Pseudomonas Exotoxin A Based Toxins Targeting Epidermal Growth Factor Receptor for the Treatment of Prostate Cancer
Abstract
:1. Introduction
2. Results
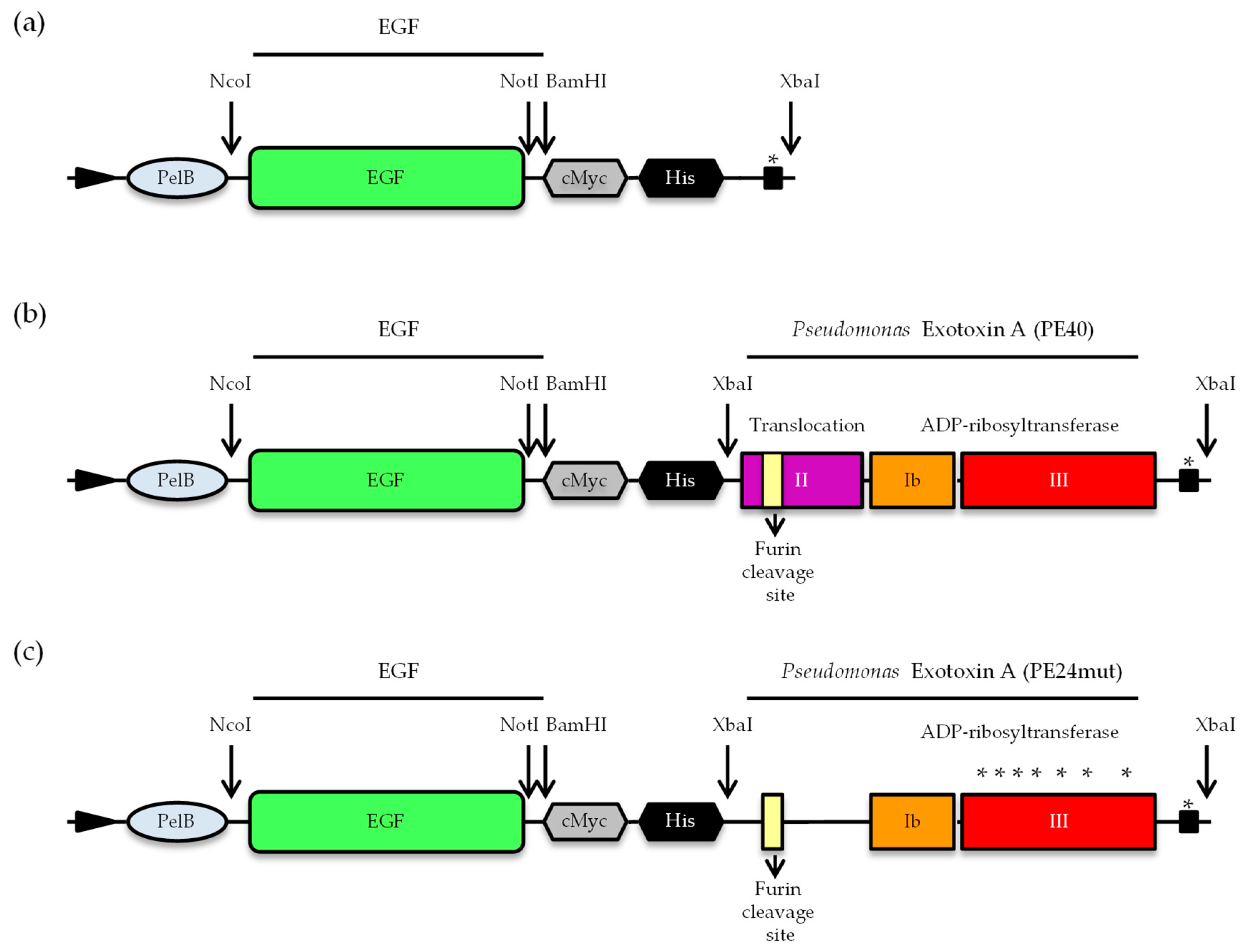
2.1. Cloning, Expression and Purification of EGF and the Targeted Toxins EGF-PE40 and EGF-PE24mut
2.2. Binding of EGF-PE40 and EGF-PE24mut to Different PCa Cell Lines
2.3. EGF-PE40 and EGF-PE24mut Inhibit Protein Biosynthesis
2.4. EGF-PE40 and EGF-PE24mut Reduce Cell Viability and Induce Apoptosis
3. Discussion
4. Materials and Methods
4.1. Cells and Chemicals
4.2. Cloning, Expression and Purification of EGF and the Anti-EGFR Immunotoxins
4.3. Preparation of Cell Lysates
4.4. SDS-PAGE and Western Blot
4.5. Flow Cytometry
4.6. Inhibition of Protein Biosynthesis
4.7. WST-1 Cell Viability Assay
4.8. Bright-Field Microscopy
4.9. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: Globocan estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Litwin, M.S.; Tan, H.J. The diagnosis and treatment of prostate cancer: A review. JAMA 2017, 317, 2532–2542. [Google Scholar] [CrossRef] [PubMed]
- Cha, H.R.; Lee, J.H.; Ponnazhagan, S. Revisiting immunotherapy: A focus on prostate cancer. Cancer Res. 2020, 80, 1615–1623. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fay, E.K.; Graff, J.N. Immunotherapy in prostate cancer. Cancers 2020, 12, 1752. [Google Scholar] [CrossRef] [PubMed]
- Elsässer-Beile, U.; Bühler, P.; Wolf, P. Targeted therapies for prostate cancer against the prostate specific membrane antigen. Curr. Drug Targets 2009, 10, 118–125. [Google Scholar] [CrossRef] [PubMed]
- Cimadamore, A.; Cheng, M.; Santoni, M.; Lopez-Beltran, A.; Battelli, N.; Massari, F.; Galosi, A.B.; Scarpelli, M.; Montironi, R. New prostate cancer targets for diagnosis, imaging, and therapy: Focus on prostate-specific membrane antigen. Front. Oncol. 2018, 8, 653. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, X.; Guo, Z.; Liu, Y.; Si, T.; Yu, H.; Li, B.; Tian, W. Prostate stem cell antigen and cancer risk, mechanisms and therapeutic implications. Expert Rev. Anticancer Ther. 2014, 14, 31–37. [Google Scholar] [CrossRef]
- Antonarakis, E.S.; Carducci, M.A.; Eisenberger, M.A. Novel targeted therapeutics for metastatic castration-resistant prostate cancer. Cancer Lett. 2010, 291, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Lorenzo, G.D.; Bianco, R.; Tortora, G.; Ciardiello, F. Involvement of growth factor receptors of the epidermal growth factor receptor family in prostate cancer development and progression to androgen independence. Clin. Prostate Cancer 2003, 2, 50–57. [Google Scholar] [CrossRef]
- Guérin, O.; Fischel, J.L.; Ferrero, J.M.; Bozec, A.; Milano, G. EGFR targeting in hormone-refractory prostate cancer: Current appraisal and prospects for treatment. Pharmaceuticals 2010, 3, 2238–2247. [Google Scholar] [CrossRef] [Green Version]
- Burgess, A.W. Egfr family: Structure physiology signalling and therapeutic targets. Growth Factors 2008, 26, 263–274. [Google Scholar] [CrossRef]
- Ogiso, H.; Ishitani, R.; Nureki, O.; Fukai, S.; Yamanaka, M.; Kim, J.H.; Saito, K.; Sakamoto, A.; Inoue, M.; Shirouzu, M.; et al. Crystal structure of the complex of human epidermal growth factor and receptor extracellular domains. Cell 2002, 110, 775–787. [Google Scholar] [CrossRef] [Green Version]
- Ferguson, K.M. Structure-based view of epidermal growth factor receptor regulation. Ann. Rev. Biophys. 2008, 37, 353–373. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harris, R.C.; Chung, E.; Coffey, R.J. EGF receptor ligands. Exp. Cell Res. 2003, 284, 2–13. [Google Scholar] [CrossRef]
- Wee, P.; Wang, Z. Epidermal growth factor receptor cell proliferation signaling pathways. Cancers 2017, 9, 52. [Google Scholar] [CrossRef] [Green Version]
- Bonaccorsi, L.; Nosi, D.; Muratori, M.; Formigli, L.; Forti, G.; Baldi, E. Altered endocytosis of epidermal growth factor receptor in androgen receptor positive prostate cancer cell lines. J. Mol. Endocrinol. 2007, 38, 51–66. [Google Scholar] [CrossRef] [Green Version]
- Day, K.C.; Lorenzatti Hiles, G.; Kozminsky, M.; Dawsey, S.J.; Paul, A.; Broses, L.J.; Shah, R.; Kunja, L.P.; Hall, C.; Palanisamy, N.; et al. Her2 and EGFR overexpression support metastatic progression of prostate cancer to bone. Cancer Res. 2017, 77, 74–85. [Google Scholar] [CrossRef] [Green Version]
- DeHaan, A.M.; Wolters, N.M.; Keller, E.T.; Ignatoski, K.M. Egfr ligand switch in late stage prostate cancer contributes to changes in cell signaling and bone remodeling. Prostate 2009, 69, 528–537. [Google Scholar] [CrossRef] [Green Version]
- Chang, Y.S.; Chen, W.Y.; Yin, J.J.; Sheppard-Tillman, H.; Huang, J.; Liu, Y.N. Egf receptor promotes prostate cancer bone metastasis by downregulating mir-1 and activating twist1. Cancer Res. 2015, 75, 3077–3086. [Google Scholar] [CrossRef] [Green Version]
- De Muga, S.; Hernandez, S.; Agell, L.; Salido, M.; Juanpere, N.; Lorenzo, M.; Lorente, J.A.; Serrano, S.; Lloreta, J. Molecular alterations of EGFR and PTEN in prostate cancer: Association with high-grade and advanced-stage carcinomas. Modern Pathol. 2010, 23, 703–712. [Google Scholar] [CrossRef]
- Di Lorenzo, G.; Tortora, G.; D’Armiento, F.P.; De Rosa, G.; Staibano, S.; Autorino, R.; D’Armiento, M.; De Laurentiis, M.; De Placido, S.; Catalano, G.; et al. Expression of epidermal growth factor receptor correlates with disease relapse and progression to androgen-independence in human prostate cancer. Clin. Cancer Res. 2002, 8, 3438–3444. [Google Scholar]
- Schlomm, T.; Kirstein, P.; Iwers, L.; Daniel, B.; Steuber, T.; Walz, J.; Chun, F.H.; Haese, A.; Kollermann, J.; Graefen, M.; et al. Clinical significance of epidermal growth factor receptor protein overexpression and gene copy number gains in prostate cancer. Clin. Cancer Res. 2007, 13, 6579–6584. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hour, T.C.; Chung, S.D.; Kang, W.Y.; Lin, Y.C.; Chuang, S.J.; Huang, A.M.; Wu, W.J.; Huang, S.P.; Huang, C.Y.; Pu, Y.S. Egfr mediates docetaxel resistance in human castration-resistant prostate cancer through the akt-dependent expression of abcb1 (mdr1). Arch. Toxicol. 2015, 89, 591–605. [Google Scholar] [CrossRef] [PubMed]
- Gravis, G.; Bladou, F.; Salem, N.; Gonçalves, A.; Esterni, B.; Walz, J.; Bagattini, S.; Marcy, M.; Brunelle, S.; Viens, P. Results from a monocentric phase ii trial of erlotinib in patients with metastatic prostate cancer. Ann. Oncol. 2008, 19, 1624–1628. [Google Scholar] [CrossRef] [PubMed]
- Canil, C.M.; Moore, M.J.; Winquist, E.; Baetz, T.; Pollak, M.; Chi, K.N.; Berry, S.; Ernst, D.S.; Douglas, L.; Brundage, M.; et al. Randomized phase ii study of two doses of gefitinib in hormone-refractory prostate cancer: A trial of the national cancer institute of Canada-clinical trials group. J. Clin. Oncol 2005, 23, 455–460. [Google Scholar] [CrossRef] [PubMed]
- Gross, M.; Higano, C.; Pantuck, A.; Castellanos, O.; Green, E.; Nguyen, K.; Agus, D.B. A phase ii trial of docetaxel and erlotinib as first-line therapy for elderly patients with androgen-independent prostate cancer. BMC Cancer 2007, 7, 142. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Horti, J.; Widmark, A.; Stenzl, A.; Federico, M.H.; Abratt, R.P.; Sanders, N.; Pover, G.M.; Bodrogi, I. A randomized, double-blind, placebo-controlled phase ii study of vandetanib plus docetaxel/prednisolone in patients with hormone-refractory prostate cancer. Cancer Biother. Radiopharm. 2009, 24, 175–180. [Google Scholar] [CrossRef] [PubMed]
- Cathomas, R.; Rothermundt, C.; Klingbiel, D.; Bubendorf, L.; Jaggi, R.; Betticher, D.C.; Brauchli, P.; Cotting, D.; Droege, C.; Winterhalder, R.; et al. Efficacy of cetuximab in metastatic castration-resistant prostate cancer might depend on egfr and pten expression: Results from a phase ii trial (sakk 08/07). Clin. Cancer Res. 2012, 18, 6049–6057. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Niesen, J.; Stein, C.; Brehm, H.; Hehmann-Titt, G.; Fendel, R.; Melmer, G.; Fischer, R.; Barth, S. Novel egfr-specific immunotoxins based on panitumumab and cetuximab show in vitro and ex vivo activity against different tumor entities. J. Cancer Res. Clinic. Oncol. 2015, 141, 2079–2095. [Google Scholar] [CrossRef]
- Wiley, H.S. Trafficking of the erbb receptors and its influence on signaling. Exp. Cell Res. 2003, 284, 78–88. [Google Scholar] [CrossRef]
- Simon, N.; FitzGerald, D. Immunotoxin therapies for the treatment of epidermal growth factor receptor-dependent cancers. Toxins 2016, 8, 137. [Google Scholar] [CrossRef] [Green Version]
- Yip, W.L.; Weyergang, A.; Berg, K.; Tønnesen, H.H.; Selbo, P.K. Targeted delivery and enhanced cytotoxicity of cetuximab-saporin by photochemical internalization in egfr-positive cancer cells. Mol. Pharm. 2007, 4, 241–251. [Google Scholar] [CrossRef] [PubMed]
- Mazor, R.; Pastan, I. Immunogenicity of immunotoxins containing pseudomonas exotoxin a: Causes, consequences, and mitigation. Front. Immunol. 2020, 11, 1261. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Onda, M.; Lee, B.; Kreitman, R.J.; Hassan, R.; Xiang, L.; Pastan, I. Recombinant immunotoxin engineered for low immunogenicity and antigenicity by identifying and silencing human b-cell epitopes. Proc. Natl. Acad. Sci. USA 2012, 109, 11782–11787. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wolf, P.; Elsasser-Beile, U. Pseudomonas exotoxin a: From virulence factor to anti-cancer agent. Int. J. Med. Microbiol. 2009, 299, 161–176. [Google Scholar] [CrossRef] [PubMed]
- Yarmolinsky, M.B.; Haba, G.L. Inhibition by puromycin of amino acid incorporation into protein. Proc. Natl. Acad. Sci. USA 1959, 45, 1721–1729. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nathans, D. Puromycin inhibition of protein synthesis: Incorporation of puromycin into peptide chains. Proc. Natl. Acad. Sci USA 1964, 51, 585–592. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Russell, P.J.; Kingsley, E.A. Human prostate cancer cell lines. Methods Mol. Med. 2003, 81, 21–39. [Google Scholar]
- Mazor, R.; Onda, M.; Pastan, I. Immunogenicity of therapeutic recombinant immunotoxins. Immunol. Rev. 2016, 270, 152–164. [Google Scholar] [CrossRef] [Green Version]
- Azemar, M.; Schmidt, M.; Arlt, F.; Kennel, P.; Brandt, B.; Papadimitriou, A.; Groner, B.; Wels, W. Recombinant antibody toxins specific for erbb2 and egf receptor inhibit the in vitro growth of human head and neck cancer cells and cause rapid tumor regression in vivo. Int. J. Cancer 2000, 86, 269–275. [Google Scholar] [CrossRef]
- Michalska, M.; Wolf, P. Pseudomonas exotoxin a: Optimized by evolution for effective killing. Front. Microbiol. 2015, 6, 963. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van Ness, B.G.; Howard, J.B.; Bodley, J.W. Adp-ribosylation of elongation factor 2 by diphtheria toxin. Nmr spectra and proposed structures of ribosyl-diphthamide and its hydrolysis products. J. Biol. Chem. 1980, 255, 10710–10716. [Google Scholar] [PubMed]
- Ghosh, A.; Heston, W.D. Tumor target prostate specific membrane antigen (psma) and its regulation in prostate cancer. J. Cell Biochem. 2004, 91, 528–539. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, H.; Weng, A.; Gilabert-Oriol, R. Augmenting the efficacy of immunotoxins and other targeted protein toxins by endosomal escape enhancers. Toxins 2016, 8, 200. [Google Scholar] [CrossRef] [PubMed]
- Kondow-McConaghy, H.M.; Muthukrishnan, N.; Erazo-Oliveras, A.; Najjar, K.; Juliano, R.L.; Pellois, J.P. Impact of the endosomal escape activity of cell-penetrating peptides on the endocytic pathway. ACS Chem. Biol. 2020, 15, 2355–2363. [Google Scholar] [CrossRef] [PubMed]
- Jerjes, W.; Theodossiou, T.A.; Hirschberg, H.; Hogset, A.; Weyergang, A.; Selbo, P.K.; Hamdoon, Z.; Hopper, C.; Berg, K. Photochemical internalization for intracellular drug delivery. From basic mechanisms to clinical research. J. Clin. Med. 2020, 9, 528. [Google Scholar] [CrossRef] [Green Version]
- Fuchs, H.; Niesler, N.; Trautner, A.; Sama, S.; Jerz, G.; Panjideh, H.; Weng, A. Glycosylated triterpenoids as endosomal escape enhancers in targeted tumor therapies. Biomedicines 2017, 5, 14. [Google Scholar] [CrossRef] [Green Version]
- Kipriyanov, S.M.; Moldenhauer, G.; Little, M. High level production of soluble single chain antibodies in small-scale escherichia coli cultures. J. Immunol. Methods 1997, 200, 69–77. [Google Scholar] [CrossRef]
- Wolf, P.; Alt, K.; Wetterauer, D.; Buhler, P.; Gierschner, D.; Katzenwadel, A.; Wetterauer, U.; Elsasser-Beile, U. Preclinical evaluation of a recombinant anti-prostate specific membrane antigen single-chain immunotoxin against prostate cancer. J. Immunother. 2010, 33, 262–271. [Google Scholar] [CrossRef]
- Michalska, M.; Schultze-Seemann, S.; Bogatyreva, L.; Hauschke, D.; Wetterauer, U.; Wolf, P. In vitro and in vivo effects of a recombinant anti-psma immunotoxin in combination with docetaxel against prostate cancer. Oncotarget 2016, 7, 22531–22542. [Google Scholar] [CrossRef] [Green Version]




| LNCaP | DU145 | PC-3 | CHO | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| IC50(nM) | IC50(nM) | IC50(nM) | IC50(nM) | |||||||||
| 24h | 48h | 72h | 24h | 48h | 72h | 24h | 48h | 72h | 24h | 48h | 72h | |
| EGF-PE40 | 2.31 | 0.04 | 0.008 | 2.71 | 0.31 | 0.18 | 9.67 | 0.64 | 0.86 | >14.5 | >14.5 | >14.5 |
| EGF-PE24mut | >20.6 | 1.91 | 0.97 | >20.6 | 8.52 | 5.27 | >20.6 | 8.80 | 9.59 | >20.6 | >20.6 | >20.6 |
| Erlotinib | nd | >5750 | nd | nd | nd | >5750 | nd | nd | >5750 | nd | nd | >5750 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fischer, A.; Wolf, I.; Fuchs, H.; Masilamani, A.P.; Wolf, P. Pseudomonas Exotoxin A Based Toxins Targeting Epidermal Growth Factor Receptor for the Treatment of Prostate Cancer. Toxins 2020, 12, 753. https://doi.org/10.3390/toxins12120753
Fischer A, Wolf I, Fuchs H, Masilamani AP, Wolf P. Pseudomonas Exotoxin A Based Toxins Targeting Epidermal Growth Factor Receptor for the Treatment of Prostate Cancer. Toxins. 2020; 12(12):753. https://doi.org/10.3390/toxins12120753
Chicago/Turabian StyleFischer, Alexandra, Isis Wolf, Hendrik Fuchs, Anie Priscilla Masilamani, and Philipp Wolf. 2020. "Pseudomonas Exotoxin A Based Toxins Targeting Epidermal Growth Factor Receptor for the Treatment of Prostate Cancer" Toxins 12, no. 12: 753. https://doi.org/10.3390/toxins12120753





