huang, C.-C.; Chiang, P.-H.; Chancellor, M.B. Intravesical botulinum toxin a administration produces analgesia against acetic acid induced bladder pain responses in rats. J. Urol. 2004, 172 Pt 1, 1529–1532. [Google Scholar] [CrossRef]
- Niku, S.D.; Stein, P.C.; Scherz, H.C.; Parsons, C.L. A new method for cytodestruction of bladder epithelium using protamine sulfate and urea. J. Urol. 1994, 152, 1025–1028. [Google Scholar] [CrossRef]
- Meng, J.; Wang, J.; Steinhoff, M.; Dolly, J.O. TNFα induces co-trafficking of TRPV1/TRPA1 in VAMP1-containing vesicles to the plasmalemma via Munc18-1/syntaxin1/SNAP-25 mediated fusion. Sci. Rep. 2016, 6, 1–15. Available online: http://www.nature.com/scientificreports/ (accessed on 11 June 2021). [CrossRef]
- Zhang, X.; Strassman, A.M.; Novack, V.; Brin, M.F.; Burstein, R. Extracranial Injections of Botulinum Neurotoxin Type A Inhibit Intracranial Meningeal Nociceptors’ Responses to Stimulation of TRPV1 and TRPA1 Channels: Are We Getting Closer to Solving This Puzzle? Cephalalgia 2016, 36, 875–886. [Google Scholar] [CrossRef] [PubMed]
- Xiao, L.; Cheng, J.; Zhuang, Y.; Qu, W.; Muir, J.; Liang, H.; Zhang, D. Botulinum toxin type A reduces hyperalgesia and TRPV1 expression in rats with neuropathic pain. Pain Med. 2013, 14, 276–286. [Google Scholar] [CrossRef]
- Fan, C.; Chu, X.; Wang, L.; Shi, H.; Li, T. Botulinum toxin type A reduces TRPV1 expression in the dorsal root ganglion in rats with adjuvant-arthritis pain. Toxicon 2017, 133, 116–122. [Google Scholar] [CrossRef]
- Morenilla-Palao, C.; Planells-Cases, R.; García-Sanz, N.; Ferrer-Montiel, A. Regulated exocytosis contributes to protein kinase C potentiation of vanilloid receptor activity. J. Biol. Chem. 2004, 279, 25665–25672. [Google Scholar] [CrossRef]
- Xiao, L.; Cheng, J.; Dai, J.; Zhang, D. Botulinum Toxin Decreases Hyperalgesia and Inhibits P2 × 3 Receptor Over-Expression in Sensory Neurons Induced by Ventral Root Transection in Rats. Pain Med. 2011, 12, 1385–1394. Available online: https://academic.oup.com/painmedicine/article/12/9/1385/1899274 (accessed on 12 June 2021). [CrossRef] [PubMed]
- Ha, U.-S.; Park, E.Y.; Kim, J.C. Effect of Botulinum Toxin on Expression of Nerve Growth Factor and Transient Receptor Potential Vanilloid 1 in Urothelium and Detrusor Muscle of Rats With Bladder Outlet Obstruction-induced Detrusor Overactivity. Urology 2011, 78, 721.e1–721.e6. Available online: https://linkinghub.elsevier.com/retrieve/pii/S0090429511005620 (accessed on 11 June 2021). [CrossRef]
- Rong, W.F.; Burnstock, G. Activation of ureter nociceptors by exogenous and endogenous ATP in guinea pig. Neuropharmacology 2004, 47, 1093–1101. [Google Scholar] [CrossRef]
- Daly, D.; Rong, W.; Chess-Williams, R.; Chapple, C.; Grundy, D. Bladder afferent sensitivity in wild-type and TRPV1 knockout mice. J. Physiol. 2007, 583, 663–674. [Google Scholar] [CrossRef] [PubMed]
- Brierley, S.M.; Hughes, P.A.; Page, A.J.; Kwan, K.Y.; Martin, C.M.; O’Donnell, T.A.; Cooper, N.J.; Harrington, A.M.; Adam, B.; Liebregts, T. The ion channel TRPA1 is required for normal mechanosensation and is modulated by algesic stimuli. Gastroenterology 2009, 137, 2084–2095e3. [Google Scholar] [CrossRef]
- Du, S.; Araki, I.; Yoshiyama, M.; Nomura, T.; Takeda, M. Transient receptor potential channel A1 involved in sensory transduction of rat urinary bladder through C-fiber pathway. Urology 2007, 70, 826–831. [Google Scholar] [CrossRef] [PubMed]
- Bossowska, A.; Majewski, M. Botulinum Toxin Type A-Induced Changes in the Chemical Coding of dorsal Root Ganglion Neurons Supplying the Porcine Urinary Bladder. Polish J. Vet. Sci. 2012, 15, 345–353. Available online: http://yadda.icm.edu.pl/yadda/element/bwmeta1.element.agro-918029d9-07ec-4d0e-a186-b3b269270805 (accessed on 22 December 2021). [CrossRef] [PubMed]
- Welch, M.J.; Purkiss, J.R.; Foster, K.A. Sensitivity of embryonic rat dorsal root ganglia neurons to Clostridium botulinum neurotoxins. Toxicon 2000, 38, 245–258. [Google Scholar] [CrossRef]
- Purkiss, J.; Welch, M.; Doward, S.; Foster, K. Capsaicin-stimulated release of substance P from cultured dorsal root ganglion neurons: Involvement of two distinct mechanisms. Biochem. Pharmacol. 2000, 59, 1403–1406. [Google Scholar] [CrossRef]
- Kim, J.C.; Park, E.Y.; Seo, S.I.; Park, Y.H.; Hwang, T.K. Nerve Growth Factor and Prostaglandins in the Urine of Female Patients With Overactive Bladder. J. Urol. 2006, 175, 1773–1776. Available online: https://pubmed.ncbi.nlm.nih.gov/16600756/ (accessed on 14 June 2021). [CrossRef]
- Qu, H.C.; Zhang, W.; Yan, S.; Liu, Y.L.; Wang, P. Urinary Nerve Growth Factor Could Be a Biomarker for Interstitial Cystitis/Painful Bladder Syndrome: A Meta-Analysis. PLoS ONE 2014, 9, 106321. Available online: http://www.plosone.org (accessed on 14 June 2021). [CrossRef]
- Dmitrieva, N.; McMahon, S.B. Sensitisation of visceral afferents by nerve growth factor in the adult rat. Pain 1996, 66, 87–97. [Google Scholar] [CrossRef]
- Dmitrieva, N.; Shelton, D.; Rice, A.S.C.; McMahon, S.B. The role of nerve growth factor in a model of visceral inflammation. Neuroscience 1997, 78, 449–459. [Google Scholar] [CrossRef]
- Liu, H.T.; Chancellor, M.B.; Kuo, H.C. Urinary Nerve Growth Factor Levels are Elevated in Patients with Detrusor Overactivity and Decreased in Responders to Detrusor Botulinum Toxin-A Injection. Eur. Urol. 2009, 56, 700–707. [Google Scholar] [CrossRef]
- Blöchl, A. SNAP-25 and Syntaxin, But Not Synaptobrevin 2, Cooperate in the Regulated Release of Nerve Growth Factor. Neuroreport 1998, 9, 1701–1705. Available online: https://pubmed.ncbi.nlm.nih.gov/9665586/ (accessed on 15 June 2021). [CrossRef]
- Heymach, J.V.; Krüttgen, A.; Suter, U.; Shooter, E.M. The regulated secretion and vectorial targeting of neurotrophins in neuroendocrine and epithelial cells. J. Biol. Chem. 1996, 271, 25430–25437. [Google Scholar] [CrossRef]
- Beckel, J.M.; Birder, L.A. Differential Expression and Function of Nicotinic Acetylcholine Receptors in the Urinary Bladder Epithelium of the Rat. J. Physiol. 2012, 590, 1465–1480. Available online: https://physoc.onlinelibrary.wiley.com/doi/full/10.1113/jphysiol.2011.226860 (accessed on 15 June 2021). [CrossRef] [PubMed]
- Chess-Williams, R. Muscarinic Receptors of the Urinary Bladder: Detrusor, Urothelial and Prejunctional. Auton. Autacoid Pharmacol. 2002, 22, 133–145. Available online: https://onlinelibrary.wiley.com/doi/full/10.1046/j.1474-8673.2002.00258.x (accessed on 15 June 2021). [CrossRef] [PubMed]
- Elneil, S.; Skepper, J.N.; Kidd, E.J.; Williamson, J.G.; Ferguson, D.R. Distribution of P2 × 1 and P2 × 3 Receptors in the Rat and Human Urinary Bladder. Pharmacology 2001, 63, 120–128. Available online: https://www.karger.com/Article/FullText/56122 (accessed on 15 June 2021). [CrossRef] [PubMed]
- Chess-Williams, R.; Sellers, D.J.; Brierley, S.M.; Grundy, D.; Grundy, L. Purinergic Receptor Mediated Calcium Signalling in Urothelial cells. Sci. Rep. 2019, 9, 1–10. [Google Scholar] [CrossRef]
- Ochodnický, P.; Humphreys, S.; Eccles, R.; Poljakovic, M.; Wiklund, P.; Michel, M.C. Expression Profiling of G-protein-coupled Receptors in Human Urothelium and Related Cell Lines. BJU Int. 2012, 110, E293–E300. Available online: https://bjui-journals.onlinelibrary.wiley.com/doi/full/10.1111/j.1464-410X.2012.011145.x (accessed on 15 June 2021). [CrossRef]
- Hanna-Mitchell, A.T.; Beckel, J.M.; Barbadora, S.; Kanai, A.J.; de Groat, W.C.; Birder, L.A. Non-neuronal acetylcholine and urinary bladder urothelium. Life Sci. 2007, 80, 2298–2302. [Google Scholar] [CrossRef] [PubMed]
- Knight, G.E.; Bodin, P.; de Groat, W.C.; Burnstock, G. ATP Is Released from Guinea Pig Ureter Epithelium on Distension. Am. J. Physiol.-Ren. Physiol. 2002, 282, F281–F288. Available online: https://pubmed.ncbi.nlm.nih.gov/11788442/ (accessed on 18 June 2021). [CrossRef] [PubMed][Green Version]
- Aizawa, N.; Igawa, Y.; Nishizawa, O.; Wyndaele, J.J. Effects of nitric oxide on the primary bladder afferent activities of the rat with and without intravesical acrolein treatment. Eur. Urol. 2011, 59, 264–271. [Google Scholar] [CrossRef]
- Kim, Y.J.; Kim, J.H.; Lee, K.J.; Choi, M.M.; Kim, Y.H.; Rhie, G.E.; Yoo, C.K.; Cha, K.; Shin, N.R. Botulinum Neurotoxin Type A Induces TLR2-Mediated Inflammatory Responses in Macrophages. PLoS ONE 2015, 10, e0120840. Available online: https://pubmed.ncbi.nlm.nih.gov/25853816/ (accessed on 20 December 2021). [CrossRef]
- Hu, L.; Feng, Y.; Liu, W.; Jin, L.; Nie, Z. Botulinum Toxin Type A suppresses arterial vasoconstriction by regulating calcium sensitization and the endothelium-dependent endothelial nitric oxide synthase/soluble guanylyl cyclase/cyclic guanosine monophosphate pathway: An in vitro study. Exp. Biol. Med. 2019, 244, 1475. [Google Scholar] [CrossRef]
- Wankel, B.; Ouyang, J.; Guo, X.; Hadjiolova, K.; Miller, J.; Liao, Y.; Tham, D.K.L.; Romih, R.; Andrade, L.R.; Gumper, I. Sequential and compartmentalized action of Rabs, SNAREs, and MAL in the apical delivery of fusiform vesicles in urothelial umbrella cells. Mol. Biol. Cell 2016, 27, 1621–1634. [Google Scholar] [CrossRef]
- Born, M.; Pahner, I.; Ahnert-Hilger, G.; Jöns, T. The maintenance of the permeability barrier of bladder facet cells requires a continuous fusion of discoid vesicles with the apical plasma membrane. Eur. J. Cell Biol. 2003, 82, 343–350. [Google Scholar] [CrossRef]
- Khandelwal, P.; Ruiz, W.G.; Apodaca, G. Compensatory endocytosis in bladder umbrella cells occurs through an integrin-regulated and RhoA-and dynamin-dependent pathway. EMBO J. 2010, 29, 1961–1975. Available online: https://www.embopress.org/doi/full/10.1038/emboj.2010.91 (accessed on 14 June 2021). [CrossRef] [PubMed]
- Coelho, A.; Dinis, P.; Pinto, R.; Gorgal, T.; Silva, C.; Silva, A.; Silva, J.; Cruz Célia, D.; Cruz, F.; Avelino, A. Distribution of the high-affinity binding site and intracellular target of botulinum toxin type A in the human bladder. Eur. Urol. 2010, 57, 884–890. [Google Scholar] [CrossRef]
- Chuang, Y.-C.; Tyagi, P.; Huang, C.-C.; Yoshimura, N.; Wu, M.; Kaufman, J.; Chancellor, M.B. Urodynamic and immunohistochemical evaluation of intravesical botulinum toxin A delivery using liposomes. J. Urol. 2009, 182, 786–792. [Google Scholar] [CrossRef] [PubMed]
- Kuo, H.C.; Liu, H.T.; Chuang, Y.C.; Birder, L.A.; Chancellor, M.B. Pilot study of liposome-encapsulated onabotulinumtoxinA for patients with overactive bladder: A single-center study. Eur. Urol. 2014, 65, 1117–1124. [Google Scholar] [CrossRef]
- Coelho, A.; Cruz, F.; Cruz Célia, D.; Avelino, A. Spread of onabotulinumtoxinA after bladder injection. Experimental study using the distribution of cleaved SNAP-25 as the marker of the toxin action. Eur. Urol. 2012, 61, 1178–1184. [Google Scholar] [CrossRef] [PubMed]
- Lucioni, A.; Bales, G.T.; Lotan, T.L.; McGehee, D.S.; Cook, S.P.; Rapp, D.E. Botulinum toxin type A inhibits sensory neuropeptide release in rat bladder models of acute injury and chronic inflammation. BJU Int. 2008, 101, 366–370. [Google Scholar] [CrossRef]
- Rapp, D.E.; Turk, K.W.; Bales, G.T.; Cook, S.P. Botulinum toxin type a inhibits calcitonin gene-related peptide release from isolated rat bladder. J. Urol. 2006, 175, 1138–1142. [Google Scholar] [CrossRef]
- Vaidyanathan, V.v.; Yoshino, K.; Jahnz, M.; Dörries, C.; Bade, S.; Nauenburg, S.; Niemann, H.; Binz, T. Proteolysis of SNAP-25 isoforms by botulinum neurotoxin types A, C, and E: Domains and amino acid residues controlling the formation of enzyme-substrate complexes and cleavage. J. Neurochem. 1999, 72, 327–337. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, A.; Li, G.; Alexander, E.A.; Schwartz, J.H. Role of SNAP-23 in trafficking of H -ATPase in cultured inner medullary collecting duct cells. Am. J. Physiol.-Cell Physiol. 2001, 280, C775–C781. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Foran, P.; Shone, C.C.; Foster, K.A.; Melling, J.; Dolly, J.O. Botulinum Neurotoxin B Inhibits Insulin-Stimulated Glucose Uptake into 3T3-L1 Adipocytes and Cleaves Cellubrevin Unlike Type A Toxin Which Failed To Proteolyze the SNAP-23 Present. Biochemistry 1997, 36, 5719–5728. Available online: https://pubs.acs.org/sharingguidelines (accessed on 9 August 2021). [CrossRef]
- Liu, H.T.; Chen, S.H.; Chancellor, M.B.; Kuo, H.C. Presence of cleaved synaptosomal-associated protein-25 and decrease of purinergic receptors p2 × 3 in the bladder urothelium influence efficacy of botulinum toxin treatment for overactive bladder syndrome. PLoS ONE 2015, 10, e0134803. Available online: https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0134803 (accessed on 6 June 2021).
- Schulte-Baukloh, H.; Zurawski, T.H.; Knispel, H.H.; Miller, K.; Haferkamp, A.; Dolly, J.O. Persistence of the synaptosomal-associated protein-25 cleavage product after intradetrusor botulinum toxin A injections in patients with myelomeningocele showing an inadequate response to treatment. BJU Int. 2007, 100, 1075–1080. [Google Scholar] [CrossRef]
- El Shatoury, M.G.; DeYoung, L.; Turley, E.; Yazdani, A.; Dave, S. Early experimental results of using a novel delivery carrier, hyaluronan-phosphatidylethanolamine (HA-PE), which may allow simple bladder instillation of botulinum toxin A as effectively as direct detrusor muscle injection. J. Pediatr. Urol. 2000, 14, 172.e1–172.e6. [Google Scholar] [CrossRef]
- Chuang, Y.-C.; Huang, C.-C.; Yoshimura, N.; Tyagi, P.; Chancellor, M.B.; Chiang, P.-H. Effects of botulinum toxin a on snap25 level and bladder function in a cyclophosphamide induced cystitis model in rats. J. Urol. 2008, 179, 61. Available online: https://www.auajournals.org/doi/abs/10.1016/S0022-5347%2808%2960180-9 (accessed on 18 October 2021). [CrossRef]
- Rhéaume, C.; Cai, B.B.; Wang, J.; Fernández-Salas, E.; Aoki, K.R.; Francis, J.; Broide, R.S. A Highly Specific Monoclonal Antibody for Botulinum Neurotoxin Type A-Cleaved SNAP25. Toxins 2015, 7, 2354. [Google Scholar] [CrossRef]
- Oliveira, R.; Coelho, A.; Charrua, A.; Avelino, A.; Cruz, F. Expression of cleaved SNAP-25 after bladder wall injection of onabotulinumtoxina or abobotulinumtoxina: A comparative study in the mice. Neurourol. Urodyn. 2017, 36, 86–90. Available online: https://pubmed.ncbi.nlm.nih.gov/26472491/ (accessed on 6 June 2021). [CrossRef] [PubMed]
- Giannantoni, A.; Proietti, S.; Vianello, A.; Amantini, C.; Santoni, G.; Porena, M. 793 Assessment of botulinum a toxin high affinity sv2 receptors on normal human urothelial cells. J. Urol. 2011, 185, 319. Available online: https://www.auajournals.org/doi/abs/10.1016/j.juro.2011.02.611 (accessed on 7 June 2021). [CrossRef]
- Yiangou, Y.; Anand, U.; Otto, W.R.; Sinisi, M.; Fox, M.; Birch, R.; Foster, K.A.; Mukerji, G.; Akbar, A.; Agarwal, S.K.; et al. Increased levels of SV2A botulinum neurotoxin receptor in clinical sensory disorders and functional effects of botulinum toxins A and E in cultured human sensory neurons. J. Pain Res. 2011, 4, 347–355. [Google Scholar] [PubMed]
- Ray, F.R.; Moore, K.H.; Hansen, M.A.; Barden, J.A. Loss of purinergic P2X receptor innervation in human detrusor and subepithelium from adults with sensory urgency. Cell Tissue Res. 2003, 314, 351–359. Available online: https://link.springer.com/article/10.1007/s00441-003-0788-z (accessed on 24 June 2021). [CrossRef]
- Chuang, Y.C.; Kaufmann, J.H.; Chancellor, D.D.; Chancellor, M.B.; Kuo, H.C. Bladder instillation of liposome encapsulated onabotulinumtoxina improves overactive bladder symptoms: A prospective, multicenter, double-blind, randomized trial. J. Urol. 2014, 192, 1743–1749. Available online: https://pubmed.ncbi.nlm.nih.gov/25046622/ (accessed on 10 June 2021). [CrossRef] [PubMed]
- Jacky, B.P.S.; Garay, P.E.; Dupuy, J.; Nelson, J.B.; Cai, B.; Molina, Y.; Wang, J.; Steward, L.E.; Broide, R.S.; Francis, J.; et al. Identification of Fibroblast Growth Factor Receptor 3 (FGFR3) as a Protein Receptor for Botulinum Neurotoxin Serotype A (BoNT/A). PLoS Pathog. 2013, 9, 1003369. Available online: https://www.plospathogens.org (accessed on 10 June 2021). [CrossRef]
- Bomba-Warczak, E.; Vevea, J.D.; Brittain, J.M.; Figueroa-Bernier, A.; Tepp, W.H.; Johnson, E.A.; Yeh, F.L.; Chapman, E.R. Interneuronal Transfer and Distal Action of Tetanus Toxin and Botulinum Neurotoxins A and D in Central Neurons. Cell Rep. 2016, 16, 1974–1987. [Google Scholar] [CrossRef]
- Akanksha, M.; Sandhya, S. Role of FGFR3 in urothelial carcinoma. Iran. J. Pathol. 2019, 14, 148–155. [Google Scholar] [CrossRef][Green Version]
- Rahnama’i, M.S.; van Koeveringe, G.A.; Hohnen, R.; Ona, S.; van Kerrebroeck, P.E.V.; de Wachter, S.G.G. Distribution of phosphodiesterase type 5 (PDE5) in the lateral wall of the guinea pig urinary bladder. BJU Int. 2013, 112, 246–257. Available online: https://onlinelibrary.wiley.com/doi/full/10.1111/bju.12031 (accessed on 7 October 2021). [CrossRef]
- Jositsch, G.; Papadakis, T.; Haberberger, R.V.; Wolff, M.; Wess, J.; Kummer, W. Suitability of muscarinic acetylcholine receptor antibodies for immunohistochemistry evaluated on tissue sections of receptor gene-deficient mice. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2009, 379, 389–395. [Google Scholar] [CrossRef]
- Lawrence, G.W.; Aoki, K.R.; Dolly, J.O. Excitatory cholinergic and purinergic signaling in bladder are equally susceptible to botulinum neurotoxin a consistent with co-release of transmitters from efferent fibers. J. Pharmacol. Exp. Ther. 2010, 334, 1080–1086. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, R.; Yunoki, T.; Naito, S.; Yoshimura, N. Differential effects of botulinum neurotoxin A on bladder contractile responses to activation of efferent nerves, smooth muscles and afferent nerves in rats. J. Urol. 2012, 188, 1993–1999. [Google Scholar] [CrossRef]
- MacKenzie, I.; Burnstock, G.; Dolly, J.O. The effects of purified botulinum neurotoxin type A on cholinergic, adrenergic and non-adrenergic, atropine-resistant autonomic neuromuscular transmission. Neuroscience 1982, 7, 997–1006. [Google Scholar] [CrossRef]
- Maignel-Ludop, J.; Huchet, M.; Krupp, J. Botulinum Neurotoxins Serotypes A and B induce paralysis of mouse striated and smooth muscles with different potencies. Pharmacol. Res. Perspect. 2017, 5, 289. Available online: https://bpspubs.onlinelibrary.wiley.com/doi/full/10.1002/prp2.289 (accessed on 8 June 2021). [CrossRef] [PubMed]
- Van Uhm, J.I.M.; Beckers, G.M.A.; van der Laarse, W.J.; Meuleman, E.J.H.; Geldof, A.A.; Nieuwenhuijzen, J.A. Development of an in vitro model to measure bioactivity of botulinum neurotoxin A in rat bladder muscle strips. BMC Urol. 2014, 14, 1–7. Available online: https://bmcurol.biomedcentral.com/articles/10.1186/1471-2490-14-37 (accessed on 8 June 2021). [CrossRef]
- Smith, C.P.; Boone, T.B.; de Groat, W.C.; Chancellor, M.B.; Somogyi, G.T. Effect of stimulation intensity and botulinum toxin isoform on rat bladder strip contractions. Brain Res. Bull. 2003, 61, 165–171. [Google Scholar] [CrossRef]
- Howles, S.; Curry, J.; McKay, I.; Reynard, J.; Brading, A.F. Lack of effectiveness of botulinum neurotoxin A on isolated detrusor strips and whole bladders from mice and guinea-pigs in vitro. BJU Int. 2009, 104, 1524–1530. Available online: https://bjui-journals.onlinelibrary.wiley.com/doi/full/10.1111/j.1464-410X.2009.08619.x (accessed on 8 June 2021). [CrossRef] [PubMed]
- Valvassori, S.S.; Dal-Pont, G.C.; Resende, W.R.; Varela, R.B.; Lopes-Borges, J.; Cararo, J.H.; Quevedo, J. Validation of the animal model of bipolar disorder induced by Ouabain: Face, construct and predictive perspectives. Transl. Psychiatry 2019, 9, 1–11. [Google Scholar] [CrossRef]
- Crowe, R.; Burnstock, G. A histochemical and immunohistochemical study of the autonomic innervation of the lower urinary tract of the female pig. Is the pig a good model for the human bladder and urethra? J. Urol. 1989, 141, 414–422. [Google Scholar] [CrossRef]
- Lepiarczyk, E.; Bossowska, A.; Majewski, M. Changes in chemical coding of sympathetic chain ganglia (SChG) neurons supplying porcine urinary bladder after botulinum toxin (BTX) treatment. Cell Tissue Res. 2015, 360, 263–272. [Google Scholar] [CrossRef]
- Stromberga, Z.; Chess-Williams, R.; Moro, C. The five primary prostaglandins stimulate contractions and phasic activity of the urinary bladder urothelium, lamina propria and detrusor. BMC Urol. 2020, 20, 48. Available online: https://bmcurol.biomedcentral.com/articles/10.1186/s12894-020-00619-0 (accessed on 22 December 2021). [CrossRef] [PubMed]
- Wyndaele, J.J.; de Wachter, S. Cystometrical Sensory Data from a Normal Population: Comparison of Two Groups of Young Healthy Volunteers Examined with 5 Years Interval. Eur. Urol. 2002, 42, 34–38. [Google Scholar] [CrossRef]
- Jeffery, S.; Fynes, M.; Lee, F.; Wang, K.; Williams, L.; Morley, R. Efficacy and complications of intradetrusor injection with botulinum toxin A in patients with refractory idiopathic detrusor overactivity. BJU Int. 2007, 100, 1302–1306. Available online: https://pubmed.ncbi.nlm.nih.gov/17979928/ (accessed on 21 December 2021). [CrossRef] [PubMed]
- Yoshiyama, M.; Mochizuki, T.; Nakagomi, H.; Miyamoto, T.; Kira, S.; Mizumachi, R.; Sokabe, T.; Takayama, Y.; Tominaga, M.; Takeda, M. Functional roles of TRPV1 and TRPV4 in control of lower urinary tract activity: Dual analysis of behavior and reflex during the micturition cycle. Am. J. Physiol.-Ren. Physiol. 2015, 308, F1128–F1134. [Google Scholar] [CrossRef] [PubMed]
- Bayliss, M.; Wu, C.; Newgreen, D.; Mundy, A.R.; Fry, C.H. A quantitative study of atropine-resistant contractile responses in human detrusor smooth muscle, from stable, unstable and obstructed bladders. J. Urol. 1999, 162, 1833–1839. Available online: https://www.auajournals.org/doi/abs/10.1016/S0022-5347%2805%2968247-X (accessed on 8 June 2021). [CrossRef]
- Gill, D.M. Bacterial toxins: A table of lethal amounts. Microbiol. Rev. 1982, 46, 86. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC373212 (accessed on 16 September 2021). [CrossRef]
- Nageib, M.; El-Hefnawy, A.S.; Zahran, M.H.; El-Tabey, N.A.; Sheir, K.Z.; Shokeir, A.A. Delivery of intravesical botulinum toxin A using low-energy shockwaves in the treatment of overactive bladder: A preliminary clinical study. Arab. J. Urol. 2019, 17, 216–220. Available online: https://www.tandfonline.com/action/journalInformation?journalCode=taju20 (accessed on 18 June 2021). [CrossRef]
- Krhut, J.; Navratilova, M.; Sykora, R.; Jurakova, M.; Gärtner, M.; Mika, D.; Pavliska, L.; Zvara, P. Intravesical instillation of onabotulinum toxin A embedded in inert hydrogel in the treatment of idiopathic overactive bladder: A double-blind randomized pilot study. Scand. J. Urol. 2016, 50, 200–205. Available online: https://www.tandfonline.com/action/journalInformation?journalCode=isju20 (accessed on 18 June 2021). [CrossRef]
- Fraser, M.O.; Chuang, Y.C.; Tyagi, P.; Yokoyama, T.; Yoshimura, N.; Huang, L.; de Groat, W.C.; Chancellor, M.B. Intravesical liposome administration-A novel treatment for hyperactive bladder in the rat. Urology 2003, 61, 656–663. [Google Scholar] [CrossRef]
- Peters, K.M.; Hasenau, D.; Killinger, K.A.; Chancellor, M.B.; Anthony, M.; Kaufman, J. Liposomal bladder instillations for IC/BPS: An open-label clinical evaluation. Int. Urol. Nephrol. 2014, 3, 2291–2295. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.C.; Su, C.H.; Tain, Y.L.; Tsai, C.N.; Yu, C.C.; Chuang, Y.C. Potential Orphan Drug Therapy of Intravesical Liposomal Onabotulinumtoxin-A for Ketamine-Induced Cystitis by Mucosal Protection and Anti-inflammation in a Rat Model. Sci. Rep. 2018, 8, 1–9. Available online: www.nature.com/scientificreports/ (accessed on 24 June 2021). [CrossRef] [PubMed]
- Chuang, Y.C.; Kuo, H.C. A Prospective, Multicenter, Double-Blind, Randomized Trial of Bladder Instillation of Liposome Formulation OnabotulinumtoxinA for Interstitial Cystitis/Bladder Pain Syndrome. J. Urol. 2017, 198, 376–382. [Google Scholar] [CrossRef] [PubMed]
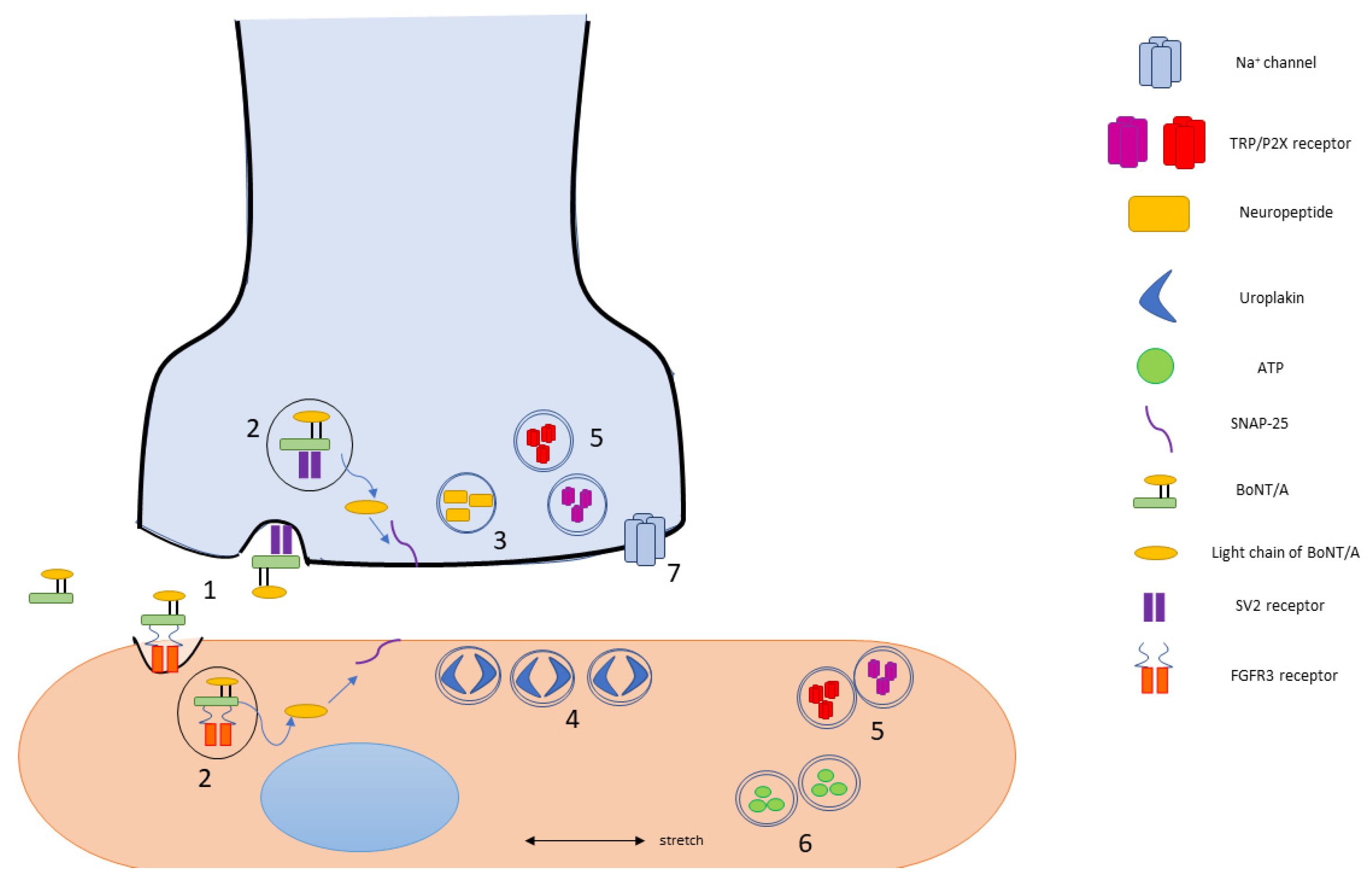
| Bladder Disorder | Definition | |
|---|---|---|
| OAB | Overactive bladder | A syndrome characterized by urinary urgency (the sudden need to urinate that cannot be deferred to later), frequency (needing to urinate more often), and nocturia (sleep disturbances caused by increased need to urinate) which can be accompanied by incontinence (inability to hold your bladder). |
| NDO | Neurogenic detrusor overactivity | Involuntary contraction of the smooth muscle of the bladder (the detrusor) during the storage phase. Defined as neurogenic when it occurs due to a neurological condition such as spinal cord injury. |
| IDO | Idiopathic detrusor overactivity | Involuntary contraction of the detrusor with no known cause. |
| IC/PBS | Interstitial cystitis/painful bladder syndrome | Pain that accompanies bladder filling, which may lead to frequency and nocturia. For the diagnosis of interstitial cystitis, cystoscopy and histological assessments are necessary. |
| BOO | Bladder outlet obstruction | When an obstruction in the urethra (most commonly an enlarged prostate) makes voiding difficult and can increase detrusor pressure due to the increased residual volume in the bladder. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ibrahim, H.; Maignel, J.; Hornby, F.; Daly, D.; Beard, M. BoNT/A in the Urinary Bladder—More to the Story than Silencing of Cholinergic Nerves. Toxins 2022, 14, 53. https://doi.org/10.3390/toxins14010053
Ibrahim H, Maignel J, Hornby F, Daly D, Beard M. BoNT/A in the Urinary Bladder—More to the Story than Silencing of Cholinergic Nerves. Toxins. 2022; 14(1):53. https://doi.org/10.3390/toxins14010053
Chicago/Turabian StyleIbrahim, Hodan, Jacquie Maignel, Fraser Hornby, Donna Daly, and Matthew Beard. 2022. "BoNT/A in the Urinary Bladder—More to the Story than Silencing of Cholinergic Nerves" Toxins 14, no. 1: 53. https://doi.org/10.3390/toxins14010053
APA StyleIbrahim, H., Maignel, J., Hornby, F., Daly, D., & Beard, M. (2022). BoNT/A in the Urinary Bladder—More to the Story than Silencing of Cholinergic Nerves. Toxins, 14(1), 53. https://doi.org/10.3390/toxins14010053





