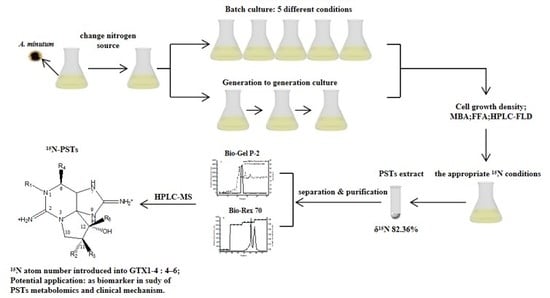
15N Stable Isotope Labeling PSTs in Alexandrium minutum for Application of PSTs as Biomarker
Abstract
1. Introduction
2. Results
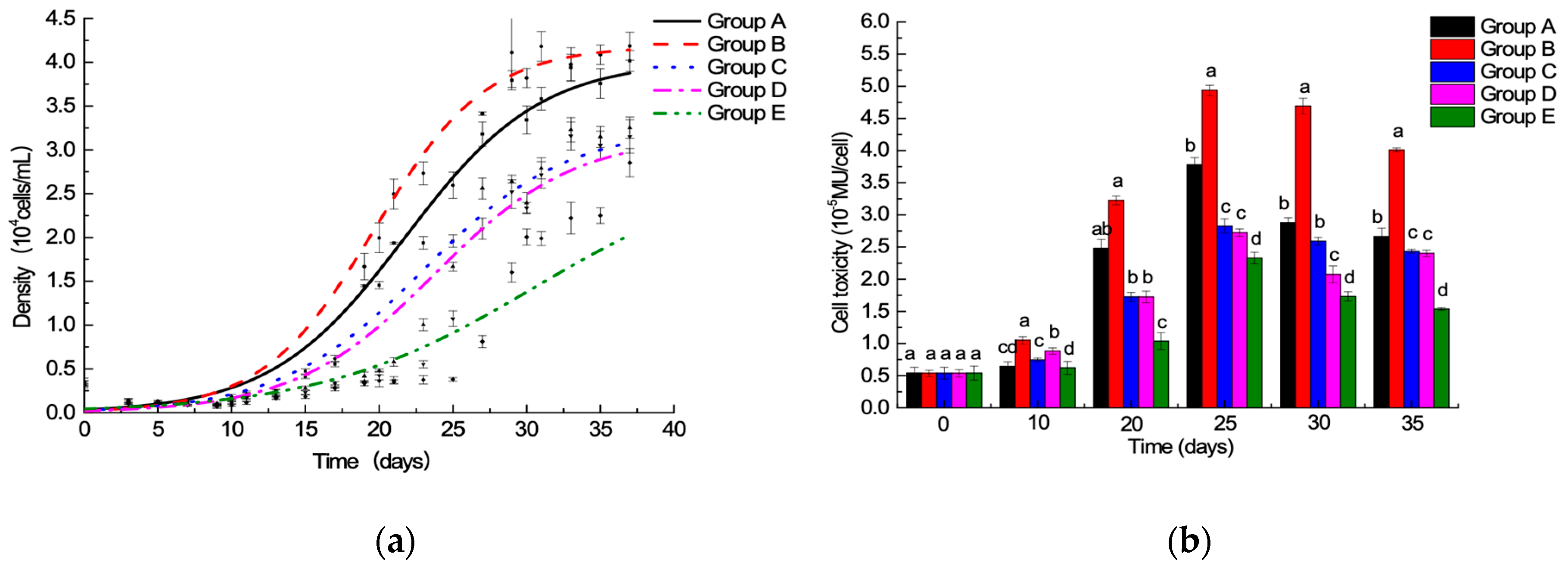
2.1. Batch Culture: 15N/P Influence in Algal Growth and Toxicity
2.2. Generation to Generation Culture: 15N/P Influence in Algal Growth and Toxicity
2.3. 15N/P Effect on Algae PSTs Profile
2.4. 15N Labeling Abundance Change of 15N-PSTs
15N atom% = 2/(I29/I30 + 2) × 100 (15N atom% > 10%)
2.5. The Efficiency of 15N-PSTs Separation and Purification
2.6. 15N Atom Number Identification of 15N-PSTs
3. Discussion
3.1. Effect 15N/P of on A. minutum Culture
3.2. The Replacement of Stable Isotope 15N
4. Conclusions
5. Materials and Methods
5.1. Chemicals and Analytical Standards
5.2. Algal Culture
5.3. 15N-PSTs Extraction
5.4. 15N-PSTs Separation and Purification
5.5. 15N Abundance Analysis of 15N-PSTs Extracts
5.6. PSTs Toxicity Test by the Mouse Bioassay
5.7. 15N-PSTs Detection by the Fast Fluorimetric Assay (FFA)
5.8. 15N-PSTs Determination by HPLC-FLD
5.9. 15N-PSTs Quantification by HPLC-MS
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hoagland, P.A.; Anderson, D.M.; Kaoru, Y.; White, A.W. The Economic Effects of Harmful Algal Blooms in the United States: Estimates, Assessment Issues, and Information Needs. Estuaries 2002, 25, 819–837. [Google Scholar] [CrossRef]
- Gustaafm, H. Ocean climate change, phytoplankton community responses, and harmful algal blooms: A formidable predictive challenge. J. Phycol. 2010, 46, 220–235. [Google Scholar] [CrossRef]
- Rolton, A.; Vignier, J.; Soudant, P.; Shumway, S.E.; Bricelj, V.M.; Volety, A.K. Effects of the red tide dinoflagellate, Karenia brevis, on early development of the eastern oyster Crassostrea virginica and northern quahog Mercenaria mercenaria. Aquat. Toxicol. 2014, 155, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Brooks, B.W.; Lazorchak, J.M.; Howard, M.D.; Johnson, M.V.; Morton, S.L.; Perkins, D.A.; Reavie, E.D.; Scott, G.I.; Smith, S.A.; Steevens, J.A. Are harmful algal blooms becoming the greatest inland water quality threat to public health and aquatic ecosystems? Environ. Toxicol. Chem. 2016, 35, 6–13. [Google Scholar] [CrossRef] [PubMed]
- Grattan, L.M.; Holobaugh, S.; Morris, J.G. Harmful algal blooms and public health. Harmful Algae 2016, 57, 2–8. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhang, S.F.; Lin, L.; Wang, D.Z. Whole Transcriptomic Analysis Provides Insights into Molecular Mechanisms for Toxin Biosynthesis in a Toxic Dinoflagellate Alexandrium catenella (ACHK-T). Toxins 2017, 9, 213. [Google Scholar] [CrossRef]
- Li, A.; Chen, H.; Qiu, J.; Lin, H.; Gu, H. Determination of multiple toxins in whelk and clam samples collected from the Chukchi and Bering seas. Toxicon 2016, 109, 84–93. [Google Scholar] [CrossRef]
- Jiang, T.; Xu, Y.; Li, Y.; Jiang, T.; Wu, F.; Zhang, F. Seasonal dynamics of Alexandrium tamarense and occurrence of paralytic shellfish poisoning toxins in bivalves in Nanji Islands, East China Sea. Mar. Freshw. Res. 2014, 65, 350. [Google Scholar] [CrossRef]
- Fabioux, C.; Sulistiyani, Y.; Haberkorn, H.; Hegaret, H.; Amzil, Z.; Soudant, P. Exposure to toxic Alexandrium minutum activates the detoxifying and antioxidant systems in gills of the oyster Crassostrea gigas. Harmful Algae 2015, 48, 55–62. [Google Scholar] [CrossRef]
- Xie, W.; Liu, X.; Yang, X.; Zhang, C.; Bian, Z. Accumulation and depuration of paralytic shellfish poisoning toxins in the oyster Ostrea rivularis Gould—Chitosan facilitates the toxin depuration. Food Control 2013, 30, 446–452. [Google Scholar] [CrossRef]
- Ding, L.; Qiu, J.; Li, A. Proposed Biotransformation Pathways for New Metabolites of Paralytic Shellfish Toxins Based on Field and Experimental Mussel Samples. J. Agric. Food Chem. 2017, 65, 5494–5502. [Google Scholar] [CrossRef] [PubMed]
- Mat, A.M.; Klopp, C.; Payton, L.; Jeziorski, C.; Chalopin, M.; Amzil, Z.; Tran, D.; Wikfors, G.H.; Hegaret, H.; Soudant, P.; et al. Oyster transcriptome response to Alexandrium exposure is related to saxitoxin load and characterized by disrupted digestion, energy balance, and calcium and sodium signaling. Aquat. Toxicol. 2018, 199, 127–137. [Google Scholar] [CrossRef] [PubMed]
- Han, S.; Yan, S.; Chen, K.; Zhang, Z.; Zed, R.; Zhang, J.; Song, W.; Liu, H. 15N isotope fractionation in an aquatic food chain: Bellamya aeruginosa (Reeve) as an algal control agent. J. Environ. Sci. 2010, 22, 242–247. [Google Scholar] [CrossRef]
- Chahrour, O.; Cobice, D.; Malone, J. Stable isotope labelling methods in mass spectrometry-based quantitative proteomics. J. Pharm. Biomed. Anal. 2015, 113, 2–20. [Google Scholar] [CrossRef] [PubMed]
- Brandsma, J.; Bailey, A.P.; Koster, G.; Gould, A.P.; Postle, A.D. Stable isotope analysis of dynamic lipidomics. Biochim. Biophys. Acta 2017, 1862, 792–796. [Google Scholar] [CrossRef][Green Version]
- Schneider, L.; Maher, W.A.; Potts, J.; Taylor, A.M.; Batley, G.E.; Krikowa, F.; Adamack, A.; Chariton, A.A.; Gruber, B. Trophic transfer of metals in a seagrass food web: Bioaccumulation of essential and non-essential metals. Mar. Pollut. Bull. 2018, 131, 468. [Google Scholar] [CrossRef] [PubMed]
- He, Y.-L.; Luo, Y.-B.; Chen, H.; Hou, H.-W.; Hu, Q.-Y. Research Progress in Analysis of Small Molecule Metabolites in Bio-matrices by Stable Isotope Coded Derivatization Combining with Liquid Chromatography–tandem Mass Spectrometry. Chin. J. Anal. Chem. 2017, 45, 1066–1077. [Google Scholar] [CrossRef]
- Thomas, M.; Huck, N.; Hoehenwarter, W.; Conrath, U.; Beckers, G.J.M. Combining Metabolic 15N Labeling with Improved Tandem MOAC for Enhance; Springer: New York, NY, USA, 2015; pp. 81–96. [Google Scholar]
- Lehmann, W.D. A timeline of stable isotopes and mass spectrometry in the life sciences. Mass Spectrom. Rev. 2016, 36. [Google Scholar] [CrossRef]
- Bequette, B.J.; Backwell, F.R.C.; Calder, A.G.; Metcalf, J.A.; Beever, D.E.; Macrae, J.C.; Lobley, G.E. Application of a U-13C-Labeled Amino Acid Tracer in Lactating Dairy Goats for Simultaneous Measurements of the Flux of Amino Acids in Plasma and the Partition of Amino Acids to the Mammary Gland. J. Dairy Sci. 1997, 80, 2842–2853. [Google Scholar] [CrossRef]
- Cox, J.; Kyle, D.; Radmer, R.; Delente, J. Stable-isotope-labeled biochemicals from microalgae. Trends Biotechnol. 1988, 6, 279–282. [Google Scholar] [CrossRef]
- Fernández, F.G.A.; Alias, C.B.; Pérez, J.A.S.; Sevilla, J.M.F.; González, M.J.I.; Grima, E.M. Production of 13C polyunsaturated fatty acids from the microalga Phaeodactylum tricornutum. J. Appl. Phycol. 2003, 15, 229–237. [Google Scholar] [CrossRef]
- Acien Fernandez, F.G.; Fernandez Sevilla, J.M.; Egorova-Zachernyuk, T.A.; Molina Grima, E. Cost-effective production of 13C, 15N stable isotope-labelled biomass from phototrophic microalgae for various biotechnological applications. Biomol. Eng. 2005, 22, 193–200. [Google Scholar] [CrossRef] [PubMed]
- Carcelén, J.N.; Marchante-Gayón, J.M.; González, P.R.; Valledor, L.; Cañal, M.J.; Alonso, J.I.G. A cost-effective approach to produce 15N-labelled amino acids employing Chlamydomonas reinhardtii CC503. Microb. Cell Factories 2017, 16, 146. [Google Scholar] [CrossRef] [PubMed]
- Wiese, M.; D’Agostino, P.M.; Mihali, T.K.; Moffitt, M.C.; Neilan, B.A. Neurotoxic alkaloids: Saxitoxin and its analogs. Mar. Drugs 2010, 8, 2185–2211. [Google Scholar] [CrossRef] [PubMed]
- Hwang, D.F.; Lu, Y.H. Influence of environmental and nutritional factors on growth, toxicity, and toxin profile of dinoflagellate Alexandrium minutum. Toxicon 2000, 38, 1491–1503. [Google Scholar] [CrossRef]
- Hui, G.; Jianting, Y.; Zhongmin, S.; Delin, D. Effects of salinity and nutrients on the growth and chlorophyll fluorescence of Caulerpa lentillifera. Chin. J. Oceanol. Limnol. 2015, 33, 410–418. [Google Scholar] [CrossRef]
- Boyer, G.L.; Sullivan, J.J.; Andersen, R.J.; Harrison, P.J.; Taylor, F.J.R. Effects of nutrient limitation on toxin production and composition in the marine dinoflagellate Protogonyaulax tamarensis. Mar. Boil. 1987, 96, 123–128. [Google Scholar] [CrossRef]
- Hamasaki, K.; Horie, M.; Tokimitsu, S.; Toda, T.; Taguchi, S. Variability in Toxicity of the Dinoflagellate Alexandrium Tamarense Isolated from Hiroshima Bay, Western Japan, as a Reflection of Changing Environmental Conditions. J. Plankton Res. 2001, 23, 271–278. [Google Scholar] [CrossRef]
- Wang, D.-Z.; Hsieh, D.P.H. Growth and toxin production in batch cultures of a marine dinoflagellate Alexandrium tamarense HK9301 isolated from the South China Sea. Harmful Algae 2005, 4, 401–410. [Google Scholar] [CrossRef]
- Anderson, D.M.; Kulis, D.M.; Sullivan, J.J.; Hall, S.; Lee, C. Dynamics and physiology of saxitoxin production by the dinoflagellates Alexandrium spp. Mar. Boil. 1990, 104, 511–524. [Google Scholar] [CrossRef]
- Dell’Aversano, C.; Hess, P.; Quilliam, M.A. Hydrophilic interaction liquid chromatography–mass spectrometry for the analysis of paralytic shellfish poisoning (PSP) toxins. J. Chromatogr. A 2005, 1081, 190–201. [Google Scholar] [CrossRef] [PubMed]
- Guéguen, M.; Baron, R.; Bardouil, M.; Truquet, P.; Haberkorn, H.; Lassus, P.; Barillé, L.; Amzil, Z. Modelling of paralytic shellfish toxin biotransformations in the course of Crassostrea gigas detoxification kinetics. Ecol. Model. 2011, 222, 3394–3402. [Google Scholar] [CrossRef]
- Brandsma, J.; Postle, A.D. Analysis of the regulation of surfactant phosphatidylcholine metabolism using stable isotopes. Ann. Anat. Anat. Anz. 2017, 211, 176–183. [Google Scholar] [CrossRef] [PubMed]
- Cho, Y.; Tsuchiya, S.; Omura, T.; Koike, K.; Oikawa, H.; Konoki, K.; Oshima, Y.; Yotsu-Yamashita, M. Metabolomic study of saxitoxin analogues and biosynthetic intermediates in dinoflagellates using (15)N-labelled sodium nitrate as a nitrogen source. Sci. Rep. 2019, 9, 3460. [Google Scholar] [CrossRef] [PubMed]
- Roelke, D.L.; Eldridge, P.M.; Cifuentes, L.A. A Model of Phytoplankton Competition for Limiting and Nonlimiting Nutrients: Implications for Development of Estuarine and Nearshore Management Schemes. Estuaries 1999, 22, 92–104. [Google Scholar] [CrossRef]
- Hii, K.S.; Lim, P.T.; Kon, N.F.; Takata, Y.; Usup, G.; Leaw, C.P. Physiological and transcriptional responses to inorganic nutrition in a tropical Pacific strain of Alexandrium minutum: Implications for the saxitoxin genes and toxin production. Harmful Algae 2016, 56, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Lim, P.T.; Leaw, C.P.; Kobiyama, A.; Ogata, T. Growth and toxin production of tropical Alexandrium minutum Halim (Dinophyceae) under various nitrogen to phosphorus ratios. J. Appl. Phycol. 2010, 22, 203–210. [Google Scholar] [CrossRef]
- Sauer, M.L.A.; Xu, B.; Sutton, F. Metabolic labeling with stable isotope nitrogen ( 15 N) to follow amino acid and protein turnover of three plastid proteins in Chlamydomonas reinhardtii. Proteome Sci. 2014, 12, 1–9. [Google Scholar] [CrossRef]
- Chou, H.N.; Chen, Y.M.; Chen, C.Y. Variety of PSP toxins in four culture strains of Alexandrium minutum collected from southern Taiwan. Toxicon 2004, 43, 337–340. [Google Scholar] [CrossRef]
- Miao, Y.P.; Zhou, H.N.; Wen, R. Isolation and purification of gonyautoxins from Alexandrium mimutum Halim. Acta Pharm. Sin. 2004, 39, 52–55. [Google Scholar] [CrossRef]
- Laycock, M.V.; Thibault, P.; Ayer, S.W.; Walter, J.A. Isolation and purification procedures for the preparation of paralytic shellfish poisoning toxin standards. Neurogastroenterol. Motil. 2010, 2, 175–183. [Google Scholar] [CrossRef]
- AOAC. AOAC Official Method 959.08: Paralytic Shellfish Poison, Biological Method; AOAC: Arlington, VA, USA, 2000. [Google Scholar]
- Gerdts, G.; Hummert, C.; Donner, G.; Luckas, B.; Schütt, C. A fast fluorimetric assay (FFA) for the detection of saxitoxin in natural phytoplankton samples. Mar. Ecol. Prog. 2002, 230, 29–34. [Google Scholar] [CrossRef]
- Van De Riet, J.; Gibbs, R.S.; Muggah, P.M.; Rourke, W.A.; Macneil, J.D.; Quilliam, M.A. Liquid chromatography post-column oxidation (PCOX) method for the determination of paralytic shellfish toxins in mussels, clams, oysters, and scallops: Collaborative study. J. AOAC Int. 2011, 94, 1154–1176. [Google Scholar] [PubMed]
| Culture Method | 1.0 Time of the f/2 Medium Standard 15N/P Concentration | 1.5 Times of the f/2 Medium Standard 15N/P Concentration | |
|---|---|---|---|
| PSTs 15N Abundance (Atom%) | |||
| Batch culture/d | 0 | 0.47 | 0.47 |
| 20 | 26.26 | 26.43 | |
| 30 | 57.16 | 70.26 | |
| Generation to generation culture/generation | 1 | 37.60 | - |
| 2 | 58.32 | - | |
| 3 | 62.46 | - | |
| No. n generation | - | 82.36 | |
| Toxin | Molecular Formula | [M+H]+ | Fragment Ion | Loss of | Fragment Ion after Labeling |
|---|---|---|---|---|---|
| GTX1 | C10H17N7O9S | 412 | 332; 314 | -SO3; -SO3-H2O | 334.3; 318.3; 319.3 |
| GTX4 | C10H17N7O9S | 412 | 332; 314;253 | -SO3; -SO3-H2O; -SO3-H2O-NH3-CO2 | 318.3; 319.3; 338.3 |
| GTX2 | C10H17N7O8S | 396 | 316; 298 | -SO3; -SO3-H2O; | 322.1; 321.1; 305.1 |
| GTX3 | C10H17N7O8S | 396 | 316; 298; 220 | -SO3; -SO3-H2O; -SO3-2H2O-NH3-NHCO | 302.3; 303.3 |
| Reagent | Working Solution (mg/L) | Stock Solution (g/L) | |
|---|---|---|---|
| A: | NaNO3 | 75 | 75 |
| B: | NaH2PO4·H2O | 5 | 5 |
| C: | Na2SiO3·9H2O | 20 | 20 |
| D: | Na2EDTA | 4.36 | 4.36 |
| E: | FeCl3·6H2O | 3.16 | 3.16 |
| F: | CuSO4·5H2O | 0.01 | 0.01 |
| ZnSO4·7H2O | 0.023 | 0.023 | |
| CoCL2·6H2O | 0.012 | 0.012 | |
| MnCL2·4H2O | 0.18 | 0.18 | |
| Na2MoO4·2H2O | 0.07 | 0.07 | |
| G: | Vitamin B1 | 0.1 | 0.01 |
| Vitamin B12 | 0.5 × 10−3 | 0.5 × 10−4 | |
| Vitamin H | 0.5 × 10−3 | 0.5 × 10−4 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xie, W.; Li, M.; Song, L.; Zhang, R.; Hu, X.; Liang, C.; Yang, X. 15N Stable Isotope Labeling PSTs in Alexandrium minutum for Application of PSTs as Biomarker. Toxins 2019, 11, 211. https://doi.org/10.3390/toxins11040211
Xie W, Li M, Song L, Zhang R, Hu X, Liang C, Yang X. 15N Stable Isotope Labeling PSTs in Alexandrium minutum for Application of PSTs as Biomarker. Toxins. 2019; 11(4):211. https://doi.org/10.3390/toxins11040211
Chicago/Turabian StyleXie, Wancui, Min Li, Lin Song, Rui Zhang, Xiaoqun Hu, Chengzhu Liang, and Xihong Yang. 2019. "15N Stable Isotope Labeling PSTs in Alexandrium minutum for Application of PSTs as Biomarker" Toxins 11, no. 4: 211. https://doi.org/10.3390/toxins11040211
APA StyleXie, W., Li, M., Song, L., Zhang, R., Hu, X., Liang, C., & Yang, X. (2019). 15N Stable Isotope Labeling PSTs in Alexandrium minutum for Application of PSTs as Biomarker. Toxins, 11(4), 211. https://doi.org/10.3390/toxins11040211