Harmful Effects and Control Strategies of Aflatoxin B1 Produced by Aspergillus flavus and Aspergillus parasiticus Strains on Poultry: Review
Abstract
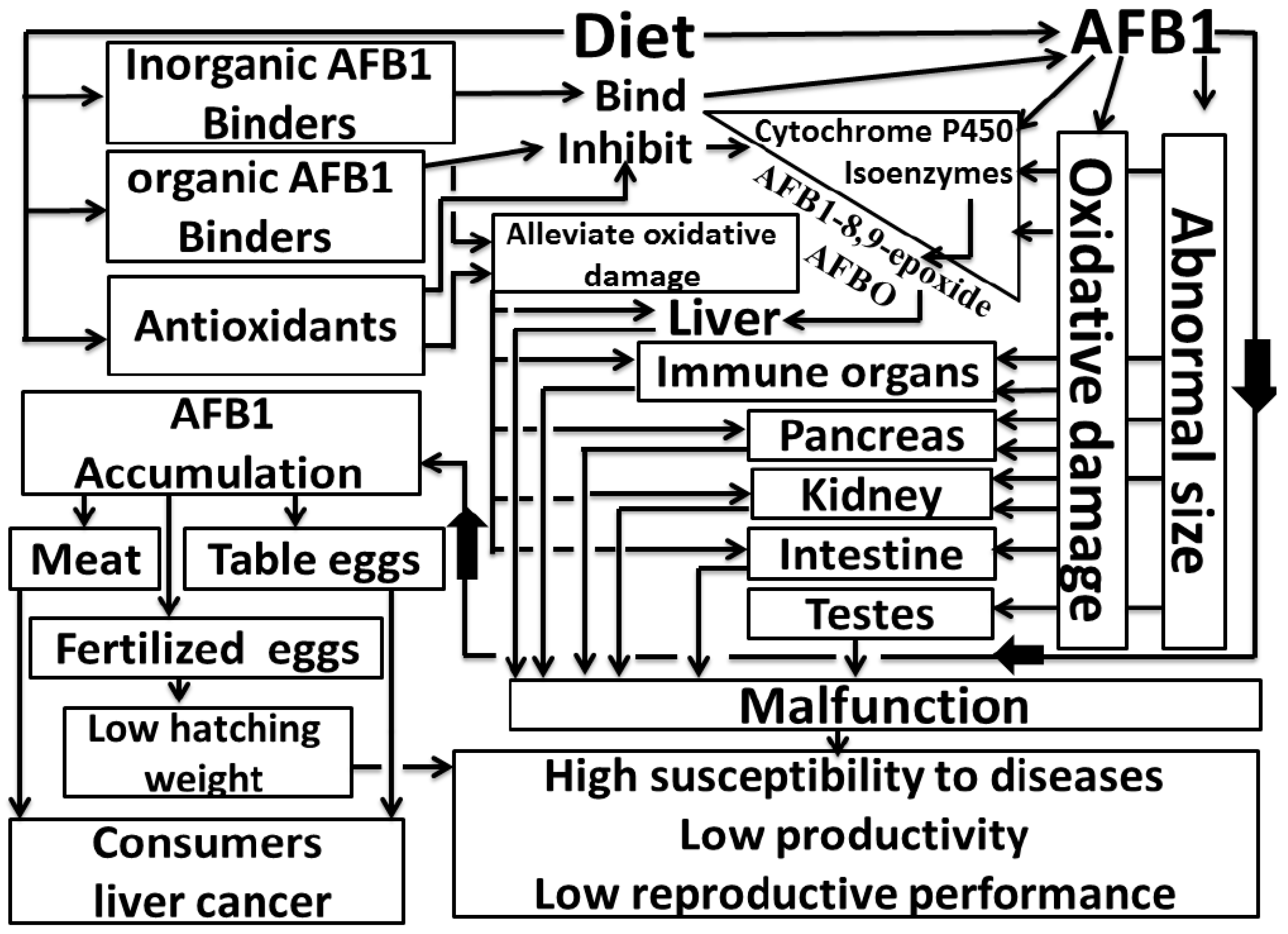
1. Introduction
2. Effects of Aflatoxin B1 Produced by Aspergillus flavus and Aspergillus parasiticus Strains on Poultry
2.1. Productivity
2.2. Egg and Meat Quality
2.3. Bones
2.4. Immune Organs
2.5. Pancreas
2.6. Intestine
2.7. Liver
2.8. Kidney
2.9. Reproductive Organs
3. Nutritional Factors for Counteracting AFB1
3.1. Inorganic AFB1 Binders
3.2. Organic AFB1 Binders
3.2.1. Yeast
3.2.2. Probiotic
3.3. Antioxidants
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Abbasi, F.; Liu, J.; Zhang, H.; Shen, X.; Luo, X. Effects of feeding corn naturally contaminated with aflatoxin on growth performance, apparent ileal digestibility, serum hormones levels and gene expression of Na+, K+-ATPase in ducklings. Asian-Aust. J. Anim. Sci. 2018, 31, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Abdallah, M.F.; Girgin, G.; Baydar, T.; Krska, R.; Sulyok, M. Occurrence of multiple mycotoxins and other fungal metabolites in animal feed and maize samples from Egypt using LC-MS/MS. J. Sci. Food Agric. 2017, 97, 4419–4428. [Google Scholar] [CrossRef]
- Streit, E.; Naehrer, K.; Rodrigues, I.; Schatzmayr, G. Mycotoxin occurrence in feed and feed raw materials worldwide-long term analysis with special focus on Europe and Asia. J. Sci. Food Agric. 2013, 93, 2892–2899. [Google Scholar] [CrossRef]
- Guerre, P. Worldwide mycotoxins exposure in pig and poultry feed formulations. Toxins 2016, 8, 350. [Google Scholar] [CrossRef]
- Arafa, A.S.; Bloomer, R.J.; Wilson, H.R.; Simpson, C.F.; Harms, R.H. Susceptibility of various poultry species to dietary aflatoxin. Br. Poult. Sci. 1981, 22, 431–436. [Google Scholar] [CrossRef] [PubMed]
- Sweeney, M.J.; Dobson, A.D.W. Mycotoxin production by Aspergillus, Fusarium and Penicillium species. Int. J. Food Microbiol. 1998, 43, 141–158. [Google Scholar] [CrossRef]
- Pitt, J.I.; Miller, J.D. A concise history of mycotoxin research. J. Agric. Food Chem. 2017, 65, 7021–7033. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, C.F.; Rosmaninho, J.F.; Castro, A.L.; Butkeraitis, P.; Reis, T.A.; Corrêa, B. Aflatoxin residues in eggs of laying Japanese quail after long-term administration of rations containing low levels of aflatoxin B1. Food Addit. Contam. 2003, 20, 648–653. [Google Scholar] [CrossRef]
- Pandey, I.; Chauhan, S.S. Studies on production performance and toxin residues in tissues and eggs of layer chickens fed on diets with various concentrations of aflatoxin AFB1. Br. Poult. Sci. 2007, 48, 713–723. [Google Scholar] [CrossRef] [PubMed]
- Hussain, Z.; Khan, M.Z.; Khan, A.; Javed, I.; Saleemi, M.K.; Mahmood, S.; Asi, M.R. Residues of aflatoxin B1 in broiler meat: Effect of age and dietary aflatoxin B1 levels. Food Chem. Toxicol. 2010, 48, 3304–3307. [Google Scholar] [CrossRef] [PubMed]
- Khlangwiset, P.; Shephard, G.S.; Wu, F. Aflatoxins and growth impairment: A review. Crit. Rev. Toxicol. 2011, 41, 740–755. [Google Scholar] [CrossRef] [PubMed]
- De Ruyck, K.; De Boevre, M.; Huybrechts, I.; De Saeger, S. Dietary mycotoxins, co-exposure, and carcinogenesis in humans: Short review. Mutat. Res. Rev. Mutat. Res. 2015, 766, 32–41. [Google Scholar] [CrossRef] [PubMed]
- Wu, F.; Groopman, J.D.; Pestka, J.J. Public health impacts of foodborne mycotoxins. Annu. Rev Food Sci. Technol. 2014, 5, 351–372. [Google Scholar] [CrossRef] [PubMed]
- Lesnierowski, G.; Stangierski, J. What’s new in chicken egg research and technology for human health promotion? A review. Trends Food Sci. Technol. 2018, 71, 46–51. [Google Scholar] [CrossRef]
- Drewnowski, A. The Nutrient Rich Foods Index helps to identify healthy, affordable foods. Am. J. Clin. Nutr. 2010, 91, 1095S–1101S. [Google Scholar] [CrossRef] [PubMed]
- Al-Khalifa, H.; Al-Naser, A. Ostrich meat: Production, quality parameters, and nutritional comparison to other types of meats. J. Appl. Poult. Res. 2014, 23, 784–790. [Google Scholar] [CrossRef]
- Paleari, M.A.; Camisasca, S.; Beretta, G.; Renon, P.; Corsico, P.; Bertolo, G.; Crivelli, G. Ostrich meat: Physico-chemical characteristics and comparison with turkey and bovine meat. Meat Sci. 1998, 8, 205–210. [Google Scholar] [CrossRef]
- Zdanowska-Sąsiadek, Ż.; Marchewka, J.; Horbańczuk, J.O.; Wierzbicka, A.; Lipińska, P.; Jóźwik, A.; Atanasov, A.G.; Huminiecki, Ł.; Sieroń, A.; Sieroń, K.; et al. Nutrients Composition in Fit Snacks Made from Ostrich, Beef and Chicken Dried Meat. Molecules 2018, 23, 1267. [Google Scholar] [CrossRef] [PubMed]
- Barbut, S. Convenience breaded poultry meat products–New developments. Trends Food Sci. Technol. 2012, 26, 14–20. [Google Scholar] [CrossRef]
- Valverde, D.; Laca, A.; Estrada, L.N.; Paredes, B.; Rendueles, M.; Díaz, M. Egg yolk fractions as basic ingredient in the development of new snack products. Int. J. Gastron. Food Sci. 2016, 3, 23–29. [Google Scholar] [CrossRef]
- Huang, X.; Ahn, D.U. How Can the Value and Use of Egg Yolk Be Increased? J. Food Sci. 2019. [Google Scholar] [CrossRef] [PubMed]
- Organization for Economic Co-operation and Development (OECD). Table 3A1.4. World Meat Projections. OECD-FAO Agricultural Outlook 2017–2026. Available online: https://doi.org/10.1787/agr_outlook-2017-table70-en (accessed on 6 February 2019).
- European Commission. Commission Regulation (EU) No 165/2010 of 26 February 2010 amending Regulation (EC) No 1881/2006 setting maximum levels for certain contaminants in foodstuffs as regards aflatoxins. Off. J. Eur. Union 2010, L50/8–L50/12. [Google Scholar]
- Kim, J.G.; Lee, Y.W.; Kim, P.G.; Roh, W.S.; Shintani, H. Reduction of aflatoxins by Korean soybean paste and its effect on cytotoxicity and reproductive toxicity-part 3. Inhibitory effects of Korean soybean paste (Doen-jang) on aflatoxin toxicity in laying hens and aflatoxin accumulation in their eggs. J. Food Prot. 2003, 66, 866–873. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.; Wang, J.; Deng, Q.; Gu, K.; Wang, J. Detoxification of aflatoxin B1 by lactic acid bacteria and hydrated sodium calcium aluminosilicate in broiler chickens. Livest. Sci. 2018, 208, 28–32. [Google Scholar] [CrossRef]
- Magnoli, A.P.; Monge, M.P.; Miazzo, R.D.; Cavaglieri, L.R.; Magnoli, C.E.; Merkis, C.I.; Cristofolini, A.L.; Dalcero, A.M.; Chiacchiera, S.M. Effect of low levels of aflatoxin B1 on performance, biochemical parameters, and aflatoxin B1 in broiler liver tissues in the presence of monensin and sodium bentonite. Poult. Sci. 2011, 90, 48–58. [Google Scholar] [CrossRef]
- Bintvihok, A.; Kositcharoenkul, S. Effect of dietary calcium propionate on performance, hepatic enzyme activities and aflatoxin residues in broilers fed a diet containing low levels of aflatoxin B1. Toxicon 2006, 47, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Salem, R.; El-Habashi, N.; Fadl, S.E.; Sakr, O.A.; Elbialy, Z.I. Effect of probiotic supplement on aflatoxicosis and gene expression in the liver of broiler chicken. Environ. Toxicol. Pharmacol. 2018, 60, 118–127. [Google Scholar] [CrossRef]
- Saminathan, M.; Selamat, J.; Abbasi Pirouz, A.; Abdullah, N.; Zulkifli, I. Effects of Nano-Composite Adsorbents on the Growth Performance, Serum Biochemistry, and Organ Weights of Broilers Fed with Aflatoxin-Contaminated Feed. Toxins 2018, 10, 345. [Google Scholar] [CrossRef] [PubMed]
- Rajput, S.A.; Sun, L.; Zhang, N.; Mohamed Khalil, M.; Gao, X.; Ling, Z.; Zhu, L.; Khan, F.A.; Zhang, J.; Qi, D. Ameliorative Effects of Grape Seed Proanthocyanidin Extract on Growth Performance, Immune Function, Antioxidant Capacity, Biochemical Constituents, Liver Histopathology and Aflatoxin Residues in Broilers Exposed to Aflatoxin B1. Toxins 2017, 9, 371. [Google Scholar] [CrossRef] [PubMed]
- Shannon, T.A.; Ledoux, D.R.; Rottinghaus, G.E.; Shaw, D.P.; Daković, A.; Marković, M. The efficacy of raw and concentrated bentonite clay in reducing the toxic effects of aflatoxin in broiler chicks. Poult. Sci. 2017, 96, 1651–1658. [Google Scholar] [CrossRef]
- Stanley, V.G.; Winsman, M.; Dunkley, C.; Ogunleye, T.; Daley, M.; Krueger, W.F.; Sefton, A.E.; Hinton Jr, A. The impact of yeast culture residue on the suppression of dietary aflatoxin on the performance of broiler breeder hens. J. Appl. Poult. Res. 2004, 13, 533–539. [Google Scholar] [CrossRef]
- Sakamoto, M.I.; Murakami, A.E.; Fernandes, A.M.; Ospina-Rojas, I.C.; Nunes, K.C.; Hirata, A.K. Performance and serum biochemical profile of Japanese quail supplemented with silymarin and contaminated with aflatoxin B1. Poult. Sci. 2018, 97, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Manafi, M. Toxicity of aflatoxin B1 on laying Japanese quails (Coturnix coturnix japonica). J. Appl. Anim. Res. 2018, 46, 953–959. [Google Scholar] [CrossRef]
- Chen, X.; Horn, N.; Cotter, P.F.; Applegate, T.J. Growth, serum biochemistry, complement activity, and liver gene expression responses of Pekin ducklings to graded levels of cultured aflatoxin B1. Poult. Sci. 2014, 93, 2028–2036. [Google Scholar] [CrossRef] [PubMed]
- Rauber, R.H.; Dilkin, P.; Giacomini, L.Z.; de Almeida, C.A.; Mallmann, C.A. Performance of turkey poults fed different doses of aflatoxins in the diet. Poult. Sci. 2007, 86, 1620–1624. [Google Scholar] [CrossRef] [PubMed]
- Diaz, G.J.; Cortes, A.; Botero, L. Evaluation of the ability of a feed additive to ameliorate the adverse effects of aflatoxins in turkey poults. Br. Poult. Sci. 2009, 50, 240–250. [Google Scholar] [CrossRef] [PubMed]
- Lozano, M.C.; Diaz, G.J. Microsomal and cytosolic biotransformation of aflatoxin B1 in four poultry species. Br. Poult. Sci. 2006, 47, 734–741. [Google Scholar] [CrossRef] [PubMed]
- Diaz, G.J.; Murcia, H.W.; Cepeda, S.M. Cytochrome P450 enzymes involved in the metabolism of aflatoxin B1 in chickens and quail. Poult. Sci. 2010, 89, 2461–2469. [Google Scholar] [CrossRef] [PubMed]
- Gregorio, M.C.D.; Bordin, K.; Souto, P.C.M.D.C.; Corassin, C.H.; Oliveira, C.A.F. Comparative biotransformation of aflatoxin B1 in swine, domestic fowls, and humans. Toxin Rev. 2015, 34, 142–150. [Google Scholar] [CrossRef]
- Williams, J.G.; Deschl, U.; Williams, G.M. DNA damage in fetal liver cells of turkey and chicken eggs dosed with aflatoxin B1. Arch. Toxicol. 2011, 85, 1167–1172. [Google Scholar] [CrossRef]
- Gülbahçe Mutlu, E.; Arslan, E.; Öznurlu, Y.; Özparlak, H. The effects of aflatoxin B1 on growth hormone regulated gene-1 and interaction between DNA and aflatoxin B1 in broiler chickens during hatching. Biotech. Histochem. 2018, 93, 463–470. [Google Scholar] [CrossRef] [PubMed]
- Jahanian, E.; Mahdavi, A.H.; Asgary, S.; Jahanian, R. Effect of dietary supplementation of mannanoligosaccharides on growth performance, ileal microbial counts, and jejunal morphology in broiler chicks exposed to aflatoxins. Livest. Sci. 2016, 190, 123–130. [Google Scholar] [CrossRef]
- Jahanian, E.; Mahdavi, A.H.; Asgary, S.; Jahanian, R. Effects of dietary inclusion of silymarin on performance, intestinal morphology and ileal bacterial count in aflatoxin-challenged broiler chicks. J. Anim. Physiol. Anim. Nutr. 2017, 101, e43–e54. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.; Wang, J.Q.; Jia, S.C.; Chen, Y.K.; Wang, J.P. Effect of yeast cell wall on the growth performance and gut health of broilers challenged with aflatoxin B1 and necrotic enteritis. Poult. Sci. 2018, 97, 477–484. [Google Scholar] [CrossRef] [PubMed]
- Khan, W.A.; Khan, M.Z.; Khan, A.; Hassan, Z.U.; Rafique, S.; Saleemi, M.K.; Ahad, A. Dietary vitamin E in White Leghorn layer breeder hens: A strategy to combat aflatoxin B1-induced damage. Avian Pathol. 2014, 43, 389–395. [Google Scholar] [CrossRef] [PubMed]
- Bagherzadeh Kasmani, F.; Karimi Torshizi, M.A.; Allameh, A.; Shariatmadari, F. A novel aflatoxin-binding Bacillus probiotic: Performance, serum biochemistry, and immunological parameters in Japanese quail. Poult. Sci. 2012, 91, 1846–1853. [Google Scholar] [CrossRef]
- Rasouli-Hiq, A.A.; Bagherzadeh-Kasmani, F.; Mehri, M.; Karimi-Torshizi, M.A. Nigella sativa (black cumin seed) as a biological detoxifier in diet contaminated with aflatoxin B1. J. Anim. Physiol. Anim. Nutr. 2017, 26, 229–238. [Google Scholar] [CrossRef]
- Mahmood, S.; Younus, M.; Aslam, A.; Anjum, A.A. Toxicological effects of aflatoxin B1 on growth performance, humoral immune response and blood profile of Japanese quail. J. Anim. Plant Sci. 2017, 27, 833–840. [Google Scholar]
- Han, X.Y.; Huang, Q.C.; Li, W.F.; Jiang, J.F.; Xu, Z.R. Changes in growth performance, digestive enzyme activities and nutrient digestibility of cherry valley ducks in response to aflatoxin B1 levels. Livest. Sci. 2008, 119, 216–220. [Google Scholar] [CrossRef]
- Hassan, Z.U.; Khan, M.Z.; Khan, A.; Javed, I.; Hussain, Z. Effects of individual and combined administration of ochratoxin A and aflatoxin B1 in tissues and eggs of White Leghorn breeder hens. J. Sci. Food Agric. 2012, 92, 1540–1544. [Google Scholar] [CrossRef]
- Denli, M.; Blandon, J.C.; Guynot, M.E.; Salado, S.; Perez, J.F. Effects of dietary AflaDetox on performance, serum biochemistry, histopathological changes, and aflatoxin residues in broilers exposed to aflatoxin B1. Poult. Sci. 2009, 88, 1444–1451. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.H.; Li, W.; Wang, X.H.; Han, M.Y.; Muhammad, I.; Zhang, X.Y.; Sun, X.Q.; Cui, X.X. Water-soluble substances of wheat: A potential preventer of aflatoxin B1-induced liver damage in broilers. Poult. Sci. 2019, 98, 136–149. [Google Scholar] [CrossRef] [PubMed]
- Goetz, M.E.; Luch, A. Reactive species: A cell damaging rout assisting to chemical carcinogens. Cancer Lett. 2008, 266, 73–83. [Google Scholar] [CrossRef] [PubMed]
- Klaunig, J.E.; Wang, Z.; Pu, X.; Zhou, S. Oxidative stress and oxidative damage in chemical carcinogenesis. Toxicol. Appl. Pharmacol. 2011, 254, 86–99. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Ma, Q.; Ma, S.; Zhang, J.; Jia, R.; Ji, C.; Zhao, L. Ameliorating effects of Bacillus subtilis ANSB060 on growth performance, antioxidant functions, and aflatoxin residues in ducks fed diets contaminated with aflatoxins. Toxins 2017, 9, 1. [Google Scholar] [CrossRef] [PubMed]
- Migliorini, M.J.; Da Silva, A.S.; Santurio, J.M.; Bottari, N.B.; Gebert, R.R.; Reis, J.H.; Volpato, A.; Morsch, V.M.; Baldissera, M.D.; Stefani, L.M.; et al. The Protective Effects of an Adsorbent against Oxidative Stress in Quails Fed Aflatoxin-Contaminated Diet. Acta Sci. Vet. 2017, 45, 1–7. [Google Scholar] [CrossRef]
- Julian, R.J. Production and growth related disorders and other metabolic diseases of poultry—A review. Vet. J. 2005, 169, 350–369. [Google Scholar] [CrossRef] [PubMed]
- Silversides, F.G.; Singh, R.; Cheng, K.M.; Korver, D.R. Comparison of bones of 4 strains of laying hens kept in conventional cages and floor pens. Poult. Sci. 2012, 91, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Rath, N.C.; Huff, G.R.; Huff, W.E.; Balog, J.M. Factors regulating bone maturity and strength in poultry. Poult. Sci. 2000, 79, 1024–1032. [Google Scholar] [CrossRef] [PubMed]
- Mueller, W.J.; Schraer, R.; Scharer, H. Calcium metabolism and skeletal dynamics of laying pullets. J. Nutr. 1964, 84, 20–26. [Google Scholar] [CrossRef] [PubMed]
- Whitehead, C.C. Overview of bone biology in the egg-laying hen. Poult. Sci. 2004, 83, 193–199. [Google Scholar] [CrossRef] [PubMed]
- Huff, W.E.; Doerr, J.A.; Hamilton, P.B.; Hamann, D.D.; Peterson, R.E.; Ciegler, A. Evaluation of bone strength during aflatoxicosis and ochratoxicosis. Appl. Environ. Microbiol. 1980, 40, 102–107. [Google Scholar]
- Raju, M.V.L.N.; Rama Rao, S.V.; Radhika, K.; Panda, A.K. Effect of amount and source of supplemental dietary vegetable oil on broiler chickens exposed to aflatoxicosis. Br. Poult. Sci. 2005, 46, 587–594. [Google Scholar] [CrossRef]
- Sur, E.; Celik, I. Effects of aflatoxin B1 on the development of the bursa of Fabricius and blood lymphocyte acid phosphatase of the chicken. Br. Poult. Sci. 2003, 44, 558–566. [Google Scholar] [CrossRef]
- Oznurlu, Y.; Celik, I.; Sur, E.; Ozaydın, T.; Oğuz, H.; Altunbaş, K. Determination of the effects of aflatoxin B1 given in ovo on the proximal tibial growth plate of broiler chickens: Histological, histometric and immunohistochemical findings. Avian Pathol. 2012, 41, 469–477. [Google Scholar] [CrossRef]
- Yin, H.B.; Chen, C.H.; Darre, M.J.; Donoghue, A.M.; Donoghue, D.J.; Venkitanarayanan, K. Phytochemicals reduce aflatoxin-induced toxicity in chicken embryos. Poult. Sci. 2017, 96, 3725–3732. [Google Scholar] [CrossRef]
- Gündüz, N.; Oznurlu, Y. Adverse effects of aflatoxin B1 on skeletal muscle development in broiler chickens. Br. Poult. Sci. 2014, 55, 684–692. [Google Scholar] [CrossRef] [PubMed]
- Bai, S.; Wang, L.; Luo, Y.; Ding, X.; Yang, J.; Bai, J.; Zhang, K.; Wang, J. Effects of Corn Naturally Contaminated with Aflatoxins on Performance, Calcium and Phosphorus Metabolism, and Bone Mineralization of Broiler Chicks. J. Poult. Sci. 2013, 51, 157–164. [Google Scholar] [CrossRef]
- Dos Anjos, F.R.; Ledoux, D.R.; Rottinghaus, G.E.; Chimonyo, M. Efficacy of adsorbents (bentonite and diatomaceous earth) and turmeric (Curcuma longa) in alleviating the toxic effects of aflatoxin in chicks. Br. Poult. Sci. 2015, 56, 459–469. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Espinosa, D.; Cervantes-Aguilar, F.J.; Del Río-García, J.C.; Villarreal-Barajas, T.; Vázquez-Durán, A.; Méndez-Albores, A. Ameliorative Effects of Neutral Electrolyzed Water on Growth Performance, Biochemical Constituents, and Histopathological Changes in Turkey Poults during Aflatoxicosis. Toxins 2017, 9, 104. [Google Scholar] [CrossRef] [PubMed]
- Mendieta, C.R.; Gómez, G.V.; Del Río, J.C.G.; Cuevas, A.C.; Arce, J.M.; Ávila, E.G. Effect of the addition of saccharomyces cerevisiae yeast cell walls to diets with mycotoxins on the performance and immune responses of broilers. J. Poult. Sci. 2018, 55, 38–46. [Google Scholar] [CrossRef]
- Yuan, S.; Wu, B.; Yu, Z.; Fang, J.; Liang, N.; Zhou, M.; Huang, C.; Peng, X. The mitochondrial and endoplasmic reticulum pathways involved in the apoptosis of bursa of Fabricius cells in broilers exposed to dietary aflatoxin B1. Oncotarget 2016, 7, 65295–65306. [Google Scholar] [CrossRef]
- Zhu, P.; Zuo, Z.; Zheng, Z.; Wang, F.; Peng, X.; Fang, J.; Cui, H.; Gao, C.; Song, H.; Zhou, Y.; et al. Aflatoxin B1 affects apoptosis and expression of death receptor and endoplasmic reticulum molecules in chicken spleen. Oncotarget 2017, 8, 99531–99540. [Google Scholar]
- Bhatti, S.A.; Khan, M.Z.; Saleemi, M.K.; Saqib, M.; Khan, A.; Ul-Hassan, Z. Protective role of bentonite against aflatoxin B1-and ochratoxin A-induced immunotoxicity in broilers. J. Immunotoxicol. 2017, 14, 66–76. [Google Scholar] [CrossRef] [PubMed]
- Peng, X.; Zhang, K.; Bai, S.; Ding, X.; Zeng, Q.; Yang, J.; Fang, J.; Chen, K. Histological lesions, cell cycle arrest, apoptosis and T cell subsets changes of spleen in chicken fed aflatoxin-contaminated corn. Int. J. Environ. Res. Public Health 2014, 11, 8567–8580. [Google Scholar] [CrossRef] [PubMed]
- Karaman, M.; Özen, H.; Tuzcu, M.; Ciğremiş, Y.; Önder, F.; Özcan, K. Pathological, biochemical and haematological investigations on the protective effect of α-lipoic acid in experimental aflatoxin toxicosis in chicks. Br. Poult. Sci. 2010, 51, 132–141. [Google Scholar] [CrossRef] [PubMed]
- Peng, X.; Bai, S.; Ding, X.; Zhang, K. Pathological impairment, cell cycle arrest and apoptosis of thymus and bursa of fabricius induced by aflatoxin-contaminated corn in Broilers. Int. J. Environ. Res. Public Health 2017, 14, 77. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Shu, G.; Peng, X.; Fang, J.; Cui, H.; Chen, J.; Wang, F.; Chen, Z.; Zuo, Z.; Deng, J.; et al. Protective role of sodium selenite on histopathological lesions, decreased T-cell subsets and increased apoptosis of thymus in broilers intoxicated with aflatoxin B1. Food Chem. Toxicol. 2013, 59, 446–454. [Google Scholar] [CrossRef] [PubMed]
- Ul-Hassan, Z.; Zargham Khan, M.; Khan, A.; Javed, I. Immunological status of the progeny of breeder hens kept on ochratoxin A (OTA)-and aflatoxin B1 (AFB1)-contaminated feeds. J. Immunotoxicol. 2012, 9, 381–391. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Zhang, K.Y.; Chen, D.W.; Ding, X.M.; Feng, G.D.; Ao, X. Effects of vitamin E and selenium yeast on growth performance and immune function in ducks fed maize naturally contaminated with aflatoxin B1. Livest. Sci. 2013, 152, 200–207. [Google Scholar] [CrossRef]
- Tessari, E.N.C.; Oliveira, C.A.F.; Cardoso, A.L.S.P.; Ledoux, D.R.; Rottinghaus, G.E. Effects of aflatoxin B1 and fumonisin B1 on body weight, antibody titres and histology of broiler chicks. Br. Poult. Sci. 2006, 47, 357–364. [Google Scholar] [CrossRef]
- Bailey, C.A.; Latimer, G.W.; Barr, A.C.; Wigle, W.L.; Haq, A.U.; Balthrop, J.E.; Kubena, L.F. Efficacy of montmorillonite clay (NovaSil PLUS) for protecting full-term broilers from aflatoxicosis. J. Appl. Poult. Res. 2006, 15, 198–206. [Google Scholar] [CrossRef]
- Matur, E.; Ergul, E.; Akyazi, I.; Eraslan, E.; Cirakli, Z.T. The effects of Saccharomyces cerevisiae extract on the weight of some organs, liver, and pancreatic digestive enzyme activity in breeder hens fed diets contaminated with aflatoxins. Poult. Sci. 2010, 89, 2213–2220. [Google Scholar] [CrossRef] [PubMed]
- Şimşek, N.; Ergun, L.; Ergun, E.; Alabay, B.; Essiz, D. The effects of experimental aflatoxicosis on the exocrine pancreas in quails (Coturnix coturnix japonica). Arch. Toxicol. 2007, 81, 583–588. [Google Scholar] [PubMed]
- Feng, G.D.; He, J.; Ao, X.; Chen, D.W. Effects of maize naturally contaminated with aflatoxin B1 on growth performance, intestinal morphology, and digestive physiology in ducks. Poult. Sci. 2017, 96, 1948–1955. [Google Scholar]
- Marchioro, A.; Mallmann, A.O.; Diel, A.; Dilkin, P.; Rauber, R.H.; Blazquez, F.J.H.; Oliveira, M.G.A.; Mallmann, C.A. Effects of aflatoxins on performance and exocrine pancreas of broiler chickens. Avian Dis. 2013, 57, 280–284. [Google Scholar]
- Chen, X.; Naehrer, K.; Applegate, T.J. Interactive effects of dietary protein concentration and aflatoxin B1 on performance, nutrient digestibility, and gut health in broiler chicks. Poult. Sci. 2016, 95, 1312–1325. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Murdoch, R.; Zhang, Q.; Shafer, D.J.; Applegate, T.J. Effects of dietary protein concentration on performance and nutrient digestibility in Pekin ducks during aflatoxicosis. Poult. Sci. 2016, 95, 834–841. [Google Scholar] [CrossRef] [PubMed]
- Applegate, T.J.; Schatzmayr, G.; Pricket, K.; Troche, C.; Jiang, Z. Effect of aflatoxin culture on intestinal function and nutrient loss in laying hens. Poult. Sci. 2009, 88, 1235–1241. [Google Scholar] [CrossRef]
- Yunus, A.W.; Razzazi-Fazeli, E.; Bohm, J. Aflatoxin B1 in affecting broiler’s performance, immunity, and gastrointestinal tract: A review of history and contemporary issues. Toxins 2011, 3, 566–590. [Google Scholar] [CrossRef] [PubMed]
- Yamauchi, K.E. Review on chicken intestinal villus histological alterations related with intestinal function. J. Poult. Sci. 2002, 39, 229–242. [Google Scholar] [CrossRef]
- Peng, X.; Zhang, S.; Fang, J.; Cui, H.; Zuo, Z.; Deng, J. Protective roles of sodium selenite against aflatoxin B1-induced apoptosis of jejunum in broilers. Int. J. Environ. Res. Public Health 2014, 11, 13130–13143. [Google Scholar] [CrossRef]
- Zheng, Z.; Zuo, Z.; Zhu, P.; Wang, F.; Yin, H.; Peng, X.; Fang, J.; Cui, H.; Gao, C.; Song, H.; et al. A study on the expression of apoptotic molecules related to death receptor and endoplasmic reticulum pathways in the jejunum of AFB1-intoxicated chickens. Oncotarget 2017, 8, 89655–89664. [Google Scholar] [CrossRef]
- Yin, H.; Jiang, M.; Peng, X.; Cui, H.; Zhou, Y.; He, M.; Zuo, Z.; Ouyang, P.; Fan, J.; Fang, J. The molecular mechanism of G2/M cell cycle arrest induced by AFB1 in the jejunum. Oncotarget 2016, 7, 35592–35606. [Google Scholar] [CrossRef]
- Fang, J.; Zheng, Z.; Yang, Z.; Peng, X.; Zuo, Z.; Cui, H.; Ouyang, P.; Shu, G.; Chen, Z.; Huang, C. Ameliorative effects of selenium on the excess apoptosis of the jejunum caused by AFB1 through death receptor and endoplasmic reticulum pathways. Toxicol. Res. 2018, 7, 1108–1119. [Google Scholar] [CrossRef] [PubMed]
- Fang, J.; Yin, H.; Zheng, Z.; Zhu, P.; Peng, X.; Zuo, Z.; Cui, H.; Zhou, Y.; Ouyang, P.; Geng, Y.; et al. The molecular mechanisms of protective role of Se on the G2/M phase arrest of jejunum caused by AFB1. Biol. Trace Elem. Res. 2018, 181, 142–153. [Google Scholar] [CrossRef]
- Liu, N.; Wang, J.Q.; Liu, Z.Y.; Wang, Y.C.; Wang, J.P. Comparison of probiotics and clay detoxifier on the growth performance and enterotoxic markers of broilers fed diets contaminated with aflatoxin B1. J. Appl. Poult. Res. 2018, 27, 341–348. [Google Scholar] [CrossRef]
- Wang, F.; Zuo, Z.; Chen, K.; Gao, C.; Yang, Z.; Zhao, S.; Li, J.; Song, H.; Peng, X.; Fang, J.; et al. Histopathological Injuries, Ultrastructural Changes, and Depressed TLR Expression in the Small Intestine of Broiler Chickens with Aflatoxin B1. Toxins 2018, 10, 131. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Fang, J.; Peng, X.; Cui, H.; Zuo, Z.; Deng, J.; Chen, Z.; Geng, Y.; Lai, W.; Shu, G.; et al. Effects of sodium selenite on aflatoxin B1-induced decrease of ileal IgA+ cell numbers and immunoglobulin contents in broilers. Biol. Trace Elem. Res. 2014, 160, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Jiang, M.; Fang, J.; Peng, X.; Cui, H.; Yu, Z. Effect of aflatoxin B1 on IgA+ cell number and immunoglobulin mRNA expression in the intestine of broilers. Immunopharmacol. Immunotoxicol. 2015, 37, 450–457. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Zuo, Z.; Zhu, P.; Zheng, Z.; Peng, X.; Fang, J.; Cui, H.; Zhou, Y.; Ouyang, P.; Geng, Y.; et al. Sodium selenite prevents suppression of mucosal humoral response by AFB1 in broiler’s cecal tonsil. Oncotarget 2017, 8, 54215–54226. [Google Scholar] [PubMed]
- Wang, F.; Zuo, Z.; Chen, K.; Peng, X.; Fang, J.; Cui, H.; Shu, G.; He, M.; Tang, L. Selenium Rescues Aflatoxin B1-Inhibited T Cell Subsets and Cytokine Levels in Cecal Tonsil of Chickens. Biol. Trace Elem. Res. 2019, 188, 461–467. [Google Scholar] [CrossRef] [PubMed]
- Fouad, A.M.; El-Senousey, H.K. Nutritional factors affecting abdominal fat deposition in poultry: A review. Asian-Aust. J. Anim. Sci. 2014, 27, 1057–1068. [Google Scholar] [CrossRef] [PubMed]
- Hiramoto, K.; Muramatsu, T.; Okumura, J. Effect of methionine and lysine deficiencies on protein synthesis in the liver and oviduct and in the whole body of laying hens. Poult. Sci. 1990, 69, 84–89. [Google Scholar] [CrossRef]
- Shanmugasundaram, R.; Selvaraj, R.K. Vitamin D-1α-hydroxylase and vitamin D-24-hydroxylase mRNA studies in chickens. Poult. Sci. 2012, 91, 1819–1824. [Google Scholar] [CrossRef]
- Siloto, E.V.; Oliveira, E.F.A.; Sartori, J.R.; Fascina, V.B.; Martins, B.A.B.; Ledoux, D.R.; Rottinghaus, G.E.; Sartori, D.R.S. Lipid metabolism of commercial layers fed diets containing aflatoxin, fumonisin, and a binder. Poult. Sci. 2013, 92, 2077–2083. [Google Scholar] [CrossRef]
- Tejada-Castaneda, Z.I.; Avila-Gonzalez, E.; Casaubon-Huguenin, M.T.; Cervantes-Olivares, R.A.; Vásquez-Peláez, C.; Hernandez-Baumgarten, E.M.; Moreno-Martínez, E. Biodetoxification of aflatoxin-contaminated chick feed. Poult. Sci. 2008, 87, 1569–1576. [Google Scholar] [CrossRef] [PubMed]
- Yarru, L.P.; Settivari, R.S.; Gowda, N.K.S.; Antoniou, E.; Ledoux, D.R.; Rottinghaus, G.E. Effects of turmeric (Curcuma longa) on the expression of hepatic genes associated with biotransformation, antioxidant, and immune systems in broiler chicks fed aflatoxin. Poult. Sci. 2009, 88, 2620–2627. [Google Scholar] [CrossRef]
- Ma, Q.G.; Gao, X.; Zhou, T.; Zhao, L.H.; Fan, Y.; Li, X.Y.; Lei, Y.P.; Ji, C.; Zhang, J.Y. Protective effect of Bacillus subtilis ANSB060 on egg quality, biochemical and histopathological changes in layers exposed to aflatoxin B1. Poult. Sci. 2012, 91, 2852–2857. [Google Scholar] [CrossRef] [PubMed]
- Liao, S.; Shi, D.; Clemons-Chevis, C.L.; Guo, S.; Su, R.; Qiang, P.; Tang, Z. Protective role of selenium on aflatoxin B1-induced hepatic dysfunction and apoptosis of liver in ducklings. Biol. Trace Elem. Res. 2014, 162, 296–301. [Google Scholar] [CrossRef]
- Ma, Q.; Li, Y.; Fan, Y.; Zhao, L.; Wei, H.; Ji, C.; Zhang, J. Molecular mechanisms of lipoic acid protection against aflatoxin B1-induced liver oxidative damage and inflammatory responses in broilers. Toxins 2015, 7, 5435–5447. [Google Scholar] [CrossRef]
- Muhammad, I.; Wang, X.; Li, S.; Li, R.; Zhang, X. Curcumin confers hepatoprotection against AFB1-induced toxicity via activating autophagy and ameliorating inflammation involving Nrf2/HO-1 signaling pathway. Mol. Biol. Rep. 2018, 45, 1775–1785. [Google Scholar] [CrossRef] [PubMed]
- Muhammad, I.; Wang, H.; Sun, X.; Wang, X.; Han, M.; Lu, Z.; Cheng, P.; Hussain, M.A.; Zhang, X. Dual role of dietary curcumin through attenuating AFB1-induced oxidative stress and liver injury via modulating liver phase-I and phase-II enzymes involved in AFB1 bioactivation and detoxification. Front. Pharmacol. 2018, 9, 554. [Google Scholar] [CrossRef] [PubMed]
- Walzem, R.L.; Simon, C.; Morishita, T.; Lowenstine, L.; Hansen, R.J. Fatty liver hemorrhagic syndrome in hens overfed a purified diet. Selected enzyme activities and liver histology in relation to liver hemorrhage and reproductive performance. Poult. Sci. 1993, 72, 1479–1491. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.; Hester, P.Y.; Hu, J.Y.; Yan, F.F.; Dennis, R.L.; Cheng, H.W. Effect of perches on liver health of hens. Poult. Sci. 2014, 93, 1618–1622. [Google Scholar] [CrossRef] [PubMed]
- Şehu, A.; Cakir, S.; Cengiz, Ö.; Eşsiz, D. MYCOTOX® and aflatoxicosis in quails. Br. Poult. Sci. 2005, 46, 520–524. [Google Scholar] [CrossRef] [PubMed]
- Gholami-Ahangaran, M.; Rangsaz, N.; Azizi, S. Evaluation of turmeric (Curcuma longa) effect on biochemical and pathological parameters of liver and kidney in chicken aflatoxicosis. Pharm. Biol. 2016, 54, 780–787. [Google Scholar] [CrossRef] [PubMed]
- Sun, D.; Li, C.; Gao, J.; Li, S.; Wang, H. Effects of selenium deficiency on principal indexes of chicken kidney function. Biol. Trace Elem. Res. 2015, 164, 58–63. [Google Scholar] [CrossRef] [PubMed]
- Neeff, D.V.; Ledoux, D.R.; Rottinghaus, G.E.; Bermudez, A.J.; Dakovic, A.; Murarolli, R.A.; Oliveira, C.A.F. In vitro and in vivo efficacy of a hydrated sodium calcium aluminosilicate to bind and reduce aflatoxin residues in tissues of broiler chicks fed aflatoxin B1. Poult. Sci. 2013, 92, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Özen, H.; Karaman, M.; Çiğremiş, Y.; Tuzcu, M.; Özcan, K.; Erdağ, D. Effectiveness of melatonin on aflatoxicosis in chicks. Res. Vet. Sci. 2009, 86, 485–489. [Google Scholar] [CrossRef] [PubMed]
- Uyar, A.; Yener, Z.; Dogan, A. Protective effects of Urtica dioica seed extract in aflatoxicosis: Histopathological and biochemical findings. Br. Poult. Sci. 2016, 57, 235–245. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Wang, F.; Liang, N.; Wang, C.; Peng, X.; Fang, J.; Cui, H.; Mughal, M.J.; Lai, W. Effect of selenium supplementation on apoptosis and cell cycle blockage of renal cells in broilers fed a diet containing aflatoxin B1. Biol. Trace Elem. Res. 2015, 168, 242–251. [Google Scholar] [CrossRef] [PubMed]
- Liang, N.; Wang, F.; Peng, X.; Fang, J.; Cui, H.; Chen, Z.; Lai, W.; Zhou, Y.; Geng, Y. Effect of sodium selenite on pathological changes and renal functions in broilers fed a diet containing aflatoxin B1. Int. J. Environ. Res. Public Health 2015, 12, 11196–11208. [Google Scholar] [CrossRef]
- Karaman, M.; Basmacioglu, H.; Ortatatli, M.; Oguz, H. Evaluation of the detoxifying effect of yeast glucomannan on aflatoxicosis in broilers as assessed by gross examination and histopathology. Br. Poult. Sci. 2005, 46, 394–400. [Google Scholar] [CrossRef] [PubMed]
- Huang, Q.; Gao, X.; Liu, P.; Lin, H.; Liu, W.; Liu, G.; Zhang, J.; Deng, G.; Zhang, C.; Cao, H.; et al. The relationship between liver-kidney impairment and viral load after nephropathogenic infectious bronchitis virus infection in embryonic chickens. Poult. Sci. 2017, 96, 1589–1597. [Google Scholar] [CrossRef] [PubMed]
- Chen, N.N.; Liu, B.; Xiong, P.W.; Guo, Y.; He, J.N.; Hou, C.C.; Ma, L.X.; Yu, D.Y. Safety evaluation of zinc methionine in laying hens: Effects on laying performance, clinical blood parameters, organ development, and histopathology. Poult. Sci. 2018, 97, 1120–1126. [Google Scholar] [CrossRef] [PubMed]
- Ortatatli, M.; Ciftci, M.K.; Tuzcu, M.; Kaya, A. The effects of aflatoxin on the reproductive system of roosters. Res. Vet. Sci. 2002, 72, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Bagherzadeh Kasmani, F.; Torshizi, K.; Mehri, M. Effect of Brevibacillus laterosporus Probiotic on Hematology, Internal Organs, Meat Peroxidation and Ileal Microflora in Japanese Quails Fed Aflatoxin B1. J. Agric. Sci. Technol. 2018, 20, 459–468. [Google Scholar]
- Eraslan, G.; Akdoğan, M.; LİMAN, B.C.; Kanbur, M.; DELİBAŞ, N. Effects of dietary aflatoxin and hydrate sodium calcium aluminosilicate on triiodothyronine, thyroxine, thyrotrophin and testosterone levels in quails. Turk. J. Vet. Anim. Sci. 2006, 30, 41–45. [Google Scholar]
- Doerr, J.A.; Ottinger, M.A. Delayed reproductive development resulting from aflatoxicosis in juvenile Japanese quail. Poult. Sci. 1980, 59, 1995–2001. [Google Scholar] [CrossRef] [PubMed]
- Wolzak, A.; Pearson, A.M.; Coleman, T.H.; Pestka, J.J.; Gray, J.I.; Chen, C. Aflatoxin carryover and clearance from tissues of laying hens. Food Chem. Toxicol. 1986, 24, 37–41. [Google Scholar] [CrossRef]
- European Commission. Commission Directive (EU) No 2003/100/EC of 31 October 2003 amending Annex I to Directive 2002/32/EC of the European Parliament and of the Council on undesirable substances in animal feed. Off. J. Eur. Union 2003, L285/33–L285/37. [Google Scholar]
- Oguz, H.; Kurtoglu, V. Effect of clinoptilolite on performance of broiler chickens during experimental aflatoxicosis. Br. Poult. Sci. 2000, 41, 512–517. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Horn, N.; Applegate, T.J. Efficiency of hydrated sodium calcium aluminosilicate to ameliorate the adverse effects of graded levels of aflatoxin B1 in broiler chicks. Poult. Sci. 2014, 93, 2037–2047. [Google Scholar] [CrossRef]
- Bhatti, S.A.; Khan, M.Z.; Hassan, Z.U.; Saleemi, M.K.; Saqib, M.; Khatoon, A.; Akhter, M. Comparative efficacy of Bentonite clay, activated charcoal and Trichosporon mycotoxinivorans in regulating the feed-to-tissue transfer of mycotoxins. J. Sci. Food Agric. 2018, 98, 884–890. [Google Scholar] [CrossRef]
- Lala, A.O.; Oso, A.O.; Ajao, A.M.; Idowu, O.M.; Oni, O.O. Effect of supplementation with molecular or nano-clay adsorbent on growth performance and haematological indices of starter and grower turkeys fed diets contaminated with varying dosages of aflatoxin B1. Livest. Sci. 2015, 178, 209–215. [Google Scholar] [CrossRef]
- Lala, A.O.; Ajayi, O.L.; Oso, A.O.; Ajao, M.O.; Oni, O.O.; Okwelum, N.; Idowu, O.M.O. Effect of dietary supplementation with clay-based binders on biochemical and histopathological changes in organs of turkey fed with aflatoxin-contaminated diets. J. Anim. Physiol. Anim. Nutr. 2016, 100, 1191–1202. [Google Scholar] [CrossRef] [PubMed]
- Shetty, P.H.; Jespersen, L. Saccharomyces cerevisiae and lactic acid bacteria as potential mycotoxin decontaminating agents. Trends Food. Sci Technol. 2006, 17, 48–55. [Google Scholar] [CrossRef]
- Magnoli, A.P.; Rodriguez, M.C.; Pereyra, M.G.; Poloni, V.L.; Peralta, M.F.; Nilson, A.J.; Miazzo, R.D.; Bagnis, G.; Chiacchiera, S.M.; Cavaglieri, L.R. Use of yeast (Pichia kudriavzevii) as a novel feed additive to ameliorate the effects of aflatoxin B1 on broiler chicken performance. Mycotoxin Res. 2017, 33, 273–283. [Google Scholar] [CrossRef] [PubMed]
- Pizzolitto, R.P.; Armando, M.R.; Salvano, M.A.; Dalcero, A.M.; Rosa, C.A. Evaluation of Saccharomyces cerevisiae as an antiaflatoxicogenic agent in broiler feedstuffs. Poult. Sci. 2013, 92, 1655–1663. [Google Scholar] [CrossRef]
- Bovo, F.; Franco, L.T.; Kobashigawa, E.; Rottinghaus, G.E.; Ledoux, D.R.; Oliveira, C.A.F. Efficacy of beer fermentation residue containing Saccharomyces cerevisiae cells for ameliorating aflatoxicosis in broilers. Poult. Sci. 2015, 94, 934–942. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Zhao, L.; Ma, Q.; Li, X.; Shi, H.; Zhou, T.; Zhang, J.; Ji, C. Effects of Bacillus subtilis ANSB060 on growth performance, meat quality and aflatoxin residues in broilers fed moldy peanut meal naturally contaminated with aflatoxins. Food Chem. Toxicol. 2013, 59, 748–753. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Zhao, L.; Ji, C.; Li, X.; Jia, R.; Xi, L.; Zhang, J.; Ma, Q. Protective effects of bacillus subtilis ansb060 on serum biochemistry, histopathological changes and antioxidant enzyme activities of broilers fed moldy peanut meal naturally contaminated with aflatoxins. Toxins 2015, 7, 3330–3343. [Google Scholar] [CrossRef] [PubMed]
- Jia, R.; Ma, Q.; Fan, Y.; Ji, C.; Zhang, J.; Liu, T.; Zhao, L. The toxic effects of combined aflatoxins and zearalenone in naturally contaminated diets on laying performance, egg quality and mycotoxins residues in eggs of layers and the protective effect of Bacillus subtilis biodegradation product. Food Chem. Toxicol. 2016, 90, 142–150. [Google Scholar] [CrossRef] [PubMed]
- Khanian, M.; Karimi-Torshizi, M.A.; Allameh, A. Alleviation of aflatoxin-related oxidative damage to liver and improvement of growth performance in broiler chickens consumed Lactobacillus plantarum 299v for entire growth period. Toxicon 2019, 158, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Fang, J.; Peng, X.; Cui, H.; Chen, J.; Wang, F.; Chen, Z.; Zuo, Z.; Deng, J.; Lai, W.; et al. Effect of selenium supplementation on aflatoxin B1-induced histopathological lesions and apoptosis in bursa of Fabricius in broilers. Food Chem. Toxicol. 2014, 74, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Peng, X.; Fang, J.; Cui, H.; Zuo, Z.; Deng, J.; Chen, Z.; Geng, Y.; Lai, W.; Tang, L.; et al. Effects of dietary selenium on histopathological changes and T cells of spleen in broilers exposed to aflatoxin B1. Int. J. Environ. Res. Public Health 2014, 11, 1904–1913. [Google Scholar] [CrossRef] [PubMed]
- Hu, P.; Zuo, Z.; Wang, F.; Peng, X.; Guan, K.; Li, H.; Fang, J.; Cui, H.; Su, G.; Ouyang, P.; et al. The Protective Role of Selenium in AFB1-Induced Tissue Damage and Cell Cycle Arrest in Chicken’s Bursa of Fabricius. Biol. Trace Elem. Res. 2018, 185, 486–496. [Google Scholar] [CrossRef] [PubMed]
- Fang, J.; Zhu, P.; Yang, Z.; Peng, X.; Zuo, Z.; Cui, H.; Ouyang, P.; Shu, G.; Chen, Z.; Huang, C.; et al. Selenium Ameliorates AFB1-Induced Excess Apoptosis in Chicken Splenocytes Through Death Receptor and Endoplasmic Reticulum Pathways. Biol. Trace Elem. Res. 2018, 187, 273–283. [Google Scholar] [CrossRef]
- Li, Y.; Ma, Q.G.; Zhao, L.H.; Wei, H.; Duan, G.X.; Zhang, J.Y.; Ji, C. Effects of lipoic acid on immune function, the antioxidant defense system, and inflammation-related genes expression of broiler chickens fed aflatoxin contaminated diets. Int. J. Mol. Sci. 2014, 15, 5649–5662. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.Y.; Qi, M.; Zhao, L.; Zhu, M.K.; Guo, J.; Liu, J.; Gu, C.Q.; Rajput, S.A.; Krumm, C.S.; Qi, D.S.; et al. Curcumin prevents aflatoxin B1 hepatoxicity by inhibition of cytochrome P450 isozymes in chick liver. Toxins 2016, 8, 327. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.H.; Zhang, N.Y.; Zhu, M.K.; Zhao, L.; Zhou, J.C.; Qi, D.S. Prevention of Aflatoxin B1 Hepatoxicity by Dietary Selenium Is Associated with Inhibition of Cytochrome P450 Isozymes and Up-Regulation of 6 Selenoprotein Genes in Chick Liver3. J. Nutr. 2016, 146, 655–661. [Google Scholar] [CrossRef] [PubMed]
- Gowda, N.K.S.; Ledoux, D.R.; Rottinghaus, G.E.; Bermudez, A.J.; Chen, Y.C. Efficacy of turmeric (Curcuma longa), containing a known level of curcumin, and a hydrated sodium calcium aluminosilicate to ameliorate the adverse effects of aflatoxin in broiler chicks. Poult. Sci. 2008, 87, 1125–1130. [Google Scholar] [CrossRef] [PubMed]
| Bird | Aflatoxin Dose * | Fungal Strain | Reference |
|---|---|---|---|
| Chickens | 2000 | A. parasiticus (NRLL 2999) | [31] |
| Chickens | 22 | A. parasiticus (FRR 2999) | [29] |
| Chickens | 2 | A. parasiticus (PTCC 5286) | [43,44] |
| Chickens | 1000 | A. flavus (NRRL 3357) | [30] |
| Chickens | 250 | A. flavus (KP137700) | [28] |
| Chickens | 40 | A. flavus (Chinese isolate) | [45] |
| Laying hens | 2500 | A. parasiticus (SRRC 148) | [9] |
| Laying hens | 500 | A. parasiticus (ATCC 15517) | [24] |
| Breeder hens | 3000 | A. parasiticus (NRRL 2999) | [32] |
| Breeder hens | 500 | A. flavus (NRRL 6540; CECT 2687) | [46] |
| Quail | 2500 | A. parasiticus (PTCC 5286) | [47,48] |
| Quail | 1500 | A. parasiticus (NRRL 2999) | [33,34] |
| Quail | 500 | A. flavus | [49] |
| Ducks | 1100 | A. parasiticus (NRRL 2999) | [35] |
| Ducks | 20 | A. flavus (CICC 2219) | [50] |
| Turkeys | 500 | A. parasiticus (NRRL 2999) | [36,37] |
| Bird | Aflatoxin Dose (μg/kg) | Fungal Strain | Relative Weights of Organs | Reference | ||
|---|---|---|---|---|---|---|
| Spleen | Bursa | Thymus | ||||
| Chickens | 40 | A. flavus (Chinese isolate) | + | − | − | [25] |
| Chickens | 22 | A. parasiticus (FRR 2999) | + | ± | ND | [29] |
| Chickens | 4000 | A. parasiticus (NRRL 2999) | + | ND | ND | [83] |
| Offspring of breeder hens | 5000 | A. flavus (NRRL 6540 CECT 2687) | − | − | ± | [80] |
| Turkeys | 330 | A. flavus (UNIGRAS 1231) | ± | ± | ND | [71] |
| Turkeys | 500 | A. parasiticus (NRRL 2999) | ± | ± | ND | [37] |
| Bird | Aflatoxin Dose (μg/kg) | Fungal Strain | Relative Weight of Liver | Reference |
|---|---|---|---|---|
| Chickens | 40 | A. flavus (Chinese isolate) | + | [25] |
| Chickens | 250 | A. flavus (KP137700) | + | [28] |
| Chickens | 1000 | A. flavus (NRRL 3357) | + | [30] |
| Ducks | 20 | A. flavus (CICC 2219) | + | [50] |
| Ducks | 1100 | A. parasiticus (NRRL 2999) | ± | [35] |
| Turkeys | 330 | A. flavus (UNIGRAS 1231) | − | [71] |
| Turkeys | 500 | A. parasiticus (NRRL 2999) | + | [37] |
| Laying hens | 500 | A. parasiticus (ATCC 15517) | + | [24] |
| Laying hens | 1000 | A. parasiticus | + | [107] |
| Laying hens | 2500 | A. parasiticus (SRRC 148) | + | [9] |
| Quail | 2500 | A. parasiticus (PTCC 5286) | + | [48] |
| Item | Amount (g/kg) | Aflatoxin Dose (μg/kg) | Bird | Reference |
|---|---|---|---|---|
| Clinoptilolite | 15.0 | 2500 (A. parasiticus NRRL 2999) | chickens | [134] |
| Hydrated sodium calcium aluminosilicate | 5.0 | 2000 (A. parasiticus NRRL 2999) | chickens | [135] |
| Hydrated sodium calcium aluminosilicate | 3.0 | 40 (A. flavus) | chickens | [25] |
| Bentonite | 7.5 | 2000 (A. parasiticus NRRL 2999) | chickens | [70] |
| Bentonite | 7.5 | 600 (A. flavus NRRL 6540; CECT 2687) | chickens | [136] |
| Nano-composite magnetic graphene oxide with chitosan | 5.0 | 22 (A. parasiticus FRR 2999) | chickens | [29] |
| Yeast cell walls 1 | 1.5 | 350 (naturally contaminated) | chickens | [72] |
| Yeast 2 | 1.0 | 100 (A. parasiticus NRRL 2999) | chickens | [140] |
| Probiotic 3 | 1.0 | 250 (A.flavus KP137700) | chickens | [28] |
| Probiotic 4 | 2.0 | 70 (naturally contaminated) | chickens | [143,144] |
| Probiotic 5 | 1.0 | 22 (naturally contaminated) | ducks | [56] |
| Probiotic 6 | 1.0 | 123 (naturally contaminated) | hens | [145] |
| Alpha-lipoic acid | 300 a | 300 (A. parasiticus NRRL 2999) | chickens | [77] |
| Urtica diocia seed extract | 300 b | 1000 (A. parasiticus NRRL 2999) | chickens | [122] |
| Grape seed proanthocyanidin extract | 250 a | 1000 (A. flavus NRRL 3357) | chickens | [30] |
| Curcuminoids | 74.0 a | 1000 (A. parasiticus NRRL 2999) | chickens | [154] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fouad, A.M.; Ruan, D.; El-Senousey, H.K.; Chen, W.; Jiang, S.; Zheng, C. Harmful Effects and Control Strategies of Aflatoxin B1 Produced by Aspergillus flavus and Aspergillus parasiticus Strains on Poultry: Review. Toxins 2019, 11, 176. https://doi.org/10.3390/toxins11030176
Fouad AM, Ruan D, El-Senousey HK, Chen W, Jiang S, Zheng C. Harmful Effects and Control Strategies of Aflatoxin B1 Produced by Aspergillus flavus and Aspergillus parasiticus Strains on Poultry: Review. Toxins. 2019; 11(3):176. https://doi.org/10.3390/toxins11030176
Chicago/Turabian StyleFouad, Ahmed Mohamed, Dong Ruan, HebatAllah Kasem El-Senousey, Wei Chen, Shouqun Jiang, and Chuntian Zheng. 2019. "Harmful Effects and Control Strategies of Aflatoxin B1 Produced by Aspergillus flavus and Aspergillus parasiticus Strains on Poultry: Review" Toxins 11, no. 3: 176. https://doi.org/10.3390/toxins11030176
APA StyleFouad, A. M., Ruan, D., El-Senousey, H. K., Chen, W., Jiang, S., & Zheng, C. (2019). Harmful Effects and Control Strategies of Aflatoxin B1 Produced by Aspergillus flavus and Aspergillus parasiticus Strains on Poultry: Review. Toxins, 11(3), 176. https://doi.org/10.3390/toxins11030176





