Daphnia magna Exudates Impact Physiological and Metabolic Changes in Microcystis aeruginosa
Abstract
1. Introduction
2. Results
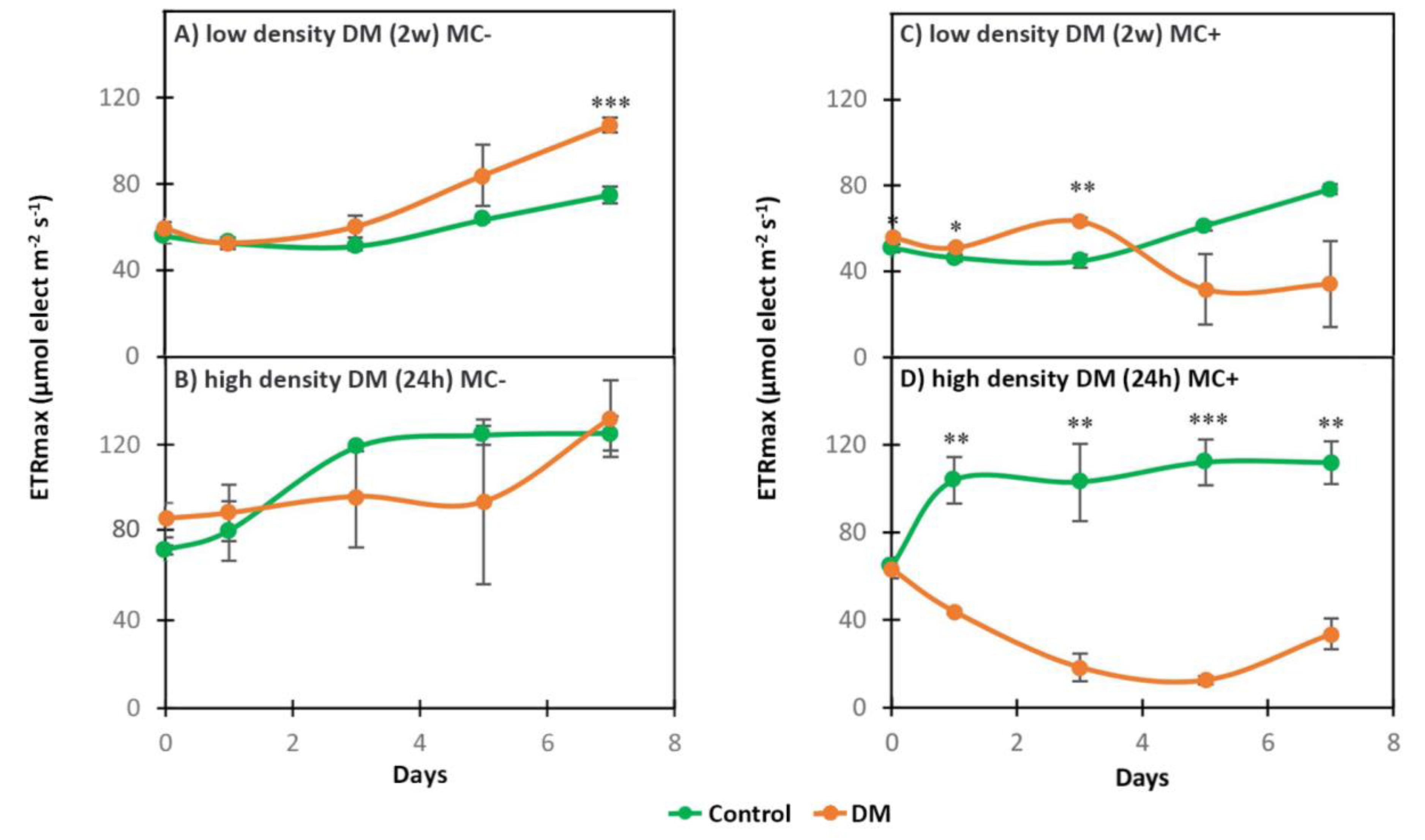
2.1. Photosynthetic Activity
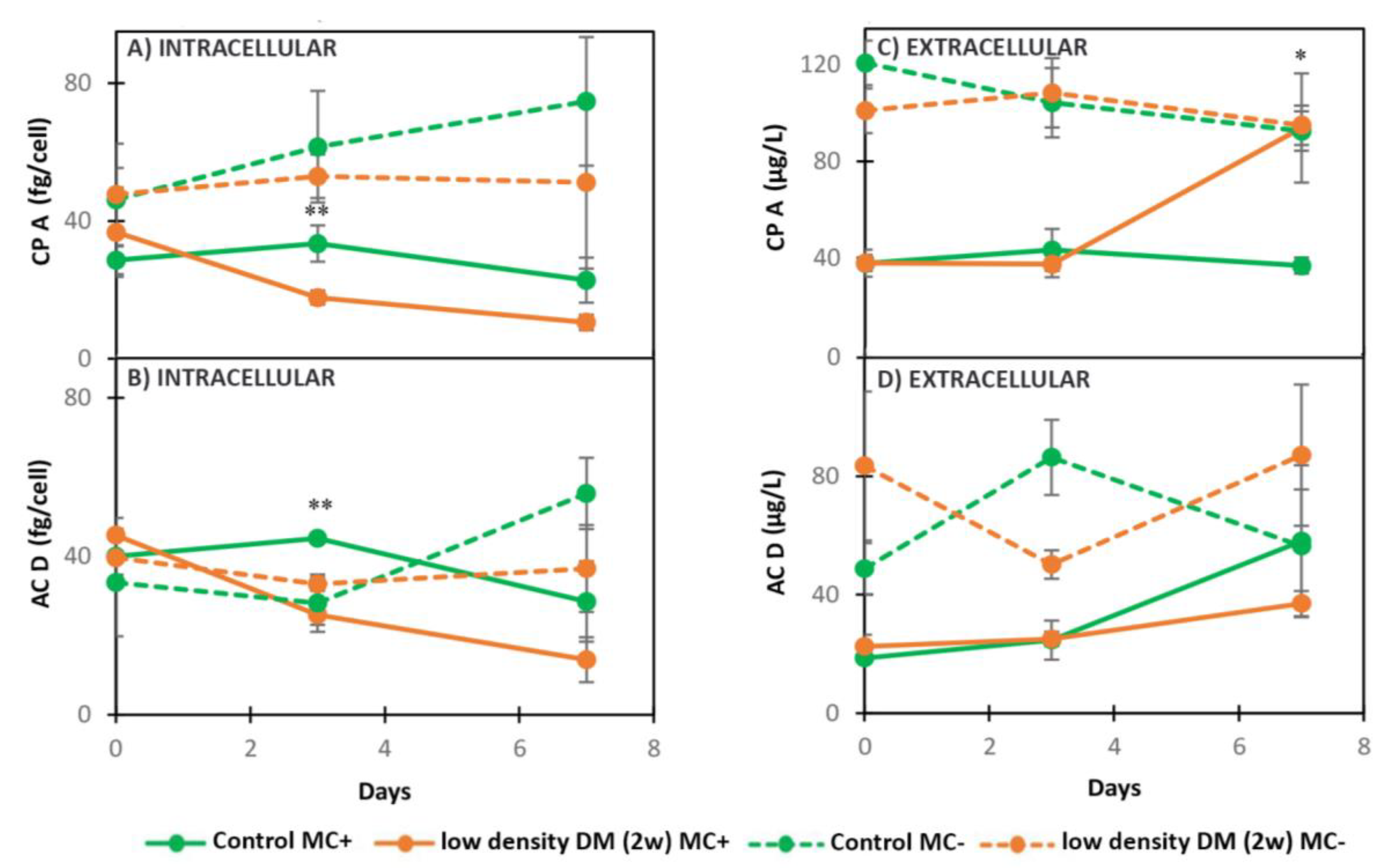
2.2. Dynamics of Intracellular and Extracellular Metabolites in MC+ and MC-
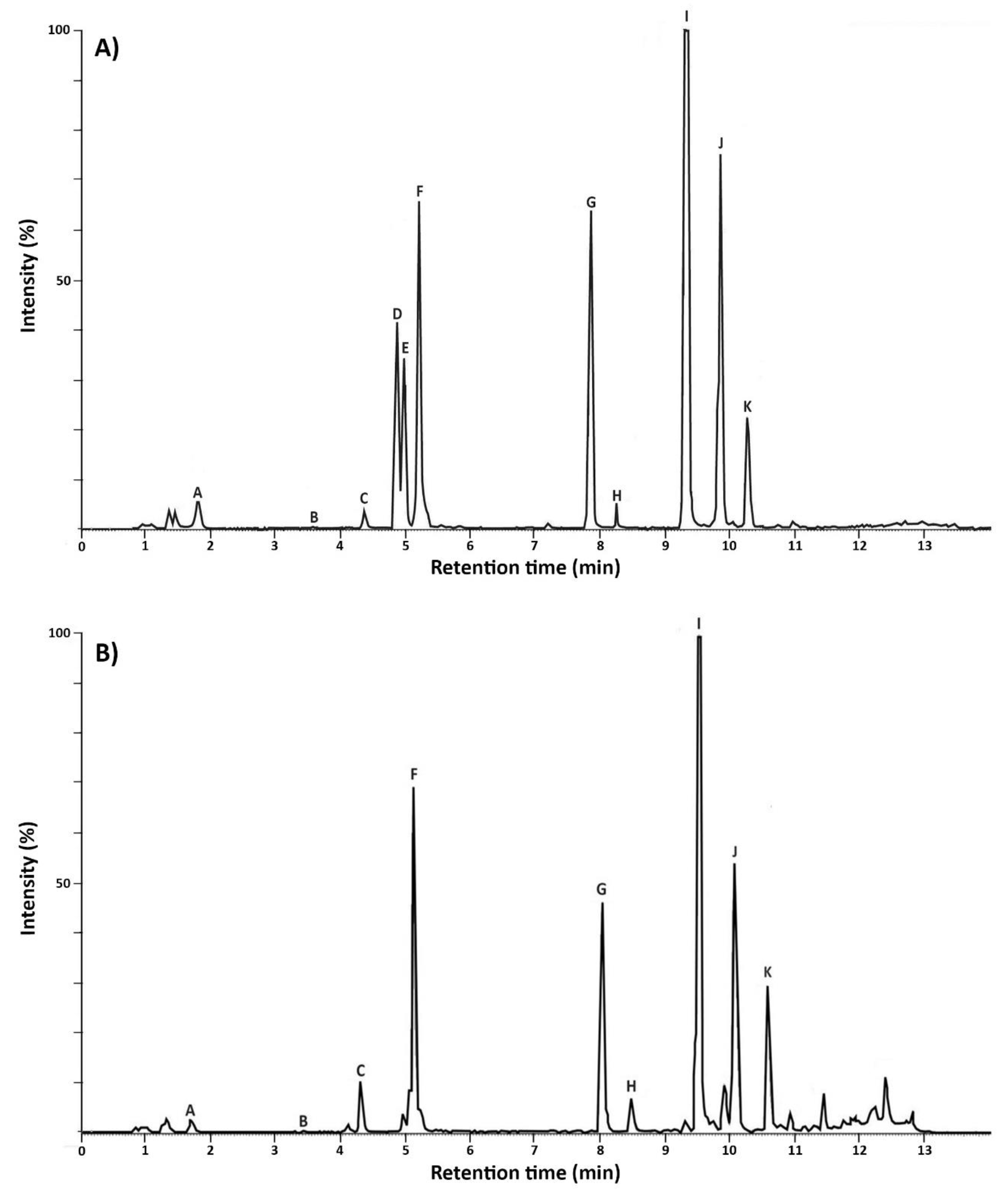
2.2.1. Metabolic Profiles of MC+ and MC–
2.2.2. Dynamics of Intracellular and Extracellular Metabolites in MC+ and MC- When Exposed to Low Density DM (2w)
2.2.3. Dynamics of Intracellular and Extracellular Metabolites in MC+ When Exposed to High Density DM (24 h)
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Culture Conditions
5.1.1. Cyanobacterial Cultures
5.1.2. D. Magna
5.2. Experimental Design
5.3. Photosynthetic Activity
5.4. Cyanobacterial Cell Density
5.5. Analysis of Cyanobacterial Secondary Metabolites
5.6. Statistics
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| MC+ | M. aeruginosa PCC7806 |
| MC- | M. aeruginosa PCC7806 mcy- |
| DM | Daphnia medium |
| low density DM (1w) | spent medium in which 75 neonatal individuals of D. magna/L were cultured for 1 week |
| low density DM (2w) | spent medium in which 75 all age individuals of D. magna/L were cultured for 2 weeks |
| high density DM (24 h) | spent medium in which 200 all age individuals of D.magna/L were cultured for 24 h |
| MC-LR | microcystin LR |
| des-MC-LR | des microcystin LR |
| CP | cyanopeptolin |
| AC | aerucyclamide |
| CP A | cyanopeptolin A |
| AC D | aerucyclamide D |
| Control MC+ | M. aeruginosa PCC7806 cultivated in BG11 |
| Control MC- | M. aeruginosa PCC7806 mcy- cultivated in BG11 |
References
- Holland, A.; Kinnear, S. Interpreting the Possible Ecological Role(s) of Cyanotoxins: Compounds for Competitive Advantage and/or Physiological Aide? Mar. Drugs 2013, 11, 2239–2258. [Google Scholar] [CrossRef] [PubMed]
- Asselman, J.; Hochmuth, J.D.; De Schamphelaere, K.A.C. A comparison of the sensitivities of Daphnia magna and Daphnia pulex to six different cyanobacteria. Harmful Algae 2014, 39, 1–7. [Google Scholar] [CrossRef]
- Ekvall, M.K.; Urrutia-Cordero, P.; Hansson, L.A. Linking cascading effects of fish predation and zooplankton grazing to reduced cyanobacterial biomass and toxin levels following biomanipulation. PLoS ONE 2014, 9, e112956. [Google Scholar] [CrossRef] [PubMed]
- Urrutia-Cordero, P.; Ekvall, M.K.; Hansson, L.-A. Controlling Harmful Cyanobacteria: Taxa-Specific Responses of Cyanobacteria to Grazing by Large-Bodied Daphnia in a Biomanipulation Scenario. PLoS ONE 2016, 11, e0153032. [Google Scholar] [CrossRef] [PubMed]
- Rohrlack, T.; Dittmann, E.; Börner, T.; Christoffersen, K. Effects of Cell-Bound Microcystins on Survival and Feeding of Daphnia spp. Appl. Environ. Microbiol. 2001, 67, 3523–3529. [Google Scholar] [CrossRef] [PubMed]
- von Elert, E.; Zitt, A.; Schwarzenberger, A. Inducible tolerance to dietary protease inhibitors in Daphnia magna. J. Exp. Biol. 2012, 215, 2051–2059. [Google Scholar] [CrossRef] [PubMed]
- Lampert, W. Inhibitory and Toxic Effects of Blue green Algae on Daphnia. Int. Rev. Gesamten Hydrobiol. Hydrogr. 1981, 66, 285–298. [Google Scholar] [CrossRef]
- Demott, W.R.; Zhang, Q.-X.; Carmichael, W.W. Effects of toxic cyanobacteria and purified toxins on the survival and feeding of a copepod and three species of Daphnia. Limnol. Oceanogr. 1991, 36, 1346–1357. [Google Scholar] [CrossRef]
- Dao, T.S.; Do-Hong, L.C.; Wiegand, C. Chronic effects of cyanobacterial toxins on Daphnia magna and their offspring. Toxicon 2010, 55, 1244–1254. [Google Scholar] [CrossRef]
- Mowe, M.A.D.; Mitrovic, S.M.; Lim, R.P.; Furey, A.; Yeo, D.C.J. Tropical cyanobacterial blooms: A review of prevalence, problem taxa, toxins and influencing environmental factors. J. Limnol. 2015, 74, 205–224. [Google Scholar] [CrossRef]
- MacKintosh, C.; Beattie, K.A.; Klumpp, S.; Cohen, P.; Codd, G.A. Cyanobacterial microcystin-LR is a potent and specific inhibitor of protein phosphatases 1 and 2A from both mammals and higher plants. FEBS Lett. 1990, 264, 187–192. [Google Scholar] [CrossRef]
- DeMott, W.R.; Dhawale, S. Inhibition of in vitro protein phosphatase activity in three zooplankton species by microcystin-LR, a toxin from cyanobacteria. Arch. Hydrobiol. 1995, 134, 417–424. [Google Scholar]
- Zurawell, R.W.; Chen, H.; Burke, J.M.; Prepas, E.E. Hepatotoxic cyanobacteria: A review of the biological importance of microcystins in freshwater environments. J. Toxicol. Environ. Health B 2005, 8, 1–37. [Google Scholar] [CrossRef] [PubMed]
- Amado, L.L.; Monserrat, J.M. Oxidative stress generation by microcystins in aquatic animals: Why and how. Environ. Int. 2010, 36, 226–235. [Google Scholar] [CrossRef] [PubMed]
- Wiegand, C.; Pflugmacher, S. Ecotoxicological effects of selected cyanobacterial secondary metabolites: A short review. Toxicol. Appl. Pharmacol. 2005, 203, 201–218. [Google Scholar] [CrossRef] [PubMed]
- Kultschar, B.; Llewellyn, C. Secondary Metabolites in Cyanobacteria. In Secondary Metabolites—Sources and Applications; InTech: London, UK, 2018. [Google Scholar]
- Neilan, B.A.; Pearson, L.A.; Muenchhoff, J.; Moffitt, M.C.; Dittmann, E. Environmental conditions that influence toxin biosynthesis in cyanobacteria. Environ. Microbiol. 2013, 15, 1239–1253. [Google Scholar] [CrossRef]
- Herms, D.A.; Mattson, W.J. The Dilemma of Plants: To Grow or Defend. Q. Rev. Biol. 1992, 67, 283–335. [Google Scholar] [CrossRef]
- Kaebernick, M.; Neilan, B.A. Ecological and molecular investigations of cyanotoxin production. FEMS Microbiol. Ecol. 2001, 35, 1–9. [Google Scholar] [CrossRef]
- Alexova, R.; Fujii, M.; Birch, D.; Cheng, J.; Waite, T.D.; Ferrari, B.C.; Neilan, B.A. Iron uptake and toxin synthesis in the bloom-forming Microcystis aeruginosa under iron limitation. Environ. Microbiol. 2011, 13, 1064–1077. [Google Scholar] [CrossRef]
- Dziallas, C.; Grossart, H.P. Increasing oxygen radicals and water temperature select for toxic Microcystis sp. PLoS ONE 2011, 6, e25569. [Google Scholar] [CrossRef]
- Zilliges, Y.; Kehr, J.C.; Meissner, S.; Ishida, K.; Mikkat, S.; Hagemann, M.; Kaplan, A.; Börner, T.; Dittmann, E. The cyanobacterial hepatotoxin microcystin binds to proteins and increases the fitness of Microcystis under oxidative stress conditions. PLoS ONE 2011, 6, e17615. [Google Scholar] [CrossRef]
- Kaplan, A.; Harel, M.; Kaplan-Levy, R.N.; Hadas, O.; Sukenik, A.; Dittmann, E. The languages spoken in the water body (or the biological role of cyanobacterial toxins). Front. Microbiol. 2012, 3, 138. [Google Scholar] [CrossRef]
- Jang, M.-H.; Ha, K.; Lucas, M.C.; Joo, G.-J.; Takamura, N. Changes in microcystin production by Microcystis aeruginosa exposed to phytoplanktivorous and omnivorous fish. Aquat. Toxicol. 2004, 68, 51–59. [Google Scholar] [CrossRef]
- Jang, M.H.; Jung, J.M.; Takamura, N. Changes in microcystin production in cyanobacteria exposed to zooplankton at different population densities and infochemical concentrations. Limnol. Oceanogr. 2007, 52, 1454–1466. [Google Scholar] [CrossRef]
- Sadler, T.; von Elert, E. Physiological interaction of Daphnia and Microcystis with regard to cyanobacterial secondary metabolites. Aquat. Toxicol. 2014, 156, 96–105. [Google Scholar] [CrossRef]
- Pérez-morales, A.; Sarma, S.S.S.; Nandini, S. Producción de microcistinas en Microcystis inducida por Daphnia pulex (Cladocera) y Brachionus calyciflorus (Rotifera). Hidrobiológica 2015, 25, 411–415. [Google Scholar]
- Omidi, A.; Esterhuizen-Londt, M.; Pflugmacher, S. Still challenging: The ecological function of the cyanobacterial toxin microcystin—What we know so far. Toxin Rev. 2018, 37, 87–105. [Google Scholar] [CrossRef]
- Chislock, M.F.; Doster, E.; Zitomer, R.A.; Wilson, A.E. Eutrophication: Causes, Consequences, and Controls in Aquatic Ecosystems. Nat. Educ. Knowl. 2013, 4, 10. [Google Scholar]
- Briand, E.; Yéprémian, C.; Humbert, J.F.; Quiblier, C. Competition between microcystin- and non-microcystin-producing Planktothrix agardhii (cyanobacteria) strains under different environmental conditions. Environ. Microbiol. 2008, 10, 3337–3348. [Google Scholar] [CrossRef]
- Van de Waal, D.B.; Verspagen, J.M.H.; Finke, J.F.; Vournazou, V.; Immers, A.K.; Kardinaal, W.E.A.; Tonk, L.; Becker, S.; Van Donk, E.; Visser, P.M.; et al. Reversal in competitive dominance of a toxic versus non-toxic cyanobacterium in response to rising CO2. ISME J. 2011, 5, 1438–1450. [Google Scholar] [CrossRef]
- Yeung, A.C.Y.; D’Agostino, P.M.; Poljak, A.; McDonald, J.; Bligh, M.W.; Waite, T.D.; Neilan, B.A. Physiological and proteomic responses of continuous cultures of Microcystis aeruginosa PCC 7806 to changes in iron bioavailability and growth rate. Appl. Environ. Microbiol. 2016, 82, 5918–5929. [Google Scholar] [CrossRef]
- Paerl, H.W.; Otten, T.G. Blooms bite the hand that feeds them. Science 2013, 342, 433–434. [Google Scholar] [CrossRef]
- Schuurmans, J.M.; Brinkmann, B.W.; Makower, A.K.; Dittmann, E.; Huisman, J.; Matthijs, H.C.P. Microcystin interferes with defense against high oxidative stress in harmful cyanobacteria. Harmful Algae 2018, 78, 47–55. [Google Scholar] [CrossRef]
- Izydorczyk, K.; Jurczak, T.; Wojtal-frankiewicz, A.; Skowron, A.; Mankiewicz-Boczek, J.; Tarczyńska, M. Influence of abiotic and biotic factors on microcystin content in Microcystis aeruginosa cells in a eutrophic temperate reservoir. J. Plankton Res. 2008, 30, 393–400. [Google Scholar] [CrossRef]
- Briand, E.; Bormans, M.; Gugger, M.; Dorrestein, P.C.; Gerwick, W.H. Changes in secondary metabolic profiles of Microcystis aeruginosa strains in response to intraspecific interactions. Environ. Microbiol. 2016, 18, 384–400. [Google Scholar] [CrossRef]
- Briand, E.; Reubrecht, S.; Mondeguer, F.; Sibat, M.; Hess, P.; Amzil, Z.; Bormans, M. Chemically mediated interactions between Microcystis and Planktothrix: Impact on their growth, morphology and metabolic profiles. Environ. Microbiol. 2019, 21, 1552–1566. [Google Scholar] [CrossRef]
- van Gremberghe, I.; Vanormelingen, P.; Van der Gucht, K.; Mancheva, A.; D’hondt, S.; De Meester, L.; Vyverman, W. Influence of Daphnia infochemicals on functional traits of Microcystis strains (cyanobacteria). Hydrobiologia 2009, 635, 147–155. [Google Scholar] [CrossRef]
- Chislock, M.F.; Sarnelle, O.; Jernigan, L.M.; Wilson, A.E. Do high concentrations of microcystin prevent Daphnia control of phytoplankton? Water Res. 2013, 47, 1961–1970. [Google Scholar] [CrossRef]
- Rantala, A.; Fewer, D.P.; Hisbergues, M.; Rouhiainen, L.; Vaitomaa, J.; Börner, T.; Sivonen, K. Phylogenetic evidence for the early evolution of microcystin synthesis. Proc. Natl. Acad. Sci. USA 2004, 101, 568–573. [Google Scholar] [CrossRef]
- Wilken, S.; Wiezer, S.; Huisman, J.; Van Donk, E. Microcystins do not provide anti-herbivore defence against mixotrophic flagellates. Aquat. Microb. Ecol. 2010, 59, 207–216. [Google Scholar] [CrossRef]
- Ger, K.A.; Urrutia-Cordero, P.; Frost, P.C.; Hansson, L.A.; Sarnelle, O.; Wilson, A.E.; Lürling, M. The interaction between cyanobacteria and zooplankton in a more eutrophic world. Harmful Algae 2016, 54, 128–144. [Google Scholar] [CrossRef]
- Jang, M.H.; Ha, K.; Takamura, N. Microcystin production by Microcystis aeruginosa exposed to different stages of herbivorous zooplankton. Toxicon 2008, 51, 882–889. [Google Scholar] [CrossRef]
- Van Gremberghe, I.; Vanormelingen, P.; Vanelslander, B.; Van Der Gucht, K.; D’Hondt, S.; De Meester, L.; Vyverman, W. Genotype-dependent interactions among sympatric Microcystis strains mediated by Daphnia grazing. Oikos 2009, 118, 1647–1658. [Google Scholar] [CrossRef]
- Becker, S. Biotic factors in induced defence revisited: Cell aggregate formation in the toxic cyanobacterium Microcystis aeruginosa PCC 7806 is triggered by spent Daphnia medium and disrupted cells. Hydrobiologia 2010, 644, 159–168. [Google Scholar] [CrossRef]
- Jang, M.-H.; Ha, K.; Joo, G.-J.; Takamura, N. Toxin production of cyanobacteria is increased by exposure to zooplankton. Freshw. Biol. 2003, 48, 1540–1550. [Google Scholar] [CrossRef]
- Pineda-Mendoza, R.M.; Zúñiga, G.; Martínez-Jerónimo, F. Infochemicals released by Daphnia magna fed on Microcystis aeruginosa affect mcyA gene expression. Toxicon 2014, 80, 78–86. [Google Scholar] [CrossRef]
- Van Donk, E. Chemical information transfer in freshwater plankton. Ecol. Inform. 2007, 2, 112–120. [Google Scholar] [CrossRef]
- Welker, M.; Von Döhren, H. Cyanobacterial peptides—Nature’s own combinatorial biosynthesis. FEMS Microbiol. Rev. 2006, 30, 530–563. [Google Scholar] [CrossRef]
- Rohrlack, T.; Christoffersen, K.; Kaebernick, M.; Neilan, B.A. Cyanobacterial protease inhibitor microviridin J causes a lethal molting disruption in Daphnia pulicaria. Appl. Environ. Microbiol. 2004, 70, 5047–5050. [Google Scholar] [CrossRef]
- Bister, B.; Keller, S.; Baumann, H.I.; Nicholson, G.; Weist, S.; Jung, G.; Süssmuth, R.D.; Jüttner, F. Cyanopeptolin 963A, a chymotrypsin inhibitor of Microcystis PCC 7806. J. Nat. Prod. 2004, 67, 1755–1757. [Google Scholar] [CrossRef]
- Von Elert, E.; Oberer, L.; Merkel, P.; Huhn, T.; Blom, J.F. Cyanopeptolin 954, a chlorine-containing chymotrypsin inhibitor of Microcystis aeruginosa NIVA Cya 43. J. Nat. Prod. 2005, 68, 1324–1327. [Google Scholar] [CrossRef]
- Ishida, K.; Christiansen, G.; Yoshida, W.Y.; Kurmayer, R.; Welker, M.; Valls, N.; Bonjoch, J.; Hertweck, C.; Börner, T.; Hemscheidt, T.; et al. Biosynthesis and Structure of Aeruginoside 126A and 126B, Cyanobacterial Peptide Glycosides Bearing a 2-Carboxy-6-Hydroxyoctahydroindole Moiety. Chem. Biol. 2007, 14, 565–576. [Google Scholar] [CrossRef]
- Elkobi-Peer, S.; Singh, R.K.; Mohapatra, T.M.; Tiwari, S.P.; Carmeli, S. Aeruginosins from a microcystis sp. bloom material collected in Varanasi, India. J. Nat. Prod. 2013, 76, 1187–1190. [Google Scholar] [CrossRef]
- Lürling, M. Daphnia growth on microcystin-producing and microcystin-free Microcystis aeruginosa in different mixtures with the green alga Scenedesmus obliquus. Limnol. Oceanogr. 2003, 48, 2214–2220. [Google Scholar] [CrossRef]
- Czarnecki, O.; Henning, M.; Lippert, I.; Welker, M. Identification of peptide metabolites of Microcystis (Cyanobacteria) that inhibit trypsin-like activity in planktonic herbivorous Daphnia (Cladocera). Environ. Microbiol. 2006, 8, 77–87. [Google Scholar] [CrossRef]
- O’Neil, J.M.; Davis, T.W.; Burford, M.A.; Gobler, C.J. The rise of harmful cyanobacteria blooms: The potential roles of eutrophication and climate change. Harmful Algae 2012, 14, 313–334. [Google Scholar] [CrossRef]
- Briand, E.; Bormans, M.; Quiblier, C.; Salençon, M.J.; Humbert, J.F. Evidence of the cost of the production of microcystins by Microcystis aeruginosa under differing light and nitrate environmental conditions. PLoS ONE 2012, 7, e29981. [Google Scholar] [CrossRef]
- Harke, M.J.; Jankowiak, J.G.; Morrell, B.K.; Gobler, C.J. Transcriptomic responses in the bloom-forming cyanobacterium Microcystis induced during exposure to zooplankton. Appl. Environ. Microbiol. 2017, 83, e02832-16. [Google Scholar] [CrossRef]
- Pannard, A.; Pédrono, J.; Bormans, M.; Briand, E.; Claquin, P.; Lagadeuc, Y. Production of exopolymers (EPS) by cyanobacteria: Impact on the carbon-to-nutrient ratio of the particulate organic matter. Aquat. Ecol. 2016, 50, 29–44. [Google Scholar] [CrossRef]
- Miles, C.O.; Sandvik, M.; Nonga, H.E.; Ballot, A.; Wilkins, A.L.; Rise, F.; Jaabaek, J.A.H.; Loader, J.I. Conjugation of Microcystins with Thiols Is Reversible: Base-Catalyzed Deconjugation for Chemical Analysis. Chem. Res. Toxicol. 2016, 29, 860–870. [Google Scholar] [CrossRef]
- Alexova, R.; Dang, T.C.; Fujii, M.; Raftery, M.J.; Waite, T.D.; Ferrari, B.C.; Neilan, B.A. Specific global responses to N and Fe nutrition in toxic and non-toxic Microcystis aeruginosa. Environ. Microbiol. 2016, 18, 401–413. [Google Scholar] [CrossRef]
- Sarnelle, O. Initial conditions mediate the interaction between Daphnia and bloom-forming cyanobacteria. Limnol. Oceanogr. 2007, 52, 2120–2127. [Google Scholar] [CrossRef]
- Kurmayer, R.; Deng, L.; Entfellner, E. Role of toxic and bioactive secondary metabolites in colonization and bloom formation by filamentous cyanobacteria Planktothrix. Harmful Algae 2016, 54, 69–86. [Google Scholar] [CrossRef]
- Janssen, E.M.-L. Cyanobacterial peptides beyond microcystins—A review on co-occurrence, toxicity, and challenges for risk assessment. Water Res. 2019, 151, 488–499. [Google Scholar] [CrossRef]
- Dittmann, E.; Fewer, D.P.; Neilan, B.A. Cyanobacterial toxins: Biosynthetic routes and evolutionary roots. FEMS Microbiol. Rev. 2013, 37, 23–43. [Google Scholar] [CrossRef]
- Weckesser, J.; Martin, C.; Jakobi, C. Cyanopeptolins, depsipeptides from cyanobacteria. Syst. Appl. Microbiol. 1996, 19, 133–138. [Google Scholar] [CrossRef]
- Agrawal, M.K.; Zitt, A.; Bagchi, D.; Weckesser, J.; Bagchi, S.N.; Von Elert, E. Characterization of proteases in guts of Daphnia magna and their inhibition by Microcystis aeruginosa PCC 7806. Environ. Toxicol. 2005, 20, 314–322. [Google Scholar] [CrossRef]
- Available online: https://research.pasteur.fr/en/team/collection-of-cyanobacteria/ (accessed on 17 July 2019).
- Dittmann, E.; Neilan, B.A.; Erhard, M.; Von Döhren, H.; Börner, T. Insertional mutagenesis of a peptide synthetase gene that is responsible for hepatotoxin production in the cyanobacterium Microcystis aeruginosa PCC 7806. Mol. Microbiol. 2003, 26, 779–787. [Google Scholar] [CrossRef]
- Bittencourt-Oliveira, M.; do, C.; Chia, M.A.; de Oliveira, H.S.B.; Cordeiro Araújo, M.K.; Molica, R.J.R.; Dias, C.T.S. Allelopathic interactions between microcystin-producing and non-microcystin-producing cyanobacteria and green microalgae: Implications for microcystins production. J. Appl. Phycol. 2014, 27, 275–284. [Google Scholar] [CrossRef]
- Harel, M.; Weiss, G.; Lieman-Hurwitz, J.; Gun, J.; Lev, O.; Lebendiker, M.; Temper, V.; Block, C.; Sukenik, A.; Zohary, T.; et al. Interactions between Scenedesmus and Microcystis may be used to clarify the role of secondary metabolites. Environ. Microbiol. Rep. 2013, 5, 97–104. [Google Scholar] [CrossRef]
- Aminot, A.; Chaussepied, M. Manuel de Analyses Chimiques en Milieu Marin; Centre National Pour L’Exploitation des Océans: Paris, France, 1983; p. 395. [Google Scholar]
- Henriksen, P.; Carmichael, W.W.; An, J.; Moestrup, O. Detection of an anatoxin-a(s)-like anticholinesterase in natural blooms and cultures of cyanobacteria/blue-green algae from Danish lakes and in the stomach contents of poisoned birds. Toxicon 1997, 35, 901–913. [Google Scholar] [CrossRef]
- Murphy, J.; Riley, J.P. A modified single solution method for the determination of phosphate in natural waters. Anal. Chim. Acta. 1962, 27, 31–36. [Google Scholar] [CrossRef]
- Schreiber, U.; Bilger, W.; Hormann, H.; Neubauer, C. Chlorophyll fluorescence as diagnostic tool: Basics and some aspects of practical relevance. In Photosynthesis: A Comprehensive Treatise; Raghavendra, A.S., Ed.; Cambridge University Press: Cambridge, UK, 1998; pp. 320–336. [Google Scholar]
- Eilers, P.H.C.; Peeters, J.C.H. A model for the relationship between light intensity and the rate of photosynthesis in phytoplankton. Ecol. Model. 1988, 42, 199–215. [Google Scholar] [CrossRef]
- Sadler, T.; von Elert, E. Dietary exposure of Daphnia to microcystins: No in vivo relevance of biotransformation. Aquat. Toxicol. 2014, 150, 73–82. [Google Scholar] [CrossRef]
- Mayumi, T.; Kato, H.; Imanishi, S.; Kawasaki, Y.; Hasegawa, M.; Harada, K.I. Structural characterization of microcystins by LC/MS/MS under ion trap conditions. J. Antibiot. 2006, 59, 710–719. [Google Scholar] [CrossRef]
- Martin, C.; Oberer, L.; Buschdtt, M.; Weckesser, J. Cyanopeptolins, new depsipeptides from the cyanobacterium Microcystis sp. PCC 7806. J. Antibiot. 1993, 46, 1550–1556. [Google Scholar] [CrossRef]
- Portmann, C.; Blom, J.F.; Kaiser, M.; Brun, R.; Jüttner, F.; Gademann, K.; Jüttner, F.; Gademann, K. Isolation of aerucyclamides C and D and structure revision of microcyclamide 7806A: Heterocyclic ribosomal peptides from Microcystis aeruginosa PCC 7806 and their antiparasite evaluation. J. Nat. Prod. 2008, 71, 1891–1896. [Google Scholar] [CrossRef]
- Portmann, C.; Blom, J.F.; Gademann, K.; Jüttner, F.; Jüttner, F.; Jüttner, F. Aerucyclamides A and B: Isolation and synthesis of toxic ribosomal heterocyclic peptides from the cyanobacterium Microcystis aeruginosa PCC 7806. J. Nat. Prod. 2008, 71, 1193–1196. [Google Scholar] [CrossRef]
- Fastner, J.; Erhard, M.; Von Döhren, H. Determination of Oligopeptide Diversity within a Natural Population of Microcystis spp. (Cyanobacteria) by Typing Single Colonies by Matrix-Assisted Laser Desorption Ionization-Time of Flight Mass Spectrometry. Appl. Environ. Microbiol. 2001, 67, 5069–5076. [Google Scholar] [CrossRef]
- Simonian, M.H.; Capp, M.W. Purification of poly(A)-messenger ribonucleic acid by reversed-phase high-performance liquid chromatography. J. Chromatogr. A 1983, 266, 351–358. [Google Scholar] [CrossRef]


| Day 0 | Day 1 | Day 3 | Day 5 | Day 7 | |
|---|---|---|---|---|---|
| Low Density DM (2w) Experiment | |||||
| Control MC+/Control MC- | NS | * | NS | NS | NS |
| low density DM (2w) MC+/low density DM (2w) MC- | NS | NS | NS | ** | *** |
| Control MC+/low density DM (2w) MC+ | + | + | ++ | NS | NS |
| Control MC-/low density DM (2w) MC- | NS | NS | NS | NS | *** |
| High Density DM (24 h) Experiment | |||||
| Control MC+/Control MC- | NS | NS | NS | NS | NS |
| high density DM (24 h) MC+/high density DM (24 h) MC- | ** | ** | *** | NS | ¤¤ |
| Control MC+/low density DM (24 h) MC+ | NS | ++ | ++ | +++ | ++ |
| Control MC-/low density DM (24 h) MC- | NS | NS | NS | NS | NS |
| Low Density DM (2w) Experiment | Day 0 | Day 3 | Day 7 | Day 0 | Day 3 | Day 7 |
|---|---|---|---|---|---|---|
| CP A Intracellular | AC D Intracellular | |||||
| Control MC+/Control MC- | NS | * | * | NS | ** | * |
| low density DM (2w) MC+/ low density DM (2w) MC- | NS | ** | NS | NS | NS | NS |
| Control MC+/ low density DM (2w) MC+ | NS | ++ | NS | NS | ++ | NS |
| Control MC-/ low density DM (2w) MC- | NS | NS | NS | NS | NS | NS |
| CP A Extracellular | AC D Extracellular | |||||
| Control MC+/Control MC- | NS | ** | ** | ¤ | ** | NS |
| low density DM (2w) MC+/ low density DM (2w) MC- | ** | ** | NS | NS | *** | NS |
| Control MC+/ low density DM (2w) MC+ | NS | NS | + | NS | NS | NS |
| Control MC-/ low density DM (2w) MC- | NS | NS | NS | NS | NS | NS |
| MC-LR Intracellular | des-MC-LR Intracellular | |||||
| Control MC+/ low density DM (2w) MC+ | NS | ++ | NS | NS | ++ | NS |
| MC-LR Extracellular | des-MC-LR Extracellular | |||||
| Control MC+/ low density DM (2w) MC+ | NS | NS | NS | NS | NS | NS |
| High Density DM (24 h) Experiment | Day 0 | Day 3 | Day 7 | Day 0 | Day 3 | Day 7 |
|---|---|---|---|---|---|---|
| MC-LR Intracellular | des-MC-LR Intracellular | |||||
| Control MC+/ low density DM (24 h) MC+ | NS | ++ | NS | NS | ++ | ++ |
| MC-LR extracellular | des-MC-LR Extracellular | |||||
| Control MC+/ low density DM (24 h) MC+ | NS | NS | NS | NS | NS | + |
| D. magna Spent Medium Characteristics (One Batch of DM per Daphnia Treatment Group) | Cyanobacterial Cell Density After 1 Week Pre-Cultivation for MC+ and MC- | Cyanobacterial Cell Density at the Beginning of the Experiment for MC+ and MC- |
|---|---|---|
| 75 neonatal individuals of D. magna/L cultivated for 1 week (low density DM (1w)) | 1 × 107 cells/mL | 2 × 106 cells/mL |
| 75 all age individuals of D. magna/L cultivated for 2 weeks (low density DM (2w)) | 1 × 107 cells/mL | 2 × 106 cells/mL |
| 200 all age individuals of D. magna/L cultivated for 24 h (high density DM (24 h)) | 2 × 106 cells/mL | 5 × 105 cells/mL |
| Peptide Class | Peptide Sub-Class | m/z [M+H]+ | References |
|---|---|---|---|
| Microcystins | |||
| MC-LR | 996 | [79] | |
| Des-MCLR | 982 | [79] | |
| Cyanopeptolins (CP) | |||
| CP 963A | 947 | [51] | |
| CP A | 958 | [80] | |
| CP B | 930 | [80] | |
| Aerucyclamides (AC) | |||
| AC C | 517 | [81] | |
| AC B | 533 | [82] | |
| AC A | 535 | [82] | |
| AC D | 587 | [81] | |
| Aeruginosins | |||
| Aeruginosin 684 | 685 | [36] | |
| Aeruginosin 602 | 603 | [83] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bojadzija Savic, G.; Edwards, C.; Briand, E.; Lawton, L.; Wiegand, C.; Bormans, M. Daphnia magna Exudates Impact Physiological and Metabolic Changes in Microcystis aeruginosa. Toxins 2019, 11, 421. https://doi.org/10.3390/toxins11070421
Bojadzija Savic G, Edwards C, Briand E, Lawton L, Wiegand C, Bormans M. Daphnia magna Exudates Impact Physiological and Metabolic Changes in Microcystis aeruginosa. Toxins. 2019; 11(7):421. https://doi.org/10.3390/toxins11070421
Chicago/Turabian StyleBojadzija Savic, Gorenka, Christine Edwards, Enora Briand, Linda Lawton, Claudia Wiegand, and Myriam Bormans. 2019. "Daphnia magna Exudates Impact Physiological and Metabolic Changes in Microcystis aeruginosa" Toxins 11, no. 7: 421. https://doi.org/10.3390/toxins11070421
APA StyleBojadzija Savic, G., Edwards, C., Briand, E., Lawton, L., Wiegand, C., & Bormans, M. (2019). Daphnia magna Exudates Impact Physiological and Metabolic Changes in Microcystis aeruginosa. Toxins, 11(7), 421. https://doi.org/10.3390/toxins11070421






