Evaluation of Cytotoxicity and Mould Contamination of Selected Plants from Meadows Covered by the Agri-Environmental Program
Abstract
:1. Introduction
2. Results
2.1. Fungal Contamination of Meadows
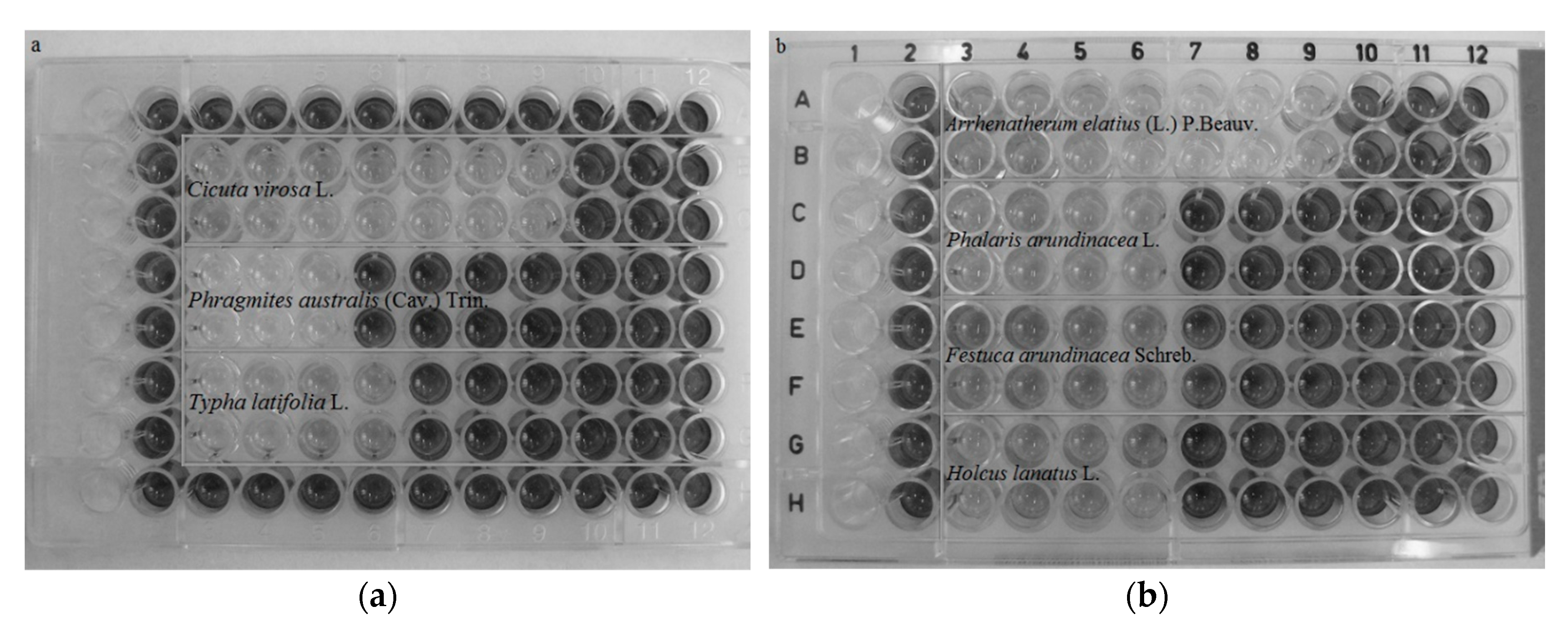
2.2. Cytotoxicity of Meadows
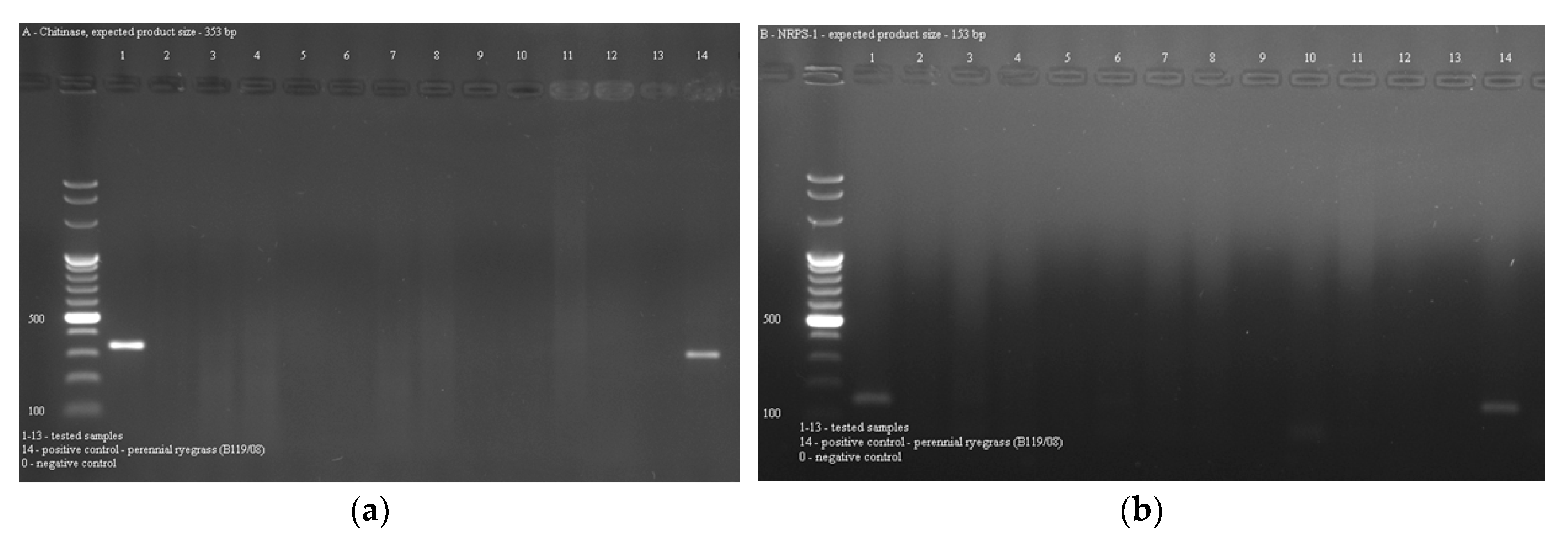
2.3. Occurrence of Epichloë Endophytes in Grasses
3. Discussion
4. Materials and Methods
4.1. Plant Material Sampling
4.2. Analysis of the Fungal Contamination
4.3. Identification of Moulds
4.4. MTT Assay
4.5. PCR Detection of Endophytes in Grasses
Author Contributions
Funding
Conflicts of Interest
References
- Mazurek, K. Użytki Zielone w Żywieniu Zwierząt; Opolski Ośrodek Doradztwa Rolniczego: Łosiów, Poland, 2015; Volume 8, pp. 1–28. [Google Scholar]
- Wasilewski, Z. Trawy przydatne na pastwiskach i ich użytkowanie. Agro Serwis Trawy I Rośliny Motylkowate 2005, 9, 46–50. [Google Scholar]
- Falkowski, M.; Kukułka, I.; Kozłowski, S. Właściwości Chemiczne Roślin Łąkowych; University Publisher Poznan University of Life Sciences: Poznań, Poland, 2000. [Google Scholar]
- Rutkowska, B. Atlas Roślin Łąkowych i Pastwiskowych; PWRiL: Warszawa, Poland, 1984. [Google Scholar]
- Filipek, J. Projekt klasyfikacji roślin łąkowych i pastwiskowych na podstawie liczb wartości użytkowej. Zesz. Nauk Rol. 1973, 4, 59–68. [Google Scholar]
- Klapp, E. Łąki i Pastwiska; PWRiL: Warszawa, Poland, 1962. [Google Scholar]
- Grajewski, J. Mikotoksyny i Grzyby Pleśniowe—Zagrożenia dla Człowieka i Zwierząt; University Publisher Kazimierz Wielki University: Bydgoszcz, Poland, 2006. [Google Scholar]
- Weidenbörner, M. Mycotoxins in Feedstuffs, 2nd ed.; Springer: Berlin/Heidelberg, Germany, 2012. [Google Scholar]
- Falkowski, M.; Kukułka, I. Właściwości fitochemiczne traw. In Trawy Polskie; Falkowski, M., Ed.; PWRiL: Warszawa, Poland, 1982; pp. 63–74. [Google Scholar]
- Prończuk, M. Endofity traw—Znaczenie, występowanie i metody wykrywania. Biul. Ihar 2005, 235, 279–309. [Google Scholar]
- Leuchtmann, A.; Bacon, C.W.; Schardl, C.L.; White, J.F., Jr.; Tadych, M. Nomenclatural realignment of Neotyphodium species with genus Epichloë. Mycologia 2014, 106, 202–215. [Google Scholar] [CrossRef]
- Pańka, D.; Jeske, M.; Troczyński, M. Occurrence of Neotyphodium and Epichloë fungi in meadow fescue and red fescue in Poland and screening of endophyte isolates as potential biological control agents. Acta Sci. Pol. Hortoru 2013, 12, 67–83. [Google Scholar]
- Wiewióra, B. Grzyby z rodzaju Neotyphodium—Endofity traw. Post. Mikrobiol. 2014, 53, 71–80. [Google Scholar]
- Koczwara, K.; Pańka, D.; Lisiecki, K.; Juda, M. Możliwość wykorzystania endofitów w biologicznej ochronie roślin. J. Educ. Health Sport. 2015, 5, 333–340. [Google Scholar]
- Grajewski, J. Możliwości Inaktywacji Ochratoksyny A w Badaniach In Vitro oraz In Vivo u Kurcząt; University Publisher Wydawnictwo Akademii Bydgoskiej: Bydgoszcz, Poland, 2003. [Google Scholar]
- Wiewióra, B.; Żurek, G. Endofity Traw—Zagrożenie Naszych łąk i Pastwisk? Informative elaboration carried out as part of the Multiannual Program 2008—2013; IHAR-PIB: Radzików, Poland, 2013. [Google Scholar]
- Broda, B.; Mowszowicz, J. Przewodnik do Oznaczania Roślin Leczniczych, Trujących i Użytkowych; Wydawnictwo Lekarskie PZWL: Warszawa, Poland, 1985. [Google Scholar]
- Pisulewska, E. Tajemnice ziół—Zastosowanie w Żywności, Żywieniu, Dietetyce i Kosmetologii; Krakowska Wyższa Szkoła Promocji Zdrowia: Kraków, Poland, 2016. [Google Scholar]
- Witczak, A.; Sikorski, Z. (Eds.) Toxins and Other Harmful Compounds in Foods; CRC Press, Taylor & Francis Group: Boca Raton, FL, USA, 2017. [Google Scholar]
- Bohne, B.; Dietze, P. Rośliny Trujące: 170 Gatunków Roślin Ozdobnych i Dziko Rosnących; Bellona Sp. z o.o. [Ltd.]: Warszawa, Poland, 2008. [Google Scholar]
- Ożarowski, A.; Jaroniewski, W. Rośliny Lecznicze i ich Praktyczne Zastosowanie; Instytut Wydawniczy Związków Zawodowych: Warszawa, Poland, 1987. [Google Scholar]
- Mowszowicz, J. Przewodnik do Oznaczania Roślin Trujących i Szkodliwych; PWRiL: Warszawa, Poland, 1982. [Google Scholar]
- Siegel, M.R.; Bush, L.P. Toxin production in grass/endophyte association. In The Mycota, V. Plant Relationships, Part B; Carroll, G.C., Tudzynski, P., Eds.; Springer: Berlin, Germany, 1997; pp. 185–208. [Google Scholar]
- Bonos, S.A.; Wilson, M.M.; Meyer, W.A.; Funk, C.R. Suppression of red thread in fine fescues through endophyte-mediated resistance. Appl. Turfgrass Sci. 2005. [Google Scholar] [CrossRef]
- Clarke, B.B.; White, J.F., Jr.; Hurley, R.H.; Torres, M.S.; Sun, S.; Huff, D.R. Endophyte mediated suppression of dollar spot disease in fine fescues. Plant Dis. 2006, 90, 994–998. [Google Scholar] [CrossRef]
- Tian, P.; Nan, Z.; Li, C.; Spangenberg, G. Effect of endophyte Neotyphodium lolii on susceptibility and host physiological response of perennial ryegrass to fungal pathogens. Eur. J. Plant Pathol. 2008, 122, 593–602. [Google Scholar] [CrossRef]
- Elmi, A.A.; West, C.P. Endophyte infection effects on stomatal conductance, osmotic adjustment and drought recovery of tall fescue. New Phytol. 1995, 131, 61–67. [Google Scholar] [CrossRef]
- Djonović, S.; Vittone, G.; Mendoza-Herrera, A.; Kenerley, C.M. Enhanced biocontrol activity of Trichoderma virens transformants constitutively coexpressing beta-1,3- and beta-1,6-glucanase genes. Mol. Plant Pathol. 2007, 8, 469–480. [Google Scholar] [CrossRef] [PubMed]
- Verma, M.; Brar, S.K.; Tyagi, R.D.; Surampalli, R.Y.; Val’ero, J.R. Antagonistic fungi, Trichoderma spp.: Panoply of biological control. Biochem. Eng. J. 2007, 37, 1–20. [Google Scholar] [CrossRef]
- Chaverri, P.; Gazis, R.O.; Samuels, G.J. Trichoderma amazonicum, a new endophytic species on Hevea brasiliensis and H. guianensis from the Amazon basin. Mycologia 2011, 103, 139–151. [Google Scholar] [CrossRef]
- Samuels, G.J.; Ismaiel, A. Trichoderma evansii and T. lieckfeldtiae: Two new T. hamatum-like species. Mycologia 2009, 101, 142–156. [Google Scholar] [CrossRef] [PubMed]
- Hanada, R.E.; deJorge Souza, T.; Pomella, A.W.; Hebbar, K.P.; Pereira, J.O.; Ismaiel, A.; Samuels, G.J. Trichoderma martiale sp. nov., a new endophyte from sapwood of Theobroma cacao with a potential for biological control. Mycol. Res. 2008, 112, 1335–1343. [Google Scholar] [CrossRef] [PubMed]
- Moricca, S.; Ragazzi, A.; Assante, G. Biocontrol of Rust Fungi by Cladosporium tenuissimum. In Rust Diseases of Willow and Poplar; Pei, M.H., McCracken, A.R., Eds.; CABI Publishing: Wallingford, UK, 2005; pp. 213–229. [Google Scholar]
- Bensaci, O.A.; Daoud, H.; Lombarkia, N.; Rouabah, K. Formulation of the endophytic fungus Cladosporium oxysporum Berk. & M.A. Curtis, isolated from Euphorbia bupleuroides subsp. luteola, as a new biocontrol tool against the black bean aphid (Aphis fabae Scop.). J. Plant Prot. Res. 2015, 55, 80–87. [Google Scholar]
- Prończuk, M. Choroby traw w uprawie na nasiona. Agro Serwis Trawy i Rośliny Motylkowate 2005, 9, 76–80. [Google Scholar]
- Weidenbörner, M. Encyclopedia of Food Mycotoxins; Springer: Berlin/Heidelberg, Germany; New York, NY, USA, 2001. [Google Scholar]
- Arseniuk, E.; Martyniak, J. Polskie trawy i koniczyny w unijnych warunkach. Agro Serwis Trawy I Rośliny Motylkowate 2005, 9, 3–10. [Google Scholar]
- Marcinkowska, J. Oznaczanie Rodzajów Grzybów sensu lato Ważnych w Fitopatologii; PWRiL: Warszawa, Poland, 2012. [Google Scholar]
- Fassatiová, O. Grzyby Mikroskopowe w Mikrobiologii Technicznej; WNT: Warszawa, Poland, 1983. [Google Scholar]
- Twarużek, M.; Ałtyn, I.; Zastempowska, E.; Grajewski, J. Mikotoksyny w TMR Objawy i zapobieganie. Agrobydło 2017, 3, 22–25. [Google Scholar]
- Blood, D.C. Poradnik Lekarza Weterynarii; Wydawnictwo Lekarskie PZWL: Warszawa, Poland, 1994. [Google Scholar]
- Womack, E.D.; Sparks, D.L.; Brown, A.E. Aflatoxin M1 in milk and milk products: A short review. World Mycotoxin J. 2015, 9, 305–315. [Google Scholar] [CrossRef]
- Zastempowska, E.; Grajewski, J.; Twarużek, M. Food-borne pathogens and contaminants in raw milk—A review. Ann. Anim. Sci. 2016, 16, 623–639. [Google Scholar] [CrossRef]
- Hoogenboom, R.L.; Mulder, P.P.J.; Zeilmaker, M.J.; Van den Top, H.J.; Remmelink, G.J.; Brandon, E.F.A.; Klijnstra, M.D.; Meijer, G.A.L.; Schothorst, R.; Van Egmond, H.P. Carry-over of pyrrolizidine alkaloids from feed to milk in dairy cows. Food Addit. Contam. 2011, 28, 359–372. [Google Scholar] [CrossRef]
- Hoerger, C.C.; Schenzel, J.; Strobel, B.W.; Bucheli, T.D. Analysis of selected phytotoxins and mycotoxins in environmental samples. Anal. Bioanal. Chem. 2009, 395, 1261–1289. [Google Scholar] [CrossRef]
- Łyszczarz, R.; Dembek, R.; Suś, R.; Zimmer-Grajewska, M.; Kornacki, P. Renowacja łąk trwałych położonych na glebach torfowo-murszowych. Itp Water-Environ. Rural Areas 2010, 10, 129–148. [Google Scholar]
- Dembek, R.; Łyszczarz, R.; Suś, R. Ocena wartości paszowej łąk użytkowanych według zaleceń programów rolnośrodowiskowych. Ekol. I Tech. 2012, 3, 162–169. [Google Scholar]
- PN ISO 7954:1999. Mikrobiologia—Ogólne zasady oznaczania drożdży i pleśni—Metoda płytkowa w 25 stopniach C; Polski Komitet Normalizacyjny: Warszawa, Poland, 1999. [Google Scholar]
- PN-EN ISO 6887-1:2000. Mikrobiologia żywności i pasz—Przygotowanie próbek, zawiesiny wyjściowej i rozcieńczeń dziesięciokrotnych do badań mikrobiologicznych—Ogólne zasady przygotowania zawiesiny wyjściowej i rozcieńczeń dziesięciokrotnych; Polski Komitet Normalizacyjny: Warszawa, Poland, 2000. [Google Scholar]
- PN-R-64791:1994. Pasze—Wymagania i badania mikrobiologiczne; Polski Komitet Normalizacyjny: Warszawa, Poland, 1994. [Google Scholar]
- Hanelt, M.; Gareis, M.; Kollarczik, B. Cytotoxicity of mycotoxins evaluated by the MTT-cell culture assay. Mycopathologia 1994, 128, 167–174. [Google Scholar] [CrossRef]
- Gareis, M. Diagnostischer Zellkulturtest (MTT-Test) für den Nachweis von zytotoxischen Kontaminanten und Rückständen. J. Verbr. Lebensm. 2006, 1, 354–363. [Google Scholar] [CrossRef]
- Doyle, J.J.; Doyle, J.L. Isolation of plant DNA from fresh tissue. Focus 1990, 12, 13–15. [Google Scholar]
- Rasmussen, S.; Parsons, A.J.; Bassett, S.; Christensen, M.J.; Hume, D.E.; Johnson, L.J.; Johnson, R.D.; Simpson, W.R.; Stacke, C.; Voisey, C.R.; et al. High nitrogen supply and carbohydrate content reduce fungal endophyte and alkaloid concentration in Lolium perenne. New Phytol. 2007, 173, 787–797. [Google Scholar] [CrossRef]
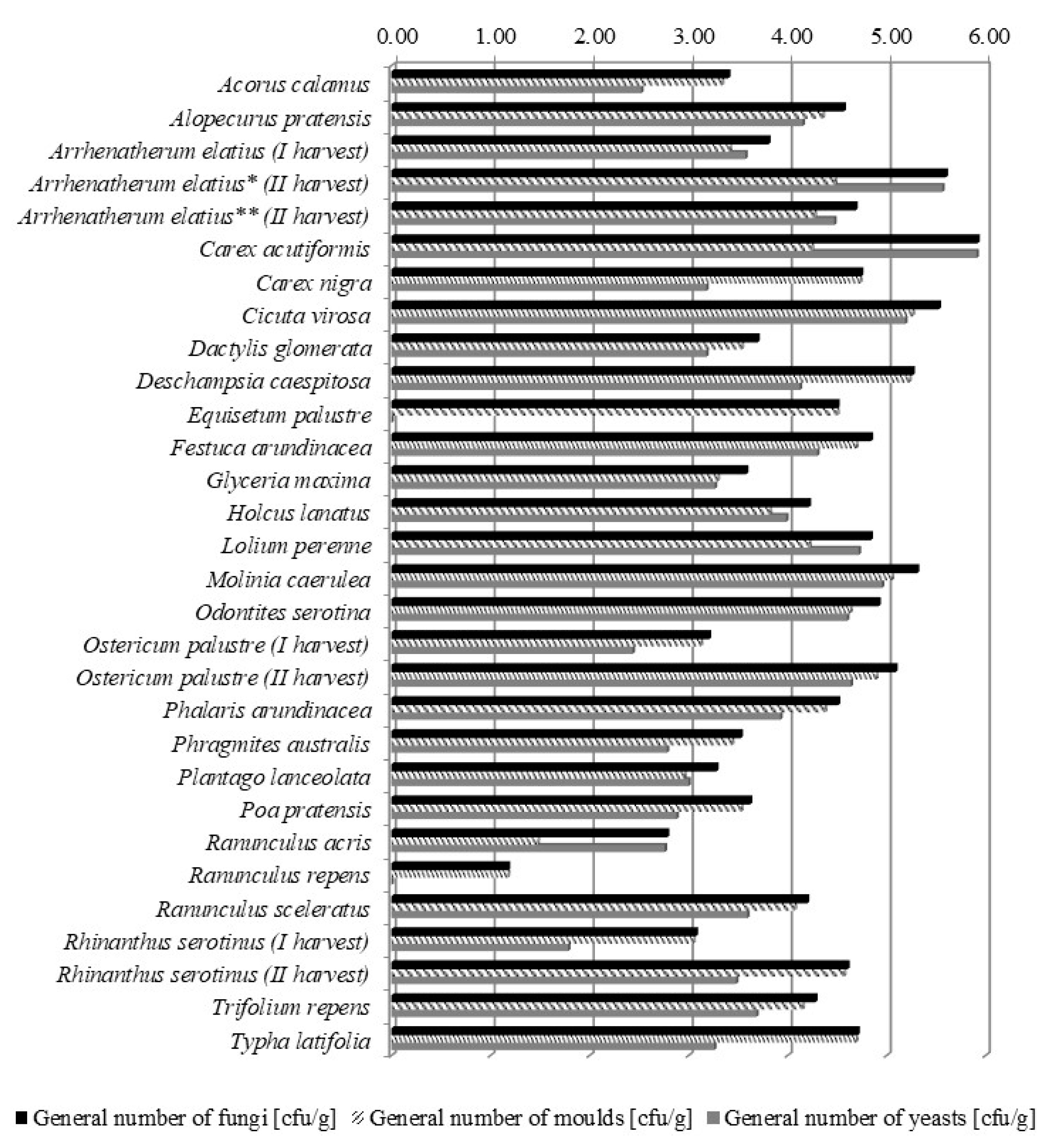
| Species | UVN a According to Filipek [5] from 10 to −3 and Klapp [6] from 8 to −1) | Cytotoxicity IC50 (mg/mL) (Classes of Cytotoxicity b) | General Number of Fungi (Moulds and Yeasts) (cfu/g) | Contribution of Moulds Genera (%) | Toxic Factors | |
|---|---|---|---|---|---|---|
| Factor | Reference | |||||
| I harvest—June/July | ||||||
| Poaceae | ||||||
| Alopecurus pratensis L. | 9 (7) | 50 (++) | 3.7 × 104 ± 2.3 × 103 | Pythium (61) Humicola (11) Phytophthora (11) Torula (8) not identified (9) | no data | - |
| Arrhenatherum elatius (L.) P.Beauv. | 9 (7) | 6.25 (+++) | 6.4 × 103 ± 1.3 × 103 | Humicola (34) Alternaria c (15) Cladosporium c (13) Ulocladium (8) Phoma (6) Fusarium c (1) Pythium (1) not identified (22) | saponins | [9] |
| Dactylis glomerata L. | 9 (7) | 50 (++) | 5.0 × 103 ± 8.6 × 102 | Torula (46) Pythium (16) Alternaria c (11) Cladosporium c (8) Humicola (8) Bipolaris (2) not identified (9) | endophytes | [13] |
| Deschampsia caespitosa (L.) P.Beauv. | 3-0 (3) | 50 (++) | 1.8 × 105 ± 6.5 × 104 | Humicola (45) Alternaria c (10) Fusarium c (6) not identified (39) | endophytes | [16] |
| Festuca arundinacea Schreb. | 6 (4) | 50 (++) | 6.9 × 104 ± 9.7 × 103 | Humicola (82) Cladosporium c (3) Pythium (3) Alternaria c (2) Epicoccum c (1) not identified (9) | perlolyrine, cyanogenic glycosides | [9] |
| alkaloids produced by endophytes (lolitrem, ergovaline) | [10,13,16] | |||||
| Glyceria maxima (Hartm.) Holmb. | 5 (4) | 100 (+) | 3.8 × 103 ± 8.8 × 102 | Chrysosporium (48) Humicola (21) Pythium (20) Alternaria c (3) Cladosporium c (3) Phytophthora (2) not identified (3) | cyanogenic glycosides | [9] |
| Holcus lanatus L. | 5 (4) | 50 (++) | 1.6 × 104 ± 4.8 × 103 | Pythium (27) Alternaria c (23) Cladosporium c (9) Humicola (9) Epicoccum c (5) not identified (27) | aconitic acid, cyanogenic glycosides, Pithomyces chartarum (fungus) | [9] |
| endophytes | [13] | |||||
| Lolium perenne L. | 10 (8) | 50 (++) | 6.9 × 104 ± 9.4 × 103 | Humicola (45) Cladosporium c (15) Nigrospora (5) not identified (35) | perlolyrine, cyanogenic glycosides, alkaloids (mainly lolitrem, ergovaline, peramine), ergot alkaloids -clavines, lysergic acid, lysergic acid amides, ergopeptides produced by endophytes) | [16] |
| Phalaris arundinacea L. | 7 (5) | 50 (++) | 3.2 × 104 ± 4.1 × 103 | Humicola (56) Alternaria c (16) Torula (10) Phoma (3) Fusarium c (1) not identified (14) | 9 alkaloids including gramine and tryptamine derivatives (N-methyltryptamine), hordein | [9] |
| Phragmites australis (Cav.) Trin. | 1 (2) | 100 (+) | 3.4 × 103 ± 4.0 × 102 | Cladosporiumc (59) Alternaria c (23) Fusarium c (8) Penicillium c (2) Trichoderma (1) not identified (7) | no data | - |
| Poa pratensis L. | 10 (8) | 50 (++) | 4.2 × 103 ± 7.9 × 102 | Humicola (44) Cladosporium c (12) Pythium (9) Alternaria c (4) Diplodia (4) Torula (4) Mucor c (2) Colletotrichum (1) Phytophthora (1) not identified (19) | endophytes | [16] |
| Cyperaceae | ||||||
| Carex acutiformis Ehrh. | 1–0 (1) | nd (−) | 8.5 × 105 ± 1.3 × 104 | Pythium (31) Humicola (24) Fusarium c (23) Alternaria c (16) Cladosporium c (3) not identified (3) | flavones (glycoflavon, tricin) | [17] |
| Carex nigra (L.) Reichard | 1 (1) | nd (−) | 5.5 × 104 ± 9.6 × 103 | Humicola (84) Alternaria c (3) Fusarium c (2) not identified (11) | flavones (tricin) | [17] |
| Other species | ||||||
| Acorus calamus L. | 0 (no data) | 50 (++) | 2.5 × 103 ± 2.3 × 102 | Alternariac (53) Fusarium c (37) Mucor c (4) Acremonium (4) Aspergillus c (2) | asarones | [18] |
| Cicuta virosa L. | −3 (−1) | 6.25 (++) | 3.4 × 105 ± 4.0 × 104 | Cladosporiumc (32) Alternaria c (29) Mucor c (25) Fusarium c (7) Penicillium c (5) Aspergillus c (2) | cicutoxin (aliphatic alcohol) | [19] |
| aconitine, benzoaconitine, neopeline, aconine, napeline | [1,20] | |||||
| Equisetum palustre L. | −2 (−1) | 100 (+) | 3.2 × 104 ± 4.0 × 103 | Torula (100) | alkaloids (palustrine, palustridine, nicotine), aconitic acid | [17] |
| Ostericum palustre Besser | no data species spreading on two-cut meadows | 50 (++) | 1.6 × 103 ± 4.6 × 102 | Alternariac (20.5) Humicola (15) Phoma (14) Chaetomium (9) Nigrospora (9) Morteriella (5) Aspergillus c (2) Botryotrichum (2) Aureobasidium (2) not identified (20.5) | no data | - |
| Plantago lanceolata L. | 7–5 (6) | 6.25 (++) | 1.9 × 103 ± 4.7 × 102 | Alternariac (27) Chaetomium (17) Humicola (17) Fusarium c (10) Drechslera (6) Mucor c (6) not identified (17) | iridoid glycoside (aucubin), flavonoids, tannins, organic acids, mucous compounds, pectins | [21] |
| iridoid glycosides (aucubin and catalpol), phenolic acids | [17] | |||||
| Ranunculus acris L. | 1 (1) | 12.5 (++) | 6.1 × 102 ± 2.2 × 102 | Alternariac (30) Nigrospora (30) Humicola (20) not identified (20) | glycoside (ranunculin), flavonoids (vitexin and neovitexin) | [17] |
| saponins, essential oil, protoanemonin, cyanogenic compounds | [1] | |||||
| Ranunculus repens L. | 2 (2) | 3.125 (+++) | < 20 ± 1.9 × 101 | Phoma (40) Humicola (40) not identified (20) | glycoside (ranunculin; aglycon: protoanemonin), flavonoids (vitexin and neovitexin) | [1,17] |
| Rhinanthus serotinus (Schönh) Oborny | 1 (−1) | 25 (++) | 1.2 × 103 ± 4.2 × 102 | Alternariac (51) Penicillium c (24) Pythium (8) Humicola (5.5) Mucor c (5.5) Arthrinium (3) not identified (3) | aucubin derivatives | [17] |
| Trifolium repens L. | 10 (8) | 50 (++) | 1.9 × 104 ± 7.4 × 103 | Humicola (28) Alternaria c (11) Cladosporium c (8) Mucor c (6) Pythium (4) not identified (43) | tannins, essential oil | [21] |
| cyanohydrin glycosides (linamarin) and flavonoids (quercetin and isoquercetin) | [17] | |||||
| Typha latifolia L. | 1 (no data) | 50 (++) | 5.1 × 104 ± 1.1 × 104 | Alternariac (91) Torula (4) Fusarium c (3) Cladosporium c (2) | no data | - |
| II harvest—August | ||||||
| Poaceae | ||||||
| Arrhenatherum elatius (L.) P.Beauv.—generative phase | 9 (7) | 6.25 (++) | 4.0 × 105 ± 1.1 × 105 | Cladosporiumc (28) Acremonium (19) Alternaria c (17) Fusarium c (10) Pythium (6) Mucor c (3) not identified (17) | saponins | [9] |
| Arrhenatherum elatius (L.) P.Beauv.—vegetative phase | 9 (7) | 1.563 (+++) | 4.8 × 104 ± 1.3 × 104 | Alternariac (26) Cladosporium c (14) Mucor c (13) Fusarium c (6) Humicola (5) Rhizopus c (3) Aspergillus c (2) Penicillium c (2) not identified (29) | saponins | [9] |
| Molinia caereluea (L.) Moench | 2 (2) | 25 (++) | 2.0 × 105 ± 4.5 × 104 | Torula (32) Alternaria c (29) Cladosporium c (18) Fusarium c (9) Rhizopus c (9) Pythium (3) | cyanogenic glycosides, ergot | [9] |
| Other species | ||||||
| Odontites serotina (Lam.) Rchb. | 1 (−1) | 12.5 (++) | 8.3 × 104 ± 2.7 × 104 | Humicola (37) Cladosporium c (33) Alternaria c (15) Pythium (3) not identified (12) | iridoid glycosides (aucubin, catalpol), agnuzide | [17] |
| Ostericum palustre Besser | no data | 50 (++) | 1.2 × 105 ± 1.0 × 104 | Cladosporiumc (80) Alternaria c (8) Fusarium c (8) Epicoccum c (1.5) Humicola (1) not identified (1.5) | no data | - |
| Ranunculus sceleratus L. | −1 (no data) | 50 (++) | 1.5 × 104 ± 1.9 × 103 | Alternariac (69) Fusarium c (21) Mucor c (10) | glycoside (ranunculin), protoanemonin and anemonin | [22] |
| Rhinanthus serotinus (Schönh) Oborny | 1 (−1) | 25 (++) | 4.0 × 104 ± 9.8 × 103 | Humicola (56) Cladosporium c (17) Alternaria c (8) Stemphylium (2) Epicoccum c (1) Mucor c (1) not identified (15) | aucubin derivatives | [17] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Twarużek, M.; Dembek, R.; Pańka, D.; Soszczyńska, E.; Zastempowska, E.; Grajewski, J. Evaluation of Cytotoxicity and Mould Contamination of Selected Plants from Meadows Covered by the Agri-Environmental Program. Toxins 2019, 11, 228. https://doi.org/10.3390/toxins11040228
Twarużek M, Dembek R, Pańka D, Soszczyńska E, Zastempowska E, Grajewski J. Evaluation of Cytotoxicity and Mould Contamination of Selected Plants from Meadows Covered by the Agri-Environmental Program. Toxins. 2019; 11(4):228. https://doi.org/10.3390/toxins11040228
Chicago/Turabian StyleTwarużek, Magdalena, Romuald Dembek, Dariusz Pańka, Ewelina Soszczyńska, Ewa Zastempowska, and Jan Grajewski. 2019. "Evaluation of Cytotoxicity and Mould Contamination of Selected Plants from Meadows Covered by the Agri-Environmental Program" Toxins 11, no. 4: 228. https://doi.org/10.3390/toxins11040228
APA StyleTwarużek, M., Dembek, R., Pańka, D., Soszczyńska, E., Zastempowska, E., & Grajewski, J. (2019). Evaluation of Cytotoxicity and Mould Contamination of Selected Plants from Meadows Covered by the Agri-Environmental Program. Toxins, 11(4), 228. https://doi.org/10.3390/toxins11040228






