Highly Sensitive and Specific Detection of Staphylococcal Enterotoxins SEA, SEG, SEH, and SEI by Immunoassay
Abstract
1. Introduction
2. Results
2.1. Production and Characterization of 79 Monoclonal Antibodies Directed against SEA, SEG, SEH, or SEI Toxins
2.2. Development of Four Sandwich Enzyme Immunoassays for the Specific Individual Detection of SEA, SEG, SEH, and SEI Toxins
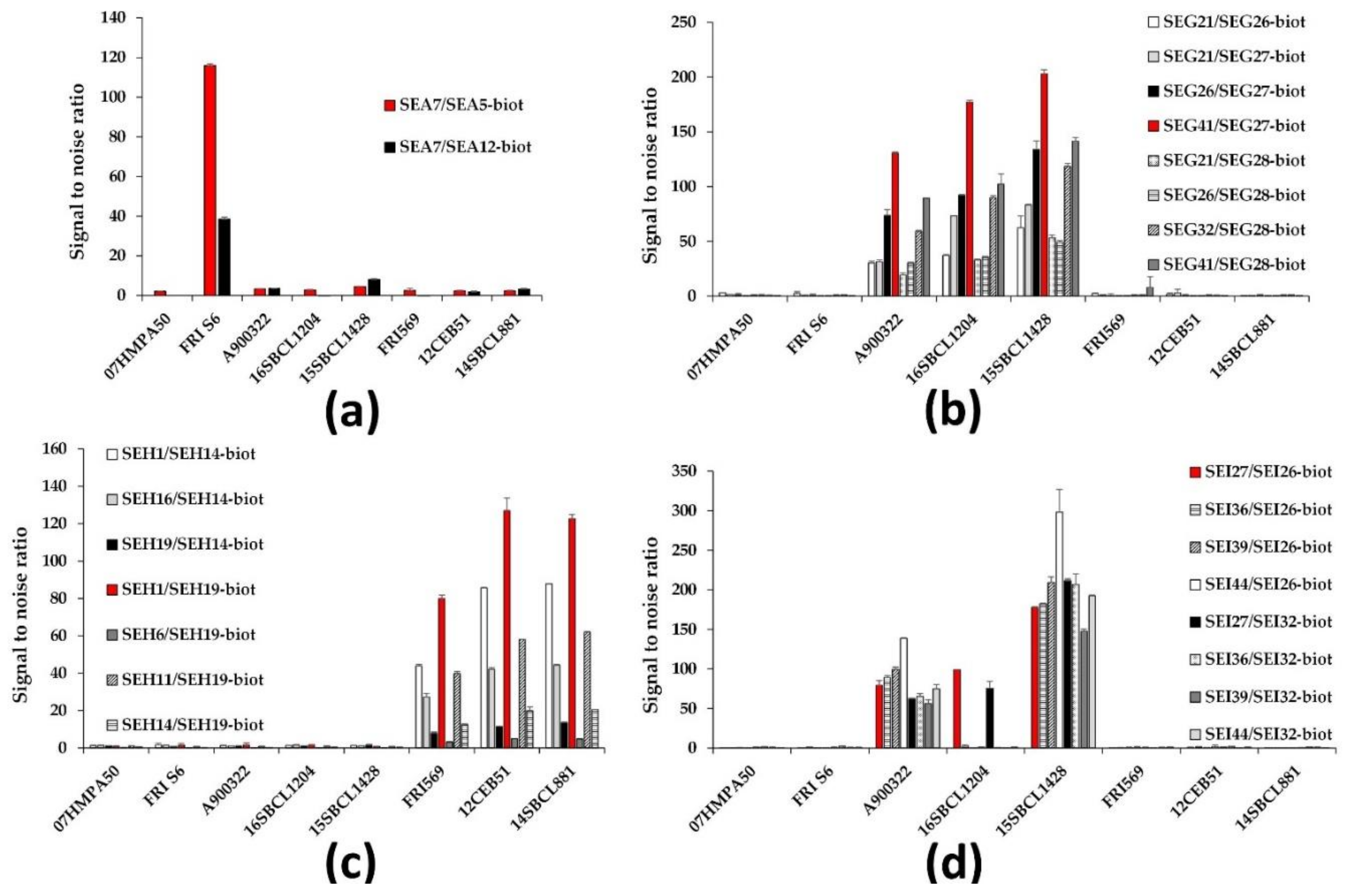
2.2.1. Selection of Best mAb Pairs
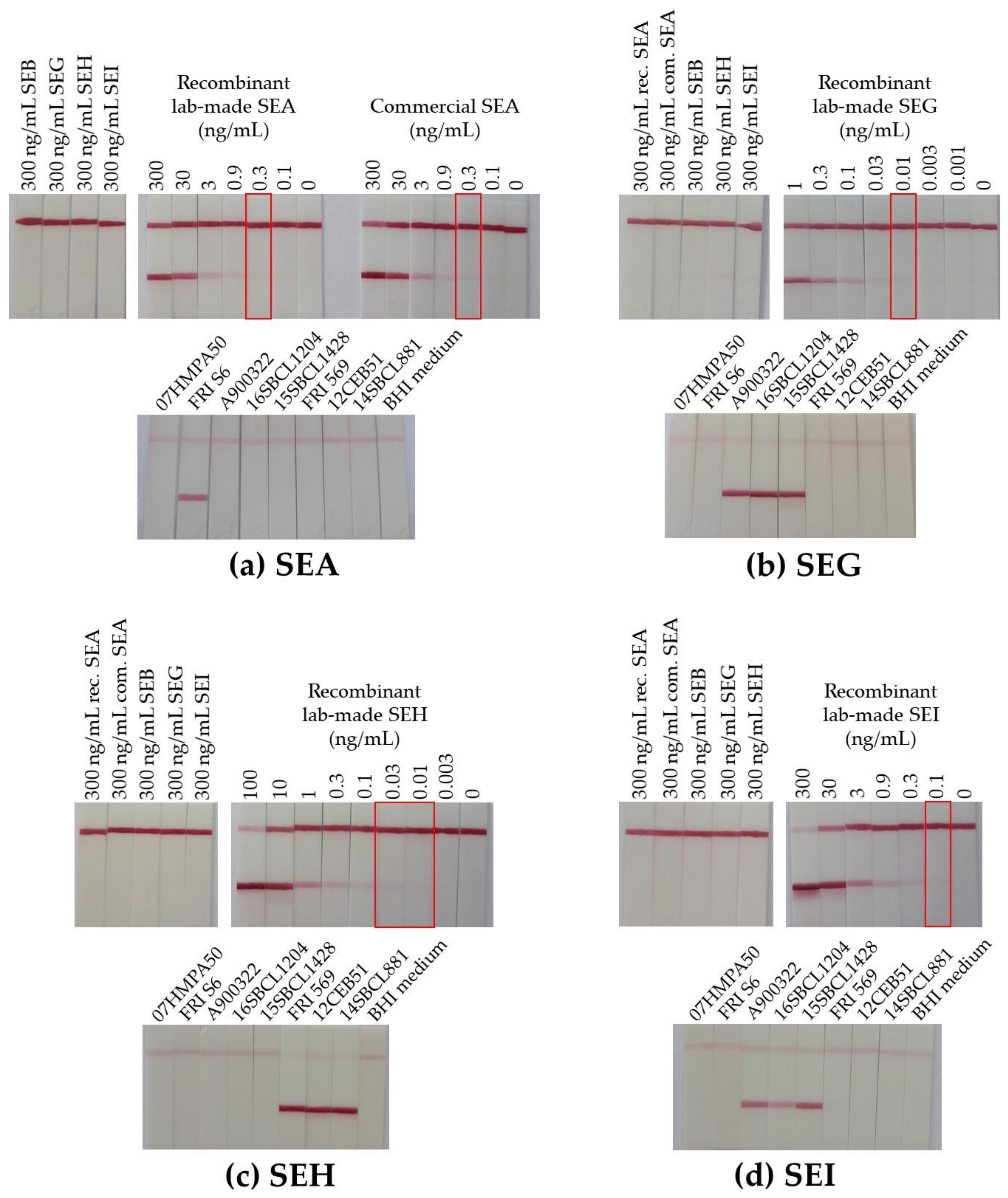
2.2.2. Performances of the Immunoassays
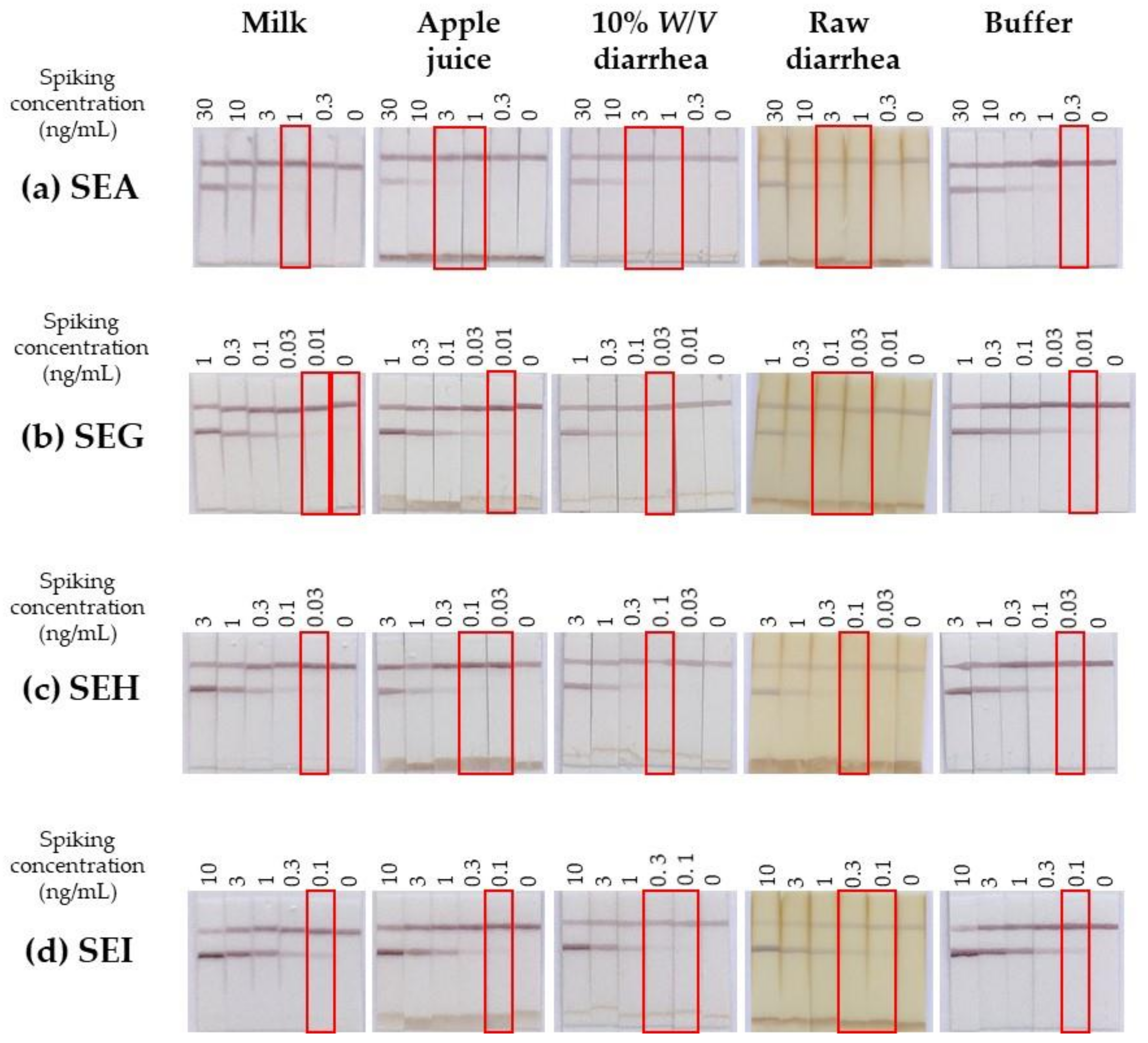
2.2.3. Robustness in Complex Matrices
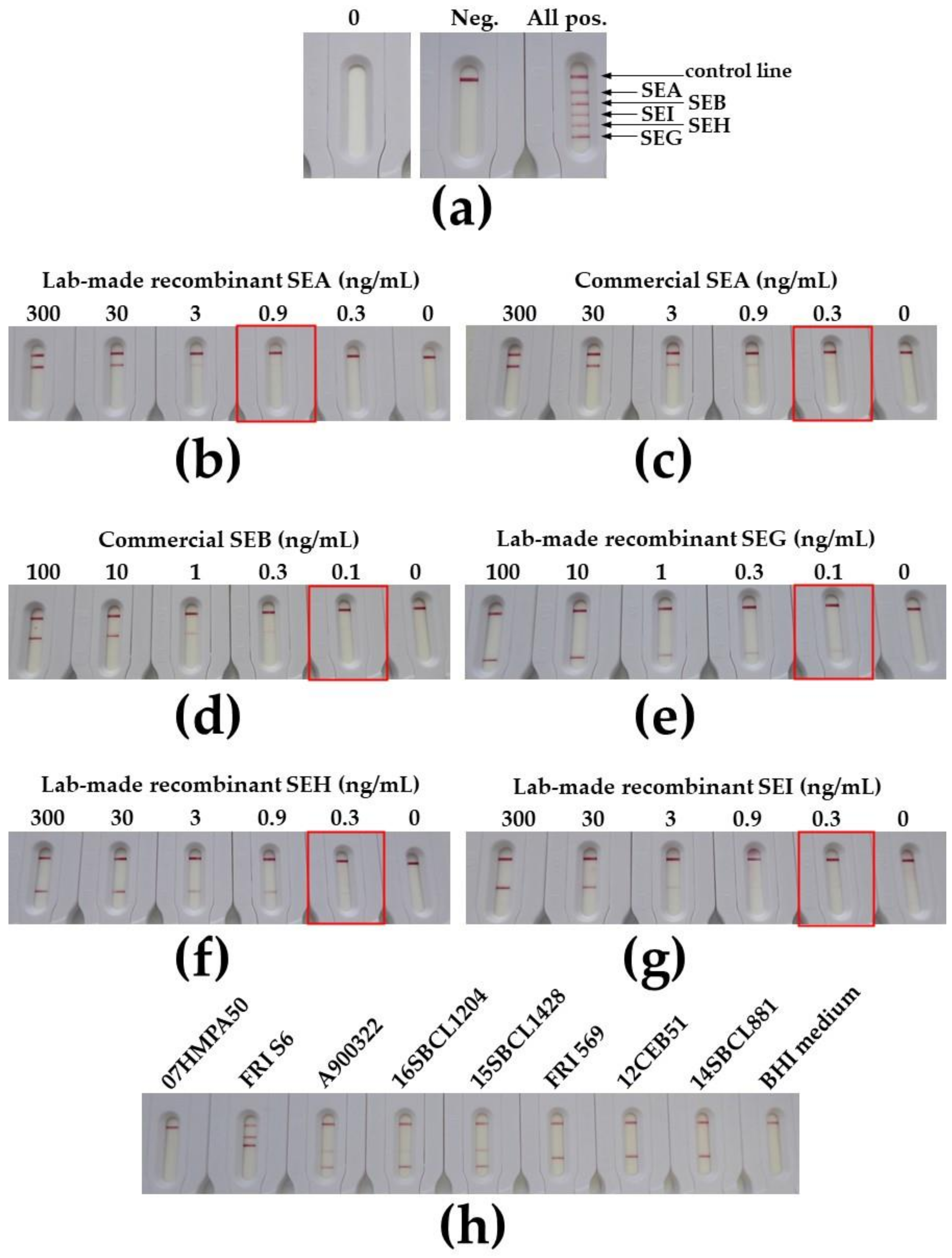
2.3. Development of Immunochromatographic Tests for the Monoplex and Multiplex Detection of SEA, SEB, SEG, SEH, and SEI Toxins
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Bacterial Cultures
5.2. Recombinant His-Tagged SEA, SEG, SEH, and SEI Production and Purification
5.3. Production of Monoclonal Antibodies against SEA, SEG, SEH, and SEI
5.4. Labeling of Proteins with Biotin
5.5. Evaluation of Polyclonal Response and Screening of mAbs in Hybridoma Supernatants
5.6. Determination of mAb Affinity
5.7. Sandwich Enzyme Immunoassays
5.8. Theoretical Limits of Detection and Quantification
5.9. Repeatability and Reproducibility Assays
5.10. Food and Clinical Sample Preparation
5.11. Immunochromatographic Assays
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kadariya, J.; Smith, T.C.; Thapaliya, D. Staphylococcus Aureus and Staphylococcal Food-Borne Disease: An Ongoing Challenge in Public Health. Biomed. Res. Int. 2014, 2014, 827965. [Google Scholar] [CrossRef] [PubMed]
- Hennekinne, J.-A.; De Buyser, M.-L.; Dragacci, S. Staphylococcus Aureus and Its Food Poisoning Toxins: Characterization and Outbreak Investigation. FEMS Microbiol. Rev. 2012, 36, 815–836. [Google Scholar] [CrossRef]
- Commission Regulation (EC) No. 1441/2007 of 5 December 2007 Amending Regulation (EC) No. 2073/2005 Regarding the Microbiological Criteria Applicable to Foodstuffs. Off. J. Eur. Union 2007, L322, 12–29.
- Commission Regulation (EC) No 178/2002 of the European Parliament and of the Council of 28 January 2002 Amended by the Regulation (EC) No 1642/2003 of 22 July 2003 and Commission Regulation (EC) No 575/2006 of 7 April 2006 Laying down the General Principles and Requirements of Food Law, Establishing the European Food Safety Authority and Laying down Procedures in Matters of Food Safety. Off. J. Eur. Union 2002, L31, 14–15.
- Le Loir, Y.; Baron, F.; Gautier, M. Staphylococcus Aureus and Food Poisoning. Genet. Mol. Res. 2003, 2, 63–76. [Google Scholar]
- Evenson, M.L.; Hinds, M.W.; Bernstein, R.S.; Bergdoll, M.S. Estimation of Human Dose of Staphylococcal Enterotoxin A from a Large Outbreak of Staphylococcal Food Poisoning Involving Chocolate Milk. Int. J. Food Microbiol. 1988, 7, 311–316. [Google Scholar] [CrossRef]
- Guillier, L.; Bergis, H.; Guillier, F.; Noel, V.; Auvray, F.; Hennekinne, J.-A. Dose-Response Modelling of Staphylococcal Enterotoxins Using Outbreak Data. Procedia Food Sci. 2016, 7, 129–132. [Google Scholar] [CrossRef]
- Nia, Y.; Mutel, I.; Assere, A.; Lombard, B.; Auvray, F.; Hennekinne, J.-A. Review Over a 3-Year Period of European Union Proficiency Tests for Detection of Staphylococcal Enterotoxins in Food Matrices. Toxins 2016, 8, 107. [Google Scholar] [CrossRef]
- Munson, S.H.; Tremaine, M.T.; Betley, M.J.; Welch, R.A. Identification and Characterization of Staphylococcal Enterotoxin Types G and I from Staphylococcus Aureus. Infect. Immun. 1998, 66, 3337–3348. [Google Scholar] [CrossRef]
- Hu, D.-L.; Omoe, K.; Shimoda, Y.; Nakane, A.; Shinagawa, K. Induction of Emetic Response to Staphylococcal Enterotoxins in the House Musk Shrew (Suncus Murinus). Infect. Immun. 2003, 71, 567–570. [Google Scholar] [CrossRef]
- Ono, H.K.; Omoe, K.; Imanishi, K.; Iwakabe, Y.; Hu, D.-L.; Kato, H.; Saito, N.; Nakane, A.; Uchiyama, T.; Shinagawa, K. Identification and Characterization of Two Novel Staphylococcal Enterotoxins, Types S and T. Infect. Immun. 2008, 76, 4999–5005. [Google Scholar] [CrossRef]
- Omoe, K.; Hu, D.-L.; Ono, H.K.; Shimizu, S.; Takahashi-Omoe, H.; Nakane, A.; Uchiyama, T.; Shinagawa, K.; Imanishi, K. Emetic Potentials of Newly Identified Staphylococcal Enterotoxin-like Toxins. Infect. Immun. 2013, 81, 3627–3631. [Google Scholar] [CrossRef]
- Ono, H.K.; Hirose, S.; Naito, I.; Sato’o, Y.; Asano, K.; Hu, D.-L.; Omoe, K.; Nakane, A. The Emetic Activity of Staphylococcal Enterotoxins, SEK, SEL, SEM, SEN and SEO in a Small Emetic Animal Model, the House Musk Shrew. Microbiol. Immunol. 2017, 61, 12–16. [Google Scholar] [CrossRef]
- Ono, H.K.; Hirose, S.; Narita, K.; Sugiyama, M.; Asano, K.; Hu, D.-L.; Nakane, A. Histamine Release from Intestinal Mast Cells Induced by Staphylococcal Enterotoxin A (SEA) Evokes Vomiting Reflex in Common Marmoset. PLoS Pathog. 2019, 15, e1007803. [Google Scholar] [CrossRef]
- Wilson, G.J.; Seo, K.S.; Cartwright, R.A.; Connelley, T.; Chuang-Smith, O.N.; Merriman, J.A.; Guinane, C.M.; Park, J.Y.; Bohach, G.A.; Schlievert, P.M.; et al. A Novel Core Genome-Encoded Superantigen Contributes to Lethality of Community-Associated MRSA Necrotizing Pneumonia. PLoS Pathog. 2011, 7, e1002271. [Google Scholar] [CrossRef]
- Ono, H.K.; Sato’o, Y.; Narita, K.; Naito, I.; Hirose, S.; Hisatsune, J.; Asano, K.; Hu, D.-L.; Omoe, K.; Sugai, M.; et al. Identification and Characterization of a Novel Staphylococcal Emetic Toxin. Appl. Environ. Microbiol. 2015, 81, 7034–7040. [Google Scholar] [CrossRef] [PubMed]
- Spoor, L.E.; Richardson, E.; Richards, A.C.; Wilson, G.J.; Mendonca, C.; Gupta, R.K.; McAdam, P.R.; Nutbeam-Tuffs, S.; Black, N.S.; O’ Gara, J.P.; et al. Recombination-Mediated Remodelling of Host-Pathogen Interactions during Staphylococcus Aureus Niche Adaptation. Microb. Genom. 2015, 1, e000036. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.-F.; Yang, X.-Y.; Zhang, J.; Qin, X.; Huang, X.; Cui, Y.; Zhou, M.; Shi, C.; French, N.P.; Shi, X. Identification and Characterization of Two Novel Superantigens among Staphylococcus Aureus Complex. Int. J. Med. Microbiol. 2018, 308, 438–446. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Duan, N.; Gu, H.; Hao, L.; Ye, H.; Gong, W.; Wang, Z. A Review of the Methods for Detection of Staphylococcus Aureus Enterotoxins. Toxins 2016, 8, 176. [Google Scholar] [CrossRef] [PubMed]
- Vernozy-Rozand, C.; Mazuy-Cruchaudet, C.; Bavai, C.; Richard, Y. Comparison of Three Immunological Methods for Detecting Staphylococcal Enterotoxins from Food. Lett. Appl. Microbiol. 2004, 39, 490–494. [Google Scholar] [CrossRef]
- Bennett, R.W. Staphylococcal Enterotoxin and Its Rapid Identification in Foods by Enzyme-Linked Immunosorbent Assay-Based Methodology. J. Food Prot. 2005, 68, 1264–1270. [Google Scholar] [CrossRef]
- Rose, S.A.; Bankes, P.; Stringer, M.F. Detection of Staphylococcal Enterotoxins in Dairy Products by the Reversed Passive Latex Agglutination (SET-RPLA) Kit. Int. J. Food Microbiol. 1989, 8, 65–72. [Google Scholar] [CrossRef]
- Standard EN ISO 19020:2017: Microbiology of the Food Chain—Horizontal Method for the Immunoenzymatic Detection of Staphylococcal Enterotoxins in Foodstuffs; ISO: Geneva, Switzerland, 2017.
- Ciupescu, L.-M.; Auvray, F.; Nicorescu, I.M.; Meheut, T.; Ciupescu, V.; Lardeux, A.-L.; Tanasuica, R.; Hennekinne, J.-A. Characterization of Staphylococcus Aureus Strains and Evidence for the Involvement of Non-Classical Enterotoxin Genes in Food Poisoning Outbreaks. FEMS Microbiol. Lett. 2018, 365. [Google Scholar] [CrossRef]
- McLauchlin, J.; Narayanan, G.L.; Mithani, V.; O’ Neill, G. The Detection of Enterotoxins and Toxic Shock Syndrome Toxin Genes in Staphylococcus Aureus by Polymerase Chain Reaction. J. Food Prot. 2000, 63, 479–488. [Google Scholar] [CrossRef]
- Kérouanton, A.; Hennekinne, J.A.; Letertre, C.; Petit, L.; Chesneau, O.; Brisabois, A.; De Buyser, M.L. Characterization of Staphylococcus Aureus Strains Associated with Food Poisoning Outbreaks in France. Int. J. Food Microbiol. 2007, 115, 369–375. [Google Scholar] [CrossRef]
- Yan, X.; Wang, B.; Tao, X.; Hu, Q.; Cui, Z.; Zhang, J.; Lin, Y.; You, Y.; Shi, X.; Grundmann, H. Characterization of Staphylococcus Aureus Strains Associated with Food Poisoning in Shenzhen, China. Appl. Environ. Microbiol. 2012, 78, 6637–6642. [Google Scholar] [CrossRef] [PubMed]
- Hait, J.; Tallent, S.; Melka, D.; Keys, C.; Bennett, R. Prevalence of Enterotoxins and Toxin Gene Profiles of Staphylococcus Aureus Isolates Recovered from a Bakery Involved in a Second Staphylococcal Food Poisoning Occurrence. J. Appl. Microbiol. 2014, 117, 866–875. [Google Scholar] [CrossRef]
- Johler, S.; Giannini, P.; Jermini, M.; Hummerjohann, J.; Baumgartner, A.; Stephan, R. Further Evidence for Staphylococcal Food Poisoning Outbreaks Caused by Egc-Encoded Enterotoxins. Toxins 2015, 7, 997–1004. [Google Scholar] [CrossRef]
- Umeda, K.; Nakamura, H.; Yamamoto, K.; Nishina, N.; Yasufuku, K.; Hirai, Y.; Hirayama, T.; Goto, K.; Hase, A.; Ogasawara, J. Molecular and Epidemiological Characterization of Staphylococcal Foodborne Outbreak of Staphylococcus Aureus Harboring Seg, Sei, Sem, Sen, Seo, and Selu Genes without Production of Classical Enterotoxins. Int. J. Food Microbiol. 2017, 256, 30–35. [Google Scholar] [CrossRef]
- Su, Y.C.; Wong, A.C. Detection of Staphylococcal Enterotoxin H by an Enzyme-Linked Immunosorbent Assay. J. Food Prot. 1996, 59, 327–330. [Google Scholar] [CrossRef]
- Sakai, F.; Ihara, H.; Aoyama, K.; Igarashi, H.; Yanahira, S.; Ohkubo, T.; Asao, T.; Kozaki, S. Characteristics of Enterotoxin H-Producing Staphylococcus Aureus Isolated from Clinical Cases and Properties of the Enterotoxin Productivity. J. Food Prot. 2008, 71, 1855–1860. [Google Scholar] [CrossRef] [PubMed]
- Omoe, K.; Ishikawa, M.; Shimoda, Y.; Hu, D.-L.; Ueda, S.; Shinagawa, K. Detection of Seg, Seh, and Sei Genes in Staphylococcus Aureus Isolates and Determination of the Enterotoxin Productivities of S. Aureus Isolates Harboring Seg, Seh, or Sei Genes. J. Clin. Microbiol. 2002, 40, 857–862. [Google Scholar] [CrossRef]
- Ono, H.K.; Hachiya, N.; Suzuki, Y.; Naito, I.; Hirose, S.; Asano, K.; Omoe, K.; Nakane, A.; Hu, D.-L. Development of an Immunoassay for Detection of Staphylococcal Enterotoxin-Like J, A Non-Characterized Toxin. Toxins 2018, 10, 458. [Google Scholar] [CrossRef]
- Nagaraj, S.; Ramlal, S.; Kingston, J.; Batra, H.V. Development of IgY Based Sandwich ELISA for the Detection of Staphylococcal Enterotoxin G (SEG), an Egc Toxin. Int. J. Food Microbiol. 2016, 237, 136–141. [Google Scholar] [CrossRef]
- Rudenko, N.V.; Karatovskaya, A.P.; Noskov, A.N.; Shepelyakovskaya, A.O.; Shchannikova, M.P.; Loskutova, I.V.; Artyemieva, O.A.; Nikanova, D.A.; Gladyr, E.A.; Brovko, F.A. Immunochemical Assay with Monoclonal Antibodies for Detection of Staphylococcal Enterotoxin H. J. Food Drug Anal. 2018, 26, 741–750. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhu, A.; Tang, J.; Tang, C.; Chen, J.; Liu, J. Identification and Measurement of Staphylococcal Enterotoxin-like Protein I (SEll) Secretion from Staphylococcus Aureus Clinical Isolate. J. Appl. Microbiol. 2016, 121, 539–546. [Google Scholar] [CrossRef] [PubMed]
- Rubina, A.Y.; Filippova, M.A.; Feizkhanova, G.U.; Shepeliakovskaya, A.O.; Sidina, E.I.; Boziev, K.M.; Laman, A.G.; Brovko, F.A.; Vertiev, Y.V.; Zasedatelev, A.S.; et al. Simultaneous Detection of Seven Staphylococcal Enterotoxins: Development of Hydrogel Biochips for Analytical and Practical Application. Anal. Chem. 2010, 82, 8881–8889. [Google Scholar] [CrossRef]
- Jørgensen, H.J.; Mathisen, T.; Løvseth, A.; Omoe, K.; Qvale, K.S.; Loncarevic, S. An Outbreak of Staphylococcal Food Poisoning Caused by Enterotoxin H in Mashed Potato Made with Raw Milk. FEMS Microbiol. Lett. 2005, 252, 267–272. [Google Scholar] [CrossRef]
- Ikeda, T.; Tamate, N.; Yamaguchi, K.; Makino, S. Mass Outbreak of Food Poisoning Disease Caused by Small Amounts of Staphylococcal Enterotoxins A and H. Appl. Environ. Microbiol. 2005, 71, 2793–2795. [Google Scholar] [CrossRef]
- Hait, J.M.; Nguyen, A.T.; Tallent, S.M. Analysis of the VIDAS® Staph Enterotoxin III (SET3) for Detection of Staphylococcal Enterotoxins G, H, and I in Foods. J. AOAC Int. 2018, 101, 1482–1489. [Google Scholar] [CrossRef] [PubMed]
- Zeleny, R.; Nia, Y.; Schimmel, H.; Mutel, I.; Hennekinne, J.-A.; Emteborg, H.; Charoud-Got, J.; Auvray, F. Certified Reference Materials for Testing of the Presence/Absence of Staphylococcus Aureus Enterotoxin A (SEA) in Cheese. Anal. Bioanal. Chem. 2016, 408, 5457–5465. [Google Scholar] [CrossRef]
- Zeleny, R.; Emteborg, H.; Charoud-Got, J.; Schimmel, H.; Nia, Y.; Mutel, I.; Ostyn, A.; Herbin, S.; Hennekinne, J.-A. Development of a Reference Material for Staphylococcus Aureus Enterotoxin A in Cheese: Feasibility Study, Processing, Homogeneity and Stability Assessment. Food Chem. 2015, 168, 241–246. [Google Scholar] [CrossRef]
- Available online: https://crm.jrc.ec.europa.eu/p/40456/40486/By-Analyte-Group/Microbiological-Properties-Pathogens/IRMM-359-CHEESE-POWDER-SEA/IRMM-359 (accessed on 9 February 2021).
- Flemming, K.; Ackermann, G. Prevalence of Enterotoxin Producing Staphylococcus Aureus in Stools of Patients with Nosocomial Diarrhea. Infection 2007, 35, 356–358. [Google Scholar] [CrossRef]
- Wang, W.; Liu, L.; Xu, L.; Kuang, H.; Zhu, J.; Xu, C. Gold-Nanoparticle-Based Multiplexed Immunochromatographic Strip for Simultaneous Detection of Staphylococcal Enterotoxin A, B, C, D, and E. Part. Part. Syst. Charact. 2016, 33, 388–395. [Google Scholar] [CrossRef]
- Khreich, N.; Lamourette, P.; Boutal, H.; Devilliers, K.; Créminon, C.; Volland, H. Detection of Staphylococcus Enterotoxin B Using Fluorescent Immunoliposomes as Label for Immunochromatographic Testing. Anal. Biochem. 2008, 377, 182–188. [Google Scholar] [CrossRef] [PubMed]
- Bee, C.; Abdiche, Y.N.; Pons, J.; Rajpal, A. Determining the Binding Affinity of Therapeutic Monoclonal Antibodies towards Their Native Unpurified Antigens in Human Serum. PLoS ONE 2013, 8, e80501. [Google Scholar] [CrossRef]
- Mahmood, R.; Khan, S.A. Role of Upstream Sequences in the Expression of the Staphylococcal Enterotoxin B Gene. J. Biol. Chem. 1990, 265, 4652–4656. [Google Scholar] [CrossRef]
- Tholen, D.; Linnet, K.; Kondratovich, M.; Armbruster, D.; Garrett, P.; Jones, R.; Kroll, M.H.; Lequin, R.; Pankratz, T.; Scassellati, G.A.; et al. Protocols for Determination of Limits of Detection and Limits of Quantitation; Approved Guidelines; NCCLS document EP17-A; NCCLS: Wayne, PA, USA, 2004; ISBN 1-56238-551-8. [Google Scholar]
- Andreasson, U.; Perret-Liaudet, A.; van Waalwijk van Doorn, L.J.C.; Blennow, K.; Chiasserini, D.; Engelborghs, S.; Fladby, T.; Genc, S.; Kruse, N.; Kuiperij, H.B.; et al. A Practical Guide to Immunoassay Method Validation. Front. Neurol. 2015, 6, 179. [Google Scholar] [CrossRef]
- Blaiotta, G.; Ercolini, D.; Pennacchia, C.; Fusco, V.; Casaburi, A.; Pepe, O.; Villani, F. PCR Detection of Staphylococcal Enterotoxin Genes in Staphylococcus Spp. Strains Isolated from Meat and Dairy Products. Evidence for New Variants of SeG and SeI in S. Aureus AB-8802. J. Appl. Microbiol. 2004, 97, 719–730. [Google Scholar] [CrossRef]
- Macori, G.; Bellio, A.; Bianchi, D.M.; Chiesa, F.; Gallina, S.; Romano, A.; Zuccon, F.; Cabrera-Rubio, R.; Cauquil, A.; Merda, D.; et al. Genome-Wide Profiling of Enterotoxigenic Staphylococcus Aureus Strains Used for the Production of Naturally Contaminated Cheeses. Genes 2020, 11, 33. [Google Scholar] [CrossRef]
- O’ Farrell, B. Lateral Flow Immunoassay Systems: Evolution from the Current State of the Art to the Next Generation of Highly Sensitive, Quantitative Rapid Assays. In The Immunoassay Handbook; Elsevier: Amsterdam, The Netherlands, 2013; pp. 89–107. [Google Scholar] [CrossRef]
- Freed, R.C.; Evenson, M.L.; Reiser, R.F.; Bergdoll, M.S. Enzyme-Linked Immunosorbent Assay for Detection of Staphylococcal Enterotoxins in Foods. Appl. Environ. Microbiol. 1982, 44, 1349–1355. [Google Scholar] [CrossRef]
- Shinagawa, K.; Kanazawa, T.; Matsusaka, N.; Sugii, S.; Nagata, K. Murine Monoclonal Antibodies Reactive with Staphylococcal Enterotoxins A, B, C2, D, and E. FEMS Microbiol. Lett. 1991, 64, 35–39. [Google Scholar] [CrossRef][Green Version]
- Liang, B.; Zhang, Y.; Liu, A.; Zhou, Y.; Chen, F.; Wang, X. Production of a Monoclonal Antibody by Simultaneous Immunization of Staphylococcal Enterotoxin A and B. Appl. Biochem. Biotechnol. 2011, 164, 831–840. [Google Scholar] [CrossRef]
- Kuang, H.; Wang, W.; Xu, L.; Ma, W.; Liu, L.; Wang, L.; Xu, C. Monoclonal Antibody-Based Sandwich ELISA for the Detection of Staphylococcal Enterotoxin A. Int. J. Environ. Res. Public Health 2013, 10, 1598–1608. [Google Scholar] [CrossRef]
- Lin, Y.S.; Largen, M.T.; Newcomb, J.R.; Rogers, T.J. Production and Characterisation of Monoclonal Antibodies Specific for Staphylococcal Enterotoxin B. J. Med. Microbiol. 1988, 27, 263–270. [Google Scholar] [CrossRef]
- Goyache, J.; Orden, J.A.; Blanco, J.L.; Domenech, A.; Hernandez, J.; Suarez, G.; Gomez-Lucia, E. Determination of the Reactivities and Cross-Reactivities of Monoclonal Antibodies against Staphylococcal Enterotoxin A by Indirect ELISA and Immunoblot Including a Semiautomated Electrophoresis System. Lett. Appl. Microbiol. 1992, 14, 217–220. [Google Scholar] [CrossRef]
- Köhler, G.; Milstein, C. Continuous Cultures of Fused Cells Secreting Antibody of Predefined Specificity. Nature 1975, 256, 495–497. [Google Scholar] [CrossRef] [PubMed]
- Grassi, J.; Frobert, Y.; Pradelles, P.; Chercuitte, F.; Gruaz, D.; Dayer, J.-M.; Poubelle, P.E. Production of Monoclonal Antibodies against Interleukin-1α and -1β: Development of Two Enzyme Immunometric Assays (EIA) Using Acetylcholinesterase and Their Application to Biological Media. J. Immunol. Methods 1989, 123, 193–210. [Google Scholar] [CrossRef]
- Grassi, J.; Créminon, C.; Frobert, Y.; Frétier, P.; Turbica, I.; Rezaei, H.; Hunsmann, G.; Comoy, E.; Deslys, J.P. Specific Determination of the Proteinase K-Resistant Form of the Prion Protein Using Two-Site Immunometric Assays. Application to the Post-Mortem Diagnosis of BSE. Arch. Virol. Suppl. 2000, 197–205. [Google Scholar] [CrossRef]
- Boutal, H.; Naas, T.; Devilliers, K.; Oueslati, S.; Dortet, L.; Bernabeu, S.; Simon, S.; Volland, H. Development and Validation of a Lateral Flow Immunoassay for Rapid Detection of NDM-Producing Enterobacteriaceae. J. Clin. Microbiol. 2017, 55, 2018–2029. [Google Scholar] [CrossRef]


| S. aureus Strain | SE Genes | Origin and Source |
|---|---|---|
| 07HMPA50 | Nontoxic coagulase-positive S. aureus | Commercial Camembert 1998 (NRL for CPS, ANSES, France) |
| FRI S6 | sea, seb | Reference strain Food Research Institute, Frozen shrimp (M. S. Bergdoll [49]) |
| A900322 | seg, sei, sep | Reference strain (Clinical strain from a patient with TSS, NRC for staphylococcus, France) |
| 16SBCL1204 | seg, sei | Food poisoning outbreak, rice chicken (NRL for CPS, ANSES, France) |
| 15SBCL1428 | seg, sei | Food poisoning outbreak, cheese (NRL for CPS, ANSES, France) |
| FRI 569 | seh | Reference strain Food Research Institute Nasal swab (Y.-C. Su, A.C.L. Wong [31]) |
| 12CEB51 | seh | Food poisoning outbreak, Pot au feu (NRL for CPS, ANSES, France) |
| 14SBCL881 | seh | Food poisoning outbreak, mixed salad (NRL for CPS, ANSES, France) |
| Target Toxin | mAb Pair | Theoretical LoD (pg/mL) | Theoretical LoQ (pg/mL) |
|---|---|---|---|
| Lab-made rec. SEA | SEA7/SEA5-biot | 2 | 10 |
| Commercial SEA | 3 | 14 | |
| Lab-made rec. SEA | SEA7/SEA12-biot | 7 | 35 |
| Commercial SEA | 24 | 100 | |
| Lab-made rec. SEG | SEG21/SEG26-biot | 6 | 23 |
| SEG21/SEG27-biot | 5 | 16 | |
| SEG26/SEG27-biot | N.C. | N.C. | |
| SEG41/SEG27-biot | 8 | 24 | |
| SEG21/SEG28-biot | 30 | 105 | |
| SEG26/SEG28-biot | 3 | 9 | |
| SEG32/SEG28-biot | 4 | 9 | |
| SEG41/SEG28-biot | 4 | 10 | |
| Lab-made rec. SEH | SEH1/SEH14-biot | 3 | 11 |
| SEH16/SEH14-biot | 13 | 50 | |
| SEH19/SEH14-biot | 47 | 135 | |
| SEH1/SEH19-biot | 2 | 11 | |
| SEH6/SEH19-biot | 148 | 346 | |
| SEH11/SEH19-biot | 5 | 22 | |
| SEH14/SEH19-biot | 53 | 179 | |
| Lab-made rec. SEI | SEI27/SEI26-biot | 63 | 163 |
| SEI36/SEI26-biot | 59 | 163 | |
| SEI39/SEI26-biot | 34 | 103 | |
| SEI44/SEI26-biot | 58 | 160 | |
| SEI27/SEI32-biot | 57 | 184 | |
| SEI36/SEI32-biot | 41 | 122 | |
| SEI39/SEI32-biot | 41 | 131 | |
| SEI44/SEI32-biot | 30 | 95 |
| Capture/Tracer mAb Pair | |||||||
|---|---|---|---|---|---|---|---|
| SEA7/SEA5-biot | SEG41/SEG27-biot | SEH1/SEH19-biot | SEI27/SEI26-biot | ||||
| Lab-Made Recombinant SEA | Commercial SEA | Lab-Made Recombinant SEG | Lab-Made Recombinant SEH | Lab-Made Recombinant SEI | |||
| Calculated estimation using fitting curves | Theoretical LoD | (pg/mL) | 2.0 ± 1.3 (n = 20) | 5.4 ± 0.7 (n = 5) | 0.21 ± 0.09 (n = 12) | 0.59 ± 0.26 (n = 12) | 3.0 ± 1.5 (n = 12) |
| (fM) | 70.7 ± 46.0 (n = 20) | 199.4 ± 25.8 (n = 5) | 7.4 ± 3.2 (n = 12) | 22.4 ± 9.9 (n = 12) | 114.9 ± 57.4 (n = 12) | ||
| Theoretical LoQ | (pg/mL) | 6.0 ± 3.0 (n = 20) | 17.3 ± 6.1 (n = 5) | 0.66 ± 0.16 (n = 12) | 1.73 ± 0.72 (n = 12) | 10.1 ± 5.1 (n = 12) | |
| (fM) | 212.1 ± 106.0 (n = 20) | 638.8 ± 225.3 (n = 5) | 23.4 ± 5.7 (n = 12) | 65.7 ± 27.4 (n = 12) | 386.7 ± 195.3 (n = 12) | ||
| Experimental evaluation | Experimental LoD | (pg/mL) | 4.1 (100%, n = 28) | 12.3 (100%, n = 5) | 0.41 (100%, n = 17) | 1.37 (100%, n = 17) | 4.1 (94.1%, n = 17) |
| (fM) | 144.9 (100%, n = 28) | 454.2 (100%, n = 5) | 14.5 (100%, n = 17) | 52.1 (100%, n = 17) | 157.0 (94.1%, n = 17) | ||
| Experimental LoQ | (pg/mL) | 12.3 (100%, n = 28) | 37.0 (100%, n = 5) | 1.23 (100%, n = 17) | 4.12 (94.1%, n = 17) | 37.0 (100%, n = 17) | |
| (fM) | 434.8 (100%, n = 28) | 1366.3 (100%, n = 5) | 43.6 (100%, n = 17) | 156.6 (94.1%, n = 17) | 1416.8 (100%, n = 17) | ||
| Target Toxin | Quality Control Standard Concentration | Accuracy (bias) | Repeatability (CV) | In-House Reproducibility (CV) |
|---|---|---|---|---|
| Commercial SEA | 25 pg/mL (0.92 pM) | 101.3% | 14.1% | 15.9% |
| 50 pg/mL (1.85 pM) | 97.0% | 6.1% | 13.8% | |
| 100 pg/mL (3.69 pM) | 93.3% | 6.4% | 7.2% | |
| 300 pg/mL (11.08 pM) | 97.1% | 4.6% | 4.7% | |
| 1000 pg/mL (36.93 pM) | 96.7% | 5.2% | 5.5% | |
| Lab-made recombinant SEA | 25 pg/mL (0.88 pM) | 95.0% | 8.2% | 9.5% |
| 50 pg/mL (1.77 pM) | 95.5% | 7.0% | 8.0% | |
| 100 pg/mL (3.53 pM) | 91.5% | 6.8% | 6.7% | |
| 300 pg/mL (10.60 pM) | 92.1% | 7.9% | 7.9% | |
| 1000 pg/mL (35.35 pM) | 91.3% | 9.6% | 8.9% | |
| Lab-made recombinant SEG | 3 pg/mL (0.11 pM) | 90.7% | 6.9% | 8.2% |
| 5 pg/mL (0.18 pM) | 91.6% | 8.3% | 9.4% | |
| 10 pg/mL (0.35 pM) | 90.5% | 6.5% | 8.3% | |
| 30 pg/mL (1.06 pM) | 93.0% | 6.5% | 8.9% | |
| 60 pg/mL (2.12 pM) | 94.7% | 6.9% | 9.8% | |
| Lab-made recombinant SEH | 5 pg/mL (0.19 pM) | 87.1% | 9.1% | 10.6% |
| 10 pg/mL (0.38 pM) | 89.0% | 7.1% | 12.6% | |
| 30 pg/mL (1.14 pM) | 88.0% | 6.1% | 7.6% | |
| 100 pg/mL (3.80 pM) | 89.9% | 5.4% | 6.3% | |
| 300 pg/mL (11.4 pM) | 88.3% | 7.0% | 7.8% | |
| Lab-made recombinant SEI | 30 pg/mL (1.15 pM) | 88.8% | 8.6% | 8.8% |
| 50 pg/mL (1.91 pM) | 90.8% | 6.4% | 7.8% | |
| 100 pg/mL (3.83 pM) | 89.9% | 5.1% | 6.8% | |
| 300 pg/mL (11.49 pM) | 88.9% | 5.1% | 6.1% | |
| 1000 pg/mL (38.29 pM) | 88.3% | 3.7% | 5.3% |
| mAb pair | Target SE | Measured SE Concentrations (ng/mL eq. Recombinant SE) in Culture Supernatant from Strain: | |||||||
|---|---|---|---|---|---|---|---|---|---|
| 07HMPA50 | FRI S6 | A900322 | 16SBCL1204 | 15SBCL1428 | FRI 569 | 12CEB51 | 14SBCL881 | ||
| SEA7/SEA5-biot | SEA | <LoD | 0.18 ± 0.02 | <LoD | LoD | <LoD | <LoD | <LoD | <LoD |
| SEB27/SEB26-biot | SEB | <LoD | 13400 ± 2263 | <LoD | <LoD | <LoD | <LoD | <LoD | <LoD |
| SEG41/SEG27-biot | SEG | <LoD | <LoD | 2.6 ± 0.9 | 5.3 ± 1.6 | 3.3 ± 0.4 | <LoD | <LoD | <LoD |
| SEH1/SEH19-biot | SEH | <LoD | <LoD | <LoD | <LoD | <LoD | 103.6 ± 41.2 | 43.9 ± 14.4 | 82.7 ± 33.5 |
| SEI27/SEI126-biot | SEI | <LoD | <LoD | 8.1 ± 7.7 | 1.9 ± 0.6 | 5.7 ± 0.1 | <LoD | <LoD | <LoD |
| Context | Sample | EN ISO 19020 | Enzyme Immunoassays | Expected Results | ||||
|---|---|---|---|---|---|---|---|---|
| (Qualitative Detection) | ||||||||
| Vidas SETII (TV(a)) | Ridascreen SET Total (AU(b)) | SEA (pg/g) | SEG (pg/g) | SEH (pg/g) | SEI (pg/g) | |||
| Certified reference materials [42,43,44] | IRMM-359a cheese | not detected | not detected | <LoD (c) | <LoD (c) | <LoD (c) | <LoD (c) | SEA, SEG, SEH, and SEI not detected |
| Vidas SET II: SEA to SEE not detected | ||||||||
| <LoD (d) | <LoD (d) | <LoD (d) | <LoD (d) | Ridascreen ST: SEA to SEE not detected | ||||
| IRMM-359b cheese | 1.35 TV (positive) | 0.96 AU (positive) | 38.4 (c) | <LoD (c) | <LoD(c) | <LoD (c) | 42 pg SEA/g (interval: 29–59 pg/g) | |
| Vidas SET II: 1.14 TV (0.47–1.53 TV) | ||||||||
| 41.8 (d) | <LoD (d) | <LoD(d) | <LoD (d) | Ridascreen ST: 0.61 AU (0.28– 1.11 AU) | ||||
| IRMM-359c cheese | 2.08 TV (positive) | 2.03 AU (positive) | 115.4 (c) | <LoD (c) | <LoD (c) | <LoD (c) | 102 pg SEA/g (interval: 81–145 pg/g) | |
| Vidas SET II: 1.97 TV (1.10–2.42 TV) | ||||||||
| 110.5 (d) | <LoD(d) | <LoD (d) | <LoD (d) | Ridascreen ST: 1.36 AU (0.45–2.31 AU) | ||||
| Samples from French CPS NRL collection | Emmental cheese 55 | 1.45 TV (positive) | 0.92 AU (positive) | 123.3 (c) | <LoD (c) | <LoD (c) | <LoD (c) | 80 pg SEA/g (spiking concentration) |
| Emmental Cheese 469 | 1.45 TV (positive) | 0.92 AU (positive) | 185.4 (c) | <LoD (c) | <LoD (c) | <LoD (c) | 80 pg SEA/g (spiking concentration) | |
| Morbier cheese 08BAC553 | 1.06 TV (positive) | 0.66 AU (positive) | <LoD (c) | <LoD (c) | <LoD (c) | <LoD (c) | Natural contamination with 180 pg SED/g | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Féraudet Tarisse, C.; Goulard-Huet, C.; Nia, Y.; Devilliers, K.; Marcé, D.; Dambrune, C.; Lefebvre, D.; Hennekinne, J.-A.; Simon, S. Highly Sensitive and Specific Detection of Staphylococcal Enterotoxins SEA, SEG, SEH, and SEI by Immunoassay. Toxins 2021, 13, 130. https://doi.org/10.3390/toxins13020130
Féraudet Tarisse C, Goulard-Huet C, Nia Y, Devilliers K, Marcé D, Dambrune C, Lefebvre D, Hennekinne J-A, Simon S. Highly Sensitive and Specific Detection of Staphylococcal Enterotoxins SEA, SEG, SEH, and SEI by Immunoassay. Toxins. 2021; 13(2):130. https://doi.org/10.3390/toxins13020130
Chicago/Turabian StyleFéraudet Tarisse, Cécile, Céline Goulard-Huet, Yacine Nia, Karine Devilliers, Dominique Marcé, Chloé Dambrune, Donatien Lefebvre, Jacques-Antoine Hennekinne, and Stéphanie Simon. 2021. "Highly Sensitive and Specific Detection of Staphylococcal Enterotoxins SEA, SEG, SEH, and SEI by Immunoassay" Toxins 13, no. 2: 130. https://doi.org/10.3390/toxins13020130
APA StyleFéraudet Tarisse, C., Goulard-Huet, C., Nia, Y., Devilliers, K., Marcé, D., Dambrune, C., Lefebvre, D., Hennekinne, J.-A., & Simon, S. (2021). Highly Sensitive and Specific Detection of Staphylococcal Enterotoxins SEA, SEG, SEH, and SEI by Immunoassay. Toxins, 13(2), 130. https://doi.org/10.3390/toxins13020130





