2-Phenylethyl Isothiocyanate Exerts Antifungal Activity against Alternaria alternata by Affecting Membrane Integrity and Mycotoxin Production
Abstract
1. Introduction
2. Results
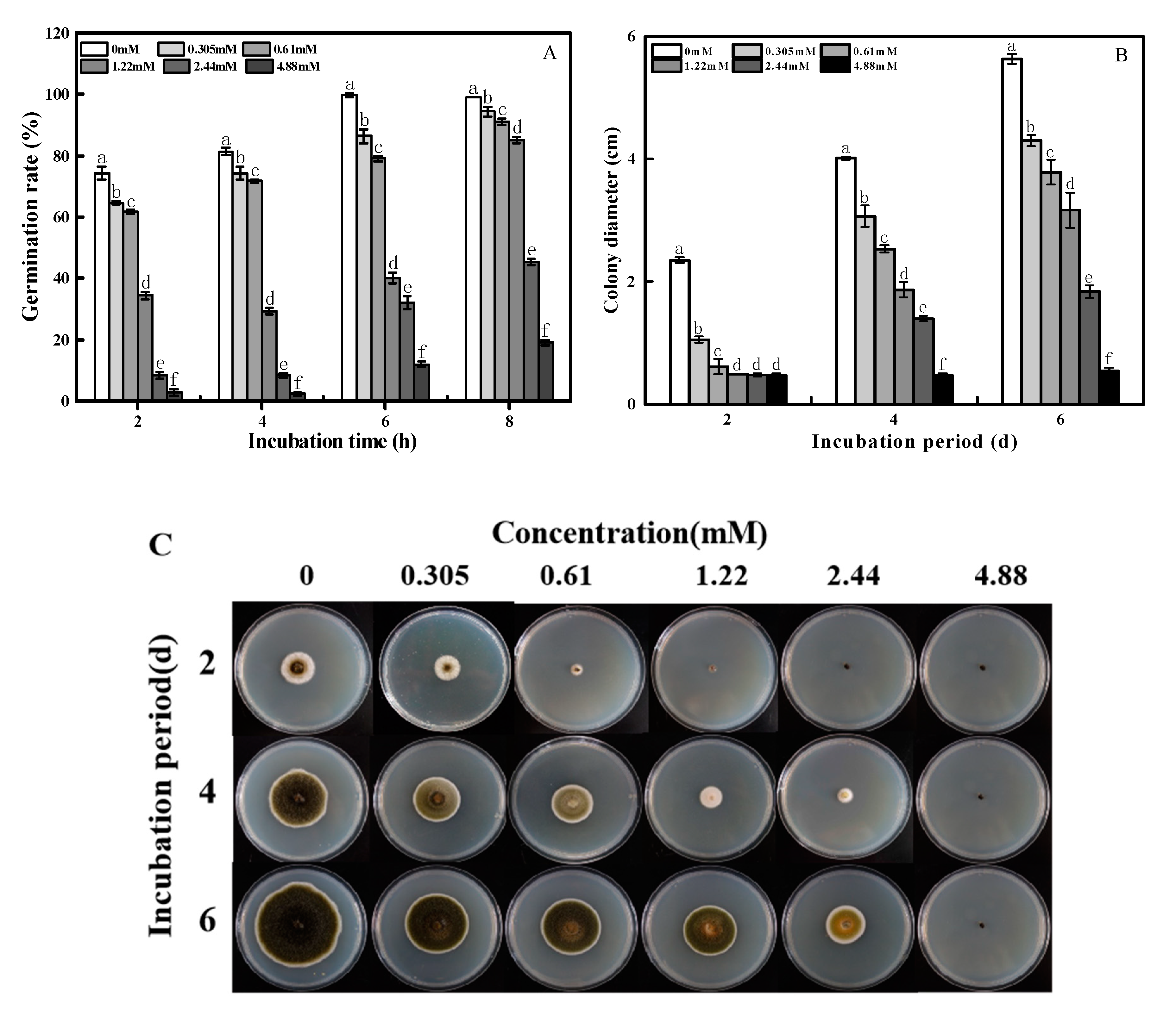
2.1. Effects of 2-PEITC on Spore Germination and Mycelial Growth of A. alternata
2.2. Inhibition of on the Lesion Development of Black Spot in Pear Fruit
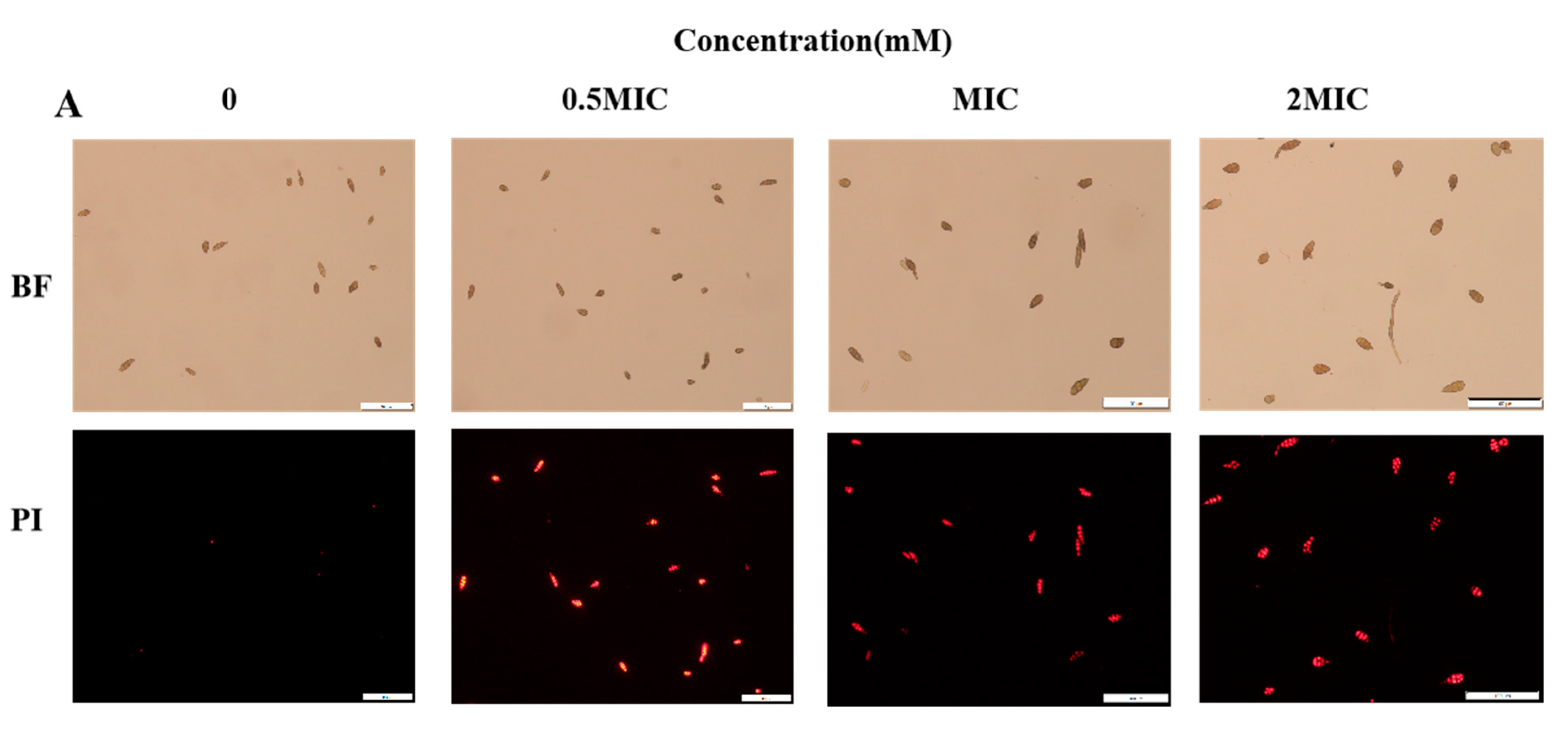
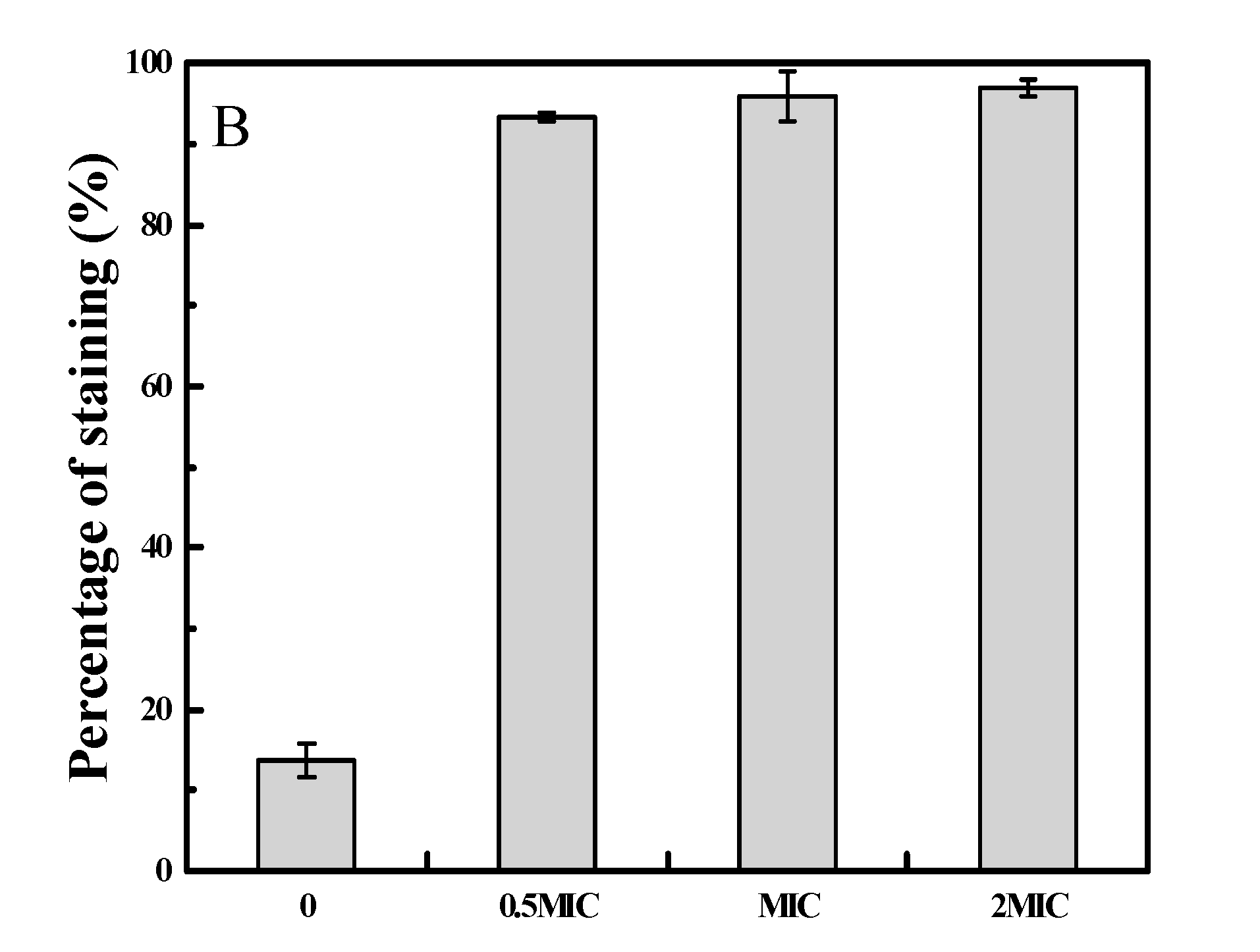
2.3. 2-PEITC Damaged Plasma Membrane Integrity of A. alternata
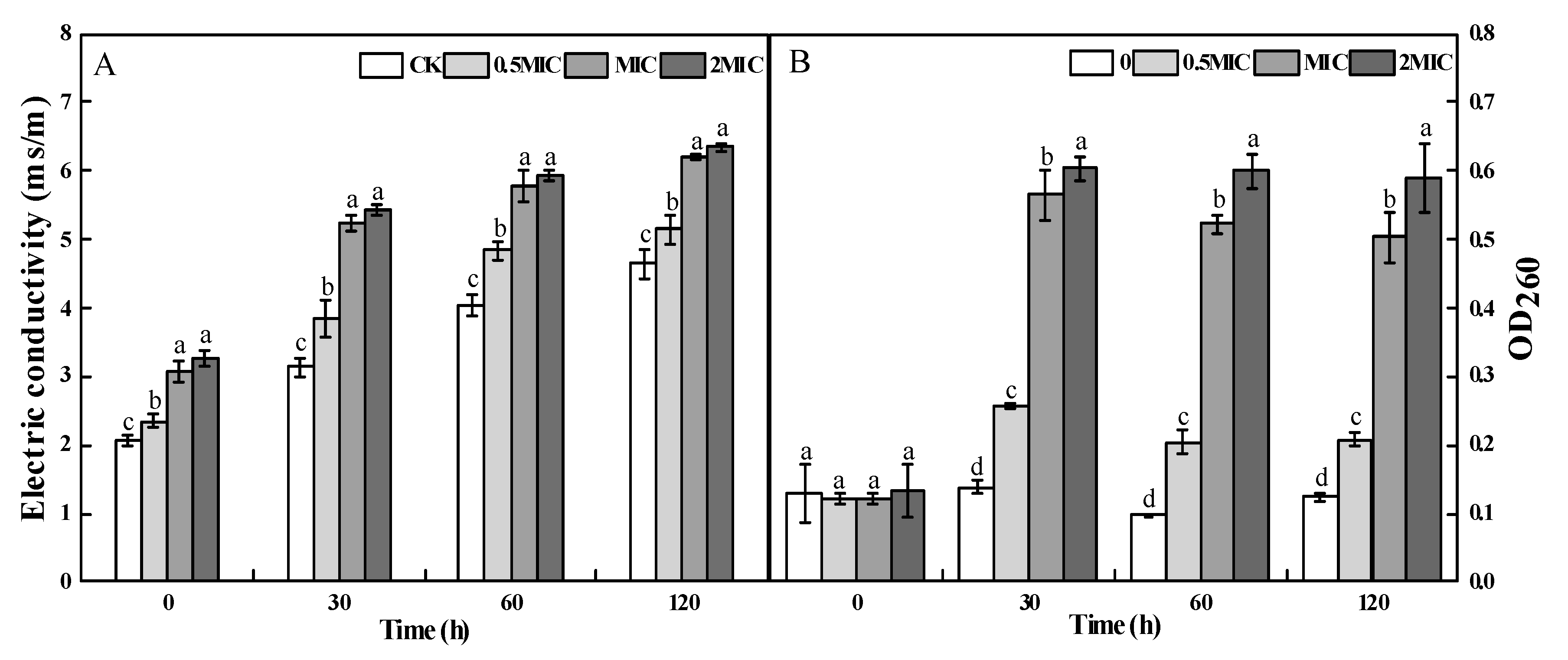
2.4. Cellular Leakage of A. alternata after 2-PEITC Treatment
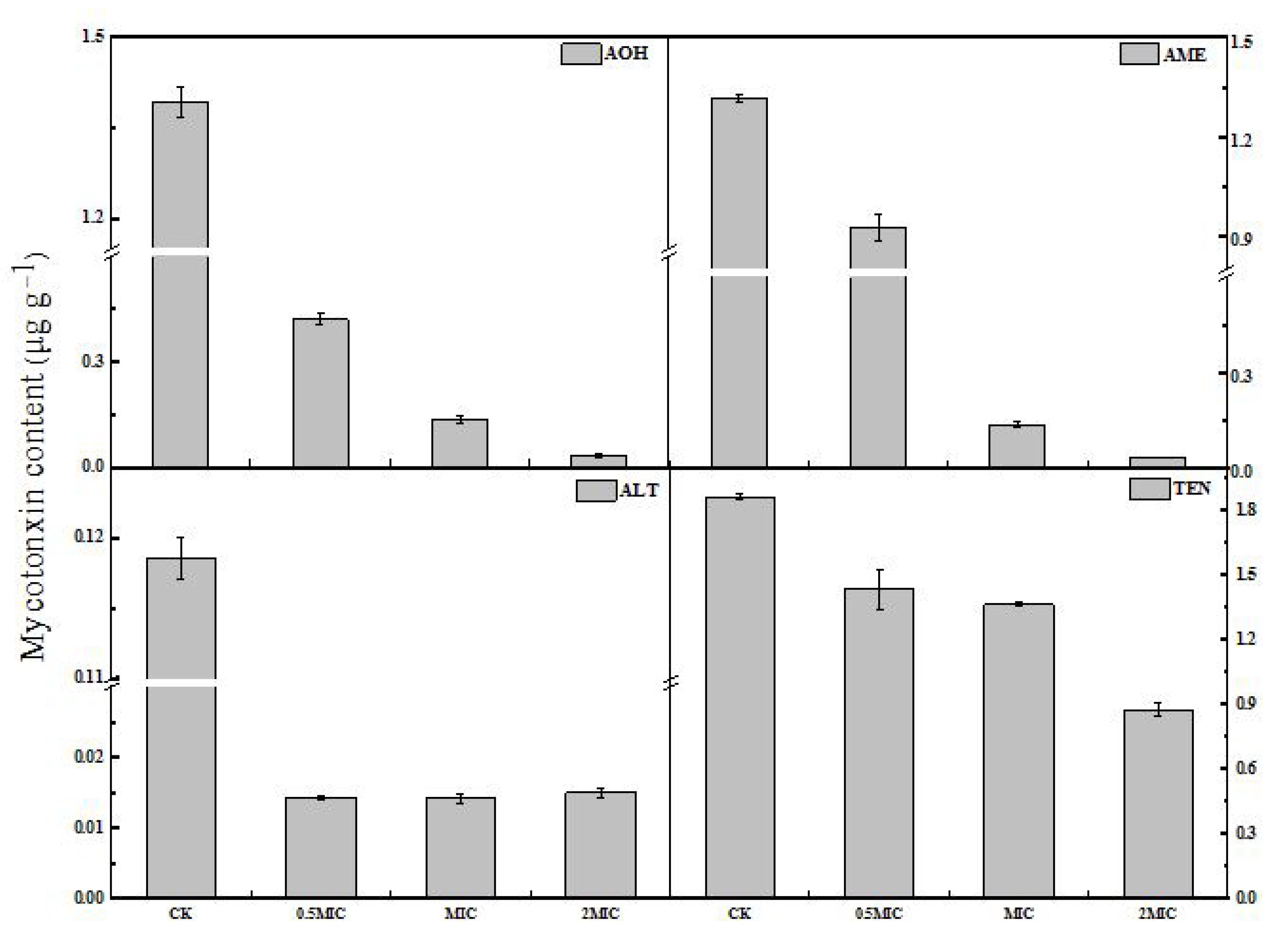
2.5. Production of Mycotoxins by A. alternata upon 2-PEITC Treatment
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Fruit and Pathogens
5.2. Mycotoxin Standards and Chemicals
5.3. In Vitro Inhibitory Effects of 2-PEITC on Growth of A. alternata
5.4. Effect of 2-PEITC on A. alternata in Pear Fruit
5.5. Determination of Plasma Membrane Integrity
5.6. Detection of Cellular Electrolyte Leakage and Nucleic Acid Content
5.7. Mycotoxin Extraction and HPLC-MS Analysis
5.8. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Vilaplana, R.; Páez, D.; Valencia-Chamorro, S. Control of black rot caused by Alternaria alternata in yellow pitahaya (Selenicereus megalanthus) through hot water dips. LWT Food Sci. Technol. 2017, 82, 162–169. [Google Scholar] [CrossRef]
- Yang, J.; Sun, C.; Zhang, Y.; Fu, D.; Zheng, X.; Yu, T. Induced resistance in tomato fruit by γ-aminobutyric acid for the control of Alternaria rot caused by Alternaria Alternata. Food Chem. 2017, 221, 1014–1020. [Google Scholar] [CrossRef] [PubMed]
- Estiarte, N.; Crespo-Sempere, A.; Marín, S.; Sanchis, V.; Ramos, A.J. Exploring polyamine metabolismo of Alternaria alternata to target new substances to control the fungal infection. Food Microbiol. 2017, 65, 193–204. [Google Scholar] [CrossRef] [PubMed]
- Gabriel, M.F.; Uriel, N.; Teifoori, F.; Postigo, I.; Suñén, E.; Martínez, J. The major Alternaria alternata allergen, Alta1: A reliable and specific marker of fungal contamination in citrus fruit. Int. J. Food Microbiol. 2017, 257, 26–30. [Google Scholar] [CrossRef] [PubMed]
- Castro, J.C.; Endo, E.H.; Souza, M.R.; Zanqueta, E.B.; Polonio, J.C.; Pamphile, J.A.; UedaNakamura, T.; Nakamura, C.V.; Dias Filho, B.P.; Abreu Filho, B.A. Bioactivity of essential oils in the control of Alternaria alternata in dragon fruit (Hylocereusundatus Haw.). Ind. Crop. Prod. 2017, 97, 101–109. [Google Scholar] [CrossRef]
- Troncoso-Rojas, R.; Corral-Acosta, Y.; Alberto, S.; Raymundo, G.; Aguilar-Valenzuela, A.; Ojeda-Contreras, J. Postharvest treatment of isothiocyanates to control alternaria rot in netted melon. Phytoparasitica 2009, 37, 445–451. [Google Scholar] [CrossRef]
- Li, Y.C.; Bi, Y.; An, L.Z. Occurrence and latent infection of Alternaria rot of Pingguoli pear (Pyrusbretschneideri Rehd. cv. Pingguoli) fruit in Gansu, China. J. Phytopathol. 2007, 155, 56–60. [Google Scholar] [CrossRef]
- Estiarte, N.; Crespo-Sempere, A.; Marín, S.; Sanchis, V.; Ramos, A.J. Effect of 1-methylcypropopene on the development of black mold disease and its potential effect on alternariol and alternariolmonomethyl ether biosynthesis on tomatoes infected with Alternaria alternata. Int. J. Food Microbiol. 2016, 236, 74–82. [Google Scholar] [CrossRef]
- Andersen, B.; Nielsen, K.F.; Pinto, V.F.; Patriarca, A. Characterization of Alternaria strains from Argentinean blueberry, tomato, walnut and wheat. Int. J. Food Microbiol. 2015, 196, 1–10. [Google Scholar] [CrossRef]
- Braghini, R.; Sucupira, M.; Rocha, L.O.; Reis, T.A.; Aquino, S.; Corrêa, B. Effects of gamma radiation on the growth of Alternaria alternata and on the production of alternariol and alternariolmonomethyl ether in sunflower seeds. Food Microbiol. 2009, 26, 927–931. [Google Scholar] [CrossRef]
- Piercey, M.J.; Mazzanti, G.; Budge, S.M.; Delaquis, P.J.; Paulson, A.T.; Hansen, L.T. Antimicrobial activity of cyclodextrin entrapped allyl isothiocyanate in a model system and packaged fresh-cut onions. Food Micobiol. 2012, 30, 213–218. [Google Scholar] [CrossRef] [PubMed]
- Elad, Y.; Pertot, I.; Cores-Prado, A.M.; Stewart, A. Plant hosts of Botrytis spp. In Botrytis—The Fungus, the Pathogen and Its Management in Agricultural Systems; Fillinger, S., Elad, Y., Eds.; Springer: Berlin, Germany, 2016; pp. 413–486. [Google Scholar]
- Soylu, E.M.; Kose, F. Antifungal activities of essential oils against citrus black rot disease agent Alternaria alternata. J. Essent. Oil Bear. Plants 2015, 18, 894–903. [Google Scholar] [CrossRef]
- Tian, S.P.; Torres, R.; Ballester, A.R.; Li, B.Q.; Vilanova, L.; González-Candelas, L. Molecular aspects in pathogen-fruit interactions: Virulence and resistance. Postharvest Biol. Technol. 2016, 122, 11–21. [Google Scholar] [CrossRef]
- Kurt, S.; Güneş, U.; Soylu, E.M. In vitro and in vivo antifungal activity of synthetic pure isothiocyanates against Sclerotinia sclerotiorum. Pest Manag. Sci. 2011, 67, 869–875. [Google Scholar] [CrossRef]
- Motisi, N.; Montfort, F.; Romillac, N.; Lucas, P. Duration of control of two soilborne pathogens following incorporation of above and below-ground residues of brassica juncea into soil. Plant Pathol. 2010, 58, 470–478. [Google Scholar] [CrossRef]
- Fahey, J.W.; Zalcmann, A.T.; Talalay, P. The chemical diversity and distribution of glucosinolates and isothiocyanates among plants. Phytochemistry 2001, 56, 5–51. [Google Scholar] [CrossRef]
- Gaurav, K.; Hardeep, S.T.; Sonam, M.; Jitendra, K.S.; Anil, T.; Sardul, S.S. Isothiocyanates: A class of bioactive metabolites with chemopreventive potential. Tumor Biol. 2015, 36, 4005–4016. [Google Scholar]
- Besma, A.; Lucas, C.R.; Riedl, K.M.; Clinton, S.K.; Amir, M. Cruciferous vegetables, isothiocyanates and bladder cancer prevention. Mol. Nutr. Food Res. 2018, 1, 272–282. [Google Scholar]
- Melina, M.; Tom, A.; Rodrigo, F.; Vasilis, Z.; Aglaia, P.; Panayiotidis, M.I. From chemo-prevention to epigenetic regulation: The role of isothiocyanates in skin cancer prevention. Pharmacol. Ther. 2018, 190, 187–201. [Google Scholar]
- Kris-Etherton, P.M.; Hecker, K.D.; Bonanome, A.; Coval, S.M.; Binkoski, A.E.; Hilpert, K.F.; Griel, A.E.; Etherton, T.D. Bioactive compounds in foods: Their role in the prevention of cardiovascular disease and cancer. Am. J. Med. 2002, 113, 71–88. [Google Scholar] [CrossRef]
- Dhingra, O.D.; Costa, M.L.N.; Silva, G.J. Potential of allylisothiocyanate to control Rhizoctoniasolani seedling damping off and seedling blight in transplant production. J. Phytopathol. 2004, 152, 352–357. [Google Scholar] [CrossRef]
- Zasada, I.A.; Ferris, H. Sensitivity of Meloidogynejavanica and Tylenchulussemipenetrans to isothiocyanates in laboratory assays. Phytopathology 2003, 93, 747–750. [Google Scholar] [CrossRef]
- Buskov, S.; Serra, B.; Rosa, E.; Sorensen, H.; Sorensen, J.C. Effects of intact glucosinolates and products produced from glucosinolates in myrosinase-catalyzed hydrolysis on the potato cyst nematode (GloboderarostochiensiscvWoll). J. Agric. Food Chem. 2002, 50, 690–695. [Google Scholar] [CrossRef]
- Haristoy, X.; Fahey, J.W.; Scholtus, I.; Lozniewski, A. Evaluation of the antimicrobial effects of several isothiocyanates on Helicobacter pylori. Planta Med. 2005, 71, 326–330. [Google Scholar] [CrossRef]
- Nadarajah, D.; Han, J.H.; Holley, R.A. Use of mustard flour to inactivate Escherichia coli O157:H7 in ground beef under nitrogen flushed packaging. Int. J. Food Mocrobiol. 2005, 99, 257–267. [Google Scholar] [CrossRef]
- Agrawal, A.A.; Kurashige, N.S. A role for isothiocyanates in plant resistance against the specialist Herbivore Pierisrapae. J. Chem. Ecol. 2003, 29, 1403–1415. [Google Scholar] [CrossRef]
- Peterse, J.; Belz, R.; Waker, F.; Hurle, K. Weed suppression by release of isothiocyanates from turnip-rape mulch. Agron. J. 2001, 93, 37–43. [Google Scholar] [CrossRef]
- Tiznado-Hernández, M.; Troncoso-Rojas, R. Control of fungal diseases with isothiocyanates. Stewart Postharvest Rev. 2006, 2, 1–14. [Google Scholar]
- Mari, M.; Leoni, O.; Bernardi, R.; Neri, F.; Palmieri, S. Control of brown rot on stonefruit by synthetic and glucosinolate-derived isothiocyanates. Postharvest Biol. Technol. 2008, 47, 61–67. [Google Scholar] [CrossRef]
- Ugolini, L.; Martini, C.; Lazzeri, L.; Lorenzo, D.; Mari, M. Control of postharvest grey mould (Botrytis cinerea per.: Fr.) on strawberries by glucosinolate-derived allyl-isothiocyanate treatments. Postharvest Biol. Technol. 2014, 90, 34–39. [Google Scholar] [CrossRef]
- Quiles, J.M.; Manyes, L.; Luciano, F.; Manes, J.; Meca, G. Influence of the antimicrobial compound allylisothiocyanate against the Aspergillus parasiticus growth and its aflatoxins production in pizza crust. Food Chem. Toxicol. 2015, 83, 222–228. [Google Scholar] [CrossRef]
- Clemente, I.; Aznar, M.; Nerín, C. Effect of an active label based on benzyl isothiocyanate on the morphology and ochratoxins production of, Aspergillus ochraceus. Food Res. Int. 2017, 101, 61–72. [Google Scholar] [CrossRef]
- Hu, P.; Wang, A.S.; Engledow, A.S.; Hollister, E.B.; Rothlisberger, K.L.; Matocha, J.E. Inhibition of the germination and growth of Phymatotrichopsis omnivora (cotton root rot) by oilseed meals and isothiocyanates. Appl. Soil Ecol. 2011, 49, 68–75. [Google Scholar] [CrossRef]
- Troncoso-Rojas, R.; Sanchez-Estrada, A.; Ruelas, C.; Garcia, H.S.; Tiznado-Hernandez, M.E. Effect of benzyl isothiocyanate on tomato fruit infection development by Alternaria Alternata. J. Sci. Food Agric. 2005, 85, 1427–1434. [Google Scholar] [CrossRef]
- Mari, M.; Leoni, O.; Iori, R.; Cembali, T. Antifungal vapour-phase activity of allyl-isothiocyanate against Penicillium expansum on pears. Plant Pathol. 2002, 51, 231–236. [Google Scholar] [CrossRef]
- Smith, B.J.; Kirkegaard, J.A. In-vitro inhibition of soil microorganisms by 2-phenylethyl isothiocyanate. Plant Pathol. 2002, 51, 585–593. [Google Scholar] [CrossRef]
- Aires, A.; Mota, V.R.; Saavedra, M.J.; Rosa, E.A.S.; Bennett, R.N. The antimicrobial effects of glucosinolates and their respective enzymatic hydrolysis products on bacteria isolated from the human intestinal tract. J. Appl. Microbiol. 2009, 106, 2086–2095. [Google Scholar] [CrossRef]
- Tao, N.G.; Ouyang, Q.; Jia, L. Citral inhibits mycelial growth of penicillium italicum by a membrane damage mechanism. Food Control 2014, 382, 116–121. [Google Scholar] [CrossRef]
- Linde, J.H.; Combrinck, S.; Regnier, T.J.C.; Virijevic, S. Chemical composition and antifungal activity of the essential oils of Lippiarehmannii from South Africa. S. Afr. J. Bot. 2010, 76, 37–42. [Google Scholar] [CrossRef]
- Zhang, J.; Tian, H.; Sun, H. Antifungal activity of trans-2-hexenal against Penicillium cyclopium by a membrane damage mechanism. J. Food Biochem. 2017, 41, e12289. [Google Scholar] [CrossRef]
- Yun, D.G.; Lee, D.G. Silymarin exerts antifungal effects via membrane-targeted mode of action by increasing permeability and inducing oxidative stress. BBA Biomembr. 2017, 1859, 467–474. [Google Scholar] [CrossRef] [PubMed]
- Mari, M.; Iori, R.; Leoni, O.; Marchi, A. In vitro activity of glucosinolate-derived isothiocyanates against post-harvest fruit pathogens. Ann. Appl. Biol. 1993, 123, 155–164. [Google Scholar] [CrossRef]
- Harvey, S.G.; Hannahan, H.N.; Sams, C.E. Indian mustard and allylisothiocyanate inhibit Sclerotium rolfsii. J. Am. Soc. Hortic. Sci. 2002, 127, 27–31. [Google Scholar] [CrossRef]
- Huang, Y.; Sekhon, N.S.; Borninski, J.; Chen, N.; Rubinsky, B. Instantaneous, quantitative single-cell viability assessment by electrical evaluation of cell membrane integrity with microfabricated devices. Sens. Actuators A Phys. 2003, 105, 31–39. [Google Scholar] [CrossRef]
- Zhou, H.; Tao, N.G.; Jia, L. Antifungal activity of citral, octanal and α-terpineol against Geotrichumcitri-aurantii. Food Control 2014, 37, 277–283. [Google Scholar] [CrossRef]
- Ji, D.; Chen, T.; Ma, D.; Liu, J.; Xu, Y.; Tian, S. Inhibitory effects of methyl thujate on mycelial growth of, Botrytis cinerea, and possible mechanisms. Postharvest Biol. Technol. 2018, 142, 46–54. [Google Scholar] [CrossRef]
- Helal, G.A.; Sarhan, M.M.; Abu Shahla, A.N.K.; Abou El-Khair, E.K. Effects of Cymbopogoncitratus L. essential oil on the growth: Morphogenesis and aflatoxin production of Aspergillus flavus ML2-strain. J. Basic Microbiol. 2007, 47, 5–15. [Google Scholar] [CrossRef]
- Howlett, B.J. Secondary metabolite toxins and nutrition of plant pathogenic fungi. Curr. Opin. Plant Biol. 2006, 9, 371–375. [Google Scholar] [CrossRef]
- Troncoso-Rojas, R.; Tiznado-Hernández, M.E. Alternaria alternata (Block rot, black spot). In Postharvest Decay; Bautista-Banos, S., Ed.; Academic Press: Cambridge, MA, USA, 2014; pp. 147–187. [Google Scholar]
- López, P.; Venema, D.; de Rijk, T.; de Kok, A.; Sholten, J.M.; Mol, H.G.J.; de Nijs, M. Occurrence of Alternaria toxins in food products in the Netherlands. Food Control 2016, 60, 196–204. [Google Scholar] [CrossRef]
- Xu, L.; Tao, N.G.; Yang, W. Cinnamaldehyde damaged the cell membrane of, Alternaria alternata, and induced the degradation of mycotoxins, in vivo. Ind. Crop. Prod. 2018, 112, 427–433. [Google Scholar] [CrossRef]
- Wu, H.; Zhang, X.; Zhang, G.A.; Zeng, S.Y.; Lin, K.C. Antifungal vapour, hase activity of a combination of allyl isothiocyanate and ethyl isothiocyanate against botrytis cinerea and penicillium expansum infection on apples. J. Phytopathol. 2011, 159, 450–455. [Google Scholar]
- Li, X.; Jin, Z.; Wang, J. Complexation of allyl isothiocyanate by a- and b-cyclodextrin and its controlled release characteristics. Food Chem. 2007, 103, 461–466. [Google Scholar] [CrossRef]
- Wu, H.; Xue, N.; Hou, C.; Feng, J.; Zhang, X. Microcapsule preparation of allyl isothiocyanate and its application on mature green tomato preservation. Food Chem. 2015, 175, 344–349. [Google Scholar] [CrossRef] [PubMed]
- Tahir, I.I.; Nybom, H.; Ahmadi-Afzadi, M.; Røen, K.; Sehic, J.; Røen, D. Susceptibility to blue mold caused by Penicillium expansum in apple cultivars adapted to a cool climate. Eur. J. Hortic. Sci. 2015, 80, 117–127. [Google Scholar] [CrossRef]
- Jiang, Y.; Wang, D.; Li, F.; Li, D.P.; Huang, Q.R. Cinnamon essential oil Pickering emulsion stabilized by zein-pectincomposite nanoparticles: Characterization, antimicrobial effect andadvantages in storage application. Int. J. Biol. Macromol. 2019. [Google Scholar] [CrossRef]
- Li, H.; Zhang, Z.Q.; He, C.; Qin, G.Z.; Tian, S.P. Comparative proteomics reveals the potential targets of BcNoxR, a putative regulatory subunit of NADPH oxidase of Botrytis cinerea. Mol. Plant Microbe. Interact. 2016, 29, 990–1003. [Google Scholar] [CrossRef]
- Liu, J.; Zong, Y.Y.; Qin, G.Z.; Li, B.Q.; Tian, S.P. Plasma membrane damage contributes to antifungal activity of silicon against Penicillium digitatum. Curr. Microbiol. 2010, 61, 274–279. [Google Scholar] [CrossRef]
- Shao, X.; Cheng, S.; Wang, H.; Yu, D.; Mungai, C. The possible mechanism of antifungal action of tea tree oil on Botrytis cinerea. J. Appl. Microbiol. 2013, 114, 1642–1649. [Google Scholar] [CrossRef]
- Paul, S.; Dubey, R.C.; Maheswari, D.K.; Kang, S.C. Trachyspermumammi (L.) fruit essential oil influencing on membrane permeability and surface characteristics in inhibiting food-borne pathogens. Food Control 2001, 22, 725–731. [Google Scholar] [CrossRef]
- Wang, M.; Jiang, N.; Xian, H.; Wei, D.; Shi, L.; Feng, X. A single-step solid phaseextraction for the simultaneous determination of 8 mycotoxins in fruit by ultra-highperformance liquid chromatography tandem mass spectrometry. J. Chromatogr. A 2016, 1429, 22–29. [Google Scholar] [CrossRef]

| Target Compounds | Ionization Mode | Parent Ion | Qualitative Ion | Keep Time | Quantitative Ion | Fragmentation Voltage | Collision Voltage |
|---|---|---|---|---|---|---|---|
| Altemariol (AOH) | ESI+ | 257.0 | 213.0 | 2.37 | 147.2 | 40 | 32 |
| Altermariol monomethylether (AME) | ESI+ | 271.0 | 256.0 | 2.85 | 228.0 212.9 | 32 | 20 |
| Allenuene (ALT) | ESI+ | 293.1 | 257.2 | 3.33 | 239.1 | 85 | 15 |
| Tentoxin (TEN) | ESI+ | 415.2 | 312.3 | 3.66 | 189.0 | 110 | 30 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, M.; Li, Y.; Bi, Y.; Wang, T.; Dong, Y.; Yang, Q.; Zhang, T. 2-Phenylethyl Isothiocyanate Exerts Antifungal Activity against Alternaria alternata by Affecting Membrane Integrity and Mycotoxin Production. Toxins 2020, 12, 124. https://doi.org/10.3390/toxins12020124
Zhang M, Li Y, Bi Y, Wang T, Dong Y, Yang Q, Zhang T. 2-Phenylethyl Isothiocyanate Exerts Antifungal Activity against Alternaria alternata by Affecting Membrane Integrity and Mycotoxin Production. Toxins. 2020; 12(2):124. https://doi.org/10.3390/toxins12020124
Chicago/Turabian StyleZhang, Miao, Yongcai Li, Yang Bi, Tiaolan Wang, Yupeng Dong, Qian Yang, and Tingting Zhang. 2020. "2-Phenylethyl Isothiocyanate Exerts Antifungal Activity against Alternaria alternata by Affecting Membrane Integrity and Mycotoxin Production" Toxins 12, no. 2: 124. https://doi.org/10.3390/toxins12020124
APA StyleZhang, M., Li, Y., Bi, Y., Wang, T., Dong, Y., Yang, Q., & Zhang, T. (2020). 2-Phenylethyl Isothiocyanate Exerts Antifungal Activity against Alternaria alternata by Affecting Membrane Integrity and Mycotoxin Production. Toxins, 12(2), 124. https://doi.org/10.3390/toxins12020124






