Generation of a Broadly Cross-Neutralizing Antibody Fragment against Several Mexican Scorpion Venoms
Abstract
1. Introduction
2. Results
2.1. In Vitro Maturation of Antibody Fragments
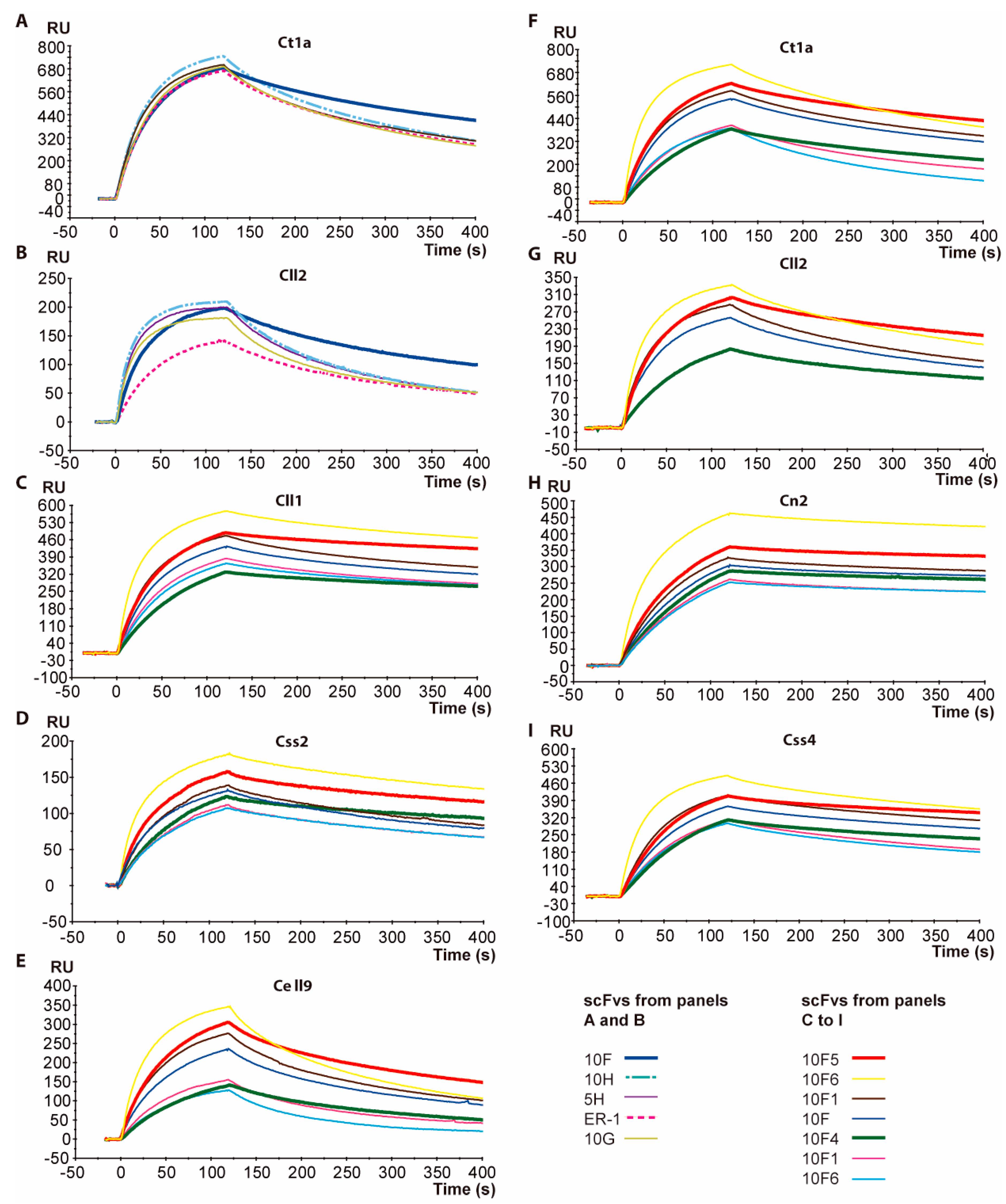
2.2. Affinity Determination of scFv 10FG2
2.3. Toxin Neutralization Assays
2.4. Venom Neutralization and Rescue Assays
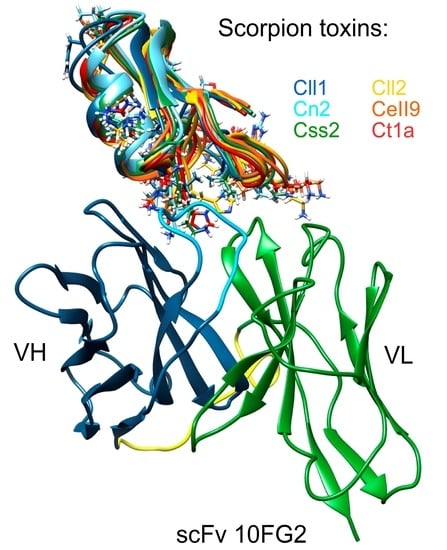
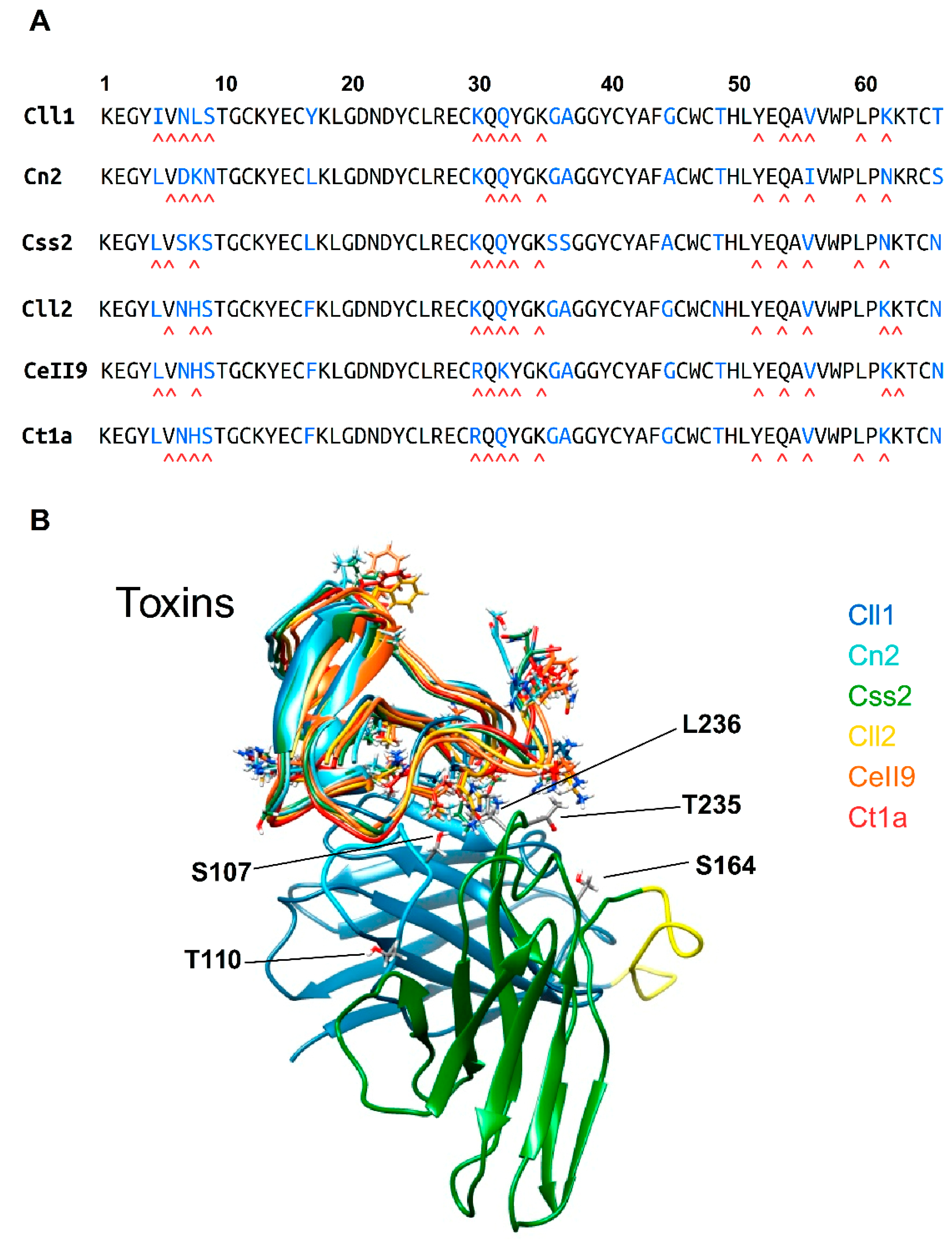
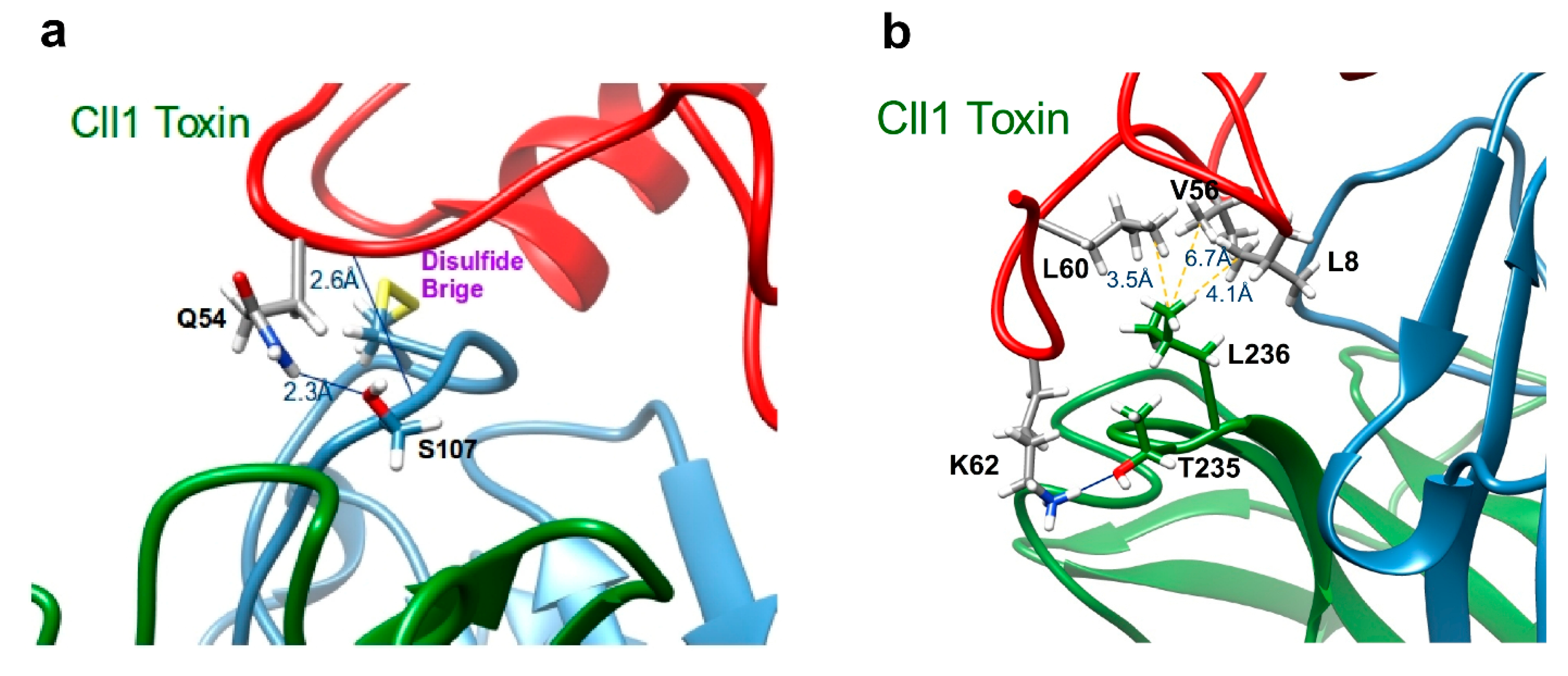
2.5. Structural Analysis of the Interaction of scFv 10FG2 with Toxins
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. In Vitro Maturation
5.1.1. Library of scFv ER-1 by Site-Directed Saturation Mutagenesis
5.1.2. Biopanning Process and Evaluation
5.1.3. Combinatorial Mutagenesis; Oligo Design and Construction of Variants of scFv 10F
5.2. Comparison of the scFv Binding Properties by Surface Plasmon Resonance
5.3. Surface Plasmon Resonance Evaluations
5.4. Toxin Neutralization Assays
5.6. Neutralization Assays with Whole Fresh Scorpion Venoms
5.6.1. Classical Protection
5.6.2. Rescue Test
5.7. Modeling and Structural Analyses of scFv 10FG2–Toxin Complexes
6. Patents
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Riano-Umbarila, L.; Rodriguez-Rodriguez, E.R.; Santibanez-Lopez, C.E.; Guereca, L.; Uribe-Romero, S.J.; Gomez-Ramirez, I.V.; Carcamo-Noriega, E.N.; Possani, L.D.; Becerril, B. Updating knowledge on new medically important scorpion species in Mexico. Toxicon 2017, 138, 130–137. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez de la Vega, R.C.; Possani, L.D. Overview of scorpion toxins specific for Na+ channels and related peptides: Biodiversity, structure-function relationships and evolution. Toxicon 2005, 46, 831–844. [Google Scholar] [CrossRef] [PubMed]
- Possani, L.D.; Becerril, B.; Delepierre, M.; Tytgat, J. Scorpion toxins specific for Na+-channels. Eur. J. Biochem. 1999, 264, 287–300. [Google Scholar] [CrossRef] [PubMed]
- Chippaux, J.P. Emerging options for the management of scorpion stings. Drug Des. Dev. Ther. 2012, 6, 165–173. [Google Scholar] [CrossRef] [PubMed]
- Espino-Solis, G.P.; Riano-Umbarila, L.; Becerril, B.; Possani, L.D. Antidotes against venomous animals: State of the art and prospectives. J. Proteom. 2009, 72, 183–199. [Google Scholar] [CrossRef] [PubMed]
- Schiavon, E.; Sacco, T.; Cassulini, R.R.; Gurrola, G.; Tempia, F.; Possani, L.D.; Wanke, E. Resurgent current and voltage sensor trapping enhanced activation by a beta-scorpion toxin solely in Nav1.6 channel. Significance in mice Purkinje neurons. J. Biol. Chem. 2006, 281, 20326–20337. [Google Scholar] [CrossRef] [PubMed]
- Schiavon, E.; Pedraza-Escalona, M.; Gurrola, G.B.; Olamendi-Portugal, T.; Corzo, G.; Wanke, E.; Possani, L.D. Negative-shift activation, current reduction and resurgent currents induced by beta-toxins from Centruroides scorpions in sodium channels. Toxicon 2012, 59, 283–293. [Google Scholar] [CrossRef]
- Olamendi-Portugal, T.; Restano-Cassulini, R.; Riano-Umbarila, L.; Becerril, B.; Possani, L.D. Functional and immuno-reactive characterization of a previously undescribed peptide from the venom of the scorpion Centruroides limpidus. Peptides 2017, 87, 34–40. [Google Scholar] [CrossRef]
- Kini, R.M.; Sidhu, S.S.; Laustsen, A.H. Biosynthetic Oligoclonal Antivenom (BOA) for Snakebite and Next-Generation Treatments for Snakebite Victims. Toxins 2018, 10, 534. [Google Scholar] [CrossRef]
- Zamudio, F.; Saavedra, R.; Martin, B.M.; Gurrola-Briones, G.; Herion, P.; Possani, L.D. Amino acid sequence and immunological characterization with monoclonal antibodies of two toxins from the venom of the scorpion Centruroides noxius Hoffmann. Eur. J. Biochem. 1992, 204, 281–292. [Google Scholar] [CrossRef]
- Olamendi-Portugal, T.; Bartok, A.; Zamudio-Zuniga, F.; Balajthy, A.; Becerril, B.; Panyi, G.; Possani, L.D. Isolation, chemical and functional characterization of several new K+-channel blocking peptides from the venom of the scorpion Centruroides tecomanus. Toxicon 2016, 115, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Martin, M.F.; Garcia y Perez, L.G.; el Ayeb, M.; Kopeyan, C.; Bechis, G.; Jover, E.; Rochat, H. Purification and chemical and biological characterizations of seven toxins from the Mexican scorpion, Centruroides suffusus suffusus. J. Biol. Chem. 1987, 262, 4452–4459. [Google Scholar] [PubMed]
- Chen, Z.; Fischer, E.R.; Kouiavskaia, D.; Hansen, B.T.; Ludtke, S.J.; Bidzhieva, B.; Makiya, M.; Agulto, L.; Purcell, R.H.; Chumakov, K. Cross-neutralizing human anti-poliovirus antibodies bind the recognition site for cellular receptor. Proc. Natl. Acad. Sci. USA 2013, 110, 20242–20247. [Google Scholar] [CrossRef]
- Xu, H.; Schmidt, A.G.; O’Donnell, T.; Therkelsen, M.D.; Kepler, T.B.; Moody, M.A.; Haynes, B.F.; Liao, H.X.; Harrison, S.C.; Shaw, D.E. Key mutations stabilize antigen-binding conformation during affinity maturation of a broadly neutralizing influenza antibody lineage. Proteins 2015, 83, 771–780. [Google Scholar] [CrossRef] [PubMed]
- Rani, M.; Bolles, M.; Donaldson, E.F.; Van Blarcom, T.; Baric, R.; Iverson, B.; Georgiou, G. Increased antibody affinity confers broad in vitro protection against escape mutants of severe acute respiratory syndrome coronavirus. J. Virol. 2012, 86, 9113–9121. [Google Scholar] [CrossRef] [PubMed]
- Cockburn, J.J.; Navarro Sanchez, M.E.; Fretes, N.; Urvoas, A.; Staropoli, I.; Kikuti, C.M.; Coffey, L.L.; Arenzana Seisdedos, F.; Bedouelle, H.; Rey, F.A. Mechanism of dengue virus broad cross-neutralization by a monoclonal antibody. Structure 2012, 20, 303–314. [Google Scholar] [CrossRef] [PubMed]
- Sui, J.; Li, W.; Murakami, A.; Tamin, A.; Matthews, L.J.; Wong, S.K.; Moore, M.J.; Tallarico, A.S.; Olurinde, M.; Choe, H.; et al. Potent neutralization of severe acute respiratory syndrome (SARS) coronavirus by a human mAb to S1 protein that blocks receptor association. Proc. Natl. Acad. Sci. USA 2004, 101, 2536–2541. [Google Scholar] [CrossRef]
- Ekiert, D.C.; Kashyap, A.K.; Steel, J.; Rubrum, A.; Bhabha, G.; Khayat, R.; Lee, J.H.; Dillon, M.A.; O’Neil, R.E.; Faynboym, A.M.; et al. Cross-neutralization of influenza A viruses mediated by a single antibody loop. Nature 2012, 489, 526–532. [Google Scholar] [CrossRef]
- Ledsgaard, L.; Jenkins, T.P.; Davidsen, K.; Krause, K.E.; Martos-Esteban, A.; Engmark, M.; Rordam Andersen, M.; Lund, O.; Laustsen, A.H. Antibody Cross-Reactivity in Antivenom Research. Toxins 2018, 10, 393. [Google Scholar] [CrossRef]
- Riano-Umbarila, L.; Contreras-Ferrat, G.; Olamendi-Portugal, T.; Morelos-Juarez, C.; Corzo, G.; Possani, L.D.; Becerril, B. Exploiting cross-reactivity to neutralize two different scorpion venoms with one single chain antibody fragment. J. Biol. Chem. 2011, 286, 6143–6151. [Google Scholar] [CrossRef]
- Riano-Umbarila, L.; Olamendi-Portugal, T.; Morelos-Juarez, C.; Gurrola, G.B.; Possani, L.D.; Becerril, B. A novel human recombinant antibody fragment capable of neutralizing Mexican scorpion toxins. Toxicon 2013, 76, 370–376. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Rodriguez, E.R.; Olamendi-Portugal, T.; Serrano-Posada, H.; Arredondo-Lopez, J.N.; Gomez-Ramirez, I.; Fernandez-Taboada, G.; Possani, L.D.; Anguiano-Vega, G.A.; Riano-Umbarila, L.; Becerril, B. Broadening the neutralizing capacity of a family of antibody fragments against different toxins from Mexican scorpions. Toxicon 2016, 119, 52–63. [Google Scholar] [CrossRef]
- Riano-Umbarila, L.; Ledezma-Candanoza, L.M.; Serrano-Posada, H.; Fernandez-Taboada, G.; Olamendi-Portugal, T.; Rojas-Trejo, S.; Gomez-Ramirez, I.V.; Rudino-Pinera, E.; Possani, L.D.; Becerril, B. Optimal Neutralization of Centruroides noxius Venom Is Understood through a Structural Complex between Two Antibody Fragments and the Cn2 Toxin. J. Biol. Chem. 2016, 291, 1619–1630. [Google Scholar] [CrossRef] [PubMed]
- Martin, B.M.; Carbone, E.; Yatani, A.; Brown, A.M.; Ramirez, A.N.; Gurrola, G.B.; Possani, L.D. Amino acid sequence and physiological characterization of toxins from the venom of the scorpion Centruroides limpidus tecomanus Hoffmann. Toxicon 1988, 26, 785–794. [Google Scholar] [CrossRef]
- Riano-Umbarila, L.; Juarez-Gonzalez, V.R.; Olamendi-Portugal, T.; Ortiz-Leon, M.; Possani, L.D.; Becerril, B. A strategy for the generation of specific human antibodies by directed evolution and phage display. An example of a single-chain antibody fragment that neutralizes a major component of scorpion venom. FEBS J. 2005, 272, 2591–2601. [Google Scholar] [CrossRef] [PubMed]
- Pintar, A.; Possani, L.D.; Delepierre, M. Solution structure of toxin 2 from centruroides noxius Hoffmann, a beta-scorpion neurotoxin acting on sodium channels. J. Mol. Biol. 1999, 287, 359–367. [Google Scholar] [CrossRef]
- Saucedo, A.L.; del Rio-Portilla, F.; Picco, C.; Estrada, G.; Prestipino, G.; Possani, L.D.; Delepierre, M.; Corzo, G. Solution structure of native and recombinant expressed toxin CssII from the venom of the scorpion Centruroides suffusus suffusus, and their effects on Nav1.5 sodium channels. Biochim. Biophys. Acta 2012, 1824, 478–487. [Google Scholar] [CrossRef]
- Laustsen, A.H.; Maria Gutierrez, J.; Knudsen, C.; Johansen, K.H.; Bermudez-Mendez, E.; Cerni, F.A.; Jurgensen, J.A.; Ledsgaard, L.; Martos-Esteban, A.; Ohlenschlaeger, M.; et al. Pros and cons of different therapeutic antibody formats for recombinant antivenom development. Toxicon 2018, 146, 151–175. [Google Scholar] [CrossRef]
- Alagon, A.C.; Guzman, H.S.; Martin, B.M.; Ramirez, A.N.; Carbone, E.; Possani, L.D. Isolation and characterization of two toxins from the Mexican scorpion Centruroides limpidus limpidus Karsch. Comp. Biochem. Physiol. B 1988, 89, 153–161. [Google Scholar] [CrossRef]
- Ramirez, A.N.; Gurrola, G.B.; Martin, B.M.; Possani, L.D. Isolation of several toxins from the venom of the scorpion Centruroides limpidus tecomanus Hoffmann. Toxicon 1988, 26, 773–783. [Google Scholar] [CrossRef]
- Lin, K.; Simossis, V.A.; Taylor, W.R.; Heringa, J. A simple and fast secondary structure prediction method using hidden neural networks. Bioinformatics 2005, 21, 152–159. [Google Scholar] [CrossRef] [PubMed]
- Bowers, K.J.; Chou, D.E.; Xu, H.; Dror, R.O.; Eastwood, M.P.; Gregersen, B.A.; Klepeis, J.L.; Kolossvary, I.; Moraes, M.A.; Sacerdoti, F.D.; et al. Scalable algorithms for molecular dynamics simulations on commodity clusters. In Proceedings of the 2006 ACM/IEEE Conference on Supercomputing, Tampa, FL, USA, 11–17 November 2006; pp. 1–13. [Google Scholar]
- Tina, K.G.; Bhadra, R.; Srinivasan, N. PIC: Protein Interactions Calculator. Nucleic Acids Res. 2007, 35, W473–W476. [Google Scholar] [CrossRef] [PubMed]
- Schymkowitz, J.; Borg, J.; Stricher, F.; Nys, R.; Rousseau, F.; Serrano, L. The FoldX web server: An online force field. Nucleic Acids Res. 2005, 33, W382–W388. [Google Scholar] [CrossRef] [PubMed]
| Species | Toxin | Sequence | % |
|---|---|---|---|
| C. noxius | Cn2 | KEGYLVDKNTGCKYECLKLGDNDYCLRECKQQYGKGAGGYCYAFACWCTHLYEQAIVWPLPNKRCS | 6.8 |
| C. limpidus | Cll1 | KEGYIVNLSTGCKYECYKLGDNDYCLRECKQQYGKGAGGYCYAFGCWCTHLYEQAVVWPLPKKTCT | 1.5 |
| Cll2 | KEGYLVNHSTGCKYECFKLGDNDYCLRECKQQYGKGAGGYCYAFGCWCNHLYEQAVVWPLPKKTCN | 3.5 | |
| Cl13 | KEGYLVDYHTGCKYTCAKLGDNDYCVRECRLRYYQSAHGYCYAFACWCTHLYEQAVVRPLPNKRCR | 2.1 | |
| C. suffussus | Css2 | KEGYLVSKSTGCKYECLKLGDNDYCLRECKQQYGKSSGGYCYAFACWCTHLYEQAVVWPLPNKTCN | 2.8 |
| C. tecomanus | Ct1a | KEGYLVNHSTGCKYECFKLGDNDYCLRECRQQYGKGAGGYCYAFGCWCTHLYEQAVVWPLPKKTCN | 1.8 |
| C. elegans | Cell9 | KEGYLVNHSTGCKYECFKLGDNDYCLRECRQKYGKGAGGYCYAFGCWCTHLYEQAVVWPLPKKTCN | 2.9 |
| C. infamatus | Cii1 | KEGYLVNHSTGCKYECYKLGDNDYCLRECKQQYGKGAGGYCYAFGCWCTHLYEQAVVWPLPKKTCN | ND |
| scFv | VH | VL | REF. | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CDR2 | CDR3 | FW1 | FW3 | CDR3 | ||||||||
| C1 | 54 S | 56 D | 57 N | 105 M | 107 A | 110 Y | 164 S | 204 A | 208 D | 235 L | 236 I | [25] |
| RU1 | G | G | L | H | [23] | |||||||
| ER-1 | G | G | S | L | T | I | G | [22] | ||||
| 10F | G | G | S | L | T | I | G | T | L | α | ||
| 10F1 | G | G | S | L | T | I | G | G | T | L | α | |
| 10F2 | G | G | S | L | N | G | G | T | L | α | ||
| 10F3 | G | G | S | L | N | G | T | L | α | |||
| 10F4 | G | G | S | L | S | N | G | G | T | L | α | |
| 10F5 | G | G | S | L | S | T | G | G | T | L | α | |
| 10F6 | G | G | S | L | S | N | G | T | L | α | ||
| (A) | |||||
| TOXIN | kon(1/Ms) ×105 | koff(1/s) ×10−5 | KD(M) ×10−9 | TR (min) | KD (M) scFv × 10−9 ER |
| Cll1 | 2.65 ± 0.65 | 6.3 ± 1.3 | 0.23 ± 0.005 | 264.5 | 1.3 |
| Cn2 | 1.50 ± 0.30 | 10.2 ± 0.8 | 0.70 ± 0.075 | 163.4 | 1.0 |
| Css2 | 1.95 ± 0.05 | 55.0 ± 5.0 | 2.75 ± 0.25 | 30.0 | ND |
| Cll2 | 2.20 ± 0.50 | 90.0 ± 10.0 | 4.20 ± 0.40 | 18.5 | 5.8 |
| CeII9 | 1.50 ± 0.10 | 92.5 ± 4.5 | 6.10 ± 0.10 | 18.0 | ND |
| Ct1a | 1.70 ± 0.81 | 100.0 ± 0.01 | 8.00 ± 4.00 | 16.7 | 29.0 |
| (B) | |||||
| Toxin | µg/20 g Mouse | Alive/Total | |||
| Control | Pre-Incubated Mix | ||||
| Cll1 | 1.7 | 1/8 | 8/8 | ||
| Cll2 | 1.5 | 0/8 | 8/8 | ||
| CeII9 | 3.0 | 0/8 | 8/8 | ||
| Ct1a | 2.0 | 0/8 | 8/8 | ||
| (A) | |||||||
| Scorpion Venom | Control | Alive/Total LD50 Number and Molar Ratios | |||||
| 2 LD50 | scFv µg/mouse | 1 LD50 1:10 | 2 LD50 1:5 | 3 LD50 1:3.3 | 4 LD50 1:2.5 | 5 LD50 1:2 | |
| C. hirsutipalpus | 0/6 | 44.5 | 6/6 | 6/6 | 6/6 | 3/6 | ND |
| C. infamatus | 0/6 | 36.4 | 6/6 | 6/6 | 6/6 | 6/6 | 6/6 |
| C. sp nov. B | 0/6 | 73.0 | 6/6 | 6/6 | 5/6 | N/D | ND |
| C. suffusus | 0/6 | 33.2 | 6/6 | 6/6 | 6/6 | 6/6 | ND |
| C. noxius | 0/6 | 9.5 | 6/6 | 6/6 | 6/6 | 6/6 | ND |
| C. sp nov. A | 0/6 | 49.4 | 6/6 | 1/6 | ND | ND | ND |
| (B) | |||||||
| Venom | Alive/Total | Molar Ratio | µg of Venom/µg of scFv/20 g Mouse | ||||
| C. hirsutipalpus | 6/6 | 1:10 | 35.1/133.4 | ||||
| C. infamatus | 6/6 | 1:10 | 28.8/109.4 | ||||
| C.sp nov. B | 6/6 | 1:10 | 57.6/218.9 | ||||
| C. noxius | 5/6 | 1:10 | 7.5/28.5 | ||||
| C. suffusus | 5/6 | 1:10 | 26.5/99.8 | ||||
| C. noxius | 6/6 | 1:20 | 7.5/57.0 | ||||
| C. suffusus | 6/6 | 1:20 | 26.5/199.6 | ||||
| Residues of scFv 10FG2 | Residues of Cll1 Toxin | Residues of Cn2 Toxin | Residues of Css2 Toxin | Residues of Cll2 Toxin | Residues of Ct1a Toxin | Residues of Cell9 Toxin |
|---|---|---|---|---|---|---|
| Y59 | L8 | |||||
| L105 | I5 | L5 | L5 | |||
| L105 | V6 | V6 | V6 | V6 | V6 | V6 |
| L105 | L8 | |||||
| L105 | Y33 | Y33 | Y33 | Y33 | Y33 | Y33 |
| L105 | A55 | |||||
| L105 | V56 | I56 | V56 | V56 | V56 | V56 |
| W231 | V56 | I56 | V56 | V56 | V56 | |
| L236 | L8 | |||||
| L236 | V56 | I56 | V56 | V56 | V56 | |
| L236 | L60 | L60 | L60 | L60 | ||
| Hydrogen Bonds | ||||||
| S31(O) | K35(NZ) | K35(NZ) | K35(NZ) | K35(NZ) | K35(NZ) | K35(NZ) |
| S31(OH) | K35(NZ) | K35(NZ) | K35(NZ) | K35(NZ) | ||
| S52(OH) | Q31(O) | Q31(O) | Q31(O) | |||
| S52(OH) | Q31(OE1) | Q31(OE1) | Q31(OE1) | Q31(OE1) | Q31(OE1) | Q31(OE1) |
| G54(N) | Q31(OE1) | Q31(OE1) | Q31(OE1) | Q31(OE1) | Q31(OE1) | Q31(OE1) |
| G55(N) | Q31(OE1) | |||||
| G56(N) | Q31(OE1) | Q31(OE1) | Q31(OE1) | Q31(OE1) | Q31(OE1) | Q31(OE1) |
| Y53(OH) | K30(O) | K30(O) | K30(O) | R30(O) | R30(O) | |
| S57(OH) | Q32(OE1) | Q32(OE1) | K32(NZ) | Q32(OE1) | ||
| S57(OH) | Q31(O) | Q31(O) | ||||
| Y59(OH) | Q32(OE1) | Q32(OE1) | Q32(OE1) | Q32(OE1) | Q32(OE1) | |
| Y59(OH) | Q32(NE2) | Q32(NE2) | K32(NZ) | Q32(NE2) | ||
| Y59(OH) | D7(O) | |||||
| Y59(OH) | N7(OD1) | N7(OD1) | ||||
| Y59(OH) | N7(ND2) | |||||
| Y59(OH) | L8(N) | K8(N) | K8(N) | H8(N) | H8(N) | H8(N) |
| Y60(O) | K8(NZ) | K8(NZ) | ||||
| D62(OD1) | K8(NZ) | |||||
| D62(OD1) | K63(NZ) | |||||
| K65(NZ) | S9(O) | N9(OD1) | S9(O) | S9(O) | ||
| K65(NZ) | S9(OH) | |||||
| R101(NH2) | Q54(OE1) | Q54(OE1) | Q54(OE1) | Q54(OE1) | Q54(OE1) | Q54(OE1) |
| R101(NH1) | Q54(NE2) | Q54(NE2) | Q54(OE1) | Q54(OE1) | ||
| D102(OD1) | Y52(OH) | Y52(OH) | Y52(OH) | Y52(OH) | Y52(OH) | Y52(OH) |
| D102(OD2) | K35(NZ) | K35(NZ) | K35(NZ) | K35(NZ) | K35(NZ) | K35(NZ) |
| D102(OD2) | K35(N) | K35(N) | K35(N) | K35(N) | K35(N) | K35(N) |
| L104(N) | Q32(O) | Q32(O) | Q32(O) | Q32(O) | K32(O) | Q32(O) |
| L105(N) | Q32(O) | Q32(O) | Q32(O) | Q32(O) | K32(O) | Q32(O) |
| L105(O) | V56(N) | L56(N) | V56(N) | V56(N) | V56(N) | V56(N) |
| S107(N) | Q54(O) | Q54(O) | Q54(O) | Q54(O) | Q54(O) | Q54(O) |
| S107(OH) | Q54(O) | Q54(O) | Q54(O) | Q54(O) | Q54(O) | |
| S107(OH) | Q54(NE2) | Q54(NE2) | Q54(NE2) | Q54(NE2) | ||
| D108(OD2) | Q54(NE2) | Q54(NE2) | Q54(NE2) | Q54(NE2) | Q54(NE2) | |
| D108(OD1) | Q54(NE2) | |||||
| S165(O) | K62(NZ) | |||||
| N171(OD1) | Q54(NE2) | |||||
| T172(OH) | Q54(OE1) | |||||
| T172(N) | Q54(OE1) | |||||
| AS190(OH) | Q54(NE2) | |||||
| D233(O) | K62(NZ) | N62(ND2) | N62(OD1) | K62(NZ) | ||
| D233(OD2) | N62(ND2) | K62(NZ) | ||||
| S234(OH) | K62(NZ) | N62(NZ) | K62(NZ) | K62(NZ) | ||
| T235(O) | K8(NZ) | K8(NZ) | ||||
| T235(OH) | K62(NZ) | N62(ND2) | K62(NZ) | K62(NZ) | ||
| T235(OH) | K63(NZ) | |||||
| L236(O) | K8(NZ) | K8(NZ) | ||||
| G237(O) | K8(NZ) | |||||
| Ionic Interactions within Six Angstroms | ||||||
| D102 | K35 | |||||
| Cation–Pi Interactions within Six Angstroms | ||||||
| Y53 | K35 | |||||
| R101 | Y52 | |||||
| Number contacts α | 42 | 36 | 34 | 27 | 29 | 36 |
| Hydrogen Bonds δ | 10.3 | 10.8 | 10.3 | 10.4 | 9.1 | 10.4 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Riaño-Umbarila, L.; Gómez-Ramírez, I.V.; Ledezma-Candanoza, L.M.; Olamendi-Portugal, T.; Rodríguez-Rodríguez, E.R.; Fernández-Taboada, G.; Possani, L.D.; Becerril, B. Generation of a Broadly Cross-Neutralizing Antibody Fragment against Several Mexican Scorpion Venoms. Toxins 2019, 11, 32. https://doi.org/10.3390/toxins11010032
Riaño-Umbarila L, Gómez-Ramírez IV, Ledezma-Candanoza LM, Olamendi-Portugal T, Rodríguez-Rodríguez ER, Fernández-Taboada G, Possani LD, Becerril B. Generation of a Broadly Cross-Neutralizing Antibody Fragment against Several Mexican Scorpion Venoms. Toxins. 2019; 11(1):32. https://doi.org/10.3390/toxins11010032
Chicago/Turabian StyleRiaño-Umbarila, Lidia, Ilse V. Gómez-Ramírez, Luis M. Ledezma-Candanoza, Timoteo Olamendi-Portugal, Everardo Remi Rodríguez-Rodríguez, Guillermo Fernández-Taboada, Lourival D. Possani, and Baltazar Becerril. 2019. "Generation of a Broadly Cross-Neutralizing Antibody Fragment against Several Mexican Scorpion Venoms" Toxins 11, no. 1: 32. https://doi.org/10.3390/toxins11010032
APA StyleRiaño-Umbarila, L., Gómez-Ramírez, I. V., Ledezma-Candanoza, L. M., Olamendi-Portugal, T., Rodríguez-Rodríguez, E. R., Fernández-Taboada, G., Possani, L. D., & Becerril, B. (2019). Generation of a Broadly Cross-Neutralizing Antibody Fragment against Several Mexican Scorpion Venoms. Toxins, 11(1), 32. https://doi.org/10.3390/toxins11010032