Biomonitoring of Deoxynivalenol and Deoxynivalenol-3-glucoside in Human Volunteers: Renal Excretion Profiles
Abstract
1. Introduction
2. Results
2.1. General Results
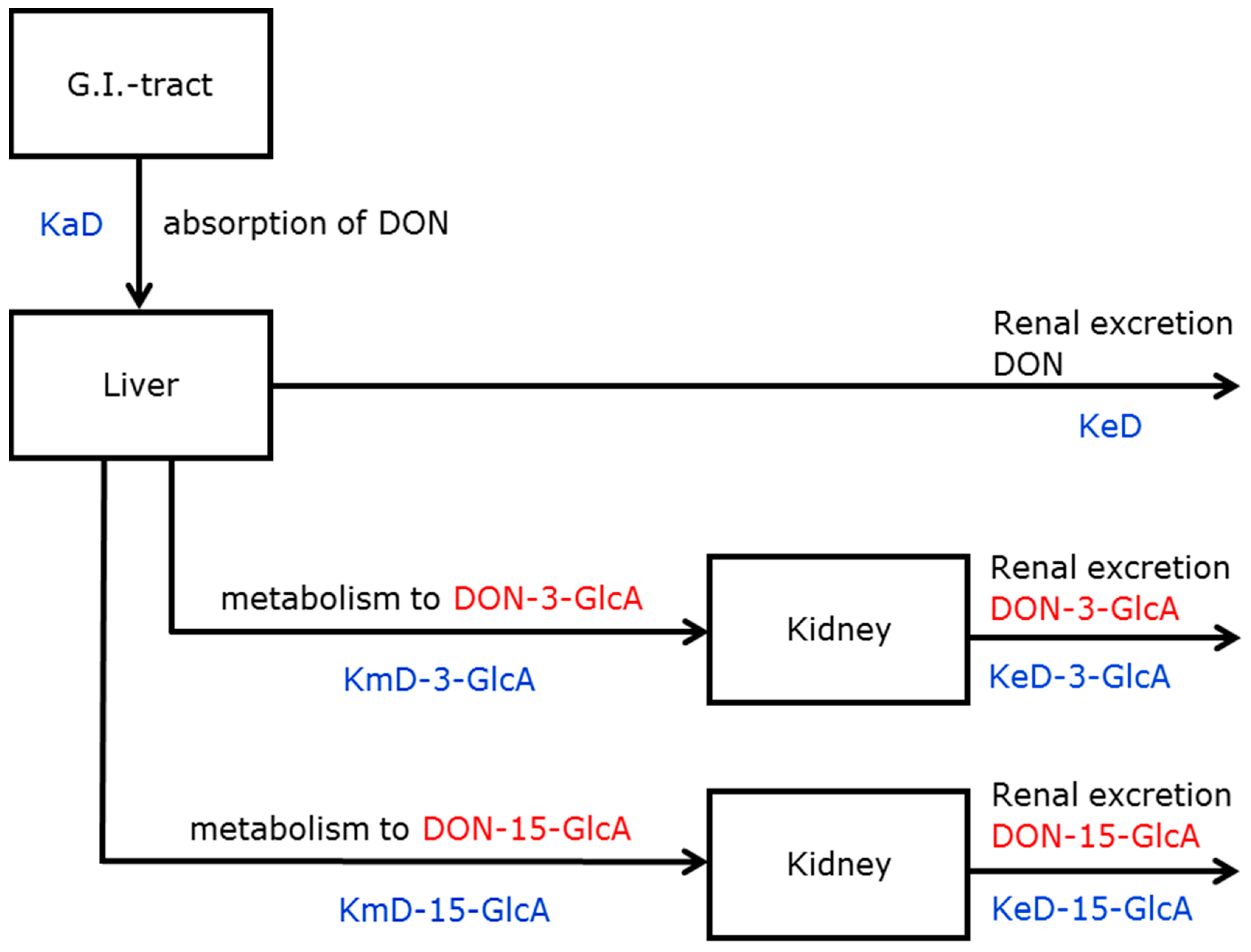
2.2. Deoxynivalenol (DON) Kinetics
2.2.1. Model Selection
2.2.2. Parameter Estimation
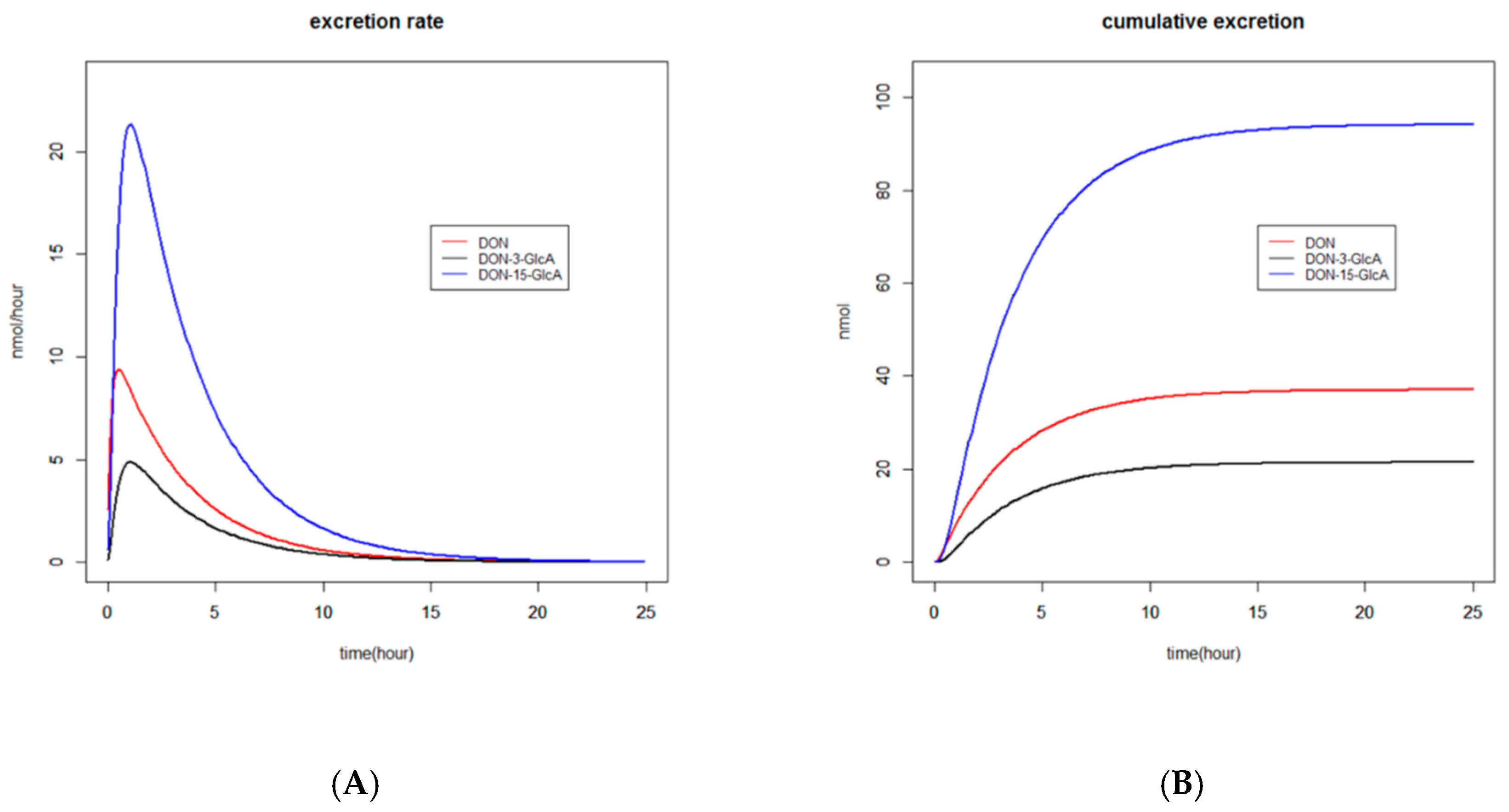
2.2.3. Graphs
2.2.4. Recovery
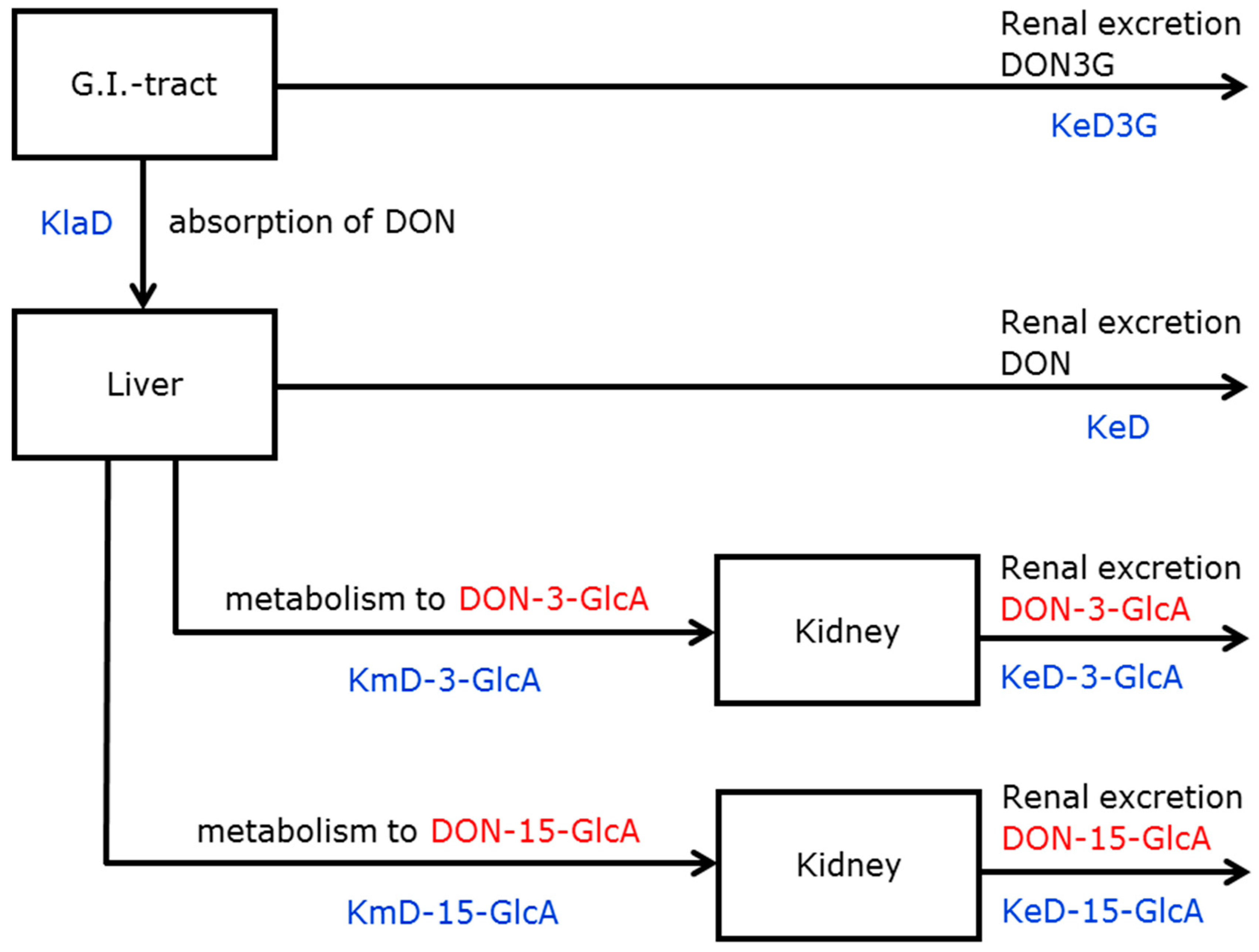
2.3. DON-3-glucoside (DON3G) Kinetics
2.3.1. Model Selection
2.3.2. Parameter Estimation
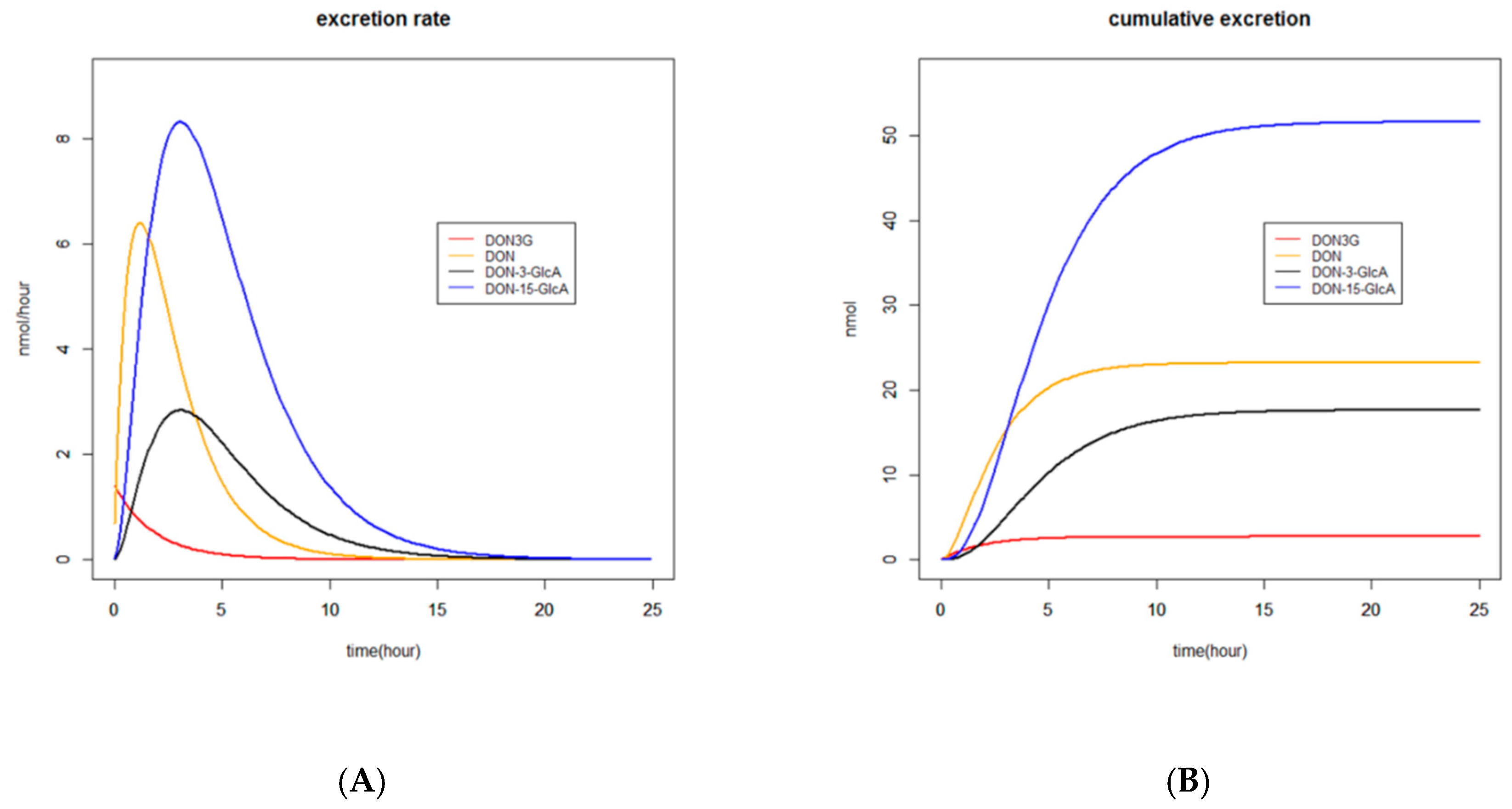
2.3.3. Graphs
2.3.4. Recovery
2.4. Biomonitoring
2.4.1. Preferred Biomarker and Reversed Dosimetry Factor
2.4.2. Collection Period
3. Discussion
3.1. Absorption, Distribution, Metabolism and Excretion (ADME) of DON and DON3G
3.1.1. Absorption and Distribution
3.1.2. Metabolism
3.1.3. Excretion
3.2. Modelling
3.2.1. Model Selection
3.2.2. Parametrisation
3.3. Biomonitoring
3.3.1. Preferred Biomarker and Exposure Calculation
3.3.2. Preferred Collection Period
4. Conclusions
5. Materials and Methods
5.1. Preparation of the DON/DON3G Dose
5.2. Study Design
5.3. Sample Preparation and Liquid Chromatography coupled to Tandem Mass Spectrometry LC-MS/MS Analysis
5.4. Modelling
5.4.1. Models for DON
5.4.2. Models for DON3G
5.5. Calculations and Statistical Evaluations
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Marin, S.; Ramos, A.J.; Cano-Sancho, G.; Sanchis, V. Mycotoxins: Occurrence, toxicology, and exposure assessment. Food Chem. Toxicol. 2013, 60, 218–237. [Google Scholar] [CrossRef] [PubMed]
- Berthiller, F.; Crews, C.; Dall‘Asta, C.; Saeger, S.D.; Haesaert, G.; Karlovsky, P.; Oswald, I.P.; Seefelder, W.; Speijers, G.; Stroka, J. Masked mycotoxins: A review. Mol. Nutr. Food Res. 2013, 57, 165–186. [Google Scholar] [CrossRef] [PubMed]
- De Boevre, M.; Jacxsens, L.; Lachat, C.; Eeckhout, M.; Di Mavungu, J.D.; Audenaert, K.; Maene, P.; Haesaert, G.; Kolsteren, P.; De Meulenaer, B.; et al. Human exposure to mycotoxins and their masked forms through cereal-based foods in Belgium. Toxicol. Lett. 2013, 218, 281–292. [Google Scholar] [CrossRef] [PubMed]
- Joint FAO/WHO Expert Committee on Food Additives (JECFA). Safety Evaluation of Certain Contaminants in Food. Prepared by the 72nd Meeting of the Joint FAO/WHO Expert Committee on Food Additives; WHO Food Additives Series 63; JECFA: Geneva, Switzerland, 2011. [Google Scholar]
- European Food Safety Authority (EFSA). Scientifc Opinion on the Risks to Human and Animal Health Related to the Presence of Deoxynivalenol and Its Acetylated and Modified Forms in Food and Feed; EFSA: Parma, Italy, 2017; Volume 15, p. 04718. [Google Scholar]
- Brera, C.; De Santis, B.; Debegnach, F.; Miano, B.; Moretti, G.; Lanzone, A.; Del Sordo, G.; Buonsenso, D.; Chiaretti, A.; Hardie, L.; et al. Experimental Study of Deoxynivalenol Biomarkers in Urine; EFSA: Parma, Italy, 2015. [Google Scholar]
- Warth, B.; Sulyok, M.; Berthiller, F.; Schuhmacher, R.; Krska, R. New insights into the human metabolism of the Fusarium mycotoxins deoxynivalenol and zearalenone. Toxicol. Lett. 2013, 220, 88–94. [Google Scholar] [CrossRef] [PubMed]
- Nagl, V.; Woechtl, B.; Schwartz-Zimmermann, H.E.; Hennig-Pauka, I.; Moll, W.D.; Adam, G.; Berthiller, F. Metabolism of the masked mycotoxin deoxynivalenol-3-glucoside in pigs. Toxicol. Lett. 2014, 229, 190–197. [Google Scholar] [CrossRef] [PubMed]
- Broekaert, N.; Devreese, M.; van Bergen, T.; Schauvliege, S.; De Boevre, M.; De Saeger, S.; Vanhaecke, L.; Berthiller, F.; Michlmayr, H.; Malachova, A.; et al. In vivo contribution of deoxynivalenol-3-beta-D-glucoside to deoxynivalenol exposure in broiler chickens and pigs: Oral bioavailability, hydrolysis and toxicokinetics. Arch. Toxicol. 2017, 91, 699–712. [Google Scholar] [CrossRef] [PubMed]
- Hostetler, G.L.; Ralston, R.A.; Schwartz, S.J. Flavones: Food Sources, Bioavailability, Metabolism, and Bioactivity. Adv. Nutr. 2017, 8, 423–435. [Google Scholar] [CrossRef] [PubMed]
- Williamson, G.; Clifford, M.N. Role of the small intestine, colon and microbiota in determining the metabolic fate of polyphenols. Biochem. Pharmacol. 2017, 139, 24–39. [Google Scholar] [CrossRef]
- Vidal, A.; Claeys, L.; Mengelers, M.; Vanhoorne, V.; Vervaet, C.; Huybrechts, B.; De Saeger, S.; De Boevre, M. Humans significantly metabolize and excrete the mycotoxin deoxynivalenol and its modified form deoxynivalenol-3-glucoside within 24 h. Sci. Rep. 2018, 8, 5255. [Google Scholar] [CrossRef]
- Vidal, A.; Mengelers, M.; Yang, S.; De Saeger, S.; De Boevre, M. Mycotoxin Biomarkers of Exposure: A Comprehensive Review. Compr. Rev. Food Sci. Food Saf. 2018, 17, 1127–1155. [Google Scholar] [CrossRef]
- Chen, Z.; Zheng, S.; Li, L.; Jiang, H. Metabolism of flavonoids in human: A comprehensive review. Curr. Drug Metab. 2014, 15, 48–61. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, I.; Marques, C.; Evora, A.; Cruz, L.; de Freitas, V.; Calhau, C.; Faria, A.; Mateus, N. Pharmacokinetics of table and Port red wine anthocyanins: A crossover trial in healthy men. Food Funct. 2017, 8, 2030–2037. [Google Scholar] [CrossRef] [PubMed]
- Actis-Goretta, L.; Dew, T.P.; Leveques, A.; Pereira-Caro, G.; Rein, M.; Teml, A.; Schafer, C.; Hofmann, U.; Schwab, M.; Eichelbaum, M.; et al. Gastrointestinal absorption and metabolism of hesperetin-7-O-rutinoside and hesperetin-7-O-glucoside in healthy humans. Mol. Nutr. Food Res. 2015, 59, 1651–1662. [Google Scholar] [CrossRef] [PubMed]
- Hollman, P.C.; de Vries, J.H.; van Leeuwen, S.D.; Mengelers, M.J.; Katan, M.B. Absorption of dietary quercetin glycosides and quercetin in healthy ileostomy volunteers. Am. J. Clin. Nutr. 1995, 62, 1276–1282. [Google Scholar] [CrossRef] [PubMed]
- Hollman, P.C.; vd Gaag, M.; Mengelers, M.J.; van Trijp, J.M.; de Vries, J.H.; Katan, M.B. Absorption and disposition kinetics of the dietary antioxidant quercetin in man. Free Radic. Biol. Med. 1996, 21, 703–707. [Google Scholar] [CrossRef]
- Berthiller, F.; Krska, R.; Domig, K.J.; Kneifel, W.; Juge, N.; Schuhmacher, R.; Adam, G. Hydrolytic fate of deoxynivalenol-3-glucoside during digestion. Toxicol. Lett. 2011, 206, 264–267. [Google Scholar] [CrossRef] [PubMed]
- Heyndrickx, E.; Sioen, I.; Huybrechts, B.; Callebaut, A.; De Henauw, S.; De Saeger, S. Human biomonitoring of multiple mycotoxins in the Belgian population: Results of the BIOMYCO study. Environ. Int. 2015, 84, 82–89. [Google Scholar] [CrossRef] [PubMed]
- Turner, P.C.; Hopton, R.P.; White, K.L.; Fisher, J.; Cade, J.E.; Wild, C.P. Assessment of deoxynivalenol metabolite profiles in UK adults. Food Chem. Toxicol. 2011, 49, 132–135. [Google Scholar] [CrossRef] [PubMed]
- Turner, P.C.; Rothwell, J.A.; White, K.L.; Gong, Y.; Cade, J.E.; Wild, C.P. Urinary deoxynivalenol is correlated with cereal intake in individuals from the United kingdom. Environ. Health Perspect. 2008, 116, 21–25. [Google Scholar] [CrossRef] [PubMed]
- Turner, P.C.; White, K.L.; Burley, V.J.; Hopton, R.P.; Rajendram, A.; Fisher, J.; Cade, J.E.; Wild, C.P. A comparison of deoxynivalenol intake and urinary deoxynivalenol in UK adults. Biomarkers 2010, 15, 553–562. [Google Scholar] [CrossRef] [PubMed]
- Janssen, E.M.; Sprong, R.C.; Wester, P.W.; Boevre, M.; Mengelers, M.J.B. Risk assessment of chronic dietary exposure to the conjugated mycotoxin deoxynivalenol-3-β-glucoside in the Dutch population. World Mycotoxin J. 2015, 8, 1–12. [Google Scholar] [CrossRef]
- Oehlert, G.W. A Note on the Delta Method. Am. Stat. 1992, 46, 27–29. [Google Scholar]
| Value | Standard Error | |
|---|---|---|
| Fabs | 0.56 | 0.06 |
| KaD | 0.31 | 0.04 |
| KmD-3-GlcA | 0.73 | 0.10 |
| KmD-15-GlcA | 3.29 | 0.39 |
| KeD | 1.28 | 0.14 |
| KeD-3-GlcA | 1.28 | 0.14 |
| KeD-15-GlcA | 2.31 | 0.47 |
| Value | Standard Error | |
|---|---|---|
| Fabs | 0.53 | 0.04 |
| KlaD | 0.67 | 0.79 |
| KmD-3-GlcA | 0.13 | 0.15 |
| KmD-15-GlcA | 0.38 | 0.45 |
| KeD3G | 0.018 | 0.017 |
| KeD | 0.17 | 0.20 |
| KeD-3-GlcA | 0.49 | 0.07 |
| KeD-15-GlcA | 0.49 | 0.07 |
| Single Dose | Substance | RDF | CI | CI-lower Limit | CI-upper Limit |
|---|---|---|---|---|---|
| DON | DON | 7.20 | 26.7 | 2.37 | 29.1 |
| DON-3-GlcA | 12.6 | 40.2 | 4.12 | 44.34 | |
| DON-15-GlcA | 2.67 | 6.56 | 1.40 | 7.96 | |
| total DON | 1.45 | 6.07 | 1.03 | 7.10 | |
| DON3G | DON3G | - | - | - | - |
| DON | 6.95 | 11.47 | 3.53 | 15.00 | |
| DON-3-GlcA | 11.21 | 26.5 | 4.47 | 31.07 | |
| DON-15-GLcA | 3.37 | 5.35 | 1.90 | 7.25 | |
| total DON | 1.73 | 2.81 | 1.18 | 3.99 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mengelers, M.; Zeilmaker, M.; Vidal, A.; De Boevre, M.; De Saeger, S.; Hoogenveen, R. Biomonitoring of Deoxynivalenol and Deoxynivalenol-3-glucoside in Human Volunteers: Renal Excretion Profiles. Toxins 2019, 11, 466. https://doi.org/10.3390/toxins11080466
Mengelers M, Zeilmaker M, Vidal A, De Boevre M, De Saeger S, Hoogenveen R. Biomonitoring of Deoxynivalenol and Deoxynivalenol-3-glucoside in Human Volunteers: Renal Excretion Profiles. Toxins. 2019; 11(8):466. https://doi.org/10.3390/toxins11080466
Chicago/Turabian StyleMengelers, Marcel, Marco Zeilmaker, Arnau Vidal, Marthe De Boevre, Sarah De Saeger, and Rudolf Hoogenveen. 2019. "Biomonitoring of Deoxynivalenol and Deoxynivalenol-3-glucoside in Human Volunteers: Renal Excretion Profiles" Toxins 11, no. 8: 466. https://doi.org/10.3390/toxins11080466
APA StyleMengelers, M., Zeilmaker, M., Vidal, A., De Boevre, M., De Saeger, S., & Hoogenveen, R. (2019). Biomonitoring of Deoxynivalenol and Deoxynivalenol-3-glucoside in Human Volunteers: Renal Excretion Profiles. Toxins, 11(8), 466. https://doi.org/10.3390/toxins11080466







