Whole-Cell Multiparameter Assay for Ricin and Abrin Activity-Based Digital Holographic Microscopy
Abstract
1. Introduction
2. Results
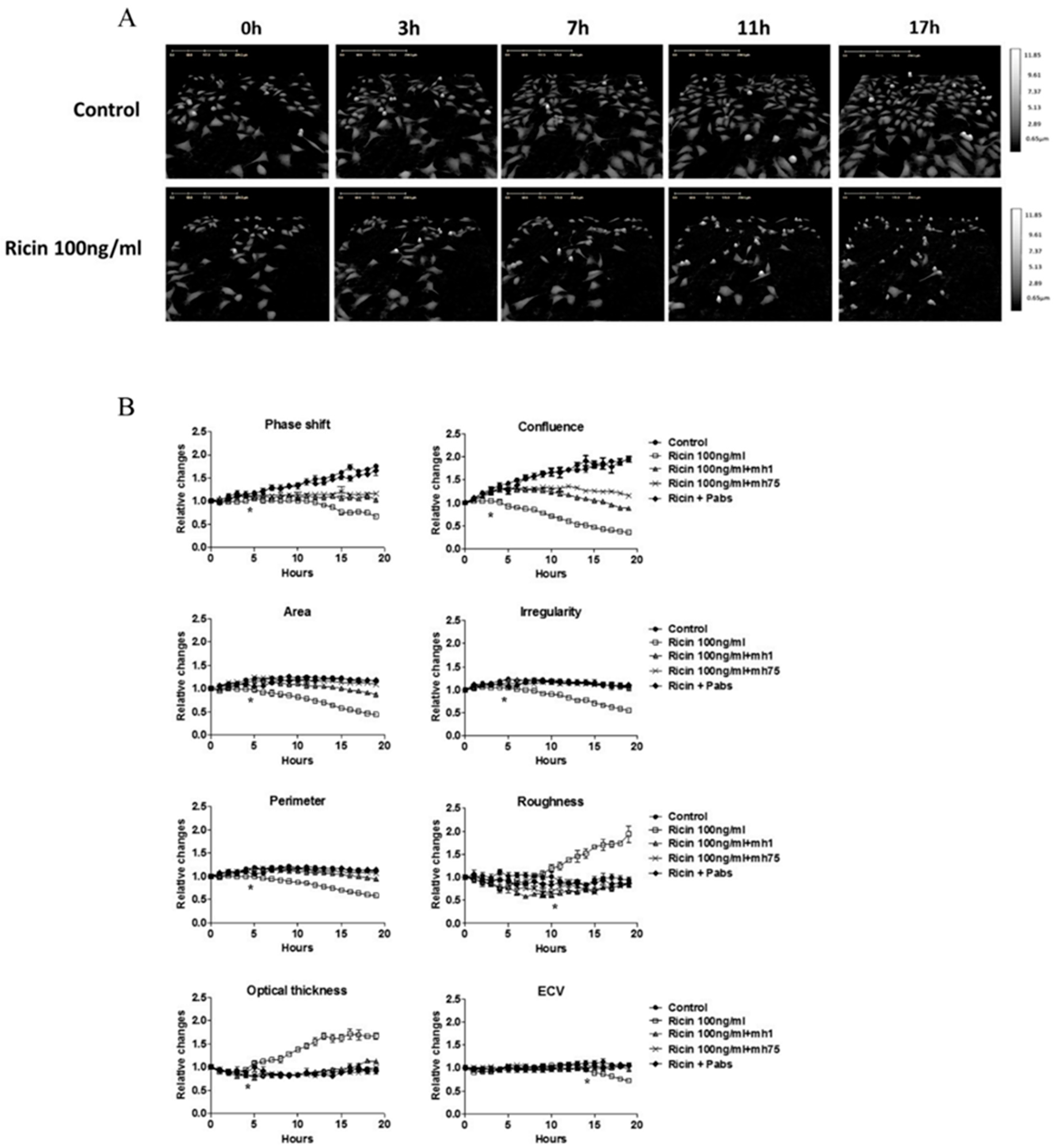
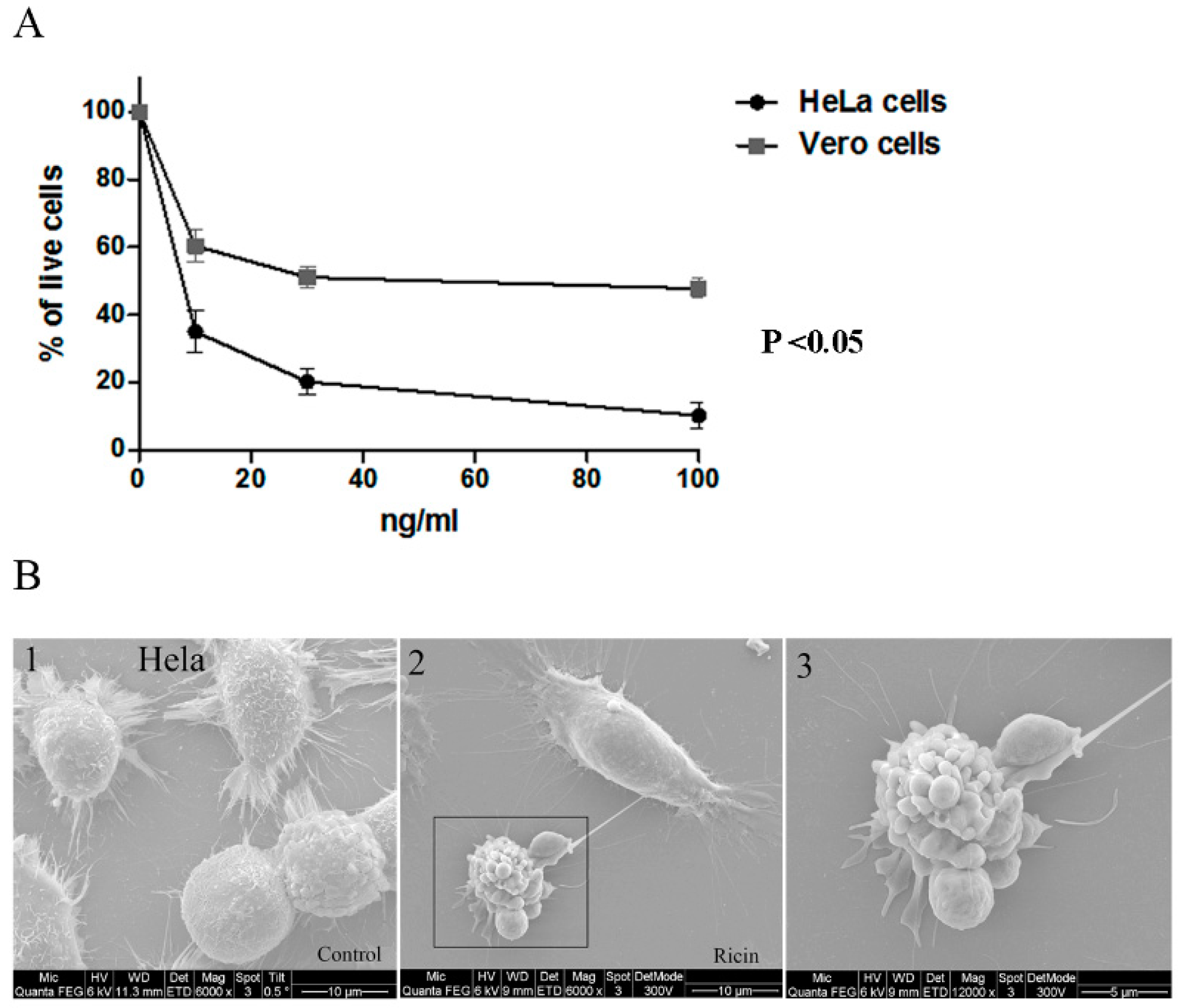
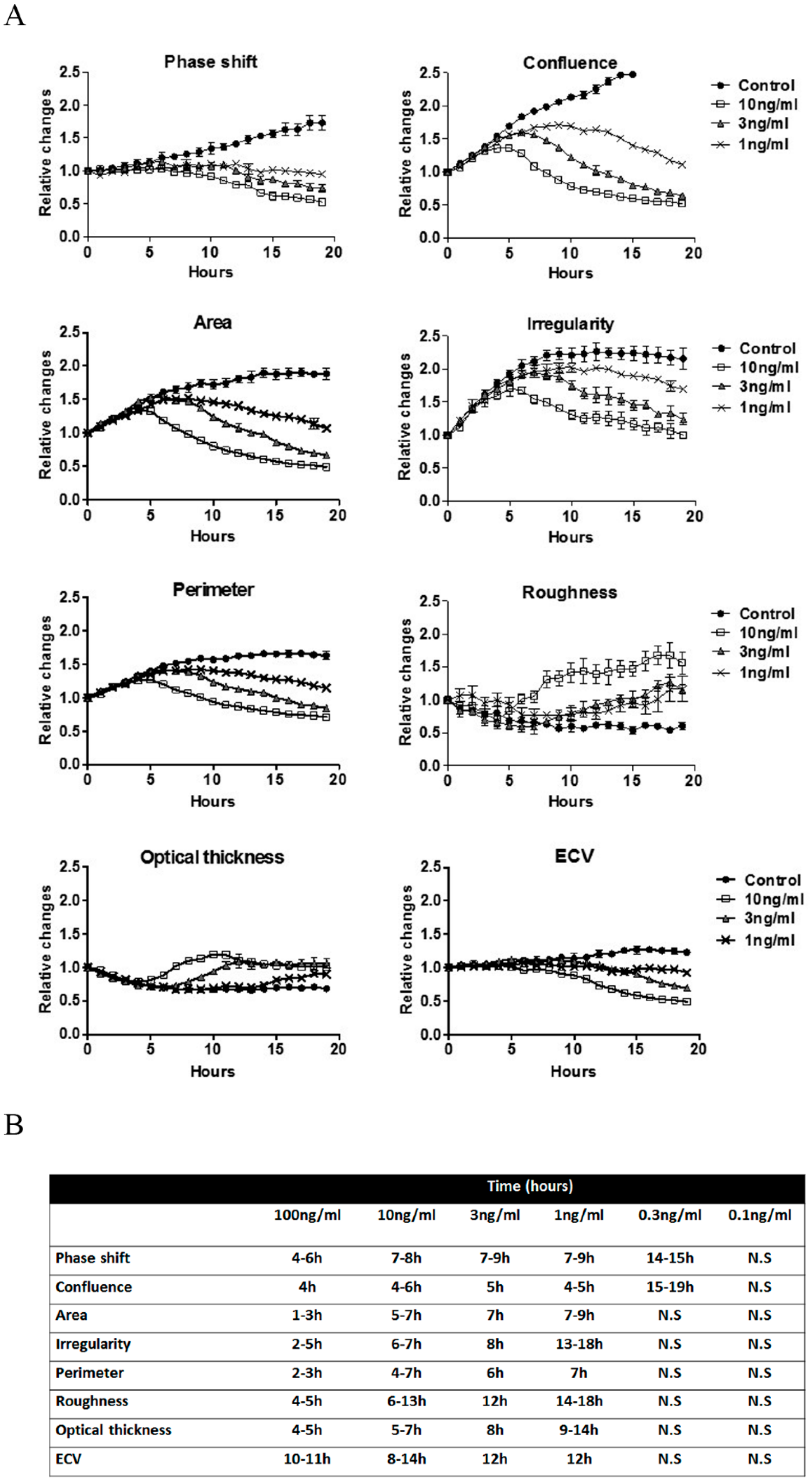
2.1. Morphological Changes in Cell Lines Observed by DHM Following Ricin Exposure
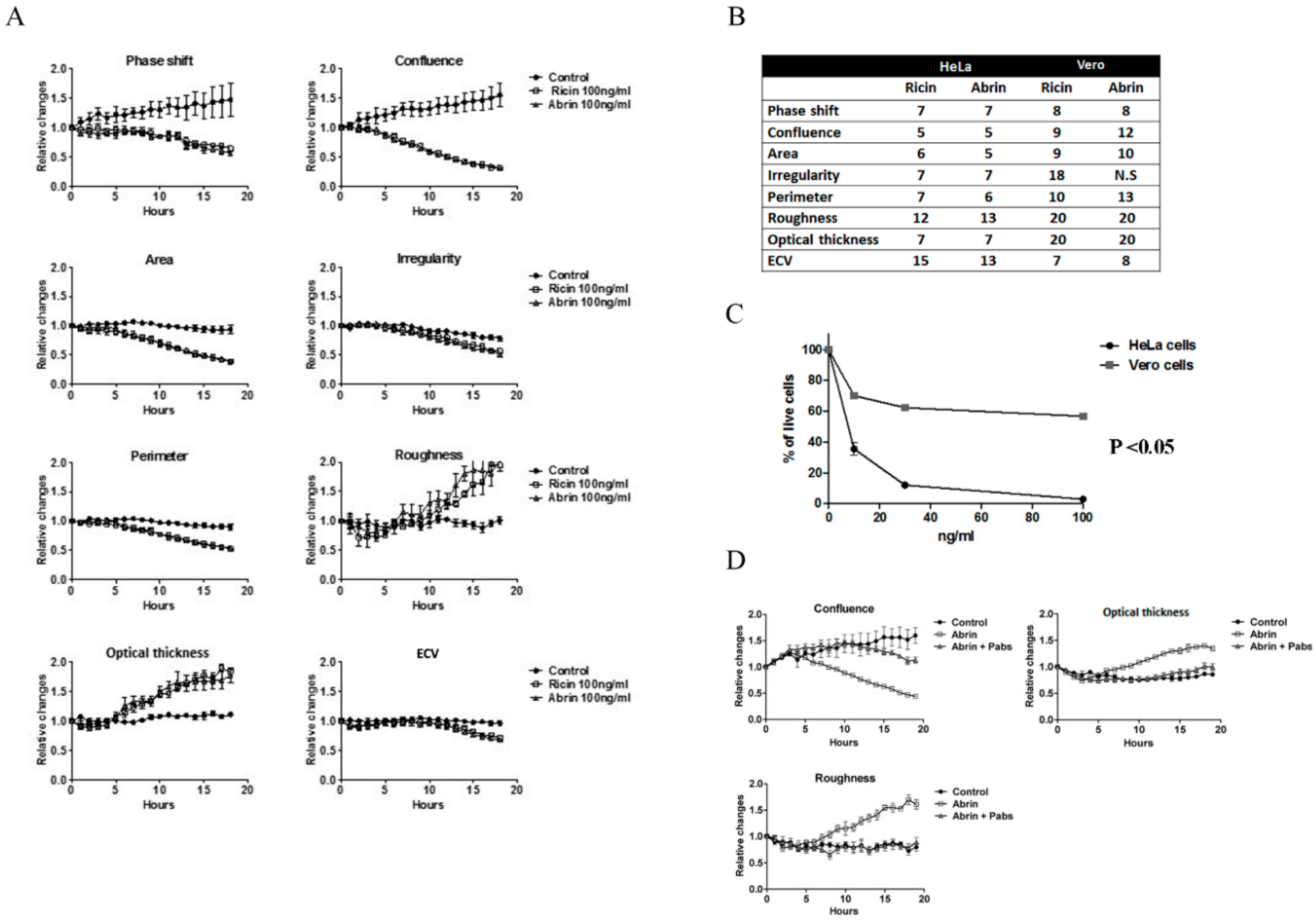
2.2. Similarities in Morphological Features during Abrin Toxicity
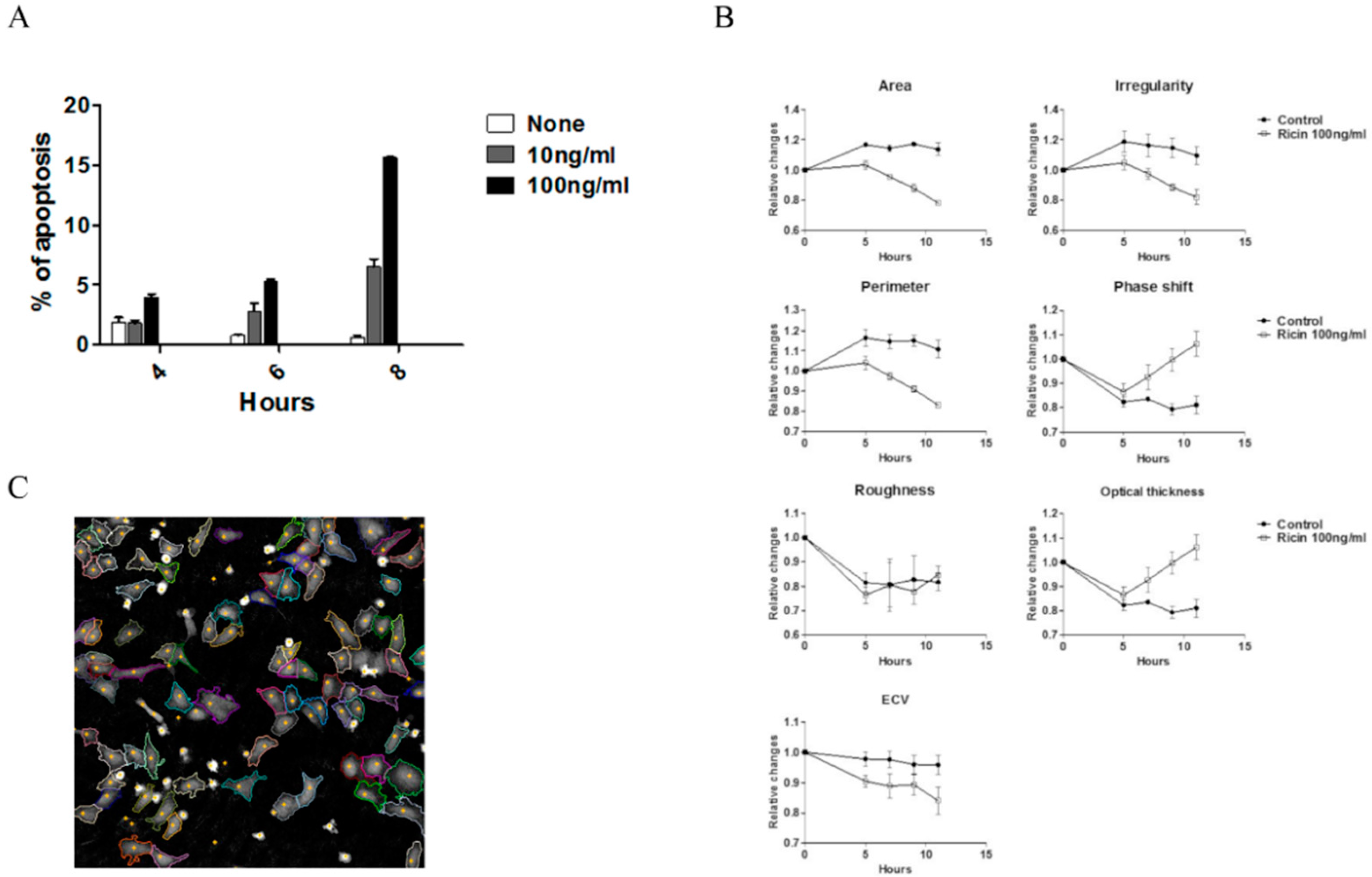
2.3. Early Morphological Changes Detected by DHM Are Attributed to Live Cells
2.4. Increased Susceptibility to RIP Intoxication during Cell Adhesion
3. Discussion
4. Materials and Methods
4.1. Toxins and Antibodies
4.2. Cell Lines
4.3. Digital Holographic Microscopy
4.4. Viability Assay
4.5. Flow Cytometry-Apoptosis Assay
4.6. Scanning Electron Microscopy
4.7. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Stirpe, F. Ribosome-inactivating proteins. Toxicon 2004, 44, 371–383. [Google Scholar] [CrossRef]
- Sperti, S.; Montanaro, L.; Mattioli, A.; Testoni, G. Relationship between elongation factor I- and elongation factor II- dependent guanosine triphosphatase activities of ribosomes. Inhibition of both activities by ricin. Biochem. J. 1975, 148, 447–451. [Google Scholar] [CrossRef] [PubMed]
- Olivieri, F.; Prasad, V.; Valbonesi, P.; Srivastava, S.; Ghosal-Chowdhury, P.; Barbieri, L.; Bolognesi, A.; Stirpe, F. A systemic antiviral resistance-inducing protein isolated from Clerodendrum inerme Gaertn. is a polynucleotide:adenosine glycosidase (ribosome-inactivating protein). FEBS Lett. 1996, 396, 132–134. [Google Scholar] [CrossRef]
- Stirpe, F.; Barbieri, L.; Battelli, M.G.; Soria, M.; Lappi, D.A. Ribosome-inactivating proteins from plants: Present status and future prospects. Biotechnology (N Y) 1992, 10, 405–412. [Google Scholar] [CrossRef]
- Endo, Y.; Tsurugi, K. RNA N-glycosidase activity of ricin A-chain. Mechanism of action of the toxic lectin ricin on eukaryotic ribosomes. J. Biol. Chem. 1987, 262, 8128–8130. [Google Scholar]
- Gal, Y.; Alcalay, R.; Sabo, T.; Noy-Porat, T.; Epstein, E.; Kronman, C.; Mazor, O. Rapid assessment of antibody-induced ricin neutralization by employing a novel functional cell-based assay. J. Immunol. Methods 2015, 424, 136–139. [Google Scholar] [CrossRef]
- Iordanov, M.S.; Pribnow, D.; Magun, J.L.; Dinh, T.H.; Pearson, J.A.; Chen, S.L.; Magun, B.E. Ribotoxic stress response: Activation of the stress-activated protein kinase JNK1 by inhibitors of the peptidyl transferase reaction and by sequence-specific RNA damage to the alpha-sarcin/ricin loop in the 28S rRNA. Mol. Cell Biol. 1997, 17, 3373–3381. [Google Scholar] [CrossRef]
- Saenz, J.B.; Doggett, T.A.; Haslam, D.B. Identification and characterization of small molecules that inhibit intracellular toxin transport. Infect. Immun. 2007, 75, 4552–4561. [Google Scholar] [CrossRef]
- Brigotti, M.; Barbieri, L.; Valbonesi, P.; Stirpe, F.; Montanaro, L.; Sperti, S. A rapid and sensitive method to measure the enzymatic activity of ribosome-inactivating proteins. Nucleic Acids Res. 1998, 26, 4306–4307. [Google Scholar] [CrossRef]
- Chen, X.Y.; Link, T.M.; Schramm, V.L. Ricin A-chain: Kinetics, mechanism, and RNA stem-loop inhibitors. Biochemistry 1998, 37, 11605–11613. [Google Scholar] [CrossRef]
- Falach, R.; Sapoznikov, A.; Gal, Y.; Israeli, O.; Leitner, M.; Seliger, N.; Ehrlich, S.; Kronman, C.; Sabo, T. Quantitative profiling of the in vivo enzymatic activity of ricin reveals disparate depurination of different pulmonary cell types. Toxicol. Lett. 2016, 258, 11–19. [Google Scholar] [CrossRef]
- Hines, H.B.; Brueggemann, E.E.; Hale, M.L. High-performance liquid chromatography-mass selective detection assay for adenine released from a synthetic RNA substrate by ricin A chain. Anal. Biochem. 2004, 330, 119–122. [Google Scholar] [CrossRef]
- Li, M.; Pestka, J.J. Comparative induction of 28S ribosomal RNA cleavage by ricin and the trichothecenes deoxynivalenol and T-2 toxin in the macrophage. Toxicol. Sci. 2008, 105, 67–78. [Google Scholar] [CrossRef]
- Stirpe, F.; Bailey, S.; Miller, S.P.; Bodley, J.W. Modification of ribosomal RNA by ribosome-inactivating proteins from plants. Nucleic Acids Res. 1988, 16, 1349–1357. [Google Scholar] [CrossRef]
- Taylor, B.E.; Irvin, J.D. Depurination of plant ribosomes by pokeweed antiviral protein. FEBS Lett. 1990, 273, 144–146. [Google Scholar] [CrossRef]
- Prigent, J.; Panigai, L.; Lamourette, P.; Sauvaire, D.; Devilliers, K.; Plaisance, M.; Volland, H.; Creminon, C.; Simon, S. Neutralising antibodies against ricin toxin. PLoS ONE 2011, 6, e20166. [Google Scholar] [CrossRef]
- Vance, D.J.; Tremblay, J.M.; Mantis, N.J.; Shoemaker, C.B. Stepwise engineering of heterodimeric single domain camelid VHH antibodies that passively protect mice from ricin toxin. J. Biol. Chem. 2013, 288, 36538–36547. [Google Scholar] [CrossRef]
- Cohen, O.; Mechaly, A.; Sabo, T.; Alcalay, R.; Aloni-Grinstein, R.; Seliger, N.; Kronman, C.; Mazor, O. Characterization and epitope mapping of the polyclonal antibody repertoire elicited by ricin holotoxin-based vaccination. Clin. Vaccine Immunol. 2014, 21, 1534–1540. [Google Scholar] [CrossRef]
- Halter, M.; Almeida, J.L.; Tona, A.; Cole, K.D.; Plant, A.L.; Elliott, J.T. A mechanistically relevant cytotoxicity assay based on the detection of cellular GFP. Assay Drug. Dev. Technol. 2009, 7, 356–365. [Google Scholar] [CrossRef]
- Wahome, P.G.; Ahlawat, S.; Mantis, N.J. Identification of small molecules that suppress ricin-induced stress-activated signaling pathways. PLoS ONE 2012, 7, e49075. [Google Scholar] [CrossRef]
- Duprez, L.; Wirawan, E.; Vanden Berghe, T.; Vandenabeele, P. Major cell death pathways at a glance. Microbes Infect 2009, 11, 1050–1062. [Google Scholar] [CrossRef] [PubMed]
- Krysko, D.V.; Vanden Berghe, T.; D’Herde, K.; Vandenabeele, P. Apoptosis and necrosis: Detection, discrimination and phagocytosis. Methods 2008, 44, 205–221. [Google Scholar] [CrossRef] [PubMed]
- Rao, P.V.; Jayaraj, R.; Bhaskar, A.S.; Kumar, O.; Bhattacharya, R.; Saxena, P.; Dash, P.K.; Vijayaraghavan, R. Mechanism of ricin-induced apoptosis in human cervical cancer cells. Biochem. Pharmacol. 2005, 69, 855–865. [Google Scholar] [CrossRef] [PubMed]
- Saxena, N.; Yadav, P.; Kumar, O. The Fas/Fas ligand apoptotic pathway is involved in abrin-induced apoptosis. Toxicol. Sci. 2013, 135, 103–118. [Google Scholar] [CrossRef] [PubMed]
- Lecoeur, H. Nuclear apoptosis detection by flow cytometry: Influence of endogenous endonucleases. Exp. Cell Res. 2002, 277, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Zamai, L.; Falcieri, E.; Marhefka, G.; Vitale, M. Supravital exposure to propidium iodide identifies apoptotic cells in the absence of nucleosomal DNA fragmentation. Cytometry 1996, 23, 303–311. [Google Scholar] [CrossRef]
- Korzeniewski, C.; Callewaert, D.M. An enzyme-release assay for natural cytotoxicity. J. Immunol. Methods 1983, 64, 313–320. [Google Scholar] [CrossRef]
- Neumann, B.; Held, M.; Liebel, U.; Erfle, H.; Rogers, P.; Pepperkok, R.; Ellenberg, J. High-throughput RNAi screening by time-lapse imaging of live human cells. Nat. Methods 2006, 3, 385–390. [Google Scholar] [CrossRef]
- Strbkova, L.; Zicha, D.; Vesely, P.; Chmelik, R. Automated classification of cell morphology by coherence-controlled holographic microscopy. J. Biomed. Opt. 2017, 22, 1–9. [Google Scholar] [CrossRef]
- Khmaladze, A.; Matz, R.L.; Epstein, T.; Jasensky, J.; Banaszak Holl, M.M.; Chen, Z. Cell volume changes during apoptosis monitored in real time using digital holographic microscopy. J. Struct. Biol. 2012, 178, 270–278. [Google Scholar] [CrossRef]
- Kühn, J.; Shaffer, E.; Mena, J.; Breton, B.; Parent, J.; Rappaz, B.; Chambon, M.; Emery, Y.; Magistretti, P.; Depeursinge, C.; et al. Label-free cytotoxicity screening assay by digital holographic microscopy. Assay Drug Dev. Technol. 2013, 11, 101–107. [Google Scholar] [CrossRef]
- Molder, A.; Sebesta, M.; Gustafsson, M.; Gisselson, L.; Wingren, A.G.; Alm, K. Non-invasive, label-free cell counting and quantitative analysis of adherent cells using digital holography. J. Microsc. 2008, 232, 240–247. [Google Scholar] [CrossRef]
- Bettenworth, D.; Lenz, P.; Krausewitz, P.; Bruckner, M.; Ketelhut, S.; Domagk, D.; Kemper, B. Quantitative stain-free and continuous multimodal monitoring of wound healing in vitro with digital holographic microscopy. PLoS ONE 2014, 9, e107317. [Google Scholar] [CrossRef]
- El-Schich, Z.; Molder, A.; Tassidis, H.; Harkonen, P.; Falck Miniotis, M.; Gjorloff Wingren, A. Induction of morphological changes in death-induced cancer cells monitored by holographic microscopy. J. Struct. Biol. 2015, 189, 207–212. [Google Scholar] [CrossRef]
- Falck Miniotis, M.; Mukwaya, A.; Gjorloff Wingren, A. Digital holographic microscopy for non-invasive monitoring of cell cycle arrest in L929 cells. PLoS ONE 2014, 9, e106546. [Google Scholar] [CrossRef]
- Marquet, P.; Rappaz, B.; Magistretti, P.J.; Cuche, E.; Emery, Y.; Colomb, T.; Depeursinge, C. Digital holographic microscopy: A noninvasive contrast imaging technique allowing quantitative visualization of living cells with subwavelength axial accuracy. Opt. Lett. 2005, 30, 468–470. [Google Scholar] [CrossRef]
- Pavillon, N.; Kuhn, J.; Moratal, C.; Jourdain, P.; Depeursinge, C.; Magistretti, P.J.; Marquet, P. Early cell death detection with digital holographic microscopy. PLoS ONE 2012, 7, e30912. [Google Scholar] [CrossRef]
- Rappaz, B.; Breton, B.; Shaffer, E.; Turcatti, G. Digital holographic microscopy: A quantitative label-free microscopy technique for phenotypic screening. Comb. Chem. High Throughput Screen 2014, 17, 80–88. [Google Scholar] [CrossRef]
- Fang, Y. Label-free biosensors for cell biology. Int. J. Electrochem. 2011, 2011. [Google Scholar] [CrossRef]
- Noy-Porat, T.; Rosenfeld, R.; Ariel, N.; Epstein, E.; Alcalay, R.; Zvi, A.; Kronman, C.; Ordentlich, A.; Mazor, O. Isolation of Anti-Ricin Protective Antibodies Exhibiting High Affinity from Immunized Non-Human Primates. Toxins 2016, 8. [Google Scholar] [CrossRef]
- Cherubin, P.; Quinones, B.; Elkahoui, S.; Yokoyama, W.; Teter, K. A Cell-Based Fluorescent Assay to Detect the Activity of AB Toxins that Inhibit Protein Synthesis. Methods Mol. Biol. 2017, 1600, 25–36. [Google Scholar] [CrossRef]
- Damiano, J.S.; Cress, A.E.; Hazlehurst, L.A.; Shtil, A.A.; Dalton, W.S. Cell adhesion mediated drug resistance (CAM-DR): Role of integrins and resistance to apoptosis in human myeloma cell lines. Blood 1999, 93, 1658–1667. [Google Scholar]
- Durand, R.E.; Sutherland, R.M. Effects of intercellular contact on repair of radiation damage. Exp. Cell Res. 1972, 71, 75–80. [Google Scholar] [CrossRef]
- Schuetz, J.D.; Schuetz, E.G. Extracellular matrix regulation of multidrug resistance in primary monolayer cultures of adult rat hepatocytes. Cell Growth Differ. 1993, 4, 31–40. [Google Scholar]
- Bauwens, A.; Bielaszewska, M.; Kemper, B.; Langehanenberg, P.; von Bally, G.; Reichelt, R.; Mulac, D.; Humpf, H.U.; Friedrich, A.W.; Kim, K.S.; et al. Differential cytotoxic actions of Shiga toxin 1 and Shiga toxin 2 on microvascular and macrovascular endothelial cells. Thromb. Haemost. 2011, 105, 515–528. [Google Scholar] [CrossRef]
- Mölder, A.L.; Persson, J.; El-Schich, Z.; Czanner, S.; Gjorloff-Wingren, A. Supervised classification of etoposide-treated in vitro adherent cells based on noninvasive imaging morphology. J. Med. Imaging (Bellingham) 2017, 4, 021106. [Google Scholar] [CrossRef]
- Song, K.; Mize, R.R.; Marrero, L.; Corti, M.; Kirk, J.M.; Pincus, S.H. Antibody to ricin a chain hinders intracellular routing of toxin and protects cells even after toxin has been internalized. PLoS ONE 2013, 8, e62417. [Google Scholar] [CrossRef] [PubMed]
- Rappaz, B.; Kuttler, F.; Breton, B.; Turcatti, G. Digital Holographic Imaging for label-free phenotypic profiling, cytotoxicity, and chloride channels target screening. Methods Pharmacol. Toxicol. 2015, 53, 307–323. [Google Scholar] [CrossRef]
- Abraham, V.C.; Towne, D.L.; Waring, J.F.; Warrior, U.; Burns, D.J. Application of a high-content multiparameter cytotoxicity assay to prioritize compounds based on toxicity potential in humans. J. Biomol. Screen. 2008, 13, 527–537. [Google Scholar] [CrossRef]
- Mery, B.; Guy, J.B.; Vallard, A.; Espenel, S.; Ardail, D.; Rodriguez-Lafrasse, C.; Rancoule, C.; Magne, N. In Vitro Cell Death Determination for Drug Discovery: A Landscape Review of Real Issues. J. Cell Death 2017, 10, 1179670717691251. [Google Scholar] [CrossRef]
- Martin, H.L.; Adams, M.; Higgins, J.; Bond, J.; Morrison, E.E.; Bell, S.M.; Warriner, S.; Nelson, A.; Tomlinson, D.C. High-content, high-throughput screening for the identification of cytotoxic compounds based on cell morphology and cell proliferation markers. PLoS ONE 2014, 9, e88338. [Google Scholar] [CrossRef] [PubMed]
- Bustillo, S.; Lucero, H.; Leiva, L.C.; Acosta, O.; Kier Joffé, E.B.; Gorodner, J.O. Cytotoxicity and morphological analysis of cell death induced by Bothrops venoms from the northeast of Argentina. J. Venom. Anim. Toxins incl. Trop. Dis 2009, 15, 28–42. [Google Scholar] [CrossRef]
- Yu, H.; Chen, K.; Wu, J.; Yang, Z.; Shi, L.; Barlow, L.L.; Aronoff, D.M.; Garey, K.W.; Savidge, T.C.; von Rosenvinge, E.C.; et al. Identification of toxemia in patients with Clostridium difficile infection. PLoS ONE 2015, 10, e0124235. [Google Scholar] [CrossRef]
- Gal, Y.; Mazor, O.; Alcalay, R.; Seliger, N.; Aftalion, M.; Sapoznikov, A.; Falach, R.; Kronman, C.; Sabo, T. Antibody/doxycycline combined therapy for pulmonary ricinosis: Attenuation of inflammation improves survival of ricin-intoxicated mice. Toxicol. Rep. 2014, 1, 496–504. [Google Scholar] [CrossRef] [PubMed]
- Sabo, T.; Gal, Y.; Elhanany, E.; Sapoznikov, A.; Falach, R.; Mazor, O.; Kronman, C. Antibody treatment against pulmonary exposure to abrin confers significantly higher levels of protection than treatment against ricin intoxication. Toxicol. Lett. 2015, 237, 72–78. [Google Scholar] [CrossRef] [PubMed]
- Otsu, N. A threshold selection method from gray-level histograms. IEEE Trans. Syst. Man Cybern 1979, 9, 62–66. [Google Scholar] [CrossRef]
- Kittler, J.; Illingworth, J. Minimum error thresholding. Pattern Recognit. 1986, 19, 41–47. [Google Scholar] [CrossRef]

| Time (h) | ||
|---|---|---|
| HeLa | Vero | |
| Phase-shift | 5–7 | 7–8 |
| Confluence | 3–5 | 9 |
| Area | 4–6 | 6–9 |
| Irregularity | 4–7 | 16–18 |
| Perimeter | 4–7 | 6–10 |
| Roughness | 11–12 | 18–20 |
| Optical thickness | 4–7 | 17–20 |
| ECV | 14–15 | 4–7 |
| Time (h) | ||
|---|---|---|
| Adherent HeLa Cells | Preadherent HeLa Cells | |
| Phase-shift | 5–7 | 4–6 |
| Confluence | 3–5 | 4 |
| Area | 4–6 | 1–3 |
| Irregularity | 4–7 | 2–5 |
| Perimeter | 4–7 | 2–3 |
| Roughness | 11–12 | 4–5 |
| Optical thickness | 4–7 | 4–5 |
| ECV | 14–15 | 10–11 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Makdasi, E.; Laskar, O.; Milrot, E.; Schuster, O.; Shmaya, S.; Yitzhaki, S. Whole-Cell Multiparameter Assay for Ricin and Abrin Activity-Based Digital Holographic Microscopy. Toxins 2019, 11, 174. https://doi.org/10.3390/toxins11030174
Makdasi E, Laskar O, Milrot E, Schuster O, Shmaya S, Yitzhaki S. Whole-Cell Multiparameter Assay for Ricin and Abrin Activity-Based Digital Holographic Microscopy. Toxins. 2019; 11(3):174. https://doi.org/10.3390/toxins11030174
Chicago/Turabian StyleMakdasi, Efi, Orly Laskar, Elad Milrot, Ofir Schuster, Shlomo Shmaya, and Shmuel Yitzhaki. 2019. "Whole-Cell Multiparameter Assay for Ricin and Abrin Activity-Based Digital Holographic Microscopy" Toxins 11, no. 3: 174. https://doi.org/10.3390/toxins11030174
APA StyleMakdasi, E., Laskar, O., Milrot, E., Schuster, O., Shmaya, S., & Yitzhaki, S. (2019). Whole-Cell Multiparameter Assay for Ricin and Abrin Activity-Based Digital Holographic Microscopy. Toxins, 11(3), 174. https://doi.org/10.3390/toxins11030174




