Aberrant Expressional Profiling of Known MicroRNAs in the Liver of Silver Carp (Hypophthalmichthys molitrix) Following Microcystin-LR Exposure Based on samllRNA Sequencing
Abstract
1. Introduction
2. Results
2.1. Solexa Sequencing of Small RNAs
2.2. Differentially Expressed Known miRNAs
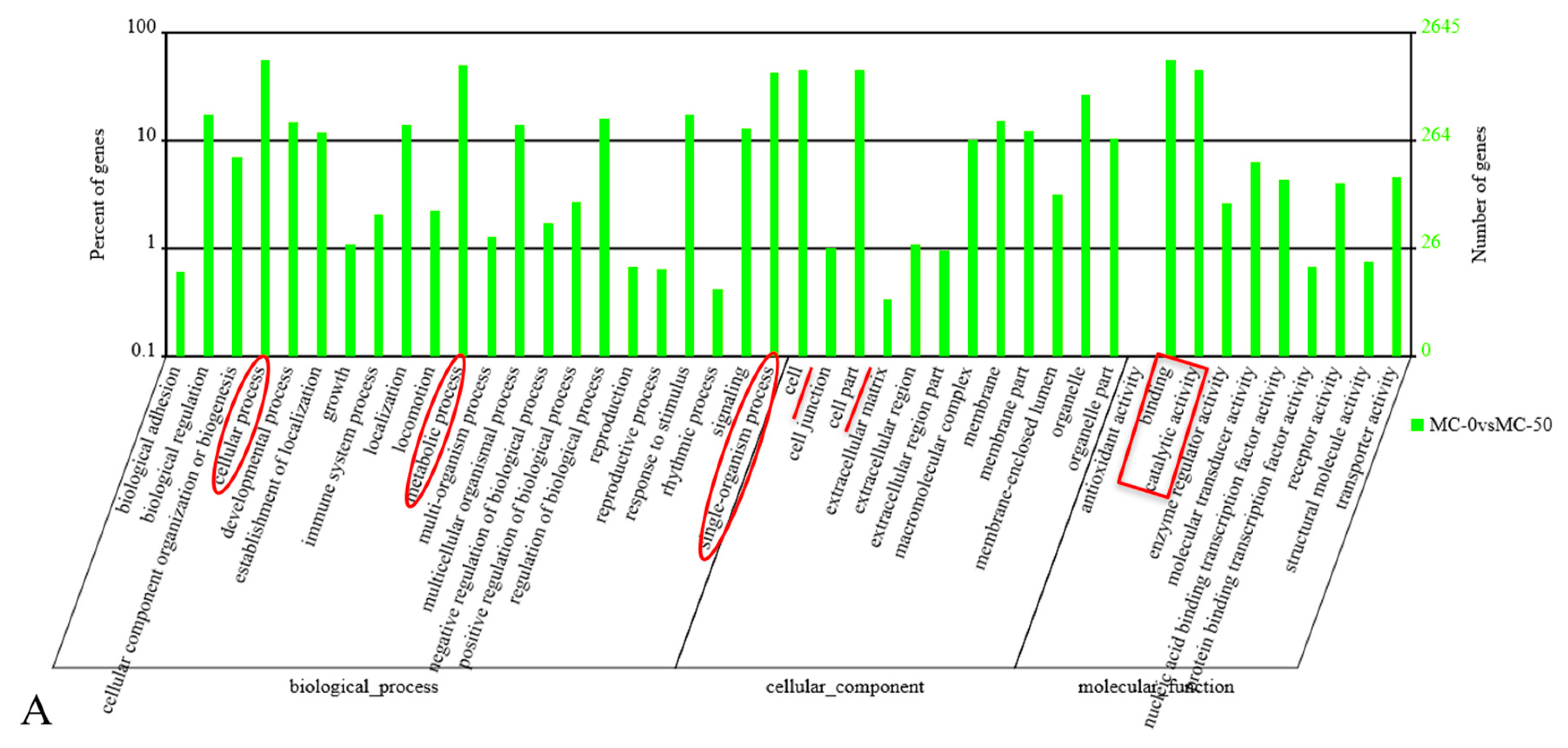
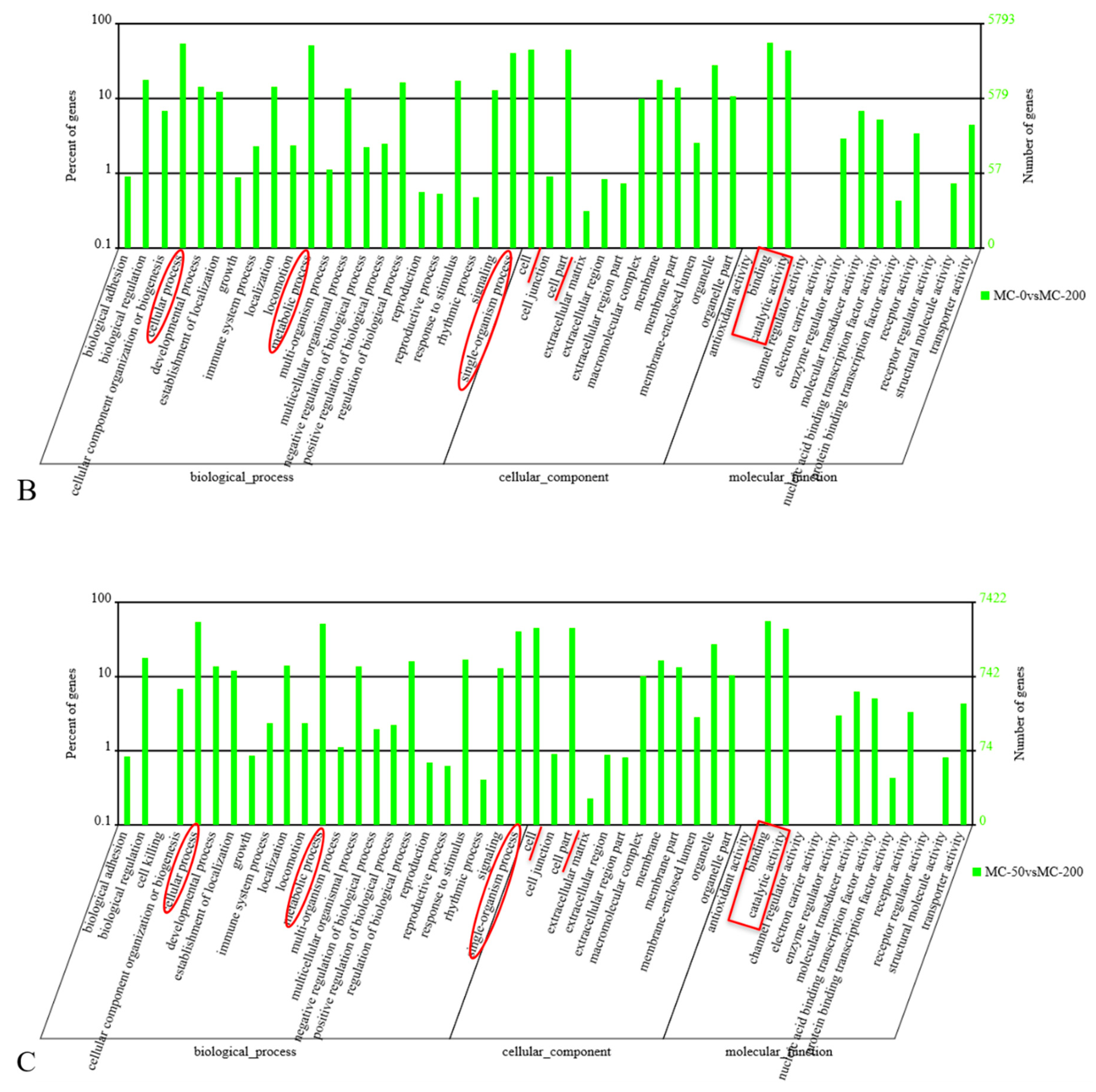
2.3. GO Analysis of the Candidate Target Genes of Differentially Expressed miRNAs
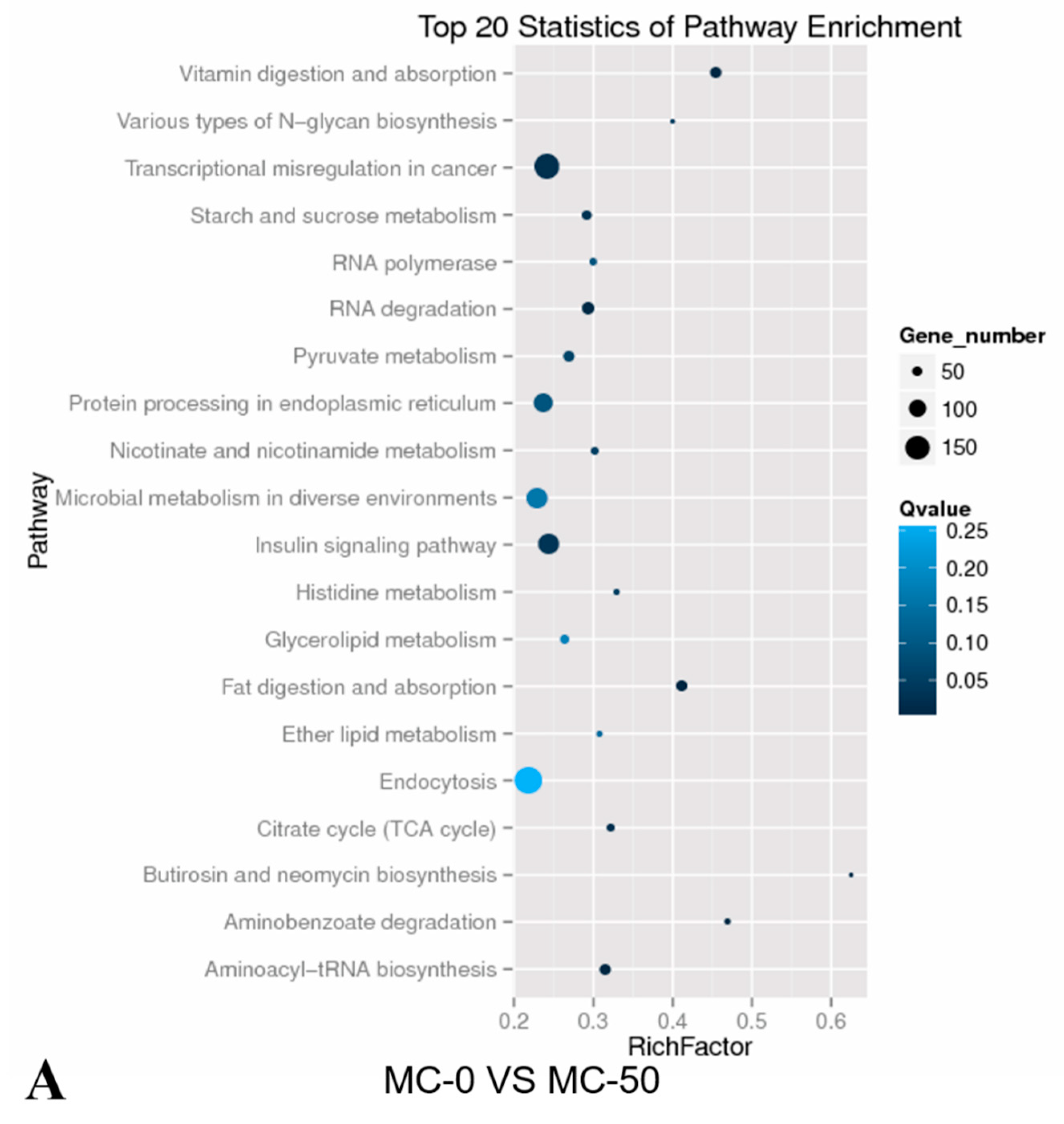
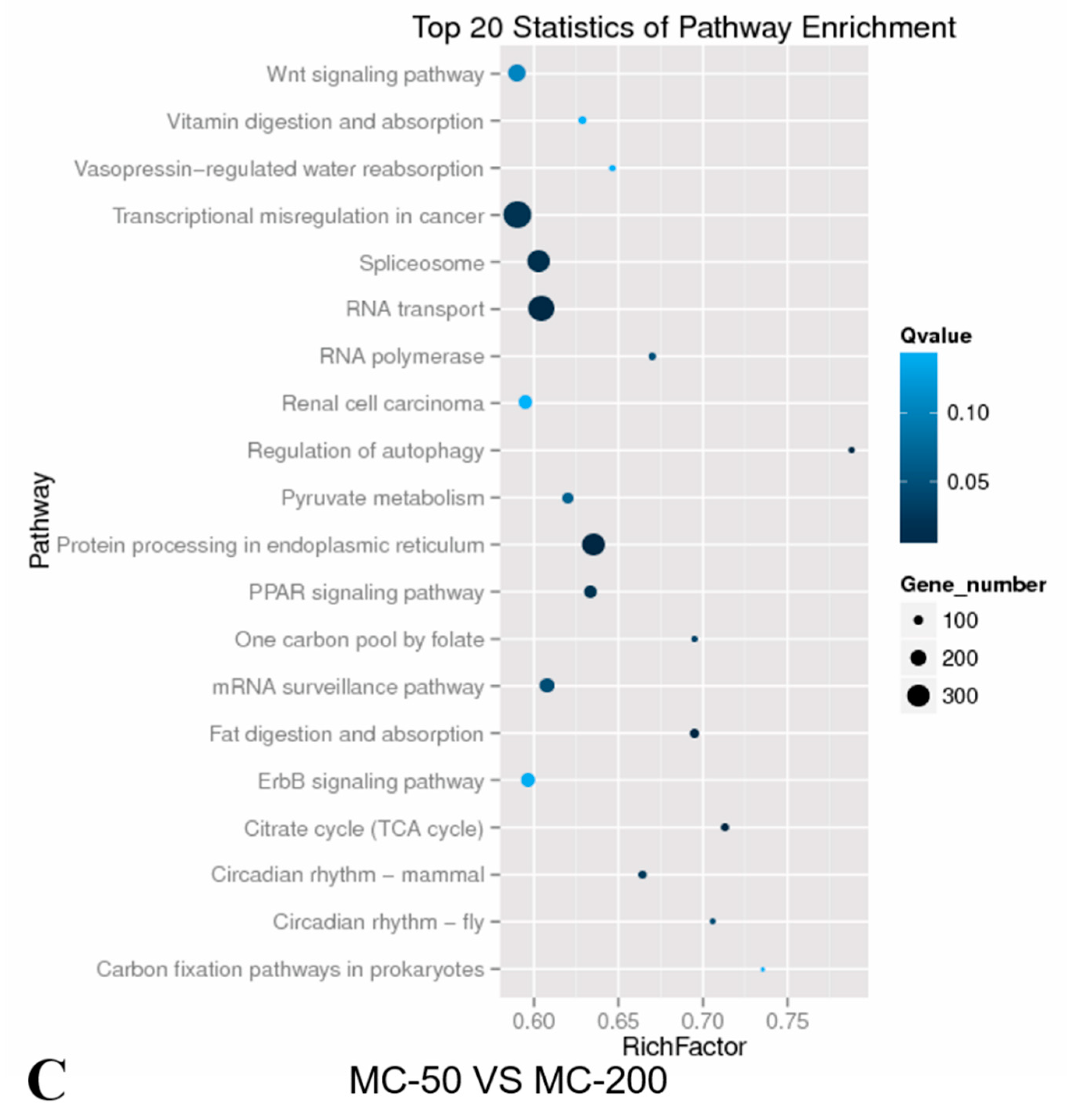
2.4. KEGG Pathway Analysis of the Candidate Target Genes of Differentially Expressed miRNAs
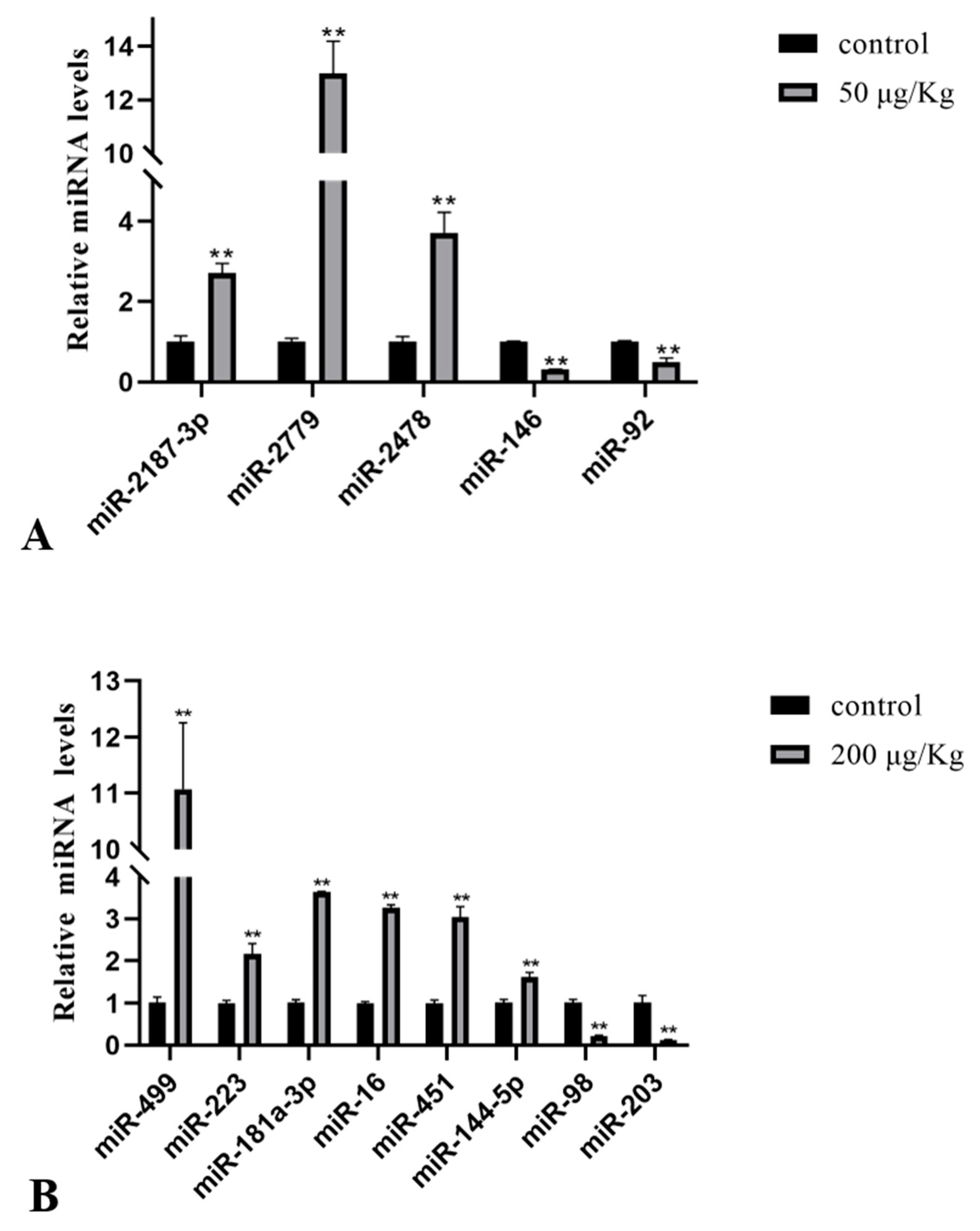
2.5. Confirmatory Study and Additional Profiling of the Selected miRNAs by qPCR
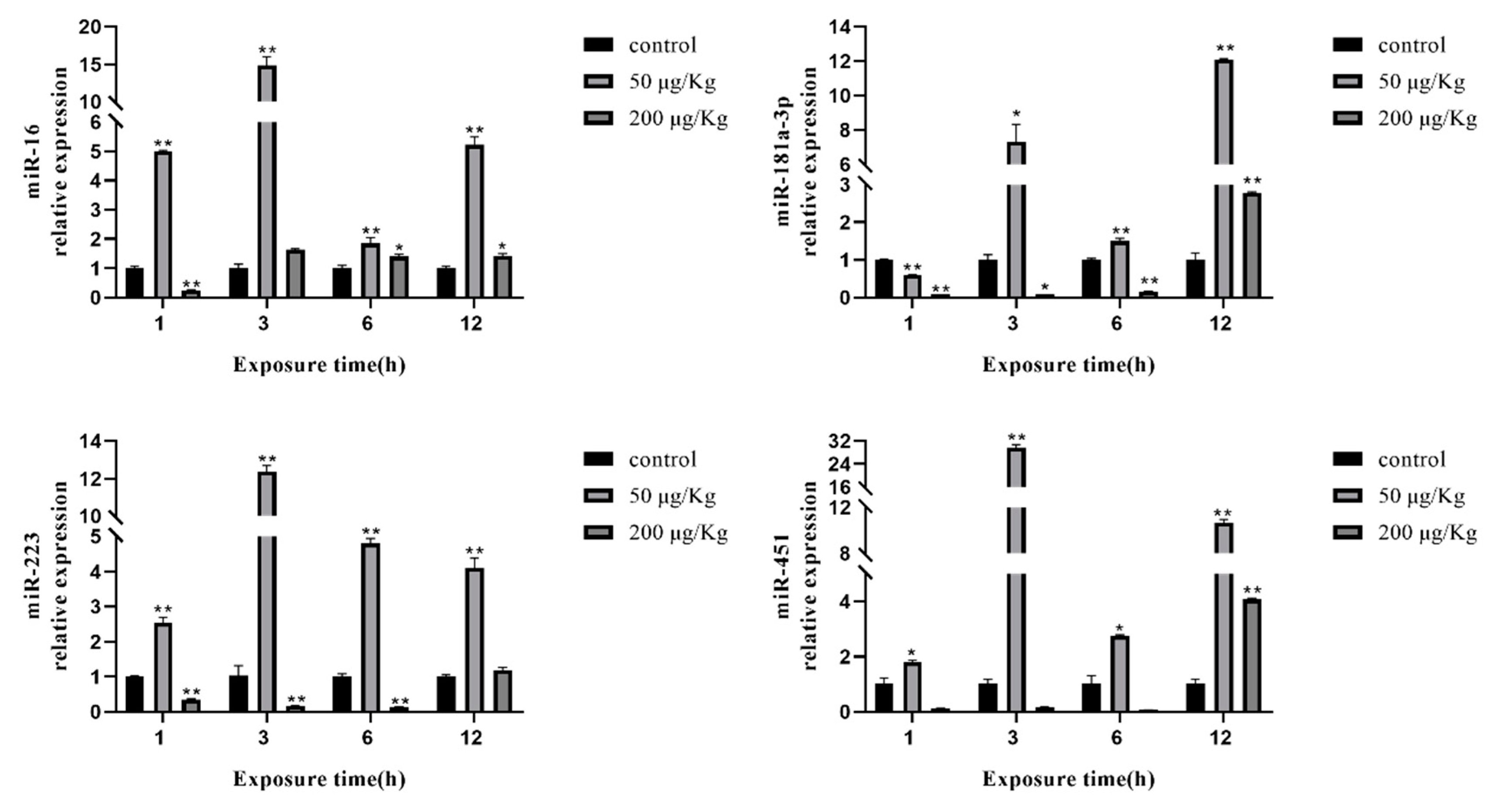
2.6. Expression of Selected miRNAs in the Liver of Silver Carp
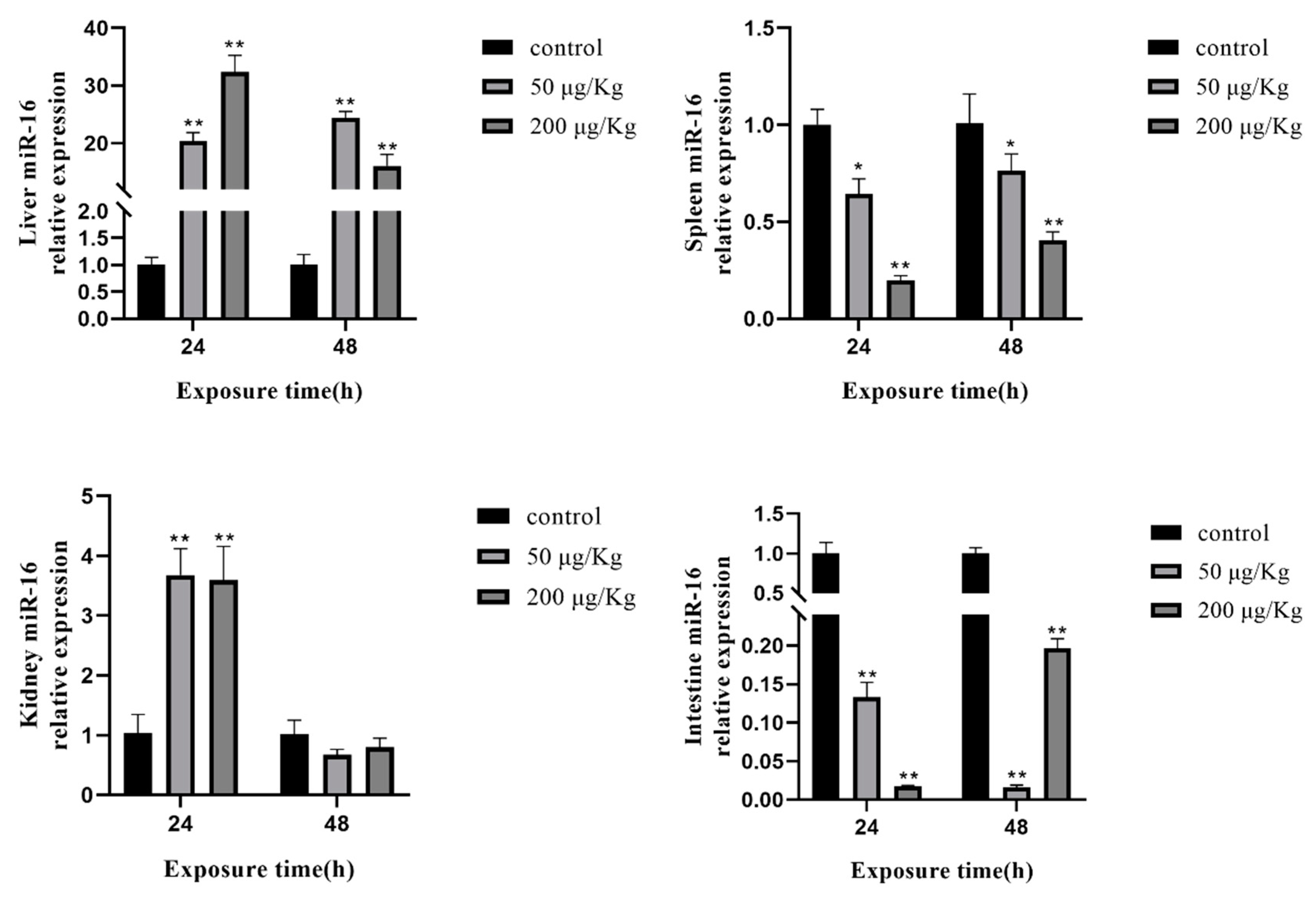
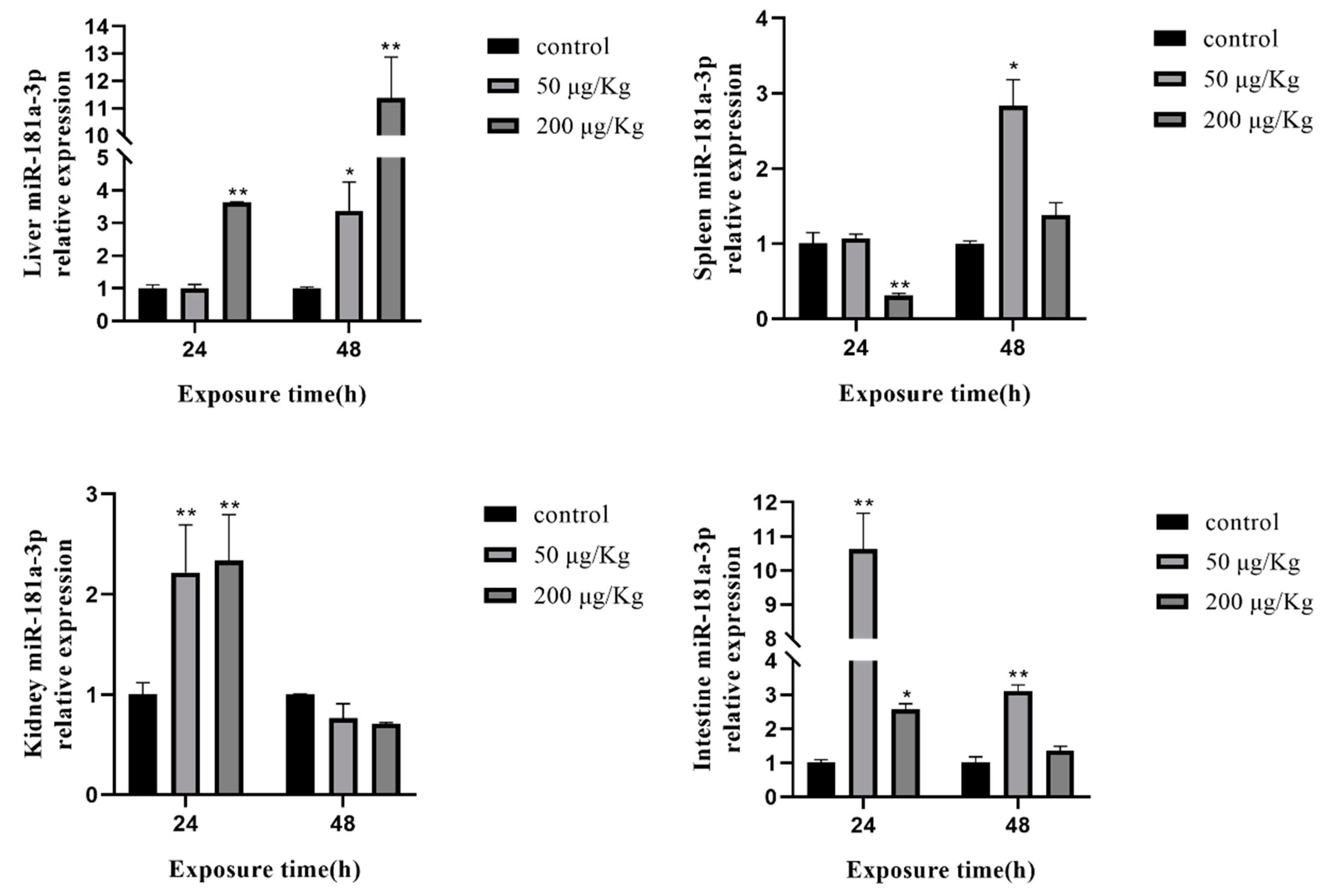
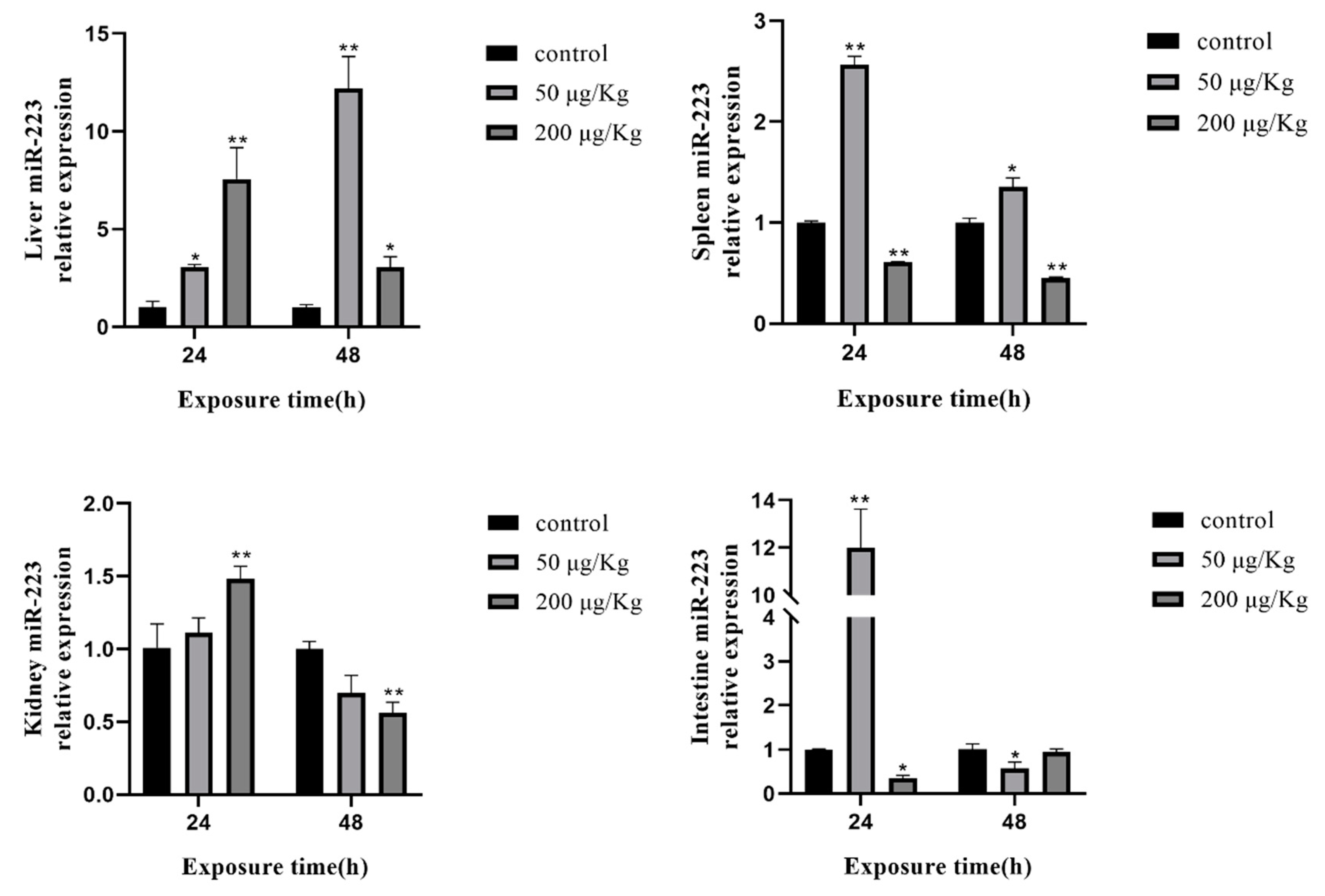
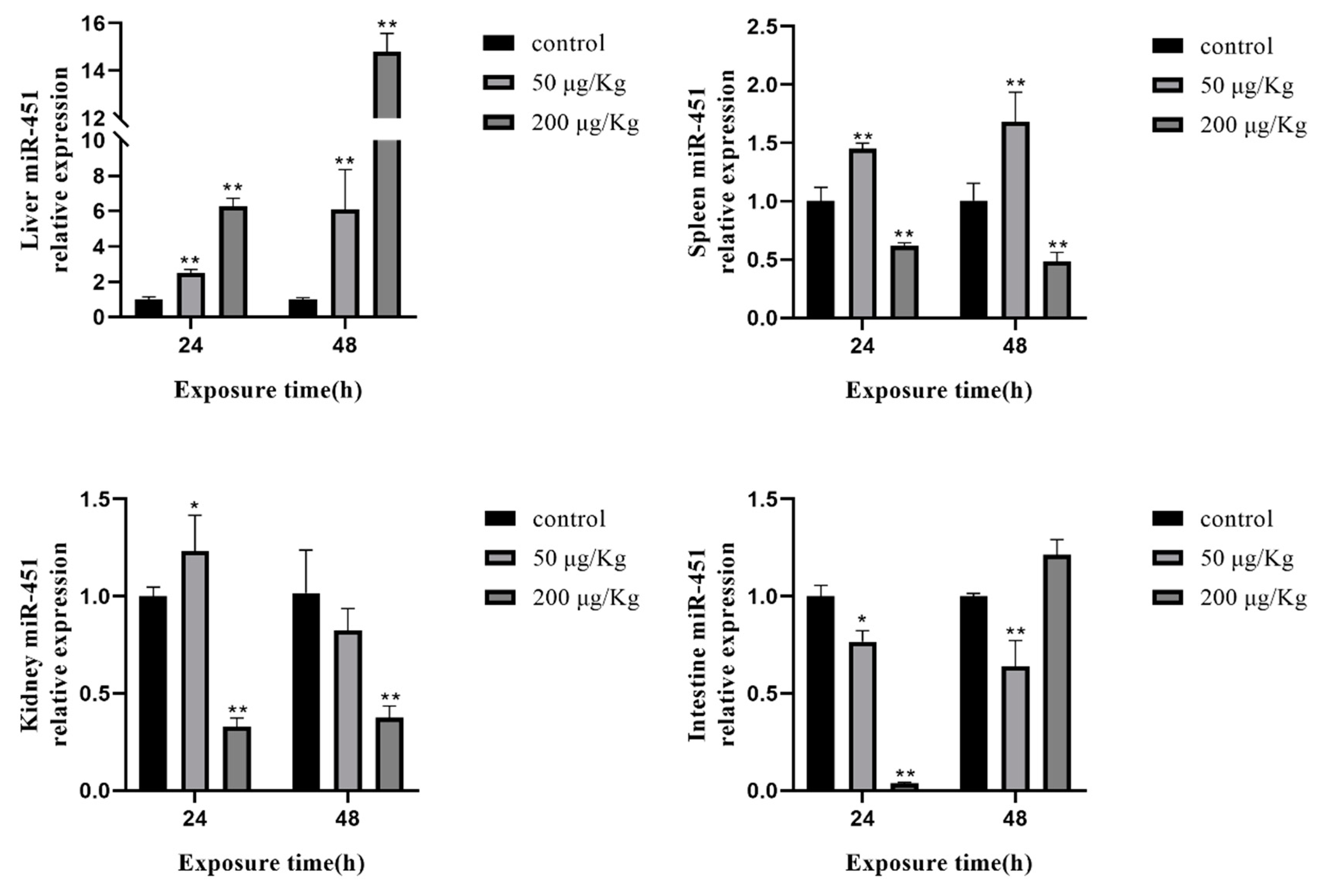
2.7. Expression of Selected miRNAs in Different Tissues of Silver Carp after Exposure to MC-LR
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Ethics Statement
5.2. Fish and Treatment
5.3. Tissue Collection and RNA Isolation
5.4. Small RNA Library Construction and Sequencing
5.5. Sequence Data Analysis
5.6. Differential miRNA Expression Analysis
5.7. miRNA Verification and Analysis by Quantitative Real-Time PCR
5.8. Known miRNA Target Gene Prediction and Function Analysis
5.9. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- DC Oliveira, E.; Castelo-Branco, R.; Silva, L.; Silva, N.; Azevedo, J.; Vasconcelos, V.; Faustino, S.; Cunha, A. First Detection of Microcystin-LR in the Amazon River at the Drinking Water Treatment Plant of the Municipality of Macapá, Brazil. Toxins 2019, 11, 669. [Google Scholar] [CrossRef]
- O’Neil, J.M.; Davis, T.W.; Burford, M.A.; Gobler, C.J. The rise of harmful cyanobacteria blooms: The potential roles of eutrophication and climate change. Harmful Algae 2012, 14, 313–334. [Google Scholar] [CrossRef]
- Hu, C.; Rzymski, P. Programmed Cell Death-Like and Accompanying Release of Microcystin in Freshwater Bloom-Forming Cyanobacterium Microcystis: From Identification to Ecological Relevance. Toxins 2019, 11, 706. [Google Scholar] [CrossRef]
- Díez-Quijada, L.; Prieto, A.I.; Guzmán-Guillén, R.; Jos, A.; Cameán Ana, M. Occurrence and toxicity of microcystin congeners other than Mc-Lr and Mc-Rr: A review. Food. Chem. Toxicol. 2019, 125, 106–132. [Google Scholar] [CrossRef]
- Paerl, H.W.; Otten, T.G. Harmful Cyanobacterial Blooms: Causes, Consequences, and Controls. Microb. Ecol. 2013, 65, 995–1010. [Google Scholar] [CrossRef]
- Chen, L.; Li, S.; Guo, X.; Xie, P.; Chen, J. The role of GSH in microcystin-induced apoptosis in rat liver: Involvement of oxidative stress and NF-κB. Environ. Toxicol. 2016, 31, 552–560. [Google Scholar] [CrossRef]
- Wörmer, L.; Huerta-Fontela, M.; Cirés, S.; Carrasco, D.; Quesada, A. Natural Photodegradation of the Cyanobacterial Toxins Microcystin and Cylindrospermopsin. Environ. Sci. Technol. 2010, 44, 3002–3007. [Google Scholar] [CrossRef] [PubMed]
- Su, X.; Steinman, A.D.; Xue, Q.; Zhao, Y.; Xie, L. Evaluating the contamination of microcystins in Lake Taihu, China: The application of equivalent total MC-LR concentration. Ecol. Indic. 2018, 89, 445–454. [Google Scholar] [CrossRef]
- Liu, J.; Wang, H.; Wang, B.; Chen, T.; Wang, X.; Huang, P.; Xu, L.; Guo, Z. Microcystin-LR promotes proliferation by activating Akt/S6K1 pathway and disordering apoptosis and cell cycle associated proteins phosphorylation in HL7702 cells. Toxicol. Lett. 2016, 240, 214–225. [Google Scholar] [CrossRef] [PubMed]
- Rinta-Kanto, J.M.; Konopko, E.A.; DeBruyn, J.M.; Bourbonniere, R.A.; Boyer, G.L.; Wilhelm, S.W. Lake Erie Microcystis: Relationship between microcystin production, dynamics of genotypes and environmental parameters in a large lake. Harmful Algae 2009, 8, 665–673. [Google Scholar] [CrossRef]
- Miller, M.A.; Kudela, R.M.; Mekebri, A.; Crane, D.; Oates, S.C.; Tinker, M.T.; Staedler, M.; Miller, W.A.; Toy-Choutka, S.; Dominik, C. Evidence for a Novel Marine Harmful Algal Bloom: Cyanotoxin (Microcystin) Transfer from Land to Sea Otters. PLoS ONE 2010, 5, e12576. [Google Scholar] [CrossRef]
- Rita, D.P.; Valeria, V.; Silvia, B.M.; Pasquale, G.; Milena, B. Microcystin Contamination in Sea Mussel Farms from the Italian Southern Adriatic Coast following Cyanobacterial Blooms in an Artificial Reservoir. J. Ecosyst. 2014, 2014, 1–11. [Google Scholar] [CrossRef]
- Chan, W.S.; Recknagel, F.; Cao, H.; Park, H.-D. Elucidation and short-term forecasting of microcystin concentrations in Lake Suwa (Japan) by means of artificial neural networks and evolutionary algorithms. Water Res. 2007, 41, 2247–2255. [Google Scholar] [CrossRef] [PubMed]
- Pouria, S.; de Andrade, A.; Barbosa, J.; Cavalcanti, R.; Barreto, V.; Ward, C.; Preiser, W.; Poon, G.K.; Neild, G.; Codd, G. Fatal microcystin intoxication in haemodialysis unit in Caruaru, Brazil. Lancet 1998, 352, 21–26. [Google Scholar] [CrossRef]
- Francis, G. Poisonous Australian Lake. Nature 1878, 18, 11–12. [Google Scholar] [CrossRef]
- Zhang, D.; Liao, Q.; Zhang, L.; Wang, D.; Luo, L.; Chen, Y.; Zhong, J.; Liu, J. Occurrence and spatial distributions of microcystins in Poyang Lake, the largest freshwater lake in China. Ecotoxicology 2015, 24, 19–28. [Google Scholar] [CrossRef]
- Yu, L.; Kong, F.; Zhang, M.; Yang, Z.; Shi, X.; Du, M. The Dynamics of Microcystis Genotypes and Microcystin Production and Associations with Environmental Factors during Blooms in Lake Chaohu, China. Toxins 2014, 6, 3238–3257. [Google Scholar] [CrossRef]
- Wu, Y.; Li, L.; Gan, N.; Zheng, L.; Ma, H.; Shan, K.; Liu, J.; Xiao, B.; Song, L. Seasonal dynamics of water bloom-forming Microcystis morphospecies and the associated extracellular microcystin concentrations in large, shallow, eutrophic Dianchi Lake. J. Environ. Sci. 2014, 26, 1921–1929. [Google Scholar] [CrossRef]
- Yu, G.; Jiang, Y.; Song, G.; Tan, W.; Zhu, M.; Li, R. Variation of Microcystis and microcystins coupling nitrogen and phosphorus nutrients in Lake Erhai, a drinking-water source in Southwest Plateau, China. Environ. Sci. Pollut. Res. 2014, 21, 9887–9898. [Google Scholar] [CrossRef]
- Zhou, L.; Yu, H.; Chen, K. Relationship between microcystin in drinking water and colorectal cancer. Biomed. Environ. Sci. 2002, 15, 166–171. [Google Scholar]
- IARC Working Group on the Evaluation of Carcinogenic Risks to Humans IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. Ingested nitrate and nitrite, and cyanobacterial peptide toxins. IARC Monogr. Eval. Carcinog. Risks Hum. 2010, 94, 448. [Google Scholar]
- Bhattacharya, M.; Ghosh, S.; Malick, R.C.; Patra, B.C.; Das, B.K. Therapeutic applications of zebrafish (Danio rerio) miRNAs linked with human diseases: A prospective review. Gene 2018, 679, 202–211. [Google Scholar] [CrossRef]
- MacFarlane, L.A.; RMurphy, P. MicroRNA: Biogenesis, Function and Role in Cancer. Curr. Genom. 2010, 11, 537–561. [Google Scholar] [CrossRef]
- Ambros, V. The functions of animal microRNAs. Nature 2004, 431, 350–355. [Google Scholar] [CrossRef]
- Miska, E.A. How microRNAs control cell division, differentiation and death. Curr. Opin. Genet. Dev. 2005, 15, 563–568. [Google Scholar] [CrossRef]
- Kim, Y.-K.; Yu, J.; Han, T.S.; Park, S.-Y.; Namkoong, B.; Kim, D.H.; Hur, K.; Yoo, M.-W.; Lee, H.-J.; Yang, H.-K. Functional links between clustered microRNAs: Suppression of cell-cycle inhibitors by microRNA clusters in gastric cancer. Nucleic Acids Res. 2009, 37, 1672–1681. [Google Scholar] [CrossRef]
- Esquela-Kerscher, A.; Slack, F.J. Oncomirs—microRNAs with a role in cancer. Nat. Rev. Cancer 2006, 6, 259–269. [Google Scholar] [CrossRef]
- Calin, G.A.; Croce, C.M. MicroRNA signatures in human cancers. Nat. Rev. Cancer 2006, 6, 857–866. [Google Scholar] [CrossRef]
- Xu, L.; Qin, W.; Zhang, H.; Wang, Y.; Dou, H.; Ding, Y.; Yang, L.; Wang, Y. Alterations in microrna expression linked to microcystin-lr-induced tumorigenicity in human WRL-68 cells. Mutat. Res. Fund. Mol. M. 2012, 743, 75–82. [Google Scholar] [CrossRef]
- Chen, L.; Yang, S.; Wen, C.; Zheng, S.; Yang, Y.; Feng, X.; Yang, F. Regulation of Microcystin-LR-Induced DNA Damage by miR-451a in HL7702 Cells. Toxins 2019, 11, 164. [Google Scholar] [CrossRef]
- Li, X.; Zhuang, X.; Xu, T.; Mao, M.; Wang, C.; Chen, Y.; Han, X.; Wu, J. Expression analysis of microRNAs and mRNAs in ovarian granulosa cells after microcystin-LR exposure. Toxicon 2017, 129, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Qu, X.; Hu, M.; Shang, Y.; Pan, L.; Jia, P.; Fu, C.; Liu, Q.; Wang, Y. Liver Transcriptome and miRNA Analysis of Silver Carp (Hypophthalmichthys molitrix) Intraperitoneally Injected with Microcystin-LR. Front. Physiol. 2018, 9, 381. [Google Scholar] [CrossRef] [PubMed]
- Carmichael, W.W.; Azevedo, S.M.F.O.; An, J.S.; Molica, R.J.R.; Jochimsen, E.M.; Lau, S.; Rinehart, K.L.; Shaw, G.R.; Eaglesham, G.K. Human fatalities from cyanobacteria: Chemical and biological evidence for cyanotoxins. Environ. Health Perspect. 2001, 109, 663–668. [Google Scholar] [CrossRef]
- Yang, S.; Chen, L.; Wen, C.; Zhang, X.; Feng, X.; Yang, F. MicroRNA expression profiling involved in MC-LR-induced hepatotoxicity using high-throughput sequencing analysis. J. Toxicol. Environ. Health Part A Curr. Issues 2018, 81, 89–97. [Google Scholar] [CrossRef]
- Ma, J.; Li, Y.; Yao, L.; Li, X. Analysis of microRNA expression profiling involved in MC-LR-induced cytotoxicity by high-throughput sequencing. Toxins 2017, 9, 23. [Google Scholar] [CrossRef]
- Eroles, P.; Tormo, E.; Pineda, B.; Espin, E.; Lluch, A. MicroRNAs in breast cancer: One More Turn in Regulation. Curr. Drug Targets 2015, 17, 1083–1100. [Google Scholar]
- Hata, A.; Lieberman, J. Dysregulation of microRNA biogenesis and gene silencing in cancer. Sci. Signal. 2015, 8, re3. [Google Scholar] [CrossRef]
- Brzuzan, P.; Woźny, M.; Wolińska, L.; Piasecka, A. Expression profiling in vivo demonstrates rapid changes in liver microRNA levels of whitefish (Coregonus lavaretus) following microcystin-LR exposure. Aquat. Toxicol. 2012, 122, 188–196. [Google Scholar] [CrossRef]
- Florczyk, M.; Brzuzan, P.; Krom, J.; Woźny, M.; Łakomiak, A. miR-122-5p as a plasma biomarker of liver injury in fish exposed to microcystin-LR. J. Fish Dis. 2016, 39, 741–751. [Google Scholar] [CrossRef]
- Łakomiak, A.; Brzuzan, P.; Jakimiuk, E.; Florczyk, M.; Woźny, M. miR-34a and bcl-2 expression in whitefish (Coregonus lavaretus) after microcystin-LR exposure. Comp. Biochem. Physiol. Part B Biochem. Mol. Biol. 2016, 193, 47–56. [Google Scholar] [CrossRef]
- Li, G.; Zhao, Y.; Wen, L.; Liu, Z.; Yan, F.; Gao, C. Identification and Characterization of MicroRNAs in the Spleen of Common Carp Immune Organ. J. Cell. Biochem. 2014, 115, 1768–1778. [Google Scholar] [CrossRef]
- White, M.F. Insulin Signaling in Health and Disease. Science 2003, 302, 1710–1711. [Google Scholar] [CrossRef]
- Sha, H.; Gu, Y.; Shen, W.; Zhang, L.; Qian, F.; Zhao, Y.; Li, H.; Zhang, T.; Lu, W. Rheinic acid ameliorates radiation-induced acute enteritis in rats through PPAR-γ/NF-κB. Genes Genom. 2019, 41, 909–917. [Google Scholar] [CrossRef]
- Li, X.; Peng, B.; Zhu, X.; Wang, P.; Sun, K.; Lei, X.; He, H.; Tian, Y.; Mo, S.; Zhang, R.; et al. MiR-210-3p inhibits osteogenic differentiation and promotes adipogenic differentiation correlated with Wnt signaling in ERα-deficient rBMSCs. J. Cell. Physiol. 2019, 234, 23475–23484. [Google Scholar] [CrossRef]
- Wang, X.; Wang, X.; Huang, Y.; Chen, X.; L., M.; Shi, L.; Li, C. Role and mechanisms of action of microRNA-21 as regards the regulation of the WNT/β-catenin signaling pathway in the pathogenesis of non-alcoholic fatty liver disease. Int. J. Mol. Med. 2019, 44, 2201–2212. [Google Scholar] [CrossRef]
- Calin, G.A.; Dumitru, C.D.; Shimizu, M.; Bichi, R.; Zupo, S.; Noch, E.; Aldler, H.; Rattan, S.; Keating, M.; Rai, K.; et al. Nonlinear partial differential equations and applications: Frequent deletions and down-regulation of micro-RNA genes miR15 and miR16 at 13q14 in chronic lymphocytic leukemia. Proc. Natl. Acad. Sci. USA 2002, 99, 15524–15529. [Google Scholar] [CrossRef]
- Rivas, M.A.; Venturutti, L.; Huang, Y.-W.; Schillaci, R.; Huang, T.H.-M.; Elizalde, P.V. Downregulation of the tumor-suppressor miR-16 via progestin-mediated oncogenic signaling contributes to breast cancer development. Breast Cancer Res. 2012, 14, R77. [Google Scholar] [CrossRef]
- Chen, T.M.; Xiao, Q.; Wang, X.J.; Wang, Z.Q.; Hu, J.W.; Zhang, Z.; Gong, Z.N.; Chen, S.L. miR-16 regulates proliferation and invasion of lung cancer cells via the ERK/MAPK signaling pathway by targeted inhibition of MAPK kinase 1 (MEK1). J. Int. Med. Res. 2019, 47, 5194–5204. [Google Scholar] [CrossRef]
- Bellon, M.; Lepelletier, Y.; Hermine, O.; Nicot, C. Deregulation of microRNA involved in hematopoiesis and the immune response in HTLV-I adult T-cell leukemia. Blood 2009, 113, 4914–4917. [Google Scholar] [CrossRef]
- Xie, W.; Li, M.; Xu, N.; Lv, Q.; Huang, N.; He, J.; Zhang, Y. miR-181a Regulates Inflammation Responses in Monocytes and Macrophages. PLoS ONE 2013, 8, e58639. [Google Scholar] [CrossRef]
- Yamada, K.; Takizawa, S.; Ohgaku, Y.; Asami, T.; Furuya, K.; Yamamoto, K.; Takahashi, F.; Hamajima, C.; Inaba, C.; Endo, K.; et al. MicroRNA 16-5p is upregulated in calorie-restricted mice and modulates inflammatory cytokines of macrophages. Gene 2020, 725, 144191. [Google Scholar] [CrossRef]
- Lei, J.; Fu, Y.; Zhuang, Y.; Zhang, K.; Lu, D. LncRNA SNHG1 alleviates IL-1β-induced osteoarthritis by inhibiting MIR-16-5p-mediated p38 MAPK and NF-κB signaling pathways. Biosci. Rep. 2019, 39, 1–10. [Google Scholar] [CrossRef]
- Wei, Y.-T.; Guo, D.-W.; Hou, X.-Z.; Jiang, D.-Q. miRNA-223 suppresses FOXO1 and functions as a potential tumor marker in breast cancer. Cell. Mol. Biol. 2017, 63, 113. [Google Scholar] [CrossRef]
- Pulikkan, J.A.; Dengler, V.; Peramangalam, P.S.; Peer Zada, A.A.; Müller-Tidow, C.; Bohlander, S.K.; Tenen, D.G.; Behre, G. Cell-cycle regulator E2F1 and microRNA-223 comprise an autoregulatory negative feedback loop in acute myeloid leukemia. Blood 2010, 115, 1768–1778. [Google Scholar] [CrossRef]
- Dong, Y.W.; Wang, R.; Cai, Q.Q.; Qi, B.; Wu, W.; Zhang, Y.H.; Wu, X.Z. Sulfatide epigenetically regulates miR-223 and promotes the migration of human hepatocellular carcinoma cells. J. Hepatol. 2014, 60, 792–801. [Google Scholar] [CrossRef]
- Wong, Q.W.-L.; Lung, R.W.M.; Law, P.T.Y.; Lai, P.B.S.; Chan, K.Y.Y.; To, K.; Wong, N. MicroRNA-223 Is Commonly Repressed in Hepatocellular Carcinoma and Potentiates Expression of Stathmin. Gastroenterology 2008, 135, 257–269. [Google Scholar] [CrossRef]
- Khordadmehr, M.; Jigari-Asl, F.; Ezzati, H.; Shahbazi, R.; Sadreddini, S.; Safaei, S.; Baradaran, B. A comprehensive review on miR-451: A promising cancer biomarker with therapeutic potential. J. Cell. Physiol. 2019, 234, 21716–21731. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, A.; Xiang, J.; Lv, Y.; Zhang, X. miR-451 acts as a suppressor of angiogenesis in hepatocellular carcinoma by targeting the IL-6R-STAT3 pathway. Oncol. Rep. 2016, 36, 1385–1392. [Google Scholar] [CrossRef]
- Mamoori, A.; Gopalan, V.; Smith, R.A.; Lam, A.K.-Y. Modulatory roles of microRNAs in the regulation of different signalling pathways in large bowel cancer stem cells. Biol. Cell. 2016, 108, 51–64. [Google Scholar] [CrossRef]
- Bitarte, N.; Bandres, E.; Boni, V.; Zarate, R.; Rodriguez, J.; Gonzalez-Huarriz, M.; Lopez, I.; Javier Sola, J.; Alonso, M.M.; Fortes, P.; et al. MicroRNA-451 Is Involved in the Self-renewal, Tumorigenicity, and Chemoresistance of Colorectal Cancer Stem Cells. Stem Cells 2011, 29, 1661–1671. [Google Scholar] [CrossRef]
- Woźny, M.; Łuczyński, M.; Wolińska, L.; Brzuzan, P.; Piasecka, A. MicroRNA expression in liver of whitefish (Coregonus lavaretus) exposed to microcystin-LR. Environ. Biotechnol. 2010, 6, 53–60. [Google Scholar]
- Svirčev, Z.; Baltić, V.; Gantar, M.; Juković, M.; Stojanović, D.; Baltić, M. Molecular aspects of microcystin-induced hepatotoxicity and hepatocarcinogenesis. J. Environ. Sci. Health Part C Environ. Carcinog. Ecotoxicol. Rev. 2010, 28, 39–59. [Google Scholar] [CrossRef]
- Wang, M.; Wang, D.; Lin, L.; Hong, H. Protein profiles in zebrafish (Danio rerio) brains exposed to chronic microcystin-LR. Chemosphere 2010, 81, 716–724. [Google Scholar] [CrossRef]
- Chen, Y.; Xu, J.; Li, Y.; Han, X. Decline of sperm quality and testicular function in male mice during chronic low-dose exposure to microcystin-LR. Reprod. Toxicol. 2011, 31, 551–557. [Google Scholar] [CrossRef]
- Li, Y.; Sheng, J.; Sha, J.; Han, X. The toxic effects of microcystin-LR on the reproductive system of male rats in vivo and in vitro. Reprod. Toxicol. 2008, 26, 239–245. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Wang, X.; Geng, Z.; Zhou, Y.; Chen, Y.; Wu, J.; Han, X. Distribution of microcystin-LR to testis of male Sprague–Dawley rats. Ecotoxicology 2013, 22, 1555–1563. [Google Scholar] [CrossRef]
- Feng, Y.; Ma, J.; Xiang, R.; Li, X. Alterations in microRNA expression in the tissues of silver carp (Hypophthalmichthys molitrix) following microcystin-LR exposure. Toxicon 2017, 128, 15–22. [Google Scholar] [CrossRef]
- Yi, S.; Gao, Z.-X.; Zhao, H.; Zeng, C.; Luo, W.; Chen, B.; Wang, W.-M. Identification and characterization of microRNAs involved in growth of blunt snout bream (Megalobrama amblycephala) by Solexa sequencing. BMC Genom. 2013, 14, 754. [Google Scholar] [CrossRef] [PubMed]
- Lewis, B.P.; Shih, I.; Jones-Rhoades, M.W.; Bartel, D.P.; Burge, C.B. Prediction of Mammalian MicroRNA Targets. Cell 2003, 115, 787–798. [Google Scholar] [CrossRef]
- Enright, A.J.; John, B.; Gaul, U.; Tuschl, T.; Sander, C.; Marks, D.S. MicroRNA targets in Drosophila. Genome Biol. 2003, 5, R1. [Google Scholar] [CrossRef] [PubMed]

| Gene | Forward Primer (5′–3′) | Reverse Primer (5′–3′) |
|---|---|---|
| U6 | GCTTCGGCAGCACATATACTAA | GCTTCACGAATTTGCGTGTCAT |
| miR-2779 | ATCCGGCTCGAAGGACTT | |
| miR-2478 | GGTCCCACTTCTGACACCAT | |
| miR-2187-3p | GGGCAGGCTATGCTAATCTATG | |
| miR-146 | GGGTGAGAACTGAATTCCATAG | |
| miR-92 | ATATTGCACTCGTCCCGGC | |
| miR-499 | GACTTGCAGTGATGTTTAGAG | |
| miR-203 | GTTTAGGACCACTTGATCAGGG | |
| miR-451 | GGGCCGTTACCATTACTGAGT | |
| miR-181a-3p | ACCATCGACCGTTGATTGTACC | |
| miR-223 | GGTCAGTTTGTCAAATACCCCA | |
| miR-144-5p | GGGACAGGATATCATCGTATACTG | |
| miR-16 miR-98 | GGCTAGCAGCACGTAAATATTGG GTTGGGGTGAGGTAGTAAGTTGT |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Feng, Y.; Chen, X.; Ma, J.; Zhang, B.; Li, X. Aberrant Expressional Profiling of Known MicroRNAs in the Liver of Silver Carp (Hypophthalmichthys molitrix) Following Microcystin-LR Exposure Based on samllRNA Sequencing. Toxins 2020, 12, 41. https://doi.org/10.3390/toxins12010041
Feng Y, Chen X, Ma J, Zhang B, Li X. Aberrant Expressional Profiling of Known MicroRNAs in the Liver of Silver Carp (Hypophthalmichthys molitrix) Following Microcystin-LR Exposure Based on samllRNA Sequencing. Toxins. 2020; 12(1):41. https://doi.org/10.3390/toxins12010041
Chicago/Turabian StyleFeng, Yiyi, Xi Chen, Junguo Ma, Bangjun Zhang, and Xiaoyu Li. 2020. "Aberrant Expressional Profiling of Known MicroRNAs in the Liver of Silver Carp (Hypophthalmichthys molitrix) Following Microcystin-LR Exposure Based on samllRNA Sequencing" Toxins 12, no. 1: 41. https://doi.org/10.3390/toxins12010041
APA StyleFeng, Y., Chen, X., Ma, J., Zhang, B., & Li, X. (2020). Aberrant Expressional Profiling of Known MicroRNAs in the Liver of Silver Carp (Hypophthalmichthys molitrix) Following Microcystin-LR Exposure Based on samllRNA Sequencing. Toxins, 12(1), 41. https://doi.org/10.3390/toxins12010041




