Critical Assessment of Mycotoxins in Beverages and Their Control Measures
Abstract
:1. Introduction
2. Major Mycotoxins in Beverages
2.1. Aflatoxins
2.2. Ochratoxin A
2.3. Patulin
2.4. Fumonisin
2.5. Trichothecenes
2.5.1. Type A Trichothecenes (T-2 Toxin and HT-2)
2.5.2. Type B Trichothecenes (Deoxynivalenol)
2.6. Zearalenone
2.7. Alternaria Toxins
3. International Standards for Mycotoxins in Fruit Juices and Beverages
4. Detection and Quantification of Mycotoxins in Beverages
4.1. HPLC Detection
4.2. Mass Spectrometry Methods for Beer Mycotoxin Detection
4.3. Biosensor and Immunosensor Mycotoxin Detection
4.4. Microchip Method for the Detection of Mycotoxins in Beverages
4.5. Biomarker Assay
4.6. Nanoparticle-Based Detection Methods
4.7. ELISA Detection System
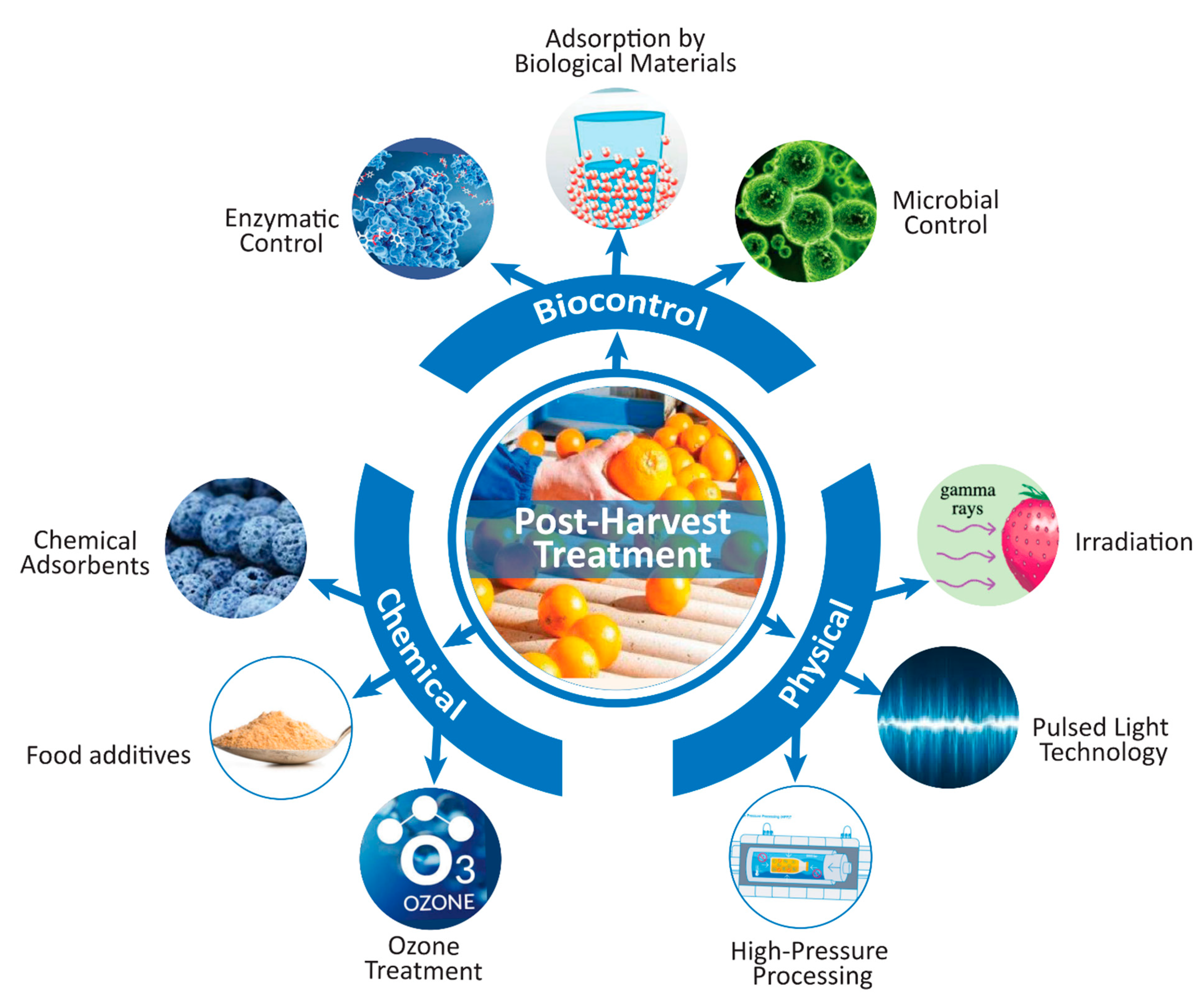
5. Mitigation Policies of Mycotoxin Contamination in Beverages
5.1. Physical Control Methods
5.1.1. Irradiation
5.1.2. Thermal Treatment
5.1.3. High-Pressure Processing
5.1.4. Pulsed Light Technology
5.2. Chemical Control Methods
5.2.1. Ozone Treatment
5.2.2. Use of Chemical Adsorbents
5.2.3. Control by Food Additives
5.3. Biological Control Methods
5.3.1. Microbiological Control
5.3.2. Antifungal Biomolecules
5.3.3. Enzymatic Control
5.3.4. Adsorption by Biological Materials
6. Critical Challenges of Mycotoxins in Beverages
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Greeff-Laubscher, M.R.; Beukes, I.; Marais, G.J.; Jacobs, K. Mycotoxin production by three different toxigenic fungi genera on formulated abalone feed and the effect of an aquatic environment on fumonisins. Mycology 2020, 11, 105–117. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jard, G.; Liboz, T.; Mathieu, F.; Guyonvarc’h, A.; Lebrihi, A. Review of mycotoxin reduction in food and feed: From prevention in the field to detoxification by adsorption or transformation. Food Addit. Contam. Part A 2011, 28, 1590–1609. [Google Scholar] [CrossRef]
- Gacem, M.A.; Gacem, H.; Telli, A.; Khelil, A.O.E.H. Mycotoxins: Decontamination and nanocontrol methods. In Nanomycotoxicology; Rai, M., Abd-Elsalam, K.A., Eds.; Academic Press: Cambridge, MA, USA, 2020; pp. 189–216. [Google Scholar]
- Amuzie, C.; Bandyopadhyay, R.; Bhat, R.; Black, R.; Burger, H.-M.; Cardwell, K.; Gelderblom, W.; Gong, Y.Y.; Groopman, J.; Kimanya, M.; et al. Effects of aflatoxins on aflatoxicosis and liver cancer. In Mycotoxin Control in Low- and Middle-Income Countries; International Agency for Research on Cancer: Lyon, France, 2015; Volume 9, pp. 13–16. [Google Scholar]
- WHO. Evaluation of Certain Contaminants in Food: Eighty-Third Report of the Joint FAO/WHO Expert Committee on Food Additives; World Health Organization: Geneva, Switzerland, 2017. [Google Scholar]
- Ostry, V.; Malir, F.; Toman, J.; Grosse, Y. Mycotoxins as human carcinogens-the IARC monographs classification. Mycotoxin Res. 2017, 33, 65–73. [Google Scholar] [CrossRef] [PubMed]
- Ashiq, S. Natural occurrence of mycotoxins in food and feed: Pakistan perspective. Compr. Rev. Food Sci. Food Saf. 2015, 14, 159–175. [Google Scholar] [CrossRef] [PubMed]
- Eskola, M.; Kos, G.; Elliott, C.T.; Hajšlová, J.; Mayar, S.; Krska, R. Worldwide contamination of food-crops with mycotoxins: Validity of the widely cited ‘FAO estimate’ of 25%. Crit. Rev. Food Sci. Nutr. 2019, 60, 2773–2789. [Google Scholar] [CrossRef] [PubMed]
- Granato, D.; Branco, G.F.; Nazzaro, F.; Cruz, A.G.; Faria, J.A.F. Functional foods and nondairy probiotic food development: Trends, concepts, and products. Compr. Rev. Food Sci. Food Saf. 2010, 9, 292–302. [Google Scholar] [CrossRef]
- Wan, J.; Chen, B.; Rao, J. Occurrence and preventive strategies to control mycotoxins in cereal-based food. Compr. Rev. Food Sci. Food Saf. 2020, 19, 928–953. [Google Scholar] [CrossRef] [Green Version]
- USFDA. Guidance for Industry: Action Levels for Poisonous or Deleterious Substances in Human Food and Animal Feed; USFDA: Silver Spring, MD, USA, 2000.
- European Commission. Regulation of the European Parliament and of the council of 29.4.2004 on the hygiene of food stuffs. Off. J. Eur. Union 2004, 852, 139–154. [Google Scholar]
- Battilani, P.; Palumbo, R.; Giorni, P.; Dall’Asta, C.; Dellafiora, L.; Gkrillas, A.; Toscano, P.; Crisci, A.; Brera, C.; De Santis, B.; et al. Mycotoxin mixtures in food and feed: Holistic, innovative, flexible risk assessment modelling approach: MYCHIF. EFSA Supporting Publ. 2020, 17, 161. [Google Scholar] [CrossRef] [Green Version]
- Oteiza, J.M.; Khaneghah, A.M.; Campagnollo, F.B.; Granato, D.; Mahmoudi, M.R.; Sant’Ana, A.S.; Gianuzzi, L. Influence of production on the presence of patulin and ochratoxin A in fruit juices and wines of Argentina. LWT Food Sci. Technol. 2017, 80, 200–207. [Google Scholar] [CrossRef]
- Bui-Klimke, T.R.; Wu, F. Ochratoxin A and human health risk: A review of the evidence. Crit. Rev. Food Sci. Nutr. 2015, 55, 1860–1869. [Google Scholar] [CrossRef] [Green Version]
- Malir, F.; Ostry, V.; Pfohl-Leszkowicz, A.; Malir, J.; Toman, J. Ochratoxin A: 50 Years of Research. Toxins 2016, 8, 191. [Google Scholar] [CrossRef] [Green Version]
- el Khoury, A.; Atoui, A. Ochratoxin A: General overview and actual molecular status. Toxins 2010, 2, 461–493. [Google Scholar] [CrossRef] [Green Version]
- Freire, L.; Braga, P.A.C.; Furtado, M.M.; Delafiori, J.; Dias-Audibert, F.L.; Pereira, G.E.; Reyes, F.G.; Catharino, R.R.; Sant’Ana, A.S. From grape to wine: Fate of ochratoxin A during red, rose, and white winemaking process and the presence of ochratoxin derivatives in the final products. Food Control 2020, 113, 107167. [Google Scholar] [CrossRef]
- Kumar, P.; Mahato, D.K.; Kamle, M.; Mohanta, T.K.; Kang, S.G. Aflatoxins: A global concern for food safety, human health and their management. Front. Microbiol. 2017, 7, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Probst, C.; Bandyopadhyay, R.; Cotty, P.J. Diversity of aflatoxin-producing fungi and their impact on food safety in sub-Saharan Africa. Int. J. Food Microbiol. 2014, 174, 113–122. [Google Scholar] [CrossRef]
- Freire, L.; Guerreiro, T.M.; Pia, A.K.R.; Lima, E.O.; Oliveira, D.N.; Melo, C.F.O.R.; Catharino, R.R.; Sant’Ana, A.S. A quantitative study on growth variability and production of ochratoxin A and its derivatives by A. carbonarius and A. niger in grape-based medium. Sci. Rep. 2018, 8, 14573. [Google Scholar] [CrossRef]
- Erdoğan, A.; Ghimire, D.; Gürses, M.; Çetin, B.; Baran, A. Patulin contamination in fruit juices and its control measures. Eur. J. Sci. Technol. 2018, 39–48. [Google Scholar] [CrossRef]
- Zhong, L.; Carere, J.; Lu, Z.; Lu, F.; Zhou, T. Patulin in apples and apple-based food products: The burdens and the mitigation strategies. Toxins 2018, 10, 475. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rheeder, J.P.; Marasas, W.F.; Vismer, H.F. Production of fumonisin analogs by Fusarium species. Appl. Environ. Microbiol. 2002, 68, 2101–2105. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pascari, X.; Ramos, A.J.; Marín, S.; Sanchís, V. Mycotoxins and beer. Impact of beer production process on mycotoxin contamination. A review. Food Res. Int. 2018, 103, 121–129. [Google Scholar]
- Liu, Y.; Galani Yamdeu, J.H.; Gong, Y.Y.; Orfila, C. A review of postharvest approaches to reduce fungal and mycotoxin contamination of foods. Compr. Rev. Food Sci. Food Saf. 2020, 19, 1521–1560. [Google Scholar] [CrossRef]
- Kuzdraliński, A.; Solarska, E.; Muszyńska, M. Deoxynivalenol and zearalenone occurence in beers analysed by an enzyme-linked immunosorbent assay method. Food Control 2013, 29, 22–24. [Google Scholar] [CrossRef]
- Miró-Abella, E.; Herrero, P.; Canela, N.; Arola, L.; Borrull, F.; Ras, R.; Fontanals, N. Determination of mycotoxins in plant-based beverages using QuEChERs and liquid chromatography-tandem mass spectrometry. Food Chem. 2017, 229, 336–372. [Google Scholar] [CrossRef] [PubMed]
- Hamed, A.M.; Arroyo-Manzanares, N.; García-Campaña, A.M.; Gámiz-Gracia, L. Determination of Fusarium toxins in functional vegetable milks applying salting-out-assisted liquid–liquid extraction combined with ultra-high-performance liquid chromatography tandem mass spectrometry. Food Addit. Contam. Part A 2017, 34, 2033–2041. [Google Scholar] [CrossRef]
- Agriopoulou, S.; Stamatelopoulou, E.; Varzakas, T. Advances in occurrence, importance, and mycotoxin control strategies: Prevention and detoxification in foods. Foods. 2020, 9, 137. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Chen, X.; Han, S.Y.; Li, M.; Ma, T.Z.; Sheng, W.J.; Zhu, X. Simultaneous analysis of 20 mycotoxins in grapes and wines from Hexi corridor region (China): Based on a QuEChERS–UHPLC–MS/MS method. Molecules 2018, 23, 1926. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meena, M.; Swapnil, P.; Upadhyay, R.S. Isolation, characterization and toxicological potential of Alternaria-mycotoxins (TeA, AOH and AME) in different Alternaria species from various regions of India. Sci. Rep. 2017, 7, 8777. [Google Scholar] [CrossRef] [Green Version]
- Meena, M.; Gupta, S.K.; Swapnil, P.; Zehra, A.; Dubey, M.K.; Upadhyay, R.S. Alternaria toxins: Potential virulence factors and genes related to pathogenesis. Front. Microbiol. 2017, 8, 1451. [Google Scholar] [CrossRef]
- Ngea, G.L.N.; Yang, Q.; Castoria, R.; Zhang, X.; Routledge, M.N.; Zhang, H. Recent trends in detecting, controlling, and detoxifying of patulin mycotoxin using biotechnology methods. Compr. Rev. Food Sci. Food Saf. 2020, 19, 2447–2472. [Google Scholar] [CrossRef]
- Iqbal, Q.; Amjad, M.; Asi, M.R.; AriÑO, A. Mold and aflatoxin reduction by gamma radiation of packed hot peppers and their evolution during storage. J. Food Prot. 2012, 75, 1528–1531. [Google Scholar] [CrossRef] [PubMed]
- Saleh, I.; Goktepe, I. The characteristics, occurrence, and toxicological effects of patulin. Food Chem. Toxicol. 2019, 129, 301–311. [Google Scholar] [CrossRef] [PubMed]
- Solairaj, D.; Guillaume Legrand, N.N.; Yang, Q.; Zhang, H. Isolation of pathogenic fungi causing postharvest decay in table grapes and in vivo biocontrol activity of selected yeasts against them. Physiol. Mol. Plant Pathol. 2020, 110, 101478. [Google Scholar] [CrossRef]
- Zouaoui, N.; Sbaii, N.; Bacha, H.; Abid-Essefi, S. Occurrence of patulin in various fruit juice marketed in Tunisia. Food Control 2015, 51, 356–360. [Google Scholar] [CrossRef]
- Ioi, J.D.; Zhou, T.; Tsao, R.; F Marcone, M. Mitigation of patulin in fresh and processed foods and beverages. Toxins 2017, 9, 157. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sadok, I.; Stachniuk, A.; Staniszewska, M. Developments in the monitoring of patulin in fruits using liquid chromatography: An overview. Food Anal. Methods 2019, 12, 76–93. [Google Scholar] [CrossRef]
- Sant’Ana, A.d.S.; Rosenthal, A.; de Massaguer, P.R. The fate of patulin in apple juice processing: A review. Food Res. Int. 2008, 41, 441–453. [Google Scholar] [CrossRef] [Green Version]
- Ramalingam, S.; Bahuguna, A.; Kim, M. The effects of mycotoxin patulin on cells and cellular components. Trends Food Sci. Technol. 2019, 83, 99–113. [Google Scholar] [CrossRef]
- Voss, K.A.; Smith, G.W.; Haschek, W.M. Fumonisins: Toxicokinetics, mechanism of action and toxicity. Anim. Feed Sci. Technol. 2007, 137, 299–325. [Google Scholar] [CrossRef]
- EFSA. Opinion of the Scientific Panel on contaminants in the food chain [CONTAM] related to fumonisins as undesirable substances in animal feed. EFSA J. 2005, 3, 235. [Google Scholar] [CrossRef]
- Wu, F.; Groopman, J.D.; Pestka, J.J. Public health impacts of foodborne mycotoxins. Annu. Rev. Food Sci. Technol. 2014, 5, 351–372. [Google Scholar] [CrossRef] [Green Version]
- Nesic, K.; Ivanovic, S.; Nesic, V. Fusarial Toxins: Secondary Metabolites of Fusarium Fungi. In Reviews of Environmental Contamination and Toxicology; Whitacre, D.M., Ed.; Springer International Publishing: Cham, Swithzerland, 2014; Volume 228, pp. 101–120. [Google Scholar]
- Marroquín-Cardona, A.G.; Johnson, N.M.; Phillips, T.D.; Hayes, A.W. Mycotoxins in a changing global environment—A review. Food Chem. Toxicol. 2014, 69, 220–230. [Google Scholar] [CrossRef]
- Piacentini, K.C.; Rocha, L.O.; Fontes, L.C.; Carnielli, L.; Reis, T.A.; Corrêa, B. Mycotoxin analysis of industrial beers from Brazil: The influence of fumonisin B(1) and deoxynivalenol in beer quality. Food Chem. 2017, 218, 64–69. [Google Scholar] [CrossRef]
- Udovicki, B.; Audenaert, K.; De Saeger, S.; Rajkovic, A. Overview on the mycotoxins incidence in Serbia in the period 2004–2016. Toxins 2018, 10, 279. [Google Scholar] [CrossRef] [Green Version]
- Ünüsan, N. Systematic review of mycotoxins in food and feeds in Turkey. Food Control 2019, 97, 1–14. [Google Scholar] [CrossRef]
- Ferrigo, D.; Raiola, A.; Causin, R. Fusarium toxins in cereals: Occurrence, legislation, factors promoting the appearance and their management. Molecules 2016, 21, 627. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Krska, R.; Malachova, A.; Berthiller, F.; van Egmond, H.P. Determination of T-2 and HT-2 toxins in food and feed: An update. World Mycotoxin J. 2014, 7, 131–142. [Google Scholar] [CrossRef]
- European Food Safety Authority. Scientific Opinion on the risks for animal and public health related to the presence of T-2 and HT-2 toxin in food and feed. EFSA J. 2011, 9, 2481. [Google Scholar] [CrossRef]
- Arroyo-Manzanares, N.; Hamed, A.M.; García-Campaña, A.M.; Gámiz-Gracia, L. Plant-based milks: Unexplored source of emerging mycotoxins. A proposal for the control of enniatins and beauvericin using UHPLC-MS/MS. Food Addit. Contam. Part B 2019, 12, 296–302. [Google Scholar] [CrossRef] [PubMed]
- Joint Food and Agriculture Organization; World Health Organization Expert Committee on Food Additives (JECFA). Evaluation of Certain Food Additives and Contaminants: Fifty-Fifth Report of the JOINT/FAO/WHO Expert Committee on Food Additives; World Health Organization: Geneva, Switzerland, 2001; p. 701. [Google Scholar]
- Nolan, P.; Auer, S.; Spehar, A.; Elliott, C.T.; Campbell, K. Current trends in rapid tests for mycotoxins. Food Addit. Contam. Part A 2019, 36, 800–814. [Google Scholar] [CrossRef] [Green Version]
- Bottalico, A.; Perrone, G. Toxigenic Fusarium species and mycotoxins associated with head blight in small-grain cereals in Europe. Eur. J. Plant Pathol. 2002, 108, 611–624. [Google Scholar] [CrossRef]
- Sobrova, P.; Adam, V.; Vasatkova, A.; Beklova, M.; Zeman, L.; Kizek, R. Deoxynivalenol and its toxicity. Interdiscip. Toxicol. 2010, 3, 94–99. [Google Scholar] [CrossRef]
- EFSA. Deoxynivalenol in food and feed: Occurrence and exposure. EFSA J. 2013, 11, 3379. [Google Scholar]
- Mishra, S.; Srivastava, S.; Dewangan, J.; Divakar, A.; Kumar Rath, S. Global occurrence of deoxynivalenol in food commodities and exposure risk assessment in humans in the last decade: A survey. Crit. Rev. Food. Sci. Nutr. 2020, 60, 1346–1374. [Google Scholar] [CrossRef]
- Piacentini, K.C.; Savi, G.D.; Olivo, G.; Scussel, V.M. Quality and occurrence of deoxynivalenol and fumonisins in craft beer. Food Control 2015, 50, 925–929. [Google Scholar] [CrossRef] [Green Version]
- EFSA. Risks to human and animal health related to the presence of deoxynivalenol and its acetylated and modified forms in food and feed. EFSA J. 2017, 15, e04718. [Google Scholar]
- Rai, A.; Das, M.; Tripathi, A. Occurrence and toxicity of a Fusarium mycotoxin, zearalenone. Crit. Rev. Food. Sci. Nutr. 2020, 60, 2710–2729. [Google Scholar] [CrossRef]
- Calori-Domingues, M.A.; Bernardi, C.M.; Nardin, M.S.; de Souza, G.V.; Dos Santos, F.G.; Stein Mde, A.; Gloria, E.M.; Dias, C.T.; de Camargo, A.C. Co-occurrence and distribution of deoxynivalenol, nivalenol and zearalenone in wheat from Brazil. Food Addit. Contam. Part B Surveill. 2016, 9, 142–151. [Google Scholar] [CrossRef]
- Tola, M.; Kebede, B. Occurrence, importance and control of mycotoxins: A review. Cogent Food Agric. 2016, 2, 1191103. [Google Scholar] [CrossRef]
- Rodriguez-Carrasco, Y.; Fattore, M.; Albrizio, S.; Berrada, H.; Manes, J. Occurrence of Fusarium mycotoxins and their dietary intake through beer consumption by the European population. Food Chem. 2015, 178, 149–155. [Google Scholar] [CrossRef]
- Commission, E. Commission Regulation (EC) No. 1881/2006 of 19 December 2006. Setting maximum levels for certain contaminants in food stuffs. Off. J. Eur. Union 2006, 364, 5–24. [Google Scholar]
- Yvv, A.K.; Renuka, R.M.; Bodaiah, B.; Mangamu, U.K.; Vijayalakshmi, M.; Poda, S. Mycotoxin strategies: Impact on global health and wealth. Pharm. Anal.Acta 2016, 7, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Scott, P.M.; Lawrence, G.A.; Lau, B.P.Y. Analysis of wines, grape juices and cranberry juices for Alternaria toxins. Mycotoxin Res. 2006, 22, 142–147. [Google Scholar] [CrossRef] [PubMed]
- Escrivá, L.; Oueslati, S.; Font, G.; Manyes, L. Alternaria mycotoxins in food and feed: An overview. J. Food Qual. 2017, 2017, 1–20. [Google Scholar] [CrossRef] [Green Version]
- Ostry, V. Alternaria mycotoxins: An overview of chemical characterization, producers, toxicity, analysis and occurrence in foodstuffs. World Mycotoxin J. 2008, 1, 175–188. [Google Scholar] [CrossRef]
- Víctor-Ortega, M.D.; Lara, F.J.; García-Campaña, A.M.; del Olmo-Iruela, M. Evaluation of dispersive liquid–liquid microextraction for the determination of patulin in apple juices using micellar electrokinetic capillary chromatography. Food Control 2013, 31, 353–358. [Google Scholar] [CrossRef]
- WHO. World Health Organization, 44th Report of the Joint FAO/WHO Expert Committee On Food Additives; WHO: Geneva, Switzerland, 1995; p. 36. [Google Scholar]
- Gonçalves, B.L.; Coppa, C.F.S.C.; de Neeff, D.V.; Corassin, C.H.; Oliveira, C.A.F. Mycotoxins in fruits and fruit-based products: Occurrence and methods for decontamination. Toxin Rev. 2019, 38, 263–272. [Google Scholar] [CrossRef]
- Singh, J.; Mehta, A. Rapid and sensitive detection of mycotoxins by advanced and emerging analytical methods: A review. Food Sci. Nutr. 2020, 8, 2183–2204. [Google Scholar] [CrossRef]
- Batrinou, A.; Houhoula, D.; Papageorgiou, E. Rapid detection of mycotoxins on foods and beverages with enzyme-linked immunosorbent assay. Qual. Assur. Saf. Crop. Foods 2020, 12, 40–49. [Google Scholar] [CrossRef] [Green Version]
- Welke, J.E.; Hoeltz, M.; Dottori, H.A.; Noll, I.B. Quantitative analysis of patulin in apple juice by thin-layer chromatography using a charge coupled device detector. Food Addit. Contam. 2009, 26, 754–758. [Google Scholar] [CrossRef]
- De Girolamo, A.; McKeague, M.; Miller, J.D.; DeRosa, M.C.; Visconti, A. Determination of ochratoxin A in wheat after clean-up through a DNA aptamer-based solid-phase extraction column. Food Chem. 2011, 127, 1378–1384. [Google Scholar] [CrossRef] [PubMed]
- Campone, L.; Piccinelli, A.L.; Rastrelli, L. Dispersive liquid–liquid microextraction combined with high-performance liquid chromatography–tandem mass spectrometry for the identification and the accurate quantification by isotope dilution assay of Ochratoxin A in wine samples. Anal. Bioanal. Chem. 2011, 399, 1279–1286. [Google Scholar] [CrossRef] [PubMed]
- Tessini, C.; Mardones, C.; von Baer, D.; Vega, M.; Herlitz, E.; Saelzer, R.; Silva, J.; Torres, O. Alternatives for sample pre-treatment and HPLC determination of ochratoxin A in red wine using fluorescence detection. Anal. Chim. Acta 2010, 660, 119–126. [Google Scholar] [CrossRef]
- Zahn, M.; Jeong, M.L.; Wang, D.; Trinh, T.; Fay, B.; Ma, W. Product-specific sample clean-up and HPLC analysis of aflatoxins for a dietary product. Phytochem. Anal. 2009, 20, 335–337. [Google Scholar] [CrossRef]
- Jiang, W.; Wang, Z.; Nölke, G.; Zhang, J.; Niu, L.; Shen, J. Simultaneous determination of aflatoxin B1 and aflatoxin M1 in food matrices by enzyme-linked immunosorbent assay. Food Anal. Methods 2013, 6, 767–774. [Google Scholar] [CrossRef]
- Aresta, A.; Vatinno, R.; Palmisano, F.; Zambonin, C.G. Determination of Ochratoxin A in wine at sub ng/mL levels by solid-phase microextraction coupled to liquid chromatography with fluorescence detection. J. Chromatogr. A 2006, 1115, 196–201. [Google Scholar] [CrossRef] [PubMed]
- MacDonald, S.J.; Anderson, S.; Brereton, P.; Wood, R.; Damant, A. Determination of zearalenone in barley, maize and wheat flour, polenta, and maize-based baby food by immunoaffinity column cleanup with liquid chromatography: Interlaboratory study. J. AOAC Int. 2005, 88, 1733–1740. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brera, C.; Debegnach, F.; Minardi, V.; Pannunzi, E.; Santis, B.D.; Miraglia, M. Immunoaffinity column cleanup with liquid chromatography for determination of aflatoxin B1 in corn samples: Interlaboratory study. J. AOAC Int. 2007, 90, 765–772. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Santini, A.; Ferracane, R.; Somma, M.C.; Aragón, A.; Ritieni, A. Multitoxin extraction and detection of trichothecenes in cereals: An improved LC-MS/MS approach. J. Sci. Food Agric. 2009, 89, 1145–1153. [Google Scholar] [CrossRef]
- Sauceda-Friebe, J.C.; Karsunke, X.Y.; Vazac, S.; Biselli, S.; Niessner, R.; Knopp, D. Regenerable immuno-biochip for screening ochratoxin A in green coffee extract using an automated microarray chip reader with chemiluminescence detection. Anal. Chim. Acta 2011, 689, 234–242. [Google Scholar] [CrossRef] [PubMed]
- Chun, H.S.; Choi, E.H.; Chang, H.-J.; Choi, S.-W.; Eremin, S.A. A fluorescence polarization immunoassay for the detection of zearalenone in corn. Anal. Chim. Acta 2009, 639, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Ren, M.; Xu, H.; Huang, X.; Kuang, M.; Xiong, Y.; Xu, H.; Xu, Y.; Chen, H.; Wang, A. Immunochromatographic assay for ultrasensitive detection of aflatoxin B1 in maize by highly luminescent quantum dot beads. ACS Appl. Mater. Interfaces 2014, 6, 14215–14222. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yuan, J.; Deng, D.; Lauren, D.R.; Aguilar, M.-I.; Wu, Y. Surface plasmon resonance biosensor for the detection of ochratoxin A in cereals and beverages. Anal. Chim. Acta 2009, 656, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.-K.; Wang, Y.-C.; Wang, H.-A.; Ji, W.-H.; Sun, J.-H.; Yan, Y.-X. An immunomagnetic-bead-based enzyme-linked immunosorbent assay for sensitive quantification of fumonisin B1. Food Control 2014, 40, 41–45. [Google Scholar] [CrossRef]
- Jodra, A.; López, M.Á.; Escarpa, A. Disposable and reliable electrochemical magnetoimmunosensor for Fumonisins simplified determination in maize-based foodstuffs. Biosens. Bioelectron. 2015, 64, 633–638. [Google Scholar] [CrossRef] [PubMed]
- Molinelli, A.; Grossalber, K.; Krska, R. A rapid lateral flow test for the determination of total type B fumonisins in maize. Anal. Bioanal. Chem. 2009, 395, 1309–1316. [Google Scholar] [CrossRef]
- Funari, R.; Della Ventura, B.; Carrieri, R.; Morra, L.; Lahoz, E.; Gesuele, F.; Altucci, C.; Velotta, R. Detection of parathion and patulin by quartz-crystal microbalance functionalized by the photonics immobilization technique. Biosens. Bioelectron. 2015, 67, 224–229. [Google Scholar] [CrossRef] [Green Version]
- Ko, J.; Lee, C.; Choo, J. Highly sensitive SERS-based immunoassay of aflatoxin B1 using silica-encapsulated hollow gold nanoparticles. J. Hazard. Mater. 2015, 285, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Radoi, A.; Targa, M.; Prieto-Simon, B.; Marty, J.-L. Enzyme-Linked Immunosorbent Assay (ELISA) based on superparamagnetic nanoparticles for aflatoxin M1 detection. Talanta 2008, 77, 138–143. [Google Scholar] [CrossRef]
- Tang, X.; Li, X.; Li, P.; Zhang, Q.; Li, R.; Zhang, W.; Ding, X.; Lei, J.; Zhang, Z. Development and application of an immunoaffinity column enzyme immunoassay for mycotoxin zearalenone in complicated samples. PLoS ONE 2014, 9, e85606. [Google Scholar] [CrossRef]
- Abdul Kadir, M.K.; Tothill, I.E. Development of an electrochemical immunosensor for fumonisins detection in foods. Toxins 2010, 2, 382–398. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schultz, C. Automating mycotoxin analysis in beverages. Column 2017, 13, 23–26. [Google Scholar]
- Zhang, K. Evaluation of automated sample preparation for mycotoxin analysis in foods. J. AOAC Int. 2020, 103, 1052–1059. [Google Scholar] [CrossRef] [PubMed]
- de Souza, G.D.; Mithofer, A.; Daolio, C.; Schneider, B.; Rodrigues-Filho, E. Identification of Alternaria alternata mycotoxins by LC-SPE-NMR and their cytotoxic effects to soybean (Glycine max) cell suspension culture. Molecules 2013, 18, 2528–2538. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tamura, M.; Uyama, A.; Mochizuki, N. Development of a multi-mycotoxin analysis in beer-based drinks by a modified QuEChERS method and ultra-high-performance liquid chromatography coupled with tandem mass spectrometry. Anal. Sci. 2011, 27, 629–635. [Google Scholar] [CrossRef] [Green Version]
- Tamura, M.; Takahashi, A.; Uyama, A.; Mochizuki, N. A method for multiple mycotoxin analysis in wines by solid-phase extraction and multifunctional cartridge purification, and ultra-high-performance liquid chromatography coupled to tandem mass spectrometry. Toxins 2012, 4, 476–486. [Google Scholar] [CrossRef]
- Rotariu, L.; Lagarde, F.; Jaffrezic-Renault, N.; Bala, C. Electrochemical biosensors for fast detection of food contaminants—Trends and perspective. Trends Analyt. Chem. 2016, 79, 80–87. [Google Scholar] [CrossRef]
- Joshi, S.; Annida, R.M.; Zuilhof, H.; Beek, T.A.V.; Nielen, M.W.F. Analysis of mycotoxins in beer using a portable nanostructured imaging surface plasmon resonance biosensor. J. Agric. Food Chem. 2016, 64, 8263–8271. [Google Scholar] [CrossRef]
- Pennacchio, A.; Ruggiero, G.; Staiano, M.; Piccialli, G.; Oliviero, G.; Lewkowicz, A.; Synak, A.; Bojarski, P.; D’Auria, S. A surface plasmon resonance based biochip for the detection of patulin toxin. Opt. Mater. 2014, 36, 1670–1675. [Google Scholar] [CrossRef]
- Pennacchio, A.; Varriale, A.; Esposito, M.G.; Scala, A.; Marzullo, V.M.; Staiano, M.; D’Auria, S. A Rapid and sensitive assay for the detection of Benzylpenicillin (PenG) in Milk. PLoS ONE 2015, 10, e0132396. [Google Scholar] [CrossRef]
- Soldatkin, O.O.; Stepurska, K.V.; Arkhypova, V.M.; Soldatkin, A.P.; El’skaya, A.V.; Lagarde, F.; Dzyadevych, S.V. Conductometric enzyme biosensor for patulin determination. Sens. Actuators B Chem. 2017, 239, 1010–1015. [Google Scholar] [CrossRef]
- Bauer, J.I.; Gross, M.; Hamscher, G.; Usleber, E. A rapid screening method for the tremorgenic indole-diterpene alkaloid mycotoxin paxilline in beer. Food Anal. Methods 2017, 11, 1051–1055. [Google Scholar] [CrossRef]
- Man, Y.; Liang, G.; Li, A.; Pan, L. Recent advances in mycotoxin determination for food monitoring via microchip. Toxins 2017, 9, 324. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shin, Y.; Perera, A.P.; Wong, C.C.; Park, M.K. Solid phase nucleic acid extraction technique in a microfluidic chip using a novel non-chaotropic agent: Dimethyl adipimidate. Lab Chip 2014, 14, 359–368. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Chen, J.; Zhou, T.; Song, L. Fabricating microstructures on glass for microfluidic chips by glass molding process. Micromachines 2018, 9, 269. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, X.; Liu, C.; Xu, Z.; Pan, Y.; Liu, J.; Du, L. An effective PDMS microfluidic chip for chemiluminescence detection of cobalt (II) in water. Microsyst. Technol. 2013, 19, 99–103. [Google Scholar] [CrossRef]
- Xuefeng, Y.; Hong, S.; Zhaolun, F. A simplified microfabrication technology for production of glass microfluidic chips. Chin. J. Anal. Chem. 2003, 31, 116–119. [Google Scholar]
- Claeys, L.; Romano, C.; De Ruyck, K.; Wilson, H.; Fervers, B.; Korenjak, M.; Zavadil, J.; Gunter, M.J.; De Saeger, S.; De Boevre, M.; et al. Mycotoxin exposure and human cancer risk: A systematic review of epidemiological studies. Compr. Rev. Food Sci. Food Saf. 2020, 19, 1449–1464. [Google Scholar] [CrossRef]
- Šarkanj, B.; Ezekiel, C.N.; Turner, P.C.; Abia, W.A.; Rychlik, M.; Krska, R.; Sulyok, M.; Warth, B. Ultra-sensitive, stable isotope assisted quantification of multiple urinary mycotoxin exposure biomarkers. Anal. Chim. Acta 2018, 1019, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Lauwers, M.; Croubels, S.; De Baere, S.; Sevastiyanova, M.; Romera Sierra, E.M.; Letor, B.; Gougoulias, C.; Devreese, M. Assessment of dried blood spots for multi-mycotoxin biomarker analysis in pigs and broiler chickens. Toxins 2019, 11, 541. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rong, X.; Sun-Waterhouse, D.; Wang, D.; Jiang, Y.; Li, F.; Chen, Y.; Zhao, S.; Li, D. The significance of regulatory microRNAs: Their roles in toxicodynamics of mycotoxins and in the protection offered by dietary therapeutics against mycotoxin-induced toxicity. Compr. Rev. Food Sci. Food Saf. 2019, 18, 48–66. [Google Scholar] [CrossRef] [Green Version]
- Rhouati, A.; Bulbul, G.; Latif, U.; Hayat, A.; Li, Z.-H.; Marty, J.L. Nano-aptasensing in mycotoxin analysis: Recent updates and progress. Toxins 2017, 9, 349. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Horky, P.; Skalickova, S.; Baholet, D.; Skladanka, J. Nanoparticles as a solution for eliminating the risk of mycotoxins. Nanomaterials 2018, 8, 727. [Google Scholar] [CrossRef] [Green Version]
- Cavaliere, C.; D’Ascenzo, G.; Foglia, P.; Pastorini, E.; Samperi, R.; Lagana, A. Determination of type B trichothecenes and macrocyclic lactone mycotoxins in field contaminated maize. Food Chem. 2005, 92, 559–568. [Google Scholar] [CrossRef]
- Reddy, K.R.N.; Reddy, C.S.; Muralidharan, K. Detection of Aspergillus spp. and aflatoxin B1 in rice in India. Food Microbiol. 2009, 26, 27–31. [Google Scholar] [CrossRef] [PubMed]
- Torres, M.R.; Sanchis, V.; Ramos, A.J. Occurrence of fumonisins in Spanish beers analyzed by an enzyme-linked immunosorbent assay method. Int. J. Food Microbiol. 1998, 39, 139–143. [Google Scholar] [CrossRef]
- Zheng, Z.; Humphrey, C.W.; King, R.S.; Richard, J.L. Validation of an ELISA test kit for the detection of total aflatoxins in grain and grain products by comparison with HPLC. Mycopathologia 2005, 159, 255–263. [Google Scholar] [CrossRef]
- Omar, S.S.; Haddad, M.A.; Parisi, S. Validation of HPLC and Enzyme-Linked Immunosorbent Assay (ELISA) techniques for detection and quantification of aflatoxins in different food samples. Foods 2020, 9, 661. [Google Scholar] [CrossRef] [PubMed]
- Beyene, A.M.; Du, X.; Schrunk, D.E.; Ensley, S.; Rumbeiha, W.K. High-performance liquid chromatography and Enzyme-Linked Immunosorbent Assay techniques for detection and quantification of aflatoxin B1 in feed samples: A comparative study. BMC Res. Notes 2019, 12, 492. [Google Scholar] [CrossRef]
- Vanhoutte, I.; Audenaert, K.; De Gelder, L. Biodegradation of mycotoxins: Tales from known and unexplored worlds. Front. Microbiol. 2016, 7, 561. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Adegoke, G.O.; Letuma, P. Strategies for the prevention and reduction of mycotoxins in developing countries. In Mycotoxin and Food Safety in Developing Countries; Intechopen: London, UK, 2013; pp. 123–136. [Google Scholar]
- Peng, W.X.; Marchal, J.L.M.; van der Poel, A.F.B. Strategies to prevent and reduce mycotoxins for compound feed manufacturing. Anim. Feed Sci. Technol. 2018, 237, 129–153. [Google Scholar] [CrossRef]
- Fernández-Cruz, M.L.; Mansilla, M.L.; Tadeo, J.L. Mycotoxins in fruits and their processed products: Analysis, occurrence and health implications. J. Adv. Res. 2010, 1, 113–122. [Google Scholar] [CrossRef] [Green Version]
- Apaolaza, V.; Hartmann, P.; D’Souza, C.; López, C.M. Eat organic—Feel good? The relationship between organic food consumption, health concern and subjective wellbeing. Food Qual. Prefer. 2018, 63, 51–62. [Google Scholar] [CrossRef]
- Rana, J.; Paul, J. Consumer behavior and purchase intention for organic food: A review and research agenda. J. Retail. Consum. Serv. 2017, 38, 157–165. [Google Scholar] [CrossRef]
- Do Amparo, H.C.; Cavichon, E.; Baratto, C.M.; Tondo, E.C.; Gelinski, J.M.L.N. Determination of patulin in apple juice from Fuji apples stored in different conditions in Southern Brazil. J. Food Res. 2012, 1, 54. [Google Scholar] [CrossRef] [Green Version]
- Diao, E.; Hou, H.; Hu, W.; Dong, H.; Li, X. Removing and detoxifying methods of patulin: A review. Trends Food Sci. Tech. 2018, 81, 139–145. [Google Scholar] [CrossRef]
- Jalili, M.; Jinap, S.; Noranizan, M.A. Aflatoxins and ochratoxin a reduction in black and white pepper by gamma radiation. Radiat. Phys. Chem. 2012, 81, 1786–1788. [Google Scholar] [CrossRef]
- Kumar, S.; Kunwar, A.; Gautam, S.; Sharma, A. Inactivation of A. ochraceus spores and detoxification of ochratoxin A in coffee beans by gamma irradiation. J. Food Sci. 2012, 77, T44–T51. [Google Scholar] [CrossRef]
- Calado, T.; Venâncio, A.; Abrunhosa, L. Irradiation for mold and mycotoxin control: A review. Compr. Rev. Food Sci. Food Saf. 2014, 13, 1049–1061. [Google Scholar] [CrossRef] [Green Version]
- Jeong, R.D.; Chu, E.H.; Lee, G.W.; Cho, C.; Park, H.J. Inhibitory effect of gamma irradiation and its application for control of postharvest green mold decay of Satsuma mandarins. Int. J. Food Microbial. 2016, 234, 1–8. [Google Scholar] [CrossRef]
- Yoon, M.; Jung, K.; Lee, K.-Y.; Jeong, J.-Y.; Lee, J.-W.; Park, H.-J. Synergistic effect of the combined treatment with gamma irradiation and sodium dichloroisocyanurate to control gray mold (Botrytis cinerea) on paprika. Radiat. Phys. Chem. 2014, 98, 103–108. [Google Scholar] [CrossRef]
- Zhu, Y.; Koutchma, T.; Warriner, K.; Zhou, T. Reduction of patulin in apple juice products by UV light of different wavelengths in the UVC range. J. Food Prot. 2014, 77, 963–971. [Google Scholar] [CrossRef] [PubMed]
- Assatarakul, K.; Churey, J.J.; Manns, D.C.; Worobo, R.W. Patulin reduction in apple juice from concentrate by UV radiation and comparison of kinetic degradation models between apple juice and apple cider. J. Food Prot. 2012, 75, 717–724. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Shukla, S.; Oh, Y.; Chung, S.H.; Kim, M. Comparative diminution of patulin content in apple juice with food-grade additives sodium bicarbonate, vinegar, mixture of sodium bicarbonate and vinegar, citric acid, baking powder, and ultraviolet irradiation. Front. Pharmacol. 2018, 9, 822. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kalagatur, N.K.; Kamasani, J.R.; Mudili, V. Assessment of detoxification efficacy of irradiation on zearalenone mycotoxin in various fruit juices by response surface methodology and elucidation of its in-vitro toxicity. Front. Microbiol. 2018, 9, 2937. [Google Scholar] [CrossRef] [Green Version]
- Hu, G.; Liu, H.; Zhu, Y.; Hernandez, M.; Koutchma, T.; Shao, S. Suppression of the formation of furan by antioxidants during UV-C light treatment of sugar solutions and apple cider. Food Chem. 2018, 269, 342–346. [Google Scholar] [CrossRef] [PubMed]
- Vidal, A.; Sanchis, V.; Ramos, A.J.; Marín, S. Thermal stability and kinetics of degradation of deoxynivalenol, deoxynivalenol conjugates and ochratoxin A during baking of wheat bakery products. Food Chem. 2015, 178, 276–286. [Google Scholar] [CrossRef] [Green Version]
- Milani, J.; Maleki, G. Effects of processing on mycotoxin stability in cereals. J. Sci. Food Agric. 2014, 94, 2372–2375. [Google Scholar] [CrossRef]
- Pinto, C.A.; Moreira, S.A.; Fidalgo, L.G.; Inácio, R.S.; Barba, F.J.; Saraiva, J.A. Effects of high-pressure processing on fungi spores: Factors affecting spore germination and inactivation and impact on ultrastructure. Compr. Rev. Food Sci. Food Saf. 2020, 19, 553–573. [Google Scholar] [CrossRef]
- Hao, H.; Zhou, T.; Koutchma, T.; Wu, F.; Warriner, K. High hydrostatic pressure assisted degradation of patulin in fruit and vegetable juice blends. Food Control 2016, 62, 237–242. [Google Scholar] [CrossRef]
- Avsaroglu, M.D.; Bozoglu, F.; Alpas, H.; Largeteau, A.; Demazeau, G. Use of pulsed-high hydrostatic pressure treatment to decrease patulin in apple juice. High Press. Res. 2015, 35, 214–222. [Google Scholar] [CrossRef]
- Syed, Q.A.; Ishaq, A.; Rahman, U.U.; Aslam, S.; Shukat, R. Pulsed electric field technology in food preservation: A Review. J. Nutri. Health Food Eng. 2017, 6, 1–5. [Google Scholar]
- Abuagela, M.O.; Iqdiam, B.M.; Mostafa, H.; Gu, L.; Smith, M.E.; Sarnoski, P.J. Assessing pulsed light treatment on the reduction of aflatoxins in peanuts with and without skin. Int. J. Food Sci. Technol. 2018, 53, 2567–2575. [Google Scholar] [CrossRef]
- Vijayalakshmi, S.; Nadanasabhapathi, S.; Kumar, R.; Sunny Kumar, S. Effect of pH and pulsed electric field process parameters on the aflatoxin reduction in model system using response surface methodology: Effect of pH and PEF on Aflatoxin Reduction. J. Food Sci. Technol. 2018, 55, 868–878. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Chen, P.; Cheng, Y.; Peng, P.; Liu, J.; Ma, Y.; Liu, Y.; Ruan, R. Deoxynivalenol Decontamination in Raw and Germinating Barley Treated by Plasma-Activated Water and Intense Pulsed Light. Food Bioproc. Tech. 2018, 12, 246–254. [Google Scholar] [CrossRef]
- Funes, G.J.; Gómez, P.L.; Resnik, S.L.; Alzamora, S.M. Application of pulsed light to patulin reduction in McIlvaine buffer and apple products. Food Control 2013, 30, 405–410. [Google Scholar] [CrossRef]
- Afsah-Hejri, L.; Hajeb, P.; Ehsani, R.J. Application of ozone for degradation of mycotoxins in food: A review. Compr. Rev. Food Sci. Food Saf. 2020, 19, 1777–1808. [Google Scholar] [CrossRef]
- Trombete, F.M.; Porto, Y.D.; Freitas-Silva, O.; Pereira, R.V.; Direito, G.M.; Saldanha, T.; Fraga, M.E. Efficacy of Ozone Treatment on Mycotoxins and Fungal Reduction in Artificially Contaminated Soft Wheat Grains. J. Food Process. Preserv. 2017, 41, e12927. [Google Scholar] [CrossRef]
- Pandiselvam, R.; Subhashini, S.; Banuu Priya, E.P.; Kothakota, A.; Ramesh, S.V.; Shahir, S. Ozone based food preservation: A promising green technology for enhanced food safety. Ozone Sci. Eng. 2019, 41, 17–34. [Google Scholar] [CrossRef]
- Torres, A.M.; Palacios, S.A.; Yerkovich, N.; Palazzini, J.M.; Battilani, P.; Leslie, J.F.; Logrieco, A.F.; Chulze, S.N. Fusarium head blight and mycotoxins in wheat: Prevention and control strategies across the food chain. World Mycotoxin J. 2019, 12, 333–355. [Google Scholar] [CrossRef]
- Diao, E.; Wang, J.; Li, X.; Wang, X.; Gao, D. Patulin degradation in apple juice using ozone detoxification equipment and its effects on quality. J. Food Process. Preserv. 2018, 42, e13645. [Google Scholar] [CrossRef]
- Appell, M.; Jackson, M.A.; Dombrink-Kurtzman, M.A. Removal of patulin from aqueous solutions by propylthiol functionalized SBA-15. J. Hazard. Mater. 2011, 187, 150–156. [Google Scholar] [CrossRef]
- Zhang, Z.; Zeng, C.; Peng, B. Adsorption properties of magnetic carbon nanotubes for patulin removal from aqueous solution systems. Food Control 2019, 102, 1–10. [Google Scholar] [CrossRef]
- Bayraç, C.; Camızcı, G. Adsorptive removal of patulin from apple juice via sulfhydryl-terminated magnetic bead-based separation. J. Hazard. Mater. 2019, 366, 413–422. [Google Scholar] [CrossRef]
- Sişman, T. The protective effect of hydrated sodium calcium aluminosilicate against the adverse effects of aflatoxin B1 on D. melanogaster. Toxicol. Ind. Health 2006, 22, 173–179. [Google Scholar] [CrossRef]
- Adeyeye, S.A.O.; Yildiz, F. Fungal mycotoxins in foods: A review. Cogent Food Agric. 2016, 2, 1–11. [Google Scholar] [CrossRef]
- Jeswal, P.; Kumar, D. Mycobiota and natural incidence of Aflatoxins, Ochratoxin A, and Citrinin in Indian Spices confirmed by LC-MS/MS. Int. J. Microbiol. 2015, 2015, 242486. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kabak, B.; Dobson, A.D.; Var, I. Strategies to prevent mycotoxin contamination of food and animal feed: A review. Crit. Rev. Food Sci. Nutr. 2006, 46, 593–619. [Google Scholar] [CrossRef] [PubMed]
- Abbasi Pirouz, A.; Abedi Karjiban, R.; Abu Bakar, F.; Selamat, J. A novel adsorbent magnetic graphene oxide modified with chitosan for the simultaneous reduction of mycotoxins. Toxins 2018, 10, 361. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Avantaggiato, G.; Havenaar, R.; Visconti, A. Assessment of the multi-mycotoxin-binding efficacy of a Carbon/Aluminosilicate-based product in an in vitro gastrointestinal model. J. Agric. Food Chem. 2007, 55, 4810–4819. [Google Scholar] [CrossRef] [PubMed]
- Scafuri, B.; Varriale, A.; Facchiano, A.; D’Auria, S.; Raggi, M.E.; Marabotti, A. Binding of mycotoxins to proteins involved in neuronal plasticity: A combined in silico/wet investigation. Sci. Rep. 2017, 7, 15156. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peng, Z.; Chen, L.; Zhu, Y.; Huang, Y.; Hu, X.; Wu, Q.; Nüssler, A.K.; Liu, L.; Yang, W. Current major degradation methods for aflatoxins: A review. Trends Food Sci. Technol. 2018, 80, 155–166. [Google Scholar] [CrossRef]
- Hawar, S.; Vevers, W.; Karieb, S.; Ali, B.K.; Billington, R.; Beal, J. Biotransformation of patulin to hydroascladiol by Lactobacillus plantarum. Food Control 2013, 34, 502–508. [Google Scholar] [CrossRef]
- Tannous, J.; Snini, S.P.; El Khoury, R.; Canlet, C.; Pinton, P.; Lippi, Y.; Alassane-Kpembi, I.; Gauthier, T.; El Khoury, A.; Atoui, A.; et al. Patulin transformation products and last intermediates in its biosynthetic pathway, E- and Z-ascladiol, are not toxic to human cells. Arch. Toxicol. 2017, 91, 2455–2467. [Google Scholar] [CrossRef]
- Sadiq, F.A.; Yan, B.; Tian, F.; Zhao, J.; Zhang, H.; Chen, W. Lactic acid bacteria as antifungal and anti-mycotoxigenic agents: A comprehensive review. Compr. Rev. Food Sci. Food Saf. 2019, 18, 1403–1436. [Google Scholar] [CrossRef] [Green Version]
- Karami, S.; Roayaei, M.; Zahedi, E.; Bahmani, M.; Mahmoodnia, L.; Hamzavi, H.; Rafieian-Kopaei, M. Antifungal effects of Lactobacillus species isolated from local dairy products. Int. J. Pharm. Investig. 2017, 7, 77–81. [Google Scholar]
- Ricelli, A.; Baruzzi, F.; Solfrizzo, M.; Morea, M.; Fanizzi, F.P. Biotransformation of patulin by Gluconobacter oxydans. Appl. Environ. Microbiol. 2007, 73, 785–792. [Google Scholar] [CrossRef] [Green Version]
- Bourdichon, F.; Casaregola, S.; Farrokh, C.; Frisvad, J.C.; Gerds, M.L.; Hammes, W.P.; Harnett, J.; Huys, G.; Laulund, S.; Ouwehand, A.; et al. Food fermentations: Microorganisms with technological beneficial use. Int. J. Food Microbiol. 2012, 154, 87–97. [Google Scholar] [CrossRef]
- Hooper, D.G.; Bolton, V.E.; Guilford, F.T.; Straus, D.C. Mycotoxin detection in human samples from patients exposed to environmental molds. Int. J. Mol. Sci. 2009, 10, 1465–1475. [Google Scholar] [CrossRef]
- Aiko, V.; Mehta, A. Occurrence, detection and detoxification of mycotoxins. J. Biosci. 2015, 40, 943–954. [Google Scholar] [CrossRef]
- Jakopović, Ž.; Hanousek Čiča, K.; Mrvčić, J.; Pucić, I.; Čanak, I.; Frece, J.; Pleadin, J.; Stanzer, D.; Zjalić, S.; Markov, K. Properties and fermentation activity of industrial yeasts Saccharomyces cerevisiae, S. uvarum, Candida utilis and Kluyveromyces marxianus exposed to AFB(1), OTA and ZEA. Food Technol. Biotechnol. 2018, 56, 208–217. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Yang, Q.; Zhang, H.; Cao, J.; Zhang, X.; Apaliya, M.T. The possible mechanisms involved in degradation of patulin by Pichia caribbica. Toxins 2016, 8, 289. [Google Scholar] [CrossRef] [PubMed]
- Ji, C.; Fan, Y.; Zhao, L. Review on biological degradation of mycotoxins. Anim. Nutr. 2016, 2, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Ianiri, G.; Pinedo, C.; Fratianni, A.; Panfili, G.; Castoria, R. Patulin degradation by the biocontrol yeast Sporobolomyces sp. is an inducible process. Toxins 2017, 9, 61. [Google Scholar] [CrossRef] [PubMed]
- Hatab, S.; Yue, T.; Mohamad, O. Removal of patulin from apple juice using inactivated lactic acid bacteria. J. Appl. Microbiol. 2012, 112, 892–899. [Google Scholar] [CrossRef] [PubMed]
- Guo, C.; Yue, T.; Hatab, S.; Yuan, Y. Ability of inactivated yeast powder to adsorb patulin from apple juice. J. Food Prot. 2012, 75, 585–590. [Google Scholar] [CrossRef]
- Yue, T.; Dong, Q.; Guo, C.; Worobo, R.W. Reducing patulin contamination in apple juice by using inactive yeast. J. Food Prot. 2011, 74, 149–153. [Google Scholar] [CrossRef]
- Coelho, A.; Celli, M.; Sataque Ono, E.; Hoffmann, F.; Pagnocca, F.; Garcia, S.; Sabino, M.; Harada, K.; Wosiacki, G.; Hirooka, E. Patulin biodegradation using Pichia ohmeri and Saccharomyces cerevisiae. World Mycotoxin J. 2008, 1, 325–331. [Google Scholar] [CrossRef]
- Piotrowska, M.; Nowak, A.; Czyzowska, A. Removal of ochratoxin A by wine Saccharomyces cerevisiae strains. Eur. Food Res. Technol. 2013, 236, 441–447. [Google Scholar] [CrossRef] [Green Version]
- Armando, M.R.; Pizzolitto, R.P.; Dogi, C.A.; Cristofolini, A.; Merkis, C.; Poloni, V.; Dalcero, A.M.; Cavaglieri, L.R. Adsorption of ochratoxin A and zearalenone by potential probiotic Saccharomyces cerevisiae strains and its relation with cell wall thickness. J. Appl. Microbial. 2012, 113, 256–264. [Google Scholar] [CrossRef]
- Salas, M.P.; Reynoso, C.M.; Céliz, G.; Daz, M.; Resnik, S.L. Efficacy of flavanones obtained from citrus residues to prevent patulin contamination. Food Res. Int. 2012, 48, 930–934. [Google Scholar] [CrossRef]
- Wang, L.; Wu, H.; Qin, G.; Meng, X. Chitosan disrupts Penicillium expansum and controls postharvest blue mold of jujube fruit. Food Control 2014, 41, 56–62. [Google Scholar] [CrossRef]
- Hassan, Y.I.; Zhou, T. Promising detoxification strategies to mitigate mycotoxins in food and feed. Toxins 2018, 10, 116. [Google Scholar] [CrossRef] [Green Version]
- Li, X.; Peng, X.; Wang, Q.; Zuo, H.; Meng, X.; Liu, B. Effective detoxification of patulin from aqueous solutions by immobilized porcine pancreatic lipase. Food Control 2017, 78, 48–56. [Google Scholar] [CrossRef]
- Tang, H.; Peng, X.; Li, X.; Meng, X.; Liu, B. Biodegradation of mycotoxin patulin in apple juice by calcium carbonate immobilized porcine pancreatic lipase. Food Control 2018, 88, 69–74. [Google Scholar] [CrossRef]
- Azam, M.S.; Yu, D.; Liu, N.; Wu, A. Degrading ochratoxin A and zearalenone mycotoxins using a multifunctional recombinant enzyme. Toxins 2019, 11, 301. [Google Scholar] [CrossRef] [Green Version]
- Whitehurst, R.J.; van Oort, M. Enzymes in Food Technology, 2nd ed.; Blackwell Publishing Ltd: Singapore, 2009; pp. 163–194. [Google Scholar]
- Liu, B.; Peng, X.; Meng, X. Effective biodegradation of mycotoxin patulin by porcine pancreatic lipase. Front. Microbiol. 2018, 9, 615. [Google Scholar] [CrossRef] [Green Version]
- Burgess, K.M.; Renaud, J.B.; McDowell, T.; Sumarah, M.W. Mechanistic Insight into the Biosynthesis and Detoxification of Fumonisin Mycotoxins. ACS Chem. Biol. 2016, 11, 2618–2625. [Google Scholar] [CrossRef]
- Vosough, P.R.; Sani, A.M.; Mehraban, M.; Karazhyan, R. In vitro effect of Lactobacillus rhamnosus GG on reduction of aflatoxin B1. Nutr. Food Sci. 2014, 44, 32–40. [Google Scholar] [CrossRef]
- Zhao, H.; Zhou, F.; Qi, Y.; Dziugan, P.; Bai, F.; Walczak, P.; Zhang, B. Screening of Lactobacillus strains for their ability to bind Benzo(a)pyrene and the mechanism of the process. Food Chem. Toxicol. 2013, 59, 67–71. [Google Scholar] [CrossRef]
- Zou, Z.-Y.; He, Z.-F.; Li, H.-J.; Han, P.-F.; Meng, X.; Zhang, Y.; Zhou, F.; Ouyang, K.-P.; Chen, X.-Y.; Tang, J. In vitro removal of deoxynivalenol and T-2 toxin by lactic acid bacteria. Food Sci. Biotechnol. 2012, 21, 1677–1683. [Google Scholar] [CrossRef]
- Assaf, J.C.; El Khoury, A.; Atoui, A.; Louka, N.; Chokr, A. A novel technique for aflatoxin M1 detoxification using chitin or treated shrimp shells: In vitro effect of physical and kinetic parameters on the binding stability. Appl. Microbiol. Biotechnol. 2018, 102, 6687–6697. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Li, Z.; Yuan, Y.; Yue, T. Bioadsorption of patulin from kiwi fruit juice onto a superior magnetic chitosan. J. Alloys Compd. 2016, 667, 101–108. [Google Scholar] [CrossRef]
- Li, J.; Liu, L.; Li, C.; Liu, L.; Tan, Y.; Meng, Y. The ability of Lactobacillus rhamnosus to bind patulin and its application in apple juice. Acta Aliment. 2020, 49, 93–102. [Google Scholar] [CrossRef]
- Liu, M.; Wang, J.; Yang, Q.; Hu, N.; Zhang, W.; Zhu, W.; Wang, R.; Suo, Y.; Wang, J. Patulin removal from apple juice using a novel cysteine-functionalized metal-organic framework adsorbent. Food Chem. 2019, 270, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Qiu, Y.; Guo, H.; Guo, C.; Zheng, J.; Yue, T.; Yuan, Y. One-step preparation of nano-Fe3O4 modified inactivated yeast for the adsorption of patulin. Food Control 2018, 86, 310–318. [Google Scholar] [CrossRef]
- Peng, X.; Liu, B.; Chen, W.; Li, X.; Wang, Q.; Meng, X.; Wang, D. Effective biosorption of patulin from apple juice by cross-linked xanthated chitosan resin. Food Control 2016, 63, 140–146. [Google Scholar] [CrossRef]
- Ge, N.; Xu, J.; Li, F.; Peng, B.; Pan, S. Immobilization of inactivated microbial cells on magnetic Fe3O4@CTS nanoparticles for constructing a new biosorbent for removal of patulin in fruit juice. Food Control 2017, 82, 83–90. [Google Scholar] [CrossRef]
- Guo, C.; Yue, T.; Yuan, Y.; Wang, Z.; Guo, Y.; Wang, L.; Li, Z. Biosorption of patulin from apple juice by caustic treated waste cider yeast biomass. Food Control 2013, 32, 99–104. [Google Scholar] [CrossRef]
- Sajid, M.; Mehmood, S.; Niu, C.; Yuan, Y.; Yue, T. Effective adsorption of patulin from apple juice by using non-cytotoxic heat-inactivated cells and spores of Alicyclobacillus strains. Toxins 2018, 10, 344. [Google Scholar] [CrossRef] [Green Version]
- Drusch, S.; Ragab, W. Mycotoxins in fruits, fruit juices, and dried fruits. J. Food Prot. 2003, 66, 1514–1527. [Google Scholar] [CrossRef] [PubMed]
- Raiola, A.; Tenore, G.C.; Manyes, L.; Meca, G.; Ritieni, A. Risk analysis of main mycotoxins occurring in food for children: An overview. Food Chem. Toxicol. 2015, 84, 169–180. [Google Scholar] [CrossRef] [PubMed]
- Ojuri, O.T.; Ezekiel, C.N.; Eskola, M.K.; Šarkanj, B.; Babalola, A.D.; Sulyok, M.; Hajšlová, J.; Elliott, C.T.; Krska, R. Mycotoxin co-exposures in infants and young children consuming household- and industrially-processed complementary foods in Nigeria and risk management advice. Food Control 2019, 98, 312–322. [Google Scholar] [CrossRef]
- Shao, S.; Zhou, T.; McGarvey, B.D. Comparative metabolomic analysis of Saccharomyces cerevisiae during the degradation of patulin using gas chromatography-mass spectrometry. Appl. Microbiol. Biotechnol. 2012, 94, 789–797. [Google Scholar] [CrossRef]
- Desmarchelier, A.; Mujahid, C.; Racault, L.; Perring, L.; Lancova, K. Analysis of patulin in pear- and apple-based foodstuffs by liquid chromatography electrospray ionization tandem mass spectrometry. J. Agric. Food Chem. 2011, 59, 7659–7665. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Li, X.; Zhang, Q. Determination of trace patulin in apple-based food matrices. Food Chem. 2017, 233, 290–301. [Google Scholar] [CrossRef] [PubMed]
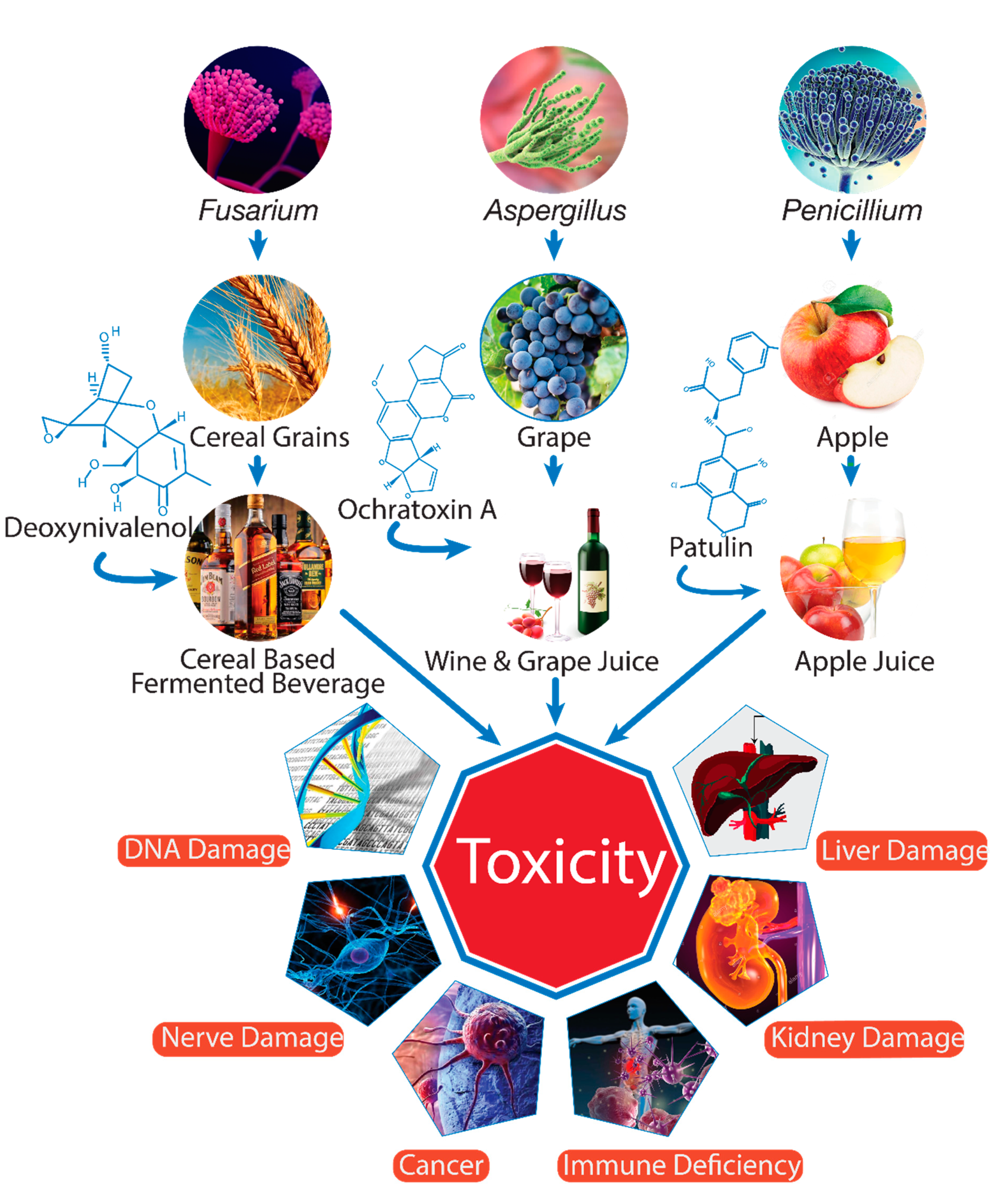
| Mycotoxins | Products Contaminated | Producing Microorganisms | References |
|---|---|---|---|
| Aflatoxins B1, B2, G1, G2 | Orange, apple juice, grape juice, grapefruit peel | Aspergillus chevallieri, A. flavus, A. niger, A. oryzae, A. parasiticus, A. repens, A. ruber, A. tamarii, and A. wentii | [19,20] |
| Ochratoxin A (OTA) | Grape juice, coffee, beer, and wine | A. ochraceus, A. carbonarius, A. niger, A. tubingensis, and Penicillium expansum | [19,21] |
| Patulin (PAT) | Fruit juices | Penicillium expansum, P. patulum, Aspergillus clavatus, Byssochlamys fulva, and B. nivea | [22,23] |
| Fumonisins (FBs) | Beer | Fusariumproliferatum, F. verticillioides, and F. nygamai | [24,25,26] |
| Trichothecenes (type B: Deoxynivalenol (DON)) | Plant-based beverages, beer | F. graminearum, F. cerealis, and F. culmorum | [25,27,28,29] |
| Trichothecenes (type A: HT-2) | Functional vegetable milks, beer | F. sporotrichioides,and F. langsethiae | [29,30] |
| Trichothecenes (type A: T-2 toxin) | Plant-based milks, beer | F. sporotrichioides, and F. langsethiae | [28,30] |
| Zearalenone (ZEN) | Beer, wine | F. graminearum, F. culmorum, F. equiseti, F. cerealis, F. verticillioides, and F. incarnatum | [25,31] |
| Alternaria toxins (TeA, AOH, AME) | Fruit juices, wine, beer | Alternaria alternate, A. tenuissima, and A. arborescens | [32,33] |
| Commodities | MLs (μg/kg) | |
|---|---|---|
| Aflatoxins | B1 | B1 + B2 + G1 +G2 |
| Dried fruits subjected to sorting or other physical treatment prior to human consumption or use as raw materials for food production | 5.0 | 10.0 |
| Dried fruits and finished products for direct human consumption or use as raw materials for food production | 2.0 | 4.0 |
| Cereal-based processed products and baby foods for young children and infants | 0.10 | – |
| Ochratoxin A | ||
| Dried vine fruits (raisins, currants, and sultanas) | 10.0 | |
| Wines (including sparkling wines and excluding liqueur wines and wines with an alcoholic strength of not less than 15% by vol) and fruit wine | 2.0 | |
| Aromatised wines, aromatized wine-based cocktails, and aromatized wine-based drinks | 2.0 | |
| Grape juice, grape nectar, grape must, and reconstituted concentrated grape must and reconstituted concentrated grape juice intended for direct human consumption | 2.0 | |
| Cereal-based processed products and baby foods for young children and infants | 0.50 | |
| Instant coffee (soluble coffee) | 10 | |
| Roasted coffee beans and ground roasted coffee, excluding soluble coffee | 5 | |
| Patulin | ||
| Fruit juices, fruit nectars, and reconstituted concentrated fruit juices | 50.0 | |
| Ciders, spirit drinks, and other fermented beverages made from apples or apple juice | 50.0 | |
| Solid apple foodstuffs including apple puree and apple compote for direct consumption | 25.0 | |
| Solid apple foodstuffs and apple juice, including apple puree and apple compote branded and promoted for young children and infants | 10.0 | |
| Baby foods other than cereal-based, processed foodstuffs for young children and infants | 10.0 | |
| Analytical Methods | Detection Method | Toxin | Applicability | LOD | References | Advantages | Disadvantages |
|---|---|---|---|---|---|---|---|
| TLC | CCD | Patulin | Apple Juice | 14 µg/L | [77] | Time saving, specific fluorescence spot on UV light | Limited plate length and environmental effects on measurement |
| HPLC | FD | OTA | Wheat | 23 pg | [78] | Fast, high resolution data, accurate and easily reproducible. Less training required | Expensive and method development could be challenging |
| MS/MS | Wine | 0.005 ng/ml | [79] | ||||
| FD | 0.09 µg/L | [80] | |||||
| AFs | Food items | 1.6-5.2 mg/kg | [81] | ||||
| UV and FD | Milk | 0.13–0.16 mg/L | [82] | ||||
| LC | FD | OTA | Wine | 0.07 ng/ml | [83] | Several mycotoxin detections, high sensitivity, provides confirmation | Expensive, required expertise In case of MS, sensitivity depends on ionization |
| ZEN | Barley, Maize, Wheat | 100 µg/Kg | [84] | ||||
| AFB1 | Corn | 2–5 ng/g | [85] | ||||
| MS/MS | Trichothecenes | Wheat and maize | 0.2–3.3 µg/Kg | [86] | |||
| Automated microarray chip reader | Chemiluminescence | OTA | Coffee | 0.3 µg/L | [87] | High throughput, multiplexed, parallel processing method | Not so common to their variability and reproducibility, require intensive labor |
| Electro-polymerization onto surface | SPR | ZEN | Corn | 0.3 ng/ml | [88] | Suitable for cereals sample, sim- plicity, portability, and ease to use, can be used in field | Optimization and validation not reported for this method |
| Immunochromatographic strip | Highly Luminescent Quantum Dot Beads | AFB1 | Maize | 0.42 pg/ml | [89] | A simple method for rapid screening, superior performance | Required expertise |
| Direct, competitive magneto-immunoassay | SPR | OTA | Beverages | 0.042 µg/L | [90] | Rapid, cost effective, and sensitive | |
| Electrochemical | FB | Maize | 0.33 µg/L | [91,92] | |||
| Lateral flow immunoassay | Colorimetric | 199 µg/Kg | [93] | Fast, one-step assay, no washing step, low cost and simple | Qualitative or semi quantitative results, sample volume governs precision | ||
| Photonics immobilization technique | Quartz-crystal microbalance (QCM) | Patulin | Apple puree | 56 ng/ml | [94] | Specific, higher sensitivity, generality, response (only requires a few minutes), flexibility, and portability | The decrease of the signal in the presence of high analyte concentrations, in situ analysis |
| Surface-enhanced Raman scattering (SERS)-based immunoassay | Silica-encapsulated hollow gold nanoparticles | AFB1 | Food | 0.1 ng/ml | [95] | Enhanced ELISA method | Hard to synthesize and expensive |
| ELISA | UV absorbance | AFM1 | Milk | 4–25 ng/L | [96] | Fast, simple, economical, high sensitivity, simultaneous analysis of multiple samples, easy to screen | Lack of precision at low concentrations, matrice interference problems, possible false-positive/negative results |
| ZEN | Maize | 0.02 µg/L | [97] | ||||
| AFB1 and AFM1 | Food items | 0.13-0.16 mg/L | [82] | ||||
| Electrochemical | FB | Corn | 5 µg/L | [98] |
| Evaluation Criteria | References |
|---|---|
| Effectiveness of the active ingredient confirmed by scientific evidence | [7] |
| High rate of addition | [165] |
| Stability over a broad pH range | [166] |
| High ability to adsorb higher mycotoxin concentrations | [164] |
| Strong affinity to adsorb low mycotoxin concentrations | [164] |
| Assertion of chemical interaction of mycotoxin with adsorbent | [167] |
| Established in vivo data with all relevant mycotoxins | [168] |
| Non-toxic, environmentally friendly component | [169] |
| Mycotoxin | Microorganism (Genus) | Strains | Matrices | Effects | Time | Reference |
|---|---|---|---|---|---|---|
| FB | Bacteria (Enterococcus) | E. faecium 21605 | Apple juice | 64% Adsorption | 24 h | [183] |
| PAT | Yeast (Saccharomyces) | S. cerevisiae strain YS3 (laboratory prepared) | Apple juice | 70% Adsorption | 24 h | [182] |
| PAT | Yeast (Saccharomyces) | S. cerevisiae strain YS3 (commercial) | Apple juice | 76% Adsorption | 24 h | [37] |
| PAT | Yeast (Saccharomyces) | S. cerevisiae YS1–YS10 | Apple juice | 50% to 7% Adsorption | 24 h | [184,185] |
| PAT | Yeast (Saccharomyces) | S. cerevisiae YS3 | Apple juice | 100% Adsorption | 36 h | [186] |
| PAT | Yeast (Saccharomyces) | S. cerevisiae | Apple juice | 90% to 96% Adsorption | 143 h | [186] |
| OTA | Yeast (Saccharomyces) | S. cerevisiae Malaga LOCK 0173 | Grape/blackcurrant juice | 85% Adsorption | 10 days | [187] |
| OTA | Yeast (Saccharomyces) | S. cerevisiae Syrena LOCK 0201 | Grape/black currant juice | 83% Adsorption | 10 days | [187] |
| OTA | Yeast (Saccharomyces) | S. cerevisiae bakery BS strain | Grape/blackcurrant juice | 64% Adsorption | 10 days | [187] |
| OTA | Yeast (Saccharomyces) | S. cerevisiae | White wine | 76% Adsorption | 90 days | [188] |
| OTA | Yeast (Saccharomyces) | S. cerevisiae | Red wine | 86% Adsorption | 90 days | [188] |
| OTA | Yeast (Saccharomyces) | S. cerevisiae | Rose wine | 90% Adsorption | 90 days | [188] |
| Bioadsorbents | Adsorption Capacity | Time (h) | Matrices | Reference |
|---|---|---|---|---|
| Zirconium-based absorbent (UiO-66(NH2) | 4.4 µg/mg | 3 | Apple juice | [204] |
| Nano-Fe3O4 modified inactivated yeast | 8.6 × 10−3 µg/mg | 3.5 | Apple juice | [205] |
| Cross-linked xanthated chitosan resin (CXCR) | 130.0 µg/mg | 18 | Apple juice | [206] |
| Inactivated microbial cells on magnetic Fe3O4@CTS nanoparticles | 90.0% | 24 | Orange juice | [207] |
| Superior magnetic chitosan | 19.4 × 10−3 µg/mg | 9 | Kiwi juice | [202] |
| Caustic treated waste cider yeast biomass | 58.3% | 24 | Apple juice | [208] |
| Non-Cytotoxic Heat-Inactivated Cells and Spores of Alicyclobacillus Strains | 12.6 × 10−3 µg/mg | 24 | Apple juice | [209] |
| Inactivated Lactobacillus rhamnosus powder | 53.39% | 4 | Apple juice | [203] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Azam, M.S.; Ahmed, S.; Islam, M.N.; Maitra, P.; Islam, M.M.; Yu, D. Critical Assessment of Mycotoxins in Beverages and Their Control Measures. Toxins 2021, 13, 323. https://doi.org/10.3390/toxins13050323
Azam MS, Ahmed S, Islam MN, Maitra P, Islam MM, Yu D. Critical Assessment of Mycotoxins in Beverages and Their Control Measures. Toxins. 2021; 13(5):323. https://doi.org/10.3390/toxins13050323
Chicago/Turabian StyleAzam, Md. Shofiul, Shafi Ahmed, Md. Nahidul Islam, Pulak Maitra, Md. Mahmudul Islam, and Dianzhen Yu. 2021. "Critical Assessment of Mycotoxins in Beverages and Their Control Measures" Toxins 13, no. 5: 323. https://doi.org/10.3390/toxins13050323








