Granulovirus GP37 Facilitated ODVs Cross Insect Peritrophic Membranes and Fuse with Epithelia
Abstract
1. Introduction
2. Results
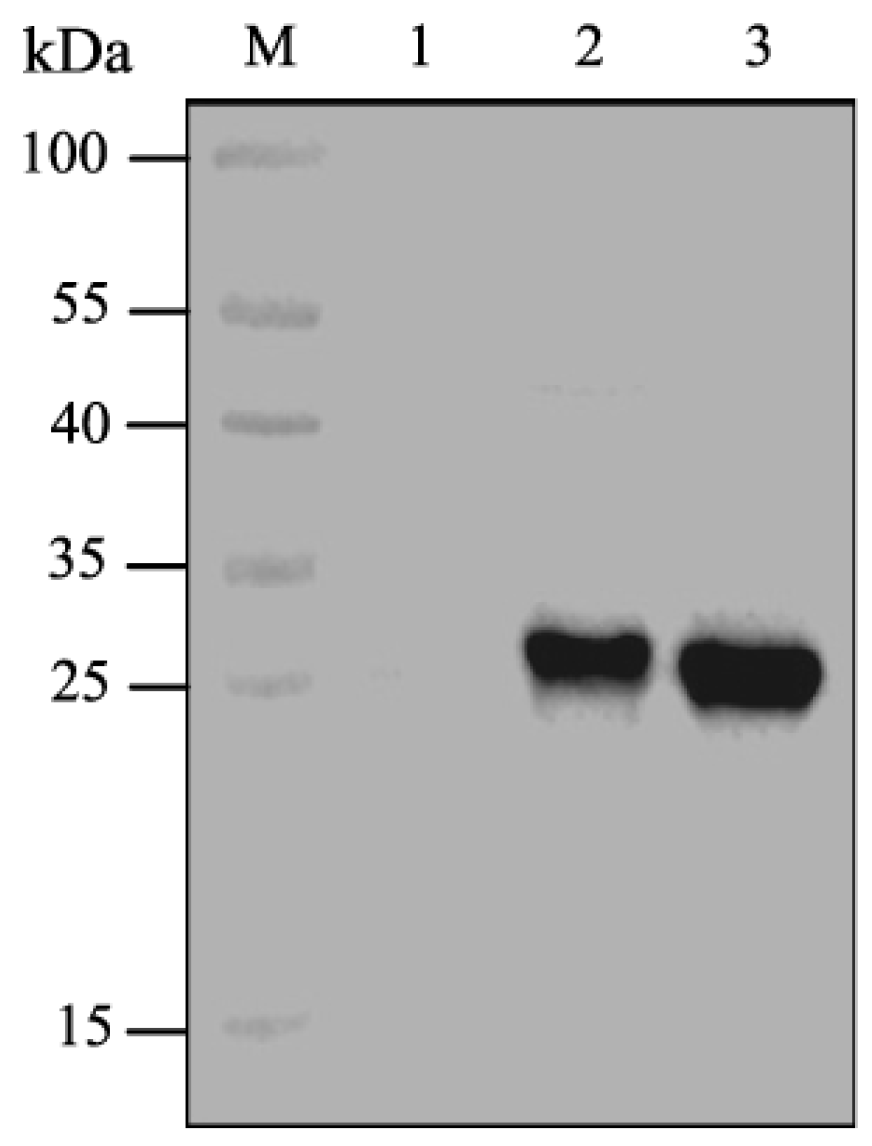
2.1. Expression of CpGV GP37
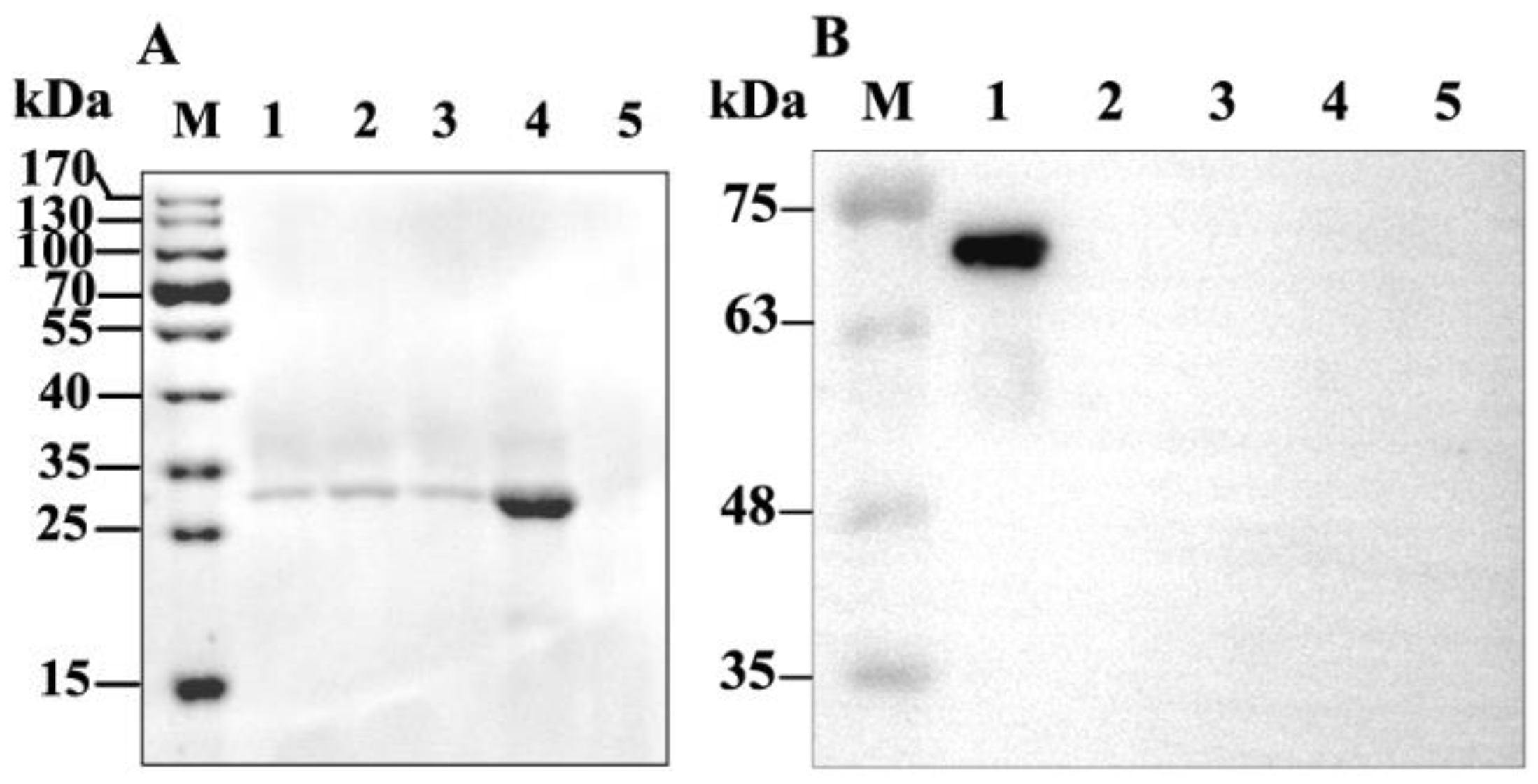
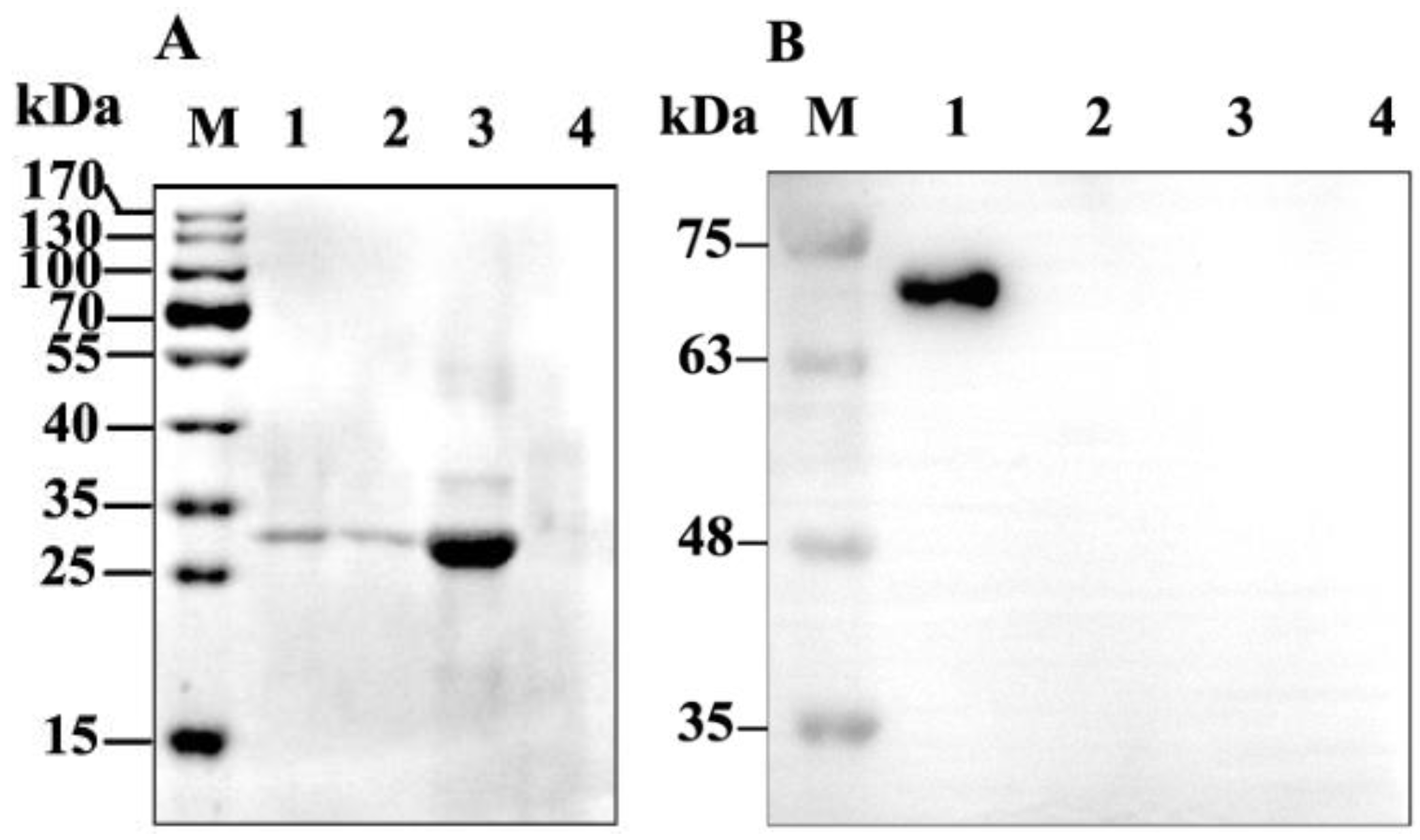
2.2. Binding of GP37 to PM
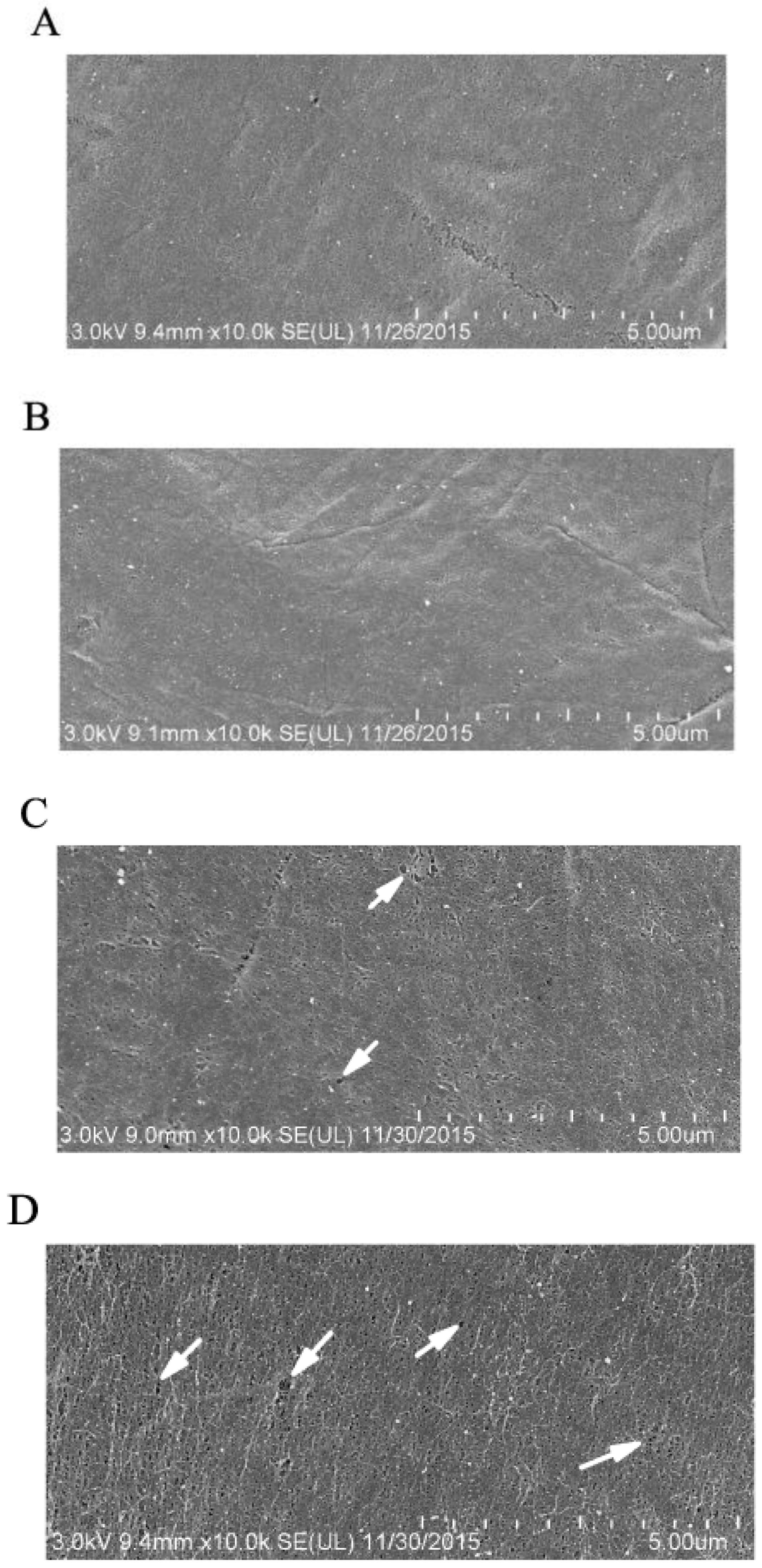
2.3. GP37 Caused Perforations on PMs
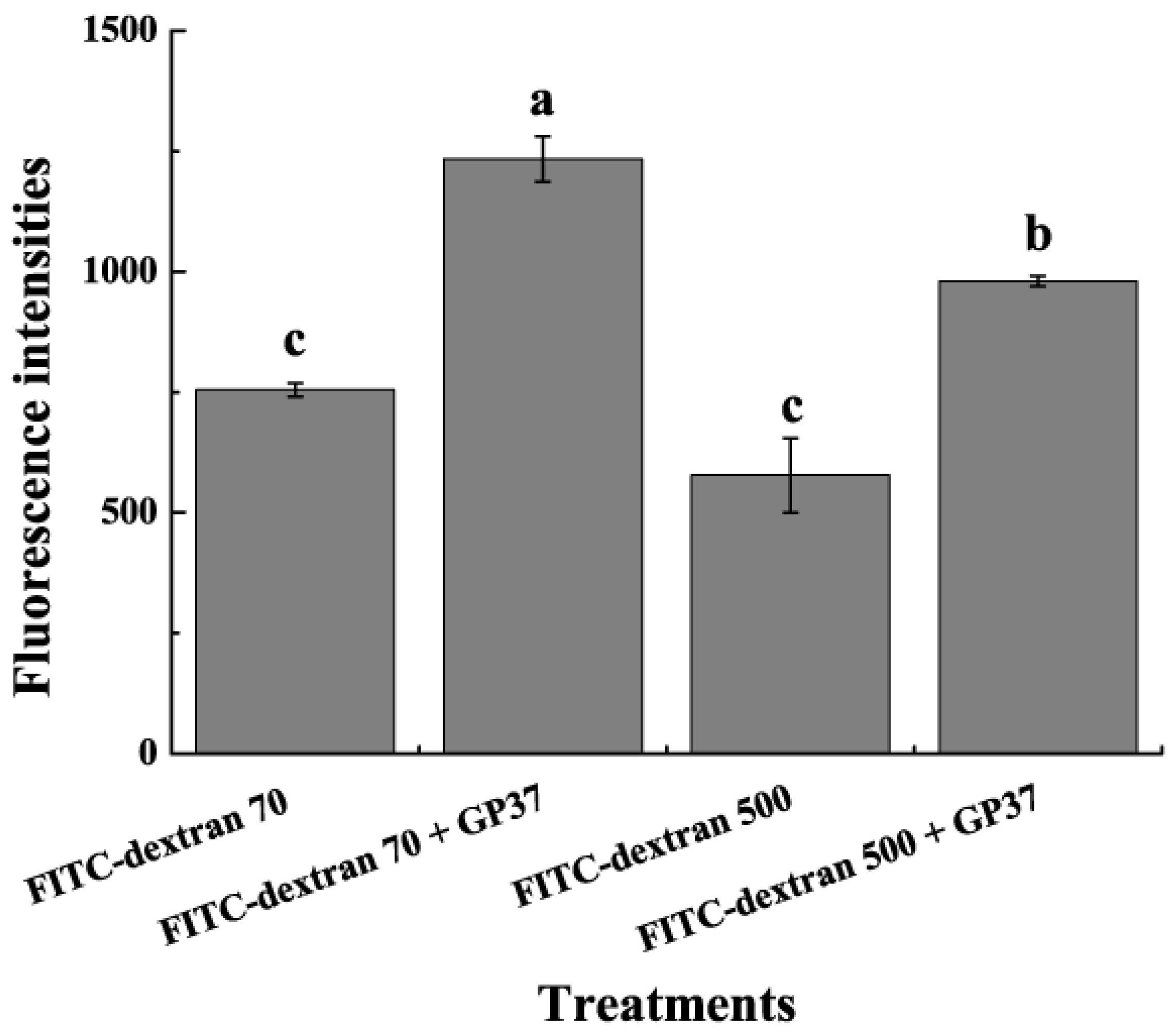
2.4. GP37 Enhanced PM Permeability
2.5. AcMNPV DNA Contents in Midguts and Intestinal Juices
2.6. GP37 Facilitated the Fusion of ODVs and Midgut Epithelia
3. Discussion
4. Materials and Methods
4.1. Insects and Viruses
4.2. Expression of CpGV GP37
4.3. In Vitro PM-Binding Experiment
4.4. In Vivo PM-Binding Experiment
4.5. Scanning Electron Microscope (SEM) Observations of PM Structures
4.6. Determination of PM Permeability
4.7. Real-Time qPCR Qualification of AcMNPV DNA Levels
4.8. Extraction of BBMVs from S. exigua Midguts
4.9. R18 Labeling of AcMNPV ODVs
4.10. Fusion of ODVs and Midgut Epithelia
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rohrmann, G.F. Baculovirus Molecular Biology, 2nd ed.; National Center for Biotechnology Information: Bethesda, MD, USA, 2011. Available online: http://www.ncbi.nlm.nih.gov/books/NBK49500/ (accessed on 5 January 2015).
- Funk, C.J.; Braunagel, S.C.; Rohrmann, G.F. Baculovirus structure. In The Baculoviruses, 1st ed.; Miller, L.K., Ed.; Plenum Press: New York, NY, USA, 1997; pp. 7–32. [Google Scholar]
- Hegedus, D.; Erlandson, M.; Gillott, C.; Toprak, U. New insights into peritrophic matrix synthesis, architecture, and function. Annu. Rev. Entomol. 2009, 54, 285–302. [Google Scholar] [CrossRef] [PubMed]
- Cheng, X.W.; Krell, P.J.; Arif, B.M. P34.8 (GP37) is not essential for baculovirus replication. J. Gen. Virol. 2001, 82, 299–305. [Google Scholar] [CrossRef] [PubMed]
- Luque, T.; Finch, R.; Crook, N.; O’Reilly, D.R.; Winstanley, D. The complete sequence of the Cydia pomonella granulovirus genome. J. Gen. Virol. 2001, 82, 2531–2547. [Google Scholar] [CrossRef] [PubMed]
- Salvador, R.; Ferrelli, M.L.; Berretta, M.F.; Mitsuhashi, W.; Biedma, M.E.; Romanowski, V.; Sciocco-Cap, A. Analysis of EpapGV gp37 gene reveals a close relationship between granulovirus and entomopoxvirus. Virus Genes 2012, 45, 610–613. [Google Scholar] [CrossRef] [PubMed]
- Mitsuhashi, W.; Kawakita, H.; Murakami, R.; Takemoto, Y.; Saiki, T.; Miyamoto, K.; Wada, S. Spindles of an entomopoxvirus facilitate its infection of the host insect by disrupting the peritrophic membrane. J. Virol. 2007, 81, 4235–4243. [Google Scholar] [CrossRef] [PubMed]
- Mitsuhashi, W.; Miyamoto, K. Disintegration of the peritrophic membrane of silkworm larvae due to spindles of an entomopoxvirus. J. Invertebr. Pathol. 2003, 82, 34–40. [Google Scholar] [CrossRef]
- Gross, C.H.; Wolgamot, G.M.; Russell, R.Q.; Pearson, M.N.; Rohrmann, G.F. A 37-kilodalton glycoprotein from a baculovirus of Orgyia pseudotsugata is localized to cytoplasmic inclusion bodies. J. Virol. 1993, 67, 469–475. [Google Scholar] [PubMed]
- Li, X.; Barrett, J.; Pang, A.; Klose, R.J.; Krell, P.J.; Arif, B.M. Characterization of an overexpressed spindle protein during a baculovirus infection. Virology 2000, 268, 56–67. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.F.; Li, C.B.; Yang, K.; Wang, L.H.; Yin, C.; Gong, Y.X.; Pang, Y. Characterization of a chitin-binding protein GP37 of Spodoptera litura multicapsid nucleopolyhedrovirus. Virus Res. 2003, 96, 113–122. [Google Scholar] [CrossRef]
- Mitsuhashi, W. Further evidence that spindles of an entomopoxvirus enhance its infectivity in a host insect. J. Invertebr. Pathol. 2002, 79, 59–61. [Google Scholar] [CrossRef]
- Mitsuhashi, W.; Sato, M.; Hirai, Y. Involvement of spindles of an entomopoxvirus (EPV) in infectivity of the EPVs to their host insect. Arch. Virol. 2000, 145, 1465–1471. [Google Scholar] [CrossRef] [PubMed]
- Chiu, E.; Hijnen, M.; Bunker, R.D.; Boudes, M.; Rajendran, C.; Aizel, K.; Oliéric, V.; Schulze-Briese, C.; Mitsuhashi, W.; Young, V.; et al. Structural basis for the enhancement of virulence by viral spindles and their in vivo crystallization. Proc. Natl. Acad. Sci. USA 2015, 112, 3973–3978. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.Y.; Ma, X.H.; Lei, C.F.; Xiao, Y.Z.; Zhang, Z.X.; Sun, X.L. Synergistic effects of Cydia pomonella granulovirus GP37 on the infectivity of nucleopolyhedroviruses and the lethality of Bacillus thuringiensis. Arch. Virol. 2011, 156, 1707–1715. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.L.; Zhao, L.J.; Ma, R.P.; Fang, W.; Hu, J.; Lei, C.F.; Sun, X.L. Improving baculovirus infectivity by efficiently embedding enhancing factors into occlusion bodies. Appl. Environ. Microb. 2017, 83, e00595-17. [Google Scholar] [CrossRef] [PubMed]
- Barbehenn, R.V.; Martin, M.M. Peritrophic envelope permeability in herbivorous insects. J. Insect Physiol. 1995, 41, 303–311. [Google Scholar] [CrossRef]
- Kuraishi, T.; Binggeli, O.; Opota, O.; Buchon, N.; Lemaitre, B. Genetic evidence for a protective role of the peritrophic matrix against intestinal bacterial infection in Drosophila melanogaster. Proc. Nat. Acad. Sci. USA 2011, 108, 15966–15971. [Google Scholar] [CrossRef] [PubMed]
- Takatsuka, J. Characterization of a nucleopolyhedrovirus of Epinotia granitalis (Lepidoptera: Tortricidae). J. Invertebr. Pathol. 2007, 96, 265–269. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.R.; Qiang, X.; Zhang, B.Q.; Tang, M.J.; Zhang, C.X. Characterization of a baculovirus newly isolated from the tea slug moth, Iragoidae fasciata. J. Microbiol. 2009, 47, 208–213. [Google Scholar] [CrossRef] [PubMed]
- Sciocco-Cap, A.; Parola, A.D.; Goldberg, A.V.; Ghiringhelli, P.D.; Romanowski, V. Characterization of a granulovirus isolated from Epinotia aporema Wals. (Lepidoptera: Tortricidae) larvae. Appl. Environ. Microb. 2001, 67, 3702–3706. [Google Scholar] [CrossRef] [PubMed]
- Horton, H.M.; Burand, J.P. Saturable attachment sites for polyhedron-derived baculovirus on insect cells and evidence for entry via direct membrane fusion. J. Virol. 1993, 67, 1860–1868. [Google Scholar] [PubMed]
- Hukuhara, T.; Wijonarko, A. Enhanced fusion of a nucleopolyhedrovirus with cultured cells by a virus enhancing factor from an entomopoxvirus. J. Invertebr. Pathol. 2001, 77, 62–67. [Google Scholar] [CrossRef] [PubMed]
- Rao, R.; Fiandra, L.; Giordana, B.; de Eguileor, M.; Congiu, T.; Burlini, N.; Arciello, S.; Corrado, G.; Pennacchio, F. AcMNPV ChiA protein disrupts the peritrophic membrane and alters midgut physiology of Bombyx mori larvae. Insect Biochem. Molec. 2004, 34, 1205–1213. [Google Scholar] [CrossRef] [PubMed]
- Zhu, R.; Liu, K.Y.; Peng, J.X.; Yang, H.; Hong, H.Z. Optical brightener M2R destroys the peritrophic membrane of Spodoptera exigua (Lepidoptera: Noctuidae) larvae. Pest. Manag. Sci. 2007, 63, 296–300. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.F.; Fang, J.C.; Liu, B.S.; Wang, J.P.; Zhong, W.F.; Wan, F.H. Enhancement of the biological activity of nucleopolyhedrovirus through disruption of the peritrophic matrix of insect larvae by chlorfluazuron. Pest. Manag. Sci. 2007, 63, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Toprak, U.; Harris, S.; Baldwin, D.; Theilmann, D.; Gillott, C.; Hegedus, D.D.; Erlandson, M.A. Role of enhancin in Mamestra configurata nucleopolyhedrovirus virulence: Selective degradation of host peritrophic matrix proteins. J. Gen. Virol. 2012, 93, 744–753. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Granados, R.R. An intestinal mucin is the target substrate for a baculovirus enhancin. Proc. Nat. Acad. Sci. USA 1997, 94, 6977–6982. [Google Scholar] [CrossRef] [PubMed]
- Hoover, K.; Humphries, M.A.; Gendron, A.R.; Slavicek, J.M. Impact of viral enhancin genes on potency of Lymantria dispar multiple nucleopolyhedrovirus in L. dispar following disruption of the peritrophic matrix. J. Invertebr. Pathol. 2010, 104, 150–152. [Google Scholar] [CrossRef] [PubMed]
- Li, H.R.; Tang, H.L.; Harrison, R.L.; Bonning, B.C. Impact of a basement membrane-degrading protease on dissemination and secondary infection of Autographa californica multiple nucleopolyhedrovirus in Heliothis virescens (Fabricus). J. Gen. Virol. 2007, 88, 1109–1119. [Google Scholar] [CrossRef] [PubMed]
- Shorey, H.H.; Hale, R.L. Mass-Rearing of the larvae of nine noctuid species on a simple artificial medium. J. Econ. Entomol. 1965, 58, 522–524. [Google Scholar] [CrossRef]
- Smith, I.R.L.; Crook, N.E. Physical maps of the genomes of four variants of Artogeia rapae granulosis virus. J. Gen. Virol. 1988, 69, 1741–1747. [Google Scholar] [CrossRef]
- Derksen, A.C.G.; Granados, R.R. Alteration of a lepidopteran peritrophic membrane by baculoviruses and enhancement of viral infectivity. Virology 1988, 167, 242–250. [Google Scholar] [CrossRef]
- Hughes, P.R.; van Beek, N.A.M.; Wood, H.A. A modified droplet feeding method for rapid assay of Bacillus thuringiensis and baculoviruses in noctuid larvae. J. Invertebr. Pathol. 1986, 48, 187–192. [Google Scholar] [CrossRef]
- Wolfersberger, M.; Luethy, P.; Maurer, A.; Parenti, P.; Sacchi, F.V.; Giordana, B.; Hanozet, G.M. Preparation and partial characterization of amino acid transporting brush border membrane vesicles from the larval midgut of the cabbage butterfly (Pieris brassicae). Comp. Biochem. Physiol. 1987, 86, 301–308. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
| Treatments | GP37 Concentrations (µg/mL) | |||||
|---|---|---|---|---|---|---|
| 2 | 6 | 10 | ||||
| Fluorescence Intensity Levels | Significant Differences | Fluorescence Intensity Levels | Significant Differences | Fluorescence Intensity Levels | Significant Differences | |
| BBMV+ODVR+GP37 | 0.0814 ± 0.0056 | a | 0.1123 ± 0.0004 | a | 0.1277 ± 0.0217 | a |
| BBMV+ODVR | 0.0858 ± 0.0061 | a | 0.0777 ± 0.0063 | b | 0.0749 ± 0.0121 | b |
| BBMV | 0.0065 ± 0.0017 | b | 0.0028 ± 0.0008 | c | 0.0036 ± 0.0021 | c |
| Primer | Primer Sequences (5′ → 3′) |
|---|---|
| gp-f1 | GATGGCGATCGCC ATGATGACGATTATGAAAAATCCC SgfI Site underlined |
| gp-f2 | AGCTGCGATCGCC ATGCCGTTGGCGAGACAGCG SgfI Site underlined |
| gp-r | GTGCGTTTAAACTTAGTGATGGTGATGGTGATGCAAATCACTTTTCGTTTGC PmeI Site underlined |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, X.; Fang, W.; Fan, R.; Zhang, L.; Lei, C.; Zhang, J.; Nian, W.; Dou, T.; An, S.; Zhou, L.; et al. Granulovirus GP37 Facilitated ODVs Cross Insect Peritrophic Membranes and Fuse with Epithelia. Toxins 2019, 11, 145. https://doi.org/10.3390/toxins11030145
Liu X, Fang W, Fan R, Zhang L, Lei C, Zhang J, Nian W, Dou T, An S, Zhou L, et al. Granulovirus GP37 Facilitated ODVs Cross Insect Peritrophic Membranes and Fuse with Epithelia. Toxins. 2019; 11(3):145. https://doi.org/10.3390/toxins11030145
Chicago/Turabian StyleLiu, Xiangyang, Wei Fang, Rui Fan, Linna Zhang, Chengfeng Lei, Jingjing Zhang, Wenkai Nian, Tao Dou, Shiheng An, Lin Zhou, and et al. 2019. "Granulovirus GP37 Facilitated ODVs Cross Insect Peritrophic Membranes and Fuse with Epithelia" Toxins 11, no. 3: 145. https://doi.org/10.3390/toxins11030145
APA StyleLiu, X., Fang, W., Fan, R., Zhang, L., Lei, C., Zhang, J., Nian, W., Dou, T., An, S., Zhou, L., & Sun, X. (2019). Granulovirus GP37 Facilitated ODVs Cross Insect Peritrophic Membranes and Fuse with Epithelia. Toxins, 11(3), 145. https://doi.org/10.3390/toxins11030145





