Feedborne Mycotoxins Beauvericin and Enniatins and Livestock Animals
Abstract
1. Introduction
2. Beauvericin
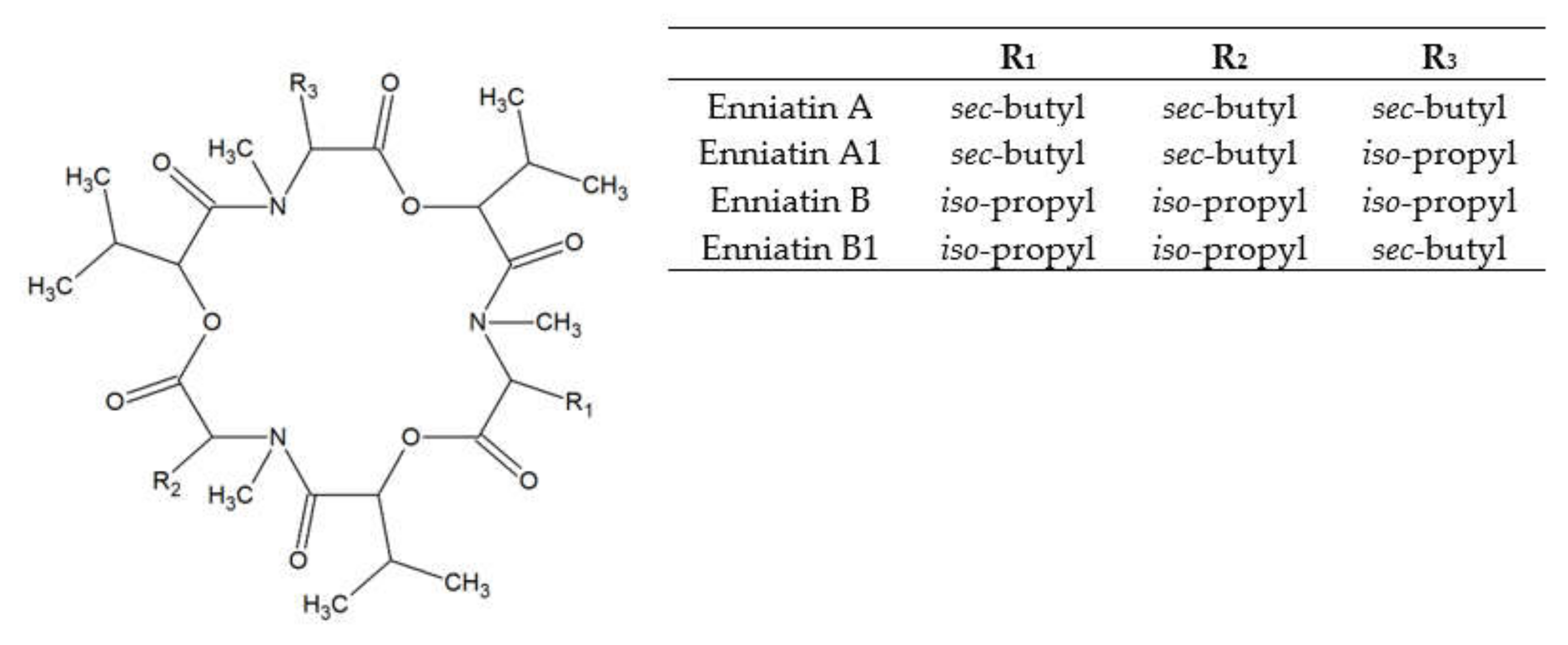
3. Enniatins
4. Presence of Beauvericin and Enniatins in Feedstuffs
5. Metabolism of Beauvericin and Enniatins
6. Occurrence of Beauvericin and Enniatins in Foods of Animal Origin
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Pitt, J.I. Toxigenic fungi and mycotoxins. Br. Med. Bull. 2000, 56, 184–192. [Google Scholar] [CrossRef] [PubMed]
- Reverberi, M.; Ricelli, A.; Zjalic, S.; Fabbri, A.A.; Fanelli, C. Natural functions of mycotoxins and control of their biosynthesis in fungi. Appl. Microbiol. Biotechnol. 2010, 87, 899–911. [Google Scholar] [CrossRef] [PubMed]
- Escrivá, L.; Font, G.; Manyes, L.; Berrada, H. Studies on the Presence of Mycotoxins in Biological Samples: An Overview. Toxins 2017, 9, 251. [Google Scholar] [CrossRef] [PubMed]
- Guerre, P. Fusariotoxins in Avian Species: Toxicokinetics, Metabolism and Persistence in Tissues. Toxins 2015, 7, 2289–2305. [Google Scholar] [CrossRef]
- Monti, S.M.; Fogliano, V.; Logrieco, A.; Ferracane, R.; Ritieni, A. Simultaneous Determination of Beauvericin, Enniatins, and Fusaproliferin by High Performance Liquid Chromatography. J. Agric. Food Chem. 2000, 48, 3317–3320. [Google Scholar] [CrossRef]
- Capriotti, A.L.; Caruso, G.; Cavaliere, C.; Foglia, P.; Samperi, R.; Laganà, A. Multiclass mycotoxin analysis in food, environmental and biological matrices with chromatography/mass spectrometry. Mass Spectrom. Rev. 2012, 31, 466–503. [Google Scholar] [CrossRef]
- Bryden, W.L. Mycotoxin contamination of the feed supply chain: Implications for animal productivity and feed security. Anim. Feed Sci. Technol. 2012, 173, 134–158. [Google Scholar] [CrossRef]
- European Food Safety Authority (EFSA). Opinion of the Scientific Panel on contaminants in the food chain [CONTAM] related to Aflatoxin B1 as undesirable substance in animal feed. EFSA J. 2004, 2, 39. [Google Scholar] [CrossRef]
- European Food Safety Authority (EFSA). Opinion of the Scientific Panel on contaminants in the food chain [CONTAM] related to Deoxynivalenol (DON) as undesirable substance in animal feed. EFSA J. 2004, 2, 73. [Google Scholar] [CrossRef]
- European Food Safety Authority (EFSA). Opinion of the Scientific Panel on contaminants in the food chain [CONTAM] related to Zearalenone as undesirable substance in animal feed. EFSA J. 2004, 2, 89. [Google Scholar] [CrossRef]
- European Food Safety Authority (EFSA). Opinion of the Scientific Panel on contaminants in the food chain [CONTAM] related to ochratoxin A (OTA) as undesirable substance in animal feed. EFSA J. 2004, 2, 101. [Google Scholar] [CrossRef]
- European Food Safety Authority (EFSA). Opinion of the Scientific Panel on contaminants in the food chain [CONTAM] related to fumonisins as undesirable substances in animal feed. EFSA J. 2005, 3, 235. [Google Scholar] [CrossRef]
- Hamill, R.L.; Higgens, C.E.; Boaz, H.E.; Gorman, M. Structure of Beauvericin, a New Depsipeptide Antibiotic Toxic to Artemia Salina. Tetrahedron Lett. 1969, 49, 4255–4258. [Google Scholar] [CrossRef]
- Gupta, S.; Krasnoff, S.B.; Underwood, N.L.; Renwick, J.A.A.; Roberts, D.W. Isolation of beauvericin as an insect toxin from Fusarium semitectum and Fusarium moniliforme var. subglutinans. Mycopathologia 1991, 115, 185–189. [Google Scholar] [CrossRef] [PubMed]
- Santini, A.; Meca, G.; Uhlig, S.; Ritieni, A. Fusaproliferin, beauvericin and enniatins: Occurrence in food—A review. World Mycotoxin J. 2012, 5, 71–81. [Google Scholar] [CrossRef]
- Paciolla, C.; Dipierro, N.; Mulè, G.; Logrieco, A.; Dipierro, S. The mycotoxins beauvericin and T-2 induce cell death and alteration to the ascorbate metabolism in tomato protoplasts. Physiol. Mol. Plant Pathol. 2004, 65, 49–56. [Google Scholar] [CrossRef]
- García-Herranz, V.; Valdehita, A.; Navas, J.M.; Fernández-Cruz, M.L. Cytotoxicity against Fish and Mammalian Cell Lines and Endocrine Activity of the Mycotoxins Beauvericin, Deoxynivalenol and Ochratoxin-A. Food Chem. Toxicol. 2019, 127, 288–297. [Google Scholar] [CrossRef]
- Juan, C.; Manyes, L.; Font, G.; Juan-García, A. Evaluation of immunologic effect of Enniatin A and quantitative determination in feces, urine and serum on treated Wistar rats. Toxicon 2014, 87, 45–53. [Google Scholar] [CrossRef]
- Calo’, L.; Fornelli, F.; Nenna, S.; Tursi, A.; Caiaffa, M.F.; Macchia, L. Beauvericin cytotoxicity to the invertebrate cell line SF-9. J. Appl. Genet. 2003, 44, 515–520. [Google Scholar]
- Macchia, L.; Caiffa, M.F.; Fornelli, F.; Calo, L.; Nenna, S.; Moretti, A.; Logrieco, A.; Tursi, A. Apoptosis induced by the Fusarium mycotoxin beauvericin in mammalian cells. Appl. Genet. 2002, 43, 363–371. [Google Scholar]
- Taevernier, L.; Veryser, L.; Roche, N.; Peremans, K.; Burvenich, C.; Delesalle, C.; De Spiegeleer, B. Human skin permeation of emerging mycotoxins (beauvericin and enniatins). J. Expo. Sci. Environ. Epidemiol. 2016, 26, 277–287. [Google Scholar] [CrossRef] [PubMed]
- Olleik, H.; Nicoletti, C.; Lafond, M.; Courvoisier-Dezord, E.; Xue, P.; Hijazi, A.; Baydoun, E.; Perrier, J.; Maresca, M. Comparative Structure–Activity Analysis of the Antimicrobial Activity, Cytotoxicity, and Mechanism of Action of the Fungal Cyclohexadepsipeptides Enniatins and Beauvericin. Toxins 2019, 11, 514. [Google Scholar] [CrossRef] [PubMed]
- Escrivá, L.; Font, G.; Manyes, L. In vivo toxicity studies of fusarium mycotoxins in the last decade: A review. Food Chem. Toxicol. 2015, 78, 185–206. [Google Scholar] [CrossRef] [PubMed]
- Meca, G.; Sospedra, I.; Soriano, J.M.; Ritieni, A.; Moretti, A.; Mañes, J. Antibacterial effect of the bioactive compound beauvericin produced by Fusarium proliferatum on solid medium of wheat. Toxicon 2010, 56, 349–354. [Google Scholar] [CrossRef]
- Madhyastha, M.S.; Marquardt, R.R.; Frohlich, A.A.; Borsa, J. Optimization of Yeast Bioassay for Trichothecene Mycotoxins. J. Food Prot. 1994, 57, 490–495. [Google Scholar] [CrossRef] [PubMed]
- Castlebury, L.A.; Sutherland, J.B.; Tanner, L.A.; Henderson, A.L.; Cerniglia, C.E. Short Communication: Use of a bioassay to evaluate the toxicity of beauvericin to bacteria. World J. Microbiol. Biotechnol. 1999, 15, 131–133. [Google Scholar] [CrossRef]
- Wu, X.-F.; Xu, R.; Ouyang, Z.-J.; Qian, C.; Shen, Y.; Wu, X.-D.; Gu, Y.-H.; Xu, Q.; Sun, Y. Beauvericin Ameliorates Experimental Colitis by Inhibiting Activated T Cells via Downregulation of the PI3K/Akt Signaling Pathway. PLoS ONE 2013, 8. [Google Scholar] [CrossRef]
- Plattner, P.A.; Nager, U. Über die Chemie des Enniatins. Experientia 1947, 3, 325–326. [Google Scholar] [CrossRef]
- Blais, L.A.; Simon, J.W.A.; Blackwell, B.A.; Greenhalgh, R.; Miller, J.D. Isolation and characterization of enniatins from Fusarium avenaceum DAOM 196490. Can. J. Chem. 1992, 70, 1281–1287. [Google Scholar] [CrossRef]
- Sy-Cordero, A.A.; Pearce, C.J.; Oberlies, N.H. Revisiting the enniatins: A review of their isolation, biosynthesis, structure determination and biological activities. J. Antibiot. 2012, 65, 541–549. [Google Scholar] [CrossRef]
- Supothina, S.; Isaka, M.; Kirtikara, K.; Tanticharoen, M.; Thebtaranonth, Y. Enniatin Production by the Entomopathogenic Fungus Verticillium hemipterigenum BCC 1449. J. Antibiot. 2004, 57, 732–738. [Google Scholar] [CrossRef] [PubMed]
- Morrison, E.; Kosiak, B.; Ritieni, A.; Aastveit, A.H.; Uhlig, S.; Bernhoft, A. Mycotoxin Production by Fusarium avenaceum Strains Isolated from Norwegian Grain and the Cytotoxicity of Rice Culture Extracts to Porcine Kidney Epithelial Cells. J. Agric. Food Chem. 2002, 50, 3070–3075. [Google Scholar] [CrossRef] [PubMed]
- Firakova, S.; Šturdíková, M.; Liptaj, T.; Prónayová, N.; Bezáková, L.; Proksa, B. Enniatins produced by Fusarium dimerum, an endophytic fungal strain. Pharmazie 2008, 63, 539–541. [Google Scholar] [CrossRef] [PubMed]
- Kabak, B.; Dobson, A.D.W.; Var, I. Strategies to Prevent Mycotoxin Contamination of Food and Animal Feed: A Review. Crit. Rev. Food Sci. Nutr. 2006, 46, 593–619. [Google Scholar] [CrossRef] [PubMed]
- Grove, J.F.; Pople, M. The insecticidal activity of beauvericin and the enniatin complex. Mycopathologia 1980, 70, 103–105. [Google Scholar] [CrossRef]
- Meca, G.; Sospedra, I.; Valero, M.A.; Mañes, J.; Font, G.; Ruiz, M.J. Antibacterial Activity of the Enniatin B, Produced by Fusarium Tricinctum in Liquid Culture, and Cytotoxic Effects on Caco-2 Cells. Toxicol. Mech. Methods 2011, 21, 503–512. [Google Scholar] [CrossRef]
- Roig, M.; Meca, G.; Marín, R.; Ferrer, E.; Mañes, J. Antibacterial Activity of the Emerging Fusarium Mycotoxins Enniatins A, A1, A2, B, B1, and B4 on Probiotic Microorganisms. Toxicon 2014, 85, 1–4. [Google Scholar] [CrossRef]
- Ivanova, L.; Skjerve, E.; Eriksen, G.S.; Uhlig, S. Cytotoxicity of enniatins A, A1, B, B1, B2 and B3 from Fusarium avenaceum. Toxicon 2006, 47, 868–876. [Google Scholar] [CrossRef]
- Lu, H.; Fernández-Franzón, M.; Font, G.; Ruiz, M.J. Toxicity evaluation of individual and mixed enniatins using an in vitro method with CHO-K1 cells. Toxicol. In Vitro 2013, 27, 672–680. [Google Scholar] [CrossRef]
- Sotnichenko, A.; Pantsov, E.; Shinkarev, D.; Okhanov, V. Hydrophobized Reversed-Phase Adsorbent for Protection of Dairy Cattle against Lipophilic Toxins from Diet. Efficiensy In Vitro and In Vivo. Toxins 2019, 11, 256. [Google Scholar] [CrossRef]
- Kouri, K.; Lemmens, M.; Lemmens-Gruber, R. Beauvericin-induced channels in ventricular myocytes and liposomes. Biochim. Biophys. Acta BBA Biomembr. 2003, 1609, 203–210. [Google Scholar] [CrossRef]
- Uhlig, S.; Ivanova, L.; Petersen, D.; Kristensen, R. Structural studies on minor enniatins from Fusarium sp. VI 03441: Novel N-methyl-threonine containing enniatins. Toxicon 2009, 53, 734–742. [Google Scholar] [CrossRef] [PubMed]
- Kouri, K.; Duchen, M.R.; Lemmens-Gruber, R. Effects of Beauvericin on the Metabolic State and Ionic Homeostasis of Ventricular Myocytes of the Guinea Pig. Chem. Res. Toxicol. 2005, 18, 1661–1668. [Google Scholar] [CrossRef] [PubMed]
- Uhlig, S.; Jestoi, M.; Parikka, P. Fusarium avenaceum—The North European situation. Int. J. Food Microbiol. 2007, 119, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Behm, C.; Degen, G.H.; Föllmann, W. The Fusarium toxin enniatin B exerts no genotoxic activity, but pronounced cytotoxicity in vitro. Mol. Nutr. Food Res. 2009, 53, 423–430. [Google Scholar] [CrossRef] [PubMed]
- Kamyar, M.; Rawnduzi, P.; Studenik, C.R.; Kouri, K.; Lemmens-Gruber, R. Investigation of the electrophysiological properties of enniatins. Arch. Biochem. Biophys. 2004, 429, 215–223. [Google Scholar] [CrossRef]
- Oliveira, C.A.F.; Ivanova, L.; Solhaug, A.; Fæste, C.K. Enniatin B1-Induced Lysosomal Membrane Permeabilization in Mouse Embryonic Fibroblasts. Mycotoxin Res. 2020, 36, 23–30. [Google Scholar] [CrossRef]
- Feed & Food Statistical Yearbook. 2018. Available online: https://fefac.eu/wp-content/uploads/2020/07/feedfood2018.pdf (accessed on 3 November 2020).
- Schenck, J.; Müller, C.; Djurle, A.; Jensen, D.F.; O’Brien, M.; Johansen, A.; Rasmussen, P.H.; Spörndly, R. Occurrence of filamentous fungi and mycotoxins in wrapped forages in Sweden and Norway and their relation to chemical composition and management. Grass Forage Sci. 2019, 74, 613–625. [Google Scholar] [CrossRef]
- Ivić, D.; Domijan, A.-M.; Peraica, M.; Miličević, T.; Cvjetković, B. Fusarium spp. Contamination of Wheat, Maize, Soybean, and Pea Grain in Croatia. Arch. Ind. Hyg. Toxicol. 2009, 60, 435–442. [Google Scholar] [CrossRef]
- Scudamore, K.A.; Livesey, C.T. Occurrence and Significance of Mycotoxins in Forage Crops and Silage: A Review. J. Sci. Food Agric. 1998, 77, 1–17. [Google Scholar] [CrossRef]
- McElhinney, C.; Danaher, M.; Elliott, C.T.; O’Kiely, P. On-farm factors relating to mycotoxin occurrence and other chemical compositional traits in grass silages in Ireland. World Mycotoxin J. 2016, 9, 505–516. [Google Scholar] [CrossRef]
- Jestoi, M.; Rokka, M.; Yli-Mattila, T.; Parikka, P.; Rizzo, A.; Peltonen, K. Presence and concentrations of the Fusarium-related mycotoxins beauvericin, enniatins and moniliformin in finnish grain samples. Food Addit. Contam. 2004, 21, 794–802. [Google Scholar] [CrossRef] [PubMed]
- Uhlig, S.; Torp, M.; Heier, B.T. Beauvericin and enniatins A, A1, B and B1 in Norwegian grain: A survey. Food Chem. 2006, 94, 193–201. [Google Scholar] [CrossRef]
- Chrpová, J.; Šíp, V.; Sumíková, T.; Salava, J.; Palicová, J.; Štočková, L.; Džuman, Z.; Hajšlová, J. Occurrence of Fusarium species and mycotoxins in wheat grain collected in the Czech Republic. World Mycotoxin J. 2016, 9, 317–327. [Google Scholar] [CrossRef]
- Svingen, T.; Lund Hansen, N.; Taxvig, C.; Vinggaard, A.M.; Jensen, U.; Have Rasmussen, P. Enniatin B and beauvericin are common in Danish cereals and show high hepatotoxicity on a high-content imaging platform. Environ. Toxicol. 2017, 32, 1658–1664. [Google Scholar] [CrossRef] [PubMed]
- Abdallah, M.F.; Girgin, G.; Baydar, T.; Krska, R.; Sulyok, M. Occurrence of multiple mycotoxins and other fungal metabolites in animal feed and maize samples from Egypt using LC-MS/MS: Toxic fungal and bacterial metabolites in feed and maize from Egypt. J. Sci. Food Agric. 2017, 97, 4419–4428. [Google Scholar] [CrossRef]
- Bertero, A.; Fossati, P.; Tedesco, D.E.A.; Caloni, F. Beauvericin and Enniatins: In Vitro Intestinal Effects. Toxins 2020, 12, 686. [Google Scholar] [CrossRef]
- Orlando, B.; Grignon, G.; Vitry, C.; Kashefifard, K.; Valade, R. Fusarium Species and Enniatin Mycotoxins in Wheat, Durum Wheat, Triticale and Barley Harvested in France. Mycotoxin Res. 2019, 35, 369–380. [Google Scholar] [CrossRef]
- Mortensen, A.; Granby, K.; Eriksen, F.D.; Cederberg, T.L.; Friis-Wandall, S.; Simonsen, Y.; Broesbøl-Jensen, B.; Bonnichsen, R. Levels and Risk Assessment of Chemical Contaminants in Byproducts for Animal Feed in Denmark. J. Environ. Sci. Health Part B 2014, 49, 797–810. [Google Scholar] [CrossRef]
- Mastanjević, K.; Lukinac, J.; Jukić, M.; Šarkanj, B.; Krstanović, V.; Mastanjević, K. Multi-(Myco)Toxins in Malting and Brewing By-Products. Toxins 2019, 11, 30. [Google Scholar] [CrossRef]
- Arroyo-Manzanares, N.; Rodríguez-Estévez, V.; Arenas-Fernández, P.; García-Campaña, A.M.; Gámiz-Gracia, L. Occurrence of Mycotoxins in Swine Feeding from Spain. Toxins 2019, 11, 342. [Google Scholar] [CrossRef] [PubMed]
- Juan, C.; Oueslati, S.; Mañes, J.; Berrada, H. Multimycotoxin Determination in Tunisian Farm Animal Feed. J. Food Sci. 2019, 84, 3885–3893. [Google Scholar] [CrossRef] [PubMed]
- Novak, B.; Rainer, V.; Sulyok, M.; Haltrich, D.; Schatzmayr, G.; Mayer, E. Twenty-Eight Fungal Secondary Metabolites Detected in Pig Feed Samples: Their Occurrence, Relevance and Cytotoxic Effects In Vitro. Toxins 2019, 11, 537. [Google Scholar] [CrossRef] [PubMed]
- Shimshoni, J.A.; Cuneah, O.; Sulyok, M.; Krska, R.; Galon, N.; Sharir, B.; Shlosberg, A. Mycotoxins in corn and wheat silage in Israel. Food Addit. Contam. Part A 2013, 30, 1614–1625. [Google Scholar] [CrossRef] [PubMed]
- Reisinger, N.; Schürer-Waldheim, S.; Mayer, E.; Debevere, S.; Antonissen, G.; Sulyok, M.; Nagl, V. Mycotoxin Occurrence in Maize Silage—A Neglected Risk for Bovine Gut Health? Toxins 2019, 11, 577. [Google Scholar] [CrossRef] [PubMed]
- Zachariasova, M.; Dzuman, Z.; Veprikova, Z.; Hajkova, K.; Jiru, M.; Vaclavikova, M.; Zachariasova, A.; Pospichalova, M.; Florian, M.; Hajslova, J. Occurrence of multiple mycotoxins in European feedingstuffs, assessment of dietary intake by farm animals. Anim. Feed Sci. Technol. 2014, 193, 124–140. [Google Scholar] [CrossRef]
- McElhinney, C.; Danaher, M.; Elliott, C.T.; O’Kiely, P. Mycotoxins in farm silages—A 2-year Irish national survey. Grass Forage Sci. 2016, 71, 339–352. [Google Scholar] [CrossRef]
- Tangni, E.K.; Pussemier, L.; Van Hove, F. Mycotoxin contaminating maize and grass silages for dairy cattle feeding: Current state and challenges. J. Anim. Sci. Adv. 2013, 23, 492–511. [Google Scholar]
- Lindblad, M.; Gidlund, A.; Sulyok, M.; Börjesson, T.; Krska, R.; Olsen, M.; Fredlund, E. Deoxynivalenol and other selected Fusarium toxins in Swedish wheat—Occurrence and correlation to specific Fusarium species. Int. J. Food Microbiol. 2013, 167, 284–291. [Google Scholar] [CrossRef]
- Fredlund, E.; Gidlund, A.; Sulyok, M.; Börjesson, T.; Krska, R.; Olsen, M.; Lindblad, M. Deoxynivalenol and other selected Fusarium toxins in Swedish oats—Occurrence and correlation to specific Fusarium species. Int. J. Food Microbiol. 2013, 167, 276–283. [Google Scholar] [CrossRef]
- Debevere, S.; De Baere, S.; Haesaert, G.; Rychlik, M.; Fievez, V.; Croubels, S. Development of an UPLC-MS/MS Method for the Analysis of Mycotoxins in Rumen Fluid with and without Maize Silage Emphasizes the Importance of Using Matrix-Matched Calibration. Toxins 2019, 11, 519. [Google Scholar] [CrossRef] [PubMed]
- Dagnac, T.; Latorre, A.; Lorenzo, B.F.; Llompart, M. Validation and application of a liquid chromatography-tandem mass spectrometry based method for the assessment of the co-occurrence of mycotoxins in maize silages from dairy farms in NW Spain. Food Addit. Contam. Part A 2016, 33, 1850–1863. [Google Scholar] [CrossRef] [PubMed]
- Panasiuk, L.; Jedziniak, P.; Pietruszka, K.; Piatkowska, M.; Bocian, L. Frequency and Levels of Regulated and Emerging Mycotoxins in Silage in Poland. Mycotoxin Res. 2019, 35, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Storm, I.; Rasmussen, R.; Rasmussen, P. Occurrence of Pre- and Post-Harvest Mycotoxins and Other Secondary Metabolites in Danish Maize Silage. Toxins 2014, 6, 2256–2269. [Google Scholar] [CrossRef]
- Wambacq, E.; Vanhoutte, I.; Audenaert, K.; De Gelder, L.; Haesaert, G. Occurrence, prevention and remediation of toxigenic fungi and mycotoxins in silage: A review: Fungi and mycotoxins in silage. J. Sci. Food Agric. 2016, 96, 2284–2302. [Google Scholar] [CrossRef]
- Hu, L.; Koehler, P.; Rychlik, M. Effect of sourdough processing and baking on the content of enniatins and beauvericin in wheat and rye bread. Eur. Food Res. Technol. 2014, 238, 581–587. [Google Scholar] [CrossRef]
- Brodal, G.; Aamot, H.U.; Almvik, M.; Hofgaard, I.S. Removal of Small Kernels Reduces the Content of Fusarium Mycotoxins in Oat Grain. Toxins 2020, 12, 346. [Google Scholar] [CrossRef]
- Tittlemier, S.A.; Blagden, R.; Chan, J.; McMillan, T.L.; Pleskach, K.; Izydorczyk, M.S. Effects of Processing Whole Oats on the Analysis and Fate of Mycotoxins and Ergosterol. World Mycotoxin J. 2020, 13, 45–56. [Google Scholar] [CrossRef]
- Meca, G.; Ritieni, A.; Mañes, J. Influence of the heat treatment on the degradation of the minor Fusarium mycotoxin beauvericin. Food Control. 2012, 28, 13–18. [Google Scholar] [CrossRef]
- Mansfield, M.A.; Jones, A.D.; Kuldau, G.A. Contamination of Fresh and Ensiled Maize by Multiple Penicillium Mycotoxins. Phytopathology 2008, 98, 330–336. [Google Scholar] [CrossRef]
- Teller, R.S.; Schmidt, R.J.; Whitlow, L.W.; Kung, L. Effect of physical damage to ears of corn before harvest and treatment with various additives on the concentration of mycotoxins, silage fermentation, and aerobic stability of corn silage. J. Dairy Sci. 2012, 95, 1428–1436. [Google Scholar] [CrossRef] [PubMed]
- Gregori, R.; Meriggi, P.; Pietri, A.; Formenti, S.; Baccarini, G.; Battilani, P. Dynamics of fungi and related mycotoxins during cereal storage in silo bags. Food Control 2013, 30, 280–287. [Google Scholar] [CrossRef]
- Schaafsma, A.W.; Limay-Rios, V.; Paul, D.E.; Miller, J.D. Mycotoxins in fuel ethanol co-products derived from maize: A mass balance for deoxynivalenol: DON in maize ethanol co-product. J. Sci. Food Agric. 2009, 89, 1574–1580. [Google Scholar] [CrossRef]
- Klosowski, G.; Blajet-Kosicka, A.; Mikulski, D.; Grajewski, J. Assessing the Potential of Reducing Mycotoxin Concentration During the Production Process of Ethanol from Maize Grain Using Pls and Classic Technology. Zywnosc Nauka Technol. Jakosc 2011, 18, 89–105. [Google Scholar] [CrossRef]
- Yoshinari, T.; Suzuki, Y.; Sugita-Konishi, Y.; Ohnishi, T.; Terajima, J. Occurrence of beauvericin and enniatins in wheat flour and corn grits on the Japanese market, and their co-contamination with type B trichothecene mycotoxins. Food Addit. Contam. Part A 2016, 33, 1620–1626. [Google Scholar] [CrossRef]
- Malachova, A.; Dzuman, Z.; Veprikova, Z.; Vaclavikova, M.; Zachariasova, M.; Hajslova, J. Deoxynivalenol, Deoxynivalenol-3-glucoside, and Enniatins: The Major Mycotoxins Found in Cereal-Based Products on the Czech Market. J. Agric. Food Chem. 2011, 59, 12990–12997. [Google Scholar] [CrossRef]
- Spanic, V.; Katanic, Z.; Sulyok, M.; Krska, R.; Puskas, K.; Vida, G.; Drezner, G.; Šarkanj, B. Multiple Fungal Metabolites Including Mycotoxins in Naturally Infected and Fusarium-Inoculated Wheat Samples. Microorganisms 2020, 8, 578. [Google Scholar] [CrossRef]
- Smith, M.-C.; Madec, S.; Coton, E.; Hymery, N. Natural Co-Occurrence of Mycotoxins in Foods and Feeds and Their in vitro Combined Toxicological Effects. Toxins 2016, 8, 94. [Google Scholar] [CrossRef]
- Mallebrera, B.; Prosperini, A.; Font, G.; Ruiz, M.J. In vitro mechanisms of Beauvericin toxicity: A review. Food Chem. Toxicol. 2018, 111, 537–545. [Google Scholar] [CrossRef]
- Prosperini, A.; Berrada, H.; Ruiz, M.J.; Caloni, F.; Coccini, T.; Spicer, L.J.; Perego, M.C.; Lafranconi, A. A Review of the Mycotoxin Enniatin B. Front. Public Health 2017, 5, 304. [Google Scholar] [CrossRef]
- Fernández-Blanco, C.; Font, G.; Ruiz, M.-J. Interaction effects of enniatin B, deoxinivalenol and alternariol in Caco-2 cells. Toxicol. Lett. 2016, 241, 38–48. [Google Scholar] [CrossRef] [PubMed]
- Tran, V.; Viktorova, J.; Augustynkova, K.; Jelenova, N.; Dobiasova, S.; Rehorova, K.; Fenclova, M.; Stranska-Zachariasova, M.; Vitek, L.; Hajslova, J.; et al. In Silico and In Vitro Studies of Mycotoxins and Their Cocktails; Their Toxicity and Its Mitigation by Silibinin Pre-Treatment. Toxins 2020, 12, 148. [Google Scholar] [CrossRef] [PubMed]
- Agahi, F.; Font, G.; Juan, C.; Juan-García, A. Individual and Combined Effect of Zearalenone Derivates and Beauvericin Mycotoxins on SH-SY5Y Cells. Toxins 2020, 12, 212. [Google Scholar] [CrossRef] [PubMed]
- Alassane-Kpembi, I.; Schatzmayr, G.; Taranu, I.; Marin, D.; Puel, O.; Oswald, I.P. Mycotoxins co-contamination: Methodological aspects and biological relevance of combined toxicity studies. Crit. Rev. Food Sci. Nutr. 2017, 57, 3489–3507. [Google Scholar] [CrossRef]
- Khoshal, A.K.; Novak, B.; Martin, P.G.P.; Jenkins, T.; Neves, M.; Schatzmayr, G.; Oswald, I.P.; Pinton, P. Co-Occurrence of DON and Emerging Mycotoxins in Worldwide Finished Pig Feed and Their Combined Toxicity in Intestinal Cells. Toxins 2019, 11, 727. [Google Scholar] [CrossRef] [PubMed]
- Kamyar, M.R.; Kouri, K.; Rawnduzi, P.; Studenik, C.; Lemmens-Gruber, R. Effects of moniliformin in presence of cyclohexadepsipeptides on isolated mammalian tissue and cells. Toxicol. In Vitro 2006, 20, 1284–1291. [Google Scholar] [CrossRef]
- EFSA Panel on Contaminants in the Food Chain (CONTAM). Scientific Opinion on the risks to human and animal health related to the presence of beauvericin and enniatins in food and feed. EFSA J. 2014, 12, 3802. [Google Scholar] [CrossRef]
- Devreese, M.; Broekaert, N.; De Mil, T.; Fraeyman, S.; De Backer, P.; Croubels, S. Pilot toxicokinetic study and absolute oral bioavailability of the Fusarium mycotoxin enniatin B1 in pigs. Food Chem. Toxicol. 2014, 63, 161–165. [Google Scholar] [CrossRef]
- Devreese, M.; De Baere, S.; De Backer, P.; Croubels, S. Quantitative determination of the Fusarium mycotoxins beauvericin, enniatin A, A1, B and B1 in pig plasma using high performance liquid chromatography-tandem mass spectrometry. Talanta 2013, 106, 212–219. [Google Scholar] [CrossRef]
- Fraeyman, S.; Devreese, M.; Antonissen, G.; De Baere, S.; Rychlik, M.; Croubels, S. Comparative Oral Bioavailability, Toxicokinetics, and Biotransformation of Enniatin B1 and Enniatin B in Broiler Chickens. J. Agric. Food Chem. 2016, 64, 7259–7264. [Google Scholar] [CrossRef]
- Devreese, M.; Girgis, G.N.; Tran, S.-T.; De Baere, S.; De Backer, P.; Croubels, S.; Smith, T.K. The effects of feed-borne Fusarium mycotoxins and glucomannan in turkey poults based on specific and non-specific parameters. Food Chem. Toxicol. 2014, 63, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Ivanova, L.; Uhlig, S.; Devreese, M.; Croubels, S.; Fæste, C.K. Biotransformation of the mycotoxin enniatin B1 in pigs: A comparative in vitro and in vivo approach. Food Chem. Toxicol. 2017, 105, 506–517. [Google Scholar] [CrossRef] [PubMed]
- Ivanova, L.; Fæste, C.K.; Van Pamel, E.; Daeseleire, E.; Callebaut, A.; Uhlig, S. Presence of enniatin B and its hepatic metabolites in plasma and liver samples from broilers and eggs from laying hens. World Mycotoxin J. 2014, 7, 167–175. [Google Scholar] [CrossRef]
- Rodríguez-Carrasco, Y.; Heilos, D.; Richter, L.; Süssmuth, R.D.; Heffeter, P.; Sulyok, M.; Kenner, L.; Berger, W.; Dornetshuber-Fleiss, R. Mouse tissue distribution and persistence of the food-born fusariotoxins Enniatin B and Beauvericin. Toxicol. Lett. 2016, 247, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Manyes, L.; Escrivá, L.; Serrano, A.B.; Rodríguez-Carrasco, Y.; Tolosa, J.; Meca, G.; Font, G. A preliminary study in Wistar rats with enniatin A contaminated feed. Toxicol. Mech. Methods 2014, 24, 179–190. [Google Scholar] [CrossRef] [PubMed]
- Debevere, S.; Cools, A.; De Baere, S.; Haesaert, G.; Rychlik, M.; Croubels, S.; Fievez, V. In Vitro Rumen Simulations Show a Reduced Disappearance of Deoxynivalenol, Nivalenol and Enniatin B at Conditions of Rumen Acidosis and Lower Microbial Activity. Toxins 2020, 12, 101. [Google Scholar] [CrossRef]
- Roig, M.; Meca, G.; Ferrer, E.; Mañes, J. Reduction of the Enniatins A, A1, B, B1 by an in Vitro Degradation Employing Different Strains of Probiotic Bacteria: Identification of Degradation Products by LC–MS–LIT. Toxicon 2013, 70, 44–53. [Google Scholar] [CrossRef]
- Ferrer E, R.M. Degradation of the Bioactive Compounds Enniatins A, A1, B, B1 Employing Different Strains of Bacillus Subtilis. J. Food Process. Technol. 2014, 5. [Google Scholar] [CrossRef]
- Meca, G.; Zhou, T.; Li, X.-Z.; Mañes, J. Beauvericin Degradation during Bread and Beer Making. Food Control 2013, 34, 1–8. [Google Scholar] [CrossRef]
- Meca, G.; Ritieni, A.; Mañes, J. Reduction in Vitro of the Minor Fusarium Mycotoxin Beauvericin Employing Different Strains of Probiotic Bacteria. Food Control 2012, 28, 435–440. [Google Scholar] [CrossRef]
- May, H.D.; Wu, Q.; Blake, C.K. Effects of the Fusarium spp. mycotoxins fusaric acid and deoxynivalenol on the growth of Ruminococcus albus and Methanobrevibacter ruminantium. Can. J. Microbiol. 2000, 46, 692–699. [Google Scholar] [CrossRef] [PubMed]
- Bacon, C.W.; Hinton, D.M.; Hinton, A. Growth-inhibiting effects of concentrations of fusaric acid on the growth of Bacillus mojavensis and other biocontrol Bacillus species. J. Appl. Microbiol. 2006, 100, 185–194. [Google Scholar] [CrossRef]
- Jestoi, M. Emerging Fusarium-Mycotoxins Fusaproliferin, Beauvericin, Enniatins, And Moniliformin—A Review. Crit. Rev. Food Sci. Nutr. 2008, 48, 21–49. [Google Scholar] [CrossRef] [PubMed]
- Sebastià, N.; Meca, G.; Soriano, J.M.; Mañes, J. Antibacterial effects of enniatins J1 and J3 on pathogenic and lactic acid bacteria. Food Chem. Toxicol. 2011, 49, 2710–2717. [Google Scholar] [CrossRef] [PubMed]
- Arab, M.; Sohrabvandi, S.; Mortazavian, A.M.; Mohammadi, R.; Tavirani, M.R. Reduction of aflatoxin in fermented milks during production and storage. Toxin Rev. 2012, 31, 44–53. [Google Scholar] [CrossRef]
- Sangsila, A.; Faucet-Marquis, V.; Pfohl-Leszkowicz, A.; Itsaranuwat, P. Detoxification of zearalenone by Lactobacillus pentosus strains. Food Control 2016, 62, 187–192. [Google Scholar] [CrossRef]
- Fuchs, S.; Sontag, G.; Stidl, R.; Ehrlich, V.; Kundi, M.; Knasmüller, S. Detoxification of patulin and ochratoxin A, two abundant mycotoxins, by lactic acid bacteria. Food Chem. Toxicol. 2008, 46, 1398–1407. [Google Scholar] [CrossRef]
- Ferrer, M.; Manyes, L.; Mañes, J.; Meca, G. Influence of prebiotics, probiotics and protein ingredients on mycotoxin bioaccessibility. Food Funct. 2015, 6, 987–994. [Google Scholar] [CrossRef] [PubMed]
- Koivisto, P.; Jonsson, M.; Jestoi, M.; Peltonen, K. Enniatin B mycotoxin is excreted as such to rat urine. Toxicol. Lett. 2015, 238, S60–S61. [Google Scholar] [CrossRef]
- Rodríguez-Carrasco, Y.; Narváez, A.; Izzo, L.; Gaspari, A.; Graziani, G.; Ritieni, A. Biomonitoring of Enniatin B1 and Its Phase I Metabolites in Human Urine: First Large-Scale Study. Toxins 2020, 12, 415. [Google Scholar] [CrossRef]
- Jestoi, M.; Rokka, M.; Järvenpää, E.; Peltonen, K. Determination of Fusarium mycotoxins beauvericin and enniatins (A, A1, B, B1) in eggs of laying hens using liquid chromatography-tandem mass spectrometry (LC–MS/MS). Food Chem. 2009, 115, 1120–1127. [Google Scholar] [CrossRef]
- Rossi, F.; Gallo, A.; Bertuzzi, T. Emerging mycotoxins in the food chain. Mediterr. J. Nutr. Metab. 2020, 13, 7–27. [Google Scholar] [CrossRef]
- Callebaut, F.; Tangni, E.K.; Debongnie, P.; Stals, E.; Huybrechts, B.; Waegeneers, N.; Delezie, E.; Van Pamel, E.; Daeseleire, E. Carry-Over of Mycotoxins to Animal Products: Case Study Poultry; Scientific Report 211/212 CODA-CERVA; Centrum voor Onderzoek in Diergeneeskunde en Agrochemie-Centre d’Étude et de Recherches Vétérinaires et Agrochemiques: Brussels, Belgium, 2011–2012; pp. 141–144. [Google Scholar]
- Tolosa, J.; Font, G.; Mañes, J.; Ferrer, E. Natural Occurrence of Emerging Fusarium Mycotoxins in Feed and Fish from Aquaculture. J. Agric. Food Chem. 2014, 62, 12462–12470. [Google Scholar] [CrossRef]
- Tolosa, J.; Font, G.; Mañes, J.; Ferrer, E. Mitigation of Enniatins in Edible Fish Tissues by Thermal Processes and Identification of Degradation Products. Food Chem. Toxicol. 2017, 101, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Nácher-Mestre, J.; Beltrán, E.; Strachan, F.; Dick, J.R.; Pérez-Sánchez, J.; Berntssen, M.H.G.; Tocher, D.R. No Transfer of the Non-Regulated Mycotoxins, Beauvericin and Enniatins, from Feeds to Farmed Fish Reared on Plant-Based Diets. Food Chem. 2020, 323, 126773. [Google Scholar] [CrossRef]
- Piątkowska, M.; Sulyok, M.; Pietruszka, K.; Panasiuk, Ł. Pilot study for the presence of fungal metabolites in sheep milk from first spring milking. J. Vet. Res. 2018, 62, 167–172. [Google Scholar] [CrossRef]
- Rubert, J.; León, N.; Sáez, C.; Martins, C.P.B.; Godula, M.; Yusà, V.; Mañes, J.; Soriano, J.M.; Soler, C. Evaluation of mycotoxins and their metabolites in human breast milk using liquid chromatography coupled to high resolution mass spectrometry. Anal. Chim. Acta 2014, 820, 39–46. [Google Scholar] [CrossRef]
- Braun, D.; Ezekiel, C.N.; Abia, W.A.; Wisgrill, L.; Degen, G.H.; Turner, P.C.; Marko, D.; Warth, B. Monitoring Early Life Mycotoxin Exposures via LC-MS/MS Breast Milk Analysis. Anal. Chem. 2018, 90, 14569–14577. [Google Scholar] [CrossRef]
- Braun, D.; Ezekiel, C.N.; Marko, D.; Warth, B. Exposure to Mycotoxin-Mixtures via Breast Milk: An Ultra-Sensitive LC-MS/MS Biomonitoring Approach. Front. Chem. 2020, 8, 423. [Google Scholar] [CrossRef]
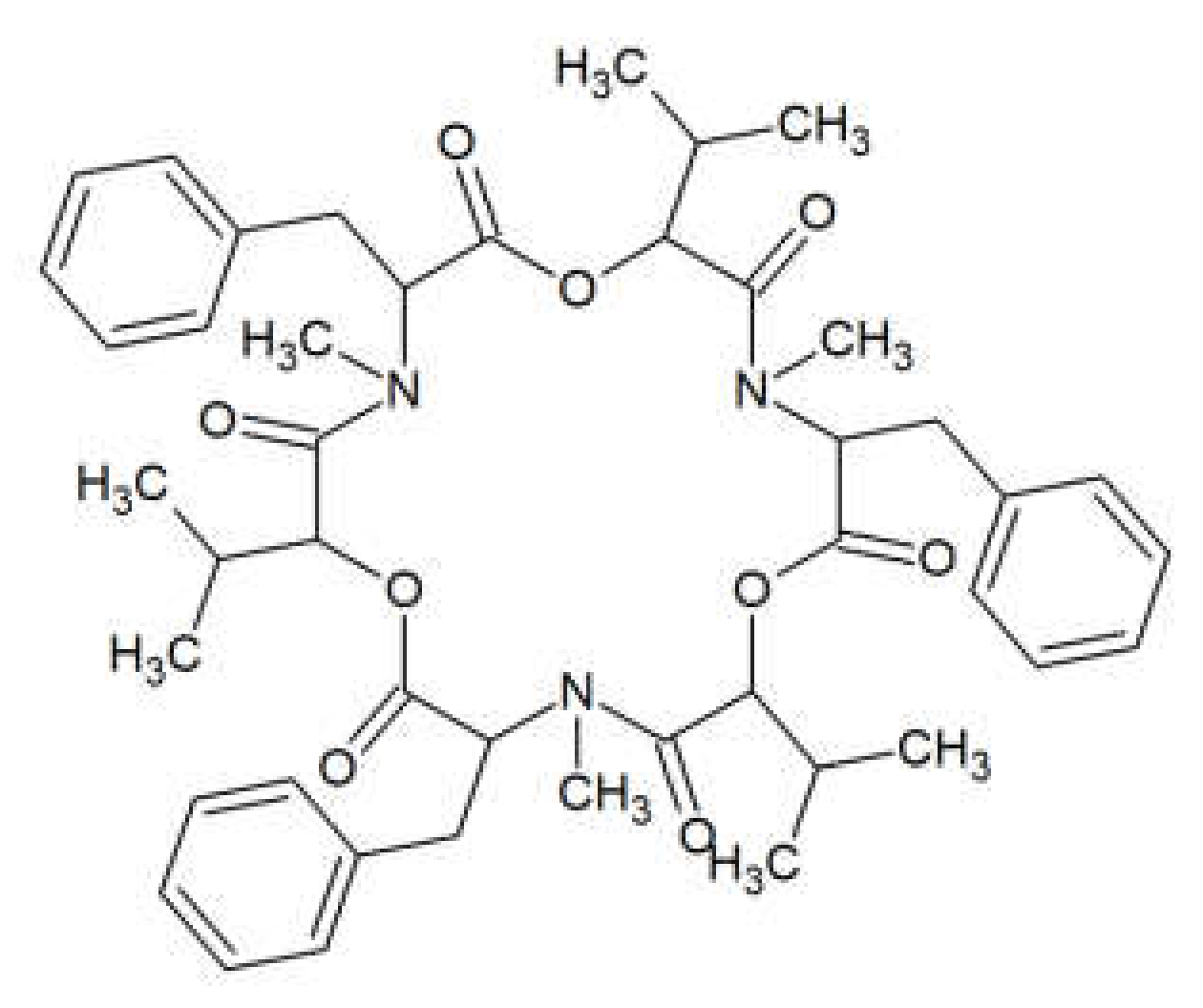
| Beauvericin | Bacillus cereus, B. mycoides, B. pumilis, B. sphaericus, Bifidobacterium adolescentis, Clostridium perfringens, Escherichia coli, Enterococcus faecium, Eubacterium biforme, Listeria monocytogenes, Paenibacillus alvei, P. azotofixans, P. macerans, P. macquariensis, P. pabuli, P. productus, P. pulvifaciens, P. validus, Peptostreptococcus anaerobius, Pseudomonas aeruginosa, Salmonella enterica, Shigella dysenteriae, Yersinia enterocolitica, | [24,26] |
| Enniatin B | Escherichia coli, E. faecium, Clostridium perfringens, Listeria monocytogenes, Pseudomonas aeruginosa, Salmonella enterica, Shygella dysenteriae, Staphylococcus aureus, Yersinia enterocolitica | [36] |
| Enniatin B1 | Bifidobacterium adolescentis | [37] |
| Mycotoxin | Bacteria | Products of Bacterial Metabolism | Source |
|---|---|---|---|
| EN A | intestinal bacteria | (EN A + K-Ile) + (EN A + K-HyLv) + | [106] |
| EN A1 | 9 bacterial and 22 S. cerevisiae strains | (EN A1 + K-Ile) + (EN A1 + Na) + (EN A1 + K-Ile) + (EN A1 + K-HyLv) + | [108] |
| EN B | 6 Bacillus subtilis strains | (EN B + K-Val-COOH) + (EN B + K-Val-H2O) + (EN B + K-HyLv) + (EN B-2HyLv-H2O) + | [109] |
| EN B | 9 bacterial and 22 S. cerevisiae strains | (EN B + K-HyLv-Val) + (EN B-HyLv-2H2O-CH3) + (EN B-HyLv) + (EN B + Na) + (EN B + K) + | [108] |
| EN B1 | 6 Bacillus subtilis strains | (EN B1 + K-Ile-Val-H2O) + (EN B1-2Val) + (EN B1 + K-HyLv) + (EN B1 + K-Ile) + (EN B1 + Na) + | [109] |
| EN B1 | 9 Bifidobacterium and Lactobacillus strains and 22 S. cerevisiae strains | (EN B1 + K-Val) + (EN B1 + Na) + (EN B1 + K-Val-H2O-2CH3) + (EN B1+K-Val-HyLv+H2O) + | [108] |
| BEA | S. cerevisiae A34 | (BEA + Na) + (BEA + K) + (BEA + H) + (BEA–N-Phe-Na) + (BEA–HyLv) + (BEA–H2O) + (BEA–HyLv–2H2O) + | [110] |
| BEA | 13 Bifidobacterium, Lactobacillus, Eubacterium, and Salmonella strains | ((BEA + Na +) + Phosphatidylcholine)) + ((BEA + Na +) + Citocoline)) + (BEA + Na) + | [111] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Křížová, L.; Dadáková, K.; Dvořáčková, M.; Kašparovský, T. Feedborne Mycotoxins Beauvericin and Enniatins and Livestock Animals. Toxins 2021, 13, 32. https://doi.org/10.3390/toxins13010032
Křížová L, Dadáková K, Dvořáčková M, Kašparovský T. Feedborne Mycotoxins Beauvericin and Enniatins and Livestock Animals. Toxins. 2021; 13(1):32. https://doi.org/10.3390/toxins13010032
Chicago/Turabian StyleKřížová, Ludmila, Kateřina Dadáková, Michaela Dvořáčková, and Tomáš Kašparovský. 2021. "Feedborne Mycotoxins Beauvericin and Enniatins and Livestock Animals" Toxins 13, no. 1: 32. https://doi.org/10.3390/toxins13010032
APA StyleKřížová, L., Dadáková, K., Dvořáčková, M., & Kašparovský, T. (2021). Feedborne Mycotoxins Beauvericin and Enniatins and Livestock Animals. Toxins, 13(1), 32. https://doi.org/10.3390/toxins13010032






