Toxicity Studies of Chanoclavine in Mice
Abstract
1. Introduction
2. Results
2.1. Acute Toxicity Testing
2.2. Short-Term Toxicity Study
2.2.1. Analysis of Mouse Diet
2.2.2. Clinical Observations and Appearance
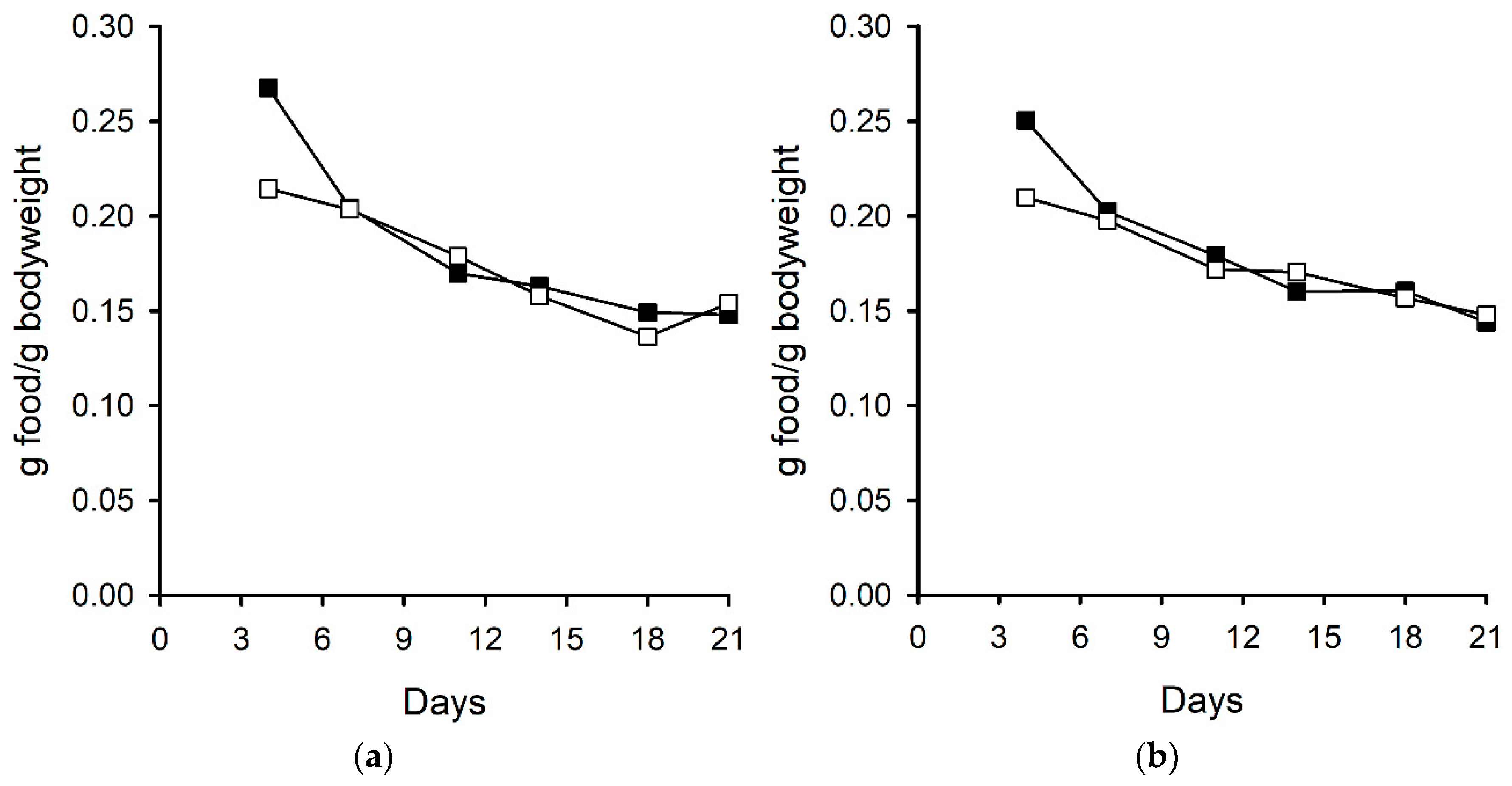
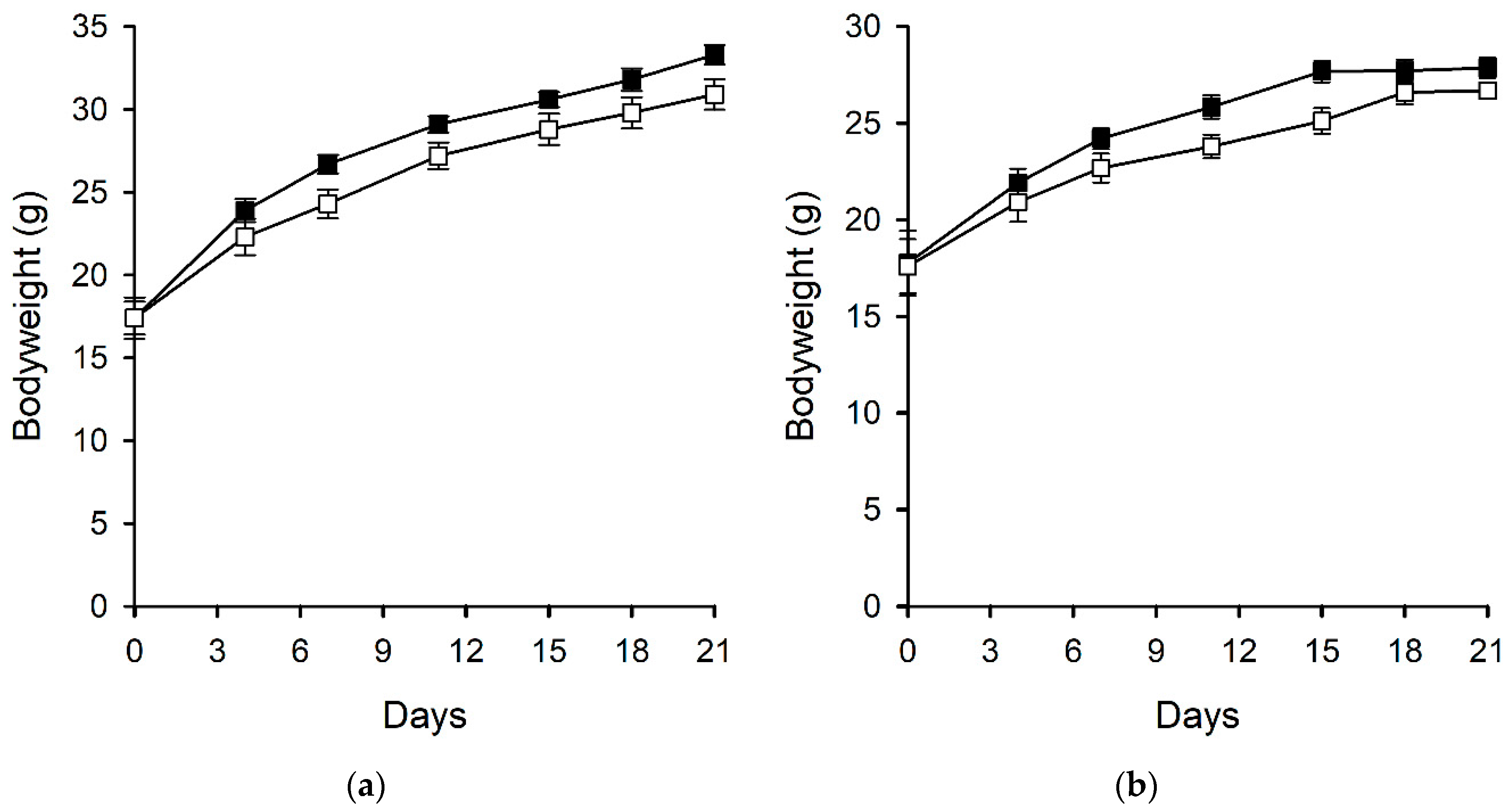
2.2.3. Bodyweight and Food Consumption
2.2.4. Hematological and Serum Biochemical Data
2.2.5. Organ Weights
2.2.6. Histological Examination
3. Discussion
4. Materials and Methods
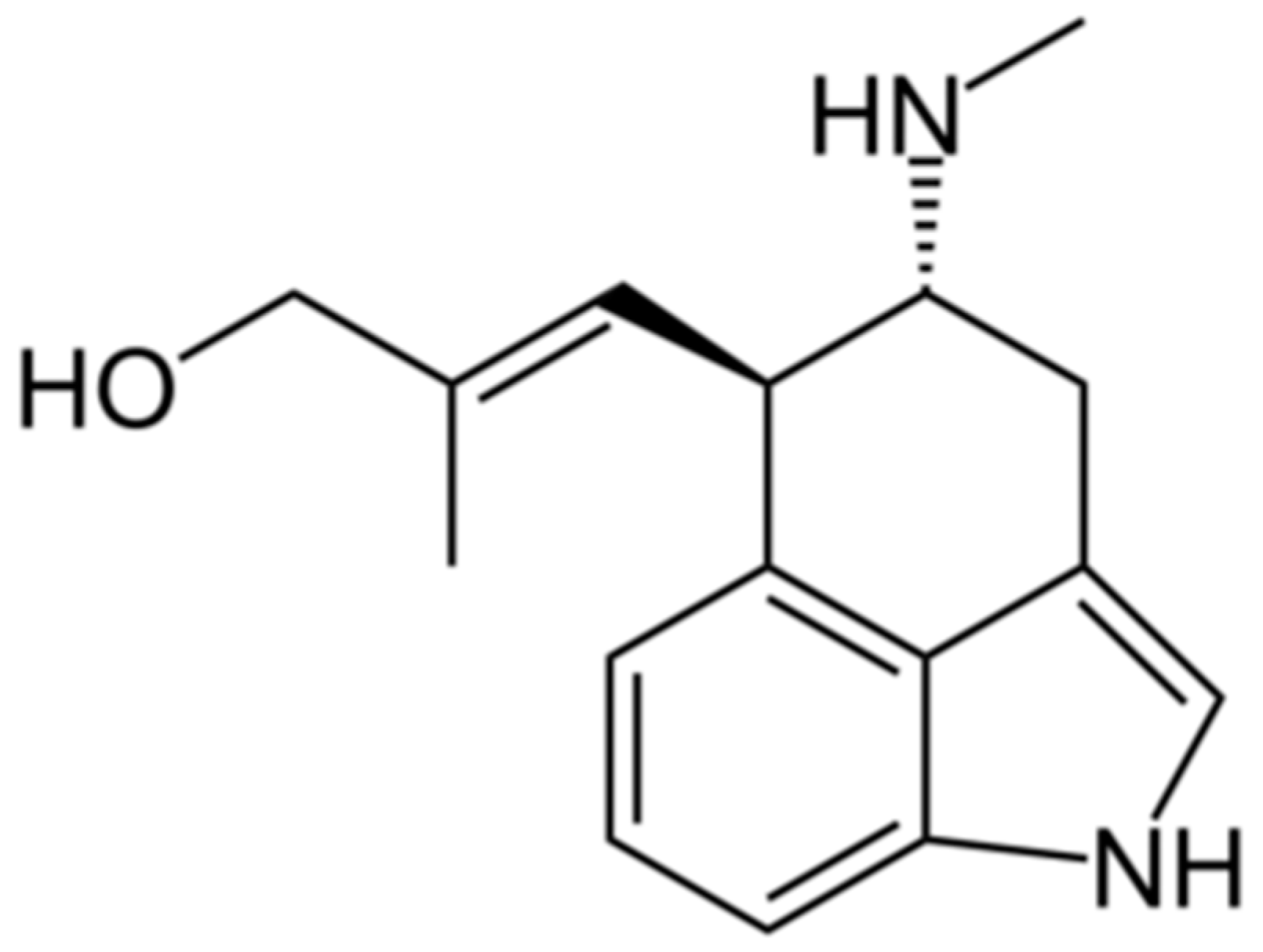
4.1. Chanoclavine
4.2. Animals
4.3. Acute Toxicity Protocol
4.4. Short-Term Toxicity Study
4.5. Measurement of Chanoclavine in Mouse Diet
4.6. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Johnson, L.J.; de Bonth, A.C.M.; Briggs, L.R.; Caradus, J.R.; Finch, S.C.; Fleetwood, D.J.; Fletcher, L.R.; Hume, D.E.; Johnson, R.D.; Popay, A.J.; et al. The exploitation of epichloae endophytes for agricultural benefit. Fungal Divers. 2013, 60, 171–188. [Google Scholar] [CrossRef]
- Rowan, D.D.; Hunt, M.B.; Gaynor, D.L. Peramine, a novel insect feeding deterrent from ryegrass infected with the endophyte Acremonium loliae. J. Chem. Soc. Chem. Commun. 1986, 935–936. [Google Scholar] [CrossRef]
- Rowan, D.D.; Dymock, J.J.; Brimble, M.A. Effect of the fungal metabolite peramine and analogs on feeding and development of Argentine stem weevil (Listrontus bonariensis). J. Chem. Ecol. 1990, 16, 1683–1695. [Google Scholar] [CrossRef]
- Rowan, D.; Gaynor, D. Isolation of feeding deterrents against Argentine stem weevil from ryegrass infected with the endophyte Acremonium loliae. J. Chem. Ecol. 1986, 12, 647–658. [Google Scholar] [CrossRef] [PubMed]
- Jensen, J.G.; Popay, A.J.; Tapper, B.A. Argentine stem weevil adults are affected by meadow fescue endophyte and its loline alkaloids. New Zealand Plant Prot. 2009, 62, 12–18. [Google Scholar]
- Reidell, W.; Kieckhefer, R.; Petroski, R.; Powell, R. Naturally occurring and synthetic loline alkaloid derivatives: Insect feeding behaviour modification and toxicity. J. Entomol. Sci. 1991, 26, 122–129. [Google Scholar] [CrossRef]
- Wilkinson, H.H.; Siegel, M.R.; Blankenship, J.D.; Mallory, A.C.; Bush, L.P.; Schardl, C.L. Contribution of fungal loline alkaloids to protection from aphids in a grass-endophyte mutualism. Mol. Plant-Microbe Interact. 2000, 13, 1027–1033. [Google Scholar] [CrossRef] [PubMed]
- Di Menna, M.E.; Finch, S.C.; Popay, A.J.; Smith, B.L. A review of the Neotyphodium lolii/Lolium perenne symbiosis and its associated effects on animal and plant health, with particular emphasis on ryegrass staggers. New Zealand Vet. J. 2012, 60, 315–328. [Google Scholar] [CrossRef]
- Fletcher, L.R.; Harvey, I.C. An association of a Lolium endophyte with ryegrass staggers. New Zealand Vet. J. 1981, 29, 185–186. [Google Scholar] [CrossRef] [PubMed]
- Bacon, C.W.; Porter, J.K.; Robbins, J.D.; Luttrell, E.S. Epichloë typhina from toxic tall fescue grasses. Appl. Environ. Micro. 1977, 34, 576–581. [Google Scholar]
- Schmidt, S.P.; Hoveland, C.S.; Clark, E.M.; Davis, N.D.; Smith, L.A.; Grimes, H.W.; Holliman, J.L. Association of an endophytic fungus with fescue toxicity in steers fed Kentucky 31 tall fescue seed or hay. J. Anim. Sci. 1982, 55, 1259–1263. [Google Scholar] [CrossRef]
- Klotz, J. Activities and effects of ergot alkaloids on livestock physiology and production. Toxins 2015, 7, 2801–2821. [Google Scholar] [CrossRef] [PubMed]
- Simpson, W.R.; Faville, M.J.; Moraga, R.A.; Williams, W.M.; McManus, M.T.; Johnson, R.D. Epichloë fungal endophytes and the formation of synthetic symbioses in Hordeeae (=Triticeae) grasses. J. Syst. Evol. 2014, 52, 794–806. [Google Scholar] [CrossRef]
- Feuillet, C.; Muehlbauer, G.J. Genetics and genomics of the Triticeae; Springer Science & Business Media: New York, NY, USA, 2009. [Google Scholar]
- Daccord, R.; Schmidt, D.; Arrigo, Y.; Gutzwiller, A. Endophytes in meadow fescue: a limiting factor in ruminant production? In Proceedings of the 2nd International Conference on Harmful and Beneficial Microorganisms in Pastures, Turf and Grassland, Paderborn, Germany, 22–24 November 1995; pp. 155–159. [Google Scholar]
- Fletcher, L.R.; Popay, A.J.; Stewart, A.V.; Tapper, B.A. Herbage and sheep production from meadow fescue with and without the endophyte Neotyphodium uncinatum. In Proceedings of the 4th International Neotyphodium/Grass Interactions Symposium, Soest, Germany, 27–29 September 2000; pp. 447–453. [Google Scholar]
- Finch, S.C.; Munday, J.S.; Munday, R.; Kerby, J.W.F. Short-term toxicity studies of loline alkaloids in mice. Food Chem. Toxicol. 2016, 94, 243–249. [Google Scholar] [PubMed]
- Young, C.; Schardl, C.; Panaccione, D.; Florea, S.; Takach, J.; Charlton, N.; Moore, N.; Webb, J.; Jaromczyk, J. Genetics, Genomics and Evolution of Ergot Alkaloid Diversity. Toxins 2015, 7, 1273–1302. [Google Scholar] [CrossRef]
- Panaccione, D.G. Origins and significance of ergot alkaloid diversity in fungi. FEMS Microbiol. Lett. 2005, 251, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Robinson, S.; Panaccione, D. Diversification of ergot alkaloids in natural and modified fungi. Toxins 2015, 7, 201–218. [Google Scholar] [CrossRef]
- Ball, O.J.P.; Miles, C.O.; Prestidge, R.A. Ergopeptine alkaloids and Neotyphodium lolii-mediated resistance in perennial ryegrass against adult Heteronychus arator (Coleoptera: Scarabaeidae). J. Econ. Entomol. 1997, 90, 1382–1391. [Google Scholar] [CrossRef]
- Clay, K.; Cheplick, G.P. Effect of ergot alkaloids from fungal endophyte-infected grasses on fall armyworm (Spodoptera frugiperda). J. Chem. Ecol. 1989, 15, 169–182. [Google Scholar] [CrossRef]
- Piper, E.; Denard, T.; Johnson, Z.; Flieger, M. Effect of chanoclavine on in vitro prolactin release. In Proceedings of the 4th International Neotyphodium/Grass Interactions Symposium, Soest, Germany, 27–29 September 2000; pp. 531–534. [Google Scholar]
- United Nations. Globally harmonized system of classification and labelling of chemicals (GHS), 4th ed.; United Nations Publications: New York, NY, USA, 2011. [Google Scholar]
- New Zealand Government—Environmental Protection Authority Correlation between GHS and New Zealand HSNO hazard classes and categories. 2009.
- Finch, S.C.; Pennell, C.G.L.; Kerby, J.W.F.; Cave, V.M. Mice find endophyte-infected seed of tall fescue unpalatable—Implications for the aviation industry. Grass Forage Sci. 2016, 71, 659–666. [Google Scholar] [CrossRef]
- Coley, A.B.; Fribourg, H.A.; Pelton, M.R.; Gwinn, K.D. Effects of tall fescue endophyte infestation on relative abundance of small mammals. J. Environ. Qual. 1995, 24, 472–475. [Google Scholar] [CrossRef]
- Matthews, A.K.; Poore, M.H.; Huntington, G.B.; Green, J.T. Intake, digestion, and N metabolism in steers fed endophyte-free, ergot alkaloid-producing endophyte-infected, or non ergot alkaloid-producing endophyte-infected fescue hay. J. Anim. Sci. 2005, 83, 1179–1185. [Google Scholar] [CrossRef]
- Panaccione, D.G.; Cipoletti, J.R.; Sedlock, A.B.; Blemings, K.P.; Schardl, C.L.; Machado, C.; Seidel, G.E. Effects of ergot alkaloids on food preference and satiety in rabbits, as assessed with gene-knockout endophytes in perennial ryegrass (Lolium perenne). J. Agric. Food Chem. 2006, 54, 4582–4587. [Google Scholar] [CrossRef] [PubMed]
- OECD guidelines for the testing of chemicals Guideline 425. Acute Oral Toxicity-Up-and-Down-Procedure (UDP); OECD: Paris, France, 2008. [Google Scholar]
- Rasmussen, S.; Lane, G.A.; Mace, W.; Parsons, A.J.; Fraser, K.; Xue, H. The use of genomics and metabolomics methods to quantify fungal endosymbionts and alkaloids in grasses. Methods Mol. Biol. 2012, 860, 213–226. [Google Scholar] [PubMed]
| Item | Control 1 | Chanoclavine 1 | SED | p-Value |
|---|---|---|---|---|
| Males | ||||
| HCT (L/L) | 0.46 ± 0.01 | 0.47 ± 0.01 | 0.01 | 0.620 |
| HB (g/L) | 140.8 ± 3.6 | 142.8 ± 4.9 | 2.72 | 0.483 |
| RBC (×1012/L) | 8.8 ± 0.30 | 8.9 ± 0.4 | 0.21 | 0.527 |
| MCV (fL) | 52.8 ± 1.5 | 53.2 ± 1.3 | 0.88 | 0.663 |
| MCH (pg) | 16.2 ± 0.4 | 16.2 ± 0.4 | 0.28 | 1.000 |
| MCHC (g/L) | 303.8 ± 2.6 | 303.6 ± 4.6 | 2.35 | 0.934 |
| WBC (×109/L) | 10.3 ± 1.6 | 10.4 ± 1.7 | 1.06 | 0.884 |
| Neutrophil (%) | 17.6 ± 1.5 | 19.4 ± 7.5 | 3.42 | 0.613 |
| Lymphocyte (%) | 75.8 ± 5.2 | 69.6 ± 8.9 | 4.62 | 0.216 |
| Monocyte (%) | 3.2 ± 1.6 | 4.8 ± 1.5 | 0.99 | 0.145 |
| Eosinophil (%) | 3.0 ± 2.0 | 3.0 ± 1.9 | 1.23 | 1.000 |
| Basophil (%) | 0.4 ± 0.6 | 3.2 ± 7.2 | 3.21 | 0.408 |
| Females | ||||
| HCT (L/L) | 0.47 ± 0.01 | 0.49 ± 0.04 | 0.02 | 0.213 |
| HB (g/L) | 145.4 ± 1.3 | 151.0 ± 12.5 | 5.61 | 0.347 |
| RBC (×1012/L) | 8.9 ± 0.1 | 9.4 ± 0.8 | 0.35 | 0.149 |
| MCV (fL) | 52.8 ± 0.8 | 51.8 ± 1.3 | 0.69 | 0.187 |
| MCH (pg) | 16.4 ± 0.5 | 16.0 ± 0.0 | 0.25 | 0.141 |
| MCHC (g/L) | 311.0 ± 3.7 | 308.6 ± 3.0 | 2.14 | 0.294 |
| WBC (×109/L) | 7.4 ± 2.1 | 8.5 ± 1.9 | 1.27 | 0.405 |
| Neutrophil (%) | 13.6 ± 3.1 a | 9.4 ± 1.7 b | 1.59 | 0.029 |
| Lymphocyte (%) | 78.0 ± 3.7 | 81.4 ± 2.4 | 1.97 | 0.122 |
| Monocyte (%) | 3.6 ± 1.1 | 4.0 ± 1.6 | 0.87 | 0.659 |
| Eosinophil (%) | 4.8 ± 1.3 | 4.6 ± 0.9 | 0.71 | 0.784 |
| Basophil (%) | 0.0 ± 0.0 | 0.6 ± 1.3 | 0.60 | 0.347 |
| Item | Control 1 | Chanoclavine 1 | SED | p-Value |
|---|---|---|---|---|
| Males | ||||
| CK (IU/L) | 294 ± 69 | 437 ± 206 | 91.6 | 0.164 |
| AST (IU/L) | 75 ± 15 | 75 ± 20 | 11.1 | 0.972 |
| ALT (IU/L) | 37 ± 12 | 35 ± 6 | 6.0 | 0.748 |
| T.Bil (µmol/L)2 | 1.8 ± 0.4 a | 0.9 ± 0.2 b | 0.2 | 0.004 |
| TP (g/L) | 54.0 ± 2.2 | 53.8 ± 2.0 | 1.4 | 0.886 |
| ALB (g/L) | 33.2 ± 0.8 | 33.0 ± 1.4 | 0.7 | 0.786 |
| Globulin (g/L) | 20.8 ± 2.9 | 20.0 ± 1.4 | 1.5 | 0.616 |
| A/G | 1.622 ± 0.230 | 1.660 ± 0.178 | 0.1 | 0.787 |
| CRN (µmol/L) | 14.0 ± 2.8 | 17.0 ± 5.8 | 2.9 | 0.328 |
| Na (mmol/L) | 146 ± 2 | 146 ± 1 | 0.9 | 0.829 |
| K (mmol/L) | 15.0 ± 1.4 | 15.3 ± 1.3 | 0.9 | 0.700 |
| Cl (mmol/L) | 103 ± 1 | 105 ± 1 | 0.6 | 0.101 |
| Females | ||||
| CK (IU/L) | 569 ± 210 | 594 ± 209 | 132.6 | 0.851 |
| AST (IU/L) | 110 ± 20 | 121 ± 17 | 11.7 | 0.366 |
| ALT (IU/L) | 31 ± 5 | 39 ± 9 | 4.5 | 0.126 |
| T.Bil (µmol/L) 2 | 1.5 ± 0.7 | 0.9 ± 0.2 | 0.3 | 0.108 |
| TP (g/L) | 54.8 ± 1.1 b | 57.8 ± 2.3 a | 1.1 | 0.029 |
| ALB (g/L) | 36.6 ± 1.1 b | 38.8 ± 1.5 a | 0.8 | 0.030 |
| Globulin (g/L) | 18.2 ± 0.8 | 19.0 ± 1.6 | 0.8 | 0.347 |
| A/G | 2.015 ± 0.140 | 2.053 ± 0.181 | 0.1 | 0.721 |
| CRN (µmol/L) | 17.0 ± 0.7 | 16.8 ± 3.7 | 1.7 | 0.908 |
| Na (mmol/L) | 148 ± 1 | 148 ± 4 | 1.8 | 0.913 |
| K (mmol/L) | 12.4 ± 1.0 | 12.8 ± 2.1 | 1.0 | 0.682 |
| Cl (mmol/L) | 106 ± 1 | 106 ± 2 | 0.9 | 0.838 |
| Brain 1 | Heart 1 | Kidneys 1 | Liver 1 | Spleen 1 | |
|---|---|---|---|---|---|
| Males | |||||
| Control | 0.467 ± 0.017 | 0.181 ± 0.011 | 0.512 ± 0.019 a | 1.832 ± 0.129 | 0.135 ± 0.029 |
| Chanoclavine | 0.453 ± 0.021 | 0.176 ± 0.019 | 0.441 ± 0.033 b | 1.656 ± 0.183 | 0.126 ± 0.017 |
| SED | 0.012 | 0.010 | 0.017 | 0.100 | 0.015 |
| p-value | 0.269 | 0.623 | 0.003 | 0.117 | 0.579 |
| Females | |||||
| Control | 0.457 ± 0.020 | 0.167 ± 0.012 | 0.355 ± 0.017 | 1.384 ± 0.203 | 0.146 ± 0.028 |
| Chanoclavine | 0.466 ± 0.010 | 0.155 ± 0.006 | 0.328 ± 0.029 | 1.280 ± 0.093 | 0.132 ± 0.020 |
| SED | 0.010 | 0.006 | 0.015 | 0.100 | 0.016 |
| p-value | 0.385 | 0.086 | 0.117 | 0.328 | 0.420 |
| Brain 1 | Heart 1 | Kidneys 1 | Liver 1 | Spleen 1 | |
|---|---|---|---|---|---|
| Males | |||||
| Control | 1.439 ± 0.039 | 0.559 ± 0.041 | 1.578 ± 0.079 a | 5.638 ± 0.259 | 0.417 ± 0.097 |
| Chanoclavine | 1.500 ± 0.110 | 0.586 ± 0.090 | 1.456 ± 0.023 b | 5.458 ± 0.251 | 0.417 ± 0.046 |
| SED | 0.052 | 0.044 | 0.037 | 0.161 | 0.048 |
| p-value | 0.280 | 0.555 | 0.011 | 0.298 | 0.999 |
| Females | |||||
| Control | 1.671 ± 0.136 | 0.610 ± 0.037 | 1.296 ± 0.059 | 5.030 ± 0.510 | 0.530 ± 0.090 |
| Chanoclavine | 1.775 ± 0.049 | 0.592 ± 0.031 | 1.250 ± 0.101 | 4.873 ± 0.322 | 0.504 ± 0.074 |
| SED | 0.065 | 0.022 | 0.052 | 0.270 | 0.052 |
| p-value | 0.147 | 0.439 | 0.404 | 0.578 | 0.626 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Finch, S.C.; Munday, J.S.; Sprosen, J.M.; Bhattarai, S. Toxicity Studies of Chanoclavine in Mice. Toxins 2019, 11, 249. https://doi.org/10.3390/toxins11050249
Finch SC, Munday JS, Sprosen JM, Bhattarai S. Toxicity Studies of Chanoclavine in Mice. Toxins. 2019; 11(5):249. https://doi.org/10.3390/toxins11050249
Chicago/Turabian StyleFinch, Sarah C., John S. Munday, Jan M. Sprosen, and Sweta Bhattarai. 2019. "Toxicity Studies of Chanoclavine in Mice" Toxins 11, no. 5: 249. https://doi.org/10.3390/toxins11050249
APA StyleFinch, S. C., Munday, J. S., Sprosen, J. M., & Bhattarai, S. (2019). Toxicity Studies of Chanoclavine in Mice. Toxins, 11(5), 249. https://doi.org/10.3390/toxins11050249






