Selection of Fusarium Trichothecene Toxin Genes for Molecular Detection Depends on TRI Gene Cluster Organization and Gene Function
Abstract
1. Introduction
2. Biosynthetic Gene Clusters
3. Trichothecene Gene Cluster Structure and Organization
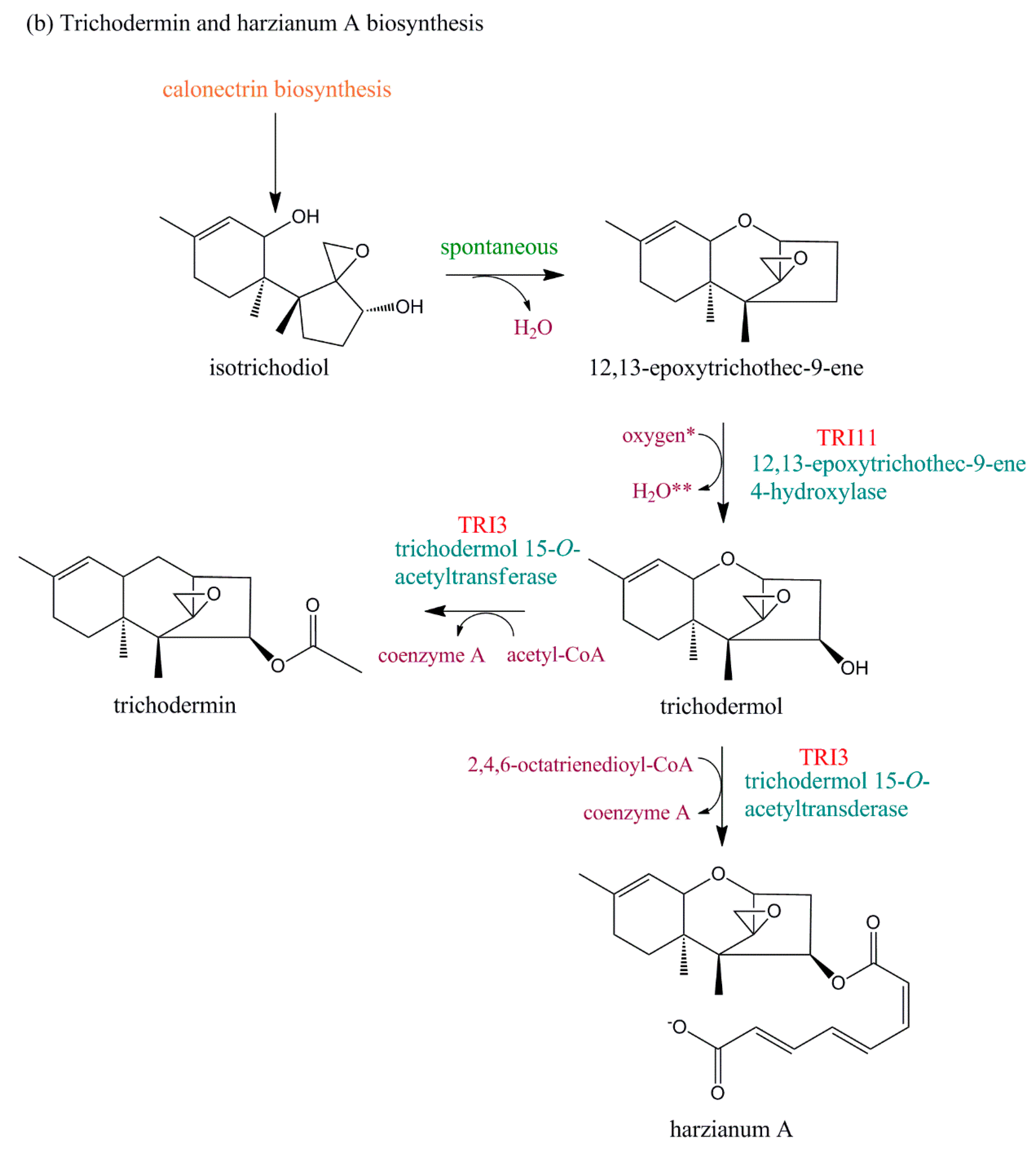
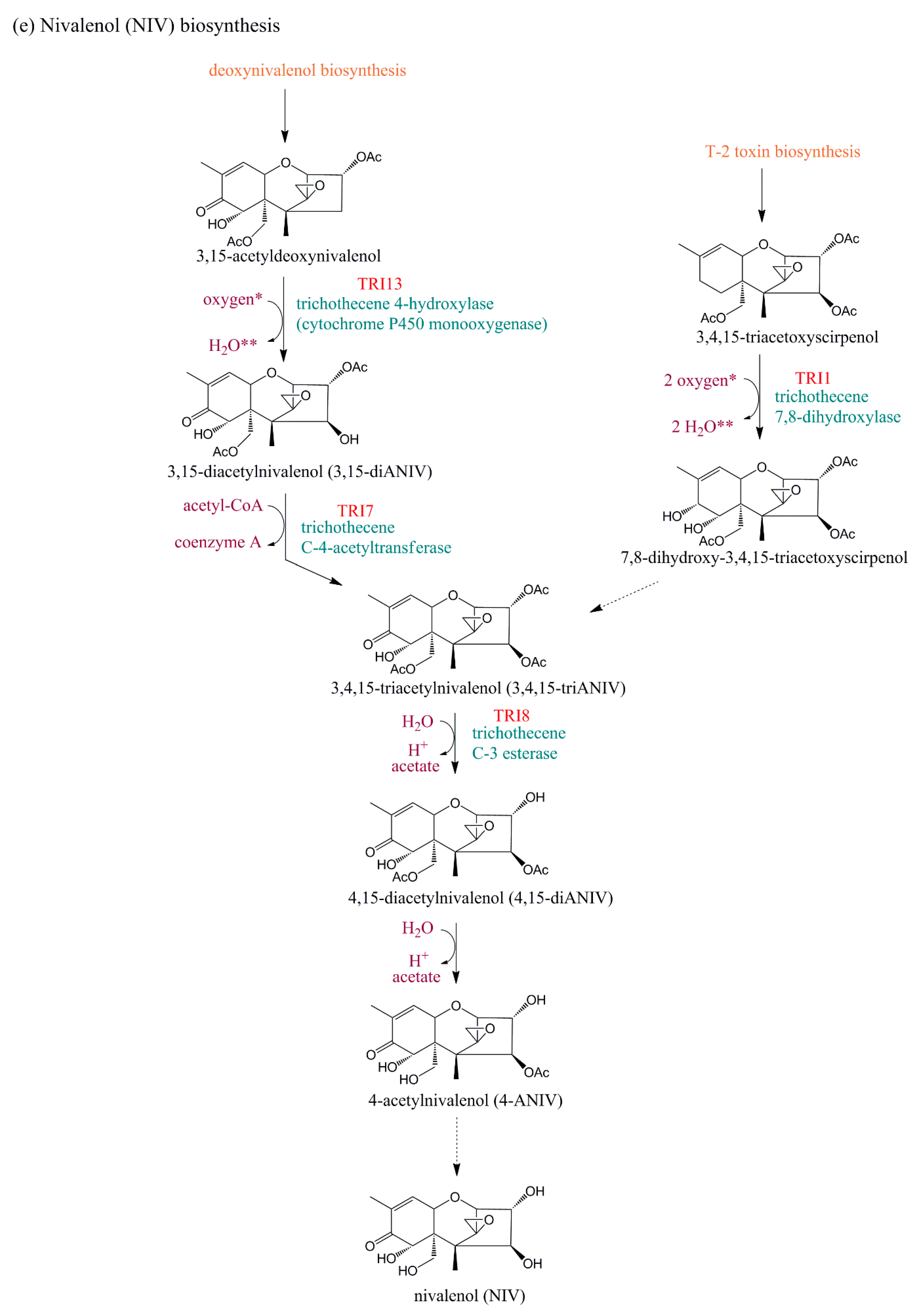
4. Evolution of TRI Genes and Production of Trichothecene Analogs
5. Using Fusarium TRI Gene Cluster for the Identification of Trichothecene Genotypes
5.1. Intra- and Interspecies Variation of TRI Genes at the Nucleotide and Amino Acid Sequence Levels
5.2. Selection of TRI Genes
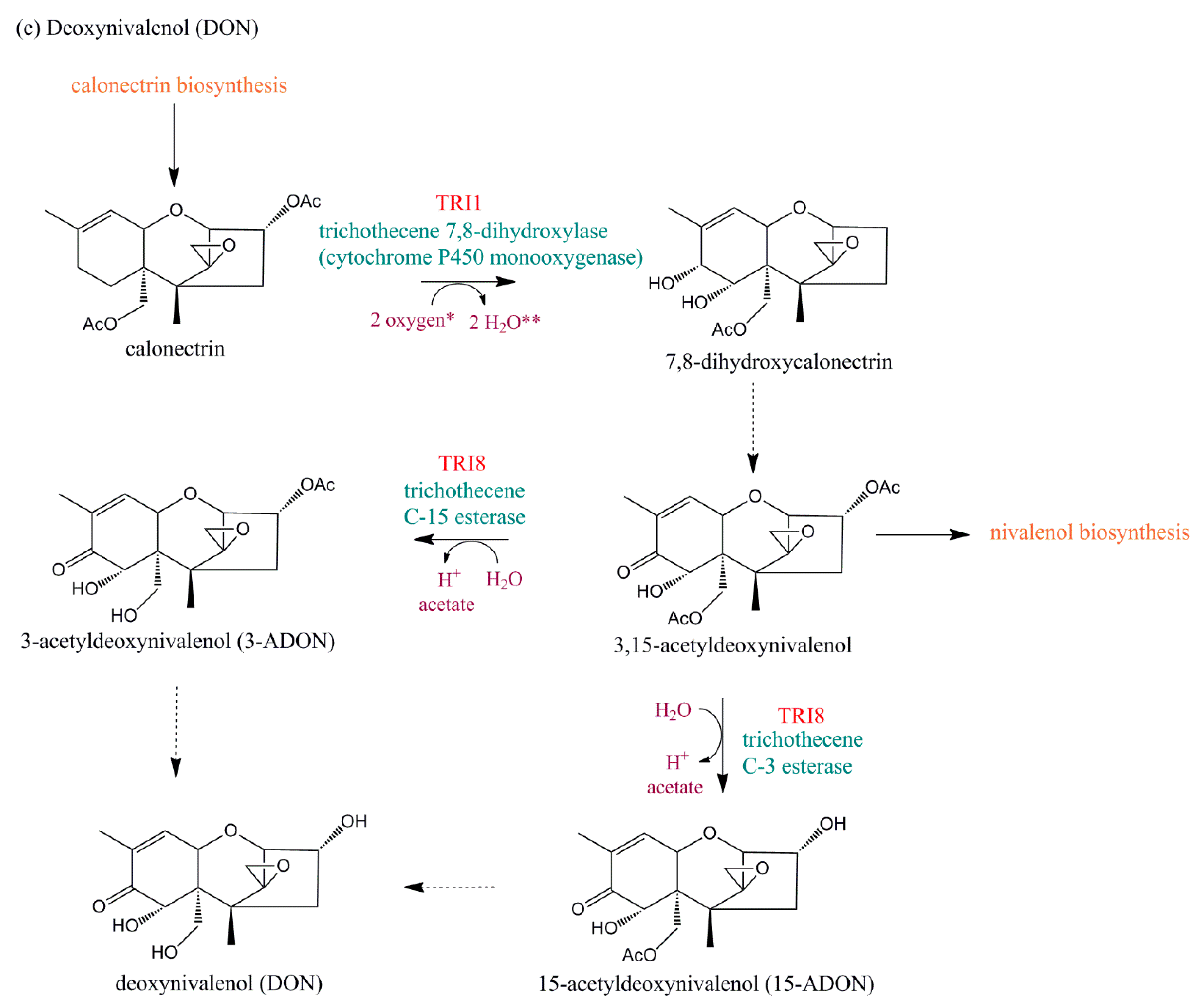
5.2.1. TRI1
5.2.2. TRI5
5.2.3. TRI6
5.2.4. TRI8
5.2.5. TRI11
5.2.6. TRI12
5.2.7. TRI13
5.2.8. TRI3 and TRI6 in Combination
5.2.9. TRI7 and TRI13 in Combination
5.2.10. TRI3 and TRI12 in Combination
6. Molecular Detection of mRNA Does Not Generally Predict its Protein Level
7. Conclusion and Future Prospects
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Moretti, A.; Susca, A.; Mulé, G.; Logrieco, A.; Proctor, R. Molecular biodiversity of mycotoxigenic fungi that threaten food safety. Int. J. Food Microbiol. 2013, 167, 57–66. [Google Scholar] [CrossRef]
- Moretti, A.; Panzarini, G.; Somma, S.; Campagna, C.; Ravaglia, S.; Logrieco, A.F.; Solfrizzo, M. Systemic growth of F. graminearum in wheat plants and related accumulation of deoxynivalenol. Toxins 2014, 6, 1308–1324. [Google Scholar] [CrossRef] [PubMed]
- Bertero, A.; Moretti, A.; Spicer, L.; Caloni, F. Fusarium Molds and Mycotoxins: Potential Species-Specific Effects. Toxins 2018, 10, 244. [Google Scholar] [CrossRef] [PubMed]
- Desjardins, A.E. From yellow rain to green wheat: 25 years of trichothecene biosynthesis research. J. Agric. Food Chem. 2009, 57, 4478–4484. [Google Scholar] [CrossRef] [PubMed]
- Lowe, R.; Jubault, M.; Canning, G.; Urban, M.; Hammond-Kosack, K.E. The induction of mycotoxins by trichothecene producing Fusarium species. In Plant Fungal Pathogens; Springer: Berlin, Germany, 2012; pp. 439–455. [Google Scholar]
- Pasquali, M.; Beyer, M.; Logrieco, A.; Audenaert, K.; Balmas, V.; Basler, R.; Boutigny, A.-L.; Chrpova, J.; Czembor, E.; Gagkaeva, T.; et al. A European database of Fusarium graminearum and F. culmorum trichothecene genotypes. Front. Microbiol. 2016, 7, 406. [Google Scholar] [CrossRef]
- Dean, R.; Van Kan, J.A.; Pretorius, Z.A.; Hammond-Kosack, K.E.; Di Pietro, A.; Spanu, P.D.; Rudd, J.J.; Dickman, M.; Kahmann, R.; Ellis, J. The Top 10 fungal pathogens in molecular plant pathology. Mol. Plant Pathol. 2012, 13, 414–430. [Google Scholar] [CrossRef] [PubMed]
- Cardwell, K.F.; Desjardins, A.; Henry, S.H.; Munkvold, G.; Robens, J. Mycotoxins: The Cost of Achieving Food Security and Food Quality. APS Features 2001. [Google Scholar] [CrossRef]
- Ueno, Y. The toxicology of mycotoxins. Crit. Rev. Toxicol. 1985, 14, 99–132. [Google Scholar] [CrossRef]
- Desjardins, A.; Hohn, T.; McCormick, S.P. Effect of gene disruption of trichodiene synthase on the virulence of Gibberella pulicaris. Mol. Plant-Microbe Int. 1992, 5, 214–222. [Google Scholar] [CrossRef]
- Desjardins, A.; Proctor, R.; McCormick, S.; Hohn, T. Reduced virulence of trichothecene antibiotic-nonproducing mutants of Gibberella zeae in wheat field tests. Fusarium Head Scab: Global Status and Future Prospects. In Proceedings of the Workshop, El Batan, Mexico, 13–17 October 1996. [Google Scholar]
- Cuzick, A.; Urban, M.; Hammond-Kosack, K. Fusarium graminearum gene deletion mutants map1 and tri5 reveal similarities and differences in the pathogenicity requirements to cause disease on Arabidopsis and wheat floral tissue. New Phytol. 2008, 177, 990–1000. [Google Scholar] [CrossRef]
- Eriksen, G.S. Fusarium Toxins in Cereals: A Risk Assessment; Nordic Council of Ministers: Copenhagen, Denmark, 1998.
- Eriksen, G.S.; Pettersson, H. Toxicological evaluation of trichothecenes in animal feed. Anim. Feed Sci. Technol. 2004, 114, 205–239. [Google Scholar] [CrossRef]
- Chappell, J.; Coates, R.M. Sesquiterpenes. In Comprehensive Natural Products II, Chemistry and Biology, 1st ed.; Mander, L., Liu, H.-W.B., Eds.; Elsevier Science and Technology: Oxford, UK, 2010; Volume 1, pp. 609–641. [Google Scholar]
- McCormick, S.P.; Stanley, A.M.; Stover, N.A.; Alexander, N.J. Trichothecenes: From simple to complex mycotoxins. Toxins 2011, 3, 802–814. [Google Scholar] [CrossRef] [PubMed]
- BIOMIN. BIOMIN Mycotoxin Survey. Available online: https://www.biomin.net/en/biomin-mycotoxin-survey/ (accessed on 16 November 2018).
- Sydenham, E.W.; Shephard, G.S.; Thiel, P.G.; Marasas, W.F.; Stockenstrom, S. Fumonisin contamination of commercial corn-based human foodstuffs. J. Agric. Food Chem. 1991, 39, 2014–2018. [Google Scholar] [CrossRef]
- Miller, J.D.; Greenhalgh, R.; Wang, Y.; Lu, M. Trichothecene chemotypes of three Fusarium species. Mycologia 1991. [Google Scholar] [CrossRef]
- Keller, N.P.; Hohn, T.M. Metabolic pathway gene clusters in filamentous fungi. Fungal Genet. Biol. 1997, 21, 17–29. [Google Scholar] [CrossRef] [PubMed]
- Keller, N.P.; Turner, G.; Bennett, J.W. Fungal secondary metabolism: From biochemistry to genomics. Nat. Rev. Microbiol. 2005, 3, 937. [Google Scholar] [CrossRef]
- Brown, D.W.; Lee, S.-H.; Kim, L.-H.; Ryu, J.-G.; Lee, S.; Seo, Y.; Kim, Y.H.; Busman, M.; Yun, S.-H.; Proctor, R.H. Identification of a 12-gene fusaric acid biosynthetic gene cluster in Fusarium species through comparative and functional genomics. Mol. Plant-Microbe Int. 2015, 28, 319–332. [Google Scholar] [CrossRef]
- Medema, M.H.; Cimermancic, P.; Sali, A.; Takano, E.; Fischbach, M.A. A systematic computational analysis of biosynthetic gene cluster evolution: Lessons for engineering biosynthesis. PLoS Comp. Biol. 2014, 10, 1–12. [Google Scholar] [CrossRef]
- Inglis, D.O.; Binkley, J.; Skrzypek, M.S.; Arnaud, M.B.; Cerqueira, G.C.; Shah, P.; Wymore, F.; Wortman, J.R.; Sherlock, G. Comprehensive annotation of secondary metabolite biosynthetic genes and gene clusters of Aspergillus nidulans, A. fumigatus, A. niger and A. oryzae. BMC Microbiol. 2013, 13, 91. [Google Scholar] [CrossRef]
- Lazarus, C.; Williams, K.; Bailey, A. Reconstructing fungal natural product biosynthetic pathways. Nat. Prod. Rep. 2014, 31, 1339–1347. [Google Scholar] [CrossRef]
- Proctor, R.H.; McCormick, S.P.; Kim, H.-S.; Cardoza, R.E.; Stanley, A.M.; Lindo, L.; Kelly, A.; Brown, D.W.; Lee, T.; Vaughan, M.M. Evolution of structural diversity of trichothecenes, a family of toxins produced by plant pathogenic and entomopathogenic fungi. PLoS Pathog. 2018, 14, e1006946. [Google Scholar] [CrossRef] [PubMed]
- Alexander, N.J.; Proctor, R.H.; McCormick, S.P. Genes, gene clusters, and biosynthesis of trichothecenes and fumonisins in Fusarium. Toxin Rev. 2009, 28, 198–215. [Google Scholar] [CrossRef]
- Maier, F.J.; Miedaner, T.; Hadeler, B.; Felk, A.; Salomon, S.; Lemmens, M.; Kassner, H.; Schaefer, W. Involvement of trichothecenes in fusarioses of wheat, barley and maize evaluated by gene disruption of the trichodiene synthase (Tri5) gene in three field isolates of different chemotype and virulence. Mol. Plant Pathol. 2006, 7, 449–461. [Google Scholar] [CrossRef] [PubMed]
- McGary, K.L.; Slot, J.C.; Rokas, A. Physical linkage of metabolic genes in fungi is an adaptation against the accumulation of toxic intermediate compounds. Proc. Nat. Acad. Sci. USA 2013, 110, 11481–11486. [Google Scholar] [CrossRef] [PubMed]
- Proctor, R.H.; McCormick, S.P.; Alexander, N.J.; Desjardins, A.E. Evidence that a secondary metabolic biosynthetic gene cluster has grown by gene relocation during evolution of the filamentous fungus Fusarium. Mol. Microbiol. 2009, 74, 1128–1142. [Google Scholar] [CrossRef] [PubMed]
- Yoder, O.; Valent, B.; Chumley, F. Genetic nomenclature and practice for plant pathogenic fungi. Phytopathology 1986, 76, 383–385. [Google Scholar] [CrossRef]
- McCormick, S.P.; Alexander, N.J.; Proctor, R.H. Trichothecene triangle: Toxins, genes, and plant disease. In Phytochemicals, Plant Growth, and the Environment; Springer: Berlin, Germany, 2013; pp. 1–17. [Google Scholar]
- Brown, D.W.; Dyer, R.B.; McCormick, S.P.; Kendra, D.F.; Plattner, R.D. Functional demarcation of the Fusarium core trichothecene gene cluster. Fungal Genet. Biol. 2004, 41, 454–462. [Google Scholar] [CrossRef]
- Cardoza, R.; Malmierca, M.; Hermosa, M.; Alexander, N.; McCormick, S.; Proctor, R.; Tijerino, A.; Rumbero, A.; Monte, E.; Gutiérrez, S. Identification of loci and functional characterization of trichothecene biosynthetic genes in the filamentous fungus Trichoderma. Appl. Environ. Microbiol. 2011, 4867–4877. [Google Scholar] [CrossRef]
- Kimura, M.; Tokai, T.; O’Donnell, K.; Ward, T.J.; Fujimura, M.; Hamamoto, H.; Shibata, T.; Yamaguchi, I. The trichothecene biosynthesis gene cluster of Fusarium graminearum F15 contains a limited number of essential pathway genes and expressed non-essential genes. FEBS Lett. 2003, 539, 105–110. [Google Scholar] [CrossRef]
- Lee, T.; Han, Y.-K.; Kim, K.-H.; Yun, S.-H.; Lee, Y.-W. Tri13 and Tri7 determine deoxynivalenol-and nivalenol-producing chemotypes of Gibberella zeae. Appl. Environ. Microbiol. 2002, 68, 2148–2154. [Google Scholar] [CrossRef]
- Rep, M.; Kistler, H.C. The genomic organization of plant pathogenicity in Fusarium species. Curr. Opin. Plant Biol. 2010, 13, 420–426. [Google Scholar] [CrossRef]
- Seong, K.-Y.; Zhao, X.; Xu, J.-R.; Güldener, U.; Kistler, H.C. Conidial germination in the filamentous fungus Fusarium graminearum. Fungal Genet. Biol. 2008, 45, 389–399. [Google Scholar] [CrossRef] [PubMed]
- Nasmith, C.G.; Walkowiak, S.; Wang, L.; Leung, W.W.; Gong, Y.; Johnston, A.; Harris, L.J.; Guttman, D.S.; Subramaniam, R. Tri6 is a global transcription regulator in the phytopathogen Fusarium graminearum. PLoS Pathog. 2011, 7, e1002266. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.; Ma, L.-J. Fusarium graminearum genomics and beyond. In Genomics of Plant-Associated Fungi: Monocot Pathogens; Springer: Berlin/Heidelberg, Germany, 2014; pp. 103–122. [Google Scholar]
- Brown, D.W.; McCormick, S.P.; Alexander, N.J.; Proctor, R.H.; Desjardins, A.E. A genetic and biochemical approach to study trichothecene diversity in Fusarium sporotrichioides and Fusarium graminearum. Fungal Genet. Biol. 2001, 32, 121–133. [Google Scholar] [CrossRef] [PubMed]
- Alexander, N.J.; McCormick, S.P.; Waalwijk, C.; Van Der Lee, T.; Proctor, R.H. The genetic basis for 3-ADON and 15-ADON trichothecene chemotypes in Fusarium. Fungal Genet. Biol. 2011, 48, 485–495. [Google Scholar] [CrossRef] [PubMed]
- Kimura, M.; Kaneko, I.; Komiyama, M.; Takatsuki, A.; Koshino, H.; Yoneyama, K.; Yamaguchi, I. Trichothecene 3-O-acetyltransferase protects both the producing organism and transformed yeast from related mycotoxins cloning and characterization of Tri101. J. Biol. Chem. 1998, 273, 1654–1661. [Google Scholar] [CrossRef] [PubMed]
- Kulik, T.; Abarenkov, K.; Buśko, M.; Bilska, K.; van Diepeningen, A.D.; Ostrowska-Kołodziejczak, A.; Krawczyk, K.; Brankovics, B.; Stenglein, S.; Sawicki, J. ToxGen: An improved reference database for the identification of type B-trichothecene genotypes in Fusarium. Peer J. 2017, 5, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Desjardins, A.E. Natural product chemistry meets genetics: When is a genotype a chemotype? J. Agric. Food Chem. 2008, 56, 7587–7592. [Google Scholar] [CrossRef]
- Pasquali, M.; Migheli, Q. Genetic approaches to chemotype determination in type B-trichothecene producing Fusaria. Int. J. Food Microbiol. 2014, 189, 164–182. [Google Scholar] [CrossRef]
- Miller, J.; Taylor, A.; Greenhalgh, R. Production of deoxynivalenol and related compounds in liquid culture by Fusarium graminearum. Can. J. Microbiol. 1983, 29, 1171–1178. [Google Scholar] [CrossRef]
- Jiao, F.; Kawakami, A.; Nakajima, T. Effects of different carbon sources on trichothecene production and Tri gene expression by Fusarium graminearum in liquid culture. FEMS Microbiol. Lett. 2008, 285, 212–219. [Google Scholar] [CrossRef] [PubMed]
- Gardiner, D.M.; Waring, P.; Howlett, B.J. The epipolythiodioxopiperazine (ETP) class of fungal toxins: Distribution, mode of action, functions and biosynthesis. Microbiology 2005, 151, 1021–1032. [Google Scholar] [CrossRef]
- Gardiner, D.M.; Kazan, K.; Manners, J.M. Nutrient profiling reveals potent inducers of trichothecene biosynthesis in Fusarium graminearum. Fungal Genet. Biol. 2009, 46, 604–613. [Google Scholar] [CrossRef] [PubMed]
- Ponts, N.; Couedelo, L.; Pinson-Gadais, L.; Verdal-Bonnin, M.-N.; Barreau, C.; Richard-Forget, F. Fusarium response to oxidative stress by H2O2 is trichothecene chemotype-dependent. FEMS Microbiol. Lett. 2009, 293, 255–262. [Google Scholar] [CrossRef] [PubMed]
- Hope, R.; Aldred, D.; Magan, N. Comparison of environmental profiles for growth and deoxynivalenol production by Fusarium culmorum and F. graminearum on wheat grain. Lett. Appl. Microbiol. 2005, 40, 295–300. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, M.L.; Chulze, S.; Magan, N. Temperature and water activity effects on growth and temporal deoxynivalenol production by two Argentinean strains of Fusarium graminearum on irradiated wheat grain. Int. J. Food Microbiol. 2006, 106, 291–296. [Google Scholar] [CrossRef] [PubMed]
- Schmidt-Heydt, M.; Magan, N.; Geisen, R. Stress induction of mycotoxin biosynthesis genes by abiotic factors. FEMS Microbiol. Lett. 2008, 284, 142–149. [Google Scholar] [CrossRef]
- Pestka, J.J.; El-Bahrawy, A.; Hart, L.P. Deoxynivalenol and 15-monoacetyl deoxynivalenol production by Fusarium graminearum R6576 in liquid media. Mycopathologia 1985, 91, 23–28. [Google Scholar] [CrossRef]
- Audenaert, K.; Vanheule, A.; Höfte, M.; Haesaert, G. Deoxynivalenol: A major player in the multifaceted response of Fusarium to its environment. Toxins 2013, 6, 1–19. [Google Scholar] [CrossRef]
- Tan, D.C.; Flematti, G.R.; Ghisalberti, E.L.; Sivasithamparam, K.; Chakraborty, S.; Obanor, F.; Jayasena, K.; Barbetti, M.J. Mycotoxins produced by Fusarium spp. associated with Fusarium head blight of wheat in Western Australia. Mycotoxin Res. 2012, 28, 89–96. [Google Scholar] [CrossRef]
- Ward, T.J.; Bielawski, J.P.; Kistler, H.C.; Sullivan, E.; O’Donnell, K. Ancestral polymorphism and adaptive evolution in the trichothecene mycotoxin gene cluster of phytopathogenic Fusarium. Proc. Nat. Acad. Sci. USA 2002, 99, 9278–9283. [Google Scholar] [CrossRef] [PubMed]
- Tokai, T.; Fujimura, M.; Inoue, H.; Aoki, T.; Ohta, K.; Shibata, T.; Yamaguchi, I.; Kimura, M. Concordant evolution of trichothecene 3-O-acetyltransferase and an rDNA species phylogeny of trichothecene-producing and non-producing fusaria and other ascomycetous fungi. Microbiology 2005, 151, 509–519. [Google Scholar] [CrossRef] [PubMed]
- Khatibi, P.A.; Newmister, S.A.; Rayment, I.; McCormick, S.P.; Alexander, N.J.; Schmale, D.G. Bioprospecting for trichothecene 3-O-acetyltransferases in the fungal genus Fusarium yields functional enzymes with different abilities to modify the mycotoxin deoxynivalenol. Appl. Environ. Microbiol. 2011, 77, 1162–1170. [Google Scholar] [CrossRef]
- Lind, A.L.; Wisecaver, J.H.; Lameiras, C.; Wiemann, P.; Palmer, J.M.; Keller, N.P.; Rodrigues, F.; Goldman, G.H.; Rokas, A. Drivers of genetic diversity in secondary metabolic gene clusters within a fungal species. PLoS Biol. 2017, 15, e2003583. [Google Scholar] [CrossRef] [PubMed]
- Villani, A.; Moretti, A.; De Saeger, S.; Han, Z.; Di Mavungu, J.D.; Soares, C.M.; Proctor, R.H.; Venâncio, A.; Lima, N.; Stea, G. A polyphasic approach for characterization of a collection of cereal isolates of the Fusarium incarnatum-equiseti species complex. Int. J. Food Microbiol. 2016, 234, 24–35. [Google Scholar] [CrossRef] [PubMed]
- Castellá, G.; Cabañes, F. Phylogenetic diversity of Fusarium incarnatum-equiseti species complex isolated from Spanish wheat. Antonie Van Leeuwenhoek 2014, 106, 309–317. [Google Scholar] [CrossRef] [PubMed]
- Ramdial, H.; Hosein, F.; Rampersad, S.N. First report of Fusarium incarnatum associated with fruit disease of bell peppers in Trinidad. Plant Dis. 2016, 100, 526. [Google Scholar] [CrossRef]
- Wisecaver, J.H.; Rokas, A. Fungal metabolic gene clusters-caravans traveling across genomes and environments. Front. Microbiol. 2015, 6, 161. [Google Scholar] [CrossRef]
- Lee, T.; Oh, D.-W.; Kim, H.-S.; Lee, J.; Kim, Y.-H.; Yun, S.-H.; Lee, Y.-W. Identification of deoxynivalenol-and nivalenol-producing chemotypes of Gibberella zeae by using PCR. Appl. Environ. Microbiol. 2001, 67, 2966–2972. [Google Scholar] [CrossRef]
- Ward, T.J.; Clear, R.M.; Rooney, A.P.; O’Donnell, K.; Gaba, D.; Patrick, S.; Starkey, D.E.; Gilbert, J.; Geiser, D.M.; Nowicki, T.W. An adaptive evolutionary shift in Fusarium head blight pathogen populations is driving the rapid spread of more toxigenic Fusarium graminearum in North America. Fungal Genet. Biol. 2008, 45, 473–484. [Google Scholar] [CrossRef]
- Kulik, T. Development of TaqMan assays for 3ADON, 15ADON and NIV Fusarium genotypes based on Tri12 gene. Cereal Res. Comm. 2011, 39, 200–214. [Google Scholar] [CrossRef]
- Nielsen, L.K.; Jensen, J.D.; Rodríguez, A.; Jørgensen, L.; Justesen, A. TRI12 based quantitative real-time PCR assays reveal the distribution of trichothecene genotypes of F. graminearum and F. culmorum isolates in Danish small grain cereals. Int. J. Food Microbiol. 2012, 157, 384–392. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.-H.; Li, H.-P.; Qu, B.; Zhang, J.-B.; Huang, T.; Chen, F.-F.; Liao, Y.-C. Development of a generic PCR detection of 3-acetyldeoxynivalenol-, 15-acetyldeoxynivalenol-and nivalenol-chemotypes of Fusarium graminearum clade. Int. J. Mol. Sci. 2008, 9, 2495–2504. [Google Scholar] [CrossRef] [PubMed]
- Desjardins, A.E.; Busman, M.; Manandhar, G.; Jarosz, A.M.; Manandhar, H.K.; Proctor, R.H. Gibberella ear rot of maize (Zea mays) in Nepal: Distribution of the mycotoxins nivalenol and deoxynivalenol in naturally and experimentally infected maize. J. Agric. Food Chem. 2008, 56, 5428–5436. [Google Scholar] [CrossRef] [PubMed]
- Kimura, M.; Tokai, T.; Takahashi-Ando, N.; Ohsato, S.; Fujimura, M. Molecular and genetic studies of Fusarium trichothecene biosynthesis: Pathways, genes, and evolution. Biosci. Biotechnol. Biochem. 2007, 71, 2105–2123. [Google Scholar] [CrossRef] [PubMed]
- Pasquali, M.; Giraud, F.; Brochot, C.; Cocco, E.; Hoffmann, L.; Bohn, T. Genetic Fusarium chemotyping as a useful tool for predicting nivalenol contamination in winter wheat. Int. J. Food Microbiol. 2010, 137, 246–253. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Zhang, Z.; Van der Lee, T.; Chen, W.; Xu, J.; Xu, J.; Yang, L.; Yu, D.; Waalwijk, C.; Feng, J. Population genetic analyses of Fusarium asiaticum populations from barley suggest a recent shift favoring 3ADON producers in southern China. Phytopathology 2010, 100, 328–336. [Google Scholar] [CrossRef]
- Mugrabi de Kuppler, A.; Steiner, U.; Sulyok, M.; Krska, R.; Oerke, E.-C. Genotyping and phenotyping of Fusarium graminearum isolates from Germany related to their mycotoxin biosynthesis. Int. J. Food Microbiol. 2011, 151, 78–86. [Google Scholar] [CrossRef]
- Castañares, E.; Albuquerque, D.R.; Dinolfo, M.I.; Pinto, V.F.; Patriarca, A.; Stenglein, S.A. Trichothecene genotypes and production profiles of Fusarium graminearum isolates obtained from barley cultivated in Argentina. Int. J. Food Microbiol. 2014, 179, 57–63. [Google Scholar] [CrossRef]
- Caspi, R.; Altman, T.; Billington, R.; Dreher, K.; Foerster, H.; Fulcher, C.A.; Holland, T.A.; Keseler, I.M.; Kothari, A.; Kubo, A.; et al. The metacyc database of metabolic pathways and enzymes and the biocyc collection of pathway/genome databases. Nucleic Acids Res. 2014, 42, D459–D471. [Google Scholar] [CrossRef]
- Bilska, K.; Jurczak, S.; Kulik, T.; Ropelewska, E.; Olszewski, J.; Żelechowski, M.; Zapotoczny, P. Species composition and trichothecene genotype profiling of Fusarium field isolates recovered from wheat in Poland. Toxins 2018, 10, 325. [Google Scholar] [CrossRef]
- Wei, S.; van der Lee, T.; Verstappen, E.; van Gent, M.; Waalwijk, C. Targeting Trichothecene Biosynthetic Genes. In Mycotoxigenic Fungi: Methods and Protocols; Antonio, M., Antonia, S., Eds.; Springer Science and Business Media: Berlin, Germany, 2017; Volume 1542, pp. 173–189. [Google Scholar]
- Quarta, A.; Mita, G.; Haidukowski, M.; Santino, A.; Mule, G.; Visconti, A. Assessment of trichothecene chemotypes of Fusarium culmorum occurring in Europe. Food Addit. Contam. 2005, 22, 309–315. [Google Scholar] [CrossRef] [PubMed]
- Edwards, S.; Pirgozliev, S.; Hare, M.; Jenkinson, P. Quantification of trichothecene-producing Fusarium species in harvested grain by competitive PCR to determine efficacies of fungicides against Fusarium head blight of winter wheat. Appl. Environ. Microbiol. 2001, 67, 1575–1580. [Google Scholar] [CrossRef]
- Wang, J.-H.; Zhang, J.-B.; Chen, F.-F.; Li, H.-P.; Ndoye, M.; Liao, Y.-C. A multiplex PCR assay for genetic chemotyping of toxigenic Fusarium graminearum and wheat grains for 3-acetyldeoxynivalenol, 15-acetyldeoxynivalenol and nivalenol mycotoxins. J. Food Agric. Environ 2012, 10, 505–511. [Google Scholar]
- Chandler, E.A.; Simpson, D.R.; Thomsett, M.A.; Nicholson, P. Development of PCR assays to Tri7 and Tri13 trichothecene biosynthetic genes, and characterisation of chemotypes of Fusarium graminearum, Fusarium culmorum and Fusarium cerealis. Physiol. Mol. Plant Pathol. 2003, 62, 355–367. [Google Scholar] [CrossRef]
- Niessen, M.L.; Vogel, R.F. Group Specific PCR-detection of potential trichothecene-producing Fusarium-species in pure cultures and cereal samples. Syst. Appl. Microbiol. 1998, 21, 618–631. [Google Scholar] [CrossRef]
- Bluhm, B.; Flaherty, J.; Cousin, M.; Woloshuk, C. Multiplex polymerase chain reaction assay for the differential detection of trichothecene-and fumonisin-producing species of Fusarium in cornmeal. J. Food Prot. 2002, 65, 1955–1961. [Google Scholar] [CrossRef]
- Talas, F.; Parzies, H.K.; Miedaner, T. Diversity in genetic structure and chemotype composition of Fusarium graminearum sensu stricto populations causing wheat head blight in individual fields in Germany. Eur. J. Plant Pathol. 2011, 131, 39–48. [Google Scholar] [CrossRef]
- Talas, F.; Kalih, R.; Miedaner, T. Within-field variation of Fusarium graminearum isolates for aggressiveness and deoxynivalenol production in wheat head blight. Phytopathology 2012, 102, 128–134. [Google Scholar] [CrossRef]
- Kim, H.-S.; Lee, T.; Dawlatana, M.; Yun, S.-H.; Lee, Y.-W. Polymorphism of trichothecene biosynthesis genes in deoxynivalenol-and nivalenol-producing Fusarium graminearum isolates. Mycol. Res. 2003, 107, 190–197. [Google Scholar] [CrossRef]
- Waalwijk, C.; Kohl, J.; De Vries, I.; Van Der Lee, T. Fusarium in Winter Tarwe (in 2007 en 2008); Plant Research International: Wageningen, The Netherlands, 2009; Volume 272, pp. 1–36. [Google Scholar]
- Pasquali, M.; Beyer, M.; Bohn, T.; Hoffmann, L. Comparative analysis of genetic chemotyping methods for Fusarium: Tri13 polymorphism does not discriminate between 3-and 15-acetylated deoxynivalenol chemotypes in Fusarium graminearum. J. Phytopathol. 2011, 159, 700–704. [Google Scholar] [CrossRef]
- Suzuki, F.; Koba, A.; Nakajima, T. Simultaneous identification of species and trichothecene chemotypes of Fusarium asiaticum and F. graminearum sensu stricto by multiplex PCR. J. Gen. Plant Pathol. 2010, 76, 31–36. [Google Scholar] [CrossRef]
- Brown, D.W.; McCormick, S.P.; Alexander, N.J.; Proctor, R.H.; Desjardins, A.E. Inactivation of a cytochrome P-450 is a determinant of trichothecene diversity in Fusarium species. Fungal Genet. Biol. 2002, 36, 224–233. [Google Scholar] [CrossRef]
- Starkey, D.E.; Ward, T.J.; Aoki, T.; Gale, L.R.; Kistler, H.C.; Geiser, D.M.; Suga, H.; Toth, B.; Varga, J.; O’Donnell, K. Global molecular surveillance reveals novel Fusarium head blight species and trichothecene toxin diversity. Fungal Genet. Biol. 2007, 44, 1191–1204. [Google Scholar] [CrossRef]
- Wilhelm, M.; Schlegl, J.; Hahne, H.; Gholami, A.M.; Lieberenz, M.; Savitski, M.M.; Ziegler, E.; Butzmann, L.; Gessulat, S.; Marx, H. Mass-spectrometry-based draft of the human proteome. Nature 2014, 509, 582. [Google Scholar] [CrossRef] [PubMed]
- Fortelny, N.; Overall, C.M.; Pavlidis, P.; Cohen Freue, G.V.C. Can we predict protein from mRNA levels? Nature 2017, 547, 19–20. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Beyer, A.; Aebersold, R. On the dependency of cellular protein levels on mRNA abundance. Cell 2016, 165, 535–550. [Google Scholar] [CrossRef] [PubMed]
- Marroquín-Cardona, A.; Johnson, N.; Phillips, T.; Hayes, A. Mycotoxins in a changing global environment—A review. Food Chem. Toxicol. 2014, 69, 220–230. [Google Scholar] [CrossRef] [PubMed]
- Pasquali, M.; Cocco, E.; Guignard, C.; Hoffmann, L. The effect of agmatine on trichothecene type B and zearalenone production in Fusarium graminearum, F. culmorum and F. poae. Peer J. 2016, 4, 1–11. [Google Scholar] [CrossRef]
- Medina, A.; Akbar, A.; Baazeem, A.; Rodriguez, A.; Magan, N. Climate change, food security and mycotoxins: Do we know enough? Fungal Biol. Rev. 2017, 31, 143–154. [Google Scholar] [CrossRef]
- Moretti, A.; Pascale, M.; Logrieco, A.F. Mycotoxin risks under a climate change scenario in Europe. Trends Food Sci. Technol. 2018. [Google Scholar] [CrossRef]
- ISPA CNR Institute of Sciences of Food Production. MYCORED PROJECT. Available online: http://www.mycored.eu/ (accessed on 16 November 2018).
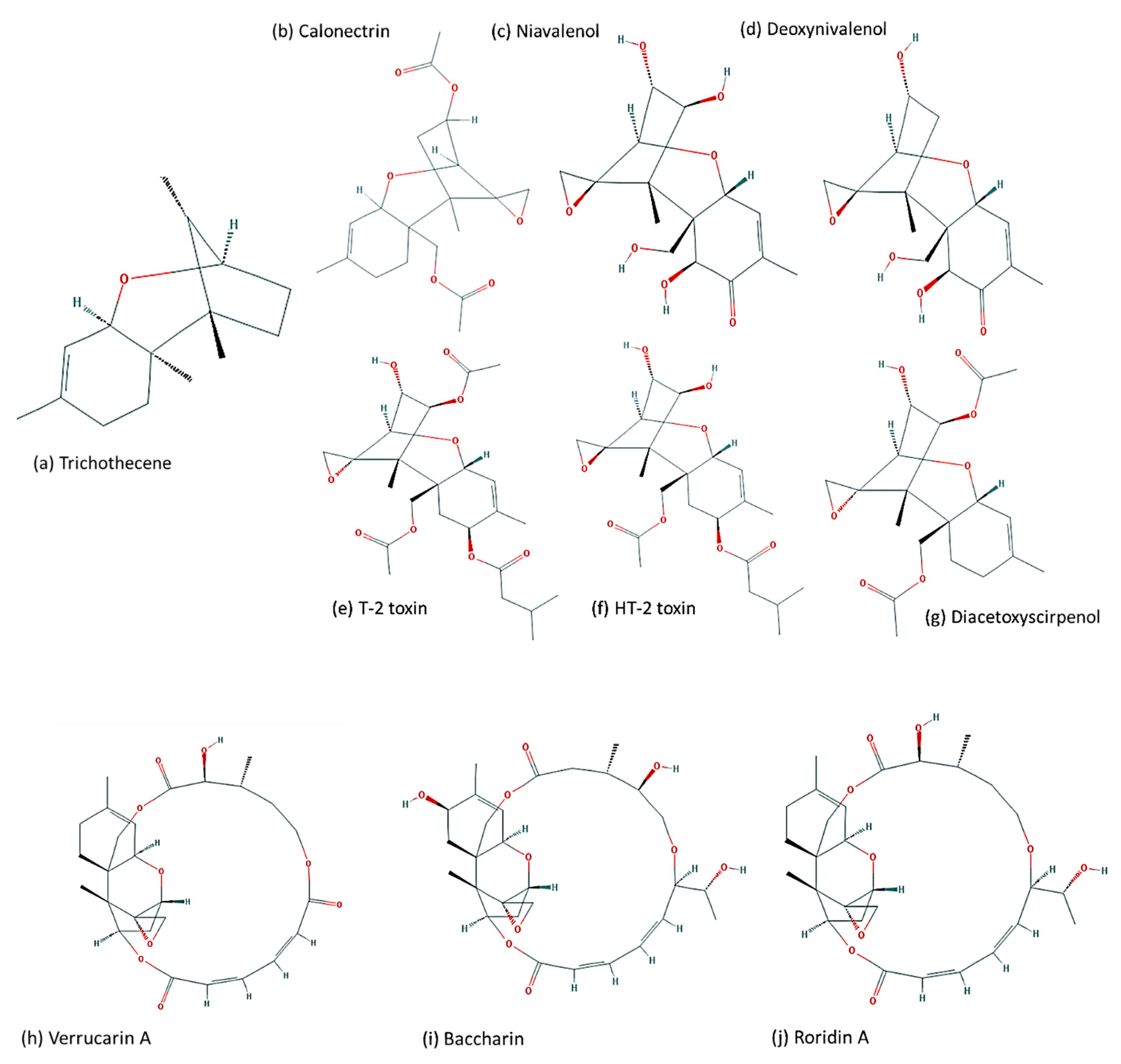
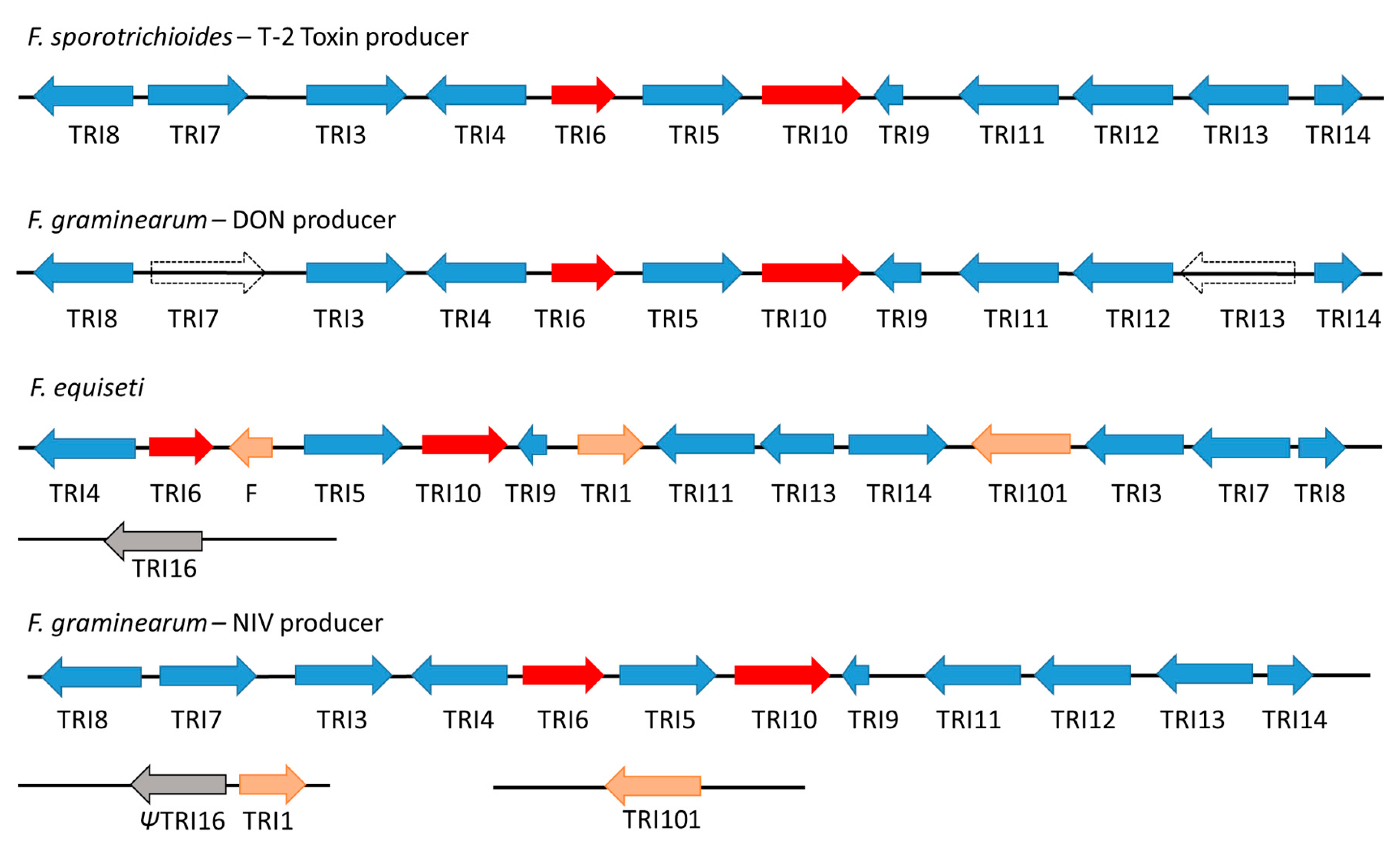
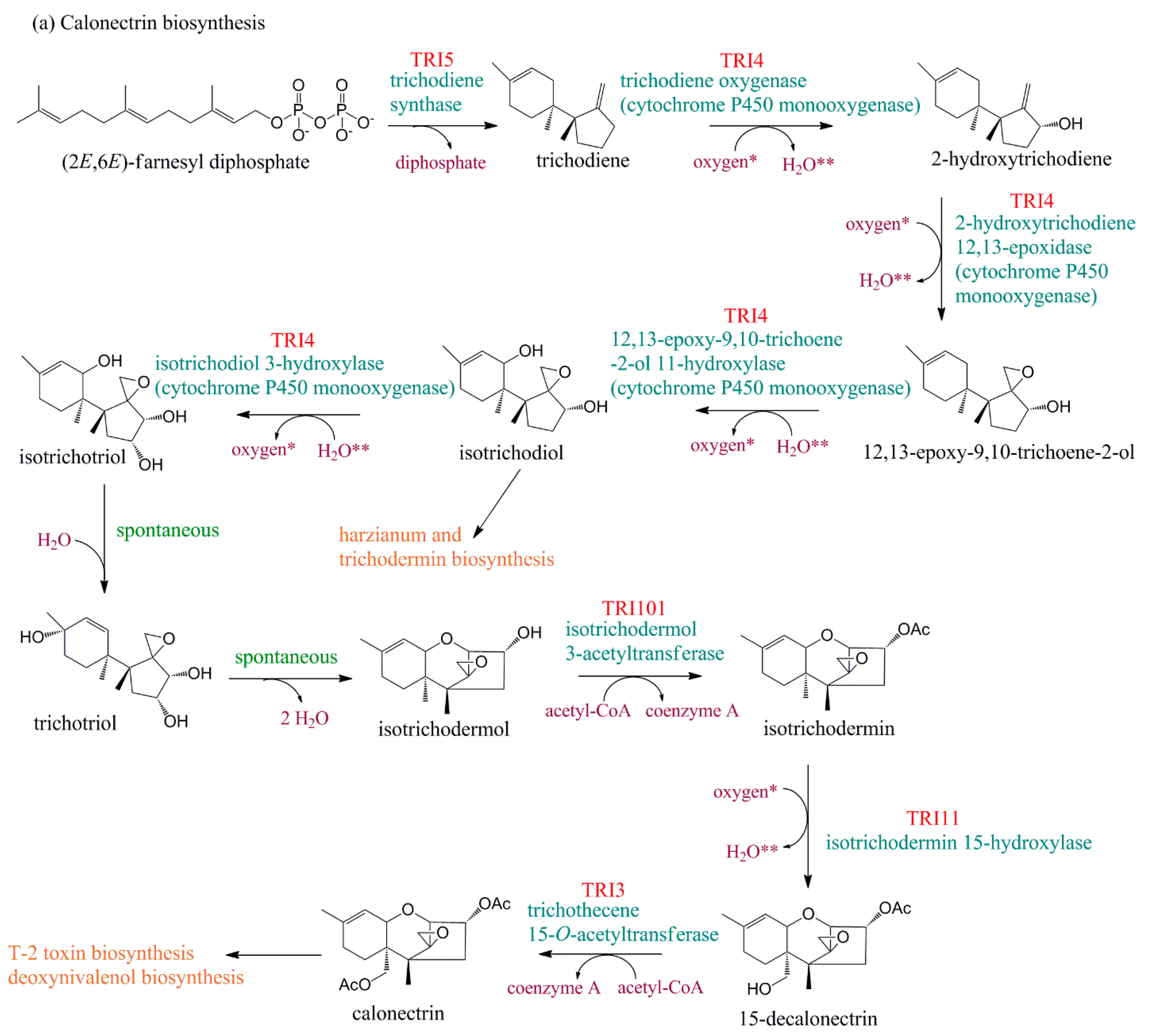
| Gene ID | Locus Tag | Gene | Protein Annotation (GenBank) | Protein Annotation [26] |
|---|---|---|---|---|
| Gene ID: 23550835 | FGSG_03532 | TRI8 | trichothecene 3-O-esterase | esterase |
| Gene ID: 23550836 | FGSG_03533 | TRI7 | hypothetical protein | acetyl transferase |
| Gene ID: 23550837 | FGSG_03534 | TRI3 | hypothetical protein | acetyl transferase |
| Gene ID: 23550838 | FGSG_03535 | TRI4 | trichodiene oxygenase | cytochrome P450 monooxygenase |
| Gene ID: 23550839 | FGSG_03536 | TRI6 | transcription factor | Zn2His2 transcription factor |
| Gene ID: 23550840 | FGSG_03537 | TRI5 | trichodiene synthase | terpene synthase |
| Gene ID: 23550841 | FGSG_03538 | TRI10 | transcription factor | transcriptional regulator |
| Gene ID: 23550842 | FGSG_03539 | TRI9 | hypothetical protein | unknown |
| Gene ID: 23550843 | FGSG_03540 | TRI11 | isotrichodermin C-15 hydroxylase | cytochrome P450 monooxygenase |
| Gene ID: 23550844 | FGSG_03541 | TRI12 | trichothecene efflux pump | major facilitator superfamily transporter |
| Gene ID: 23550845 | FGSG_03542 | TRI13 | hypothetical protein | cytochrome P450 monooxygenase |
| Gene ID: 23550846 | FGSG_03543 | TRI14 | hypothetical protein | unknown |
| NA | NA | TRI16 | NA | acyl transferase |
| NA | NA | TRI101 | NA | acetyl transferase |
| TRI Gene | Gene Function | Nucleotide (nt) Sequence % Similarity | Amino Acid (aa) Sequence % Similarity | ||||
|---|---|---|---|---|---|---|---|
| nt Sequence Length 1 | Among Species 2 | Within Species | aa Sequence Length | Among Species | Within Species | ||
| TRI8 | esterase | 625; 802 | 89–100%/N = 21 | 99–100%/N = 4 | 267 | 76–100% | 98–100% |
| TRI7 | acetyl transferase | 1045 | 94–100%/N = 19 | 98–100%/N = 18; 30 | 331 | 73–100% | 98–100% |
| TRI3 | acetyl transferase | 664; 809 | 85–100%/N = 17 | 99–100%/N = 23; 4 | 170 | 85–100% | 99–100% |
| TRI4 | cytochrome P450 monooxygenase | 1186; 1217 | 75–100/N = 13; 15 | 94–100%/N = 4 | 368 | 86–100% | 98–100% |
| TRI6 | Zn2His2 transcription factor | 450 | 84–100%/N = 18 | 98–100%/N = 18; 30 | 600 | 87–100% | 98–100% |
| TRI5 | terpene synthase | 284; 504 | 77–100%/N = 41 | 89–100%/N = 13; 16; 59 | 111 | 80–100% | 98–100% |
| TRI10 | transcriptional regulator | 89–100%/N = 39 | 420 | 70–100% | 98–100% | ||
| TRI9 | unknown | *** | *** | *** | *** | *** | *** |
| TRI11 | cytochrome P450 monooxygenase | 334 | 89–100%/N = 39 | 94–100%/N = 33 | 147 | 90–100% | 98–100% |
| TRI12 | major facilitator superfamily transporter/efflux pump | 1967 | *** | 99–100%/N = 4 | 600 | 87–100% | 97–100% |
| TRI13 | cytochrome P450 monooxygenase | 1010; 1060 | 92–100%/N = 13 | 99–100%/N = 4 | 315 | 76–100% | 98–100% |
| TRI14 | unknown | 658; 660 | 75–100%/N = 41 | 97–100%/N = 4; 11; 8 | 202 | 89–100% | 99–100% |
| TRI16 | acyl transferase | 1143 | 90–100%/N = 8 | 99–100%/N = 4 | 381 | 60–100% | 99–100% |
| TRI101 | acetyl transferase | 925; 1244 | 96–100%/N = 17; 33 | 99–100%/N = 11; 8; 6 | 444 | 79–100% | 99–100% |
| TRI1 | cytochrome P450 monooxygenase | 664; 1755 | 94–100%/N = 8 | 99–100%/N = 100 | 512 | 85–100% | 98–100% |
| Gene Target | Primer Name | Primer Sequence 5′ to 3′ | Amplicon Length/bp | Target Species | Reference |
|---|---|---|---|---|---|
| TRI3 | 3_CONS 1 | TGGCAAAGACTGGTTCAC | F. graminearum, F. asiaticum | [79] | |
| TRI3 | 3_NIV_F | GTGCACAGAATATACGAGC | 840 | F. graminearum, F. asiaticum | [79] |
| TRI3 | 3_15ADON_F | ACTGACCCAAGCTGCCATC | 610 | F. graminearum, F. asiaticum | [79] |
| TRI3 | 3_3ADON_F | CGCATTGGCTAACACATG | 243 | F. graminearum, F. asiaticum | [79] |
| TRI12 | 12_CONS | CATGAGCATGGTGATGTC | F. graminearum, F. asiaticum | [79] | |
| TRI12 | 12_NIV_F | TCTCCTCGTTGTATCTGG | 840 | F. graminearum, F. asiaticum | [79] |
| TRI12 | 12_15ADON_F | TACAGCGGTCGCAACTTC | 670 | F. graminearum, F. asiaticum | [79] |
| TRI12 | 12_3ADON_F | CTTTGGCAAGCCCGTGCA | 410 | F. graminearum, F. asiaticum | [79] |
| TRI3 | Tri3F971 | CATCATACTCGCTCTGCTG | 708for 15-ADON producers only | F. graminearum, F. culmorum, F. cerealis | [80] |
| Tri3F1325 | GCATTGGCTAACACATGA | 354for 3-ADON producers only | F. graminearum, F. culmorum, F. cerealis | [80] | |
| Tri3R1679 | TT(A/G)TAGTTTGCA TCATT(A/G)TAG | F. graminearum, F. culmorum, F. cerealis | [80] | ||
| Gene F | 3891 | GCTGTCAYAGYCAGAAGYTACGATG | 1200 | Fusarium incarnatum equiseti species complex | [62] |
| 3894 | AGAYATGBAGGACARGGCTTAGGGT | Fusarium incarnatum equiseti species complex | [62] | ||
| TRI1 | 1285 | GCGTCTCAGCTTCATCAAGGCAKCKAMTGAWTCG | 1200 | F. graminearum, F. sporotrichioides | [30] |
| 1292 | CTTGACTTSMTTGGCKGCAAAGAARCGACCA | F. graminearum, F. sporotrichioides | [30] | ||
| TRI3 | 1912 | TGTGTMGGYGCWGAGGCVATYGTTGG | F. graminearum, F. sporotrichioides | [30] | |
| 1914 | ACRGCAGCRGTCTGRCACATGGCGTA | F. graminearum, F. sporotrichioides | [30] | ||
| TRI4 | 2576 | CCAATCAGYCAYGCTRTTGGGATACTG | 1800 | F. graminearum, F. sporotrichioides | [30] |
| 2578 | ACCCGGATTTCRCCAACATGCT | F. graminearum, F. sporotrichioides | [30] | ||
| TRI5 | 1558 | GGCATGGTCGTGTACTCTTGGGTCAAGGT | 1300 | F. graminearum, F. sporotrichioides | [30] |
| 1559 | GCCTGMYCAWAGAAYTTGCRGAACTT | F. graminearum, F. sporotrichioides | [30] | ||
| TRI8 | 3904 | GACCAGNAYCACSGYCAACAGTTCAG | 1200 | Fusarium incarnatum equiseti species complex | [62] |
| 3906 | GAACAGCCRCTCCRWAACTATTGTC | Fusarium incarnatum equiseti species complex | [62] | ||
| TRI11 | 3895 | TWCCCCACAAGRAACAYCTYGARCT | 1300 | Fusarium incarnatum equiseti species complex | [62] |
| 3897 | TCCCASACTGTYCTSGCMAGCATCAT | Fusarium incarnatum equiseti species complex | [62] | ||
| TRI16 | 1472b | CCTCTCTCCCCTTGAYCAATTRAACTCT | NA | F. graminearum, F. sporotrichioides | [30] |
| 1473b | CTTCCCGATCCCRAYGAGCCTCTTACAC | F. graminearum, F. sporotrichioides | [30] | ||
| 1474b | GCCTTATMTKGGTAATGTCGTGCTKACA | F. graminearum, F. sporotrichioides | [30] | ||
| 1475b | AAGAGGCTCRTYGGGATCGGGAAGGTTC | F. graminearum, F. sporotrichioides | [30] | ||
| 1476b | CARCCGACGATGTMAGCACGACATTACC | F. graminearum, F. sporotrichioides | [30] | ||
| 1477b | CAATATACGGATACCGCACAAAGACTGG | F. graminearum, F. sporotrichioides | [30] | ||
| TRI101 2 | 109 | CCATGGGTCGCRGGCCARGTSAA | NA | F. graminearum, F. sporotrichioides | [30] |
| 178 | AACTCSCCRTCIGGYTTYTTNGGCAT | F. graminearum, F. sporotrichioides | [30] | ||
| TRI5 | HATri/F | CAGATGGAGAACTGGATGGT | 260 | F. culmorum, F. poae, F. sporotrichioides, F. graminearum, F. sambucinum | [81] |
| HATri/R | GCACAAGTGCCACGTGAC | F. culmorum, F. poae, F. sporotrichioides, F. graminearum, F. sambucinum | [81] | ||
| TRI11 | N11 | CTTGTCAGGCGGCACAGTAG | 643 for NIV-producers | F. asiaticum, F. mesoamericanum, F. cortaderiae, F. gerlachii, F. meridionale × F. asiaticum, F. meridionale, F. lunulosporum, F. cerealis, F. vorosii, F. aethiopicum, F. graminearum, F. boothii, F. asiaticum, F. brasilicum, F. austroamericanum, F. culmorum, F. pseudograminearum | [74] |
| 15D11 | AAGTATGGTCCAGTTGTCCGTATT | 424 for 3-ADON producers | F. asiaticum, F. mesoamericanum, F. cortaderiae, F. gerlachii, F. meridionale × F. asiaticu), F. meridionale, F. lunulosporum, F. cerealis, F. vorosii, F. aethiopicum, F. graminearum, F. boothii, F. asiaticum, F. brasilicum, F. austroamericanum, F. culmorum, F. pseudograminearum | [74] | |
| 3D11 | GCAA GTCTGGCGAGGCC | 342 for 15-ADON producers | F. asiaticum, F. mesoamericanum, F. cortaderiae, F. gerlachii, F. meridionale × F. asiaticu), F. meridionale, F. lunulosporum, F. cerealis, F. vorosii, F. aethiopicum, F. graminearum, F. boothii, F. asiaticum, F. brasilicum, F. austroamericanum, F. culmorum, F. pseudograminearum | [74] | |
| 11R | TCAAAGGCCAGAGCA ACCC | F. asiaticum, F. mesoamericanum, F. cortaderiae, F. gerlachii, F. meridionale × F. asiaticu), F. meridionale, F. lunulosporum, F. cerealis, F. vorosii, F. aethiopicum, F. graminearum, F. boothii, F. asiaticum, F. brasilicum, F. austroamericanum, F. culmorum, F. pseudograminearum | [74] | ||
| TRI11 | Tri11-CON | GACTGCTCATGGAGACGCTG | NA | F. graminearum | [82] |
| Tri11-3AcDON | TCCTCATGCTCG GTGGACTCG | 334 | F. graminearum | [82] | |
| Tri11-15AcDON | TGGTCCAGT TGTCCGTATT | 279 | F. graminearum | [82] | |
| Tri11-NIV | GTAGGTTCCATTGC TTGTTC | 497 | F. graminearum | [82] | |
| TRI7 | GzTri7/f1 | GGCTTTACGACTCCTCAACAATGG | ∼160 | F. graminearum | [66] |
| GzTri7/r1 | AGAGCCCTGCGAAAG(C/T)ACTGGTGC | F. graminearum | [66] | ||
| TRI7 | Tri7F340 | ATCGTGTACAAGGTTTACG | 625 | F. graminearum, F. culmorum, F. cerealis | [80] |
| Tri7R965 | TTCAAGTAACGTTCGACAAT | F. graminearum, F. culmorum, F. cerealis | [80] | ||
| TRI13 | Tri13F | CATCATGAGACTTGTKCRGTTTGGG | 1075 for NIV producers; 799 for DON-producers | F. graminearum, F. culmorum, F. cerealis | [83] |
| Tri13DONR | GCTAGATCGATTGTTGCATTGAG | 282 for DON producers | F. graminearum, F. culmorum, F. cerealis | [83] | |
| Tri13R | TTGAAAGCTCCAATGTCGTG | F. graminearum, F. culmorum, F. cerealis | [83] | ||
| Tri13NIVF | CCAAATCCGAAAACCGCAG | 312 for NIV producers | F. graminearum, F. culmorum, F. cerealis | [83] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Villafana, R.T.; Ramdass, A.C.; Rampersad, S.N. Selection of Fusarium Trichothecene Toxin Genes for Molecular Detection Depends on TRI Gene Cluster Organization and Gene Function. Toxins 2019, 11, 36. https://doi.org/10.3390/toxins11010036
Villafana RT, Ramdass AC, Rampersad SN. Selection of Fusarium Trichothecene Toxin Genes for Molecular Detection Depends on TRI Gene Cluster Organization and Gene Function. Toxins. 2019; 11(1):36. https://doi.org/10.3390/toxins11010036
Chicago/Turabian StyleVillafana, Ria T., Amanda C. Ramdass, and Sephra N. Rampersad. 2019. "Selection of Fusarium Trichothecene Toxin Genes for Molecular Detection Depends on TRI Gene Cluster Organization and Gene Function" Toxins 11, no. 1: 36. https://doi.org/10.3390/toxins11010036
APA StyleVillafana, R. T., Ramdass, A. C., & Rampersad, S. N. (2019). Selection of Fusarium Trichothecene Toxin Genes for Molecular Detection Depends on TRI Gene Cluster Organization and Gene Function. Toxins, 11(1), 36. https://doi.org/10.3390/toxins11010036





