Evidence for Naturally Produced Beauvericins Containing N-Methyl-Tyrosine in Hypocreales Fungi
Abstract
:1. Introduction
2. Results and Discussion
2.1. Mass Spectrometry and Tentative Structure Determination of Beauvericins
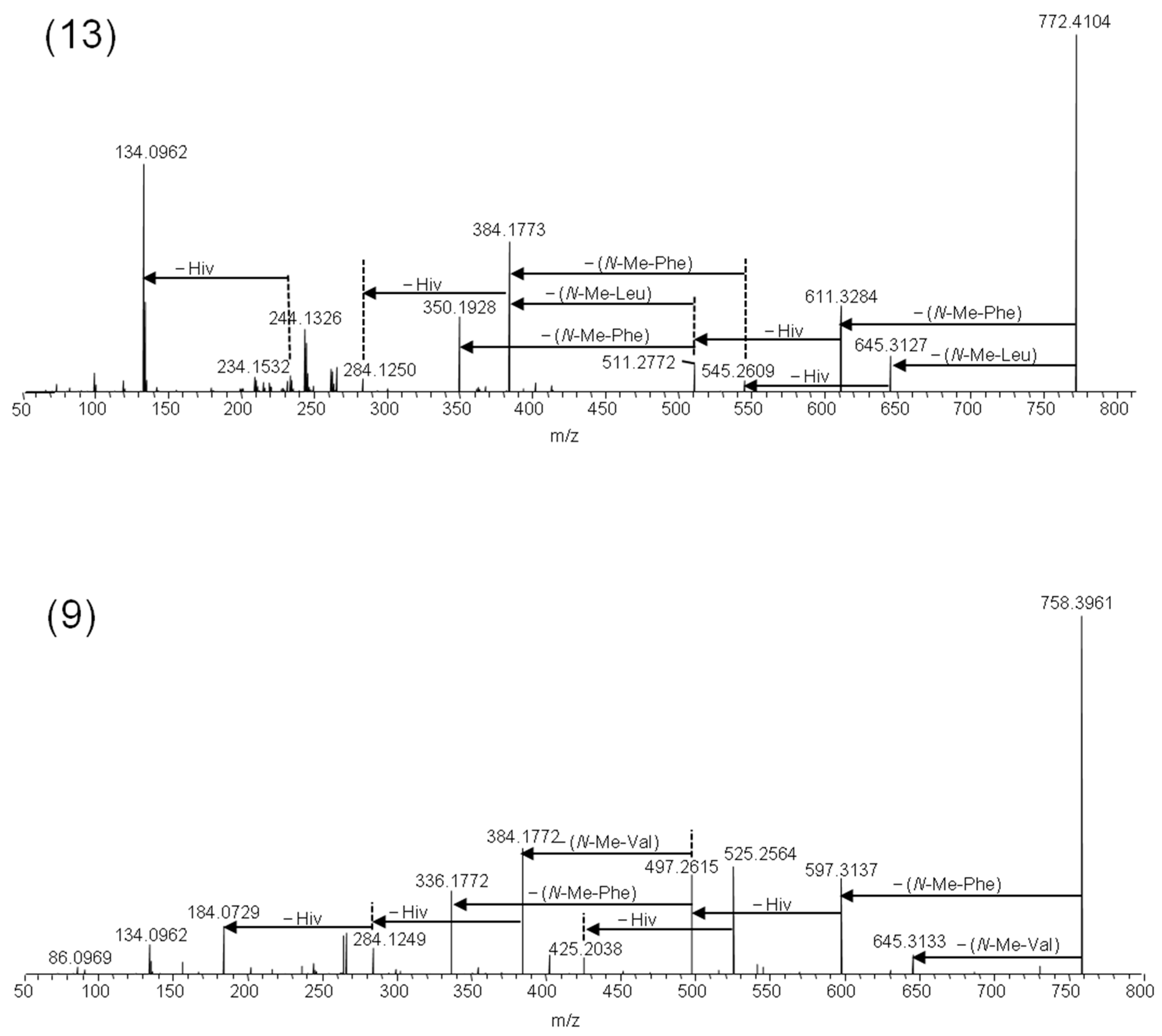
2.2. Mass Spectrometry and Tentative Structure Determination of Beauvenniatins
3. Conclusions
4. Materials and Methods
4.1. Fungal Strains
4.2. Media and Growth Conditions
4.3. DNA Extraction and Molecular Identification of Fungal Strains
4.4. Chemicals
4.5. Extraction
4.6. Liquid Chromatography–Ion-Trap Mass Spectrometry (ITMS)
4.7. Liquid Chromatography High-Resolution Mass Spectrometry (HRMS)
4.8. Chromatographic Fractionation, Acid Hydrolysis, and Amino and Hydroxy Acid Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Xu, L.J.; Liu, Y.S.; Zhou, L.G.; Wu, J.Y. Enhanced Beauvericin Production with in Situ Adsorption in Mycelial Liquid Culture of Fusarium redolens Dzf2. Process Biochem. 2009, 44, 1063–1067. [Google Scholar] [CrossRef]
- Hornbogen, T.; Glinski, M.; Zocher, R. Biosynthesis of Depsipeptide Mycotoxins in Fusarium. Eur. J. Plant Pathol. 2002, 108, 713–718. [Google Scholar] [CrossRef]
- Nilanonta, C.; Isaka, M.; Kittakoop, P.; Trakulnaleamsai, S.; Tanticharoen, M.; Thebtaranonth, Y. Precursor-Directed Biosynthesis of Beauvericin Analogs by the Insect Pathogenic Fungus Paecilomyces tenuipes Bcc 1614. Tetrahedron 2002, 58, 3355–3360. [Google Scholar] [CrossRef]
- Xu, Y.; Zhan, J.; Wijeratne, E.M.K.; Burns, A.M.; Gunatilaka, A.A.L.; Molnar, I. Cytotoxic and Antihaptotactic Beauvericin Analogues from Precursor-Directed Biosynthesis with the Insect Pathogen Beauveria bassiana Atcc 7159. J. Nat. Prod. 2007, 70, 1467–1471. [Google Scholar] [CrossRef] [PubMed]
- Fukuda, T.; Arai, M.; Tomoda, H.; Omura, S. New Beauvericins, Potentiators of Antifungal Miconazole Activity, Produced by Beauveria sp. Fki-1366—II. Structure Elucidation. J. Antibiot. 2004, 57, 117–124. [Google Scholar] [CrossRef]
- Fukuda, T.; Arai, M.; Yamaguchi, Y.; Masuma, R.; Tomoda, H.; Omura, S. New Beauvericins, Potentiators of Antifungal Miconazole Activity, Produced by Beauveria sp. Fki-1366—I. Taxonomy, Fermentation, Isolation and Biological Properties. J. Antibiot. 2004, 57, 110–116. [Google Scholar] [CrossRef]
- Logrieco, A.; Moretti, A.; Castella, G.; Kostecki, M.; Golinski, P.; Ritieni, A.; Chelkowski, J. Beauvericin Production by Fusarium Species. Appl. Environ. Microbiol. 1998, 64, 3084–3088. [Google Scholar]
- Luangsa-Ard, J.J.; Berkaew, P.; Ridkaew, R.; Hywel-Jones, N.L.; Isaka, M. A Beauvericin Hot Spot in the Genus Isaria. Mycol. Res. 2009, 113, 1389–1395. [Google Scholar] [CrossRef]
- Stepien, L.; Waskiewicz, A. Sequence Divergence of the Enniatin Synthase Gene in Relation to Production of Beauvericin and Enniatins in Fusarium Species. Toxins 2013, 5, 537–555. [Google Scholar] [CrossRef] [PubMed]
- Tomoda, H.; Huang, X.H.; Cao, J.; Nishida, H.; Nagao, R.; Okuda, S.; Tanaka, H.; Omura, S.; Arai, H.; Inoue, K. Inhibition of Acyl-Coa—Cholesterol Acyltransferase Activity by Cyclodepsipeptide Antibiotics. J. Antibiot. 1992, 45, 1626–1632. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.X.; Yan, K.Z.; Zhang, Y.; Huang, R.; Bian, J.; Zheng, C.S.; Sun, H.X.; Chen, Z.H.; Sun, N.; An, R.; et al. High-Throughput Synergy Screening Identifies Microbial Metabolites as Combination Agents for the Treatment of Fungal Infections. Proc. Natl. Acad. Sci. USA 2007, 104, 4606–4611. [Google Scholar] [CrossRef] [PubMed]
- Jow, G.M.; Chou, C.J.; Chen, B.F.; Tsai, J.H. Beauvericin Induces Cytotoxic Effects in Human Acute Lymphoblastic Leukemia Cells through Cytochrome C Release, Caspase 3 Activation: The Causative Role of Calcium. Cancer Lett. 2004, 216, 165–173. [Google Scholar] [CrossRef]
- Lin, H.I.; Lee, Y.J.; Chen, B.F.; Tsai, M.C.; Lu, J.L.; Chou, C.J.; Jow, G.M. Involvement of Bcl-2 Family, Cytochrome C and Caspase 3 in Induction of Apoptosis by Beauvericin in Human Non-Small Cell Lung Cancer Cells. Cancer Lett. 2005, 230, 248–259. [Google Scholar] [CrossRef] [PubMed]
- Isaka, M.; Yangchum, A.; Sappan, M.; Suvannakad, R.; Srikitikulchai, P. Cyclohexadepsipeptides from Acremonium sp. Bcc 28424. Tetrahedron 2011, 67, 7929–7935. [Google Scholar] [CrossRef]
- Bunyapaiboonsri, T.; Vongvilai, P.; Auncharoen, P.; Isaka, M. Cyclohexadepsipeptides from the Filamentous Fungus Acremonium sp. Bcc 2629. Helv. Chim. Acta 2012, 95, 963–972. [Google Scholar] [CrossRef]
- Santini, A.; Meca, G.; Uhlig, S.; Ritieni, A. Fusaproliferin, Beauvericin and Enniatins: Occurrence in Food—A Review. World Mycotoxin J. 2012, 5, 71–81. [Google Scholar] [CrossRef]
- Stepien, L.; Waskiewicz, A.; Urbaniak, M. Wildly Growing Asparagus (Asparagus officinalis L.) Hosts Pathogenic Fusarium Species and Accumulates Their Mycotoxins. Microb. Ecol. 2016, 71, 927–937. [Google Scholar] [CrossRef] [PubMed]
- Logrieco, A.; Rizzo, A.; Ferracane, R.; Ritieni, A. Occurrence of Beauvericin and Enniatins in Wheat Affected by Fusarium avenaceum Head Blight. Appl. Environ. Microbiol. 2002, 68, 82–85. [Google Scholar] [CrossRef] [PubMed]
- Galvez, L.; Urbaniak, M.; Waskiewicz, A.; Stepien, L.; Palmero, D. Fusarium proliferatum—Causal Agent of Garlic Bulb Rot in Spain: Genetic Variability and Mycotoxin Production. Food Microbiol. 2017, 67, 41–48. [Google Scholar] [CrossRef]
- Tuiche, M.V.; Lopes, A.A.; Silva, D.B.; Lopes, N.P.; Pupo, M.T. Direct Maldi-Tof/Tof Analyses of Unnatural Beauvericins Produced by the Endophytic Fungus Fusarium oxysporum Ss46. Rev. Bras. Farmacogn. 2014, 24, 433–438. [Google Scholar] [CrossRef]
- Kozlowska, E.; Hoc, N.; Sycz, J.; Urbaniak, M.; Dymarska, M.; Grzeszczuk, J.; Kostrzewa-Suslow, E.; Stepien, L.; Plaskowska, E.; Janeczko, T. Biotransformation of Steroids by Entomopathogenic Strains of Isaria farinosa. Microb. Cell Fact. 2018, 17, 71. [Google Scholar] [CrossRef] [PubMed]
- Gorczyca, A.; Oleksy, A.; Gala-Czekaj, D.; Urbaniak, M.; Laskowska, M.; Waskiewicz, A.; Stepien, L. Fusarium Head Blight Incidence and Mycotoxin Accumulation in Three Durum Wheat Cultivars in Relation to Sowing Date and Density. Sci. Nat. 2018, 105, 2. [Google Scholar] [CrossRef] [PubMed]
- Carbone, I.; Kohn, L.M. A method for designing primer sets for speciation studies in filamentous ascomycetes. Mycologia 1999, 91, 553–556. [Google Scholar] [CrossRef]
- Kullnig-Gradinger, C.M.; Szakacs, G.; Kubicek, C.P. Phylogeny and evolution of genus Trichoderma a multigene approach. Mycol. Res. 2002, 106, 757–767. [Google Scholar] [CrossRef]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetic. In PCR Protocols, a Guide to Methods and Applications; Innis, M.A., Gelfand, D.H., Shinsky, J.J., White, T.J., Eds.; Academic Press: San Diego, CA, USA, 1990; pp. 315–322. [Google Scholar]
- Kozlowska, E.; Urbaniak, M.; Hoc, N.; Grzeszczuk, J.; Dymarska, M.; Stepien, L.; Plaskowska, E.; Kostrzewa-Suslow, E.; Janeczko, T. Cascade Biotransformation of Dehydroepiandrosterone (Dhea) by Beauveria Species. Sci. Rep. 2018, 8, 13449. [Google Scholar] [CrossRef] [PubMed]
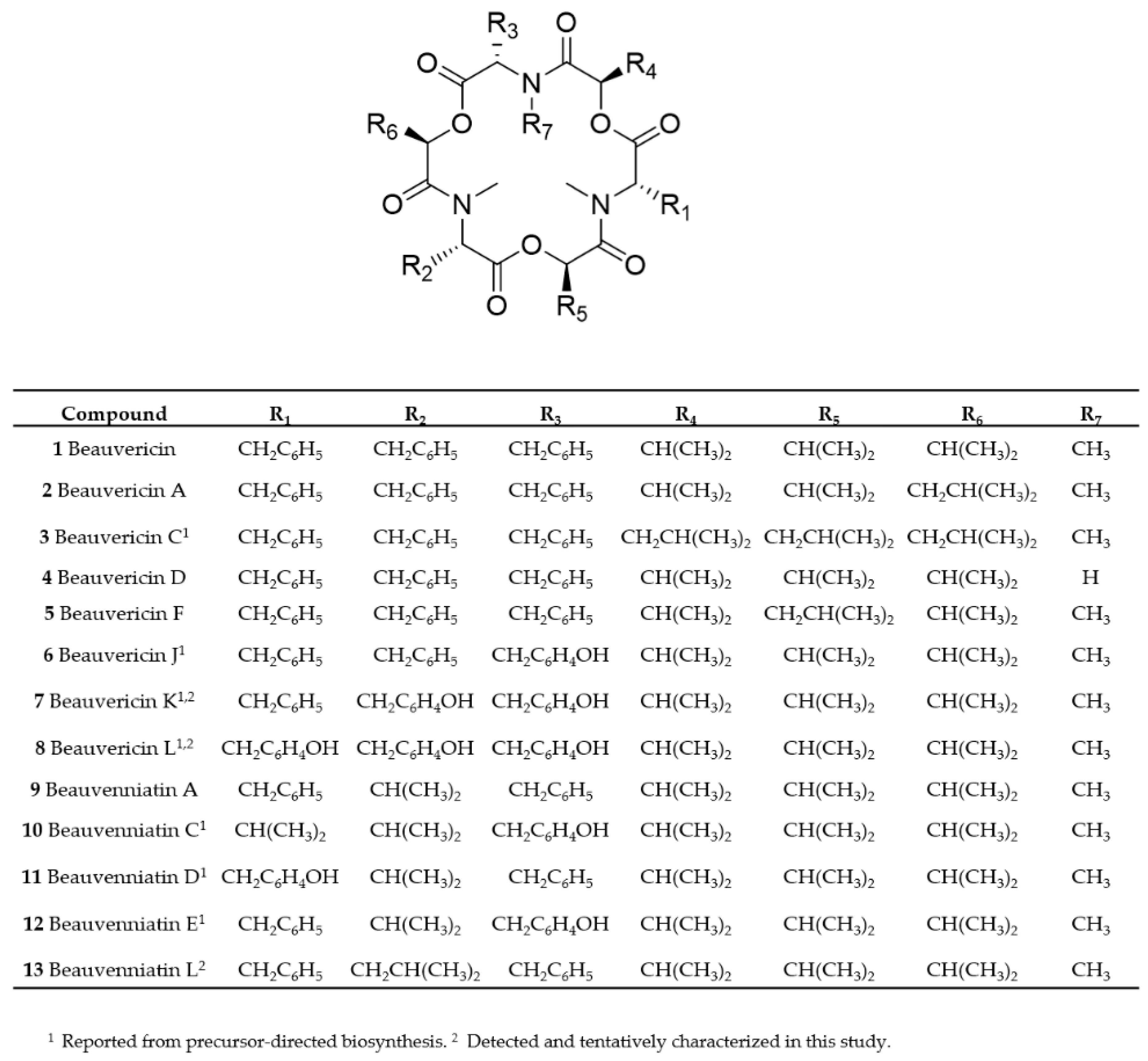
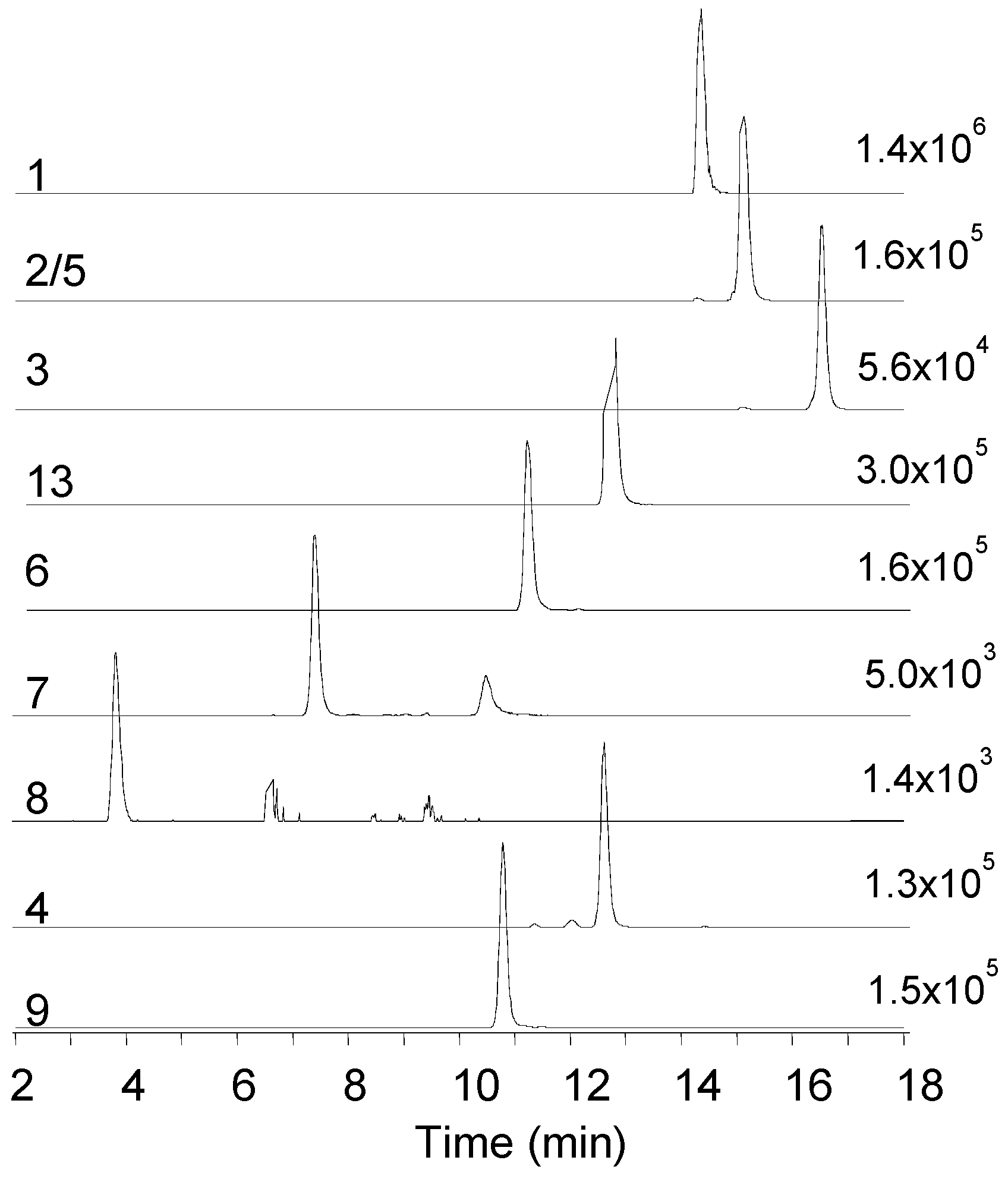
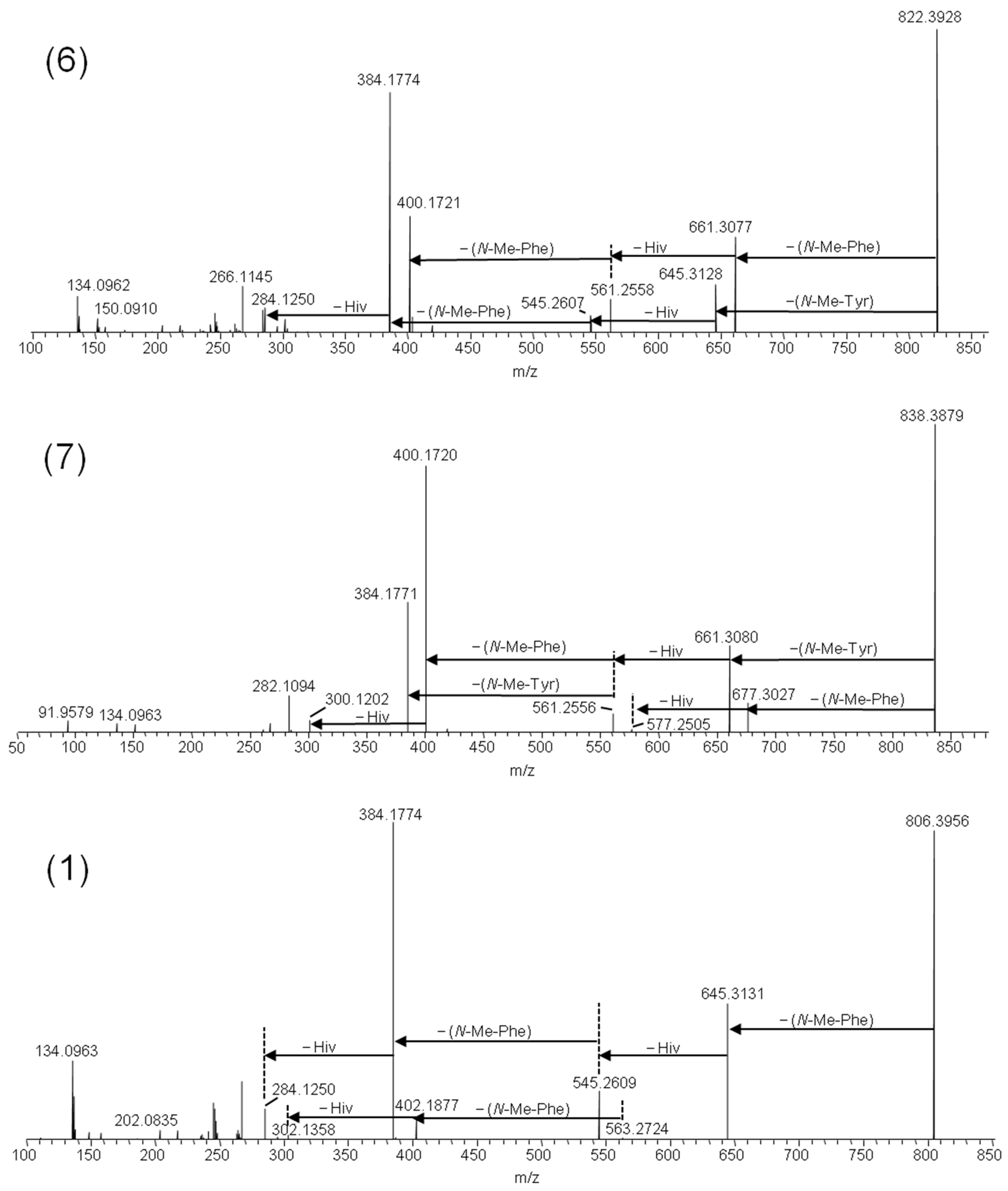
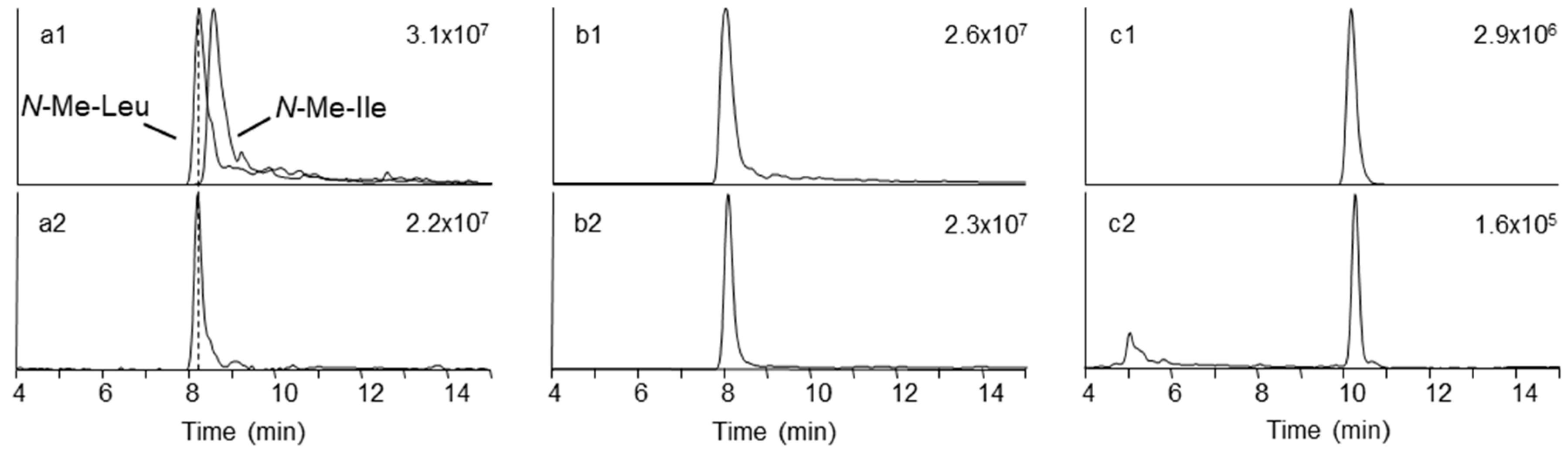
| Strain | Species | Host | Sequence Nucleotide Identity | Metabolic Profile * |
|---|---|---|---|---|
| RT 6.7 | Fusarium proliferatum | rice (Oryza sativa) | 99.78% identity to the Fusarium proliferatum acc. number JF740730.1 | beauvericin (87.0%), beauvericin A/F (0.8%), beauvericin D (3.5%), beauvenniatin A (0.8%), beauvenniatin L (3.2%), beauvericin J (4.7%), beauvericin K (trace) |
| RT 5.4 | Fusarium proliferatum | rice (Oryza sativa) | 99.78% identity to the Fusarium proliferatum acc. number JF740730.1 | beauvericin (95.6%), beauvericin D (2.6%), beauvenniatin L (0.8%), beauvericin J (0.9%) |
| MU12 | Fusarium verticillioides | banana (Musa L.) | 98.66% identity to the Fusarium verticillioides acc. number JF740717.1 | beauvericin (18.4%), beauvericin D (8.5%), beauvenniatin A (61.3%), beauvericin K (11.8%) |
| P35 | Fusarium concentricum | pineapple (Ananas comosus) | 100% identity to the Fusarium concentricum, acc. number JF740760.1 | beauvericin (77.9%), beauvericin A/F (2.2%), beauvericin D (3.6%), beauvenniatin A (3.6%), beauvenniatin L (8.6%), beauvericin J (4.1%), beauvericin K (trace), beauvericin L (trace) |
| PIN 5.5 | Fusarium proliferatum | unknown | 99.32% identity to the Fusarium proliferatum acc. number JF740730.1 | beauvericin (87.7%), beauvericin A/F (1.0%), beauvericin D (3.1%), beauvenniatin A (1.2%), beauvenniatin L (5.3%), beauvericin J (1.8%) |
| 4447 | Isaria farinosa | bark beetle (Trypodendron lineatum) | 100% identity to the Isaria farinosa, acc. number AY624181.1 | beauvericin (35.4%), beauvericin A/F (42.5%), beauvericin C (13.8%), beauvericin D (1.5%), beauvenniatin A (0.4%), beauvenniatin L (0.3%), beauvericin J (5.5%), beauvericin K (0.2%), beauvericin L (0.3%) |
| Compound | Measured (m/z) [M + NH4]+ | Measured (m/z) [M + Na]+ | Retention Time (min) | Elemental Composition of Neutral Molecule | Mass Error (ppm) [M + NH4]+ | Mass Error (ppm) [M + Na]+ |
|---|---|---|---|---|---|---|
| 1 Beauvericin | 801.4427 | 806.3956 | 14.4 | C45H57N3O9 | 0.0 | −1.5 |
| 2/5 Beauvericin A/F | 815.4582 | 820.4112 | 15.1 | C46H59N3O9 | −1.1 | −1.7 |
| 3 Beauvericin C | 843.4897 | 848.4433 | 16.5 | C48H63N3O9 | −0.4 | −1.6 |
| 4 Beauvericin D | 787.4269 | 792.3807 | 12.6 | C44H55N3O9 | −1.2 | −2.4 |
| 6 Beauvericin J | 817.4409 | 822.3928 | 11.1 | C45H57N3O10 | 2.6 | −1.0 |
| 7 Beauvericin K | 833.4332 | 838.3879 | 7.4 | C45H57N3O11 | −0.3 | −0.8 |
| 8 Beauvericin L | 849.4281 | 854.3825 | 3.8 | C45H57N3O12 | −0.0 | −1.1 |
| 9 Beauvenniatin A | 753.4432 | 758.3961 | 10.8 | C41H57N3O9 | −0.3 | −0.4 |
| 13 Beauvenniatin L | 767.4583 | 772.4104 | 12.7 | C42H59N3O9 | −0.7 | −0.8 |
| % Methanol | Beauvericin Analogue |
|---|---|
| 1 | beauvenniatin A |
| 2 | beauvericin, beauvericin A/F, beauvenniatin L, beauvericin C, beauvericin D |
| 4 | beauvericin J, beauvericin K, beauvericin L |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Urbaniak, M.; Stępień, Ł.; Uhlig, S. Evidence for Naturally Produced Beauvericins Containing N-Methyl-Tyrosine in Hypocreales Fungi. Toxins 2019, 11, 182. https://doi.org/10.3390/toxins11030182
Urbaniak M, Stępień Ł, Uhlig S. Evidence for Naturally Produced Beauvericins Containing N-Methyl-Tyrosine in Hypocreales Fungi. Toxins. 2019; 11(3):182. https://doi.org/10.3390/toxins11030182
Chicago/Turabian StyleUrbaniak, Monika, Łukasz Stępień, and Silvio Uhlig. 2019. "Evidence for Naturally Produced Beauvericins Containing N-Methyl-Tyrosine in Hypocreales Fungi" Toxins 11, no. 3: 182. https://doi.org/10.3390/toxins11030182
APA StyleUrbaniak, M., Stępień, Ł., & Uhlig, S. (2019). Evidence for Naturally Produced Beauvericins Containing N-Methyl-Tyrosine in Hypocreales Fungi. Toxins, 11(3), 182. https://doi.org/10.3390/toxins11030182







