Arecoline Promotes Migration of A549 Lung Cancer Cells through Activating the EGFR/Src/FAK Pathway
Abstract
1. Introduction
2. Results
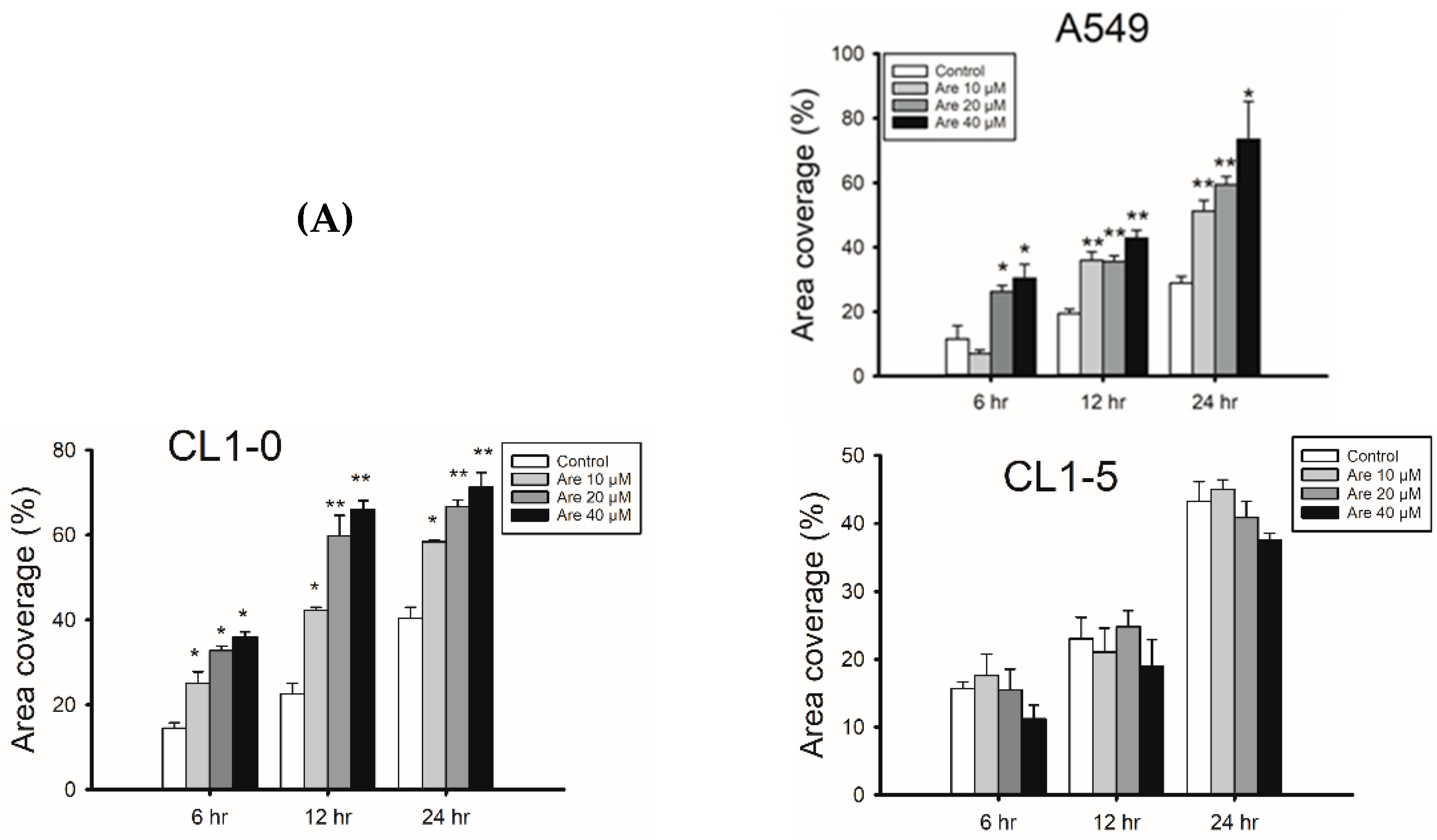
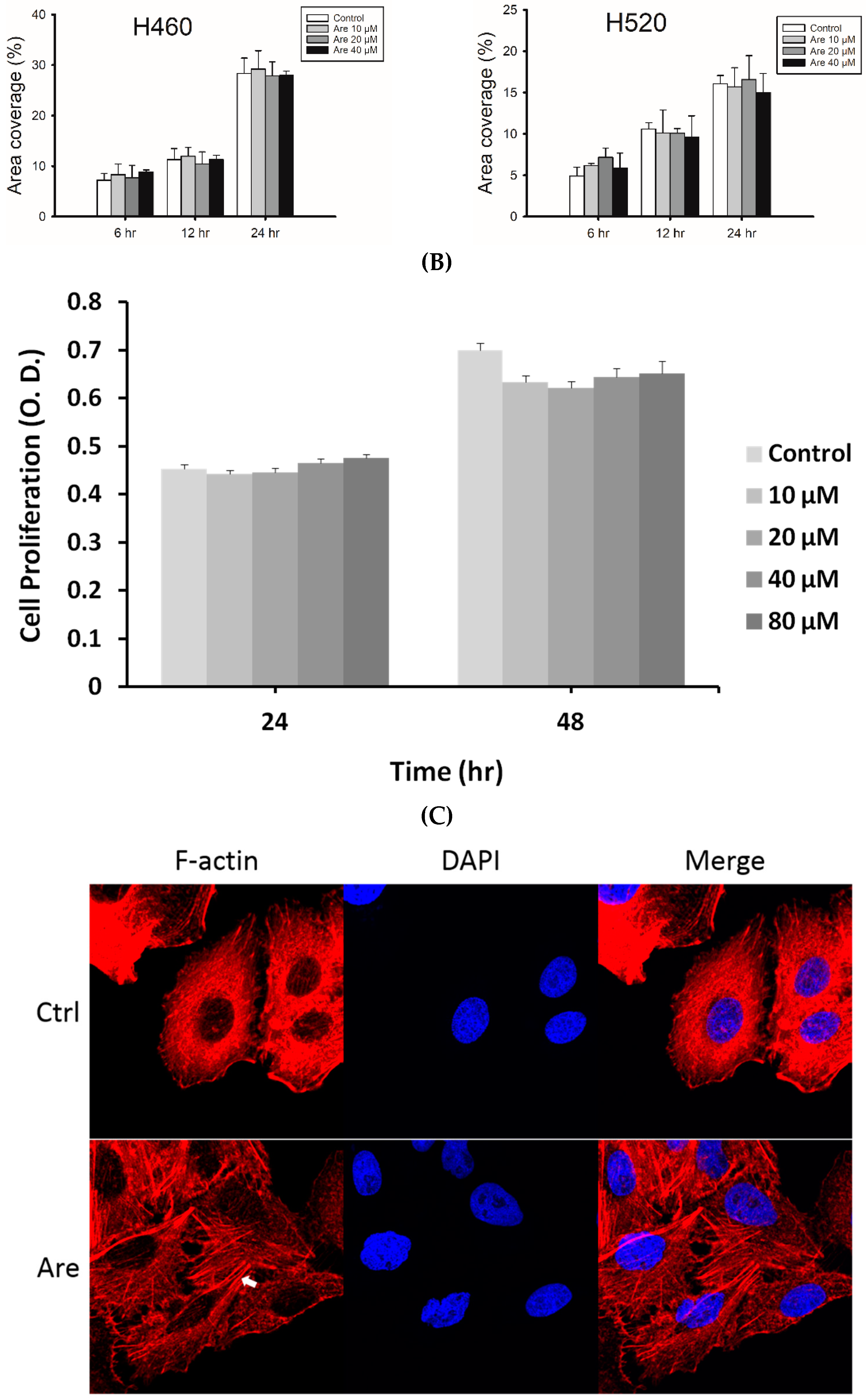
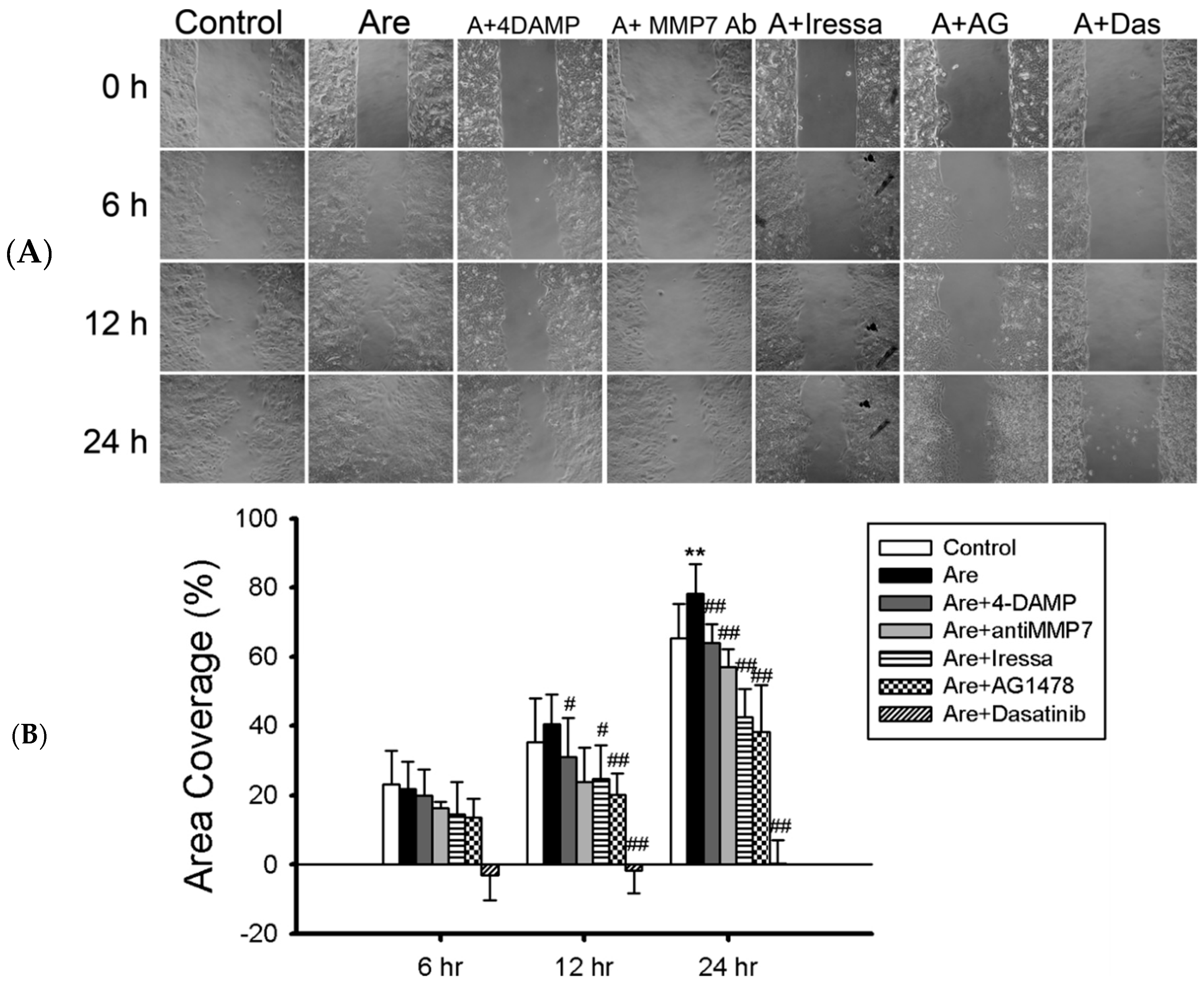
2.1. Arecoline Promotes Migration in the A549 Lung Cancer Cell Line
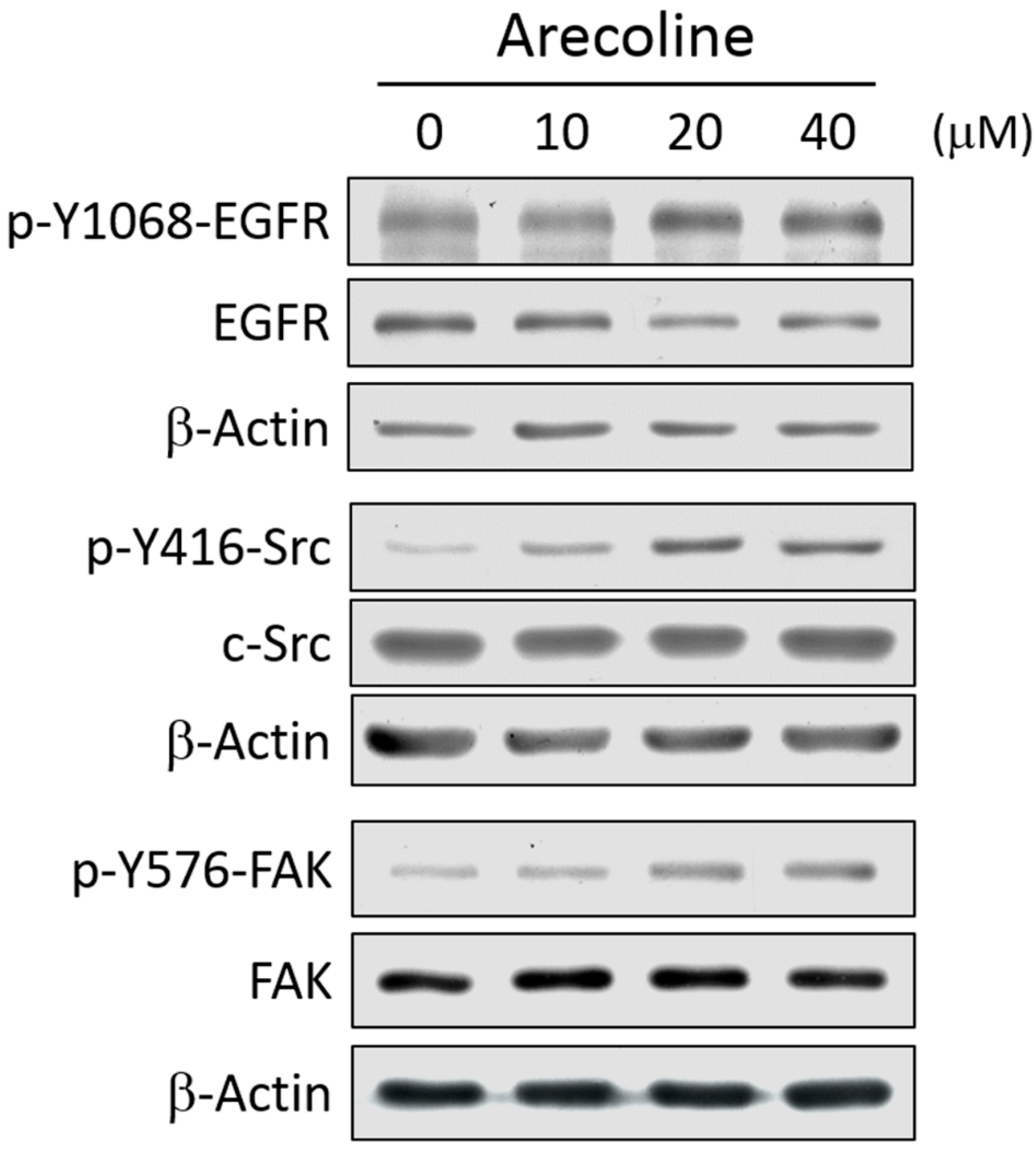
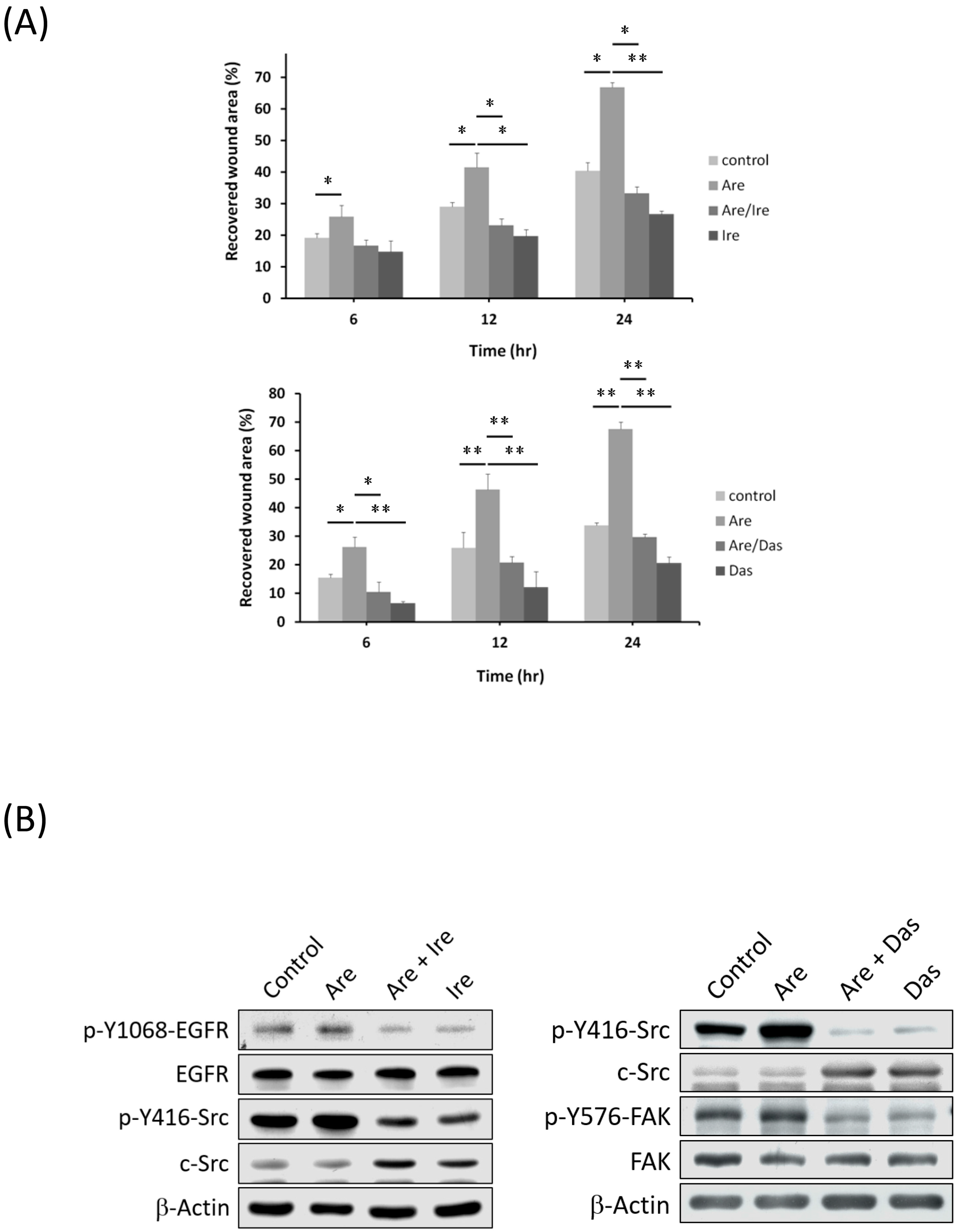
2.2. Arecoline Treatment Activates the EGFR/c-Src/FAK Signaling Pathway in the A549 Cell Line
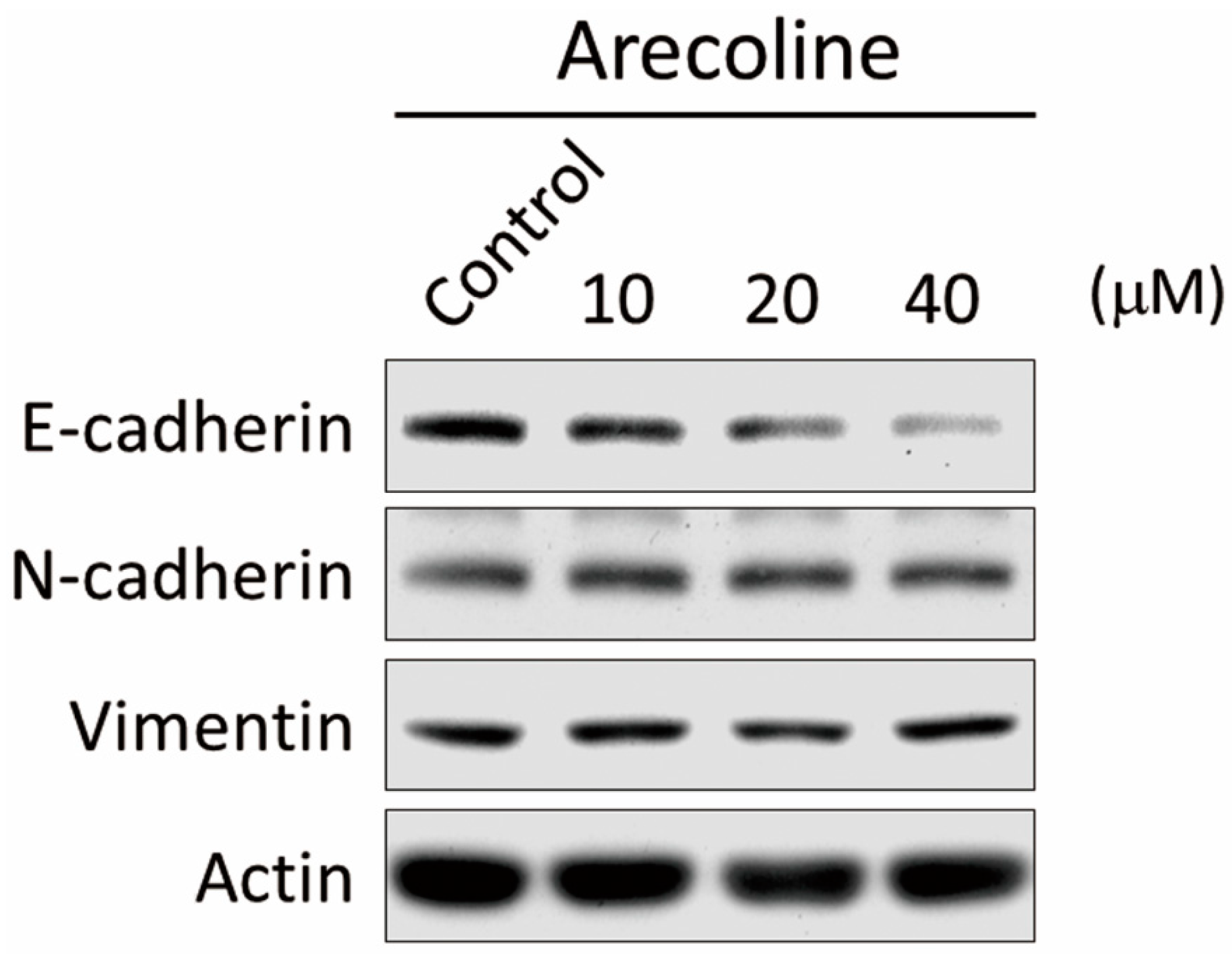
2.3. Arecoline Stimulates Epithelial–Mesenchymal Transition (EMT) Markers in the A549 Cell Line
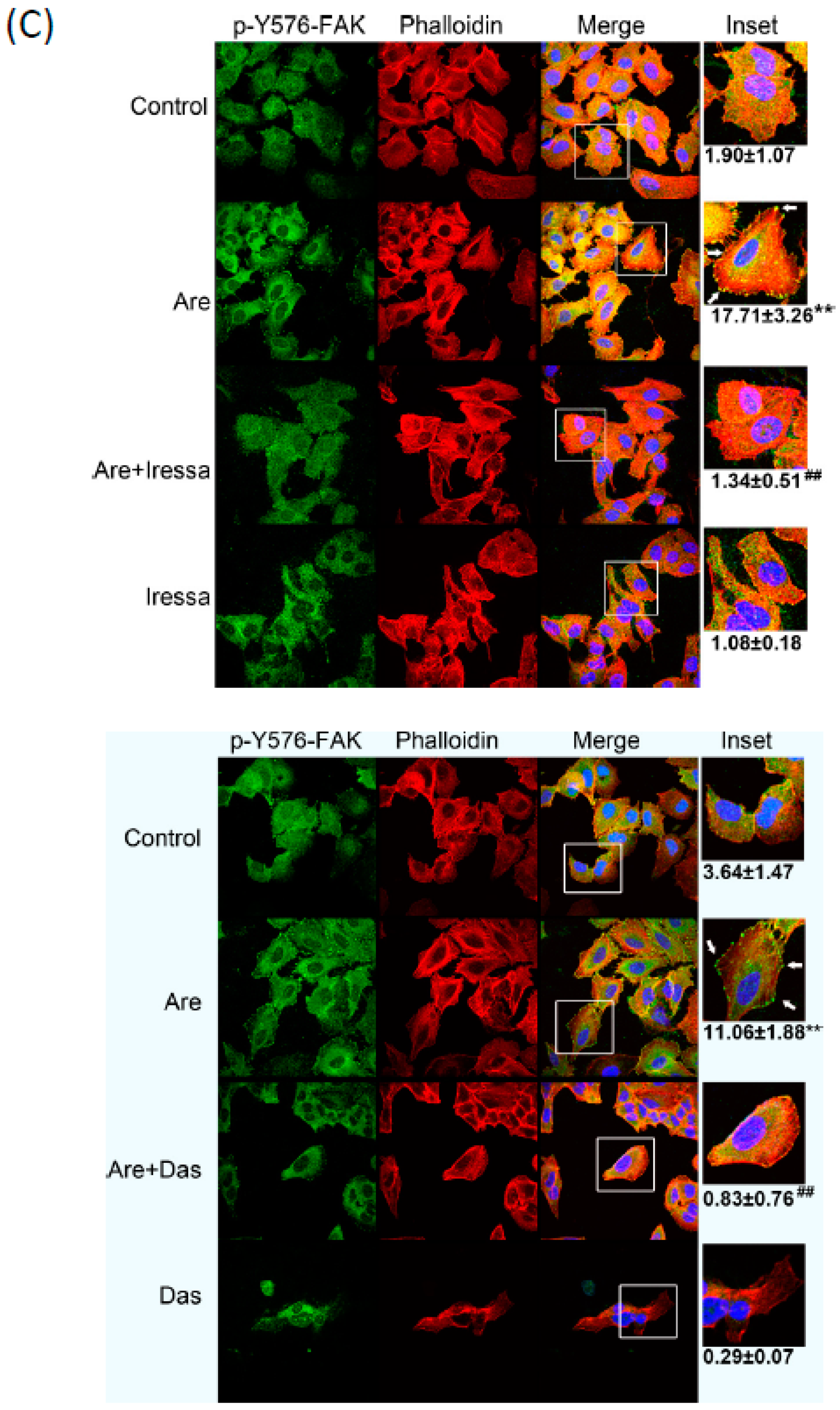
2.4. The Inhibition of EGFR or c-Src Activation Reversed Arecoline-Stimulated Migration in the A549 Cell Line
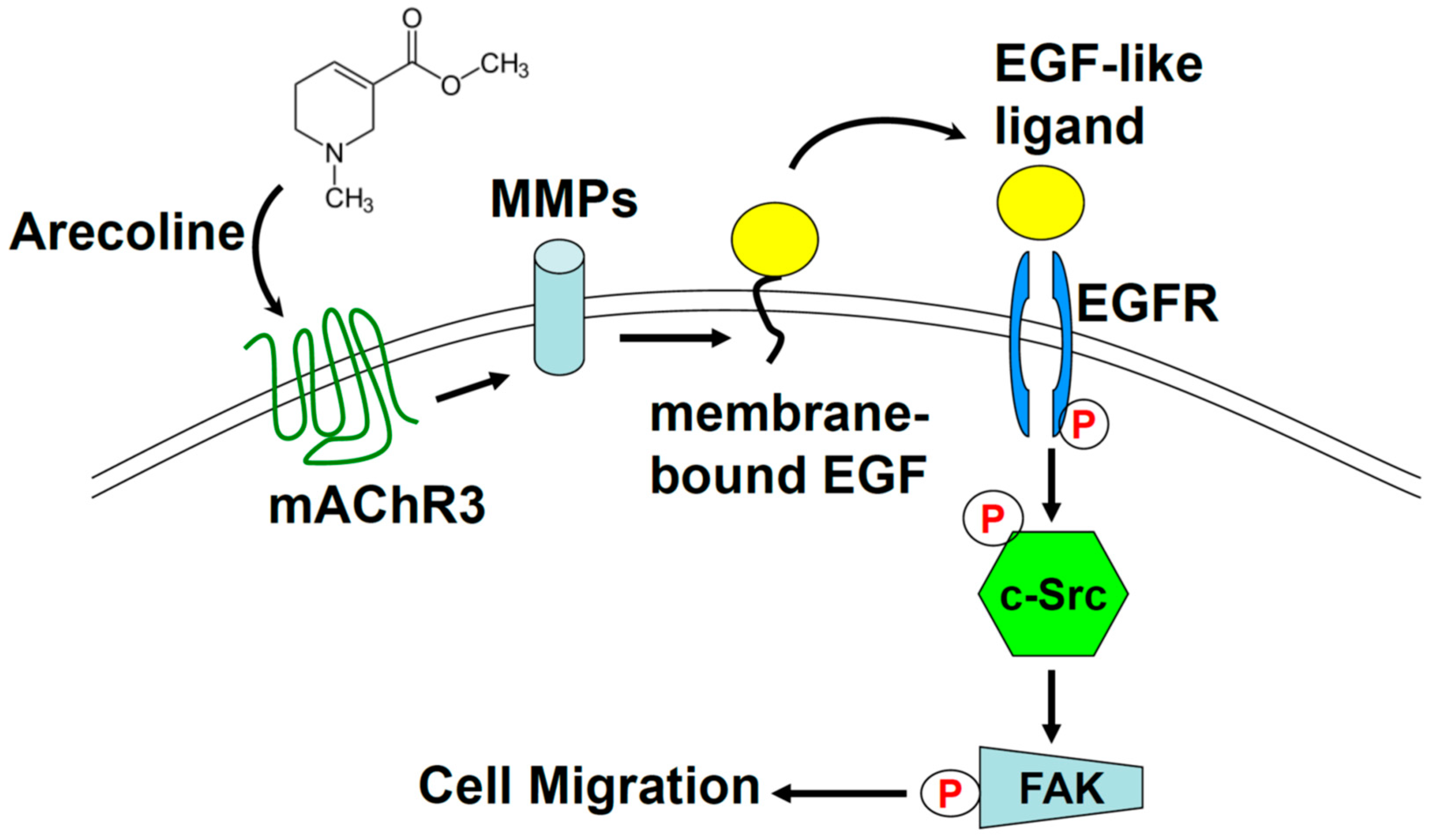
2.5. Arecoline Stimulates Lung Cancer Cell Migration through the Muscarinic Acetylcholine Receptor 3 (mAChR3) Transactivating EGFR Pathway in the A549 Cell line
3. Discussion
4. Materials and Methods
4.1. Cell Lines and Cell Culture
4.2. Cell Viability Assay
4.3. Cell Migration Assay
4.4. Immunoblotting
4.5. Immunostaining
4.6. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| EGF | Epithelial growth factor |
| EGFR | Epithelial growth factor receptor |
| EMT | Epithelial-mesenchymal transition |
| GPCR | G-protein coupled receptor |
| mAChR3 | Muscarinic acetylcholine receptor 3 |
| MMPs | Matrix metalloproteinases |
| RTK | Receptor tyrosine kinase |
References
- Chen, Y.J.; Chang, J.T.; Liao, C.T.; Wang, H.M.; Yen, T.C.; Chiu, C.C.; Lu, Y.C.; Li, H.F.; Cheng, A.J. Head and neck cancer in the betel quid chewing area: Recent advances in molecular carcinogenesis. Cancer Sci. 2008, 99, 1507–1514. [Google Scholar] [CrossRef]
- Garg, A.; Chaturvedi, P.; Gupta, P.C. A review of the systemic adverse effects of areca nut or betel nut. Indian J. Med. Paediatr. Oncol. 2014, 35, 3–9. [Google Scholar] [CrossRef]
- IARC Working Group on the Evaluation of Carcinogenic Risks to Humans. Betel-quid and areca-nut chewing and some areca-nut derived nitrosamines. IARC Monogr. Eval Carcinog Risks Hum. 2004, 85, 1–334. [Google Scholar]
- Bhide, S.V.; Shivapurkar, N.M.; Gothoskar, S.V.; Ranadive, K.J. Carcinogenicity of betel quid ingredients: Feeding mice with aqueous extract and the polyphenol fraction of betel nut. Br. J. Cancer 1979, 40, 922–926. [Google Scholar] [CrossRef] [PubMed]
- Suri, K.; Goldman, H.M.; Wells, H. Carcinogenic effect of a dimethyl sulphoxide extract of betel nut on the mucosa of the hamster buccal pouch. Nature 1971, 230, 383–384. [Google Scholar] [CrossRef]
- Shirname, L.P.; Menon, M.M.; Nair, J.; Bhide, S.V. Correlation of mutagenicity and tumorigenicity of betel quid and its ingredients. Nutr. Cancer 1983, 5, 87–91. [Google Scholar] [CrossRef]
- Ullah, M.; Cox, S.; Kelly, E.; Boadle, R.; Zoellner, H. Arecoline is cytotoxic for human endothelial cells. J. Oral Pathol. Med. 2014, 43, 761–769. [Google Scholar] [CrossRef]
- Wu, I.C.; Chen, P.H.; Wang, C.J.; Wu, D.C.; Tsai, S.M.; Chao, M.R.; Chen, B.H.; Lee, H.H.; Lee, C.H.; Ko, Y.C. Quantification of blood betel quid alkaloids and urinary 8-hydroxydeoxyguanosine in humans and their association with betel chewing habits. J. Anal. Toxicol. 2010, 34, 325–331. [Google Scholar] [CrossRef] [PubMed]
- Jeng, J.H.; Hahn, L.J.; Lin, B.R.; Hsieh, C.C.; Chan, C.P.; Chang, M.C. Effects of areca nut, inflorescence piper betle extracts and arecoline on cytotoxicity, total and unscheduled DNA synthesis in cultured gingival keratinocytes. J. Oral Pathol. Med. 1999, 28, 64–71. [Google Scholar] [CrossRef]
- Chandra, J.N.; Malviya, M.; Sadashiva, C.T.; Subhash, M.N.; Rangappa, K.S. Effect of novel arecoline thiazolidinones as muscarinic receptor 1 agonist in Alzheimer’s dementia models. Neurochem. Int. 2008, 52, 376–383. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.Y.; Liu, Y.C.; Huang, W.T.; Huang, G.C.; Chen, T.C.; Lin, M.H. Up-regulation of matrix metalloproteinase-8 by betel quid extract and arecoline and its role in 2d motility. Oral Oncol. 2007, 43, 1026–1033. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.H.; Liu, S.Y.; Lin, M.H.; Chiang, W.F.; Chen, T.C.; Huang, W.T.; Chou, D.S.; Chiu, C.T.; Liu, Y.C. Upregulation of matrix metalloproteinase-1 (mmp-1) expression in oral carcinomas of betel quid (bq) users: Roles of bq ingredients in the acceleration of tumor cell motility through mmp-1. Arch. Oral Biol. 2008, 53, 810–818. [Google Scholar] [CrossRef]
- Belo, A.; Cheng, K.; Chahdi, A.; Shant, J.; Xie, G.; Khurana, S.; Raufman, J.P. Muscarinic receptor agonists stimulate human colon cancer cell migration and invasion. Am. J. Physiol. Gastrointest Liver Physiol. 2011, 300, G749–G760. [Google Scholar] [CrossRef] [PubMed]
- Xie, G.; Cheng, K.; Shant, J.; Raufman, J.P. Acetylcholine-induced activation of m3 muscarinic receptors stimulates robust matrix metalloproteinase gene expression in human colon cancer cells. Am. J. Physiol. Gastrointest Liver Physiol. 2009, 296, G755–G763. [Google Scholar] [CrossRef]
- Xu, R.; Shang, C.; Zhao, J.; Han, Y.; Liu, J.; Chen, K.; Shi, W. Activation of m3 muscarinic receptor by acetylcholine promotes non-small cell lung cancer cell proliferation and invasion via egfr/pi3k/akt pathway. Tumour Biol. 2015, 36, 4091–4100. [Google Scholar] [CrossRef]
- Finn, R.S. Targeting src in breast cancer. Ann. Oncol. 2008, 19, 1379–1386. [Google Scholar] [CrossRef] [PubMed]
- Ishizawar, R.; Parsons, S.J. C-src and cooperating partners in human cancer. Cancer Cell 2004, 6, 209–214. [Google Scholar] [CrossRef] [PubMed]
- Lin, G.; Sun, L.; Wang, R.; Guo, Y.; Xie, C. Overexpression of muscarinic receptor 3 promotes metastasis and predicts poor prognosis in non-small-cell lung cancer. J. Thorac. Oncol. 2014, 9, 170–178. [Google Scholar] [CrossRef]
- Yang, K.; Song, Y.; Tang, Y.B.; Xu, Z.P.; Zhou, W.; Hou, L.N.; Zhu, L.; Yu, Z.H.; Chen, H.Z.; Cui, Y.Y. Machrs activation induces epithelial-mesenchymal transition on lung epithelial cells. BMC Pulm Med. 2014, 14, 53. [Google Scholar] [CrossRef]
- Tang, Z.; Du, R.; Jiang, S.; Wu, C.; Barkauskas, D.S.; Richey, J.; Molter, J.; Lam, M.; Flask, C.; Gerson, S.; et al. Dual met-egfr combinatorial inhibition against t790m-egfr-mediated erlotinib-resistant lung cancer. Br. J. Cancer 2008, 99, 911–922. [Google Scholar] [CrossRef]
- Shen, H.; Yuan, Y.; Sun, J.; Gao, W.; Shu, Y.Q. Combined tamoxifen and gefitinib in non-small cell lung cancer shows antiproliferative effects. Biomed. Pharm. 2010, 64, 88–92. [Google Scholar] [CrossRef] [PubMed]
- Tian, T.; Hao, J.; Xu, A.; Luo, C.; Liu, C.; Huang, L.; Xiao, X.; He, D. Determination of metastasis-associated proteins in non-small cell lung cancer by comparative proteomic analysis. Cancer Sci. 2007, 98, 1265–1274. [Google Scholar] [CrossRef] [PubMed]
- Tsai, H.F.; Huang, C.W.; Chang, H.F.; Chen, J.J.; Lee, C.H.; Cheng, J.Y. Evaluation of egfr and rtk signaling in the electrotaxis of lung adenocarcinoma cells under dGefct-current electric field stimulation. PLoS ONE 2013, 8, e73418. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Togo, S.; Minakata, K.; Gu, T.; Ohashi, R.; Tajima, K.; Murakami, A.; Iwakami, S.; Zhang, J.; Xie, C.; et al. Distinct roles of cholinergic receptors in small cell lung cancer cells. Anticancer Res. 2010, 30, 97–106. [Google Scholar]
- Wu, J.; Zhou, J.; Yao, L.; Lang, Y.; Liang, Y.; Chen, L.; Zhang, J.; Wang, F.; Wang, Y.; Chen, H.; et al. High expression of m3 muscarinic acetylcholine receptor is a novel biomarker of poor prognostic in patients with non-small cell lung cancer. Tumour Biol. 2013, 34, 3939–3944. [Google Scholar] [CrossRef]
- Kuhne, S.; Ockenga, W.; Banning, A.; Tikkanen, R. Cholinergic transactivation of the egfr in hacat keratinocytes stimulates a flotillin-1 dependent mapk-mediated transcriptional response. Int. J. Mol. Sci. 2015, 16, 6447–6463. [Google Scholar] [CrossRef]
- Hao, L.; Du, M.; Lopez-Campistrous, A.; Fernandez-Patron, C. Agonist-induced activation of matrix metalloproteinase-7 promotes vasoconstriction through the epidermal growth factor-receptor pathway. Circ. Res. 2004, 94, 68–76. [Google Scholar] [CrossRef]
- Cheng, K.; Zimniak, P.; Raufman, J.P. Transactivation of the epidermal growth factor receptor mediates cholinergic agonist-induced proliferation of h508 human colon cancer cells. Cancer Res. 2003, 63, 6744–6750. [Google Scholar] [CrossRef]
- Laurent-Puig, P.; Lievre, A.; Blons, H. Mutations and response to epidermal growth factor receptor inhibitors. Clin. Cancer Res. 2009, 15, 1133–1139. [Google Scholar] [CrossRef] [PubMed]
- Sieg, D.J.; Hauck, C.R.; Ilic, D.; Klingbeil, C.K.; Schaefer, E.; Damsky, C.H.; Schlaepfer, D.D. Fak integrates growth-factor and integrin signals to promote cell migration. Nat. Cell Biol. 2000, 2, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Cheng, K.; Xie, G.; Raufman, J.P. Matrix metalloproteinase-7-catalyzed release of hb-egf mediates deoxycholyltaurine-induced proliferation of a human colon cancer cell line. Biochem. Pharmacol. 2007, 73, 1001–1012. [Google Scholar] [CrossRef] [PubMed]
- Puthenedam, M.; Wu, F.; Shetye, A.; Michaels, A.; Rhee, K.J.; Kwon, J.H. Matrilysin-1 (mmp7) cleaves galectin-3 and inhibits wound healing in intestinal epithelial cells. Inflamm. Bowel Dis. 2011, 17, 260–267. [Google Scholar] [CrossRef] [PubMed]
- Raufman, J.P.; Cheng, K.; Saxena, N.; Chahdi, A.; Belo, A.; Khurana, S.; Xie, G. Muscarinic receptor agonists stimulate matrix metalloproteinase 1-dependent invasion of human colon cancer cells. Biochem. Biophys. Res. Commun. 2011, 415, 319–324. [Google Scholar] [CrossRef] [PubMed]
- Pelegrina, L.T.; Lombardi, M.G.; Fiszman, G.L.; Azar, M.E.; Morgado, C.C.; Sales, M.E. Immunoglobulin g from breast cancer patients regulates mcf-7 cells migration and mmp-9 activity by stimulating muscarinic acetylcholine receptors. J. Clin. Immunol. 2013, 33, 427–435. [Google Scholar] [CrossRef] [PubMed]
- Geback, T.; Schulz, M.M.; Koumoutsakos, P.; Detmar, M. Tscratch: A novel and simple software tool for automated analysis of monolayer cell migration assays. Biotechniques 2009, 46, 265–274. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chang, C.-H.; Chen, M.-C.; Chiu, T.-H.; Li, Y.-H.; Yu, W.-C.; Liao, W.-L.; Oner, M.; Yu, C.-T.R.; Wu, C.-C.; Yang, T.-Y.; et al. Arecoline Promotes Migration of A549 Lung Cancer Cells through Activating the EGFR/Src/FAK Pathway. Toxins 2019, 11, 185. https://doi.org/10.3390/toxins11040185
Chang C-H, Chen M-C, Chiu T-H, Li Y-H, Yu W-C, Liao W-L, Oner M, Yu C-TR, Wu C-C, Yang T-Y, et al. Arecoline Promotes Migration of A549 Lung Cancer Cells through Activating the EGFR/Src/FAK Pathway. Toxins. 2019; 11(4):185. https://doi.org/10.3390/toxins11040185
Chicago/Turabian StyleChang, Chih-Hsiang, Mei-Chih Chen, Te-Huan Chiu, Yu-Hsuan Li, Wan-Chen Yu, Wan-Ling Liao, Muhammet Oner, Chang-Tze Ricky Yu, Chun-Chi Wu, Tsung-Ying Yang, and et al. 2019. "Arecoline Promotes Migration of A549 Lung Cancer Cells through Activating the EGFR/Src/FAK Pathway" Toxins 11, no. 4: 185. https://doi.org/10.3390/toxins11040185
APA StyleChang, C.-H., Chen, M.-C., Chiu, T.-H., Li, Y.-H., Yu, W.-C., Liao, W.-L., Oner, M., Yu, C.-T. R., Wu, C.-C., Yang, T.-Y., Teng, C.-L. J., Chiu, K.-Y., Chen, K.-C., Wang, H.-Y., Yue, C.-H., Lai, C.-H., Hsieh, J.-T., & Lin, H. (2019). Arecoline Promotes Migration of A549 Lung Cancer Cells through Activating the EGFR/Src/FAK Pathway. Toxins, 11(4), 185. https://doi.org/10.3390/toxins11040185





