Tissue Accumulations of Toxic Aconitum Alkaloids after Short-Term and Long-Term Oral Administrations of Clinically Used Radix Aconiti Lateralis Preparations in Rats
Abstract
:1. Introduction
2. Results
2.1. LC/MS/MS Method Development and Validation for Simultaneous Determination of Six Toxic Aconitum Alkaloids in Different Biological Matrix
2.1.1. Optimization of Chromatographic and Mass Conditions
2.1.2. Optimization of Solid Phase Extraction (SPE) Conditions for Sample Treatment
2.1.3. Method Validation
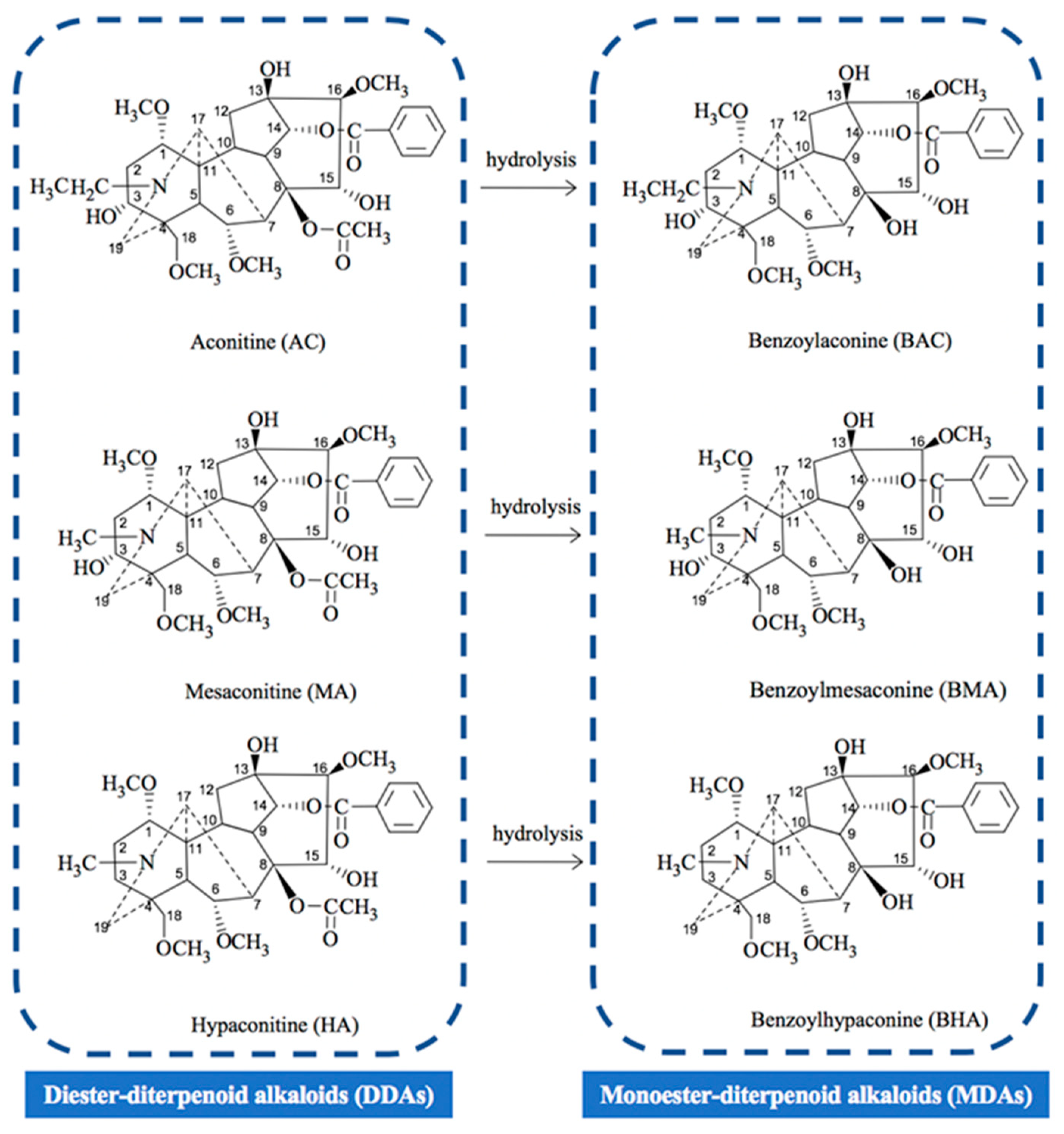
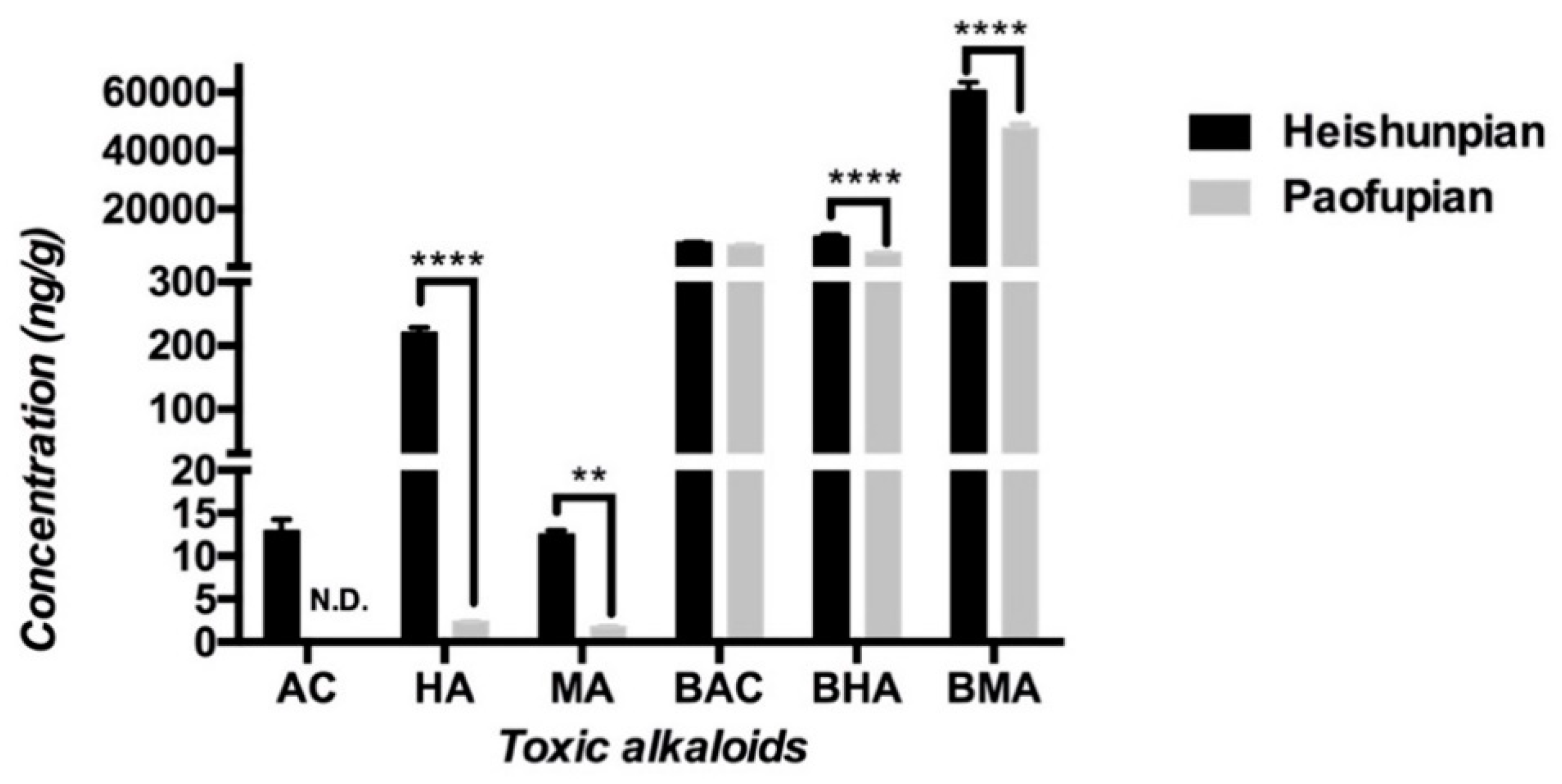
2.2. Content of Six Toxic Aconitum Alkaloids in Studied Radix Aconiti Lateralis Preparation
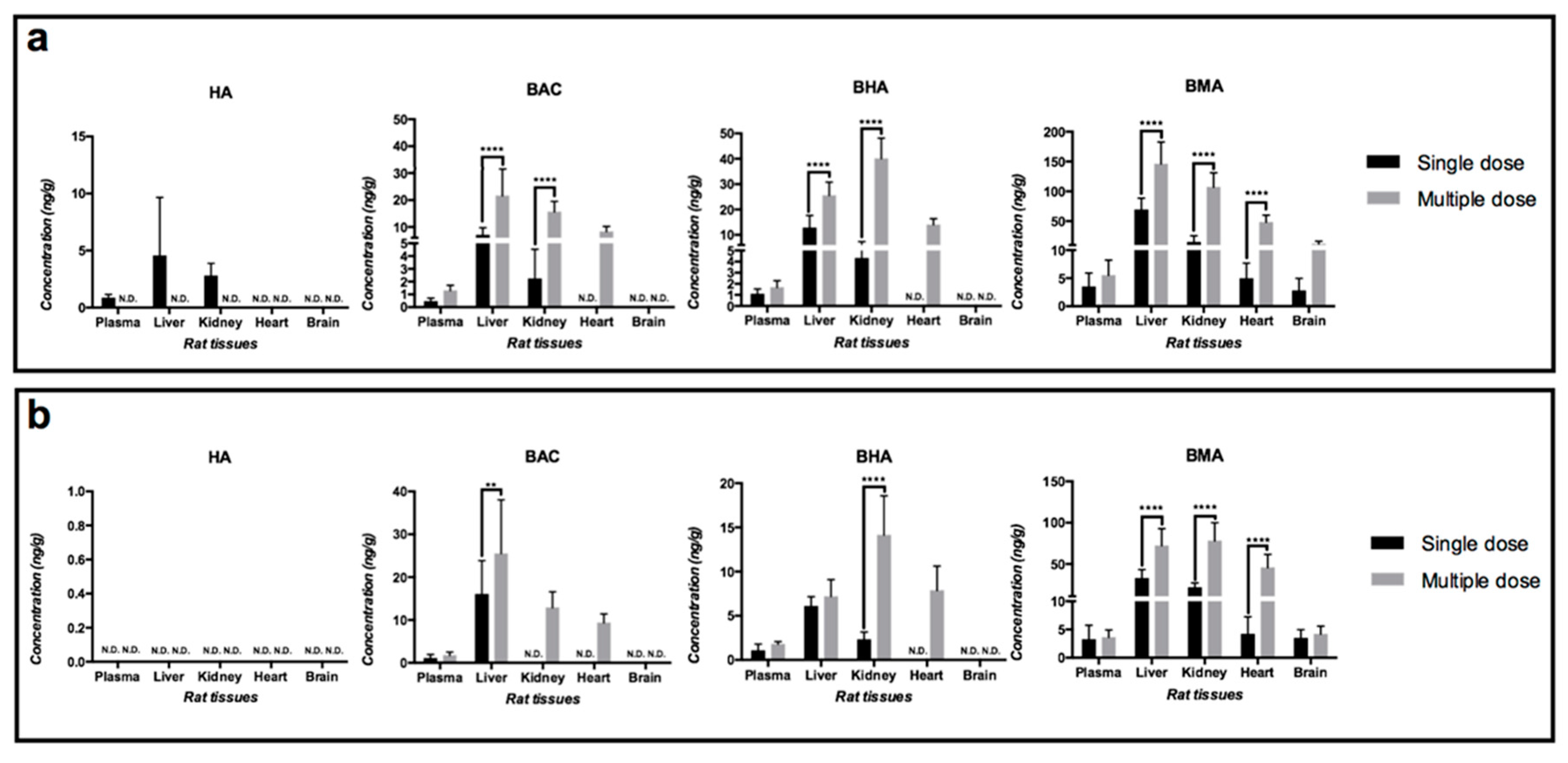
2.3. Biodistributions of Toxic Aconitum Alkaloids after Oral Administrations of the Studied Radix Aconiti Lateralis Preparations
2.4. Comparison of Dose-Normalized Toxic Aconitum Alkaloid Contents in Rat Plasma, Urine and Major Organs
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Materials, Reagents and Animals
5.2. Chromatographic and Mass Spectrometric Conditions
5.3. Preparation of Standard Solution and Quality Control Samples
5.4. Sample Preparations
5.5. LC/MS/MS Method Validation
5.5.1. Specificity
5.5.2. Linearity and Sensitivity
5.5.3. Accuracy and Precision
5.5.4. Recovery and Matrix Effect
5.5.5. Stability
5.6. Application of the Developed LC/MS/MS Method for Biodistribution Study of Radix Aconiti Lateralis Preparations in Rats
5.6.1. Quality Control of the Studied Radix Aconiti Lateralis Preparations
5.6.2. Animal Studies
5.7. Data Analyses
Author Contributions
Funding
Conflicts of Interest
Appendix A
| Tissue | Heart | Liver | Brain | Urine | Kidney | Plasma | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Compound | LLOQ ng/mL | RSD % | RE % | Linear Range ng/mL | LLOQ ng/mL | RSD % | RE % | Linear Range ng/mL | LLOQ ng/mL | RSD % | RE % | Linear Range ng/mL | LLOQ ng/mL | RSD % | RE % | Linear Range ng/mL | LLOQ ng/mL | RSD % | RE % | Linear Range ng/mL | LLOQ ng/mL | RSD % | RE % | Linear Range ng/mL |
| AC | 1 | 16.24 | −2.06 | 1–20 | 2 | 5.12 | −0.41 | 2–20 | 2 | 3.05 | 2.35 | 2–100 | 2 | 3.88 | 5.4 | 1–200 | 1 | 1.46 | 1.49 | 1–50 | 0.5 | 4.74 | 16.16 | 0.5–100 |
| HA | 1 | 12.14 | −2.93 | 1–20 | 1 | 1.7 | 6.19 | 1–20 | 2 | 7.81 | 0.39 | 2–100 | 0.5 | 6.87 | 7.27 | 0.5–200 | 0.5 | 2.66 | 2.44 | 0.5–50 | 0.5 | 7.30 | 5.84 | 0.5–100 |
| MA | 1 | 8.32 | 0.01 | 1–20 | 1 | 1.74 | 17.21 | 1–20 | 2 | 7.53 | 0.23 | 2–100 | 0.5 | 9.48 | 0.47 | 0.5–200 | 0.5 | 12.03 | −8.39 | 0.5–50 | 0.5 | 9.89 | 5.79 | 0.5–100 |
| BAC | 1 | 13.46 | 0.11 | 1–20 | 1 | 6.42 | 9.14 | 1–20 | 2 | 5.87 | −7.78 | 2–100 | 0.5 | 1.67 | −2.25 | 0.5–200 | 0.5 | 9.32 | 2.67 | 0.5–50 | 0.5 | 5.72 | 5.89 | 0.5–100 |
| BHA | 1 | 2.27 | 7.05 | 1–20 | 1 | 3.89 | 10.02 | 1–20 | 2 | 10.18 | −5.5 | 2–100 | 0.5 | 5.1 | −12.24 | 0.5–200 | 0.5 | 17.27 | 1.1 | 0.5–50 | 0.5 | 1.18 | −1.64 | 0.5–100 |
| BMA | 1 | 4.99 | 11.42 | 1–20 | 1 | 5.39 | 2.83 | 1–20 | 2 | 11.83 | −1.03 | 2–100 | 2 | 3.13 | −4.32 | 2–200 | 0.5 | 10.62 | −0.05 | 0.5–50 | 0.5 | 1.22 | 5.52 | 0.5–100 |
| Analytes | Concentration (ng/mL) | Intra-Day (%, RSD) | Inter-Day (%, RSD) | Accuracy (%, RE) | Absolute Recovery (%, Mean ± SD) | Matrix Effect (%, RSD) | Stability (%, Mean ± SD) | |||
|---|---|---|---|---|---|---|---|---|---|---|
| 4 h at Room Temperature | 12 h at 8 °C | 3 Freeze-Thaw Cycles | 30 Days at –80 °C | |||||||
| AC | 2 | 11.21 | 5.32 | 2.11 | 48.83 ± 11.47 | 10.04 | 66.64 ± 12.85 | 71.12 ± 9.60 | 105.41 ± 12.97 | 69.46 ± 0.48 |
| 5 | 0.04 | 1.55 | 0.03 | 36.19 ± 4.35 | 3.81 | N.A. | N.A. | N.A. | N.A. | |
| 10 | 0.08 | 3.99 | 0.95 | 35.58 ± 1.53 | 8.72 | 88.50 ± 10.76 | 72.77 ± 7.56 | 109.17 ± 4.33 | 129.82 ± 5.89 | |
| HA | 2 | 11.25 | 1.31 | −11.08 | 52.47 ± 4.89 | 11.25 | 80.32 ± 4.42 | 97.99 ± 14.73 | 101.72 ± 14.32 | 94.47 ± 6.18 |
| 5 | 6.54 | 4.67 | 2.95 | 48.65 ± 2.96 | 6.54 | N.A. | N.A. | N.A. | N.A. | |
| 10 | 10.52 | 8.86 | −13.62 | 34.06 ± 1.17 | 9.89 | 92.43 ± 9.29 | 75.79 ± 9.84 | 107.78 ± 7.09 | 127.00 ± 5.00 | |
| MA | 2 | 8.71 | 6.65 | −3.11 | 27.08 ± 3.41 | 9.26 | 68.43 ± 3.93 | 90.60 ± 13.11 | 98.31 ± 7.88 | 103.54 ± 1.54 |
| 5 | 9.37 | 7.02 | 0.78 | 22.02 ± 0.91 | 8.86 | N.A. | N.A. | N.A. | N.A. | |
| 10 | 10.92 | 2.07 | −1.19 | 15.93 ± 0.10 | 11.42 | 82.54 ± 9.02 | 80.64 ± 8.44 | 94.76 ± 1.83 | 125.96 ± 4.38 | |
| BAC | 2 | 11.06 | 3.06 | 1.76 | 55.06 ± 9.84 | 11.06 | 104.47 ± 10.69 | 146.84 ± 11.79 | 86.81 ± 5.92 | 86.34 ± 6.75 |
| 5 | 7.74 | 5.39 | –6.22 | 36.39 ± 4.63 | 7.74 | N.A. | N.A. | N.A. | N.A. | |
| 10 | 5.17 | 6.85 | 2.49 | 29.75 ± 2.12 | 5.17 | 115.46 ± 6.90 | 157.93 ± 13.13 | 111.46 ± 7.11 | 105.17 ± 2.94 | |
| BHA | 2 | 7.2 | 7.3 | 1.22 | 38.20 ± 7.50 | 7.2 | 102.36 ± 3.86 | 146.72 ± 5.14 | 94.32 ± 11.40 | 89.70 ± 2.61 |
| 5 | 9.19 | 7.8 | 9.41 | 25.50 ± 2.69 | 9.19 | N.A. | N.A. | N.A. | N.A. | |
| 10 | 6.24 | 3.76 | 2.16 | 19.91 ± 1.68 | 6.24 | 86.60 ± 10.07 | 131.51 ± 3.44 | 97.57 ± 6.87 | 105.87 ± 1.33 | |
| BMA | 2 | 5.56 | 7.73 | −10.38 | 59.61 ± 4.38 | 5.14 | 92.84 ± 5.27 | 185.65 ± 14.98 | 118.67 ± 6.02 | 69.01 ± 4.47 |
| 5 | 11 | 6.58 | 10.14 | 60.62 ± 1.69 | 13.23 | N.A. | N.A. | N.A. | N.A. | |
| 10 | 6.59 | 2.29 | −0.44 | 51.37 ± 2.95 | 6.59 | 78.04 ± 5.01 | 113.08 ± 5.77 | 73.44 ± 4.82 | 139.14 ± 10.84 | |
| Analytes | Concentration (ng/mL) | Intra-Day (%, RSD) | Inter-Day (%, RSD) | Accuracy (%, RE) | Absolute Recovery (%, Mean ± SD) | Matrix Effect (%, RSD) | Stability (%, Mean ± SD) | |||
|---|---|---|---|---|---|---|---|---|---|---|
| 4 h at Room Temperature | 12 h at 8 °C | 3 Freeze-Thaw Cycles | 30 Days at −80 °C | |||||||
| AC | 5 | 10.83 | 9.21 | −0.74 | 16.27 ± 1.76 | 10.83 | 109.37 ± 9.10 | 95.40 ± 4.17 | 119.10 ± 11.54 | 123.35 ± 14.20 |
| 10 | 4.2 | 2.04 | 2.89 | 15.44 ± 0.65 | 4.2 | N.A. | N.A. | N.A. | N.A. | |
| 50 | 1.8 | 0.06 | 0.6 | 16.85 ± 0.30 | 1.8 | 88.31 ± 5.74 | 99.15 ± 2.97 | 91.81 ± 2.92 | 98.03 ± 14.28 | |
| HA | 5 | 6.17 | 5.05 | 1.36 | 17.48 ± 1.08 | 6.17 | 101.74 ± 2.56 | 91.27 ± 6.35 | 108.72 ± 3.18 | 89.69 ± 4.36 |
| 10 | 7.85 | 0.08 | 0.07 | 17.00 ± 1.33 | 7.85 | N.A. | N.A. | N.A. | N.A. | |
| 50 | 1.75 | 0.06 | −3.88 | 18.35 ± 0.32 | 1.75 | 106.79 ± 1.71 | 95.52 ± 2.14 | 110.29 ± 4.89 | 95.45 ± 13.53 | |
| MA | 5 | 6.08 | 3 | −2.47 | 16.46 ± 1.00 | 6.08 | 101.76 ± 0.57 | 93.24 ± 4.46 | 120.36 ± 6.72 | 92.81 ± 10.90 |
| 10 | 2.75 | 8.07 | -0.01 | 15.78 ± 0.43 | 2.75 | N.A. | N.A. | N.A. | N.A. | |
| 50 | 3.52 | 0.05 | 0.14 | 16.94 ± 0.60 | 3.52 | 94.11 ± 4.29 | 96.41 ± 3.02 | 99.94 ± 4.80 | 92.27 ± 12.05 | |
| BAC | 5 | 9.37 | 2.81 | −1.07 | 2.64 ± 0.25 | 9.37 | 138.93 ± 7.85 | 85.36 ± 8.33 | 141.29 ± 11.86 | 127.23 ± 9.12 |
| 10 | 8.85 | 0.53 | 2.23 | 3.38 ± 0.30 | 8.85 | N.A. | N.A. | N.A. | N.A. | |
| 50 | 2.65 | 3.04 | −0.23 | 2.96 ± 0.08 | 2.65 | 97.68 ± 5.58 | 97.81 ± 4.03 | 100.15 ± 4.94 | 106.27 ± 6.59 | |
| BHA | 5 | 5.69 | 6.67 | 9.08 | 2.60 ± 0.15 | 5.69 | 141.14 ± 9.36 | 93.21 ± 7.16 | 161.20 ± 10.89 | 137.34 ± 12.78 |
| 10 | 11.98 | 5.63 | −0.17 | 4.22 ± 0.51 | 11.98 | N.A. | N.A. | N.A. | N.A. | |
| 50 | 3.61 | 3.99 | −5.59 | 3.12 ± 0.11 | 3.61 | 106.85 ± 3.50 | 95.41 ± 2.42 | 112.54 ± 3.40 | 140.18 ± 14.86 | |
| BMA | 5 | 5.51 | 2.22 | 6.12 | 2.47 ± 0.14 | 5.51 | 104.19 ± 9.20 | 87.37 ± 3.96 | 113.15 ± 9.93 | 100.35 ± 11.19 |
| 10 | 5.84 | 4.62 | −3.09 | 2.71 ± 0.16 | 5.84 | N.A. | N.A. | N.A. | N.A. | |
| 50 | 2.78 | 5.54 | −10.32 | 2.36 ± 0.07 | 2.78 | 106.43 ± 5.48 | 93.28 ± 4.63 | 93.12 ± 0.68 | 86.70 ± 6.88 | |
| Analytes | Concentration (ng/mL) | Intra-Day (%, RSD) | Inter-Day (%, RSD) | Accuracy (%, RE) | Absolute Recovery (%, Mean ± SD) | Matrix Effect (%, RSD) | Stability (%, Mean ± SD) | |||
|---|---|---|---|---|---|---|---|---|---|---|
| 4 h at Room Temperature | 12 h at 8 °C | 3 Freeze-Thaw Cycles | 30 Days at −80 °C | |||||||
| AC | 2 | 5.55 | 15.2 | −1.88 | 27.48 ± 1.53 | 5.55 | 95.56 ± 10.84 | 100.24 ± 12.29 | 105.91 ± 5.19 | 114.35 ± 9.72 |
| 5 | 7.87 | 6.76 | −1.72 | 34.63 ± 2.73 | 7.87 | N.A. | N.A. | N.A. | N.A. | |
| 10 | 8.97 | 3.06 | −1.17 | 32.03 ± 2.87 | 8.97 | 98.68 ± 6.63 | 105.49 ± 7.80 | 101.30 ± 4.77 | 111.57 ± 11.56 | |
| HA | 2 | 4.35 | 15.2 | 0.17 | 33.56 ± 1.46 | 4.35 | 93.24 ± 5.68 | 100.36 ± 7.15 | 98.89 ± 3.59 | 97.16 ± 6.95 |
| 5 | 2.9 | 2.02 | −4.82 | 31.15 ± 0.90 | 2.9 | N.A. | N.A. | N.A. | N.A. | |
| 10 | 4.34 | 1.51 | 3.29 | 33.46 ± 1.45 | 4.34 | 104.40 ± 3.42 | 104.03 ± 5.74 | 101.13 ± 4.34 | 104.66 ± 8.08 | |
| MA | 2 | 3.82 | 15.19 | 0.42 | 26.02 ± 0.99 | 3.82 | 94.97 ± 8.67 | 100.26 ± 9.16 | 100.46 ± 0.77 | 90.20 ± 10.51 |
| 5 | 3.2 | 1.59 | −13.39 | 23.74 ± 0.76 | 3.2 | N.A. | N.A. | N.A. | N.A. | |
| 10 | 4.65 | 3.26 | −0.49 | 25.60 ± 1.19 | 4.65 | 100.93 ± 5.61 | 104.19 ± 6.67 | 101.38 ± 2.04 | 93.22 ± 7.50 | |
| BAC | 2 | 3.38 | 18.14 | −0.37 | 4.80 ± 0.16 | 3.38 | 109.55 ± 14.53 | 100.56 ± 6.29 | 105.67 ± 18.95 | 225.25 ± 1.44 |
| 5 | 9.9 | 6.8 | −8.87 | 4.46 ± 0.44 | 9.9 | N.A. | N.A. | N.A. | N.A. | |
| 10 | 4.08 | 6.48 | −4.88 | 5.78 ± 0.24 | 4.08 | 103.91 ± 5.44 | 96.30 ± 2.65 | 90.73 ± 6.52 | 184.82 ± 11.92 | |
| BHA | 2 | 3.22 | 7.16 | 3.48 | 5.21 ± 0.17 | 3.22 | 89.92 ± 7.02 | 95.97 ± 8.77 | 86.65 ± 10.79 | 166.17 ± 14.83 |
| 5 | 6.26 | 4.47 | −13.7 | 3.65 ± 0.23 | 6.26 | N.A. | N.A. | N.A. | N.A. | |
| 10 | 4.5 | 4.28 | −1.36 | 7.42 ± 0.33 | 4.5 | 101.38 ± 1.24 | 100.01 ± 6.63 | 88.47 ± 3.88 | 1&7.87 ± 4.76 | |
| BMA | 2 | 9.48 | 15.56 | −1.38 | 4.15 ± 0.39 | 9.48 | 144.55 ± 14.24 | 94.36 ± 16.14 | 98.54 ± 6.93 | 229.74 ± 7.66 |
| 5 | 3.12 | 5.88 | −2.41 | 3.97 ± 0.12 | 3.12 | N.A. | N.A. | N.A. | N.A. | |
| 10 | 3.52 | 2.54 | −3.12 | 4.74 ± 0.17 | 3.52 | 83.7 ± 2.74 | 105.89 ± 5.97 | 83.46 ± 5.53 | 136.53 ± 5.32 | |
| Analytes | Concentration (ng/mL) | Intra-Day (%, RSD) | Inter-Day (%, RSD) | Accuracy (%, RE) | Absolute Recovery (%, Mean ± SD) | Matrix Effect (%, RSD) | Stability (%, Mean ± SD) | |||
|---|---|---|---|---|---|---|---|---|---|---|
| 4 h at Room Temperature | 12 h at 8 °C | 3 Freeze-Thaw Cycles | 30 Days at −80 °C | |||||||
| AC | 2 | 3.88 | 4.67 | 5.4 | 84.41 ± 4.15 | 3.88 | 87.05 ± 13.58 | 126.77 ± 4.77 | 79.91 ± 12.50 | 73.12 ± 7.69 |
| 10 | 10.24 | 11.5 | −2.94 | 93.28 ± 5.62 | 10.24 | N.A. | N.A. | N.A. | N.A. | |
| 50 | 2.7 | 1.87 | 0.31 | 98.79 ± 3.53 | 2.7 | 117.59 ± 8.17 | 101.92 ± 6.69 | 115.14 ± 5.79 | 109.04 ± 3.36 | |
| HA | 2 | 6.11 | 3.87 | 3.15 | 100.06 ± 1.70 | 6.11 | 76.89 ± 5.39 | 95.39 ± 4.69 | 84.95 ± 1.67 | 88.36 ± 2.51 |
| 10 | 6.21 | 7.69 | −10.93 | 94.11 ± 1.80 | 6.21 | N.A. | N.A. | N.A. | N.A. | |
| 50 | 3.98 | 1.62 | 1.63 | 101.24 ± 3.59 | 3.98 | 113.66 ± 2.41 | 98.69 ± 1.66 | 129.99 ± 1.85 | 107.34 ± 0.85 | |
| MA | 2 | 3.5 | 5.74 | 9.99 | 96.95 ± 3.15 | 3.5 | 80.31 ± 1.09 | 97.99 ± 4.14 | 81.96 ± 0.65 | 82.86 ± 1.14 |
| 10 | 4.12 | 6.04 | −7.28 | 92.25 ± 2.45 | 4.12 | N.A. | N.A. | N.A. | N.A. | |
| 50 | 6.59 | 3.77 | 0.46 | 98.47 ± 3.07 | 6.59 | 111.56 ± 0.51 | 98.19 ± 2.31 | 119.99 ± 1.65 | 103.40 ± 3.53 | |
| BAC | 2 | 3.73 | 6.12 | 9.14 | 106.46 ± 2.39 | 3.73 | 89.96 ± 13.19 | 100.22 ± 10.17 | 85.13 ± 11.07 | 81.30 ± 6.12 |
| 10 | 7.11 | 3 | 0.47 | 99.19 ± 0.80 | 7.11 | N.A. | N.A. | N.A. | N.A. | |
| 50 | 3.3 | 11.7 | 2.59 | 103.41 ± 1.48 | 3.3 | 102.55 ± 2.13 | 122.66 ± 4.15 | 111.31 ± 0.85 | 105.31 ± 3.33 | |
| BHA | 2 | 3.81 | 8.28 | 5.11 | 98.27 ± 2.68 | 3.81 | 76.31 ± 8.32 | 99.16 ± 6.30 | 94.79 ± 3.84 | 96.08 ± 4.23 |
| 10 | 5.89 | 4.05 | −1.71 | 96.69 ± 1.86 | 5.89 | N.A. | N.A. | N.A. | N.A. | |
| 50 | 7.29 | 6.7 | −1.64 | 100.71 ± 2.40 | 7.29 | 96.92 ± 1.62 | 116.89 ± 2.81 | 135.02 ± 1.75 | 101.46 ± 2.66 | |
| BMA | 10 | 4.99 | 7.41 | 13.21 | 100.05 ± 2.61 | 4.99 | 83.77 ± 4.06 | 103.23 ± 4.57 | 92.87 ± 11.00 | 21.21 ± 3.08 |
| 50 | 8.53 | 8.53 | 0.27 | 104.94 ± 1.96 | 8.53 | N.A. | N.A. | N.A. | N.A. | |
| 100 | 6.52 | 6.52 | 3.07 | 110.31 ± 3.53 | 6.52 | 109.94 ± 1.16 | 126.82 ± 1.82 | 103.55 ± 5.31 | 90.84 ± 0.72 | |
| Analytes | Concentration (ng/mL) | Intra-Day (%, RSD) | Inter-Day (%, RSD) | Accuracy (%, RE) | Absolute Recovery (%, Mean ± SD) | Matrix Effect (%, RSD) | Stability (%, Mean ± SD) | |||
|---|---|---|---|---|---|---|---|---|---|---|
| 4 h at Room Temperature | 12 h at 8 °C | 3 Freeze-Thaw Cycles | 30 Days at –80 °C | |||||||
| AC | 2 | 6.61 | 5.34 | −2.34 | 71.28 ± 4.71 | 6.61 | 101.30 ± 7.24 | 100.54 ± 0.29 | 112.76 ± 9.08 | 164.14±8.87 |
| 5 | 3.98 | 8.03 | -6.10 | 67.46 ± 2.69 | 3.98 | N.A. | N.A. | N.A. | N.A. | |
| 20 | 5.79 | 3.74 | 5.20 | 82.80 ± 4.80 | 5.79 | 100.21 ± 3.55 | 88.89 ± 4.26 | 108.61 ± 3.01 | 108.22 ± 0.03 | |
| HA | 2 | 3.23 | 5.56 | −9.89 | 67.00 ± 2.16 | 3.23 | 125.20 ± 7.65 | 100.01 ± 3.38 | 119.58 ± 1.07 | 118.18±7.50 |
| 5 | 2.19 | 8.19 | −14.04 | 59.65 ± 0.53 | 0.88 | N.A. | N.A. | N.A. | N.A. | |
| 20 | 4.03 | 6.06 | −1.04 | 69.54 ± 2.80 | 4.03 | 133.03 ± 4.53 | 98.81 ± 4.70 | 121.92 ± 5.95 | 114.49 ± 2.00 | |
| MA | 2 | 5.31 | 5.57 | −13.57 | 71.34 ± 3.79 | 5.31 | 118.52 ± 11.80 | 80.92 ± 3.75 | 103.79 ± 1.63 | 99.84 ± 5.24 |
| 5 | 2.00 | 2.05 | −6.56 | 88.82 ± 1.78 | 2.00 | N.A. | N.A. | N.A. | N.A. | |
| 20 | 3.74 | 5.04 | 8.29 | 92.04 ± 3.45 | 3.74 | 115.71 ± 4.75 | 87.06 ± 4.38 | 106.95 ± 1.82 | 98.85 ± 0.90 | |
| BAC | 2 | 3.24 | 4.98 | −3.11 | 82.85 ± 2.67 | 3.23 | 114.26 ± 6.75 | 113.52±9.71 | 110.02 ± 5.14 | 79.65 ± 3.17 |
| 5 | 3.63 | 7.28 | 2.41 | 111.31 ± 4.04 | 3.63 | N.A. | N.A. | N.A. | N.A. | |
| 20 | 2.55 | 3.27 | −8.37 | 90.32 ± 2.31 | 2.55 | 123.28 ± 5.74 | 95.37 ± 0.09 | 114.72 ± 2.84 | 103.64 ± 2.26 | |
| BHA | 2 | 7.46 | 7.65 | 9.60 | 80.58 ± 6.01 | 7.46 | 111.19 ± 14.68 | 94.49 ± 3.94 | 108.49 ± 3.62 | 98.50 ± 5.31 |
| 5 | 4.72 | 8.91 | −2.83 | 95.82 ± 4.52 | 4.72 | N.A. | N.A. | N.A. | N.A. | |
| 20 | 4.77 | 5.51 | −6.23 | 87.84 ± 4.19 | 4.77 | 122.77 ± 4.71 | 89.36 ± 1.46 | 112.25 ± 3.59 | 102.16 ± 2.74 | |
| BMA | 2 | 5.25 | 14.89 | 8.22 | 68.49 ± 1.99 | 2.91 | 109.38 ± 4.75 | 132.63 ± 9.45 | 128.71 ± 14.56 | 48.11 ± 7.51 |
| 5 | 9.42 | 12.40 | 5.92 | 100.65 ± 10.12 | 10.06 | N.A. | N.A. | N.A. | N.A. | |
| 20 | 2.52 | 2.15 | −13.54 | 94.23 ± 2.39 | 2.91 | 117.83 ± 5.66 | 95.58 ± 1.79 | 106.33 ± 5.24 | 98.74 ± 8.44 | |
| Analytes | Concentration (ng/mL) | Intra-Day (%, RSD) | Inter-Day (%, RSD) | Accuracy (%, RE) | Absolute Recovery (%, Mean ± SD) | Matrix Effect (%, SD) | Stability (%, Mean ± SD) | |||
|---|---|---|---|---|---|---|---|---|---|---|
| 4 h at Room Temperature | 12 h at 8 °C | 3 Freeze-Thaw Cycles | 30 Days at −80 °C | |||||||
| AC | 1 | 1.46 | 11.68 | 1.49 | 90.67 ± 9.23 | 3.20 | 102.31 ± 3.11 | 53.29 ± 5.38 | 106.24 ± 8.01 | 89.62 ± 7.12 |
| 5 | 10.43 | 10.43 | 5.16 | 79.64 ± 2.48 | 0.45 | N.A. | N.A. | N.A. | N.A. | |
| 20 | 6.53 | 6.53 | 4.7 | 81.40 ± 10.34 | 1.85 | 63.75 ± 5.51 | 44.14 ± 5.58 | 75.04 ± 6.90 | 85.86 ± 5.49 | |
| HA | 1 | 5.08 | 10.02 | 5.77 | 76.99 ± 8.35 | 1.60 | 53.22 ± 2.99 | 70.71 ± 0.52 | 47.33 ± 3.15 | 88.69 ± 8.19 |
| 5 | 5.22 | 10.64 | −1.03 | 79.36 ± 6.23 | 1.07 | N.A. | N.A. | N.A. | N.A. | |
| 20 | 0.46 | 1.38 | 0.64 | 92.53 ± 12.25 | 2.07 | 65.23 ± 3.45 | 82.41 ± 1.36 | 38.14 ± 1.10 | 87.13 ± 1.33 | |
| MA | 1 | 5.28 | 10.12 | 8.75 | 87.61 ± 5.49 | 0.95 | 74.54 ± 1.05 | 47.25 ± 5.35 | 92.18 ± 5.12 | 98.80 ± 5.34 |
| 5 | 4.78 | 11.87 | −4.37 | 78.46 ± 5.63 | 0.87 | N.A. | N.A. | N.A. | N.A. | |
| 20 | 2.18 | 1.77 | 1.86 | 98.72 ± 12.45 | 1.74 | 68.62 ± 2.81 | 34.29 ± 2.14 | 84.91 ± 0.92 | 107.02 ± 2.69 | |
| BAC | 1 | 9.8 | 12.28 | 1.64 | 101.51 ± 10.51 | 1.12 | 106.14 ± 5.48 | 49.46 ± 4.12 | 120.73 ± 8.11 | 103.41 ± 5.29 |
| 5 | 7.15 | 7.47 | −3.42 | 91.28 ± 14.16 | 1.26 | N.A. | N.A. | N.A. | N.A. | |
| 20 | 8.65 | 1.46 | 0.91 | 86.44 ± 13.89 | 1.21 | 108.06 ± 12.04 | 56.57 ± 3.82 | 127.04 ± 4.56 | 87.07 ± 5.42 | |
| BHA | 1 | 6.11 | 6.53 | 1.63 | 96.88 ± 14.86 | 1.66 | 93.91 ± 13.38 | 63.66 ± 10.36 | 98.44 ± 13.02 | 108.47 ± 2.67 |
| 5 | 9.47 | 4.83 | 0.91 | 72.70 ± 9.55 | 0.98 | N.A. | N.A. | N.A. | N.A. | |
| 20 | 6.29 | 1.76 | 1.32 | 87.82 ± 14.79 | 1.33 | 108.66 ± 2.18 | 46.05 ± 6.01 | 120.96 ± 2.55 | 92.94 ± 3.62 | |
| BMA | 1 | 10.16 | 8.06 | −2.43 | 93.99 ± 11.26 | 1.75 | 79.79 ± 1.43 | 77.59 ± 9.88 | 96.94 ± 10.13 | 97.18 ± 7.90 |
| 5 | 7.74 | 3.58 | 3.52 | 78.27 ± 8.32 | 0.64 | N.A. | N.A. | N.A. | N.A. | |
| 20 | 3.43 | 6.06 | 2.84 | 76.90 ± 13.40 | 1.01 | 97.12 ± 7.38 | 49.49 ± 10.83 | 112.43 ± 2.61 | 82.85 ± 1.04 | |
References
- Chan, T.Y.K. Aconite poisoning. Clin. Toxicol. 2009, 47, 279–285. [Google Scholar] [CrossRef] [PubMed]
- Zhou, G.; Tang, L.; Zhou, X.; Wang, T.; Kou, Z.; Wang, Z. A review on phytochemistry and pharmacological activities of the processed lateral root of Aconitum carmichaelii Debeaux. J. Ethnopharmacol. 2015, 160, 173–193. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.J. Study on “Shang Han Lun” Dosage and Related Problems. Ph.D. Thesis, Beijing University of Chinese Medicine, Beijing, China, 2007. [Google Scholar]
- He, F.; Wang, C.J.; Xie, Y.; Cheng, C.S.; Liu, Z.Q.; Liu, L.; Zhou, H. Simultaneous quantification of nine aconitum alkaloids in Aconiti Lateralis Radix Praeparata and related products using UHPLC-QQQ-MS/MS. Sci. Rep. 2017, 7, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Alert on Chinese Medicine from Department of Health. Available online: http://www.cmd.gov.hk/text/gb/important_info/alert.html (accessed on 28 May 2019).
- Aconitine Poisoning. Poisoning Watch. Volume 4, No. 1. 2011 from Department of Health. Available online: https://www.chp.gov.hk/files/pdf/poisoning_watch_vol4_num1_20111228.pdf (accessed on 28 May 2019).
- Tong, P.; Wu, C.; Wang, X.; Hu, H.; Jin, H.; Li, C.; Zhu, Y.; Shan, L.; Xiao, L. Development and assessment of a complete-detoxication strategy for Fuzi (lateral root of Aconitum carmichaelii) and its application in rheumatoid arthritis therapy. J. Ethnopharmacol. 2013, 146, 562–571. [Google Scholar] [CrossRef] [PubMed]
- Li, X.J.Y.; Luan, Y.; Sun, R. Comparative Study on Acute Toxicity of Different Components of Aconiti Lateralis Radix Praeparata on Normal Mice. Chin. J. Pharmacovigil. 2013, 10, 583. [Google Scholar]
- Chang, H.M.; But, P.P.; Yao, S.C. Pharmacology and Applications of Chinese Materia Medica; World Scientific: Singapore, 1986; Volume 1. [Google Scholar] [CrossRef]
- Singhuber, J.; Zhu, M.; Prinz, S.; Kopp, B. Aconitum in Traditional Chinese Medicine-A valuable drug or an unpredictable risk? J. Ethnopharmacol. 2009, 126, 18–30. [Google Scholar] [CrossRef] [PubMed]
- Ameri, A. The effects of Aconitum alkaloids on the central nervous system. Prog. Neurobiol. 1998, 56, 211–235. [Google Scholar] [CrossRef]
- Sun, G.B.; Sun, H.; Meng, X.B.; Hu, J.; Zhang, Q.; Liu, B.; Wang, M.; Xu, H.B.; Sun, X.B. Aconitine-induced Ca2+ overload causes arrhythmia and triggers apoptosis through p38 MAPK signaling pathway in rats. Toxicol. Appl. Pharmacol. 2014, 279, 8–22. [Google Scholar] [CrossRef]
- Grishchenko, I.I.; Naumov, A.P.; Zubov, A.N. Gating and selectivity of aconitine-modified sodium channels in neuroblastoma cells. Neuroscience 1983, 9, 549–554. [Google Scholar] [CrossRef]
- Feng, Q.; Li, X.Y.; Luan, Y.F.; Sun, S.N.L.; Sun, R. Study on effect of aqueous extracts from aconite on “dose-time-toxicity” relationships in mice hearts. Zhongguo Zhong Yao Za Zhi 2015, 40, 927–932. [Google Scholar]
- Fu, M.; Wu, M.; Qiao, Y.; Wang, Z. Toxicological mechanisms of Aconitum alkaloids. Pharmazie 2006, 61, 735–741. [Google Scholar] [PubMed]
- Lei, H.C.; Yi, J.H.; Liu, T. Observation of hepatocyte apoptosis induced by aconite poisoning. J. Health Toxicol. 2004, 18, 199–200. [Google Scholar] [CrossRef]
- Lei, H.C.; Yi, J.H. Observation of apoptosis in renal tubule epithelial cell after aconitine poisoning. J. Ind. Health Occup. Dis. 2005, 31, 83–85. [Google Scholar] [CrossRef]
- Lei, H.C.; Xiang, W.C.; Shi, L. Apoptosis in brain nerve cells of aconitine poisoning rats. J. Shandong Med. 2006, 46, 21–22. [Google Scholar]
- Mizugaki, M.; Ito, K. Aconite toxins. In Drugs and Poisons in Humans—A Handbook of Practical Analysis; Suzuki, O., Watanabe, K., Eds.; Springer Verlag Press: New York, NY, USA, 2005; pp. 456–467. [Google Scholar]
- The Compile Commission of Zhonghua Bencao of the State Administration of Traditional Chinese Medicine of the People’s Republic of China. Zhonghua Bencao; Shanghai Science and Technology Press: Shanghai, China, 1999; Volume 5, pp. 101–120.
- Xiao, P.G. New Chinese Materia Medica (Xin Bian Zhong Yao Zhi); Chemical Industry Press: Beijing, China, 2002; Volume 1, pp. 536–541, 645–660. [Google Scholar]
- Lu, X.Q. Zhongyao Paozhi Daquan; Hunan Science and Technology Press: Changsha, China, 1999; pp. 271–274. [Google Scholar]
- Hospital Authority of Hong Kong. Cluster of Aconite Poisoning after Taking Chinese Herbal Medicine. Poisoning Alert. 6 January 2017. Available online: http://www.cmd.gov.hk/html/gb/important_info/resource/Alert_on_herbal_medicine_poisoning_20170106.pdf (accessed on 26 January 2019).
- Yang, M.B.; Ji, X.Y.; Zuo, Z. Relationships between the Toxicities of Radix Aconiti Lateralis Preparata (Fuzi) and the Toxicokinetics of Its Main Diester-Diterpenoid Alkaloids. Toxins 2018, 10, 391. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; Xu, Y.Y.; Wu, Y.Y.; Wu, H.Y.; Wang, Y.; Yuan, L.; Xie, J.B.; Li, Y.B.; Zhang, Y.J. Simultaneous determination of ten Aconitum alkaloids in rat tissues by UHPLC–MS/MS and its application to a tissue distribution study on the compatibility of Heishunpian and Fritillariae thunbergii Bulbus. J. Chromatogr. B 2016, 1033–1034, 242–249. [Google Scholar] [CrossRef] [PubMed]
- Ren, M.Y.; Song, S.; Liang, D.D.; Hou, W.T.; Tan, X.M.; Luo, J.B. Comparative tissue distribution and excretion study of alkaloids from Herba Ephedrae-Radix Aconiti Lateralis extracts in rats. J. Pharm. Biomed. Anal. 2017, 134, 137–142. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.H.; Wang, Z.P.; Wen, J.; He, Y. Simultaneous determination of three aconitum alkaloids in urine by LC-MS-MS. J. Pharm. Biomed. Anal. 2007, 45, 145–148. [Google Scholar] [CrossRef]
- Industry, G. Bioanalytical Method Validation; Center for Drug Evaluation and Research (CDER), Food and Drug Administration, US Department of Health and Human Services: Washington, DC, USA, 2001.
- Song, L.; Zhang, H.; Liu, X.; Zhao, Z.L.; Chen, S.L.; Wang, Z.T.; Xu, H.X. Rapid determination of yunaconitine and related alkaloids in aconites and aconite-containing drugs by ultra high-performance liquid chromatography–tandem mass spectrometry. Biomed. Chromatogr. 2012, 26, 1567–1574. [Google Scholar] [CrossRef]
- Wong, S.K.; Tsui, S.K.; Kwan, S.Y. Analysis of proprietary Chinese medicines for the presence of toxic ingredients by LC/MS/MS. J. Pharm. Biomed. Anal. 2002, 30, 161–170. [Google Scholar] [CrossRef]
- The Chinese Pharmacopoeia Commission. Chinese Pharmacopoeia; China Medical Science Press: Beijing, China, 2015; Volume 1, pp. 191–193. [Google Scholar]
- Ye, L.; Yang, X.S.; Lu, L.L.; Chen, W.Y.; Zeng, S.; Yan, T.M.; Dong, L.N.; Peng, X.J.; Shi, J.; Liu, Z.Q. Monoester-Diterpene Aconitum Alkaloid Metabolism in human Liver Microsomes: Predominant Role of CYP3A4 and CYP3A5. Evid. Based Complement. Altern. Med. 2013, 2013, 941093. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.J.; Lin, N.; Li, F.Y.; Zhang, G.Y.; He, S.G.; Zhu, Y.F.; Ou, R.L.; Li, N.; Liu, S.Q.; Feng, L.Z.; et al. Induction of p-glycoprotein expression and activity by aconitum alkaloids: Implication for clinical drug-drug interactions. Sci. Rep. 2016, 6, 25343. [Google Scholar] [CrossRef] [PubMed]
- Niitsu, H.; Fujita, Y.; Fujita, S.; Kumagai, R.; Takamiya, M.; Aoki, Y.; Dewa, K. Distribution of Aconitum alkaloids in autopsy cases of aconite poisoning. Forensic Sci. Int. 2013, 227, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.P.; Kong, D.Z.; Du, Q.; Zhao, J.; Li, Q.; Zhang, J.H.; Li, T.; Ren, L. A conscious rat model involving bradycardia and hypotension after oral administration: A toxicokinetical study of aconitine. Xenobiotica 2017, 47, 515–525. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J. Pharmacokinetics and Cardiac Distribution of Hypaconitine in Rats. Master’s Thesis, Beijing University of Chinese Medicine, Beijing, China, 2010. [Google Scholar]
- Wang, R. Quality Evaluation of Fuzi and Pharmacokinetic Study of Aconitine. Ph.D. Thesis, Beijing University of Chinese Medicine, Beijing, China, 2007. [Google Scholar]
- Li, W.D.; Ma, C. Determination of Aconitine in Tissues of Rats by HPLC. J. Exp. Tradit. Med. Formulae 2007, 13, 56–58. [Google Scholar] [CrossRef]
- Huang, D.C. Study on Principle of Formation of the School of Warming Yang. Master’s Thesis, Shandong University of Chinese Medicine, Shandong, China, 2009. [Google Scholar]
- Li, W.D.; Ma, C. Tissue distribution of Aconitum alkaloids extracted from Radix aconiti preparata after oral administration to rats. Yao Xue Xue Bao 2005, 40, 539–543. [Google Scholar] [PubMed]
- Zhou, H.; Zhang, P.; Hou, Z.; Xie, J.; Wang, Y.; Yang, B.; Xu, Y.; Li, Y. Research on the relationships between endogenous biomarkers and exogenous toxic substances of acute toxicity in Radix Aconiti. Molecules 2016, 21, 1623. [Google Scholar] [CrossRef]
- Tang, L.; Gong, Y.; Lv, C.; Ye, L.; Liu, L.; Liu, Z.Q. Pharmacokinetics of aconitine as the targeted marker of Fuzi (Aconitum carmichaeli) following single and multiple oral administrations of Fuzi extracts in rat by UPLC/MS/MS. J. Ethnopharmacol. 2012, 141, 736–741. [Google Scholar] [CrossRef]
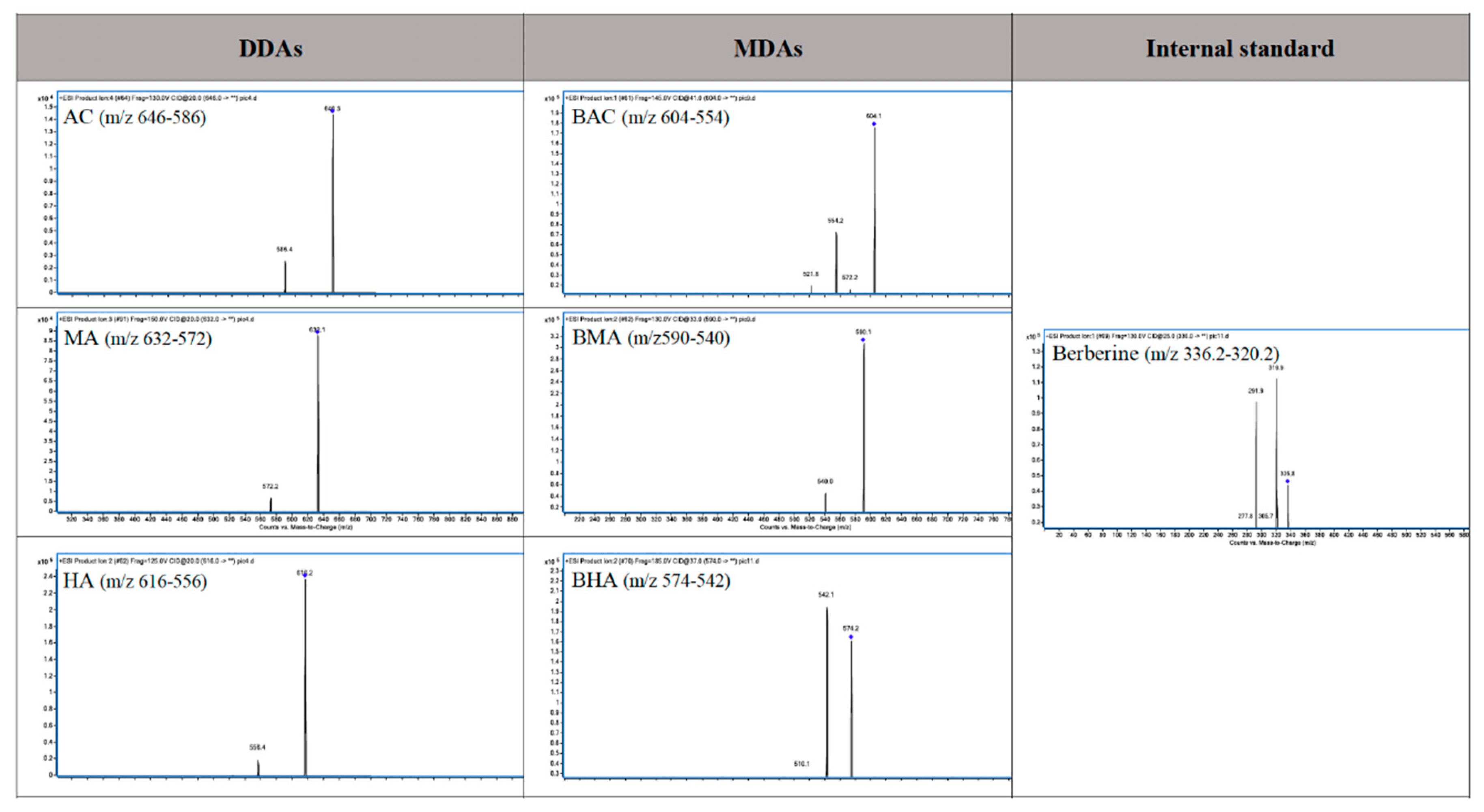
| Compound | Precursor Ion (m/z) | Product Ion (m/z) | Fragmentor (mV) | Collision Energy (mV) |
|---|---|---|---|---|
| AC | 646 | 586 | 130 | 25 |
| MA | 632 | 572 | 150 | 37 |
| HA | 616 | 556 | 125 | 33 |
| BAC | 604 | 554 | 145 | 41 |
| BMA | 590 | 540 | 130 | 33 |
| BHA | 574 | 542 | 185 | 37 |
| Berberine (IS) | 336.2 | 320.2 | 130 | 25 |
| Tissue | Liver | Kidney | Heart | Brain | Plasma | Urine | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Compound | Heishunpian | Paofupian | Heishunpian | Paofupian | Heishunpian | Paofupian | Heishunpian | Paofupian | Heishunpian | Paofupian | Heishunpian | Paofupian |
| AC | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. |
| HA | 10.89 ± 11.11 | N.D. | 1.38 ± 0.47 | N.D. | 0.09 ± 0.14 | N.D. | N.D. | N.D. | 3.91 ± 1.49 | N.D. | 17.10 ± 13.51 | N.D. |
| MA | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | 9.63 ± 6.93 | N.D. |
| Total DDAs | 10.89 ± 11.11 | N.D. | 1.38 ± 0.47 | N.D. | 0.09 ± 0.14 | N.D. | N.D. | N.D. | 3.91 ± 1.49 | N.D. | 26.73 ± 19.88 | N.D. |
| BAC | 0.44 ± 0.13 | 2.84 ± 0.12 | 0.03 ± 0.03 | N.D. | N.D. | N.D. | N.D. | N.D. | 0.05 ± 0.03 | 0.04 ± 0.00 | 0.25 ± 0.19 | 0.11 ± 0.00 |
| BHA | 0.66 ± 0.25 | 1.37 ± 0.06 | 0.04 ± 0.03 | 0.10 ± 0.00 | 0.01 ± 0.00 | N.D. | 0.01 ± 0.00 | N.D. | 0.10 ± 0.05 | 0.06 ± 0.00 | 0.28 ± 0.17 | 0.07 ± 0.00 |
| BMA | 0.64 ± 0.21 | 0.82 ± 0.04 | 0.03 ± 0.02 | 0.11 ± 0.00 | N.D. | 0.00 ± 0.00 | N.D. | 0.02 ± 0.00 | 0.06 ± 0.04 | 0.02 ± 0.00 | 0.31 ± 0.20 | 0.11 ± 0.00 |
| Total MDAs | 1.74 ± 0.47 | 5.03 ± 0.22 | 0.10 ± 0.07 | 0.21 ± 0.01 | 0.01 ± 0.01 | 0.00 ± 0.00 | **** 0.01 ± 0.01 | 0.02 ± 0.00 | 0.21 ± 0.09 | 0.11 ± 0.00 | 0.84 ± 0.53 | 0.29 ± 0.01 |
| Total toxic alkaloids | ** 12.63 ± 10.97 | 5.03 ± 0.22 | **** 1.48 ± 0.51 | 0.21 ± 0.01 | * 0.09 ± 0.14 | 0.00 ± 0.00 | N.D. | 0.02 ± 0.00 | **** 4.13 ± 1.56 | 0.11 ± 0.00 | **** 27.57 ± 20.31 | 0.29 ± 0.01 |
| Tissue | Liver | Kidney | Heart | Brain | Plasma | Urine | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Compound | Heishunpian | Paofupian | Heishunpian | Paofupian | Heishunpian | Paofupian | Heishunpian | Paofupian | Heishunpian | Paofupian | Heishunpian | Paofupian |
| AC | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. |
| HA | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | 102.96 ± 46.64 | N.D. |
| MA | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. |
| Total DDAs | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | N.D. | 102.96 ± 46.64 | N.D. |
| BAC | 3.01 ± 1.51 | 4.23 ± 0.28 | 0.51 ± 0.11 | 0.30 ± 0.02 | 0.13 ± 0.02 | 0.11 ± 0.01 | N.D. | N.D. | 0.16 ± 0.05 | 0.14 ± 0.01 | 12.18 ± 8.84 | 3.02 ± 0.20 |
| BHA | ** 2.82 ± 0.83 | 0.78 ± 0.05 | **** 1.04 ± 0.20 | 0.54 ± 0.04 | **** 0.17 ± 0.03 | 0.26 ± 0.02 | N.D. | N.D. | 0.16 ± 0.06 | 0.25 ± 0.02 | 14.38 ± 8.43 | 6.74 ± 0.45 |
| BMA | *** 2.87 ± 0.97 | 0.81 ± 0.05 | 0.49 ± 0.11 | 0.35 ± 0.02 | 0.11 ± 0.03 | 0.09 ± 0.01 | **** 0.05 ± 0.01 | 0.03 ± 0.00 | 0.09 ± 0.05 | 0.03 ± 0.00 | 8.06 ± 7.33 | 2.84 ± 0.19 |
| Total MDAs | **** 8.70 ± 2.52 | 5.82 ± 0.39 | **** 2.05 ± 0.40 | 1.21 ± 0.08 | ** 0.41 ± 0.04 | 0.45 ± 0.03 | **** 0.05 ± 0.01 | 0.03 ± 0.00 | 0.41 ± 0.10 | 0.38 ± 0.15 | 34.62 ± 23.87 | 12.60 ± 0.85 |
| Total toxic alkaloids | **** 8.70 ± 2.52 | 5.82 ± 0.39 | **** 2.05 ± 0.40 | 1.21 ± 0.08 | ** 0.41 ± 0.04 | 0.45 ± 0.03 | **** 0.05 ± 0.01 | 0.03 ± 0.00 | 0.41 ± 0.10 | 0.38 ± 0.15 | **** 137.58 ± 49.70 | 12.60 ± 0.85 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ji, X.; Yang, M.; Or, K.H.; Yim, W.S.; Zuo, Z. Tissue Accumulations of Toxic Aconitum Alkaloids after Short-Term and Long-Term Oral Administrations of Clinically Used Radix Aconiti Lateralis Preparations in Rats. Toxins 2019, 11, 353. https://doi.org/10.3390/toxins11060353
Ji X, Yang M, Or KH, Yim WS, Zuo Z. Tissue Accumulations of Toxic Aconitum Alkaloids after Short-Term and Long-Term Oral Administrations of Clinically Used Radix Aconiti Lateralis Preparations in Rats. Toxins. 2019; 11(6):353. https://doi.org/10.3390/toxins11060353
Chicago/Turabian StyleJi, Xiaoyu, Mengbi Yang, Ka Hang Or, Wan Sze Yim, and Zhong Zuo. 2019. "Tissue Accumulations of Toxic Aconitum Alkaloids after Short-Term and Long-Term Oral Administrations of Clinically Used Radix Aconiti Lateralis Preparations in Rats" Toxins 11, no. 6: 353. https://doi.org/10.3390/toxins11060353
APA StyleJi, X., Yang, M., Or, K. H., Yim, W. S., & Zuo, Z. (2019). Tissue Accumulations of Toxic Aconitum Alkaloids after Short-Term and Long-Term Oral Administrations of Clinically Used Radix Aconiti Lateralis Preparations in Rats. Toxins, 11(6), 353. https://doi.org/10.3390/toxins11060353





