Mycotoxins and Mycotoxin Producing Fungi in Pollen: Review
Abstract
1. Introduction
2. Mycotoxins in Pollen
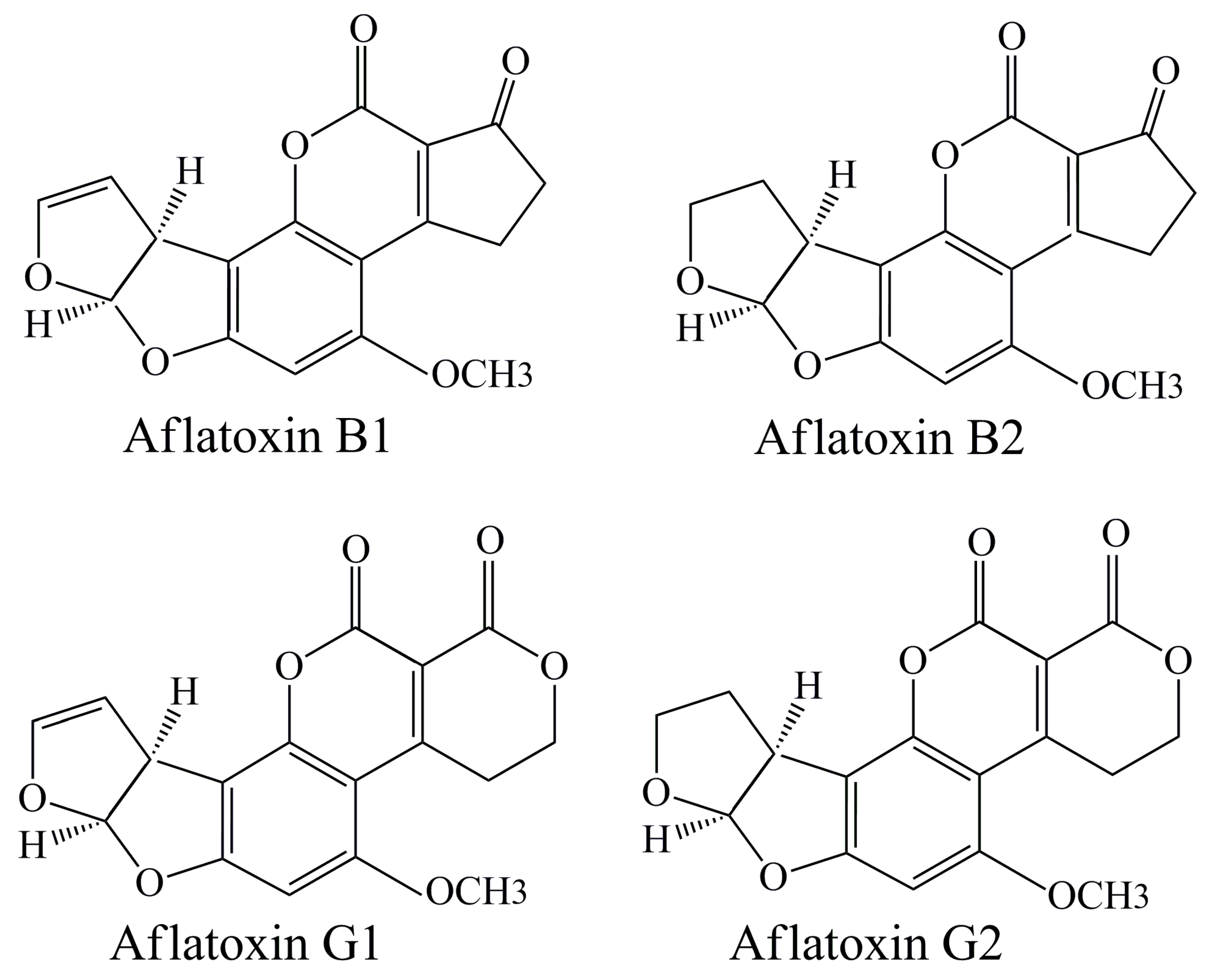
2.1. Aflatoxins
2.1.1. Contamination of Pollen with Aflatoxins—Possible Ways
- -
- There are different ways of pollen contamination with aflatoxin(s).
- -
- These toxins remain in samples with or without presence of appropriate fungi.
- -
- It is extremely important to always perform mycotoxicological analysis together with microbiological characterization of pollen.
2.1.2. Quantification of Aflatoxins in Pollen Samples
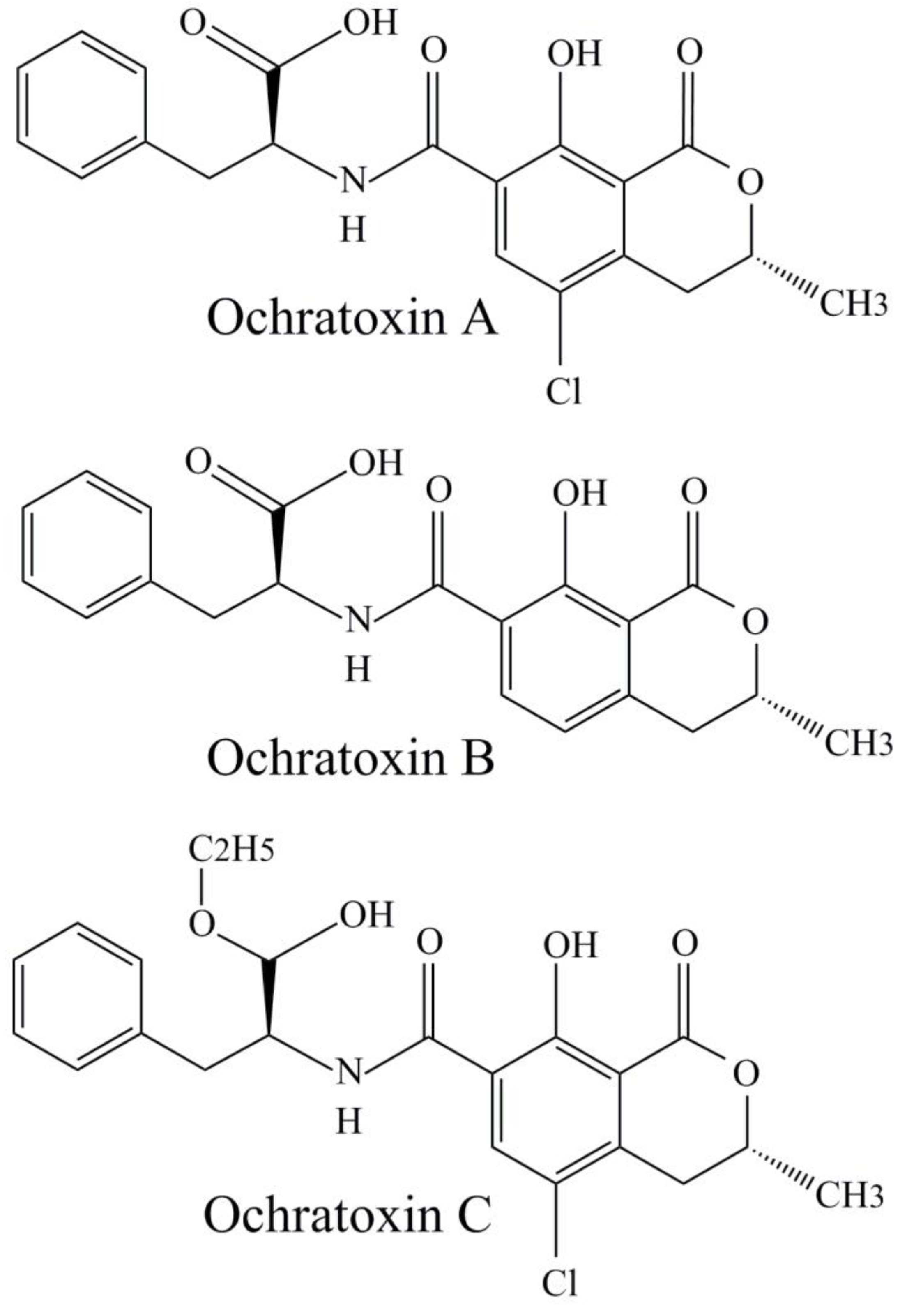
2.2. Ochratoxins
Ochratoxins in Pollen
2.3. The Other Mycotoxins Examined in Pollen
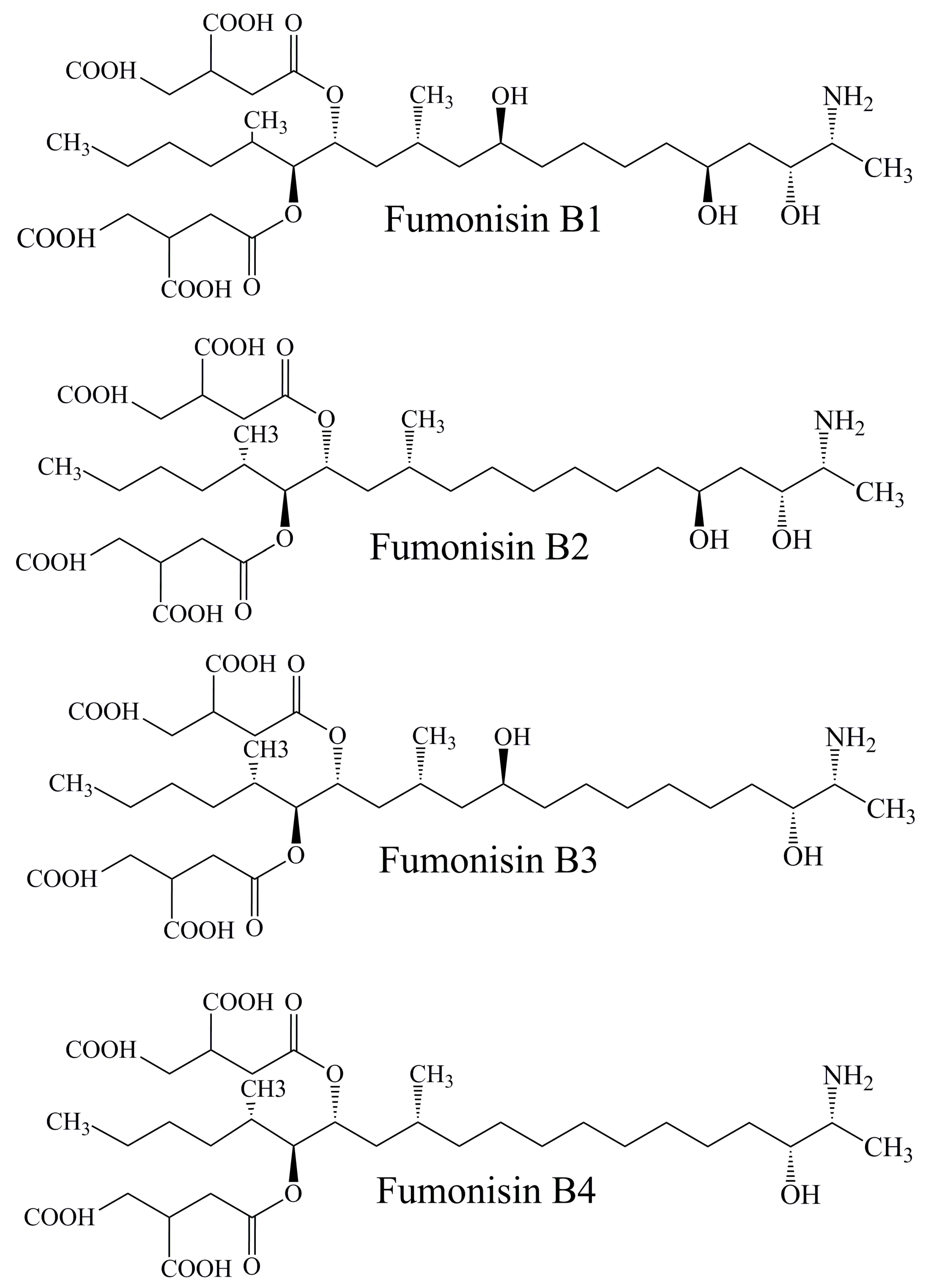
2.3.1. Fumonisins
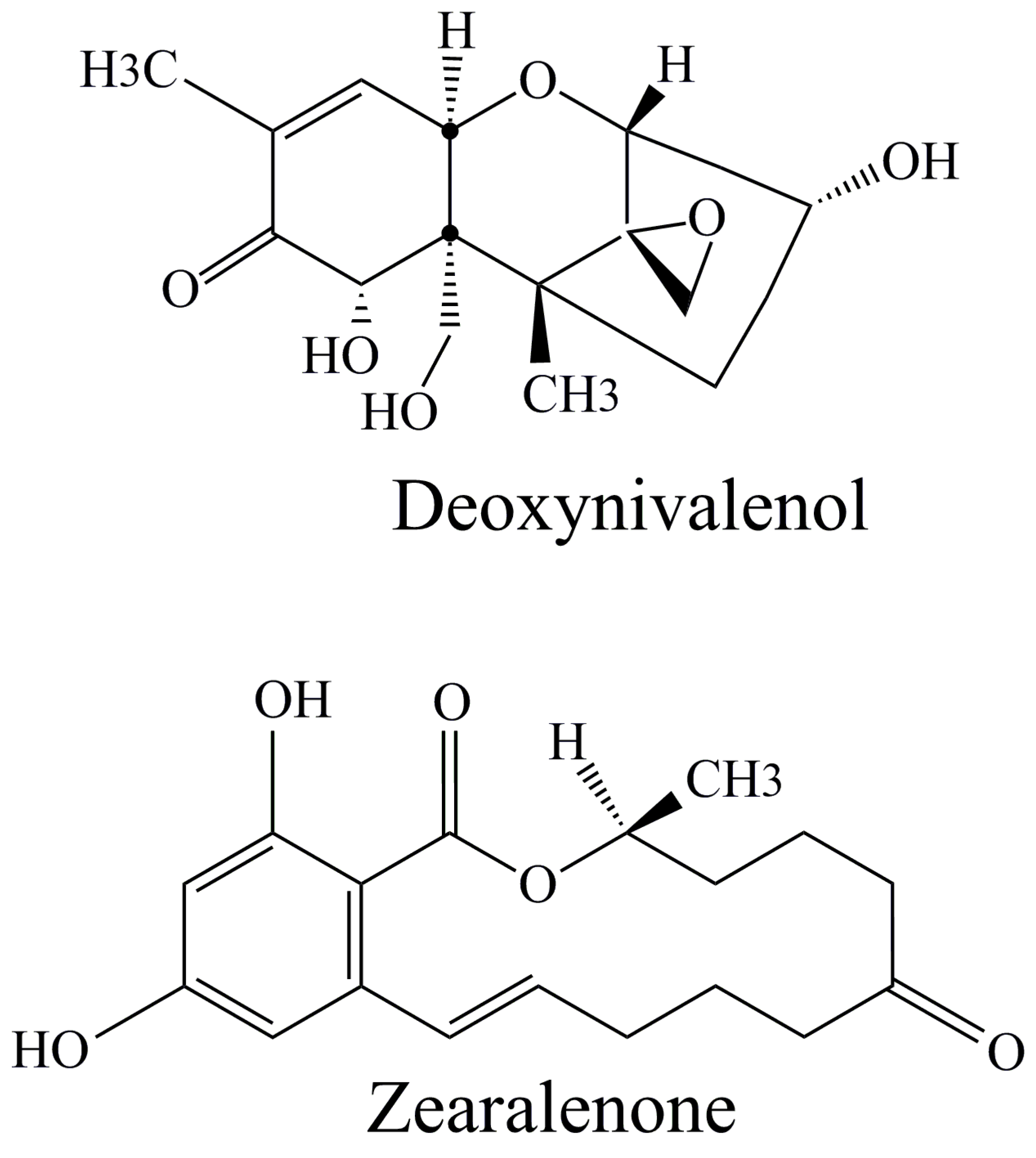
2.3.2. Zearalenone
2.3.3. Trichothecenes Group of Mycotoxins
3. Mycotoxin Producing Fungi in Pollen
4. Legislations of Mycotoxins Level in Food and Pollen
5. Conclusions and Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Borg, M.; Brownfield, L.; Twell, D. Male gametophyte development: A molecular perspective. J. Exp. Bot. 2009, 60, 1465–1478. [Google Scholar] [CrossRef]
- Campos, G.R.M.; Bogdanov, S.; Almeida-Muradian, L.B.; Szczesna, T.; Mancebo, Y.; Frigerio, C.; Ferreira, F. Pollen composition and standardization of analytical methods. J. Apic. Res. 2008, 47, 154–161. [Google Scholar] [CrossRef]
- Bogdanov, S. Pollen: Collection, harvest, composition, quality. In Bee Product Science (The Pollen Book); 2012; Chapter 1; Available online: http://www.bee-hexagon.net/pollen/collection-harvest-composition-quality/ (accessed on 23 January 2019).
- Kostić, A.Ž.; Barać, M.B.; Stanojević, S.P.; Milojković-Opsenica, D.M.; Tešić, Ž.L.; Šikoparija, B.; Radišić, P.; Prentović, M.; Pešić, M.B. Physicochemical properties and techno-functional properties of bee pollen collected in Serbia. LWT Food Sci. Technol. 2015, 62, 301–309. [Google Scholar] [CrossRef]
- Kostić, A.Ž.; Kaluđerović, L.M.; Dojčinović, B.P.; Barać, M.B.; Babić, V.B.; Mačukanović-Jocić, M.P. Preliminary investigation of mineral content of pollen collected from different Serbian maize hybrids—Is there any potential nutritional value? J. Sci. Food Agric. 2017, 97, 2803–2809. [Google Scholar] [CrossRef] [PubMed]
- Kostić, A.Ž.; Pešić, M.B.; Trbović, D.; Petronijević, R.; Dramićanin, A.; Milojković-Opsenica, D.M.; Tešić, Ž.L. Fatty acid’s profile of Serbian bee-collected pollen—Chemotaxonomic and nutritional approach. J. Apic. Res. 2017, 56, 533–542. [Google Scholar] [CrossRef]
- Kostić, A.Ž.; Mačukanović-Jocić, M.P.; Špirović Trifunović, B.D.; Vukašinović, I.Ž.; Pavlović, V.B.; Pešić, M.B. Fatty acids of maize pollen-quantification, nutritional and morphological evaluation. J. Cereal Sci. 2017, 77, 180–185. [Google Scholar] [CrossRef]
- Conte, P.; Del Caro, A.; Balestra, F.; Piga, A.; Fadda, C. Bee pollen as a functional ingredient in gluten-free bread: A physical-chemical, technological and sensory approach. LWT Food Sci. Technol. 2018, 90, 1–7. [Google Scholar] [CrossRef]
- Campos, M.; Markham, M.R.; Mitchell, K.A.; da Cuhna, A.P. An approach to the characterization of bee pollens via their flavonoid/phenolic profiles. Phytochem Anal. 1997, 8, 181–185. [Google Scholar] [CrossRef]
- Serra-Bonvehí, J.; Torrentó, S.M.; Lorente, C.E. Evaluation of polyphenolic and flavonoid compounds in honeybee-collected pollen produced in Spain. J. Agric. Food Chem. 2001, 49, 1848–1853. [Google Scholar] [CrossRef]
- Campos, M.G.; Webby, F.B.; Markham, M.R.; Mitchell, K.A.; da Cuhna, A.P. Age-induced diminution of free radical scavenging capacity in bee pollens and the contribution of constituent flavonoids. J. Agric. Food Chem. 2003, 51, 742–745. [Google Scholar] [CrossRef]
- Di Paola-Naranjo, R.D.; Sánchez, S.J.; Paramás, A.M.G.; Gonzalo, J.C.R. Liquid chromatographic-mass spectrometric analysis of anthocyanin composition of dark blue bee pollen from Echium Plantagineum. J. Chromatogr. A 2004, 1054, 205–210. [Google Scholar] [CrossRef] [PubMed]
- Almaraz Abarca, N.; Campos da Graça, M.; Ávila-Reyes, J.A.; Naranjo-Jiménez, N.; Corral, J.H.; González-Valdez, L.S. Antioxidant activity of polyphenolic extract of monofloral honeybee-collected pollen from mesquite (Prosopis juliflora, Leguminosae). J. Food Compos. Anal. 2007, 20, 119–124. [Google Scholar] [CrossRef]
- Ferreres, F.; Pereira, D.M.; Valentão, P.; Andrade, P.B. First report of noncoloured flavonoids in Echium plantagineum bee pollen: Differentation of ismomers by liquid chromatography/ion trap mass spectometry. Rapid Commun. Mass Spectrom. 2010, 24, 801–806. [Google Scholar] [CrossRef]
- Ares, A.M.; Valverde, S.; Bernal, J.L.; Nozal, M.J.; Bernal, J. Extraction and determination of bioactive compounds from bee pollen. J. Pharm. Biomed. Anal. 2018, 147, 110–124. [Google Scholar] [CrossRef] [PubMed]
- De-Melo, A.A.M.; Estevinho, L.M.; Moreira, M.M.; Delerue-Matos, C.; da Silva de Freitas, A.; Barth, O.M.; de Almeida-Muradian, L.B. Phenolic profile by HPLC-MS, biological potential, and nutritonal value of a promising food: Monofloral bee pollen. J. Food Biochem. 2018, 42, e12536. [Google Scholar] [CrossRef]
- Almeida-Muradian, L.B.; Pamplona, L.C.; Coimbra, S.; Ortrud, M.B. Chemical composition and botanical evaluation of dried bee pollen pellets. J. Food Compos. Anal. 2005, 18, 105–111. [Google Scholar] [CrossRef]
- Mărgăoan, R.; Mărghitaş, L.A.; Dezmirean, D.S.; Dulf, F.V.; Bunea, A.; Socaci, S.A.; Bobiş, O. Predominant and secondary pollen botanical origins influence the carotenoid and fatty acid profile in fresh honeybee-collected pollen. J. Agric. Food Chem. 2014, 62, 6306–6316. [Google Scholar] [CrossRef]
- Krystyjan, M.; Gumul, D.; Ziobro, R.; Korus, A. The fortification of biscuits with bee pollen and its effect on physicochemical and antioxidant properties in biscuits. LWT Food Sci. Technol. 2015, 63, 640–646. [Google Scholar] [CrossRef]
- De Florio Almeida, J.; Soares dos Reis, A.; Serafini Heldt, L.F.; Pereira, D.; Bianchin, M.; de Moura, C.; Plata-Oviedo, M.V.; Haminiuk, C.W.I.; Ribeiro, I.S.; Fernades Pinto da Luz, C.; et al. Lyophilized bee pollen extract: A natural antioxidant source to prevent lipid oxidation in refrigerated sausages. LWT Food Sci. Technol. 2017, 76, 299–305. [Google Scholar] [CrossRef]
- Kostić, A.Ž.; Pešić, M.B.; Mosić, M.D.; Dojčinović, B.P.; Natić, M.N.; Trifković, J.Đ. Mineral content of some bee-collected pollen from Serbia. Arch. Ind. Hyg. Toxicol. 2015, 66, 251–258. [Google Scholar] [CrossRef]
- Sattler, J.A.G.; de Melo Machado, A.A.; do Nascimento, K.S.; de Melo Pereira, I.L.; Mancini-Filho, J.; Sattler, A.; de Almeida-Muradian, L.B. Essential minerals and inorganic contaminants (barium, cadmium, lithium, lead and vanadium) in dried bee pollen produced in Rio Grande do Sul State, Brazil. Food Sci. Technol. 2016, 36, 505–509. [Google Scholar] [CrossRef]
- Altunaltmaz, S.S.; Tarhan, D.; Aksu, F.; Barutçu, U.B.; Or, M.E. Mineral element and heavy metal (cadmium, lead and arsenic) levels of bee pollen in Turkey. Food Sci. Technol. 2017, 37 (Suppl. S1), 136–141. [Google Scholar] [CrossRef]
- Bennet, J.W.; Klich, M. Mycotoxins. Clin. Microbiol. Rev. 2003, 16, 497–516. [Google Scholar] [CrossRef]
- Hanssen, E.; Jung, M. Control of aflatoxins in the food industry. Pure Appl. Chem. 1973, 35, 239–250. [Google Scholar] [CrossRef]
- Bosco, F.; Mollea, C. Mycotoxins in food. In Food Industrial Processes—Methods and Equipment; Valdez, B., Ed.; Intech Open Limited: London, UK, 2012; Chapter 10; pp. 169–200. ISBN 978-953-307-905-9. [Google Scholar]
- Stanković, S.; Lević, J.; Ivanović, D.; Krnjaja, V.; Stanković, G.; Tančić, S. Fumonisin B1 and its co-occurrence with other fusariotoxins in naturally-contaminated wheat grain. Food Control 2012, 23, 384–388. [Google Scholar] [CrossRef]
- Krnjaja, V.; Mandić, V.; Lević, J.; Stanković, S.; Petrović, T.; Vasić, T.; Obradović, A. Influence of N-fertilization on Fusarium head blight and mycotoxin levels in winter wheat. Crop Prot. 2015, 67, 251–256. [Google Scholar] [CrossRef]
- Abrunhosa, L.; Morales, H.; Soares, C.; Calado, T.; Vila-Cha, A.S.; Pereira, M.; Venâncio, A. A review of mycotoxins in food and feed products in Portugal and estimation of probable daily intake. Crit. Rev. Food Sci. Nutr. 2016, 56, 249–265. [Google Scholar] [CrossRef]
- Bijelić, Z.; Krnjaja, V.; Stanković, S.; Muslić-Ružić, D.; Mandić, V.; Škrbić, Z.; Lukić, M. Occurrence of moulds and mycotoxins in grass-legume silages influenced by nitrogen fertilization and phenological phase at harvest. Rom. Biotech. Lett. 2017, 22, 12907–12914. [Google Scholar]
- Krnjaja, V.; Stanković, S.; Obradović, A.; Petrović, T.; Mandić, V.; Bijelić, Z.; Božić, M. Trichothecene genotypes of Fusarium graminearum populations isolated from winter wheat crops in Serbia. Toxins 2018, 10, 460. [Google Scholar] [CrossRef]
- Smith, M.-C.; Madec, S.; Coton, E.; Hymery, N. Natural co-occurrence of mycotoxins in foods and feeds and their in vitro combined toxicological effects. Toxins 2016, 8, 94. [Google Scholar] [CrossRef] [PubMed]
- EC Commission. Setting of maximum levels for certain contaminants in foodstuffs—Regulation No. 1881/2006. Official J. of the EU. 2006, L364, 5–24. [Google Scholar]
- Van Egmond, H.P. Mycotoxins: Risks, regulations and European co-operation. J. Nat. Sci. Matica Srpska Novi Sad. 2013, 125, 7–20. [Google Scholar] [CrossRef]
- Serra-Bonvehi, J.; Escolà Jordà, R. Nutrient composition and microbiological quality of honey bee-collected pollen in Spain. J. Agric. Food Chem. 1997, 45, 725–732. [Google Scholar] [CrossRef]
- Medina, Á.; González, G.; Sáez, J.M.; Mateo, R.; Jiménez, M. Bee pollen, a substrate that stimulates ochratoxin A production by Aspergillus ochraceus Wilh. Syst. Appl. Microbiol. 2004, 27, 261–267. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Villanova, R.J.; Cordón, C.; González-Paramás, A.M.; Aparicio, P.; Garcia Rosales, M.E. Simultaneous immunoaffinity column cleanup and hplc analysis of aflatoxins and ochratoxin a in spanish bee pollen. J. Agric. Food Chem. 2004, 52, 7235–7239. [Google Scholar] [CrossRef] [PubMed]
- González, G.; Hinojo, M.J.; Mateo, R.; Medina, A.; Jiménez, M. Occurrence of mycotoxin producing fungi in bee pollen. Int. J. Food Microbiol. 2005, 105, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Zaijun, L.; Zhongyun, W.; Xiulan, S.; Yinjun, F.; Peipei, C. A sensitive and highly stable electrochemical impendace immunosensor based on the formation of silica gel-ionic liquid biocompatible film on the glassy carbon electrode for the determination of aflatoxin B1 in bee pollen. Talanta 2010, 80, 1632–1637. [Google Scholar] [CrossRef]
- Pitta, M.; Markaki, P. Study of aflatoxin B1 production by Aspergillus parasiticus in bee pollen of Greek origin. Mycotoxin Res. 2010, 26, 229–234. [Google Scholar] [CrossRef]
- Kačaniová, M.; Juráček, M.; Chlebo, R.; Kňazovická, V.; Kadasi-Horáková, M.; Kunová, S.; Lejková, J.; Haščik, P.; Mareček, J.; Šimko, M. Mycobiota and mycotoxins in bee pollen collected from different areas of Slovakia. J. Environ. Sci. Health Part B 2011, 46, 623–629. [Google Scholar] [CrossRef]
- Vidal, J.C.; Bonel, L.; Ezquerra, A.; Hernández, S.; Bertolín, J.R.; Cubel, C.; Castillo, J.R. Electrochemical affinity biosensors for detection of mycotoxins: A review. Biosens. Bioelectron. 2013, 49, 146–158. [Google Scholar] [CrossRef]
- Rodríguez-Carasco, Y.; Font, G.; Mañes, J.; Berrada, H. Determination of mycotoxins in bee pollen by gas chromatography−tandem mass spectrometry. J. Agric. Food Chem. 2013, 61, 1999–2005. [Google Scholar] [CrossRef] [PubMed]
- Petrović, T.; Nedić, N.; Paunović, D.; Rajić, J.; Matović, K.; Radulović, Z.; Krnjaja, V. Natural mycobiota and aflatoxin B1 presence in bee pollen collected in Serbia. Biotechnol. Anim. Husb. 2014, 30, 731–741. [Google Scholar] [CrossRef]
- Xue, X.; Selvaraj, J.N.; Zhao, L.; Dong, H.; Liu, F.; Liu, Y.; Li, Y. Simultaneous determination of aflatoxins and ochratoxin a in bee pollen by low-temperature fat precipitation and immunoaffinity column cleanup coupled with LC-MS/MS. Food Anal. Methods 2014, 7, 690–696. [Google Scholar] [CrossRef]
- Cirigliano, A.M.; Rodríguez, M.A.; Godeas, A.M.; Cabrera, G.M. Mycotoxins from beehive pollen mycoflora. J. Sci. Res. Rep. 2014, 3, 966–972. [Google Scholar] [CrossRef]
- Valadares Deveza, M.; Keller, K.M.; Affonso Lorenzon, M.C.; Teixeira Nunes, L.M.; Oliveira Sales, E.; Barth, O.M. Mycotoxicological and palynological profiles of commercial brands of dried bee pollen. Braz. J. Microbiol. 2015, 46, 1171–1176. [Google Scholar] [CrossRef]
- Kostić, A.Ž.; Petrović, T.S.; Krnjaja, V.S.; Nedić, N.M.; Tešić, Ž.L.; Milojković-Opsenica, D.M.; Barać, M.B.; Stanojević, S.P.; Pešić, M.B. Mold/aflatoxin contamination of honey bee collected pollen from different Serbian regions. J. Apic. Res. 2017, 56, 13–20. [Google Scholar] [CrossRef]
- Hosny, A.S.; Sabbah, F.M.; El-Bazza, Z.E. Studies on microbial decontamination of Egyptian bee pollen by γ-irradiation. Egypt Pharm. J. 2018, 17, 190–200. [Google Scholar] [CrossRef]
- Estevinho, L.M.; Dias, T.; Anjos, O. Influence of the storage conditions (frozen vs dried) in health-related lipid indexes and antioxidants of bee pollen. Eur. J. Lipid Sci. Technol. 2018, 2018, 1800393. [Google Scholar] [CrossRef]
- Vidal, A.; Mengelers, M.; Yang, S.; De Saeger, S.; De Boevre, M. Mycotoxin biomarkers of exposure: A comprehensive review. Compr. Rev. Food Sci. Food Saf. 2018, 17, 1127–1155. [Google Scholar] [CrossRef]
- Neal, G.E. Genetic implications in the metabolism and toxicity of mycotoxins. Toxicol. Lett. 1995, 82/83, 861–867. [Google Scholar] [CrossRef]
- Magan, N.; Lacey, J. Effect of temperature and pH on water realtions of field and storage fungi. Trans. Br. Mycol. Soc. 1984, 82, 71–81. [Google Scholar] [CrossRef]
- Estevinho, L.M.; Rodrigues, S.; Pereira, A.P.; Feás, X. Portugese bee pollen: Palynological study, nutritional and microbiological evaluation. Int. J. Food Sci. Technol. 2012, 47, 429–435. [Google Scholar] [CrossRef]
- Niu, G.; Johnson, R.M.; Berenbaum, M.R. Toxicity of mycotoxins to honeybees and its amelioration by propolis. Apidologie 2011, 42, 79–87. [Google Scholar] [CrossRef]
- Temiz, A.; Şener Mumcu, A.; Özkök Tüylü, A.; Sorkun, K.; Salih, B. Antifungal activity of propolis samples collected from different geographical regions of Turkey against two food-related molds, Aspergillus versicolor and Penicillium aurantiogriseum. Gida 2013, 38, 135–142. [Google Scholar] [CrossRef]
- Tao, Y.; Xie, S.; Xu, F.; Liu, A.; Wang, Y.; Chen, D.; Pan, Y.; Huang, L.; Peng, D.; Wang, X.; et al. Ochratoxin A: Toxicity, oxidative stress and metabolism (Review). Food Chem. Toxicol. 2018, 112, 320–331. [Google Scholar] [CrossRef] [PubMed]
- Cendoya, E.; Chiotta, M.L.; Zachetti, V.; Chulze, S.N.; Ramirez, M.L. Fumonisins and fumonisin-producing Fusarium occurrence in wheat and wheat by products: A review. J. Cereal Sci. 2018, 80, 158–166. [Google Scholar] [CrossRef]
- Gilliam, M. Microbiology of pollen and bee bread: The yeasts. Apidologie 1979, 10, 43–53. [Google Scholar] [CrossRef]
- Gilliam, M. Microbiology of pollen and bee bread: The genus Bacillus. Apidologie 1979, 10, 269–274. [Google Scholar] [CrossRef]
- Gilliam, M.; Prest, D.B.; Lorenz, B.J. Microbiology of pollen and bee bread: Taxonomy and enzimology of molds. Apidologie 1989, 20, 53–68. [Google Scholar] [CrossRef]
- Carelli Barreto, L.M.R.; Cunha Funari, S.R.; de Oliveira Rosi, R. Composição e qualidade do pólen apícola proveniente de sete estados Brasileiros e do distrito federal. Bol. Ind. Anim. 2005, 62, 167–175. [Google Scholar]
- Kačániová, M.; Pavličová, S.; Haščík, P.; Kociubinski, G.; Kńazovická, V.; Sudzina, M.; Sudzinova, J.; Fikselová, M. Microbial communities in bees, pollen and honey from Slovakia. Acta Microbiol. Immunol. Hung. 2009, 56, 285–295. [Google Scholar] [CrossRef] [PubMed]
- Bucio Villalobos, C.M.; López Preciado, G.; Martínez Jaime, O.A.; Torres Morales, J.J. Micoflora asociada a granos de polen recolectados por abejas domésticas (Apis mellifera L.). Rev. Electron. Nova Sci. 2010, 4, 93–103. [Google Scholar] [CrossRef]
- Brindza, J.; Gróf, J.; Bacigálová, K.; Ferianc, P.; Tóth, D. Pollen microbial colonization and food safety. Acta Chim. Slov. 2010, 3, 95–102. [Google Scholar]
- Puig-Peña, Y.; del-Risco-Ríos, C.A.; Álvarez-Rivera, V.P.; Leiva-Castillo, V.; García-Neninger, R. Comparación de la calidad microbiológica del polen apícola fresco y después de un proceso de secado. Rev. CENIC. Cienc. Biol. 2012, 43, 23–27. [Google Scholar]
- Nogueira, C.; Iglesias, A.; Feás, X.; Estevinho, M.L. Commercial bee pollen with different geographical origins: A comprehensive approach. Int. J. Mol. Sci. 2012, 13, 11173–11187. [Google Scholar] [CrossRef] [PubMed]
- Feás, X.; Pilar Vázquez-Tato, M.; Estevinho, L.; Seijas, J.A.; Iglesias, A. Organic bee pollen: Botanical origin, nutritional value, bioactive compounds, antioxidant activity and microbiological quality. Molecules 2012, 17, 8359–8377. [Google Scholar] [CrossRef] [PubMed]
- Hani, B.; Dalila, B.; Saliha, D.; Daoud, H.; Mouloud, G.; Seddik, K. Microbiological sanitary aspects of pollen. Adv. Environ. Biol. 2012, 6, 1415–1420. [Google Scholar]
- De-Melo Machado, A.A.; Estevinho, M.L.M.F.; Almeida-Muradian, L.B. A diagnosis of the microbiological quality of dehydrated bee-pollen produced in Brazil. Lett. Appl. Microbiol. 2015, 61, 477–483. [Google Scholar] [CrossRef]
- Santa Bárbara, M.; Machado, C.S.; da Silva Sodré, G.; Dias, L.G.; Estevinho, L.M.; Lopes de Carvalho, C.A. Microbiological assessment, nutritional characterization and phenolic compounds of bee pollen from Mellipona mandacaia Smith, 1983. Molecules 2015, 20, 12525–12544. [Google Scholar] [CrossRef]
- Nardoni, S.; D’Ascenzi, C.; Rocchigiani, G.; Moretti, V.; Mancianti, F. Occurrence of molds from bee pollen in Central Italy—A preliminary study. Ann. Agric. Environ. Med. 2016, 23, 103–105. [Google Scholar] [CrossRef]
- De-Melo Machado, A.A.; Fernandes Estevinho, M.L.M.; Gasparotto Sattler, J.A.; Rodrigues Souza, B.; da Silva Freitas, A.; Barth, O.M.; Bicudo Almeida-Muradian, L. Effect of processing conditions on characteristics of dehydrated bee-pollen and correlation between quality parameters. LWT Food Sci. Technol. 2016, 65, 808–815. [Google Scholar] [CrossRef]
- Grabowski, N.T.; Klein, G. Microbiology of processed edible insect products—Results of a preliminary survey. Int. J. Food Microbiol. 2017, 243, 103–107. [Google Scholar] [CrossRef] [PubMed]
- Libonatti, C.; Andersen-Puchuri, L.; Tabera, A.; Varela, S.; Passucci, J.; Basualdo, M. Caracterización microbiológica de polen comercial. Reporte preliminar. Rev. Electron. Vet. 2017, 18, 1–5. [Google Scholar]
- Aparecida Soares de Arruda, V.; Vieria dos Santos, A.; Figueiredo Sampaio, D.; da Silva Araújo, E.; de Castro Peixoto, A.L.; Fernandes Estevinho, L.M.; de Almeida-Muradian, B.L. Microbiological quality and physicochemical characterization of Brazilian bee pollen. J. Apic. Res. 2017, 56, 231–238. [Google Scholar] [CrossRef]
- Adjlane, N.; Hadj Ali, L.M.; Benamara, M.; Bounadi, O.; Haddad, N. Qualite microbiologique du pollen produit par les apiculteurs et commercialise en Algerie. Rev. Microbiol. Ind. San et Environ. 2017, 11, 31–39. [Google Scholar]
- Beev, G.; Stratev, D.; Vashin, I.; Pavlov, D.; Dinkov, D. Quality assessment of bee pollen: A cross sectional survey in Bulgaria. J. Food Qual. Hazards Control 2018, 5, 11–16. [Google Scholar] [CrossRef]
- Figueredo Santa Bárbara, M.; Santiago Machado, C.; da Silva Sodré, G.; de Lima Silva, F.; Alfredo Lopes de Carvalho, C. Microbiological and physicochemical characterization of the pollen stored by stingless bees. Braz. J. Food Technol. 2018, 21, e2017180. [Google Scholar] [CrossRef]
- Zuluaga-Domínguez, C.; Serrato-Bermudez, J.; Quicazán, M. Influence of drying-related operations on microbiological, structural and physicochemical aspects for processing of bee-pollen. Eng. Agric. Environ. Food. 2018, 11, 57–64. [Google Scholar] [CrossRef]
- Arroyo-Manzanares, N.; Huertas-Pérez, J.F.; García-Campaña, A.M.; Gámiz-Gracia, L. Mycotoxin analysis: New proposals for sample treatment. Adv. Chem. 2014, 2014, 547506. [Google Scholar] [CrossRef]
- European Food Safety Authority (EFSA). Opinion of the Scientific Panel on contaminants in the food chain of the EFSA on a request from the Commission related to ochratoxin A in food. EFSA J. 2006, 4, 365. [Google Scholar] [CrossRef]
- EPSA Panel on Contaminants in the Food Chain (Contam); Knutsen, H.K.; Barregard, L.J.A.; Bingami, M.; Bruschweiler, B.; Ceccatelli, S.; Cottrill, B.; Dinovi, M.; Edler, L.; Grasl-Kraup, B.; et al. Effect on public health of a possible increase of the maximum level for ‘aflatoxin total’ from 4 to 10 μg/kg in peanuts and processed products thereof, intended for direct human consumption or use as an ingredient in foodstuffs-statement. EFSA J. 2018, 16, 5175. [Google Scholar] [CrossRef]
- EPSA Panel on Contaminants in the Food Chain (Contam). Scientific Opinion on the risks for public health related to the presence of zearalenone in food. EFSA J. 2011, 9, 2197. [Google Scholar] [CrossRef]
- EPSA Panel on Contaminants in the Food Chain (Contam); Knutsen, H.K.; Alexander, J.; Barregard, L.J.A.; Bingami, M.; Bruschweiler, B.; Ceccatelli, S.; Cottrill, B.; Dinovi, M.; Grasl-Kraup, B.; et al. Scientific Opinion on the risks for human and animal health related to the presence of modified forms of certain mycotoxins in food and feed. EFSA J. 2014, 12, 3916. [Google Scholar] [CrossRef]
- EPSA Panel on Contaminants in the Food Chain (Contam). Risks to human and animal health related to the presence of deoxynivalenol and its acetylated and modfied forms in food and feed. EFSA J. 2017, 15, 4718. [Google Scholar] [CrossRef]
- EPSA Panel on Contaminants in the Food Chain (Contam). Scientific Opinion on the risks for animal and public health related to the presence of T-2 and HT-2 toxin in food and feed. EFSA J. 2011, 9, 2481. [Google Scholar] [CrossRef]
- Šegvić Klarić, M. Adverse effects of combined mycotoxins. Arh. Ind. Hyg. Toxikol. 2012, 63, 519–530. [Google Scholar] [CrossRef]
- Šegvić Klarić, M.; Rašić, D.; Peraica, M. Deleterious effects of mycotoxin combinations involving Ochratoxin, A. Toxins 2013, 5, 1965–1987. [Google Scholar] [CrossRef] [PubMed]
- Manafi, M.; Umakantha, B.; Mohan, K.; Narayana Swamy, H.D. Synergistic effects of two commonly contaminating mycotoxins (Aflatoxin and T-2 toxin) on biochemical parameters and immune status of broiler chickens. World Appl. Sci. J. 2012, 17, 364–367. [Google Scholar]
| No. of Examined Pollen Samples | Geographical Origin | Analytical Methods | Isolated Mycotoxins Producing Fungi Species | AF Types and Concentration Range(s) | Reference |
|---|---|---|---|---|---|
| 20 | Spain | ELISA test | / | Total AFs: below 5 μg/kg | [35] |
| 20 | Spain | HPLC (with fluorescent detection) | / | AFB1 and AFB2: below limit detection (BLD) | [37] |
| 87 + 3 | Spain + Argentina | HPLC (with fluorescent detection) | A. flavus A. parasiticus | AFB1, AFB2, AFG1 and AFG2: not determined. | [38] |
| 5 | China | Cyclic voltametry | / | AFB1: 0.00–0.52 μg/kg | [39,42] |
| 1 | Epirus (Western Greece) | HPLC (with fluorescent detection) | not detected | AFB1: not detected | [40] |
| 45 | Slovakia | ELISA test | A. flavus, A. parasiticus. | Total AFs: 13.60–16.20 μg/kg (in poppy pollen) 3.15–5.40 μg/kg (in rape pollen) 1.20–3.40 μg/kg (in sunflower pollen) | [41] |
| 33 | Serbia | ELISA test | A. flavus | AFB1: 3.49–14.02 μg/kg | [44] |
| 20 | China | LC-MS/MS | / | AFB1, AFB2, AFG1 and AFG2: below limit detection (BLD) | [45] |
| 27 | Brazil | Qualitative analysis | A. flavus | AFB1 and AFB2: not determined | [47] |
| 26 | Serbia | ELISA test | A. flavus | AFB1: 3.15–17.32 μg/kg | [48] |
| 30 | Egypt | Thin-layer chromatography | A. flavus | AFB1 AFB2, AFG1 and AFG2 were not determined. | [49] |
| 9 | Portugal | ELISA test | Not detected | Not detected AFB1 | [50] |
| No. of Contaminated/Examined Pollen Samples | Geographical Origin | Analytical Methods | Isolated Mycotoxin Producing Fungi Specie(s) | Mycotoxin Types and Concentration Range(s) | Reference |
|---|---|---|---|---|---|
| 15/45 were contaminated | Slovakia | ELISA test | F. proliferatum, A. alternata Keissl. | Total FBs: 6.30–12.60 μg/kg | [41] |
| 45 | Slovakia | ELISA test | F. graminearum | ZEN: 311.00–361.30 μg/kg (in poppy pollen) 137.10–181.60 μg/kg (in rape pollen) 115.60–147.40 μg/kg (in sunflower pollen) | [41] |
| 45 | Slovakia | ELISA test | F. graminearum, F. oxysporum, F. proliferatum, F. sporotrichioides, F. verticillioides | T-2 toxin: 113.90–299.60 μg/kg (in poppy pollen) 197.10-265.70 μg/kg (in rape pollen) 173.60–364.90 μg/kg (in sunflower pollen) | [41] |
| 45 | Slovakia | ELISA test | F. graminearum, F. oxysporum, F. proliferatum, F. sporotrichioides, F. verticillioides | DON: 183.10–273.90 μg/kg (in poppy pollen) 189.60–244.70 μg/kg (in rape pollen) 133.30–203.50 μg/kg (in sunflower pollen) | [41] |
| 2/15 | Spain | GC/MS | / | neosolaniol: 22 i.e., 30 μg/kg nivalenol: 1 μg/kg | [43] |
| No. of Examined Pollen Samples | Geographical Origin | Detected Microbial Class | Microbial Species or/and Total Microbial | Microbial Count | Observations | Reference |
|---|---|---|---|---|---|---|
| Unknown number of samples of floral and bee-collected almond pollen | unknown | Mold | No. of fungal isolates: | Mucor spp. was the dominant mold in floral pollen but not identified in bee-collected pollen. Aureobasidium pullulans, P. corylophilum, P. crustosum and Rhizopus nigricans were identified only in bee-collected pollen. | [61] | |
| Alternaria spp. | 6 | |||||
| Cladosporium spp. | 5 | |||||
| Penicillium spp. | 5 | |||||
| Aspergillus spp. | 3 | |||||
| Mucor spp. | 19 | |||||
| 90 samples of bee pollen | Spain (87 samples) Argentina (3 samples) | Mold | Aspergillus section Nigri | 1.4 × 10–2.3 × 102 cfu/g | The results show the occurrence of different mold species in pollen samples. Penicillium, Alternaria, and Aspergillus spp. were present in 90%, 86.6%, and 80% of samples, respectively. Predominant Aspergillus species was A. niger. The species of the genus Fusarium were isolated in 53.3%. | [38] |
| A.flavus +A. parasiticus | 1.7 × 10–2.5 × 10 cfu/g | |||||
| Other Aspergillus spp. | 2 × 10 cfu/g | |||||
| P. verrucosum | 1.4 × 102 cfu/g | |||||
| Other Penicillium spp. | 1.3 × 102–4.3 × 103 cfu/g | |||||
| Fusarium spp. | 16–9.5 × 101 cfu/g | |||||
| Cladosporium spp. | 6 × 10–1.4 × 103 cfu/g | |||||
| Alternaria spp. | 6 × 10–5.2 × 102 cfu/g | |||||
| Rhizopus spp. | 2 × 10–9 × 10 cfu/g | |||||
| Mucor spp. | 8–2.2 × 102 cfu/g | |||||
| Botrytis spp. | 8–3 × 10 cfu/g | |||||
| Epicoccum spp. | 5–10 cfu/g | |||||
| Yeast | Not specified | 3.6 × 102–7.3 × 103 cfu/g | ||||
| 42 samples of dehydrated bee pollen | Brazil | Mold/Yeast | Not specified | Total mold and yeast count: 102–1.3 × 104 cfu/g | About 12% of pollen samples were contaminated with mold and yeast above the limit (1×104) for a total mold and yeast proposed by Brazilian legislation. | [62] |
| 30 samples of bee pollen | Slovakia | Microscopic fungi (mold) | Alternaria spp. Cladosporium spp. Penicillium spp. Fusarium spp. Aspergillus spp. (A. flavus, A. ochraceus) Mucor spp. Trichoderma spp. Acremonium spp. Scopulariopsis spp. Rhizopus spp. Botrytis spp. | Total mold and yeast count: 1.1 × 102–4.57 × 105 cfu/g | The dominant fungi isolated from pollen samples were colonies of A. alternata, Cladosporium cladosporoides, and Penicillium spp. Also, the presence of well-known mycotoxicogenic species such as A. flavus and A. ochraceus were detected. | [63] |
| 19 samples of bee pollen | Mexico | Fungi (mold) | A. flavus | Incidence of mold genus (%): | Fungi contamination was generally low. The highest contamination was in three samples handled without packages. | [64] |
| Alternaria spp. | 3.6% | |||||
| Penicillium spp. | 2.9% | |||||
| Fusarium spp. | 2.9% | |||||
| Aspergillus spp. | 3.6% | |||||
| Mucor spp. | 3.1% | |||||
| Rhizopus spp. | 0.7% | |||||
| 8 samples of bee pollen | Slovakia | Mold | Alternaria spp. Cladosporium spp. Penicillium spp. Aspergillus spp. Mucor spp. Aureobasidium spp. Humicola spp. Monodictys spp. Paecilomyces spp. Rhizopus spp. Mortierella spp. Trichosporiella spp. Harpografium spp. Mortierella spp. | Total mold and yeast count: 107–4688 cfu/g | The results show that in all analyzed samples of pollen 21 fungal species of 13 genera of microscopic fungi were detected. The dominant identified species, over 62% of the isolates belonged to following genera: Mucor, Rhizopus, Aspergillus, Alternaria, and Paecilomyces. | [65] |
| 28 samples (fresh and dried bee pollen) | Cuba | Mold/Yeast | Not specified | Total mold and yeast count: 104–1.5 × 105 cfu/g | All samples had quantified number of mold and yeast above proposed limits (104 cfu/g for the fresh and 102 cfu/g for dried pollen). Nevertheless, in the dry pollen, a smaller number of high contaminated samples were recorded. Drying could not be used as reliable method for obtaining pollen with acceptable microbiological quality. | [66] |
| 8 samples of commercial bee pollen | Portugal (4 samples) Spain (3 samples) Unknown origin (1 sample) | Mold Yeast | Not specified Individually identified yeast | Total mold and yeast count: ˂10 to 9.4 × 102 cfu/g | All samples were contaminated with yeast and mold. Further, yeast species were identified, and results indicated the presence of five different genus of yeast which can influence the risk of food-borne illness and spoilage or can serve as an indicator of a lack of hygiene standards. | [67] |
| Unknown | Portugal | Mold/Yeast | Not specified | Total mold and yeast count: ˂104 cfu/g | Generally, yeast and mold were identified in 60% of all examined samples. pH and aw values had a strong impact on the total microbe number in pollen. | [54] |
| 22 samples of organic bee pollen | Portugal | Mold/Yeast | Not specified | Total mold and yeast count: ˂10–3560 cfu/g | In all samples of organic bee pollen, the presence of mold and yeast was detected, but their individual species were not identified. | [68] |
| 3 samples of pollen | Algeria | Mold/Yeast | Not specified | Total mold and yeast count: 5 × 104–4 × 105 cfu/g | / | [69] |
| 33 samples of bee pollen | Serbia | Mold | Alternaria spp. Mucor spp. Rhizopus spp. Cladosporium spp. Epicoccum spp. Acremonium spp. | Total mold count: 1 × 103–1 × 105 cfu/g | See Table 1. | [44] |
| 27 samples of dried bee pollen | Brazil | Mold | Total mold count: 1 × 102–5 × 102 cfu/g Incidence of mold genus (%): | Total mold count depends on growing media. | [47] | |
| Aspergillus spp. (A. flavus; A. fumigatus; A. versicolor; A. ochraceus; A. carbonarius; A. terreus; A. oryzae) | 85% | |||||
| Cladosporium spp. | 63% | |||||
| Penicillium spp. (P. citrinum; P. citreonigrum; P. glabrum; P. oxalicum) | 41% | |||||
| Alternaria spp. | 19% | |||||
| Wallemia spp. and Eurotium spp. | 11% | |||||
| Mucor spp. | 7% | |||||
| Curvularia spp., Paecilomyces spp. and Fusarium spp. (F. camptoceras) | 4% | |||||
| 45 samples of dehydrated bee pollen | Brazil | Mold Yeast | Not specified Identified different species | Total mold and yeast count: ˂10–7.67 × 103 cfu/g | / | [70] |
| 21 samples of bee pollen (Melipona bees) | Brazil | Mold/Yeast | Not specified | / | All samples were sterile without presence of any mold or yeast species. | [71] |
| 40 samples of bee pollen | Italy | Mold | Cladosporium spp. Alternaria spp. Humicola spp. Mucoraceae Acremonium spp. Penicillium spp. (P. chrysogenum; P. brevicompacticum) Aspergillus spp. (A. flavus; A. nidulans; A.miger; A. terreus) | Total mold count: 4–568 cfu/g | In all pollen samples at least one fungal isolate was detected. Cladosporium spp. was the most frequently detected mold. Aspergillus spp. and Penicillium spp., as a potentially mycotoxicogenic mold, were also identified in 8 i.e., 22 pollen samples. | [72] |
| Dehydrated (electric oven, EO) or lyophilized (L) bee pollen samples | Brazil | Mold/Yeast | Not specified | Total mold and yeast count: 99–242 cfu/g (EO) 16–935 cfu/g (L) | Number of quantified mold and yeast depended on time (April or September) of collection. | [73] |
| 26 samples of bee pollen | Serbia | Mold | Total mold count: | See Table 1 | [48] | |
| Alternaria spp. | 1 × 103 cfu/g | |||||
| Mucor spp. | 1 × 103 cfu/g | |||||
| Rhizopus spp. | 1 × 103 cfu/g | |||||
| Trichoderma spp. | 1 × 104 cfu/g | |||||
| 1 sample of bee pollen | Not known | Mold/Yeast | Not specified | Total mold and yeast count: >2l cfu/g | Presence of yeast and mold can be responsible for the potential presence of toxins in the samples. | [74] |
| 18 samples of commercial bee pollen | Argentina | Mold/Yeast | Not specified | Total mold and yeast count: ˂102 cfu/g | The total fungi number is specified for 28% of the samples. | [75] |
| 62 samples of dehydrated bee pollen | Brazil | Mold/Yeast | Not specified | Total mold and yeast count: 1.9 × 102–7.62 × 102 cfu/g | The microbial contamination is dependent on geographical origin of samples. | [76] |
| 8 samples of commercial bee pollen | Algeria | Mold/Yeast | Not specified | Total mold and yeast count: 104–2.8 × 105 cfu/g | / | [77] |
| 32 (13 fresh (F) and 19 dried (D) samples of bee pollen) | Bulgaria | Mold | Identified mold: Aspergillus spp. Fusarium spp. Penicillium spp. (P. brevicompactum) Alternaria spp. Cladosporium spp. Other species | Total mold count: 5.6 × 102 –3.7 × 104 cfu/g (F) 150–1.1 × 104 cfu/g (D) | The results show that the values for fungal colony count were significantly lower in the dried pollen samples. 136 fungal isolates were identified. Among detected isolates, genus Penicillium was dominant while the genus Fusarium was the least fungal contaminant. Dominant species isolated from 14 different samples was P. brevicompactum. | [78] |
| 19 samples of stored pollen of five stingless bee species | Brazil | Mold/Yeast | Not specified | Total mold and yeast count: 4.2 × 101 cfu/g (1 sample only) | The results show that only for the stored pollen of the stingless bee specie Frieseomellite varies it was possible to enumerate mold and yeast. | [79] |
| bee pollen samples | Colombia | Mold/Yeast | Not specified | Total mold and yeast count: 3 × 102–2 × 105 cfu/g | Number of quantified microbes is strongly dependent on applied temperature for drying of samples. | [80] |
| Food/Food Supplements | Mycotoxin(s) | MPC Value(s) | Reference |
|---|---|---|---|
| Groundnuts used as components for food production | AFB1 | 8 μg/kg | [33] |
| Sum of AFB1, AFB2, AFG1 and AFG2 | 15 μg/kg | ||
| Groundnuts for direct human consumption | AFB1 | 2 μg/kg | [33] |
| Sum of AFB1, AFB2, AFG1 and AFG2 | 4 μg/kg | ||
| Dried fruits used as components for food production | AFB1 | 5 μg/kg | [33] |
| Sum of AFB1, AFB2, AFG1 and AFG2 | 10 μg/kg | ||
| Dried fruits for direct human consumption | AFB1 | 2 μg/kg | [33] |
| Sum of AFB1, AFB2, AFG1 and AFG2 | 4 μg/kg | ||
| Raw milk used for consumption and dairy productions, infant formulae and infant-milk | AFB1 | 0 μg/kg | [33] |
| Sum of AFB1, AFB2, AFG1 and AFG2 | 0 μg/kg | ||
| Unprocessed cereals | OTA | 5 μg/kg | [33] |
| Cereals based products | OTA | 3 μg/kg | [33] |
| Instant coffee | OTA | 10 μg/kg | [33] |
| Roasted coffee | OTA | 5 μg/kg | [33] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kostić, A.Ž.; Milinčić, D.D.; Petrović, T.S.; Krnjaja, V.S.; Stanojević, S.P.; Barać, M.B.; Tešić, Ž.L.; Pešić, M.B. Mycotoxins and Mycotoxin Producing Fungi in Pollen: Review. Toxins 2019, 11, 64. https://doi.org/10.3390/toxins11020064
Kostić AŽ, Milinčić DD, Petrović TS, Krnjaja VS, Stanojević SP, Barać MB, Tešić ŽL, Pešić MB. Mycotoxins and Mycotoxin Producing Fungi in Pollen: Review. Toxins. 2019; 11(2):64. https://doi.org/10.3390/toxins11020064
Chicago/Turabian StyleKostić, Aleksandar Ž., Danijel D. Milinčić, Tanja S. Petrović, Vesna S. Krnjaja, Sladjana P. Stanojević, Miroljub B. Barać, Živoslav Lj. Tešić, and Mirjana B. Pešić. 2019. "Mycotoxins and Mycotoxin Producing Fungi in Pollen: Review" Toxins 11, no. 2: 64. https://doi.org/10.3390/toxins11020064
APA StyleKostić, A. Ž., Milinčić, D. D., Petrović, T. S., Krnjaja, V. S., Stanojević, S. P., Barać, M. B., Tešić, Ž. L., & Pešić, M. B. (2019). Mycotoxins and Mycotoxin Producing Fungi in Pollen: Review. Toxins, 11(2), 64. https://doi.org/10.3390/toxins11020064









