Osmotic-Adaptation Response of sakA/hogA Gene to Aflatoxin Biosynthesis, Morphology Development and Pathogenicity in Aspergillus flavus
Abstract
:1. Introduction
2. Results
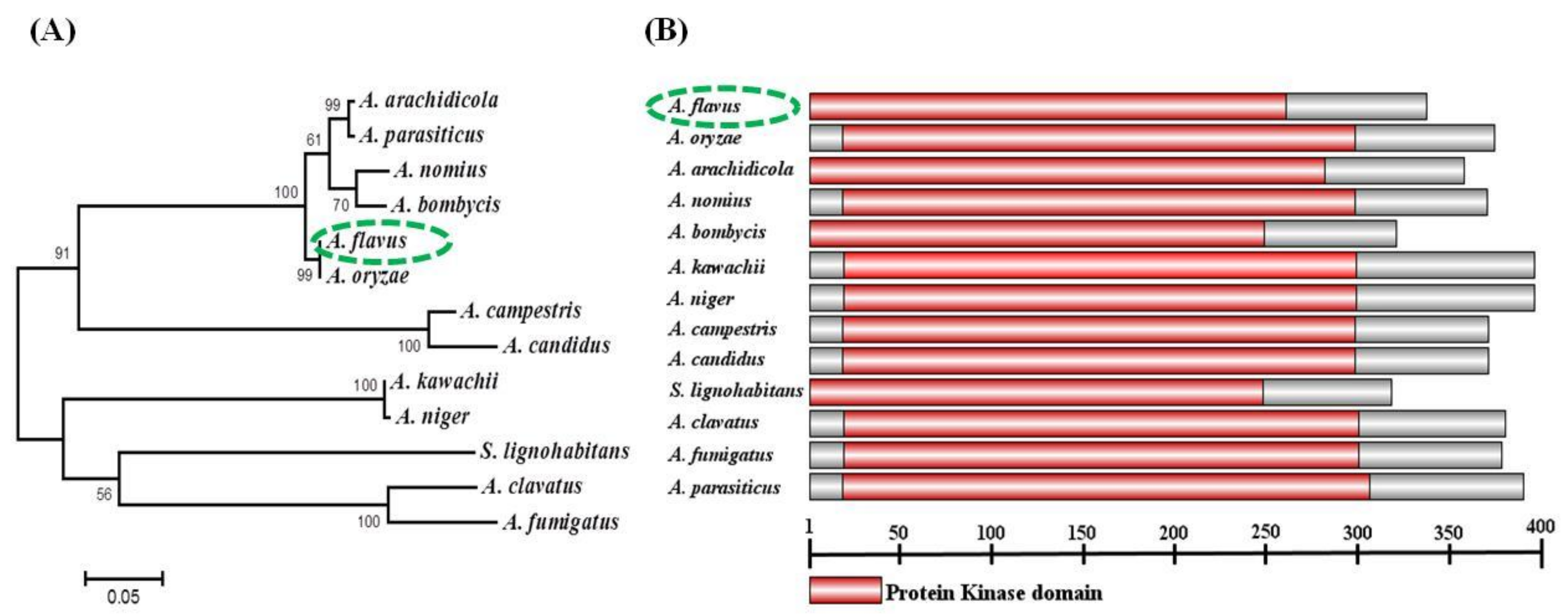
2.1. Identification and Analysis of sakA in A. flavus
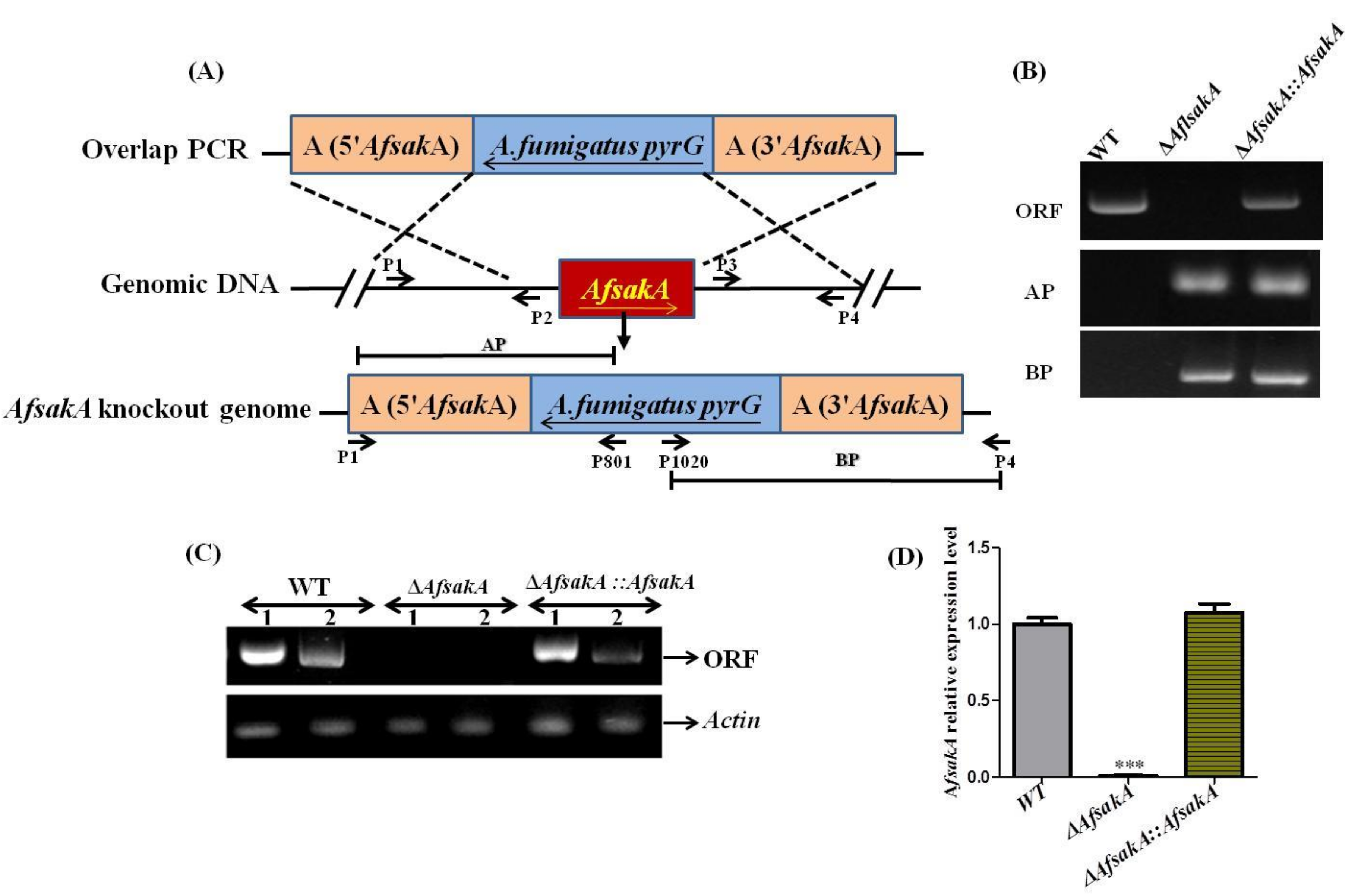
2.2. Generation of AfsakA Deletion and Complementation Mutant Strains
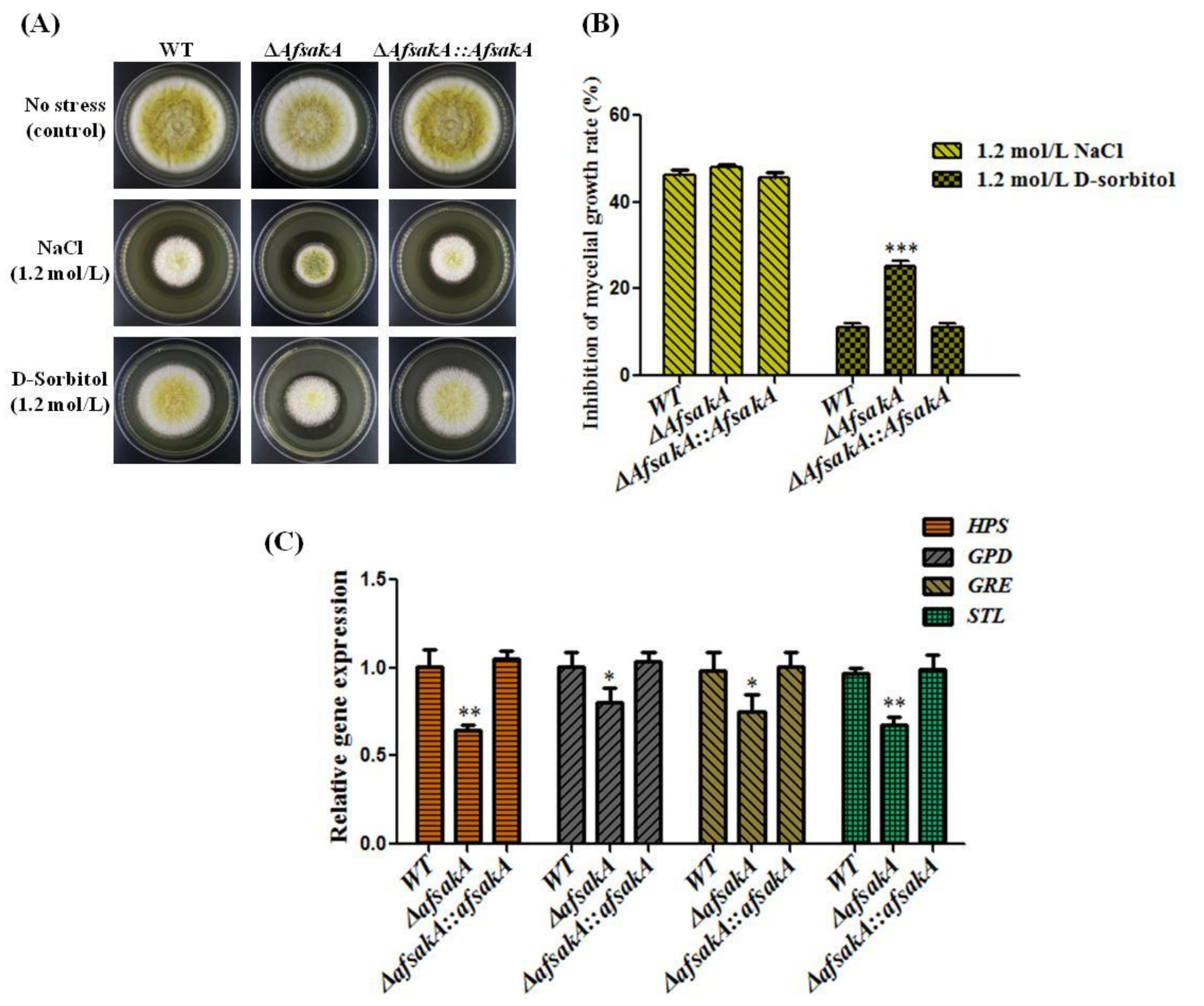
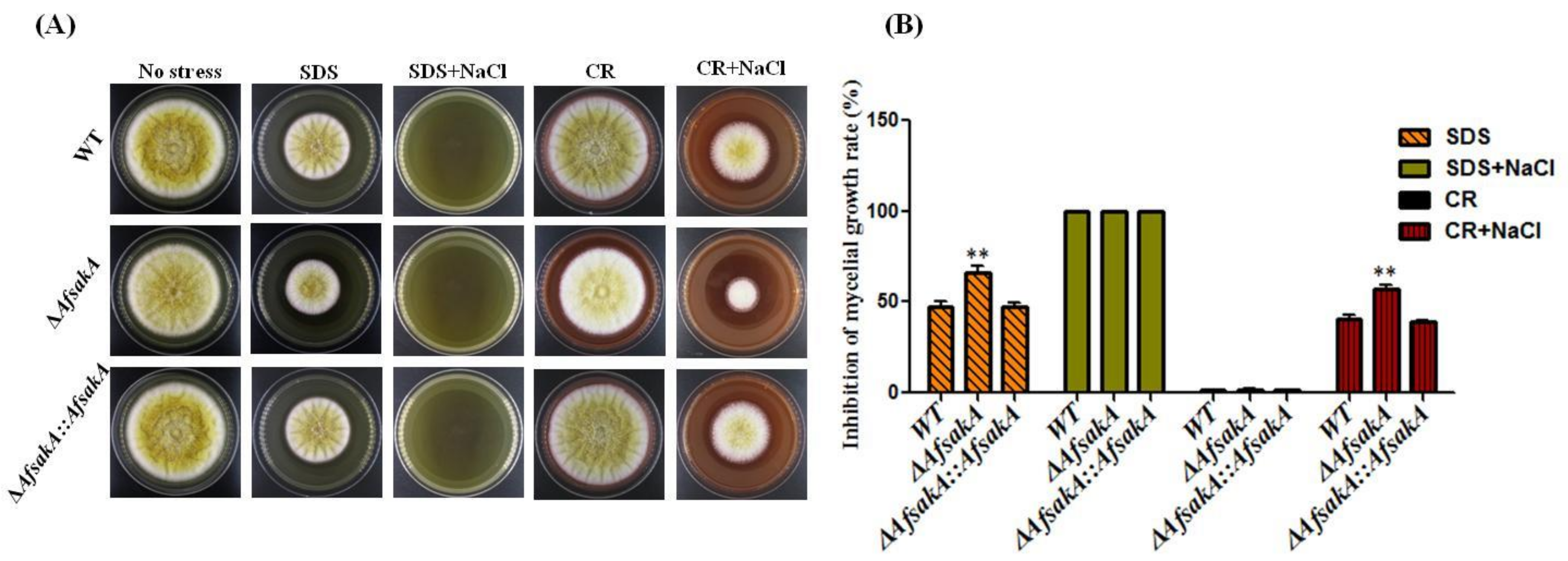
2.3. Effects of AfsakA Deletion on Osmotic Stress Response in A. flavus
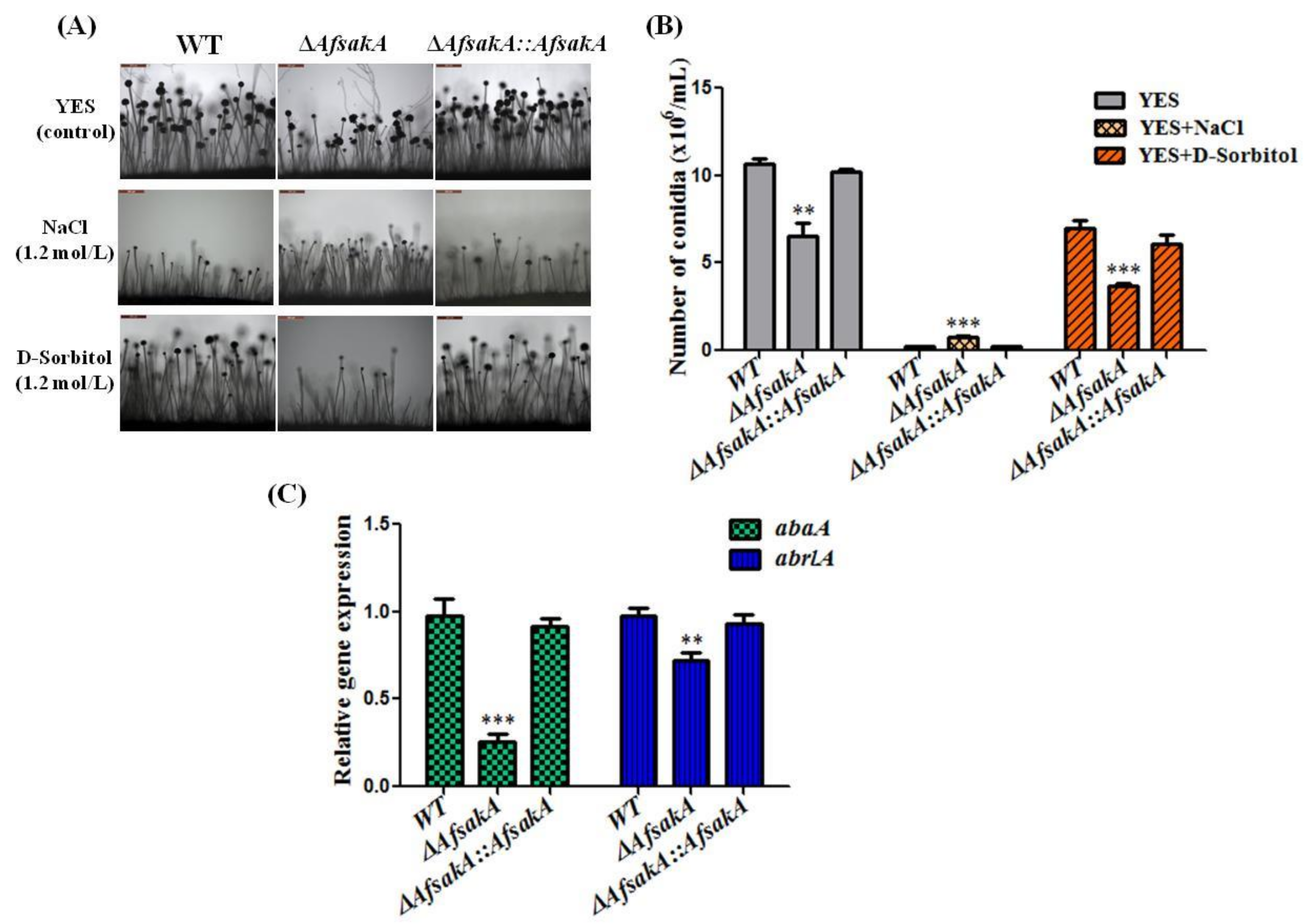
2.4. Effects of AfsakA Deletion on Conidial Production in A. flavus
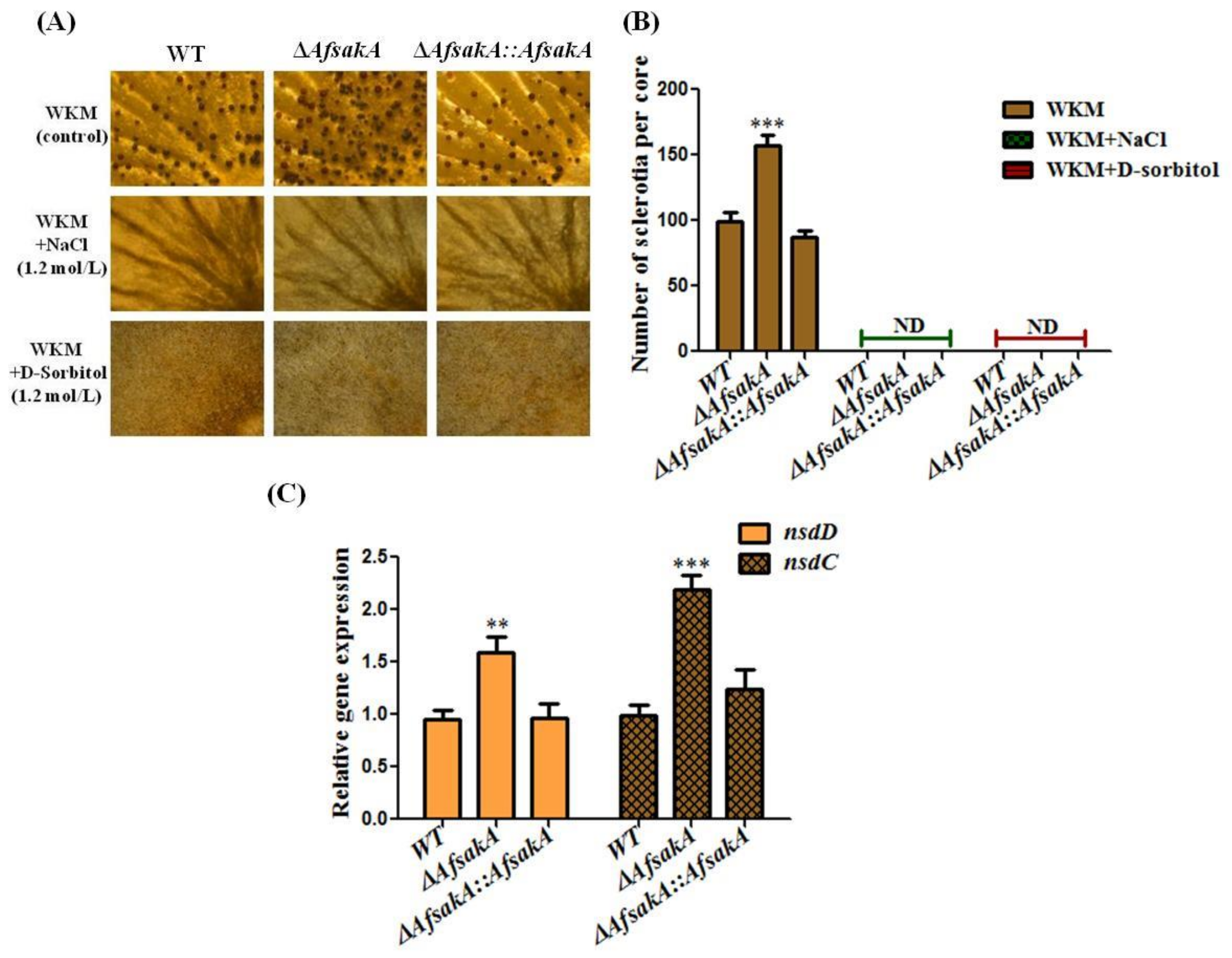
2.5. Effects of AfsakA Deletion on Sclerotia Production in A. flavus
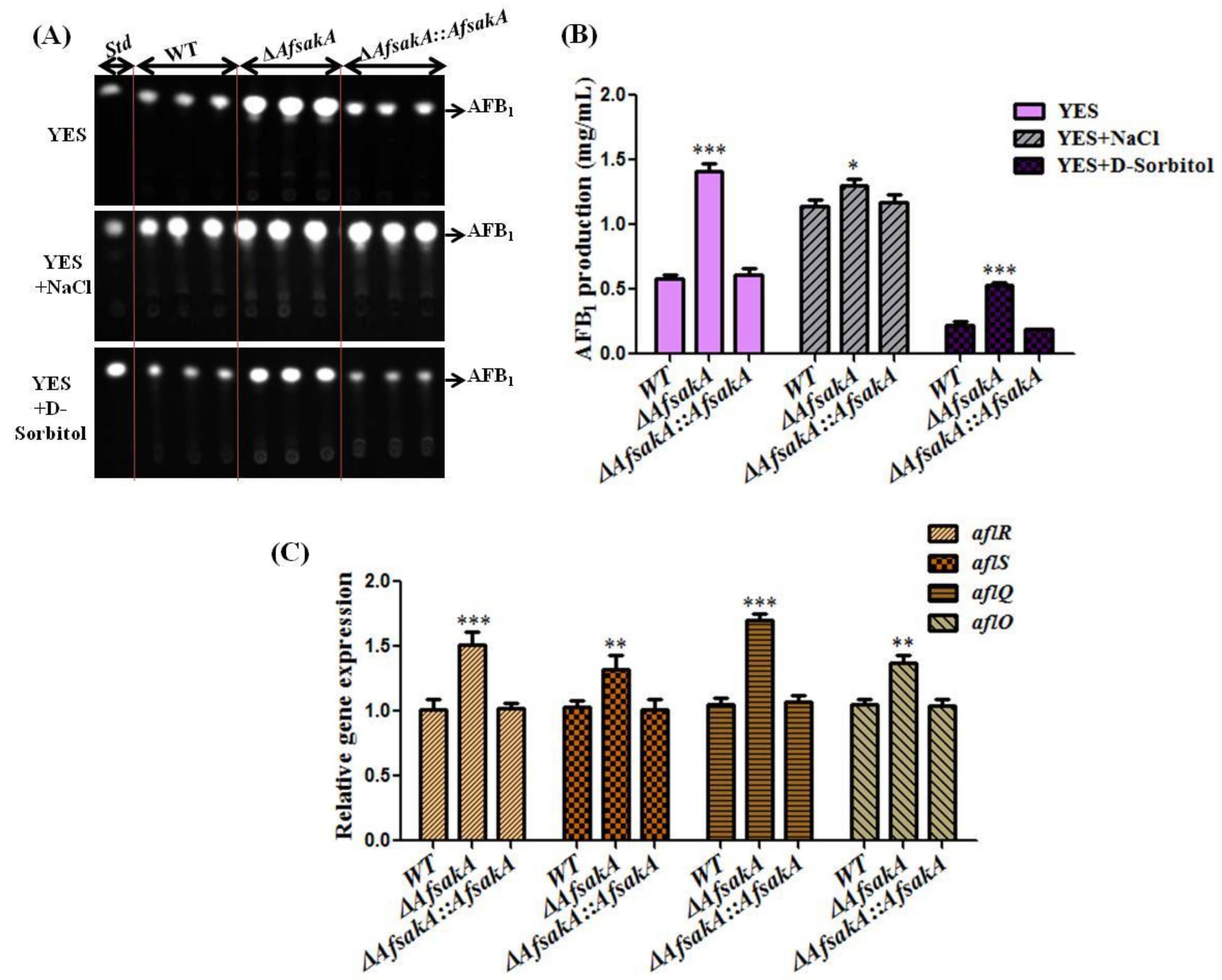
2.6. AfsakA Plays a Negative Role in Regulating AFB1 Biosynthesis in A. flavus
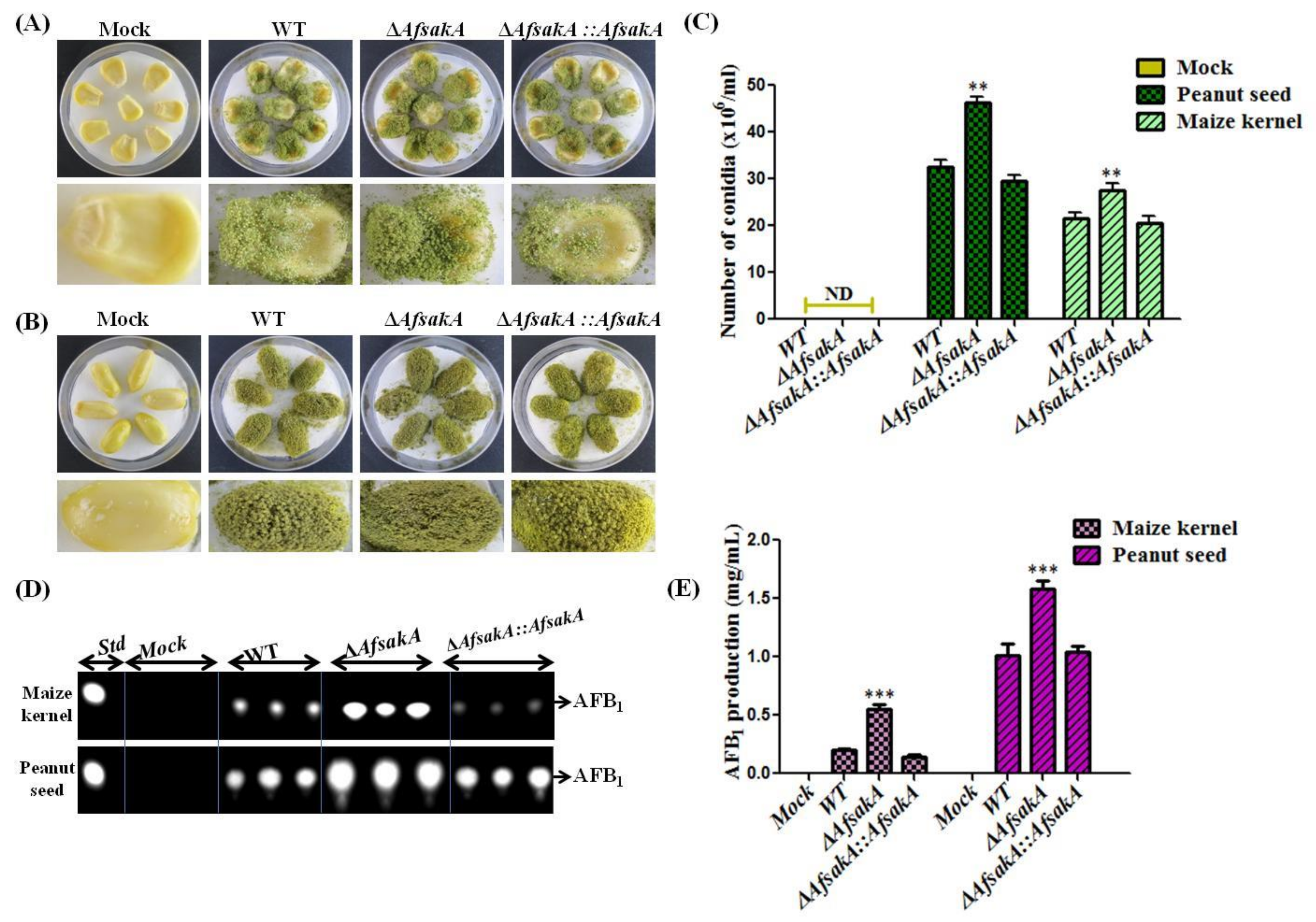
2.7. AfsakA Has an Influence on Virulence to Crop Seeds
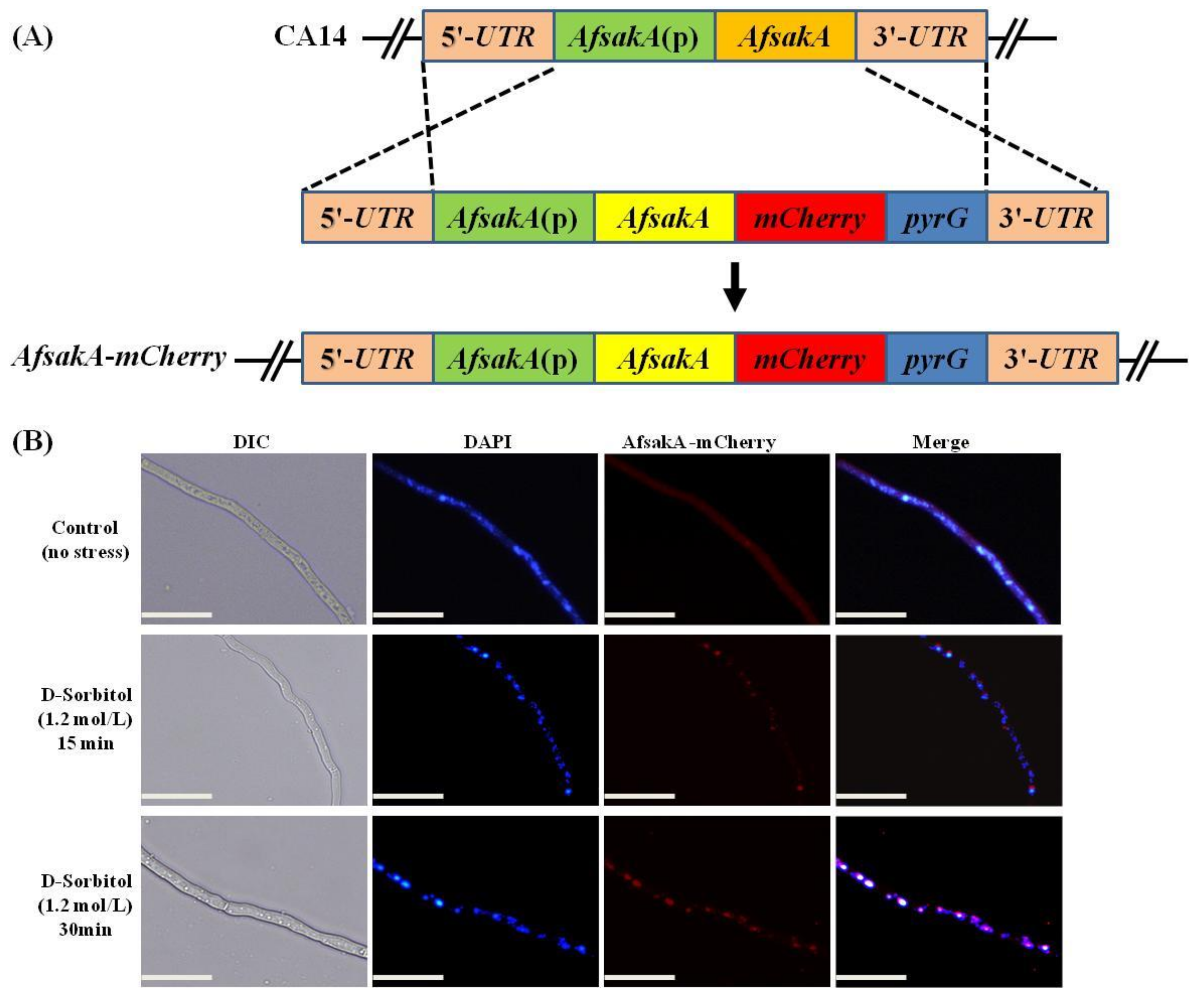
2.8. Subcellular Localization of AfSakA in A. flavus
2.9. Effects of AfsakA Deletion on Growth and Sensitivity to Stress
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Fungal Strains and Culture Conditions
5.2. Sequence and Phylogenetic Tree Analysis
5.3. Construction of AfsakA Deletion and Complementation Strains
5.4. Stress Assay
5.5. Morphological Analysis
5.6. Determination of AFB1 Production
5.7. Localization of AfsakA gene in A. flavus
5.8. Pathogenicity Test
5.9. Reverse Transcriptase Polymerase Chain Reaction (RT-PCR) and Quantitative Real Time Polymerase Chain Reaction (qRT-PCR)
5.10. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Krishnan, S.; Manavathu, E.K.; Chandrasekar, P.H. Aspergillus flavus: An emerging non-fumigatus Aspergillus species of significance. Mycoses 2009, 52, 206–222. [Google Scholar] [CrossRef] [PubMed]
- Nie, X.; Yu, S.; Qiu, M.; Wang, X.; Wang, Y.; Bai, Y.; Zhang, F.; Wang, S. Aspergillus flavus SUMO Contributes to Fungal Virulence and Toxin Attributes. J. Agric. Food Chem. 2016, 64, 6772–6782. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, P.; Venâncio, A.; Kozakiewicz, Z.; Lima, N. A polyphasic approach to the identification of aflatoxigenic and non-aflatoxigenic strains of Aspergillus section Flavi isolated from Portuguese almonds. Int. J. Food Microbiol. 2009, 129, 187–193. [Google Scholar] [CrossRef] [PubMed]
- Davari, E.; Mohsenzadeh, M.; Mohammadi, G.; Rezaeian-Doloei, R. Characterization of aflatoxigenic Aspergillus flavus and A. parasiticus strain isolates from animal feedstuffs in northeastern Iran. Iran. J. Vet. Res. 2015, 16, 150. [Google Scholar] [PubMed]
- Hedayati, M.; Pasqualotto, A.; Warn, P.; Bowyer, P.; Denning, D. Aspergillus flavus: Human pathogen, allergen and mycotoxin producer. Microbiology 2007, 153, 1677–1692. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Chang, P.-K.; Ehrlich, K.C.; Cary, J.W.; Bhatnagar, D.; Cleveland, T.E.; Payne, G.A.; Linz, J.E.; Woloshuk, C.P.; Bennett, J.W. Clustered pathway genes in aflatoxin biosynthesis. Appl. Environ. Microbiol. 2004, 70, 1253–1262. [Google Scholar] [CrossRef]
- Cleveland, T.E.; Yu, J.; Fedorova, N.; Bhatnagar, D.; Payne, G.A.; Nierman, W.C.; Bennett, J.W. Potential of Aspergillus flavus genomics for applications in biotechnology. Trends Biotechnol. 2009, 27, 151–157. [Google Scholar] [CrossRef]
- Yang, K.; Qin, Q.; Liu, Y.; Zhang, L.; Liang, L.; Lan, H.; Chen, C.; You, Y.; Zhang, F.; Wang, S. Adenylate cyclase AcyA regulates development, aflatoxin biosynthesis and fungal virulence in Aspergillus flavus. Front. Cell. Infect. Microbiol. 2016, 6, 190. [Google Scholar] [CrossRef]
- Zhang, F.; Guo, Z.; Zhong, H.; Wang, S.; Yang, W.; Liu, Y.; Wang, S. RNA-Seq-based transcriptome analysis of aflatoxigenic Aspergillus flavus in response to water activity. Toxins 2014, 6, 3187–3207. [Google Scholar] [CrossRef]
- Gonçalves, S.S.; Cano, J.F.; Stchigel, A.M.; Melo, A.S.; Godoy-Martinez, P.C.; Correa, B.; Guarro, J. Molecular phylogeny and phenotypic variability of clinical and environmental strains of Aspergillus flavus. Fungal Biol. 2012, 116, 1146–1155. [Google Scholar] [CrossRef]
- Yu, J.; Cleveland, T.E.; Nierman, W.C.; Bennett, J.W. Aspergillus flavus genomics: Gateway to human and animal health, food safety, and crop resistance to diseases. Revista Iberoamericana de Micología 2005, 22, 194–202. [Google Scholar] [CrossRef]
- Amaike, S.; Keller, N.P. Aspergillus flavus. Ann. Rev. Phytopathol. 2011, 49, 107–133. [Google Scholar] [CrossRef] [PubMed]
- Nascimento, B.; de Oliveira, A.C.M.; dos Reis, T.F.; de Castro, P.A.; Hori, J.I.; Bom, V.L.P.; de Assis, L.J.; Ramalho, L.N.Z.; Rocha, M.C.; Malavazi, I. Mitogen activated protein kinases SakAHOG1 and MpkC collaborate for Aspergillus fumigatus virulence. Mol. Microbiol. 2016, 100, 841–859. [Google Scholar] [CrossRef] [PubMed]
- Du, C.; Sarfati, J.; Latge, J.; Calderone, R. The role of the sakA (Hog1) and tcsB (sln1) genes in the oxidant adaptation of Aspergillus fumigatus. Med. Mycol. 2006, 44, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Duran, R.; Cary, J.W.; Calvo, A.M. Role of the osmotic stress regulatory pathway in morphogenesis and secondary metabolism in filamentous fungi. Toxins 2010, 2, 367–381. [Google Scholar] [CrossRef] [PubMed]
- Johnson, G.L.; Lapadat, R. Mitogen-activated protein kinase pathways mediated by ERK, JNK, and p38 protein kinases. Science 2002, 298, 1911–1912. [Google Scholar] [CrossRef]
- Nimmanee, P.; Tam, E.W.; Woo, P.C.; Vanittanakom, P.; Vanittanakom, N. Role of the Talaromyces marneffei (Penicillium marneffei) sakA gene in nitrosative stress response, conidiation and red pigment production. FEMS Microbiol. Lett. 2017, 364. [Google Scholar] [CrossRef]
- Saito, H.; Tatebayashi, K. Regulation of the osmoregulatory HOG MAPK cascade in yeast. J. Biochem. 2004, 136, 267–272. [Google Scholar] [CrossRef]
- Kawasaki, L.; Sánchez, O.; Shiozaki, K.; Aguirre, J. SakA MAP kinase is involved in stress signal transduction, sexual development and spore viability in Aspergillus nidulans. Mol. Microbiol. 2002, 45, 1153–1163. [Google Scholar] [CrossRef]
- Nimmanee, P.; Woo, P.C.; Kummasook, A.; Vanittanakom, N. Characterization of sakA gene from pathogenic dimorphic fungus Penicillium marneffei. Int. J. Med. Microbiol. 2015, 305, 65–74. [Google Scholar] [CrossRef]
- Hagiwara, D.; Takahashi-Nakaguchi, A.; Toyotome, T.; Yoshimi, A.; Abe, K.; Kamei, K.; Gonoi, T.; Kawamoto, S. NikA/TcsC histidine kinase is involved in conidiation, hyphal morphology, and responses to osmotic stress and antifungal chemicals in Aspergillus fumigatus. PLoS ONE 2013, 8, e80881. [Google Scholar] [CrossRef] [PubMed]
- May, G.; Xue, T.; Kontoyiannis, D.; Gustin, M. Mitogen activated protein kinases of Aspergillus fumigatus. Med. Mycol. 2005, 43, 83–86. [Google Scholar] [CrossRef]
- Xue, T.; Nguyen, C.K.; Romans, A.; May, G.S. A mitogen-activated protein kinase that senses nitrogen regulates conidial germination and growth in Aspergillus fumigatus. Eukaryot. Cell 2004, 3, 557–560. [Google Scholar] [CrossRef] [PubMed]
- Jaimes-Arroyo, R.; Lara-Rojas, F.; Bayram, Ö.; Valerius, O.; Braus, G.H.; Aguirre, J. The SrkA kinase is part of the SakA MAPK interactome and regulates stress responses and development in Aspergillus nidulans. Eukaryot. Cell 2015, 14, 459–510. [Google Scholar] [CrossRef] [PubMed]
- Pereira Silva, L.; Alves de Castro, P.; Dos Reis, T.F.; Paziani, M.H.; Von Zeska Kress, M.R.; Riaño-Pachón, D.M.; Hagiwara, D.; Ries, L.N.; Brown, N.A.; Goldman, G.H. Genome-wide transcriptome analysis of Aspergillus fumigatus exposed to osmotic stress reveals regulators of osmotic and cell wall stresses that are SakAHOG1 and MpkC dependent. Cell. Microbiol. 2017, 19, e12681. [Google Scholar] [CrossRef] [PubMed]
- Albertyn, J.; Hohmann, S.; Thevelein, J.M.; Prior, B.A. GPD1, which encodes glycerol-3-phosphate dehydrogenase, is essential for growth under osmotic stress in Saccharomyces cerevisiae, and its expression is regulated by the high-osmolarity glycerol response pathway. Mol. Cell. Biol. 1994, 14, 4135–4144. [Google Scholar] [CrossRef] [PubMed]
- Norbeck, J.; Påhlman, A.-K.; Akhtar, N.; Blomberg, A.; Adler, L. Purification and characterization of two isoenzymes of dl-glycerol-3-phosphatase from Saccharomyces cerevisiae identification of the corresponding GPP1 and GPP2 genes and evidence for osmotic regulation of Gpp2p expression by the osmosensing mitogen-activated protein kinase signal transduction pathway. J. Biol. Chem. 1996, 271, 13875–13881. [Google Scholar]
- Påhlman, A.-K.; Granath, K.; Ansell, R.; Hohmann, S.; Adler, L. The yeast glycerol 3-phosphatases Gpp1p and Gpp2p are required for glycerol biosynthesis and differentially involved in the cellular responses to osmotic, anaerobic, and oxidative stress. J. Biol. Chem. 2001, 276, 3555–3563. [Google Scholar] [CrossRef]
- Lillehoj, E.; Wall, J.; Bowers, E. Preharvest aflatoxin contamination: Effect of moisture and substrate variation in developing cottonseed and corn kernels. Appl. Environ. Microbiol. 1987, 53, 584–586. [Google Scholar]
- Fiedurek, J. Effect of osmotic stress on glucose oxidase production and secretion by Aspergillus niger. J. Basic Microbiol. 1998, 38, 107–112. [Google Scholar] [CrossRef]
- Hagiwara, D.; Suzuki, S.; Kamei, K.; Gonoi, T.; Kawamoto, S. The role of AtfA and HOG MAPK pathway in stress tolerance in conidia of Aspergillus fumigatus. Fungal Genet. Biol. 2014, 73, 138–149. [Google Scholar] [CrossRef] [PubMed]
- Seo, J.-A.; Guan, Y.; Yu, J.-H. FluG-dependent asexual development in Aspergillus nidulans occurs via de-repression. Genetics 2005, 172, 1535–1544. [Google Scholar] [CrossRef] [PubMed]
- Smith, M.E.; Henkel, T.W.; Rollins, J.A. How many fungi make sclerotia? Fungal Ecol. 2015, 13, 211–220. [Google Scholar] [CrossRef]
- El-Abyad, M.; Hindorf, H.; Rizk, M. Impact of salinity stress on soil-borne fungi of sugarbeet. Plant Soil 1988, 110, 27–32. [Google Scholar] [CrossRef]
- Han, K.H.; Seo, J.A.; Yu, J.H. A putative G protein-coupled receptor negatively controls sexual development in Aspergillus nidulans. Mol. Microbiol. 2004, 51, 1333–1345. [Google Scholar] [CrossRef] [PubMed]
- Mert, H.; Ekmekci, S. The effect of salinity and osmotic pressure of the medium on the growth, sporulation and changes in the total organic acid content of Aspergillus flavus and Penicillium chrysogenum. Mycopathologia 1987, 100, 85–89. [Google Scholar] [CrossRef]
- Ramos, A.; Magan, N.; Sanchis, V. Osmotic and matric potential effects on growth, sclerotia and partitioning of polyols and sugars in colonies and spores of Aspergillus ochraceus. Mycol. Res. 1999, 103, 141–147. [Google Scholar] [CrossRef]
- Rementeria, A.; López-Molina, N.; Ludwig, A.; Vivanco, A.B.; Bikandi, J.; Pontón, J.; Garaizar, J. Genes and molecules involved in Aspergillus fumigatus virulence. Rev. Iberoam. Micol. 2005, 22, 1–23. [Google Scholar] [CrossRef]
- Zhao, X.; Mehrabi, R.; Xu, J.-R. Mitogen-activated protein kinase pathways and fungal pathogenesis. Eukaryot. Cell 2007, 6, 1701–1714. [Google Scholar] [CrossRef]
- Alves de Castro, P.; Dos Reis, T.F.; Dolan, S.K.; Oliveira Manfiolli, A.; Brown, N.A.; Jones, G.W.; Doyle, S.; Riaño-Pachón, D.M.; Squina, F.M.; Caldana, C. The Aspergillus fumigatus SchASCH9 kinase modulates SakAHOG1 MAP kinase activity and it is essential for virulence. Mol. Microbiol. 2016, 102, 642–671. [Google Scholar] [CrossRef]
- Lara-Rojas, F.; Sánchez, O.; Kawasaki, L.; Aguirre, J. Aspergillus nidulans transcription factor AtfA interacts with the MAPK SakA to regulate general stress responses, development and spore functions. Mol. Microbiol. 2011, 80, 436–454. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; He, Y.; Li, X.; Fasoyin, O.E.; Hu, Y.; Liu, Y.; Yuan, J.; Zhuang, Z.; Wang, S. Histone Methyltransferase aflrmtA gene is involved in the morphogenesis, mycotoxin biosynthesis, and pathogenicity of Aspergillus flavus. Toxicon 2017, 127, 112–121. [Google Scholar] [CrossRef] [PubMed]
- Chang, P.-K.; Scharfenstein, L.L.; Mack, B.; Ehrlich, K.C. Deletion of the Aspergillus flavus orthologue of Aspergillus nidulans fluG reduces conidiation, promotes sclerotial production, but does not abolish aflatoxin biosynthesis. Appl. Environ. Microbiol. 2012, 78, 7557–7563. [Google Scholar] [CrossRef] [PubMed]
- Chang, P.-K.; Scharfenstein, L.L.; Wei, Q.; Bhatnagar, D. Development and refinement of a high-efficiency gene-targeting system for Aspergillus flavus. J. Microbiol. Methods 2010, 81, 240–246. [Google Scholar] [CrossRef]
- Zhuang, Z.; Lohmar, J.M.; Satterlee, T.; Cary, J.W.; Calvo, A.M. The master transcription factor mtfA governs aflatoxin production, morphological development and pathogenicity in the fungus Aspergillus flavus. Toxins 2016, 8, 29. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.-H.; Hamari, Z.; Han, K.-H.; Seo, J.-A.; Reyes-Domínguez, Y.; Scazzocchio, C. Double-joint PCR: A PCR-based molecular tool for gene manipulations in filamentous fungi. Fungal Genet. Biol. 2004, 41, 973–981. [Google Scholar] [CrossRef] [PubMed]
- Yang, K.; Liang, L.; Ran, F.; Liu, Y.; Li, Z.; Lan, H.; Gao, P.; Zhuang, Z.; Zhang, F.; Nie, X. The DmtA methyltransferase contributes to Aspergillus flavus conidiation, sclerotial production, aflatoxin biosynthesis and virulence. Sci. Rep. 2016, 6, 23259. [Google Scholar] [CrossRef]
- Yao, G.; Yue, Y.; Fu, Y.; Fang, Z.; Xu, Z.; Ma, G.; Wang, S. Exploration of the regulatory mechanism of secondary metabolism by comparative transcriptomics in Aspergillus flavus. Front. Microbiol. 2018, 9, 1568. [Google Scholar] [CrossRef]
- Lan, H.; Sun, R.; Fan, K.; Yang, K.; Zhang, F.; Nie, X.Y.; Wang, X.; Zhuang, Z.; Wang, S. The Aspergillus flavus histone acetyltransferase AflGcnE regulates morphogenesis, aflatoxin biosynthesis, and pathogenicity. Front. Microbiol. 2016, 7, 1324. [Google Scholar] [CrossRef]
- Fasoyin, O.E.; Wang, B.; Qiu, M.; Han, X.; Chung, K.-R.; Wang, S. Carbon catabolite repression gene creA regulates morphology, aflatoxin biosynthesis and virulence in Aspergillus flavus. Fungal Genet. Biol. 2018, 115, 41–51. [Google Scholar] [CrossRef]
- Yang, G.; Hu, Y.; Wang, S.; Fasoyin, O.E.; Yue, Y.; Qiu, Y.; Wang, X. The Aspergillus flavus Phosphatase CDC14 Regulates Development, Aflatoxin Biosynthesis and Pathogenicity. Front. Cell. Infect. Microbiol. 2018, 8, 141. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Han, X.; Bai, Y.; Lin, Z.; Qiu, M.; Nie, X.; Wang, S.; Zhang, F.; Zhuang, Z.; Yuan, J. Effects of nitrogen metabolism on growth and aflatoxin biosynthesis in Aspergillus flavus. J. Hazard. Mater. 2017, 324, 691–700. [Google Scholar] [CrossRef] [PubMed]
- Han, X.; Qiu, M.; Wang, B.; Yin, W.-B.; Nie, X.; Qin, Q.; Ren, S.; Yang, K.; Zhang, F.; Zhuang, Z. Functional analysis of the nitrogen metabolite repression regulator gene nmrA in Aspergillus flavus. Front. Microbiol. 2016, 7, 1794. [Google Scholar] [CrossRef] [PubMed]
- Wong, K.H.; Todd, R.B.; Oakley, B.R.; Oakley, C.E.; Hynes, M.J.; Davis, M.A. Sumoylation in Aspergillus nidulans: SumO inactivation, overexpression and live-cell imaging. Fungal Genet. Biol. 2008, 45, 728–737. [Google Scholar] [CrossRef] [PubMed]
- Yang, K.; Liu, Y.; Liang, L.; Li, Z.; Qin, Q.; Nie, X.; Wang, S. The high-affinity phosphodiesterase PdeH regulates development and aflatoxin biosynthesis in Aspergillus flavus. Fungal Genet. Biol. 2017, 101, 7–19. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Yang, G.; Zhang, D.; Liu, Y.; Li, Y.; Lin, G.; Guo, Z.; Wang, S.; Zhuang, Z. The PHD Transcription Factor Rum1 Regulates Morphogenesis and Aflatoxin Biosynthesis in Aspergillus flavus. Toxins 2018, 10, 301. [Google Scholar] [CrossRef] [PubMed]
- Kale, S.P.; Milde, L.; Trapp, M.K.; Frisvad, J.C.; Keller, N.P.; Bok, J.W. Requirement of LaeA for secondary metabolism and sclerotial production in Aspergillus flavus. Fungal Genet. Biol. 2008, 45, 1422–1429. [Google Scholar] [CrossRef]
- Yuan, J.; Chen, Z.; Guo, Z.; Li, D.; Zhang, F.; Shen, J.; Zhang, Y.; Wang, S.; Zhuang, Z. PbsB regulates morphogenesis, Aflatoxin B1 biosynthesis and pathogenicity of Aspergillus flavus. Front. Cell. Infect. Microbiol. 2018, 8, 162. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2− ΔΔCT method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
| Strain Name | Related Genotype | Source |
|---|---|---|
| CA14 | Δku70; niaD-; ΔpyrG | Chang et al. [44] |
| Wild-type | Δku70; niaD-; ΔpyrG::pyrG | This study |
| ΔAfsakA | Δku70; ΔAfsakA::pyrG | This study |
| ΔAfsakA::AfsakA | Δku70; ΔAfsakA::pyrG; AfsakA (p)::sakA::ptrA | This study |
| AfsakA-mCherry | Δku70; AfsakA (p)::mCherry::AfsakA::pyrG. | This study |
| Primers | Sequence (5′-3′) | Application |
|---|---|---|
| P1 | TGTTATAGGGACGCTCTG | AfsakA deletion |
| P2 | CGAACGTAGTACCCAAGAT | |
| P3 | CGGAGCATTGTCCTACAT | |
| P4 | GAGCGAATTACTGTTTGAGT | |
| P7 | CATGTCGGGACGAGTTTG | |
| P8 | GAAGTGGTTTACGGTGGT | |
| P9 | ATACGCCTGCAACGCTAA | |
| P10 | ATCCGCCTGGAGAAAGTC | |
| pyrG-F | GCCTCAAACAATGCTCTTCACCC | pyrG amplification |
| pyrG-R | GTCTGAGAGGAGGCACTGATGC | |
| P801 | CAGGAGTTCTCGGGTTGTCG | AfsakA mutant verification |
| P1080 | ATCGGCAATACCGTCCAGAAGC | |
| AfSakA-F | TGTTATAGGGACGCTCTG | AfsakA complementation |
| AfsakA-R | TCATATATCCGCCTGGAGAAAG | |
| mCherry/F | ATGGTGAGCAAGGGCGAG | AfsakA- mCherry tag construction |
| mCherry/R | GGGTGAAGAGCATTGTTTGAGGCCTACTTGTACAGCTCGTCCAT | |
| pyrG-R/R | GCCTCAAACAATGCTCTTCACCC | |
| pyrG-R/F | AGTTGGTACGAAACAGATCAGTCTGAGAGGAGGCACTGATGC | |
| AfsakA-mCherry/F | TCGGTTTGGGTGCGTTTG | |
| AfsakA-mCherry/R | CTCGCCCTTGCTCACCATGACTAGTTTGTAAAGTTTACTTTGGACTAT | |
| AfsakA-B/F | GTCTGAGAGGAGGCACTGATGCTGATCTGTTTCGTACCAACT | |
| AfsakA-B/R | TGAGACCGCCGTCCTAAC | |
| AfsakA-O/F | GGTTTGGGTGCGTTTG | |
| AfsakA-O/R | CCCGAATTTATTGTAGCG |
| Primers | Sequence (5′-3′) | Application |
|---|---|---|
| Q-AfsakA-F | CCCACTACCAAAGGCACTC | AfsakA detection |
| Q-AfsakA-R | GGCATCATTGAACGACCAG | |
| AflO-F | GATTGGGATGTGGTCATGCGATT | AflO qRT-PCR |
| AflO-R | GCCTGGGTCCGAAGAATGC | |
| AflQ-F | GTCGCATATGCCCCGGTCGG | AflQ qRT-PCR |
| AflQ-R | GGCAACCAGTCGGGTTCCGG | |
| AflR-F | AAAGCACCCTGTCTTCCCTAAC | AflR qRT-PCR |
| AflR-R | GAAGAGGTGGGTCAGTGTTTGTAG | |
| AflS-F | GCTCAGACTGACCGCCGCTC | AflR qRT-PCR |
| AflS-R | GCTCAGACTGACCGCCGCTC | |
| NsdC-F | GCCAGACTTGCCAATCAC | NsdC qRT-PCR |
| NsdC-R | CATCCACCTTGCCCTTTA | |
| NsdD-F | GGACTTGCGGGTCGTGCTA | NsdD qRT-PCR |
| NsdD-R | AGAACGCTGGGTCTGGTGC | |
| abaA-F | TCTTCGGTTGATGGATGATTTC | abaA qRT-PCR |
| abaA-R | CCGTTGGGAGGCTGGGT | |
| brlA-F | GCCTCCAGCGTCAACCTTC | brlA qRT-PCR |
| brlA-R | TCTCTTCAAATGCTCTTGCCTC | |
| HSP-F | CCGGCATACTATGTCTCGTCT | HSP qRT-PCR |
| HSP-R | TAGGGCCTTCGTCGAACA | |
| GPD-F | TGTCTCGGTGGTGTCCCTAT | GPD qRT-PCR |
| GPD-R | ACCATGGCTGATGGAAGACT | |
| GRE-F | GCGTATCGTCGTTACCTCATC | GRE qRT-PCR |
| GRE-R | CCTTCTCCTTTACCTCCTCGAT | |
| STL-F | CGTTTACCACGACCAGAGC | STL qRT-PCR |
| STL-R | AAGCTCAAGCCATGTGCAG | |
| Actin-F | ACGGTGTCGTCACAAACTGG | Actin qRT-PCR |
| Actin-R | CGGTTGGACTTAGGGTTGATAG |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tumukunde, E.; Li, D.; Qin, L.; Li, Y.; Shen, J.; Wang, S.; Yuan, J. Osmotic-Adaptation Response of sakA/hogA Gene to Aflatoxin Biosynthesis, Morphology Development and Pathogenicity in Aspergillus flavus. Toxins 2019, 11, 41. https://doi.org/10.3390/toxins11010041
Tumukunde E, Li D, Qin L, Li Y, Shen J, Wang S, Yuan J. Osmotic-Adaptation Response of sakA/hogA Gene to Aflatoxin Biosynthesis, Morphology Development and Pathogenicity in Aspergillus flavus. Toxins. 2019; 11(1):41. https://doi.org/10.3390/toxins11010041
Chicago/Turabian StyleTumukunde, Elisabeth, Ding Li, Ling Qin, Yu Li, Jiaojiao Shen, Shihua Wang, and Jun Yuan. 2019. "Osmotic-Adaptation Response of sakA/hogA Gene to Aflatoxin Biosynthesis, Morphology Development and Pathogenicity in Aspergillus flavus" Toxins 11, no. 1: 41. https://doi.org/10.3390/toxins11010041
APA StyleTumukunde, E., Li, D., Qin, L., Li, Y., Shen, J., Wang, S., & Yuan, J. (2019). Osmotic-Adaptation Response of sakA/hogA Gene to Aflatoxin Biosynthesis, Morphology Development and Pathogenicity in Aspergillus flavus. Toxins, 11(1), 41. https://doi.org/10.3390/toxins11010041






