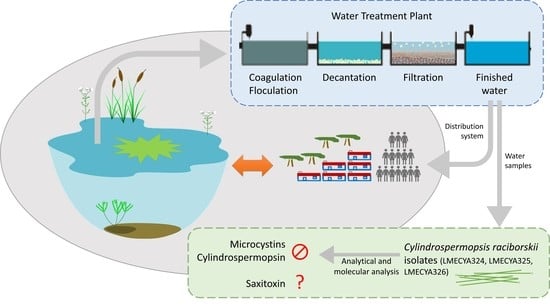
Isolation and Characterization of Cylindrospermopsis raciborskii Strains from Finished Drinking Water
Abstract
1. Introduction
2. Results
2.1. Phytoplankton Composition of Treated Water Samples
2.2. Isolation of Viable Cyanobacteria from Treated Water Samples
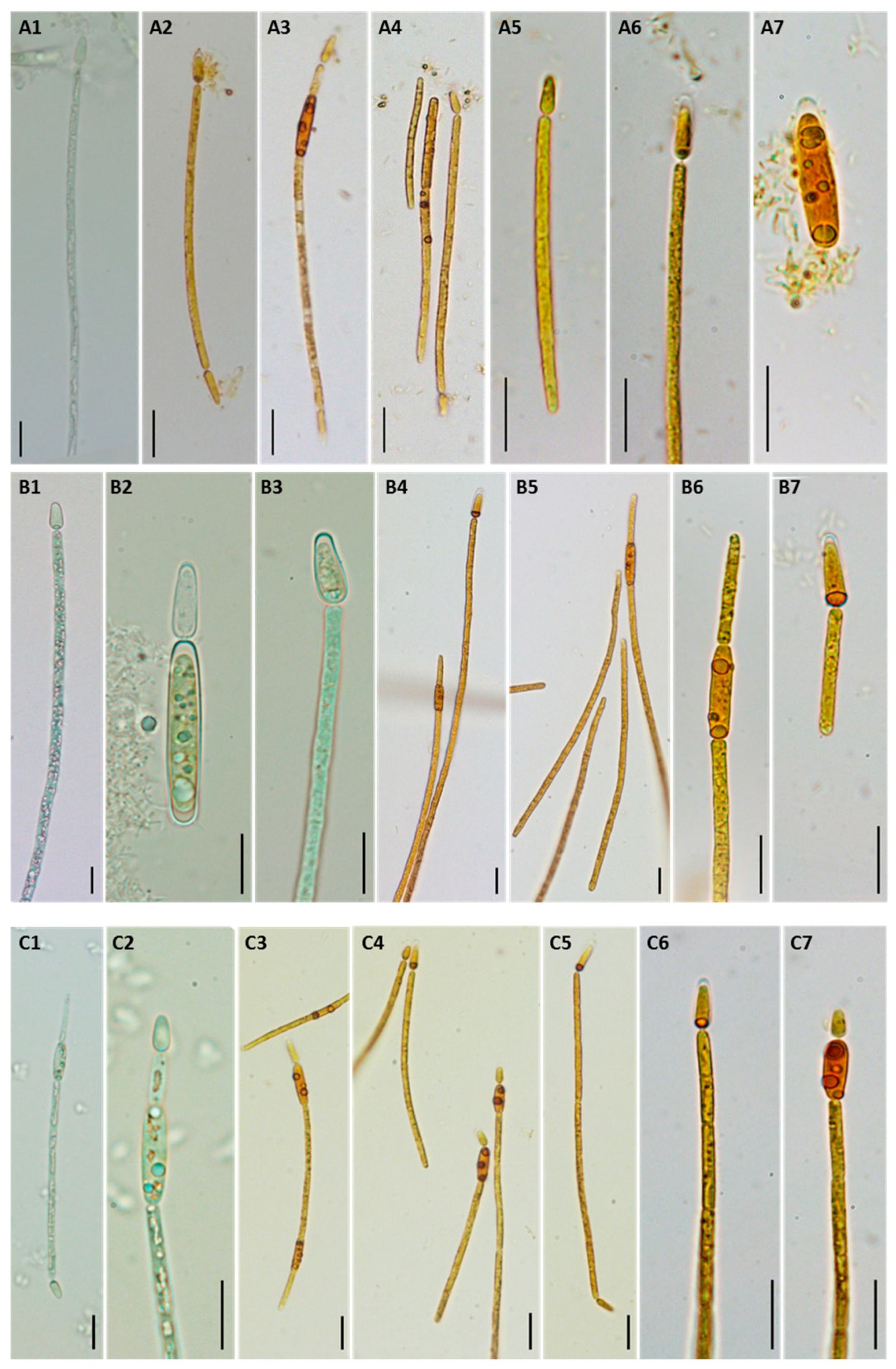
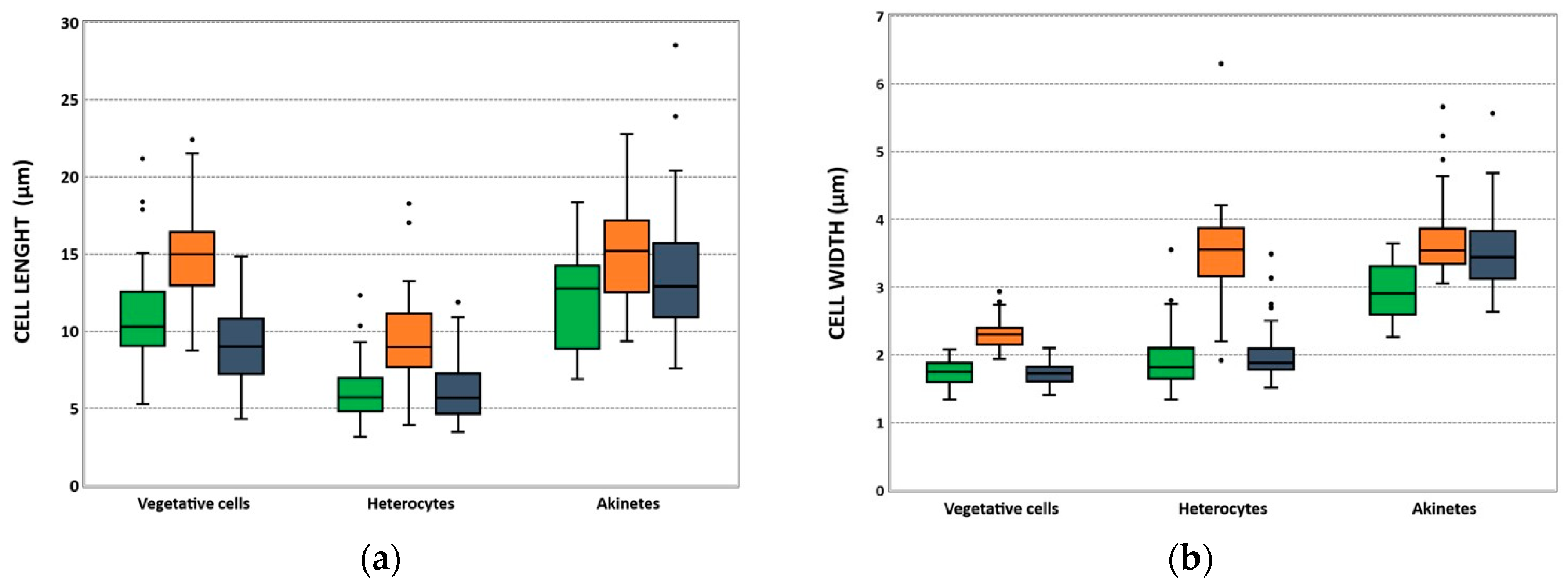
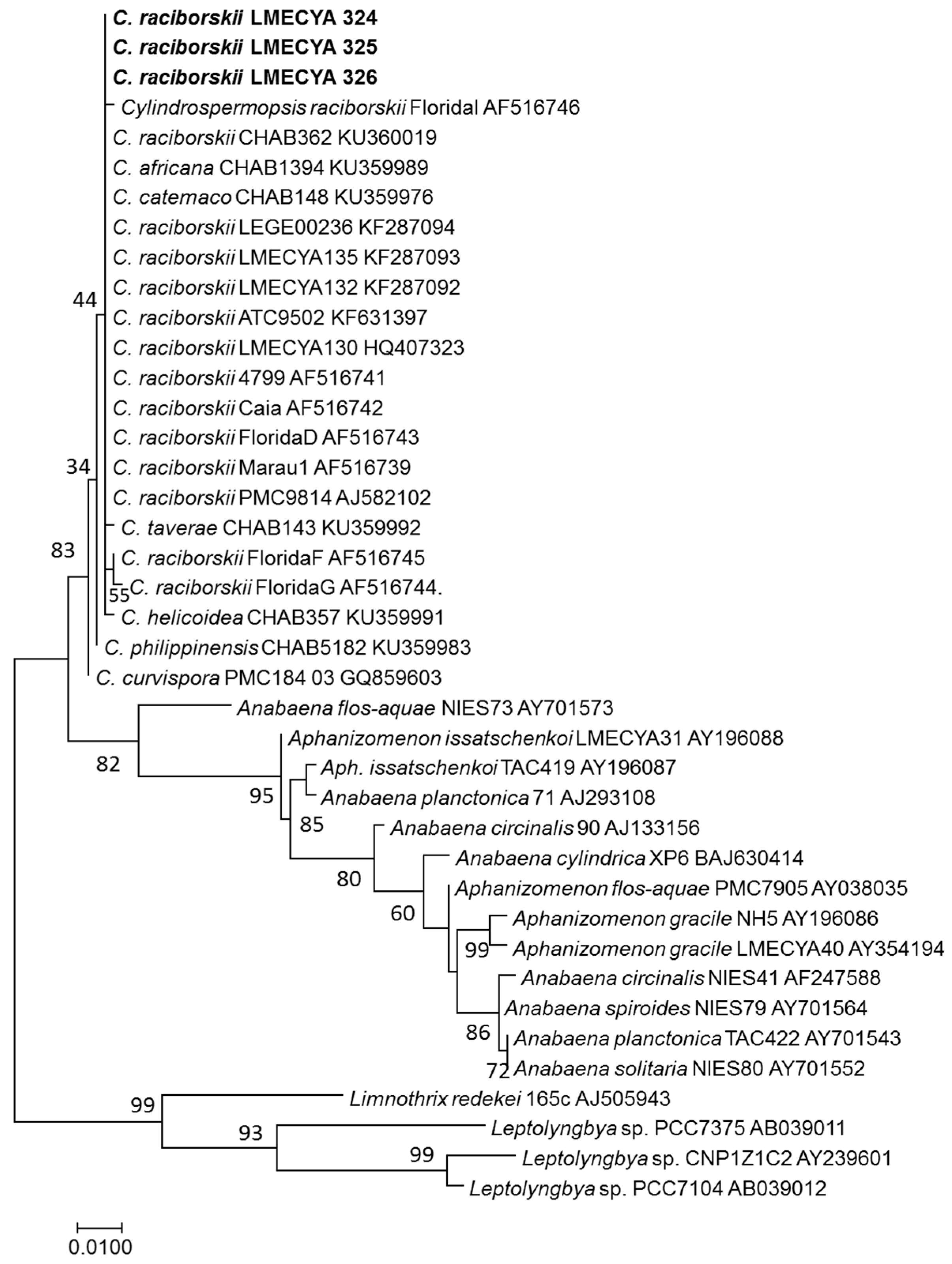
2.3. Morphological and Molecular Characterization of C. raciborskii Isolated from Treated Water Samples
2.4. Evaluation of C. raciborskii Toxicity
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Water Sampling and Phytoplankton Analysis
5.2. Isolation of Cyanobacteria from Treated Water Samples
5.3. Morphological Characterization of C. raciborskii Isolates
5.4. Molecular Identification of C. raciborskii Isolates by PCR Amplification and Sequencing of 16S rDNA Gene
5.5. Quantification of Cyanotoxins in C. raciborskii Cultures
5.6. Saxitoxin Derivatives Analysis by LC-FLD
5.7. PCR Amplification of Genes Involved in Microcystin, Cylindrospermopsin, and Saxitoxin Production
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Nunes, L.J.R.; Meireles, C.I.R.; Pinto Gomes, C.J.; Almeida Ribeiro, N.M.C. The Evolution of Climate Changes in Portugal: Determination of Trend Series and Its Impact on Forest Development. Climate 2019, 7, 78. [Google Scholar] [CrossRef]
- Carvalho, A.; Schmidt, L.; Santos, F.D.; Delicado, A. Climate change research and policy in Portugal: Climate change research and policy in Portugal. Wiley Interdiscip. Rev. Clim. Chang. 2014, 5, 199–217. [Google Scholar] [CrossRef]
- Menezes, C.; Churro, C.; Dias, E. Risk Levels of Toxic Cyanobacteria in Portuguese Recreational Freshwaters. Toxins 2017, 9, 327. [Google Scholar] [CrossRef] [PubMed]
- Ramos, S. Previsão da Ocorrência de Blooms de Cianobactérias na Albufeira do Roxo. Master’s Thesis, Faculty of Pharmacy of the University of Lisbon, Lisbon, Portugal, 2017. [Google Scholar]
- Gurung, R.P. Modelling of Eutrophication in Roxo Reservoir, Alentejo, Portugal—A System Dynamic Based Approach. Master Thesis, International Institute for Geo-Information Science and Earth Observation, Enschede, The Netherlands, 2007. [Google Scholar]
- Dias, E.; Menezes, C. Distribuição e Dinâmica de Ocorrências de Cianobactérias e Compostos Associados na Albufeira do Roxo; Instituto Nacional de Saúde Doutor Ricardo Jorge, IP: Lisbon, Portugal, 2019; pp. 1–58. Available online: http://hdl.handle.net/10400.18/6345 (accessed on 26 November 2019).
- Bellém, F.; Nunes, S.; Morais, M. Cyanobacteria Toxicity: Potential Public Health Impact in South Portugal Populations. J. Toxicol. Environ. Health A 2013, 76, 263–271. [Google Scholar] [CrossRef]
- Martins, O.; (Águas Públicas do Alentejo, Beja, Portugal). Personal communication, 2016.
- Ibelings, B.W.; Backer, L.C.; Kardinaal, W.E.A.; Chorus, I. Current approaches to cyanotoxin risk assessment and risk management around the globe. Harmful Algae 2015, 49, 63–74. [Google Scholar] [CrossRef]
- Antunes, J.T.; Leão, P.N.; Vasconcelos, V.M. Cylindrospermopsis raciborskii: Review of the distribution, phylogeography, and ecophysiology of a global invasive species. Front. Microbiol. 2015, 6, 473. [Google Scholar] [CrossRef]
- Tonetta, D.; Hennemann, M.C.; Brentano, D.M.; Petrucio, M.M. Considerations regarding the dominance of Cylindrospermopsis raciborskii under low light availability in a low phosphorus lake. Acta Bot. Bras. 2015, 29, 448–451. [Google Scholar] [CrossRef][Green Version]
- Griffiths, D.J.; Saker, M.L. The Palm Island mystery disease 20 years on: A review of research on the cyanotoxin cylindrospermopsin. Environ. Toxicol. 2003, 18, 78–93. [Google Scholar] [CrossRef]
- Ohtani, I.; Moore, R.E.; Runnegar, M.T.C. Cylindrospermopsin: A potent hepatotoxin from the blue-green alga Cylindrospermopsis raciborskii. J. Am. Chem. Soc. 1992, 114, 7941–7942. [Google Scholar] [CrossRef]
- Carneiro, R.L.; Pacheco, A.B.F.; Azevedo, S.M.F.D.O. Growth and Saxitoxin Production by Cylindrospermopsis raciborskii (Cyanobacteria) Correlate with Water Hardness. Mar. Drugs 2013, 11, 2949–2963. [Google Scholar] [CrossRef]
- Lagos, N.; Onodera, H.; Zagatto, P.A.; Andrinolo, D.; Azevedo, S.M.F.Q.; Oshima, Y. The first evidence of paralytic shellfish toxins in the freshwater cyanobacterium Cylindrospermopsis raciborskii, isolated from Brazil. Toxicon 1999, 37, 1359–1373. [Google Scholar] [CrossRef]
- Panou, M.; Zervou, S.-K.; Christophoridis, C.; Manolidi, K.; Triantis, M.T.; Kaloudis, T.; Hiskia, A.; Gkelis, S. First report of a microcystin-producing Cylindrospermopsis raciborskii strain isolated from Greece. In Proceedings of the 10th International Conference on Toxic Cyanobacteria, Wuhan, China, 23–28 October 2016. [Google Scholar]
- Pestana, C.J.; Capelo-Neto, J.; Lawton, L.; Oliveira, S.; Carloto, I.; Linhares, H.P. The effect of water treatment unit processes on cyanobacterial trichome integrity. Sci. Total Environ. 2019, 659, 1403–1414. [Google Scholar] [CrossRef] [PubMed]
- Zamyadi, A.; Dorner, S.; Sauvé, S.; Ellis, D.; Bolduc, A.; Bastien, C.; Prévost, M. Species-dependence of cyanobacteria removal efficiency by different drinking water treatment processes. Water Res. 2013, 47, 2689–2700. [Google Scholar] [CrossRef] [PubMed]
- Westrick, J.A.; Szlag, D.C.; Southwell, B.J.; Sinclair, J. A review of cyanobacteria and cyanotoxins removal/inactivation in drinking water treatment. Anal. Bioanal. Chem. 2010, 397, 1705–1714. [Google Scholar] [CrossRef] [PubMed]
- Jung, S.W.; Baek, K.H.; Yu, M.J. Treatment of taste and odor material by oxidation and adsorption. Water Sci. Technol. 2004, 49, 289–295. [Google Scholar] [CrossRef] [PubMed]
- U.S. EPA. Water Treatment Optimization for Cyanotoxins; EPA 810-B-16-007; Version 1.0; United States Environmental Protection Agency: Cincinnati, OH, USA, 2016.
- Zamyadi, A.; Coral, L.A.; Barbeau, B.; Dorner, S.; Lapolli, F.R.; Prévost, M. Fate of toxic cyanobacterial genera from natural bloom events during ozonation. Water Res. 2015, 73, 204–215. [Google Scholar] [CrossRef]
- Miao, H.; Tao, W. The mechanisms of ozonation on cyanobacteria and its toxins removal. Sep. Purif. Tech. 2009, 66, 187–193. [Google Scholar] [CrossRef]
- AFNOR. Water Quality—Guidance Standard on Enumeration of Phytoplankton Using Inverted Microscopy (Utermöhl Technique); European Committee for Standardization: Brussels, Belgium, 2006. [Google Scholar]
- Komárek, J. Süßwasserflora von Mitteleuropa; Bd. 19/3: Cyanoprokaryota: 3. Teil/3rd part: Heterocytous Genera; Springer-Verlag: Berlin/Heidelberg, Germany, 2013; p. 1131. [Google Scholar]
- Alster, A.; Kaplan-Levy, R.N.; Sukenik, A.; Zohary, T. Morphology and phylogeny of a non-toxic invasive Cylindrospermopsis raciborskii from a Mediterranean Lake. Hydrobiologia 2010, 639, 115–128. [Google Scholar] [CrossRef]
- Dvořák, P.; Hašler, P. Occurrence and morphological variability of Cylindrospermopsis raciborskii (WOLOSZ.) SEENAYYA et SUBBA RAJU (Cyanophyta, Nostocales) near Olomouc in 2006. Fottea 2007, 7, 39–42. [Google Scholar]
- Shafik, H.M.; Vörös, L.; Sprőber, P.; Présing, M.; Kovács, A.W. Some special morphological features of Cylindrospermopsis raciborskii in batch and continuous cultures. Hydrobiologia 2003, 506, 163–167. [Google Scholar] [CrossRef]
- Lionello, P.; Scarascia, L. The relation between climate change in the Mediterranean region and global warming. Reg. Environ. Chang. 2018, 18, 1481–1493. [Google Scholar] [CrossRef]
- Viceto, C.; Cardoso Pereira, S.; Rocha, A. Climate Change Projections of Extreme Temperatures for the Iberian Peninsula. Atmosphere 2019, 10, 229. [Google Scholar] [CrossRef]
- Moreira, C.; Fathalli, A.; Vasconcelos, V.; Antunes, A. Phylogeny and biogeography of the invasive cyanobacterium Cylindrospermopsis raciborskii. Arch. Microbiol. 2015, 197, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Saker, M.L.; Nogueira, I.C.G.; Vasconcelos, V. Distribution and toxicity of Cylindrospermopsis raciborskii (Cyanobacteria) in Portuguese freshwaters. Limnetica 2004, 23, 145–152. [Google Scholar]
- Águas Públicas do Alentejo Annual Report. 2017. Available online: https://www.agda.pt/images/stories/Relat%C3%B3rio_e_Contas_2017.pdf (accessed on 1 October 2019).
- He, X.; Liu, Y.-L.; Conklin, A.; Westrick, J.; Weavers, L.K.; Dionysiou, D.D.; Lenhart, J.J.; Mouser, P.J.; Szlag, D.; Walker, H.W. Toxic cyanobacteria and drinking water: Impacts, detection, and treatment. Harmful Algae 2016, 54, 174–193. [Google Scholar] [CrossRef] [PubMed]
- Cheng, X.; Shi, H.; Adams, C.D.; Timmons, T.; Ma, Y. Effects of oxidative and physical treatments on inactivation of Cylindrospermopsis raciborskii and removal of cylindrospermopsin. Water Sci. Technol. 2009, 60, 689–697. [Google Scholar] [CrossRef] [PubMed]
- Szlag, D.; Sinclair, J.; Southwell, B.; Westrick, J. Cyanobacteria and Cyanotoxins Occurrence and Removal from Five High-Risk Conventional Treatment Drinking Water Plants. Toxins 2015, 7, 2198–2220. [Google Scholar] [CrossRef] [PubMed]
- Rzymski, P.; Horyn, O.; Budzyńska, A.; Jurczak, T.; Kokociński, M.; Niedzielski, P.; Klimaszyk, P.; Falfushynska, H. A report of Cylindrospermopsis raciborskii and other cyanobacteria in the water reservoirs of power plants in Ukraine. Environ. Sci. Pollut. Res. 2018, 25, 15245–15252. [Google Scholar] [CrossRef]
- Otten, T.G.; Graham, J.L.; Harris, T.D.; Dreher, T.W. Elucidation of Taste- and Odor-Producing Bacteria and Toxigenic Cyanobacteria in a Midwestern Drinking Water Supply Reservoir by Shotgun Metagenomic Analysis. Appl. Environ. Microbiol. 2016, 82, 5410–5420. [Google Scholar] [CrossRef]
- Fonseca, A.L.; Lankoff, A.; Azevedo, S.M.F.O.; Soares, R.M. Effects on DNA and cell viability of treated water contaminated with Cylindrospermopsis raciborskii extract including cylindrospermopsin. Ecotoxicol. Environ. Saf. 2013, 8, 135–141. [Google Scholar]
- Skulberg, R.; Skulberg, O.M. Forskning med algekulturer NIVAs kultursampling av alger. Research with algal cultures. In NIVA’s Culture Collection of Algae; Norsk Institutt for Vannforskning: Oslo, Norway, 1990. [Google Scholar]
- Paulino, S.; Sam-Bento, F.; Churro, C.; Alverca, E.; Dias, E.; Valério, E.; Pereira, P. The Estela Sousa e Silva Algal Culture Collection: A resource of biological and toxicological interest. Hydrobiologia 2009, 636, 489. [Google Scholar] [CrossRef]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of Image Analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef]
- Nübel, U.; Garcia-Pichel, F.; Muyzer, G. PCR primers to amplify 16S rRNA genes from cyanobacteria. Appl. Environ. Microbiol. 1997, 63, 3327–3332. [Google Scholar] [CrossRef]
- Valério, E.; Chambel, L.; Paulino, S.; Faria, N.; Pereira, P.; Tenreiro, R. Molecular identification, typing and traceability of cyanobacteria from freshwater reservoirs. Microbiology 2009, 155, 642–656. [Google Scholar] [CrossRef]
- Tamura, K.; Nei, M. Estimation of the number of nucleotide substitutions in the control region of mitochondrial DNA in humans and chimpanzees. Mol. Biol. Evol. 1993, 10, 512–526. [Google Scholar]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular Evolutionary Genetics Analysis Version 7.0 for Bigger Datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef]
- Botelho, M.J.; Vale, C.; Grilo, R.V.; Ferreira, J.G. Uptake and release of paralytic shellfish toxins by the clam Ruditapes decussatus exposed to Gymnodinium catenatum and subsequent depuration. Mar. Environ. Res. 2012, 77, 23–29. [Google Scholar] [CrossRef]
- Botelho, M.J.; Vale, C.; Mota, A.M.; Rodrigues, S.M.; Costa, P.R.; Gonçalves, M.L.S.S. Matrix effect on paralytic shellfish toxins quantification and toxicity estimation in mussels exposed to Gymnodinium catenatum. Food Addit. Contam. Part A 2010, 27, 1724–1732. [Google Scholar] [CrossRef]
- Lawrence, J.F.; Niedzwiadek, B.; Menard, C. Quantitative determination of paralytic shellfish poisoning toxins in shellfish using prechromatographic oxidation and liquid chromatography with fluorescence detection: Collaborative study. J. AOAC Int. 2005, 88, 1714–1732. [Google Scholar]
- Valério, E.; Chambel, L.; Paulino, S.; Faria, N.; Pereira, P.; Tenreiro, R. Multiplex PCR for detection of microcystins-producing cyanobacteria from freshwater samples. Environ. Toxicol. 2010, 25, 251–260. [Google Scholar] [CrossRef]
- Fergusson, K.M.; Saint, C.P. Multiplex PCR assay for Cylindrospermopsis raciborskii and cylindrospermopsin-producing cyanobacteria. Environ. Toxicol. 2003, 18, 120–125. [Google Scholar] [CrossRef]
- Cirés, S.; Delgado, A.; González-Pleiter, M.; Quesada, A. Temperature Influences the Production and Transport of Saxitoxin and the Expression of sxt Genes in the Cyanobacterium Aphanizomenon gracile. Toxins 2017, 9, 322. [Google Scholar] [CrossRef]
- Hoff-Risseti, C.; Dörr, F.A.; Schaker, P.D.C.; Pinto, E.; Werner, V.R.; Fiore, M.F. Cylindrospermopsin and Saxitoxin Synthetase Genes in Cylindrospermopsis raciborskii Strains from Brazilian Freshwater. PLoS ONE 2013, 8, e74238. [Google Scholar] [CrossRef]
| Phytoplankton Species | Cell Densities (cells mL−1) | |
|---|---|---|
| Exit Point of WTP | City Reservoir | |
| Cyanobacteria | ||
| Cylindrospermopsis raciborskii Pseudanabaena limnetica Aphanizomenon gracile | 1472 1042 − | 651 383 579 |
| Diatoms | ||
| Nitzschia acicularis | 30 | 13 |
| Chlorophytes | ||
| Scenedesmus ellipticus | 26 | - |
| Total | 2570 | 1627 |
| Isolates | Vegetative Cell Length Mean ± SD (µm) (N) | Vegetative Cell Width Mean ± SD (µm) (N) | Heterocyte Length Mean ± SD (µm) (N) | Heterocyte Width Mean ± SD (µm) (N) | Akinete Length Mean ± SD (µm) (N) | Akinete Width Mean ± SD (µm) (N) |
|---|---|---|---|---|---|---|
| LMECYA 324 | 10.83 ± 3.22 (52) | 1.74 ± 0.16 (50) | 5.91 ± 1.62 (63) | 1.90 ± 0.37 (64) | 12.18 ± 3.01 (18) | 2.94 ± 0.41 (18) |
| LMECYA 325 | 14.75 ± 2.61 (71) | 2.29 ± 0.19 (71) | 9.35 ± 2.54 (70) | 3.49 ± 0.61 (70) | 15.16 ± 3.36 (73) | 3.68 ± 0.50 (70) |
| LMECYA 326 | 9.05 ± 2.25 (71) | 1.73 ± 0.16 (73) | 6.03 ± 1.95 (50) | 1.98 ± 0.37 (50) | 13.71 ± 4.08 (50) | 3.50 ± 0.54 (53) |
| Toxin Genes/Cyanotoxin | LMECYA 324 | LMECYA 325 | LMECYA 326 |
|---|---|---|---|
| mcyA-cd/mcyAB/mcyB genes Microcystins (ELISA) | Negative <0.15 µg L−1 | Negative <0.15 µg L−1 | Negative <0.15 µg L−1 |
| PS/PKS genes Cylindrospermopsin (ELISA) | Negative <0.1 µg L−1 | Negative <0.1 µg L−1 | Negative <0.1 µg L−1 |
| sxtA/sxtA4/sxtB/sxtM/sxtPer/sxtI genes Saxitoxin | Negative 0.0178 µg L−1 (ELISA) <1.9 µg L−1 (LC-FLD) | Negative 0.0070 µg L−1 (ELISA) < 1.9 µg L−1 (LC-FLD) | Negative 0.0052 µg L−1 (ELISA) <1.9 µg L−1 (LC-FLD) |
| Gene | Primers | Sequence (5′-3′) | Size (bp) | References |
|---|---|---|---|---|
| sxtA | jrtPKSF | GGAGTGGATTTCAACACCAGAA | 147 | Referred to in [52] |
| jrtPKSR | GTTTCCCAGACTCGTTTCAGG | |||
| sxtA4 | SXTA4-F | GGACTCGGCTTGTTGCTTC | 200 | [53] |
| SXTA4-R | CCAGACAGCACGCTTCATAA | |||
| sxtB | SXTB-F | TTTGTAGGRCAGGCACTT | 400 | [53] |
| SXTB-R | ATCATCGGTATCATCGGTAG | |||
| sxtM | qMgrF | GAAGCACGAGTCAGCCTACA | 129 | [52] |
| qMgrR | CAAAGCACCACCAGCCAAAA | |||
| sxtPer | qPERgrF | CTGGGCGAGACATTTGAGA | 116 | [52] |
| qPERgrR | GCACAGAGACAGGCGAACTA | |||
| sxtI | OCT-F | TGCCGTTTTGTGCTTAGATG | 923 | [53] |
| OCT-R | GGACGGAAGGACTCACGATA |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Menezes, C.; Valério, E.; Botelho, M.J.; Dias, E. Isolation and Characterization of Cylindrospermopsis raciborskii Strains from Finished Drinking Water. Toxins 2020, 12, 40. https://doi.org/10.3390/toxins12010040
Menezes C, Valério E, Botelho MJ, Dias E. Isolation and Characterization of Cylindrospermopsis raciborskii Strains from Finished Drinking Water. Toxins. 2020; 12(1):40. https://doi.org/10.3390/toxins12010040
Chicago/Turabian StyleMenezes, Carina, Elisabete Valério, Maria João Botelho, and Elsa Dias. 2020. "Isolation and Characterization of Cylindrospermopsis raciborskii Strains from Finished Drinking Water" Toxins 12, no. 1: 40. https://doi.org/10.3390/toxins12010040
APA StyleMenezes, C., Valério, E., Botelho, M. J., & Dias, E. (2020). Isolation and Characterization of Cylindrospermopsis raciborskii Strains from Finished Drinking Water. Toxins, 12(1), 40. https://doi.org/10.3390/toxins12010040