Efficacy of a Yeast Cell Wall Extract to Mitigate the Effect of Naturally Co-Occurring Mycotoxins Contaminating Feed Ingredients Fed to Young Pigs: Impact on Gut Health, Microbiome, and Growth
Abstract
1. Introduction
2. Results
2.1. Experiment 1
2.1.1. Growth Performance
2.1.2. Hematological Measurements
2.1.3. Serum Biochemical Measurements
2.1.4. Immunological and Oxidative Stress Measurements
2.1.5. Histomorphometry of Duodenum and Jejunum
2.2. Experiment 2
2.2.1. Growth Performance
2.2.2. Apparent Ileal Digestibility
2.2.3. Hematological Measurements
2.2.4. Serum Biochemical Measurements
2.2.5. Jejunal Histomorphometry and Crypt Cell Proliferation
2.2.6. Immunological and Oxidative Stress Measurements
2.2.7. Microbiome Analysis in Jejunal Mucosa
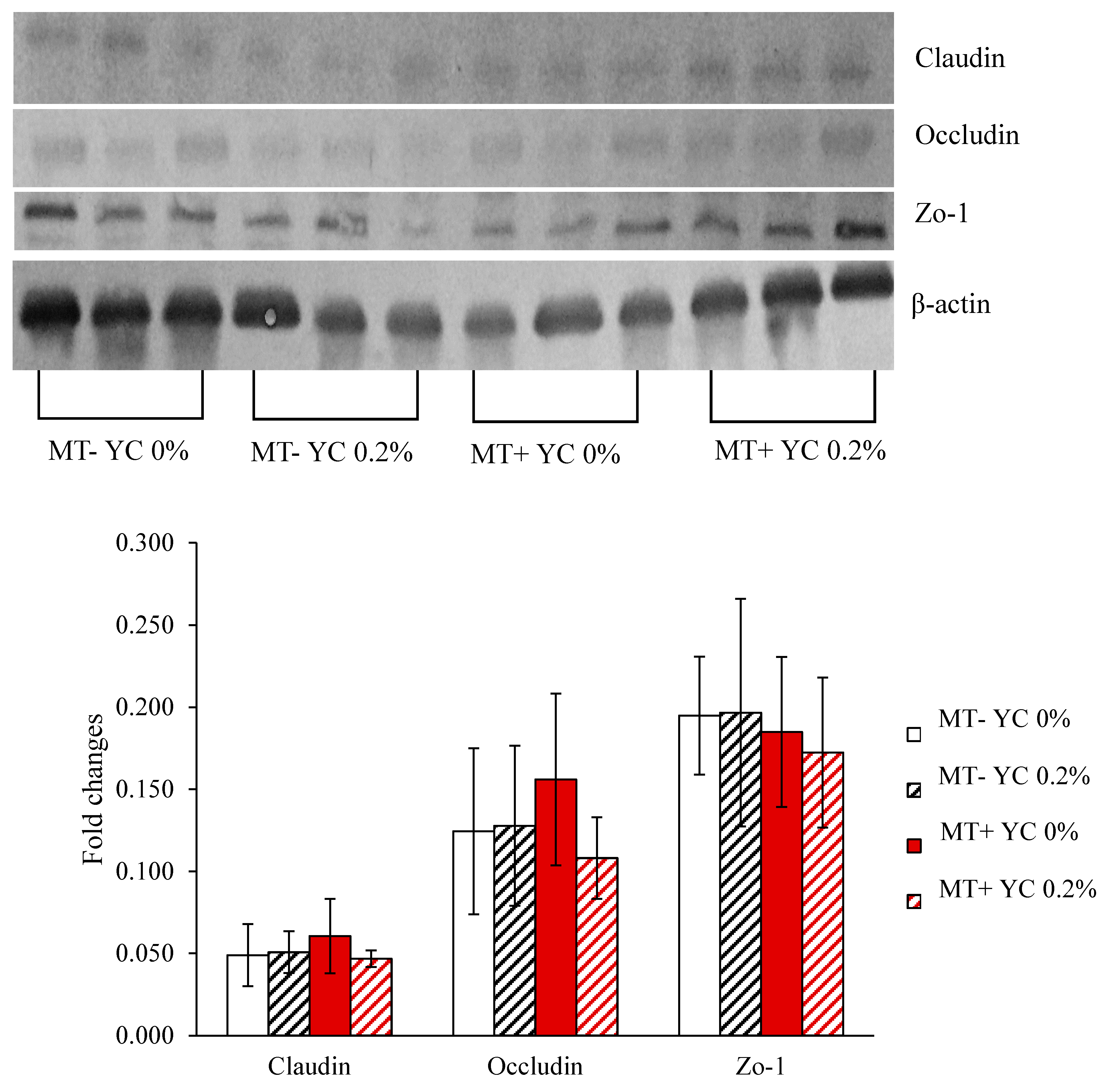
2.2.8. Tight Junction Proteins in Jejunum
3. Discussion
4. Materials and Methods
4.1. Animals and Experimental Diets
4.2. Sampling and Laboratory Analyses
4.2.1. Blood Sampling
4.2.2. Tissue, Mucosa, and Digesta Collection
4.2.3. Hematological and Biochemical Assays
4.2.4. Tumor Necrosis Factor-α
4.2.5. Immunoglobulins A and G
4.2.6. Malondialdehydes
4.2.7. 8-Hydroxy-Deoxyguanosine
4.2.8. Protein Carbonyl
4.2.9. Chemical Analysis
4.2.10. Apparent Ileal Digestibility
4.2.11. Immunohistochemistry for Ki-67 and Morphometry on Duodenum and Jejunum
4.2.12. Microbiome Analysis of Jejunal Mucosa
4.2.13. Tight Junction Proteins in Jejunal Tissue
4.3. Data Analysis and Interpretation
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Weaver, A.; See, M.; Kim, S. Protective effect of two yeast based feed additives on pigs chronically exposed to deoxynivalenol and zearalenone. Toxins 2014, 6, 3336–3353. [Google Scholar] [CrossRef] [PubMed]
- Chaytor, A.C.; See, M.T.; Hansen, J.A.; de Souza, A.L.P.; Middleton, T.F.; Kim, S.W. Effects of chronic exposure of diets with reduced concentrations of aflatoxin and deoxynivalenol on growth and immune status of pigs. J. Anim. Sci. 2011, 89, 124–135. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Park, I.; Guo, J.; Weaver, A.C.; Kim, S.W. Impacts of low level aflatoxin in feed and the use of modified yeast cell wall extract on growth and health of nursery pigs. Anim. Nutr. 2015, 1, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Weaver, A.; See, M.; Hansen, J.; Kim, Y.; De Souza, A.; Middleton, T.; Kim, S. The use of feed additives to reduce the effects of aflatoxin and deoxynivalenol on pig growth, organ health and immune status during chronic exposure. Toxins 2013, 5, 1261–1281. [Google Scholar] [CrossRef] [PubMed]
- Jouany, J.P. Methods for preventing, decontaminating and minimizing the toxicity of mycotoxins in feeds. Anim. Feed Sci. Technol. 2007, 137, 342–362. [Google Scholar] [CrossRef]
- Yiannikouris, A.; André, G.; Buléon, A.; Jeminet, G.; Canet, I.; François, J.; Bertin, G.; Jouany, J.-P. Comprehensive conformational study of key interactions involved in zearalenone complexation with β-D-glucans. Biomacromolecules 2004, 5, 2176–2185. [Google Scholar] [CrossRef]
- Yiannikouris, A.; François, J.; Poughon, L.; Dussap, C.-G.; Bertin, G.; Jeminet, G.; Jouany, J.-P. Adsorption of zearalenone by b-D-glucans in the Saccharomyces cerevisiae cell wall. J. Food Prot. 2004, 67, 1195–1200. [Google Scholar] [CrossRef]
- Yiannikouris, A.; Francois, J.; Poughon, L.; Dussap, C.-G.; Rard Bertin, G.Ä.; Jeminet, G.; Jouany, J.-P. Alkali extraction of β-D-glucans from Saccharomyces cerevisiae cell wall and study of their adsorptive properties toward zearalenone. J. Agric. Food Chem. 2004, 52, 3666–3673. [Google Scholar] [CrossRef]
- Yiannikouris, A.; André, G.; Poughon, L.; François, J.; Dussap, C.-G.; Jeminet, G.; Bertin, G.; Jouany, J.-P. Chemical and conformational study of the interactions involved in mycotoxin complexation with β-d-glucans. Biomacromolecules 2006, 7, 1147–1155. [Google Scholar] [CrossRef]
- Yiannikouris, A.; Kettunen, H.; Apajalahti, J.; Pennala, E.; Moran, C.A. Comparison of the sequestering properties of yeast cell wall extract and hydrated sodium calcium aluminosilicate in three in vitro models accounting for the animal physiological bioavailability of zearalenone. Food Addit. Contam. Part A 2013, 30, 1641–1650. [Google Scholar] [CrossRef]
- Firmin, S.; Gandia, P.; Morgavi, D.P.; Houin, G.; Jouany, J.P.; Bertin, G.; Boudra, H. Modification of aflatoxin B 1 and ochratoxin A toxicokinetics in rats administered a yeast cell wall preparation. Food Addit. Contam. Part A 2010, 27, 1153–1160. [Google Scholar] [CrossRef] [PubMed]
- Prelusky, D.B.; Gerdes, R.G.; Underhill, K.L.; Rotter, B.A.; Jui, P.Y.; Trenholm, H.L. Effects of low-level dietary deoxynivalenol on haematological and clinical parameters of the pig. Nat. Toxins 1994, 2, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Harvey, R.B.; Edrington, T.S.; Kubena, L.F.; Elissalde, M.H.; Rottinghaus, G.E. Influence of aflatoxin and fumonisin B1-containing culture material on growing barrows. Am. J. Vet. Res. 1995, 56, 1668–1672. [Google Scholar] [PubMed]
- Moeser, A.J.; Pohl, C.S.; Rajput, M. Weaning stress and gastrointestinal barrier development: Implications for lifelong gut health in pigs. Anim. Nutr. 2017, 3, 313–321. [Google Scholar] [CrossRef]
- Linghong, J.; Wang, W.; Degroote, J.; Van Noten, N.; Yan, H.; Majdeddin, M.; Van Poucke, M.; Peelman, L.; Goderis, A.; Van De Mierop, K.; et al. Mycotoxin binder improves growth rate in piglets associated with reduction of toll-like receptor-4 and increase of tight junction protein gene expression in gut mucosa. J. Anim. Sci. Biotechnol. 2017, 8, 80. [Google Scholar]
- Dersjant-Li, Y.; Verstegen, M.W.A.; Gerrits, W.J.J. The impact of low concentrations of aflatoxin, deoxynivalenol or fumonisin in diets on growing pigs and poultry. Nutr. Res. Rev. 2003, 16, 223–239. [Google Scholar] [CrossRef]
- Pestka, J.J. Deoxynivalenol: Mechanisms of action, human exposure, and toxicological relevance. Arch. Toxicol. 2010, 84, 663–679. [Google Scholar] [CrossRef]
- Pasternak, J.A.; Aiyer, V.I.A.; Hamonic, G.; Beaulieu, A.D.; Columbus, D.A.; Wilson, H.L. Molecular and physiological effects on the small intestine of weaner pigs following feeding with deoxynivalenol-contaminated feed. Toxins 2018, 10, 40. [Google Scholar] [CrossRef]
- Flannery, B.M.; Clark, E.S.; Pestka, J.J. Anorexia induction by the trichothecene deoxynivalenol (vomitoxin) is mediated by the release of the gut satiety hormone peptide YY. Toxicol. Sci. 2012, 130, 289–297. [Google Scholar] [CrossRef]
- Fioramonti, J.; Dupuy, C.; Dupuy, J.; Bueno, L. The mycotoxin, deoxynivalenol, delays gastric emptying through serotonin-3 receptors in rodents. J. Pharmacol. Exp. Ther. 1993, 266, 1255–1260. [Google Scholar]
- Flannery, B.M.; Wu, W.; Pestka, J.J. Characterization of deoxynivalenol-induced anorexia using mouse bioassay. Food Chem. Toxicol. 2011, 49, 1863–1869. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Bates, M.A.; Bursian, S.J.; Flannery, B.; Zhou, H.-R.; Link, J.E.; Zhang, H.; Pestka, J.J. Peptide YY 3-36 and 5-hydroxytryptamine mediate emesis induction by trichothecene deoxynivalenol (vomitoxin). Toxicol. Sci. 2013, 133, 186–195. [Google Scholar] [CrossRef]
- Manners, D.J.; Masson, A.J.; Patterson, J.C. The structure of a beta-(1-3)-D-glucan from yeast cell walls. Biochem. J. 1973, 135, 19–30. [Google Scholar] [CrossRef] [PubMed]
- Yiannikouris, A.; Poughon, L.; Cameleyre, X.; Dussap, C.-G.; François, J.; Bertin, G.; Jouany, J.-P. A novel technique to evaluate interactions between Saccharomyces cerevisiae cell wall and mycotoxins: Application to zearalenone. Biotechnol. Lett. 2003, 25, 783–789. [Google Scholar] [CrossRef] [PubMed]
- Xiong, X.; Yang, H.; Li, B.; Liu, G.; Huang, R.; Li, F.; Liao, P.; Zhang, Y.; Martin Nyachoti, C.; Deng, D. Dietary supplementation with yeast product improves intestinal function, and serum and ileal amino acid contents in weaned piglets. Livest. Sci. 2015, 171, 20–27. [Google Scholar] [CrossRef]
- Shen, Y.B.; Piao, X.S.; Kim, S.W.; Wang, L.; Liu, P.; Yoon, I.; Zhen, Y.G. Effects of yeast culture supplementation on growth performance, intestinal health, and immune response of nursery pigs. J. Anim. Sci. 2009, 87, 2614–2624. [Google Scholar] [CrossRef]
- Sweeney, T.; Collins, C.B.; Reilly, P.; Pierce, K.M.; Ryan, M.; O’Doherty, J.V. Effect of purified β-glucans derived from Laminaria digitata, Laminaria hyperborea and Saccharomyces cerevisiae on piglet performance, selected bacterial populations, volatile fatty acids and pro-inflammatory cytokines in the gastrointestinal tract of pig. Br. J. Nutr. 2012, 108, 1226–1234. [Google Scholar] [CrossRef]
- Freire, L.; Sant’Ana, A.S. Modified mycotoxins: An updated review on their formation, detection, occurrence, and toxic effects. Food Chem. Toxicol. 2018, 111, 189–205. [Google Scholar] [CrossRef]
- Anraku, M.; Chuang, V.T.G.; Maruyama, T.; Otagiri, M. Redox properties of serum albumin. Biochim. Biophys. Acta Gen. Subj. 2013, 1830, 5465–5472. [Google Scholar] [CrossRef]
- Latimer, K.S. (Ed.) Duncan and Prasse’s Veterinary Laboratory Medicine: Clinical Pathology, 5th ed.; John Wiley & Sons: Ames, IA, USA, 2011. [Google Scholar]
- Ruiz, M.-J.; Macáková, P.; Juan-García, A.; Font, G. Cytotoxic effects of mycotoxin combinations in mammalian kidney cells. Food Chem. Toxicol. 2011, 49, 2718–2724. [Google Scholar] [CrossRef]
- Hou, Y.-J.; Zhao, Y.-Y.; Xiong, B.; Cui, X.-S.; Kim, N.-H.; Xu, Y.-X.; Sun, S.-C. Mycotoxin-containing diet causes oxidative stress in the mouse. PLoS ONE 2013, 8, e60374. [Google Scholar] [CrossRef] [PubMed]
- Reddy, K.; Song, J.; Lee, H.-J.; Kim, M.; Kim, D.-W.; Jung, H.; Kim, B.; Lee, Y.; Yu, D.; Kim, D.-W.; et al. Effects of high levels of deoxynivalenol and zearalenone on growth performance, and hematological and immunological parameters in pigs. Toxins 2018, 10, 114. [Google Scholar] [CrossRef] [PubMed]
- Pierron, A.; Alassane-Kpembi, I.; Oswald, I.P. Impact of mycotoxin on immune response and consequences for pig health. Anim. Nutr. 2016, 2, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Garas, L.C.; Feltrin, C.; Kristina Hamilton, M.; Hagey, J.V.; Murray, J.D.; Bertolini, L.R.; Bertolini, M.; Raybould, H.E.; Maga, E.A. Milk with and without lactoferrin can influence intestinal damage in a pig model of malnutrition. Food Funct. 2016, 7, 665–678. [Google Scholar] [CrossRef]
- Aderem, A.; Ulevitch, R.J. Toll-like receptors in the induction of the innate immune response. Nature 2000, 406, 782–787. [Google Scholar] [CrossRef]
- Yang, F.; Wang, A.; Zeng, X.; Hou, C.; Liu, H.; Qiao, S. Lactobacillus reuteri I5007 modulates tight junction protein expression in IPEC-J2 cells with LPS stimulation and in newborn piglets under normal conditions. BMC Microbiol. 2015, 15, 32. [Google Scholar] [CrossRef]
- Vaahtovuo, J.; Korkeamäki, M.; Munukka, E.; Hämeenoja, P.; Vuorenmaa, J. Microbial balance index—A view on the intestinal microbiota. Livest. Sci. 2007, 109, 174–178. [Google Scholar] [CrossRef]
- Almeida, L.R.; Costa, P.S.; Nascimento, A.M.A.; Reis, M.d.P.; Barros, K.O.; Alvim, L.B.; Nunes, Á.C.; Queiroz, D.M.M.; Rocha, G.A.; Nicoli, J.R.; et al. Porcine stomachs with and without gastric ulcer differ in Lactobacillus load and strain characteristics. Can. J. Microbiol. 2018, 64, 493–499. [Google Scholar] [CrossRef]
- Sapountzis, P.; Gruntjes, T.; Otani, S.; Estevez, J.; da Costa, R.R.; Plunkett, G., 3rd; Perna, N.T.; Poulsen, M. The Enterobacterium Trabulsiella odontotermitis presents novel adaptations related to its association with fungus-growing termites. Appl. Environ. Microbiol. 2015, 81, 6577–6588. [Google Scholar] [CrossRef]
- De Angelis, M.; Siragusa, S.; Berloco, M.; Caputo, L.; Settanni, L.; Alfonsi, G.; Amerio, M.; Grandi, A.; Ragni, A.; Gobbetti, M. Selection of potential probiotic lactobacilli from pig feces to be used as additives in pelleted feeding. Res. Microbiol. 2006, 157, 792–801. [Google Scholar] [CrossRef]
- Zou, Z.-Y.; He, Z.-F.; Li, H.-J.; Han, P.-F.; Meng, X.; Zhang, Y.; Zhou, F.; Ouyang, K.-P.; Chen, X.-Y.; Tang, J. In vitro removal of deoxynivalenol and T-2 toxin by lactic acid bacteria. Food Sci. Biotechnol. 2012, 21, 1677–1683. [Google Scholar] [CrossRef]
- Haskard, C.A.; El-Nezami, H.S.; Kankaanpää, P.E.; Salminen, S.; Ahokas, J.T. Surface binding of aflatoxin B(1) by lactic acid bacteria. Appl. Environ. Microbiol. 2001, 67, 3086–3091. [Google Scholar] [CrossRef] [PubMed]
- Sato, I.; Ito, M.; Ishizaka, M.; Ikunaga, Y.; Sato, Y.; Yoshida, S.; Koitabashi, M.; Tsushima, S. Thirteen novel deoxynivalenol-degrading bacteria are classified within two genera with distinct degradation mechanisms. FEMS Microbiol. Lett. 2012, 327, 110–117. [Google Scholar] [CrossRef] [PubMed]
- Willing, B.P.; Van Kessel, A.G. Enterocyte proliferation and apoptosis in the caudal small intestine is influenced by the composition of colonizing commensal bacteria in the neonatal gnotobiotic pig. J. Anim. Sci. 2007, 85, 3256–3266. [Google Scholar] [CrossRef]
- (NRC) National Research Council. Nutrition Requirements of Swine, 11th ed.; National Academy Press: Washington, DC, USA, 2012. [Google Scholar]
- Swamy, H.V.L.N.; Smith, T.K.; MacDonald, E.J.; Karrow, N.A.; Woodward, B.; Boermans, H.J. Effects of feeding a blend of grains naturally contaminated with Fusarium mycotoxins on growth and immunological measurements of starter pigs, and the efficacy of a polymeric glucomannan mycotoxin adsorbent. J. Anim. Sci. 2003, 81, 2792–2803. [Google Scholar] [CrossRef]
- Weaver, A.C.; Kim, S.W.; Campbell, J.M.; Crenshaw, J.D.; Polo, J. Efficacy of dietary spray dried plasma protein to mitigate the negative effects on performance of pigs fed diets with corn naturally contaminated with multiple mycotoxins. J. Anim. Sci. 2014, 92, 3878–3886. [Google Scholar] [CrossRef]
- Shen, Y.B.; Weaver, A.C.; Kim, S.W. Effect of feed grade L-methionine on growth performance and gut health in nursery pigs compared with conventional DL-methionine. J. Anim. Sci. 2014, 92, 5530–5539. [Google Scholar] [CrossRef]
- Passos, A.A.; Park, I.; Ferket, P.; von Heimendahl, E.; Kim, S.W. Effect of dietary supplementation of xylanase on apparent ileal digestibility of nutrients, viscosity of digesta, and intestinal morphology of growing pigs fed corn and soybean meal based diet. Anim. Nutr. 2015, 1, 19–23. [Google Scholar] [CrossRef]
- AOAC International. AOAC International Guidelines for Laboratories Performing Microbiological and Chemical Analyses of Food and Pharmaceuticals: An Aid to Interpretation of ISO/IEC 17025: 2005; AOAC: Frederick, MD, USA, 2006. [Google Scholar]
- Myers, W.D.; Ludden, P.A.; Nayigihugu, V.; Hess, B.W. Technical Note: A procedure for the preparation and quantitative analysis of samples for titanium dioxide. J. Anim. Sci. 2004, 82, 179–183. [Google Scholar] [CrossRef]
- Almeida, J.S.; Iriabho, E.E.; Gorrepati, V.L.; Wilkinson, S.R.; Hackney, J.R.; Grüneberg, A.; Robbins, D.E. ImageJS: Personalized, participated, pervasive, and reproducible image bioinformatics in the web browser. J. Pathol. Inform. 2012, 3, 25. [Google Scholar] [CrossRef]
| Mycotoxins (MT) | - | + | p Value | |||||
|---|---|---|---|---|---|---|---|---|
| YCWE (YC) | 0% | 0.2% | 0% | 0.2% | SEM | MT | YC | MT × YC |
| Body Weight,kg | ||||||||
| Initial | 55.7 | 55.6 | 55.7 | 55.8 | 3.1 | 0.687 | 0.929 | 0.608 |
| Day 7 | 59.7 | 58.8 | 58.8 | 58.6 | 3.6 | 0.151 | 0.176 | 0.405 |
| Day 14 | 68.9 | 68.0 | 67.8 | 67.2 | 3.8 | 0.099 | 0.178 | 0.758 |
| Day 21 | 75.8 | 74.7 | 74.7 | 74.8 | 4.1 | 0.444 | 0.409 | 0.335 |
| Day 28 | 82.5 | 80.8 | 81.2 | 82.1 | 4.3 | 0.547 | 0.262 | 0.197 |
| Day 35 | 90.5 | 88.3 | 88.5 | 88.4 | 4.5 | 0.255 | 0.205 | 0.228 |
| ADG2,kg | ||||||||
| Day 0 to 7 | 1.342 | 1.078 | 1.047 | 0.937 | 0.211 | 0.048 | 0.088 | 0.470 |
| Day 7 to 14 | 1.311 | 1.303 | 1.282 | 1.229 | 0.054 | 0.271 | 0.506 | 0.626 |
| Day 14 to 21 | 0.985 | 0.956 | 0.987 | 1.083 | 0.118 | 0.221 | 0.520 | 0.231 |
| Day 21 to 28 | 0.963 | 0.880 | 0.928 | 0.933 | 0.067 | 0.879 | 0.492 | 0.442 |
| Day 28 to 35 | 1.142 | 1.074 | 1.037 | 1.012 | 0.105 | 0.220 | 0.484 | 0.745 |
| Overall | 1.124 | 1.055 | 1.057 | 1.052 | 0.071 | 0.201 | 0.178 | 0.249 |
| ADFI3,kg | ||||||||
| Day 0 to 7 | 2.733 | 2.502 | 2.597 | 2.579 | 0.142 | 0.788 | 0.256 | 0.330 |
| Day 7 to 14 | 3.075 | 2.825 | 2.828 | 2.847 | 0.170 | 0.174 | 0.166 | 0.107 |
| Day 14 to 21 | 2.889 | 2.774 | 2.813 | 2.785 | 0.232 | 0.741 | 0.472 | 0.659 |
| Day 21 to 28 | 2.588 | 2.606 | 2.430 | 2.683 | 0.233 | 0.781 | 0.358 | 0.421 |
| Day 28 to 35 | 3.093 | 2.828 | 2.872 | 2.672 | 0.252 | 0.145 | 0.073 | 0.796 |
| Overall | 2.894 | 2.733 | 2.713 | 2.721 | 0.183 | 0.165 | 0.272 | 0.225 |
| G:F4 | ||||||||
| Day 0 to 7 | 0.472 | 0.423 | 0.396 | 0.364 | 0.064 | 0.044 | 0.216 | 0.798 |
| Day 7 to 14 | 0.434 | 0.461 | 0.457 | 0.435 | 0.024 | 0.925 | 0.896 | 0.151 |
| Day 14 to 21 | 0.340 | 0.346 | 0.353 | 0.390 | 0.022 | 0.098 | 0.209 | 0.372 |
| Day 21 to 28 | 0.381 | 0.344 | 0.390 | 0.355 | 0.026 | 0.683 | 0.137 | 0.960 |
| Day 28 to 35 | 0.369 | 0.382 | 0.358 | 0.379 | 0.019 | 0.697 | 0.339 | 0.788 |
| Overall | 0.389 | 0.388 | 0.390 | 0.387 | 0.007 | 0.937 | 0.627 | 0.865 |
| Mycotoxins (MT) | - | + | p Value | |||||
|---|---|---|---|---|---|---|---|---|
| YCWE (YC) | 0% | 0.2% | 0% | 0.2% | SEM | MT | YC | MT × YC |
| RBC2, 106/µL | 6.84 | 6.90 | 7.05 | 6.80 | 0.23 | 0.687 | 0.601 | 0.387 |
| Hemogblobin, g/dL | 12.43 | 12.48 | 12.64 | 11.95 | 0.45 | 0.709 | 0.317 | 0.254 |
| Hematocrit, % | 40.91 | 40.63 | 41.05 | 38.79 | 1.38 | 0.495 | 0.189 | 0.317 |
| MCV3, fL | 59.70 | 58.70 | 58.39 | 57.22 | 1.19 | 0.124 | 0.286 | 0.988 |
| MCH4, pg | 18.22 | 18.12 | 17.96 | 17.62 | 0.36 | 0.217 | 0.527 | 0.755 |
| MCHC5, g/dL | 30.47 | 30.81 | 30.79 | 30.79 | 0.21 | 0.492 | 0.420 | 0.412 |
| Platelet count, mL | 185.9 | 144.0 | 133.7 | 152.4 | 27.1 | 0.394 | 0.618 | 0.237 |
| WBC6, 103/µL | 20.52 | 21.43 | 19.91 | 18.83 | 1.36 | 0.236 | 0.950 | 0.470 |
| Neutrophils, cell/mL | 7.89 | 6.66 | 5.89 | 5.77 | 0.72 | 0.003 | 0.139 | 0.225 |
| Lymphocytes, cell/mL | 10.97A | 13.05B | 12.47AB | 11.30AB | 0.97 | 0.868 | 0.614 | 0.099 |
| Monocytes, cell/mL | 1.04 | 1.13 | 0.96 | 1.13 | 0.15 | 0.777 | 0.397 | 0.781 |
| Eosinophils, cell/µL | 543 | 499 | 518 | 484 | 84 | 0.812 | 0.636 | 0.949 |
| Basophils, cell/µL | 109 | 128 | 130 | 66 | 45 | 0.585 | 0.545 | 0.271 |
| Mycotoxins (MT) | - | + | p Value | |||||
|---|---|---|---|---|---|---|---|---|
| YCWE (YC) | 0% | 0.2% | 0% | 0.2% | SEM | MT | YC | MT × YC |
| Total protein, g/dL | 6.44 | 6.18 | 6.49 | 6.36 | 0.12 | 0.309 | 0.090 | 0.573 |
| Albumin, g/dL | 3.60 | 3.48 | 3.62 | 3.73 | 0.08 | 0.042 | 0.950 | 0.074 |
| Globulin, g/dL | 2.84 | 2.70 | 2.87 | 2.66 | 0.11 | 0.948 | 0.106 | 0.731 |
| A to G ratio2 | 1.28 | 1.29 | 1.27 | 1.40 | 0.06 | 0.389 | 0.231 | 0.319 |
| AST3, U/L | 23.25 | 27.88 | 22.72 | 20.36 | 2.71 | 0.111 | 0.712 | 0.144 |
| ALT4, U/L | 19.38 | 20.63 | 20.03 | 17.83 | 1.76 | 0.507 | 0.769 | 0.291 |
| ALP5, U/L | 132 | 143 | 149 | 157 | 10 | 0.098 | 0.301 | 0.947 |
| CPK6, U/L | 1,230 | 1,749 | 1,119 | 1,358 | 254 | 0.287 | 0.113 | 0.552 |
| BUN7, mg/dL | 14.75 | 13.75 | 13.08 | 12.33 | 1.84 | 0.088 | 0.336 | 0.868 |
| Creatinine, mg/dL | 1.30 | 1.40 | 1.30 | 1.25 | 0.08 | 0.197 | 0.629 | 0.189 |
| BUN-to-creatinine ratio | 11.50 | 10.25 | 10.07 | 10.34 | 1.85 | 0.385 | 0.526 | 0.330 |
| Cholesterol, mg/dL | 86.13 | 85.25 | 79.89 | 74.29 | 3.65 | 0.019 | 0.354 | 0.497 |
| Glucose, mg/dL | 72.50 | 77.13 | 82.90 | 79.27 | 4.14 | 0.051 | 0.873 | 0.189 |
| Ca, mg/dL | 10.59 | 10.54 | 10.66 | 10.76 | 0.13 | 0.232 | 0.821 | 0.519 |
| Cl, mEq/L | 100.6 | 101.5 | 100.0 | 101.1 | 0.86 | 0.384 | 0.081 | 0.807 |
| Na, mEq/L | 145.4 | 147.5 | 145.8 | 146.0 | 0.65 | 0.374 | 0.064 | 0.129 |
| K, mEq/L | 5.28 | 5.35 | 5.06 | 4.95 | 0.22 | 0.064 | 0.914 | 0.566 |
| Na to K ratio | 27.63 | 27.75 | 28.96 | 29.90 | 1.06 | 0.053 | 0.553 | 0.652 |
| P, mg/dL | 9.16 | 9.03 | 8.82 | 8.86 | 0.16 | 0.100 | 0.721 | 0.577 |
| Mycotoxins (MT) | - | + | p Value | |||||
|---|---|---|---|---|---|---|---|---|
| YCWE (YC) | 0% | 0.2% | 0% | 0.2% | SEM | MT | YC | MT × YC |
| Immunological Parameters | ||||||||
| Duodenal TNF-α2, pg/mg protein | 8.79 | 6.96 | 8.69 | 8.91 | 0.81 | 0.261 | 0.331 | 0.217 |
| Jejunal TNF-α, pg/mg protein | 6.56 | 5.53 | 5.36 | 5.80 | 0.75 | 0.490 | 0.659 | 0.281 |
| Serumal TNF-α, pg/mL | 183 | 187 | 169 | 173 | 11 | 0.206 | 0.726 | 0.959 |
| Duodenal IgG3, µg/mg protein | 2.77 | 4.26 | 5.36 | 4.61 | 0.71 | 0.049 | 0.605 | 0.128 |
| Jejunal IgG, µg/mg protein | 1.75 a | 2.95 b | 2.96 b | 2.40 ab | 0.37 | 0.255 | 0.274 | 0.005 |
| Serumal IgG, mg/mL | 7.92 | 8.04 | 9.49 | 7.75 | 0.85 | 0.240 | 0.143 | 0.096 |
| Oxidative Stress Parameters | ||||||||
| Duodenal MDA4, nmol/mg protein | 0.82 | 0.73 | 0.68 | 0.96 | 0.12 | 0.701 | 0.415 | 0.123 |
| Jejunal MDA, nmol/mg protein | 1.14 | 1.20 | 0.99 | 1.08 | 0.38 | 0.561 | 0.747 | 0.959 |
| Serumal MDA, µM | 15.94 | 14.74 | 15.80 | 15.22 | 5.39 | 0.915 | 0.575 | 0.845 |
| Serumal 8-OHdG5, ng/mL | 1.29 | 0.93 | 1.66 | 0.26 | 0.49 | 0.737 | 0.049 | 0.235 |
| Mycotoxins (MT) | - | + | p Value | |||||
|---|---|---|---|---|---|---|---|---|
| YCWE (YC) | 0% | 0.2% | 0% | 0.2% | SEM | MT | YC | MT × YC |
| Duodenum | ||||||||
| Villus height (VH), µm | 516 | 484 | 498 | 491 | 19 | 0.770 | 0.306 | 0.509 |
| Villus width, µm | 115 | 112 | 111 | 108 | 5.8 | 0.323 | 0.442 | 1.000 |
| Crypt depth (CD), µm | 295 | 253 | 275 | 268 | 13 | 0.840 | 0.051 | 0.141 |
| VH to CD ratio | 1.77 | 1.93 | 1.83 | 1.83 | 0.08 | 0.761 | 0.192 | 0.233 |
| Jejunum | ||||||||
| Villus height, µm | 482 | 495 | 495 | 495 | 36 | 0.845 | 0.845 | 0.842 |
| Villus width, µm | 119 | 101 | 116 | 108 | 5.3 | 0.685 | 0.018 | 0.324 |
| Crypt depth, µm | 238 | 235 | 243 | 245 | 8.9 | 0.368 | 0.950 | 0.798 |
| VH-to-CD ratio | 2.04 | 2.11 | 2.05 | 2.01 | 0.16 | 0.758 | 0.899 | 0.646 |
| Mycotoxins (MT) | - | + | p Value | |||||
|---|---|---|---|---|---|---|---|---|
| YCWE (YC) | 0% | 0.2% | 0% | 0.2% | SEM | MT | YC | MT × YC |
| Body Weight,kg | ||||||||
| Initial | 6.0 | 6.0 | 6.0 | 6.0 | 0.2 | 1.000 | 0.632 | 0.905 |
| Day 5 | 6.1 | 5.9 | 5.9 | 5.9 | 0.3 | 0.364 | 0.258 | 0.569 |
| Day 10 | 6.9 | 6.7 | 6.8 | 6.4 | 0.3 | 0.293 | 0.057 | 0.473 |
| Day 15 | 8.5 | 8.1 | 8.2 | 7.6 | 0.4 | 0.079 | 0.037 | 0.779 |
| Day 20 | 10.8 | 10.3 | 10.2 | 9.3 | 0.4 | 0.030 | 0.066 | 0.638 |
| Day 27 | 14.5 | 14.3 | 13.7 | 14.4 | 0.6 | 0.017 | 0.156 | 0.327 |
| Day 34 | 19.6 | 18.6 | 17.4 | 16.4 | 0.7 | 0.001 | 0.129 | 0.940 |
| Day 41 | 25.4 | 24.3 | 22.2 | 22.0 | 0.8 | <0.001 | 0.370 | 0.514 |
| Day 48 | 31.7 | 30.4 | 27.6 | 27.9 | 1.0 | <0.001 | 0.589 | 0.348 |
| ADG2, kg | ||||||||
| Day 0 to 5 | 0.017 | −0.017 | −0.008 | −0.024 | 0.014 | 0.205 | 0.048 | 0.489 |
| Day 5 to 10 | 0.165 | 0.155 | 0.180 | 0.104 | 0.021 | 0.390 | 0.047 | 0.122 |
| Day 10 to 15 | 0.321 | 0.278 | 0.265 | 0.243 | 0.029 | 0.082 | 0.210 | 0.673 |
| Day 15 to 20 | 0.462 | 0.445 | 0.403 | 0.343 | 0.033 | 0.020 | 0.249 | 0.527 |
| Day 20 to 27 | 0.529 | 0.567 | 0.506 | 0.444 | 0.044 | 0.103 | 0.783 | 0.259 |
| Day 27 to 34 | 0.728 A | 0.614 AB | 0.521 B | 0.569 AB | 0.047 | 0.011 | 0.488 | 0.096 |
| Day 34 to 41 | 0.831 a | 0.819 a | 0.686 b | 0.795 a | 0.034 | 0.006 | 0.110 | 0.045 |
| Day 41 to 48 | 0.895 | 0.876 | 0.778 | 0.852 | 0.036 | 0.046 | 0.420 | 0.180 |
| Phase 1 (day 0 to 20) | 0.241 | 0.215 | 0.210 | 0.166 | 0.017 | 0.025 | 0.048 | 0.612 |
| Phase 2 (day 20 to 48) | 0.746 | 0.719 | 0.623 | 0.665 | 0.024 | 0.001 | 0.750 | 0.151 |
| Overall | 0.494 | 0.467 | 0.416 | 0.416 | 0.017 | <0.001 | 0.420 | 0.440 |
| ADFI3, kg | ||||||||
| Day 0 to 5 | 0.083 c | 0.053 d | 0.066 | 0.049 | 0.008 | 0.212 | 0.005 | 0.403 |
| Day 5 to 10 | 0.234 | 0.201 | 0.201 | 0.145 | 0.019 | 0.027 | 0.027 | 0.564 |
| Day 10 to 15 | 0.433 | 0.402 | 0.372 | 0.303 | 0.024 | 0.002 | 0.039 | 0.428 |
| Day 15 to 20 | 0.642 | 0.625 | 0.523 | 0.488 | 0.039 | 0.001 | 0.475 | 0.805 |
| Day 20 to 27 | 0.786 | 0.805 | 0.643 | 0.630 | 0.043 | <0.001 | 0.944 | 0.722 |
| Day 27 to 34 | 1.060 | 0.944 | 0.783 | 0.789 | 0.056 | <0.001 | 0.328 | 0.281 |
| Day 34 to 41 | 1.245 | 1.136 | 1.036 | 1.046 | 0.064 | 0.010 | 0.327 | 0.284 |
| Day 41 to 48 | 1.529 | 1.406 | 1.275 | 1.262 | 0.055 | <0.001 | 0.156 | 0.245 |
| Phase 1 (day 0 to 20) | 0.348 | 0.320 | 0.291 | 0.247 | 0.019 | 0.001 | 0.065 | 0.679 |
| Phase 2 (day 20 to 48) | 1.155 | 1.073 | 0.934 | 0.932 | 0.042 | <0.001 | 0.301 | 0.324 |
| Overall | 0.752 | 0.700 | 0.613 | 0.589 | 0.028 | <0.001 | 0.149 | 0.554 |
| G:F4 | ||||||||
| Day 0 to 5 | −0.043 | −1.346 | −0.299 | −1.109 | 0.542 | 0.986 | 0.054 | 0.646 |
| Day 5 to 10 | 0.718 | 0.769 | 0.873 A | 0.632 B | 0.074 | 0.904 | 0.210 | 0.055 |
| Day 10 to 15 | 0.732 | 0.693 | 0.721 | 0.806 | 0.061 | 0.352 | 0.678 | 0.256 |
| Day 15 to 20 | 0.731 | 0.705 | 0.771 | 0.709 | 0.036 | 0.547 | 0.226 | 0.632 |
| Day 20 to 27 | 0.666 | 0.695 | 0.791 | 0.703 | 0.045 | 0.140 | 0.501 | 0.190 |
| Day 27 to 34 | 0.689 | 0.640 | 0.663 | 0.716 | 0.034 | 0.447 | 0.949 | 0.117 |
| Day 34 to 41 | 0.675 | 0.731 | 0.663 | 0.769 | 0.022 | 0.547 | <0.001 | 0.263 |
| Day 41 to 48 | 0.587 | 0.626 | 0.604 | 0.678 | 0.019 | 0.078 | 0.006 | 0.366 |
| Phase 1 (day 0 to 20) | 0.693 | 0.666 | 0.716 | 0.673 | 0.032 | 0.579 | 0.186 | 0.766 |
| Phase 2 (day 20 to 48) | 0.649 | 0.673 | 0.666 | 0.717 | 0.012 | 0.016 | 0.003 | 0.287 |
| Overall | 0.660 | 0.673 | 0.679 | 0.708 | 0.012 | 0.023 | 0.079 | 0.495 |
| Mycotoxins (MT) | - | + | p Value | |||||
|---|---|---|---|---|---|---|---|---|
| YCWE (YC) | 0% | 0.2% | 0% | 0.2% | SEM | MT | YC | MT × YC |
| AID, % | ||||||||
| Dry matter | 69.7 | 70.0 | 64.0 | 70.7 | 2.39 | 0.243 | 0.106 | 0.144 |
| Crude protein | 78.5 | 78.9 | 75.5 b | 80.1 a | 1.25 | 0.479 | 0.049 | 0.091 |
| Gross energy | 70.9 AB | 70.9 AB | 65.1 B | 72.6 A | 2.35 | 0.354 | 0.097 | 0.096 |
| Ether extract | 81.8 ab | 80.8 ab | 76.6 b | 83.5 a | 1.88 | 0.483 | 0.113 | 0.032 |
| Mycotoxins (MT) | - | + | p Value | |||||
|---|---|---|---|---|---|---|---|---|
| YCWE (YC) | 0% | 0.2% | 0% | 0.2% | SEM | MT | YC | MT × YC |
| RBC2, 106/µL | ||||||||
| Day 14 | 6.84 | 6.90 | 7.05 | 6.80 | 0.23 | 0.687 | 0.601 | 0.387 |
| Day 45 | 6.86 | 6.30 | 6.91 | 6.68 | 0.17 | 0.194 | 0.023 | 0.313 |
| Hemoglobin, g/dL | ||||||||
| Day 14 | 12.43 | 12.48 | 12.64 | 11.95 | 0.45 | 0.709 | 0.317 | 0.254 |
| Day 45 | 12.11 | 11.51 | 12.53 | 12.18 | 0.27 | 0.052 | 0.086 | 0.659 |
| Hematocrit, % | ||||||||
| Day 14 | 40.91 | 40.63 | 41.05 | 38.79 | 1.38 | 0.495 | 0.189 | 0.317 |
| Day 45 | 41.58 | 38.92 | 42.33 | 40.17 | 1.15 | 0.392 | 0.043 | 0.830 |
| MCV3, fL | ||||||||
| Day 14 | 59.70 | 58.70 | 58.39 | 57.22 | 1.19 | 0.124 | 0.286 | 0.988 |
| Day 45 | 60.50 | 61.75 | 61.25 | 60.25 | 0.85 | 0.660 | 0.883 | 0.192 |
| MCH4, pg | ||||||||
| Day 14 | 18.22 | 18.12 | 17.96 | 17.62 | 0.36 | 0.217 | 0.527 | 0.755 |
| Day 45 | 17.67 | 18.31 | 18.14 | 18.32 | 0.25 | 0.330 | 0.104 | 0.347 |
| MCHC5, g/dL | ||||||||
| Day 14 | 30.47 | 30.81 | 30.79 | 30.79 | 0.21 | 0.492 | 0.420 | 0.412 |
| Day 45 | 29.21 | 29.70 | 29.66 | 30.40 | 0.41 | 0.170 | 0.147 | 0.757 |
| Platelet count, cell/mL | ||||||||
| Day 14 | 185.9 | 144.0 | 133.7 | 152.4 | 27.1 | 0.394 | 0.618 | 0.237 |
| Day 45 | 230.4 | 174.0 | 220.3 | 154.1 | 31.4 | 0.635 | 0.058 | 0.876 |
| WBC6, 103/µL | ||||||||
| Day 14 | 20.52 | 21.43 | 19.91 | 18.83 | 1.36 | 0.236 | 0.950 | 0.470 |
| Day 45 | 18.62 | 16.30 | 18.68 | 16.25 | 1.28 | 0.997 | 0.071 | 0.966 |
| Neutrophils, cell/mL | ||||||||
| Day 14 | 7.89 | 6.66 | 5.89 | 5.77 | 0.72 | 0.003 | 0.139 | 0.225 |
| Day 45 | 3.64 | 4.21 | 3.89 | 3.45 | 0.44 | 0.545 | 0.882 | 0.233 |
| Lymphocytes, cell/mL | ||||||||
| Day 14 | 10.97 | 13.05 | 12.47 | 11.30 | 0.97 | 0.868 | 0.614 | 0.100 |
| Day 45 | 13.33 | 9.96 | 12.96 | 11.37 | 1.19 | 0.662 | 0.042 | 0.452 |
| Monocytes, cell/mL | ||||||||
| Day 14 | 1.04 | 1.13 | 0.96 | 1.13 | 0.15 | 0.777 | 0.397 | 0.781 |
| Day 45 | 1.07 | 1.04 | 1.32 | 0.98 | 0.18 | 0.580 | 0.285 | 0.377 |
| Eosinophils, cell/µL | ||||||||
| Day 14 | 543 | 499 | 518 | 484 | 84 | 0.812 | 0.636 | 0.949 |
| Day 45 | 502 | 451 | 478 | 434 | 78 | 0.786 | 0.529 | 0.954 |
| Mycotoxins (MT) | - | + | p Value | |||||
|---|---|---|---|---|---|---|---|---|
| YCWE (YC) | 0% | 0.2% | 0% | 0.2% | SEM | MT | YC | MT × YC |
| Total protein, g/dL | ||||||||
| Day 14 | 4.65 | 4.64 | 4.67 | 4.57 | 0.09 | 0.742 | 0.542 | 0.606 |
| Day 45 | 5.18 | 5.14 | 5.14 | 4.87 | 0.110 | 0.077 | 0.077 | 0.163 |
| Albumin, g/dL | ||||||||
| Day 14 | 2.65 | 2.58 | 2.70 | 2.69 | 0.06 | 0.164 | 0.506 | 0.605 |
| Day 45 | 3.33 | 2.98 | 7.64 | 5.27 | 1.96 | 0.078 | 0.457 | 0.583 |
| Globulin, g/dL | ||||||||
| Day 14 | 2.00 | 2.06 | 1.97 | 1.82 | 0.08 | 0.071 | 0.539 | 0.167 |
| Day 45 | 1.84 | 3.50 | 3.88 | 1.78 | 1.16 | 0.895 | 0.850 | 0.114 |
| Albumin to globulin ratio | ||||||||
| Day 14 | 1.34 | 1.27 | 1.42 | 1.51 | 0.06 | 0.012 | 0.891 | 0.175 |
| Day 45 | 1.84 a | 1.47 b | 1.62 ab | 1.76 ab | 0.09 | 0.723 | 0.218 | 0.009 |
| AST2, U/L | ||||||||
| Day 14 | 33.92 AB | 38.83 AB | 41.42 A | 32.42 B | 3.69 | 0.884 | 0.583 | 0.067 |
| Day 45 | 21.33 | 23.42 | 31.08 | 23.50 | 4.60 | 0.193 | 0.463 | 0.201 |
| ALT3, U/L | ||||||||
| Day 14 | 22.17 | 25.50 | 22.42 | 24.33 | 1.85 | 0.806 | 0.165 | 0.704 |
| Day 45 | 22.08 | 21.50 | 22.17 | 28.75 | 2.30 | 0.120 | 0.201 | 0.128 |
| ALP4, U/L | ||||||||
| Day 14 | 381 | 352 | 375 | 332 | 21 | 0.535 | 0.091 | 0.718 |
| Day 45 | 311 | 256 | 271 | 278 | 22 | 0.693 | 0.278 | 0.165 |
| CPK5, U/L | ||||||||
| Day 14 | 1230 | 1749 | 1119 | 1358 | 254 | 0.287 | 0.113 | 0.552 |
| Day 45 | 1224 | 1161 | 1931 | 1454 | 403 | 0.223 | 0.508 | 0.610 |
| BUN6, mg/dL | ||||||||
| Day 14 | 15.42 | 17.75 | 16.50 | 17.67 | 1.61 | 0.751 | 0.270 | 0.711 |
| Day 45 | 10.83 | 11.50 | 10.33 | 11.00 | 0.52 | 0.345 | 0.210 | 1.000 |
| Creatinine, mg/dL | ||||||||
| Day 14 | 0.75 | 0.75 | 0.78 | 0.88 | 0.04 | 0.035 | 0.154 | 0.154 |
| Day 45 | 0.88 | 0.89 | 0.89 | 0.82 | 0.04 | 0.351 | 0.351 | 0.245 |
| BUN-to-creatinine ratio | ||||||||
| Day 14 | 20.42 | 24.08 | 21.33 | 20.42 | 2.09 | 0.455 | 0.455 | 0.216 |
| Day 45 | 12.67 | 13.25 | 11.75 | 13.58 | 0.791 | 0.715 | 0.135 | 0.435 |
| Cholesterol, mg/dL | ||||||||
| Day 14 | 86.13 | 85.25 | 79.89 | 74.29 | 3.65 | 0.019 | 0.354 | 0.497 |
| Day 45 | 82.83 ab | 86.25 a | 83.92 ab | 77.08 b | 2.19 | 0.072 | 0.439 | 0.024 |
| Glucose, mg/dL | ||||||||
| Day 14 | 101.3 | 104.3 | 99.1 | 99.4 | 4.0 | 0.377 | 0.680 | 0.741 |
| Day 45 | 110.8 | 113.1 | 112.9 | 113.0 | 3.9 | 0.757 | 0.719 | 0.738 |
| Ca, mg/dL | ||||||||
| Day 14 | 11.64 | 11.12 | 11.30 | 10.79 | 0.25 | 0.153 | 0.030 | 0.971 |
| Day 45 | 11.31 | 10.96 | 11.02 | 11.13 | 0.14 | 0.683 | 0.416 | 0.108 |
| Cl, mEq/L | ||||||||
| Day 14 | 100.6 | 101.5 | 100.0 | 101.1 | 0.86 | 0.384 | 0.081 | 0.807 |
| Day 45 | 101.6 | 102.8 | 101.3 | 102.1 | 0.615 | 0.442 | 0.112 | 0.726 |
| Na, mEq/L | ||||||||
| Day 14 | 144.4 | 143.3 | 145.3 | 143.5 | 0.86 | 0.534 | 0.083 | 0.678 |
| Day 45 | 141.9 | 142.8 | 140.5 | 142.3 | 0.790 | 0.217 | 0.099 | 0.552 |
| K, mEq/L | ||||||||
| Day 14 | 5.28 | 5.35 | 5.06 | 4.95 | 0.22 | 0.064 | 0.914 | 0.566 |
| Day 45 | 5.57 | 6.08 | 5.68 | 5.53 | 0.25 | 0.381 | 0.479 | 0.181 |
| Na-to-K ratio | ||||||||
| Day 14 | 27.63 | 27.75 | 28.96 | 29.90 | 1.06 | 0.053 | 0.553 | 0.652 |
| Day 45 | 26.00 | 24.00 | 25.42 | 26.42 | 1.06 | 0.363 | 0.618 | 0.140 |
| P, mg/dL | ||||||||
| Day 14 | 9.20 | 9.06 | 9.03 | 8.52 | 0.27 | 0.184 | 0.227 | 0.493 |
| Day 45 | 10.76 | 10.91 | 10.55 | 10.54 | 0.24 | 0.246 | 0.773 | 0.748 |
| Mycotoxins (MT) | - | + | p Value | |||||
|---|---|---|---|---|---|---|---|---|
| YCWE (YC) | 0% | 0.2% | 0% | 0.2% | SEM | MT | YC | MT × YC |
| Villus height (VH), μm | 521 | 525 | 509 | 520 | 4.4 | 0.047 | 0.088 | 0.448 |
| Villus width (top), μm | 91 | 89 | 91 | 100 | 3.1 | 0.096 | 0.246 | 0.130 |
| Villus width (middle), μm | 118 | 113 | 111 | 114 | 3.3 | 0.401 | 0.673 | 0.258 |
| Villus width (bottom), µm | 124 | 119 | 121 | 117 | 4.3 | 0.476 | 0.257 | 0.870 |
| Crypt depth (CD), μm | 239 | 239 | 232 | 234 | 4.3 | 0.101 | 0.764 | 0.820 |
| VH-to-CD ratio2 | 2.19 | 2.20 | 2.20 | 2.23 | 0.04 | 0.759 | 0.632 | 0.788 |
| Ki-67, % | 26.0 | 28.7 | 22.0 | 26.5 | 2.01 | 0.091 | 0.052 | 0.646 |
| Mycotoxins (MT) | - | + | p Value | |||||
|---|---|---|---|---|---|---|---|---|
| YCWE (YC) | 0% | 0.2% | 0% | 0.2% | SEM | MT | YC | MT × YC |
| Tumor necrosis factor-α | ||||||||
| Serum at day 14, pg/mL | 124 A | 124 A | 151 B | 127 A | 6.8 | 0.031 | 0.084 | 0.083 |
| Serum at day 45, pg/mL | 61 | 61 | 68 | 61 | 2.5 | 0.180 | 0.115 | 0.147 |
| Jejunal mucosa, ng/mg protein | 708 | 668 | 730 | 562 | 58 | 0.429 | 0.055 | 0.233 |
| Immunoglobulin A | ||||||||
| Serum at day 14, mg/mL | 0.42 | 0.33 | 0.41 | 0.37 | 0.05 | 0.701 | 0.183 | 0.554 |
| Serum at day 45, mg/mL | 0.57 | 0.55 | 0.67 | 0.61 | 0.06 | 0.187 | 0.515 | 0.752 |
| Jejunal mucosa, μg/mg protein | 5.83 | 5.57 | 6.85 | 6.12 | 0.24 | 0.002 | 0.037 | 0.318 |
| Immunoglobulin G | ||||||||
| Serum at day 14, mg/mL | 3.19 | 3.59 | 3.33 | 3.05 | 0.30 | 0.469 | 0.831 | 0.226 |
| Serum at day 45, mg/mL | 2.62 AB | 2.83 AB | 3.04 A | 2.51 B | 0.21 | 0.787 | 0.418 | 0.057 |
| Jejunal mucosa, μg/mg protein | 1.04 | 1.42 | 1.11 | 1.02 | 0.19 | 0.379 | 0.441 | 0.208 |
| Protein carbonyl | ||||||||
| Serum at day 14, nmol/mg protein | 1.91 | 2.00 | 2.16 | 1.85 | 0.15 | 0.730 | 0.458 | 0.189 |
| Serum at day 45, nmol/mg protein | 1.76 | 2.01 | 2.02 | 1.87 | 0.12 | 0.605 | 0.668 | 0.108 |
| Jejunal mucosa, nmol/mg protein | 2.33 | 2.31 | 2.83 a | 2.51 b | 0.08 | 0.001 | 0.026 | 0.047 |
| Malondialdehydes | ||||||||
| Serum at day 14, μM | 8.90 | 9.49 | 11.17 | 9.91 | 1.23 | 0.255 | 0.774 | 0.429 |
| Serum at day 45, μM | 8.89 | 9.57 | 10.25 | 9.83 | 1.43 | 0.542 | 0.922 | 0.682 |
| Jejunal mucosa, nmol/g protein | 511 | 542 | 488 | 579 | 78 | 0.925 | 0.401 | 0.679 |
| Mycotoxins (MT) | - | + | p Value | |||||
|---|---|---|---|---|---|---|---|---|
| YCWE (YC) | 0% | 0.2% | 0% | 0.2% | SEM | MT | YC | MT × YC |
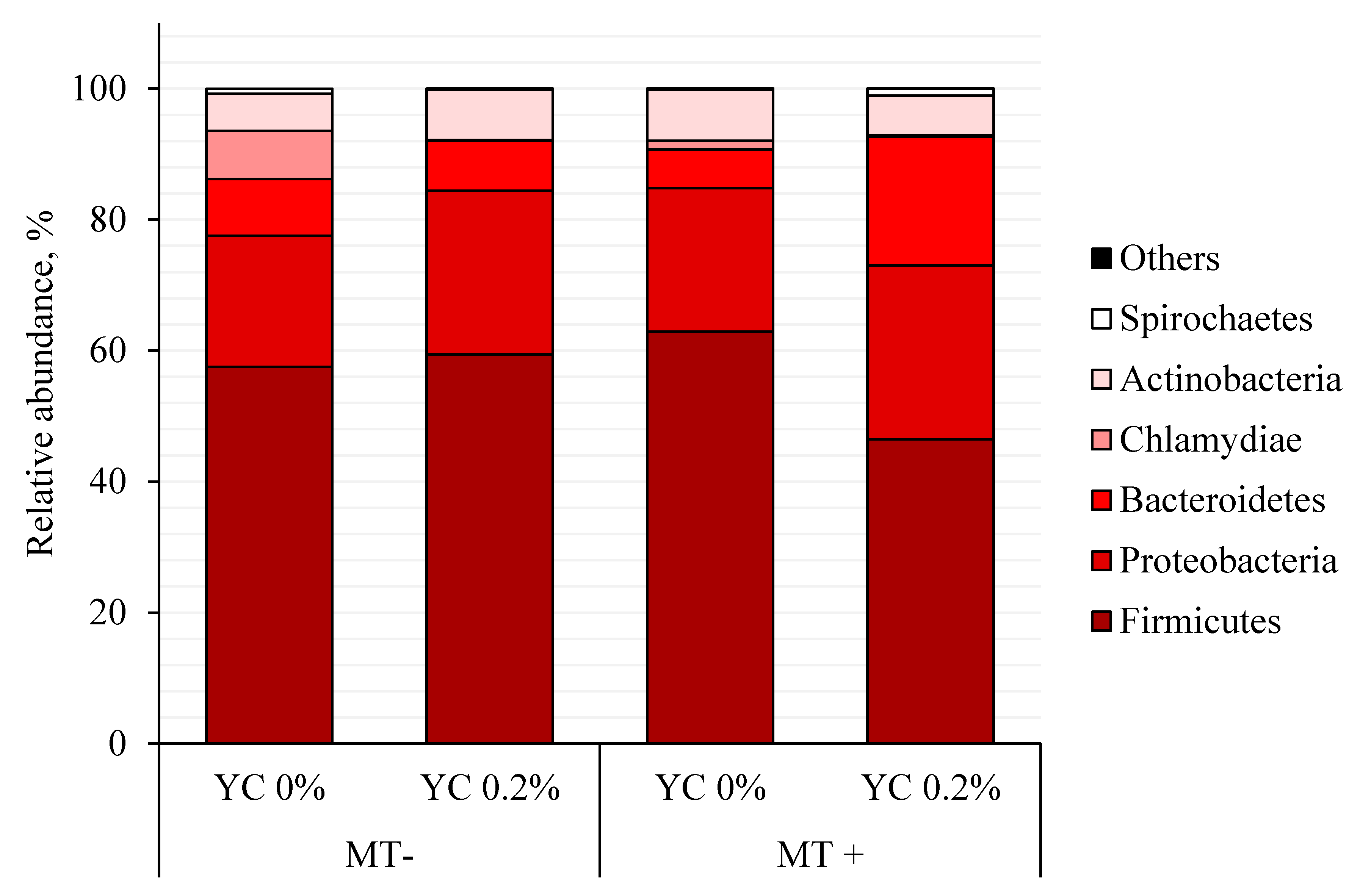
| Actinobacteria | 5.65 | 7.69 | 7.71 | 6.01 | 1.90 | 0.915 | 0.922 | 0.288 |
| Bacteroidetes | 8.64 ab | 7.66 ab | 5.93 b | 19.63 a | 3.52 | 0.182 | 0.069 | 0.037 |
| Firmicutes | 57.49 | 59.40 | 62.92 | 46.44 | 6.68 | 0.514 | 0.210 | 0.116 |
| Proteobacteria | 20.06 | 25.01 | 21.88 | 26.57 | 4.90 | 0.733 | 0.331 | 0.979 |
| Spirochaetes | 0.74 AB | 0.16 AB | 0.10 A | 1.07 B | 0.45 | 0.737 | 0.632 | 0.062 |
| Chlamydiae | 7.37 | 0.08 | 1.34 | 0.25 | 2.80 | 0.302 | 0.142 | 0.275 |
| Deinococcus-Thermus | 0.00 | 0.00 | 0.11 | 0.00 | 0.06 | 0.324 | 0.324 | 0.324 |
| Fusobacteria | 0.02 | 0.00 | 0.00 | 0.00 | 0.01 | 0.324 | 0.324 | 0.324 |
| Nitrospirae | 0.01 | 0.00 | 0.00 | 0.03 | 0.01 | 0.467 | 0.467 | 0.231 |
| Tenericutes | 0.01 | 0.00 | 0.00 | 0.01 | 0.01 | 0.965 | 0.965 | 0.161 |
| Verrucomicrobia | 0.00 | 0.00 | 0.01 | 0.00 | 0.01 | 0.324 | 0.324 | 0.324 |
| Mycotoxins (MT) | - | + | p Value | |||||
|---|---|---|---|---|---|---|---|---|
| YCWE (YC) | 0% | 0.2% | 0% | 0.2% | SEM | MT | YC | MT × YC |
| Lactobacillaceae | 13.95 | 20.02 | 32.20 | 35.86 | 6.68 | 0.011 | 0.451 | 0.851 |
| Lactobacillus | 13.95 | 20.02 | 32.20 | 35.86 | 6.68 | 0.011 | 0.451 | 0.851 |
| Clostridiaceae | 12.94 | 7.53 | 7.81 | 11.60 | 3.99 | 0.895 | 0.841 | 0.256 |
| Clostridium | 12.94 | 7.53 | 7.81 | 11.60 | 3.99 | 0.895 | 0.841 | 0.256 |
| Prevotellaceae | 14.84 | 7.77 | 11.38 | 3.73 | 3.78 | 0.295 | 0.044 | 0.935 |
| Prevotella | 14.84 | 7.77 | 11.38 | 3.73 | 3.78 | 0.295 | 0.044 | 0.935 |
| Veillonellaceae | 3.81 | 4.32 | 4.18 | 4.98 | 1.66 | 0.758 | 0.696 | 0.929 |
| Dialister | 0.10 | 0.31 | 0.22 | 0.13 | 0.10 | 0.762 | 0.576 | 0.129 |
| Mitsuokella | 0.52 | 1.56 | 1.75 | 2.75 | 0.91 | 0.192 | 0.271 | 0.981 |
| Ruminococcaceae | 2.37 | 3.54 | 2.65 | 4.57 | 2.03 | 0.707 | 0.379 | 0.831 |
| Faecalibacterium | 0.52 | 2.23 | 1.03 | 3.29 | 1.68 | 0.581 | 0.169 | 0.849 |
| Ruminococcus | 1.85 | 1.31 | 1.62 | 1.28 | 0.62 | 0.830 | 0.471 | 0.870 |
| Propionibacteriaceae | 2.46 | 5.03 | 2.59 | 2.86 | 0.93 | 0.249 | 0.110 | 0.194 |
| Propionibacterium | 0.02 | 0.00 | 0.01 | 0.00 | 0.01 | 0.542 | 0.215 | 0.542 |
| Helicobacteraceae | 2.50 | 5.10 | 0.80 | 3.92 | 2.90 | 0.623 | 0.329 | 0.930 |
| Helicobacter | 2.50 | 5.10 | 0.80 | 3.92 | 2.90 | 0.623 | 0.329 | 0.930 |
| Bacillaceae | 2.33 | 4.13 | 2.97 | 2.76 | 1.78 | 0.835 | 0.658 | 0.575 |
| Anoxybacillus | 0.50 | 1.63 | 1.32 | 0.12 | 0.69 | 0.615 | 0.957 | 0.097 |
| Bacillus | 1.65 | 2.35 | 1.55 | 2.46 | 1.66 | 0.999 | 0.631 | 0.949 |
| Moraxellaceae | 1.89 | 2.01 | 2.57 | 4.44 | 1.16 | 0.188 | 0.397 | 0.453 |
| Acinetobacter | 1.89 | 2.01 | 2.57 | 4.44 | 1.16 | 0.188 | 0.397 | 0.453 |
| Lachnospiraceae | 2.78 | 2.28 | 3.37 | 1.97 | 0.89 | 0.871 | 0.267 | 0.601 |
| Roseburia | 1.31 | 1.24 | 0.88 | 0.99 | 0.54 | 0.533 | 0.973 | 0.868 |
| Oxalobacteraceae | 1.06 | 0.25 | 0.16 | 0.34 | 0.34 | 0.244 | 0.365 | 0.160 |
| Massilia | 0.00 | 0.00 | 0.01 | 0.00 | 0.00 | 0.324 | 0.324 | 0.324 |
| Enterobacteriaceae | 3.65 | 0.83 | 1.42 | 2.90 | 1.29 | 0.919 | 0.632 | 0.097 |
| Leclercia | 0.12 | 0.00 | 0.00 | 0.00 | 0.06 | 0.324 | 0.324 | 0.324 |
| Proteus | 0.38 | 0.00 | 0.01 | 0.2 | 0.15 | 0.233 | 0.217 | 0.187 |
| Trabulsiella | 0.03 | 0.00 | 0.00 | 0.00 | 0.01 | 0.175 | 0.175 | 0.175 |
| Turicibacter | 0.34 | 0.19 | 1.35 | 0.38 | 0.52 | 0.125 | 0.144 | 0.241 |
| Chlamydiaceae | 0.04 | 8.24 | 0.70 | 0.05 | 2.79 | 0.185 | 0.184 | 0.121 |
| Chlamydia | 0.04 | 8.24 | 0.70 | 0.05 | 2.79 | 0.185 | 0.184 | 0.121 |
| Staphylococcaceae | 1.17 | 5.08 | 0.81 | 1.91 | 2.13 | 0.413 | 0.247 | 0.513 |
| Staphylococcus | 0.01 | 0.10 | 0.02 | 0.05 | 0.06 | 0.742 | 0.319 | 0.627 |
| Pseudomonadaceae | 0.68 | 3.19 | 1.61 | 1.63 | 1.04 | 0.752 | 0.212 | 0.216 |
| Pseudomonas | 0.58 | 2.96 | 1.52 | 1.43 | 1.03 | 0.760 | 0.247 | 0.212 |
| Erysipelotrichaceae | 2.72 | 0.45 | 2.93 | 0.98 | 0.92 | 0.665 | 0.017 | 0.853 |
| Streptococcaceae | 1.72 | 0.88 | 2.85 | 1.07 | 1.02 | 0.481 | 0.166 | 0.615 |
| Streptococcus | 1.72 | 0.88 | 2.85 | 1.07 | 1.02 | 0.481 | 0.166 | 0.615 |
| Paenibacillaceae | 6.17 | 0.75 | 0.17 | 0.15 | 2.08 | 0.121 | 0.198 | 0.201 |
| Succinivibrionaceae | 1.65 | 2.04 | 1.00 | 1.16 | 0.62 | 0.206 | 0.642 | 0.841 |
| Succinivibrio | 1.65 | 2.04 | 1.00 | 1.16 | 0.62 | 0.206 | 0.642 | 0.841 |
| Xanthomonadaceae | 3.88 | 0.20 | 0.35 | 0.53 | 1.79 | 0.369 | 0.326 | 0.279 |
| Stenotrophomonas | 0.04 | 0.00 | 0.00 | 0.03 | 0.02 | 0.673 | 0.673 | 0.121 |
| Others2 | 17.44 | 16.36 | 17.60 | 12.62 | 3.74 | 0.634 | 0.423 | 0.604 |
| Mycotoxins (MT) | - | + | p Value | |||||
|---|---|---|---|---|---|---|---|---|
| YCWE (YC) | 0% | 0.2% | 0% | 0.2% | SEM | MT | YC | MT × YC |
| Lactobacillus mucosae | 8.18 | 16.63 | 7.17 | 9.19 | 3.83 | 0.259 | 0.164 | 0.389 |
| Prevotella copri | 2.49 | 8.85 | 5.62 | 11.62 | 3.02 | 0.306 | 0.036 | 0.951 |
| Lactobacillus kitasatonis | 17.58 | 5.50 | 3.01 | 1.06 | 4.51 | 0.031 | 0.107 | 0.241 |
| Clostridium perfringens | 7.07 | 3.18 | 5.34 | 8.56 | 3.82 | 0.636 | 0.930 | 0.358 |
| Propionibacterium acnes | 2.85 | 2.55 | 5.00 | 2.43 | 0.92 | 0.245 | 0.104 | 0.197 |
| Lactobacillus delbrueckii | 2.35 | 2.64 | 2.57 | 1.59 | 0.50 | 0.386 | 0.476 | 0.190 |
| Chlamydia suis | 0.05 | 0.70 | 8.22 | 0.04 | 2.78 | 0.184 | 0.184 | 0.120 |
| Lactobacillus sp. | 2.04 | 2.38 | 2.01 | 1.19 | 0.49 | 0.159 | 0.582 | 0.179 |
| Lactobacillus equicursoris | 2.99 | 0.37 | 3.90 | 0.13 | 1.49 | 0.820 | 0.039 | 0.701 |
| Clostridium butyricum | 2.15 | 2.77 | 1.00 | 1.05 | 0.93 | 0.133 | 0.725 | 0.759 |
| Dialister succinatiphilus | 2.75 | 1.75 | 1.56 | 0.52 | 0.91 | 0.192 | 0.271 | 0.981 |
| Faecalibacterium prausnitzii | 1.28 | 1.62 | 1.31 | 1.85 | 0.62 | 0.830 | 0.471 | 0.870 |
| Succinivibrio dextrinosolvens | 1.16 | 1.00 | 2.04 | 1.65 | 0.62 | 0.206 | 0.642 | 0.841 |
| Massilia niabensis | 1.51 | 1.60 | 1.49 | 1.23 | 0.56 | 0.679 | 0.858 | 0.715 |
| Acinetobacter radioresistens | 2.43 | 1.49 | 0.94 | 0.72 | 0.75 | 0.139 | 0.441 | 0.631 |
| Prevotella stercorea | 0.70 | 1.95 | 0.89 | 2.03 | 0.58 | 0.813 | 0.047 | 0.921 |
| Streptococcus hyointestinalis | 0.90 | 2.04 | 0.51 | 1.52 | 0.96 | 0.597 | 0.217 | 0.938 |
| Mitsuokella jalaludinii | 0.95 | 1.02 | 1.25 | 1.57 | 0.52 | 0.414 | 0.713 | 0.808 |
| Ruminococcus gauvreauii | 2.22 | 0.29 | 1.88 | 0.38 | 1.42 | 0.917 | 0.161 | 0.859 |
| Helicobacter equorum | 0.08 | 0.00 | 2.94 | 1.65 | 1.66 | 0.182 | 0.681 | 0.718 |
| Staphylococcus sciuri | 0.00 | 0.67 | 3.52 | 0.22 | 1.79 | 0.396 | 0.465 | 0.273 |
| Turicibacter sanguinis | 0.59 | 1.18 | 0.26 | 2.37 | 0.71 | 0.544 | 0.064 | 0.286 |
| Stenotrophomonas rhizophila | 0.24 | 0.24 | 0.10 | 3.73 | 1.77 | 0.349 | 0.311 | 0.312 |
| Helicobacter mastomyrinus | 2.22 | 0.41 | 1.25 | 0.41 | 1.04 | 0.642 | 0.207 | 0.643 |
| Mitsuokella multacida | 0.99 | 0.83 | 0.96 | 1.28 | 0.55 | 0.696 | 0.875 | 0.659 |
| Leclercia adecarboxylata | 1.65 | 0.73 | 0.48 | 0.64 | 0.40 | 0.064 | 0.257 | 0.106 |
| Prevotella sp. | 0.49 | 0.54 | 1.17 | 1.17 | 0.54 | 0.160 | 0.965 | 0.958 |
| Helicobacter rappini | 1.61 | 0.39 | 0.90 | 0.39 | 0.82 | 0.665 | 0.296 | 0.659 |
| Bacillus coagulans | 1.35 | 0.81 | 0.18 | 0.51 | 0.68 | 0.285 | 0.875 | 0.529 |
| Clostridium sp. | 0.21 | 0.41 | 0.19 | 1.74 | 0.48 | 0.182 | 0.075 | 0.164 |
| Eubacterium multiforme | 0.40 | 0.22 | 0.79 | 1.10 | 0.45 | 0.170 | 0.891 | 0.593 |
| Roseburia faecis | 0.46 | 0.70 | 0.65 | 0.68 | 0.34 | 0.806 | 0.684 | 0.749 |
| Trabulsiella odontotermitis | 1.11 | 0.53 | 0.33 | 0.40 | 0.28 | 0.067 | 0.292 | 0.190 |
| Clostridium hiranonis | 0.18 | 0.13 | 0.85 | 1.11 | 0.56 | 0.118 | 0.839 | 0.771 |
| Eubacterium biforme | 0.34 | 1.37 | 0.18 | 0.30 | 0.40 | 0.124 | 0.148 | 0.255 |
| Anoxybacillus kestanbolensis | 0.06 | 0.97 | 1.06 | 0.08 | 0.48 | 0.913 | 0.945 | 0.056 |
| Lactobacillus johnsonii | 0.52 | 0.96 | 0.38 | 0.17 | 0.32 | 0.324 | 0.324 | 0.324 |
| Experiment | 1 | 2 | ||||||
|---|---|---|---|---|---|---|---|---|
| Treatments | MT- YC 0% | MT- YC 0.2% | MT+ YC 0% | MT+ YC 0.2% | MT- YC 0% | MT- YC 0.2% | MT+ YC 0% | MT+ YC 0.2% |
| Factor | ||||||||
| Mycotoxin (MT) | - | - | + | + | - | - | + | + |
| YCWE1 (YC) | - | + | - | + | - | + | - | + |
| Pigs | ||||||||
| Per treatment | 30 | 30 | 30 | 30 | 12 | 12 | 12 | 12 |
| Per pen | 3 | 3 | 3 | 3 | 1 | 1 | 1 | 1 |
| Period, d | 35 | 35 | 35 | 35 | 48 | 48 | 48 | 48 |
| Feedstuff | ||||||||
| Ground yellow corn | ||||||||
| Aflatoxins, mg/kg | ND | ND | 2.8 | 2.8 | ND | ND | 2.8 | 2.8 |
| Fumonisins, mg/kg | ND | ND | 170.2 | 170.2 | ND | ND | 170.2 | 170.2 |
| Zearalenone, mg/kg | ND | ND | 1.1 | 1.1 | ND | ND | 1.1 | 1.1 |
| Wheat, soft red | ||||||||
| Deoxynivalenol, mg/kg | - | - | - | - | ND | ND | 7.3 | 7.3 |
| Zearalenone, mg/kg | - | - | - | - | ND | ND | 1.8 | 1.8 |
| Diet2 | ||||||||
| YCWE, % | - | 0.2 | - | 0.2 | - | 0.2 | - | 0.2 |
| Aflatoxin B1, μg/kg | - | - | 180 | 180 | - | - | 180 | 180 |
| Fumonisin B1, mg/kg | - | - | 14 | 14 | - | - | 9 | 9 |
| Deoxynivalenol, mg/kg | - | - | - | - | - | - | 1 | 1 |
| Item | Basal Diet |
|---|---|
| Ingredients, % | |
| Ground yellow corn | 75.60 |
| Soybean meal, dehulled | 21.00 |
| L-Lys HCl | 0.18 |
| Poultry fat | 1.00 |
| Salt | 0.22 |
| Vitamin premix2 | 0.03 |
| Trace mineral premix3 | 0.15 |
| Dicalcium P | 1.12 |
| Ground limestone | 0.70 |
| Calculated composition | |
| Dry matter, % | 89.06 |
| Metabolizable energy, Mcal/kg | 3.35 |
| Crude protein, % | 16.49 |
| Standardized ileal digestible Lys, % | 0.85 |
| Ca, % | 0.60 |
| Available P, % | 0.27 |
| Item | Phase 1 | Phase 2 |
|---|---|---|
| Ingredient, % | ||
| Ground yellow corn | 37.5 | 48.82 |
| Soybean meal, dehulled | 22.0 | 27.0 |
| Wheat, soft red | 15.0 | 15.0 |
| Whey permeate | 12.0 | 2.0 |
| Poultry meal | 5.0 | 3.0 |
| Blood plasma | 3.3 | -- |
| L-Lys HCl | 0.45 | 0.41 |
| DL-Met | 0.17 | 0.12 |
| L-Thr | 0.13 | 0.12 |
| Salt | 0.22 | 0.22 |
| Vitamin and mineral premix2 | 0.18 | 0.18 |
| Dicalcium P | 0.45 | 0.79 |
| Ground limestone | 1.10 | 0.84 |
| Poultry fat | 2.5 | 1.50 |
| Calculated composition: | ||
| Metabolizable energy, Mcal/kg | 3.4 | 3.4 |
| Crude protein, % | 22.09 | 21.12 |
| Standardized ileal digestible Lys, % | 1.35 | 1.23 |
| Standardized ileal digestible Met + Cys | 0.75 | 0.68 |
| Standardized ileal digestible Thr, % | 0.79 | 0.73 |
| Standardized ileal digestible Trp, % | 0.23 | 0.22 |
| Standardized total tract digestible P, % | 0.40 | 0.33 |
| Mycotoxin | Corn | Wheat |
|---|---|---|
| Aflatoxin B1, mg/kg | 2.5 | ND |
| Aflatoxin B2, mg/kg | 0.1 | ND |
| Aflatoxin G1, mg/kg | 0.2 | ND |
| Deoxynivalenol (DON), mg/kg | ND | 5.5 |
| DON-3-glucoside, mg/kg | ND | 0.9 |
| 15-acetyl-DON, mg/kg | ND | 0.5 |
| 3-acetyl- DON, mg/kg | ND | 0.3 |
| Fumonisin B1, mg/kg | 142.9 | 0.1 |
| Fumonisin B2, mg/kg | 13.9 | ND |
| Fumonisin B3, mg/kg | 13.4 | ND |
| Fusarenon X, mg/kg | ND | ND |
| Gliotoxin, mg/kg | ND | ND |
| Neosolaniol, mg/kg | ND | ND |
| Nivalenol, mg/kg | 0.2 | ND |
| Ochratoxin A, mg/kg | 0.1 | ND |
| Ochratoxin B, mg/kg | ND | ND |
| Zearalenone, mg/kg | 1.1 | 1.8 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, S.W.; Holanda, D.M.; Gao, X.; Park, I.; Yiannikouris, A. Efficacy of a Yeast Cell Wall Extract to Mitigate the Effect of Naturally Co-Occurring Mycotoxins Contaminating Feed Ingredients Fed to Young Pigs: Impact on Gut Health, Microbiome, and Growth. Toxins 2019, 11, 633. https://doi.org/10.3390/toxins11110633
Kim SW, Holanda DM, Gao X, Park I, Yiannikouris A. Efficacy of a Yeast Cell Wall Extract to Mitigate the Effect of Naturally Co-Occurring Mycotoxins Contaminating Feed Ingredients Fed to Young Pigs: Impact on Gut Health, Microbiome, and Growth. Toxins. 2019; 11(11):633. https://doi.org/10.3390/toxins11110633
Chicago/Turabian StyleKim, Sung Woo, Débora Muratori Holanda, Xin Gao, Inkyung Park, and Alexandros Yiannikouris. 2019. "Efficacy of a Yeast Cell Wall Extract to Mitigate the Effect of Naturally Co-Occurring Mycotoxins Contaminating Feed Ingredients Fed to Young Pigs: Impact on Gut Health, Microbiome, and Growth" Toxins 11, no. 11: 633. https://doi.org/10.3390/toxins11110633
APA StyleKim, S. W., Holanda, D. M., Gao, X., Park, I., & Yiannikouris, A. (2019). Efficacy of a Yeast Cell Wall Extract to Mitigate the Effect of Naturally Co-Occurring Mycotoxins Contaminating Feed Ingredients Fed to Young Pigs: Impact on Gut Health, Microbiome, and Growth. Toxins, 11(11), 633. https://doi.org/10.3390/toxins11110633





