Exposure to Aerosolized Algal Toxins in South Florida Increases Short- and Long-Term Health Risk in Drosophila Model of Aging
Abstract
:1. Introduction
2. Results
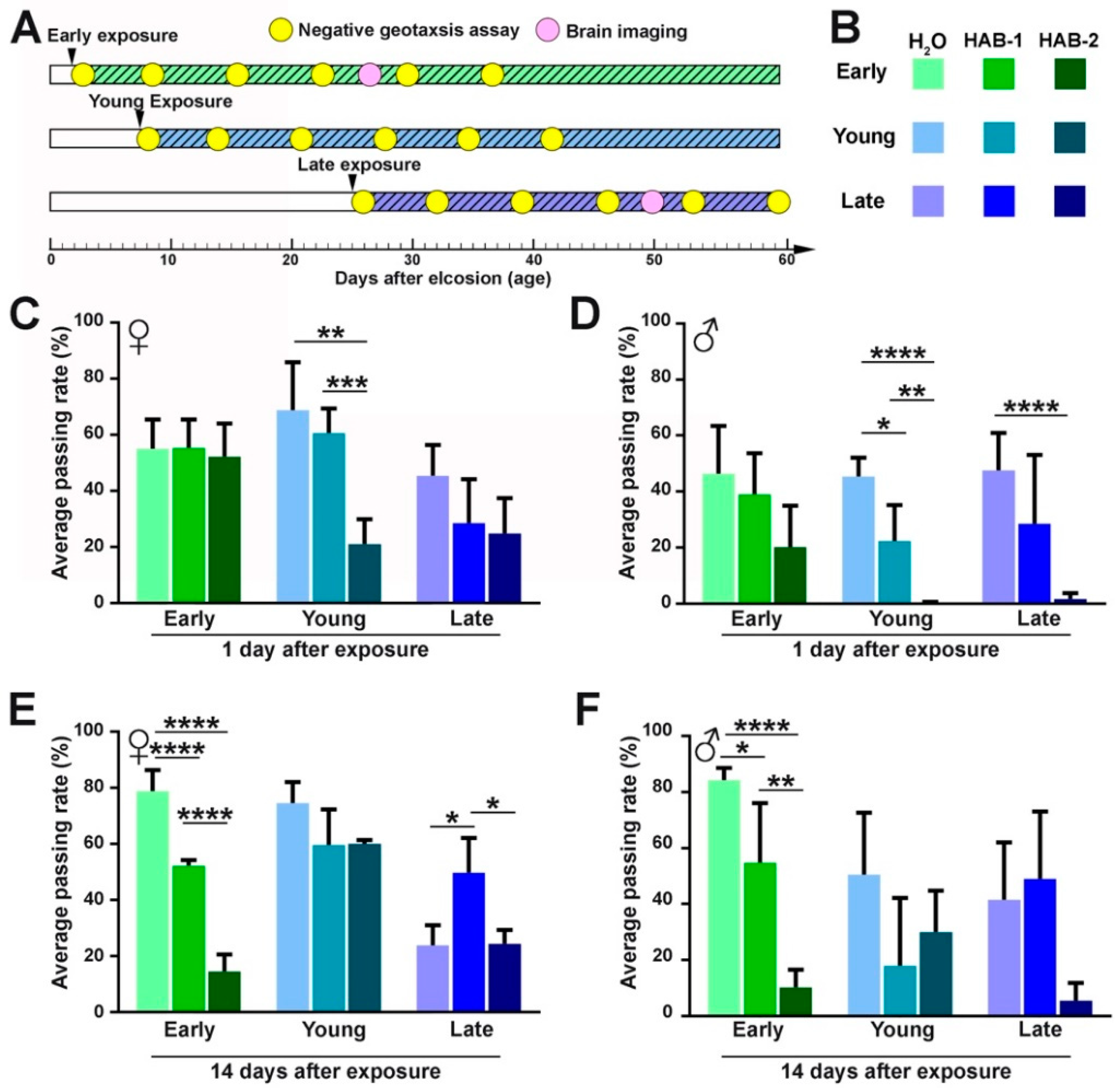
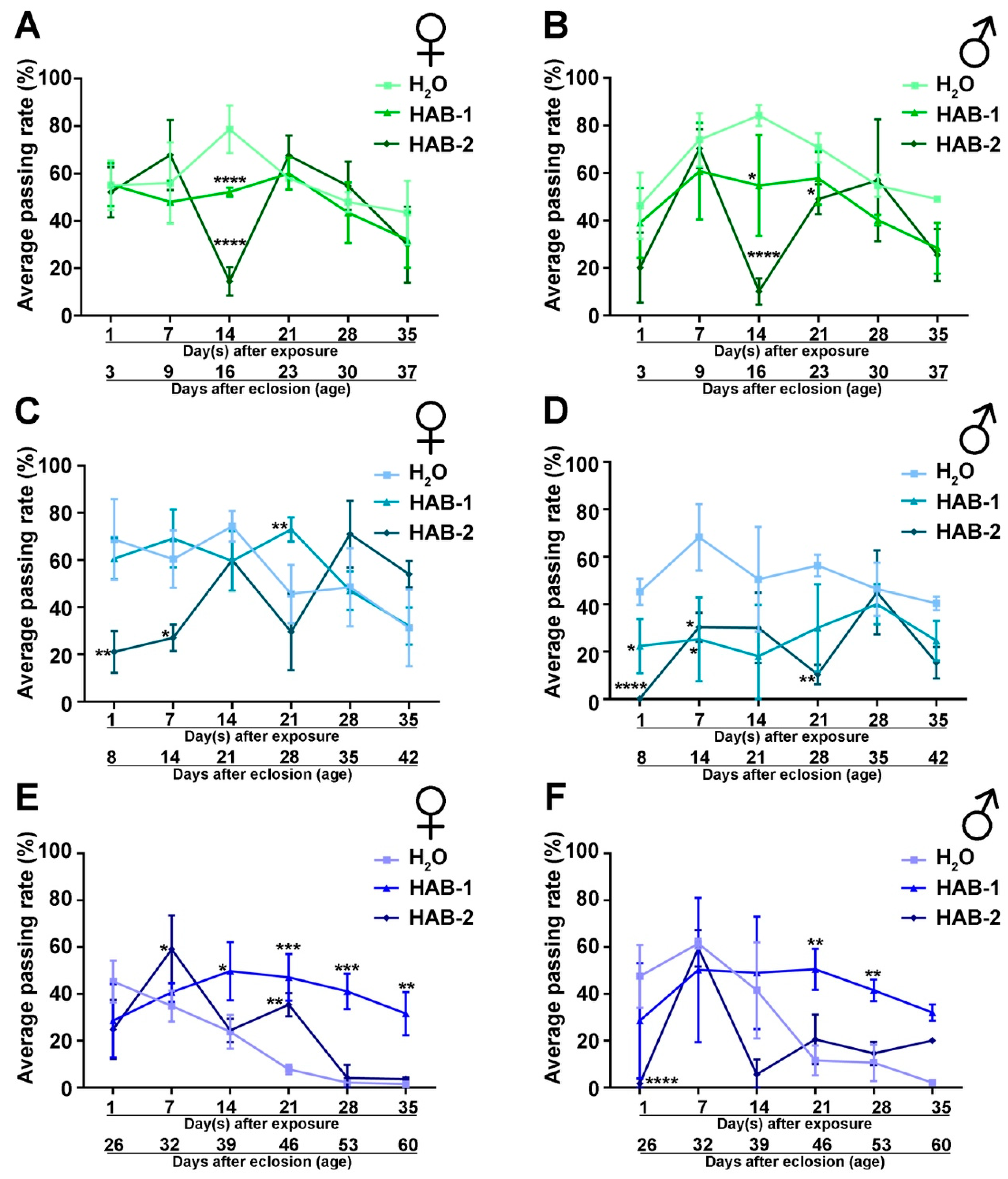
2.1. Aerosolized HABs Impairs Drosophila Locomotor Functions
2.2. Exposure to Aerosolized HABs Causes Long-Term Detriment in Neuromuscular Functions
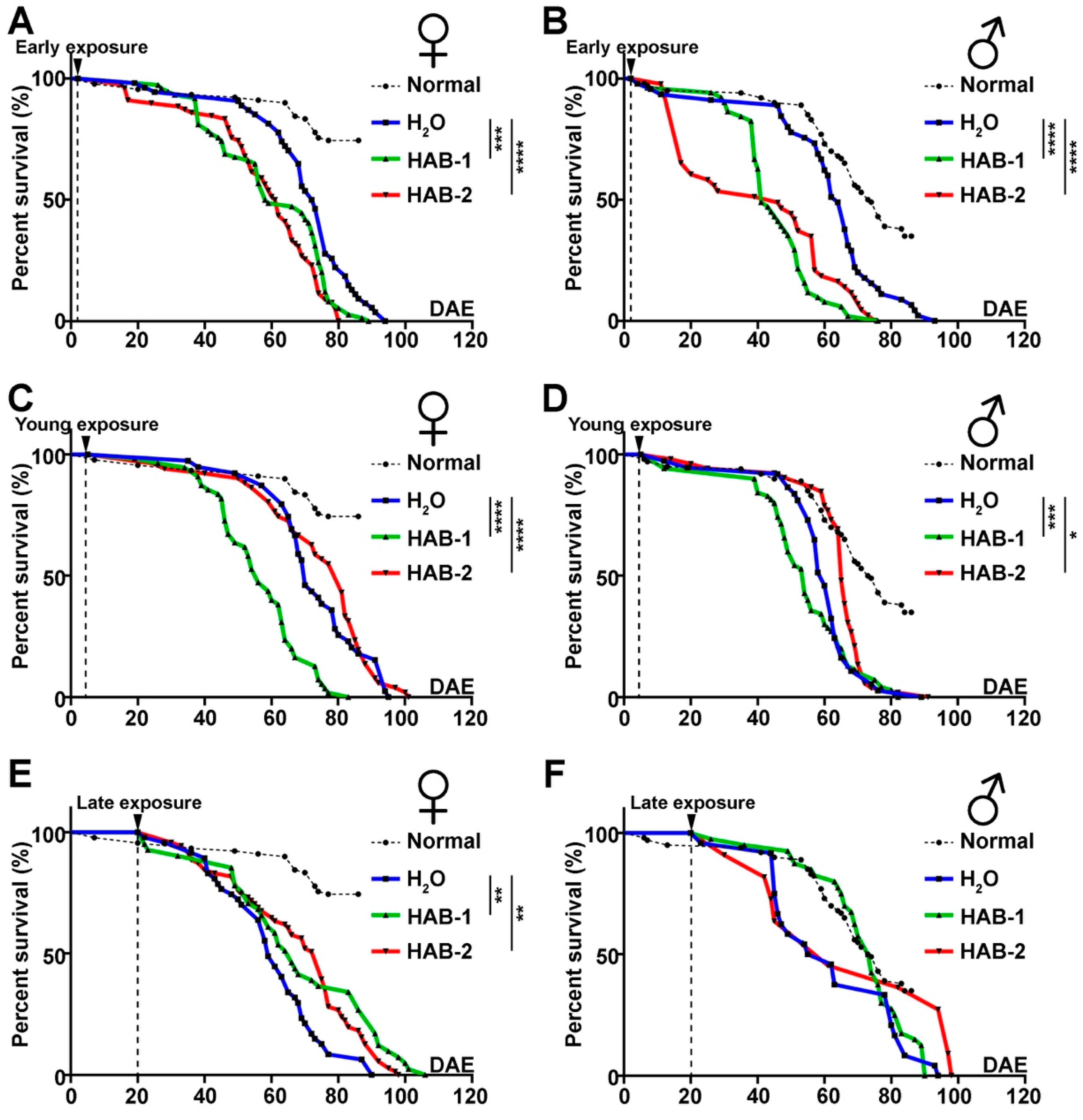
2.3. Early Exposure to HABs Particularly Reduces the Longevity of Drosophila
2.4. Aerosolized HAB Significantly Impairs Drosophila Synapse Function
2.5. BMAA and Microcystin Are Consistent Toxic Constituents of HABs
3. Discussion
3.1. Age at Exposure Is a Major Determinant of the Health Impact of Aerosolized HABs
3.2. Vulnerability of Gender Differences
3.3. Contribution of BMAA and Microcystin to the Aerosol Toxicity
3.4. Limitations and Future Directions
4. Conclusions
5. Materials and Methods
5.1. Aerosol Exposure Apparatus and Aerosol Collection
5.2. Drosophila Culture and Exposure Setup
5.3. HAB Water Collection and Toxin Sampling
5.4. Drosophila Behavior Analysis: Negative Geotaxis Assay
5.5. Drosophila Lifespan Assay
5.6. Immunofluorescence Imaging-Based Morphological Analysis of Drosophila Brain
5.7. Toxin Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hallegraeff, G.M. A review of harmful algal blooms and their apparent global increase. Phycologia 1993, 32, 79–99. [Google Scholar] [CrossRef] [Green Version]
- Anderson, D.M.; Glibert, P.M.; Burkholder, J.M. Harmful algal blooms and eutrophication: Nutrient sources, composition, and consequences. Estuaries 2002, 25, 704–726. [Google Scholar] [CrossRef]
- Hallegraeff, G.M. Harmful Algal Blooms: A Global Overview. In Manual on Harmful Marine Microalgae; UNESCO Publications: Paris, France, 2004. [Google Scholar]
- Anderson, D. HABs in a changing world: A perspective on harmful algal blooms, their impacts, and research and management in a dynamic era of climactic and environmental change. In Proceedings of the Harmful Algae 2012: 15th International Conference on Harmful Algae, CECO, Changwon, Gyeongnam, Korea, 29 October–2 November 2012; pp. 3–17. [Google Scholar]
- Schmale, D.G.; Ault, A.P.; Saad, W.; Scott, D.T.; Westrick, J.A. Perspectives on harmful algal blooms (HABs) and the cyberbiosecurity of freshwater systems. Front. Bioeng. Biotechnol. 2019, 7, 128. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brand, L.E. Human exposure to cyanobacteria and BMAA. Amyotroph. Lateral Scler. 2009, 10, 85–95. [Google Scholar] [CrossRef]
- Codd, G.; Bell, S.; Kaya, K.; Ward, C.; Beattie, K.; Metcalf, J. Cyanobacterial toxins, exposure routes and human health. Eur. J. Phycol. 1999, 34, 405–415. [Google Scholar] [CrossRef]
- Chernoff, N.; Hill, D.J.; Diggs, D.L.; Faison, B.D.; Francis, B.M.; Lang, J.R.; Larue, M.M.; Le, T.T.; Loftin, K.A.; Lugo, J.N.; et al. A critical review of the postulated role of the non-essential amino acid, β-N-methylamino-L-alanine, in neurodegenerative disease in humans. J. Toxicol. Environ. Heal. Part B 2017, 20, 183–229. [Google Scholar] [CrossRef]
- Falconer, I.R. Potential impact on human health of toxic cyanobacteria. Phycologia 1996, 35, 6–11. [Google Scholar] [CrossRef]
- Backer, L.C.; McNeel, S.V.; Barber, T.; Kirkpatrick, B.; Williams, C.; Irvin, M.; Zhou, Y.; Johnson, T.B.; Nierenberg, K.; Aubel, M.; et al. Recreational exposure to microcystins during algal blooms in two California lakes. Toxicon 2010, 55, 909–921. [Google Scholar] [CrossRef]
- Fleming, L.E.; Rivero, C.; Burns, J.; Williams, C.; Bean, J.A.; Shea, K.A.; Stinn, J. Blue green algal (cyanobacterial) toxins, surface drinking water, and liver cancer in Florida. Harmful Algae 2002, 1, 157–168. [Google Scholar] [CrossRef]
- Benson, J.M.; Hutt, J.A.; Rein, K.; Boggs, S.E.; Barr, E.B.; Fleming, L.E. The toxicity of microcystin LR in mice following 7 days of inhalation exposure. Toxicon 2005, 45, 691–698. [Google Scholar] [CrossRef] [Green Version]
- Bláha, L.; Babica, P.; Maršálek, B. Toxins produced in cyanobacterial water blooms–toxicity and risks. Interdiscip. Toxicol. 2009, 2, 36–41. [Google Scholar] [CrossRef] [Green Version]
- Downing, S.; Contardo-Jara, V.; Pflugmacher, S.; Downing, T.G. The fate of the cyanobacterial toxin β-N-methylamino-l-alanine in freshwater mussels. Ecotoxicol. Environ. Saf. 2014, 101, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Violi, J.P.; Mitrovic, S.M.; Colville, A.; Main, B.J.; Rodgers, K.J. Prevalence of β-methylamino-L-alanine (BMAA) and its isomers in freshwater cyanobacteria isolated from eastern Australia. Ecotoxicol. Environ. Saf. 2019, 172, 72–81. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Yolitz, J.; Wang, C.; Spangler, E.; Zhan, M.; Zou, S. Aging studies in drosophila melanogaster. Bioinform. MicroRNA Res. 2013, 1048, 77–93. [Google Scholar] [CrossRef] [Green Version]
- Akitake, B.; Ren, Q.; Boiko, N.; Ni, J.; Sokabe, T.; Stockand, J.D.; Eaton, B.A.; Montell, C. Coordination and fine motor control depend on Drosophila TRPγ. Nat. Commun. 2015, 6, 7288. [Google Scholar] [CrossRef] [Green Version]
- Manjila, S.; Hasan, G. Flight and Climbing Assay for Assessing Motor Functions in Drosophila. BIO-Protocol 2018, 8. [Google Scholar] [CrossRef]
- Zhu, Y.; Lazopulo, S.; Syed, S.; Zhai, R.G. Behavioral Studies in Drosophila Models of Human Diseases. In Reference Module in Neuroscience and Biobehavioral Psychology; Elsevier BV: Amsterdam, The Netherlands, 2020. [Google Scholar]
- Benzer, S. Behavioral Mutants of Drosophila isolated by countercurrent distribution. Proc. Natl. Acad. Sci. USA 1967, 58, 1112–1119. [Google Scholar] [CrossRef] [Green Version]
- Ganetzky, B.; Flanagan, J.R. On the relationship between senescence and age-related changes in two wild-type strains of Drosophila melanogaster. Exp. Gerontol. 1978, 13, 189–196. [Google Scholar] [CrossRef]
- Ali, Y.O.; Escala, W.; Ruan, K.; Zhai, R.G. Assaying locomotor, learning, and memory deficits in Drosophila models of neurodegeneration. J. Vis. Exp. 2011, e2504. [Google Scholar] [CrossRef] [Green Version]
- Zhu, Y.; Li, C.; Tao, X.; Brazill, J.M.; Park, J.; Diaz-Perez, Z.; Grace Zhai, R. Nmnat restores neuronal integrity by neutralizing mutant Huntingtin aggregate-induced progressive toxicity. Proc. Natl. Acad. Sci. USA 2019, 116, 19165–19175. [Google Scholar] [CrossRef] [Green Version]
- Riemensperger, T.; Issa, A.R.; Pech, U.; Coulom, H.; Nguyễn, M.V.; Cassar, M.; Jacquet, M.; Fiala, A.; Birman, S. A Single Dopamine Pathway Underlies Progressive Locomotor Deficits in a Drosophila Model of Parkinson Disease. Cell Rep. 2013, 5, 952–960. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ruan, K.; Zhu, Y.; Li, C.; Brazill, J.M.; Zhai, R.G. Alternative splicing of Drosophila Nmnat functions as a switch to enhance neuroprotection under stress. Nat. Commun. 2015, 6, 10057. [Google Scholar] [CrossRef] [Green Version]
- Zhou, X.; Escala, W.; Papapetropoulos, S.; Grace Zhai, R. β-N-methylamino-L-alanine induces neurological deficits and shortened life span in drosophila. Toxins 2010, 2, 2663–2679. [Google Scholar] [CrossRef] [PubMed]
- Piper, M.D.W.; Partridge, L. Protocols to study aging in drosophila. In Methods in Molecular Biology; Springer Science and Business Media LLC: Berlin, Germany, 2016; Volume 1478, pp. 291–302. [Google Scholar]
- Linford, N.J.; Bilgir, C.; Ro, J.; Pletcher, S.D. Measurement of lifespan in Drosophila melanogaster. J. Vis. Exp. 2013, e50068. [Google Scholar] [CrossRef] [Green Version]
- Adams, M.D.; Celniker, S.E.; Holt, R.A.; Evans, C.A.; Gocayne, J.D.; Amanatides, P.G.; Scherer, S.E.; Li, P.W.; Hoskins, R.A.; Galle, R.F.; et al. The genome sequence of Drosophila melanogaster. Science 2000, 287, 2185–2195. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tito, A.J.; Cheema, S.; Jiang, M.; Zhang, S. A simple one-step dissection protocol for whole-mount preparation of adult drosophila brains. J. Vis. Exp. 2016. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.K.; Ma, P.L.; Ji, S.Y.; Zhao, X.L.; Tan, J.X.; Sun, X.J.; Huang, F.D. Age-dependent alterations in the presynaptic active zone in a Drosophila model of Alzheimer’s Disease. Neurobiol. Dis. 2013, 51, 161–167. [Google Scholar] [CrossRef]
- Krzeptowski, W.; Górska-Andrzejak, J.; Kijak, E.; Görlich, A.; Guzik, E.; Moore, G.; Pyza, E.M. External and circadian inputs modulate synaptic protein expression in the visual system of Drosophila melanogaster. Front. Physiol. 2014, 5. [Google Scholar] [CrossRef] [Green Version]
- Doktór, B.; Damulewicz, M.; Krzeptowski, W.; Bednarczyk, B.; Pyza, E. Effects of pink1 mutation on synapses and behavior in the brain of drosophila melanogaster. Acta Neurobiol. Exp. 2018, 78, 231–241. [Google Scholar] [CrossRef] [Green Version]
- Olson, N.E.; Cooke, M.E.; Shi, J.H.; Birbeck, J.A.; Westrick, J.A.; Ault, A.P. Harmful Algal Bloom Toxins in Aerosol Generated from Inland Lake Water. Environ. Sci. Technol. 2020, 54, 4769–4780. [Google Scholar] [CrossRef]
- May, N.W.; Olson, N.E.; Panas, M.; Axson, J.L.; Tirella, P.S.; Kirpes, R.M.; Craig, R.L.; Gunsch, M.J.; China, S.; Laskin, A.; et al. Aerosol Emissions from Great Lakes Harmful Algal Blooms. Environ. Sci. Technol. 2018, 52, 397–405. [Google Scholar] [CrossRef] [PubMed]
- Zanchett, G.; Oliveira-Filho, E.C. Cyanobacteria and cyanotoxins: From impacts on aquatic ecosystems and human health to anticarcinogenic effects. Toxins 2013, 5, 1896–1917. [Google Scholar] [CrossRef] [PubMed]
- Rao, P.V.L.; Gupta, N.; Jayaraj, R.; Bhaskar, A.S.B.; Jatav, P.C. Age-dependent effects on biochemical variables and toxicity induced by cyclic peptide toxin microcystin-LR in mice. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2005, 140, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Beam, C.R.; Kaneshiro, C.; Jang, J.Y.; Reynolds, C.A.; Pedersen, N.L.; Gatz, M. Differences between Women and Men in Incidence Rates of Dementia and Alzheimer’s Disease. J. Alzheimer’s Dis. 2018, 64, 1077–1083. [Google Scholar] [CrossRef]
- Simunovic, F.; Yi, M.; Wang, Y.; Stephens, R.; Sonntag, K.C. Evidence for gender-specific transcriptional profiles of nigral dopamine neurons in Parkinson Disease. PLoS ONE 2010, 5, e8856. [Google Scholar] [CrossRef]
- Le Manach, S.; Khenfech, N.; Huet, H.; Qiao, Q.; Duval, C.; Marie, A.; Bolbach, G.; Clodic, G.; Djediat, C.; Bernard, C.; et al. Gender-Specific Toxicological Effects of Chronic Exposure to Pure Microcystin-LR or Complex Microcystis aeruginosa Extracts on Adult Medaka Fish. Environ. Sci. Technol. 2016, 50, 8324–8334. [Google Scholar] [CrossRef]
- Keene, W.C.; Maring, H.; Maben, J.R.; Kieber, D.J.; Pszenny, A.A.P.; Dahl, E.E.; Izaguirre, M.A.; Davis, A.J.; Long, M.S.; Zhou, X.; et al. Chemical and physical characteristics of nascent aerosols produced by bursting bubbles at a model air-sea interface. J. Geophys. Res. Space Phys. 2007, 112. [Google Scholar] [CrossRef] [Green Version]
- Gaston, C.J.; Furutani, H.; Guazzotti, S.A.; Coffee, K.R.; Bates, T.S.; Quinn, P.K.; Aluwihare, L.I.; Mitchell, B.G.; Prather, K.A. Unique ocean-derived particles serve as a proxy for changes in ocean chemistry. J. Geophys. Res. Space Phys. 2011, 116. [Google Scholar] [CrossRef]
- Moore, M.J.K.; Furutani, H.; Roberts, G.C.; Moffet, R.C.; Gilles, M.K.; Palenik, B.; Prather, K.A. Effect of organic compounds on cloud condensation nuclei (CCN) activity of sea spray aerosol produced by bubble bursting. Atmos. Environ. 2011. [Google Scholar] [CrossRef]
- Tamura, T.; Chiang, A.S.; Ito, N.; Liu, H.P.; Horiuchi, J.; Tully, T.; Saitoe, M. Aging Specifically Impairs amnesiac-Dependent Memory in Drosophila. Neuron 2003, 40, 1003–1011. [Google Scholar] [CrossRef]
- Brazill, J.M.; Zhu, Y.; Li, C.; Zhai, R.G. Quantitative cell biology of neurodegeneration in drosophila through unbiased analysis of fluorescently tagged proteins using imageJ. J. Vis. Exp. 2018, e58041. [Google Scholar] [CrossRef] [PubMed]
- Réveillon, D.; Abadie, E.; Séchet, V.; Brient, L.; Savar, V.; Bardouil, M.; Hess, P.; Amzil, Z. Beta-N-methylamino-L-alanine: LC-MS/MS optimization, screening of cyanobacterial strains and occurrence in shellfish from Thau, a French Mediterranean Lagoon. Mar. Drugs 2014, 12, 5441–5467. [Google Scholar] [CrossRef] [PubMed]
- Gambaro, A.; Barbaro, E.; Zangrando, R.; Barbante, C. Simultaneous quantification of microcystins and nodularin in aerosol samples using high-performance liquid chromatography/negative electrospray ionization tandem mass spectrometry. Rapid Commun. Mass Spectrom. 2012, 26, 1497–1506. [Google Scholar] [CrossRef] [PubMed]

| Water Sample | At the Time of Water Sample Collection | At the Time of Exposure Experiment | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Number | Date | Location | Chloro-phyll (μg/L) | Micro-cystin (μg/L) | BMAA (ng/L) | Date | Micro-cystin, water (μg/L) | BMAA, water (ng/L) | Micro-cystin, air (pg/L) |
| HAB-1 | 2 September 2018 | Cape Coral | 555 | 319.93 | <LOQ | 1 May 2019 | 0.37 | 7.2 | <LOQ |
| HAB-2 | 3 March 2019 | Lake Okeechobee | 4.3 | N.D. | N.D. | 3 April 2019 | 0.01 | 1.7 | <LOQ |
| HAB-3 | n/a | Monospecific culture of Microcystis aeruginosa | n/a | n/a | n/a | 9 January 2020 | 379.38 | N.D. | 62 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hu, J.; Liu, J.; Zhu, Y.; Diaz-Perez, Z.; Sheridan, M.; Royer, H.; Leibensperger, R., III; Maizel, D.; Brand, L.; Popendorf, K.J.; et al. Exposure to Aerosolized Algal Toxins in South Florida Increases Short- and Long-Term Health Risk in Drosophila Model of Aging. Toxins 2020, 12, 787. https://doi.org/10.3390/toxins12120787
Hu J, Liu J, Zhu Y, Diaz-Perez Z, Sheridan M, Royer H, Leibensperger R III, Maizel D, Brand L, Popendorf KJ, et al. Exposure to Aerosolized Algal Toxins in South Florida Increases Short- and Long-Term Health Risk in Drosophila Model of Aging. Toxins. 2020; 12(12):787. https://doi.org/10.3390/toxins12120787
Chicago/Turabian StyleHu, Jiaming, Jiaqi Liu, Yi Zhu, Zoraida Diaz-Perez, Michael Sheridan, Haley Royer, Raymond Leibensperger, III, Daniela Maizel, Larry Brand, Kimberly J. Popendorf, and et al. 2020. "Exposure to Aerosolized Algal Toxins in South Florida Increases Short- and Long-Term Health Risk in Drosophila Model of Aging" Toxins 12, no. 12: 787. https://doi.org/10.3390/toxins12120787
APA StyleHu, J., Liu, J., Zhu, Y., Diaz-Perez, Z., Sheridan, M., Royer, H., Leibensperger, R., III, Maizel, D., Brand, L., Popendorf, K. J., Gaston, C. J., & Zhai, R. G. (2020). Exposure to Aerosolized Algal Toxins in South Florida Increases Short- and Long-Term Health Risk in Drosophila Model of Aging. Toxins, 12(12), 787. https://doi.org/10.3390/toxins12120787






