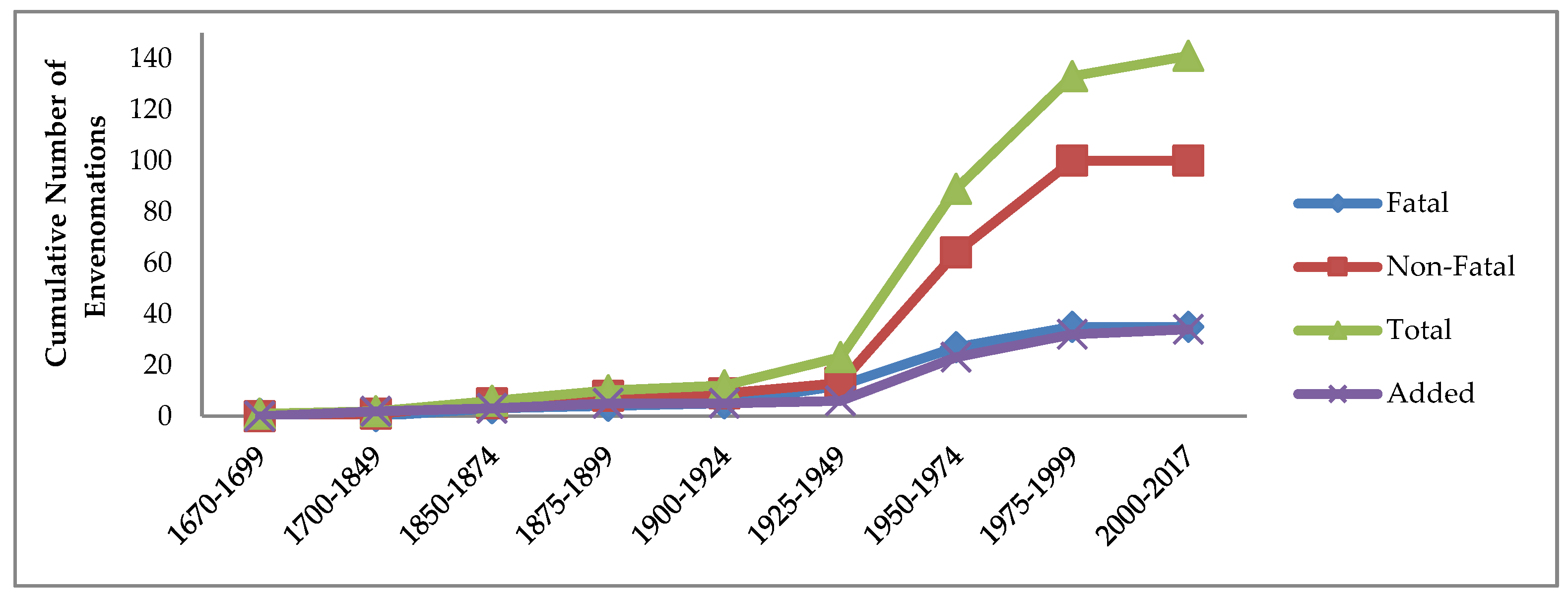
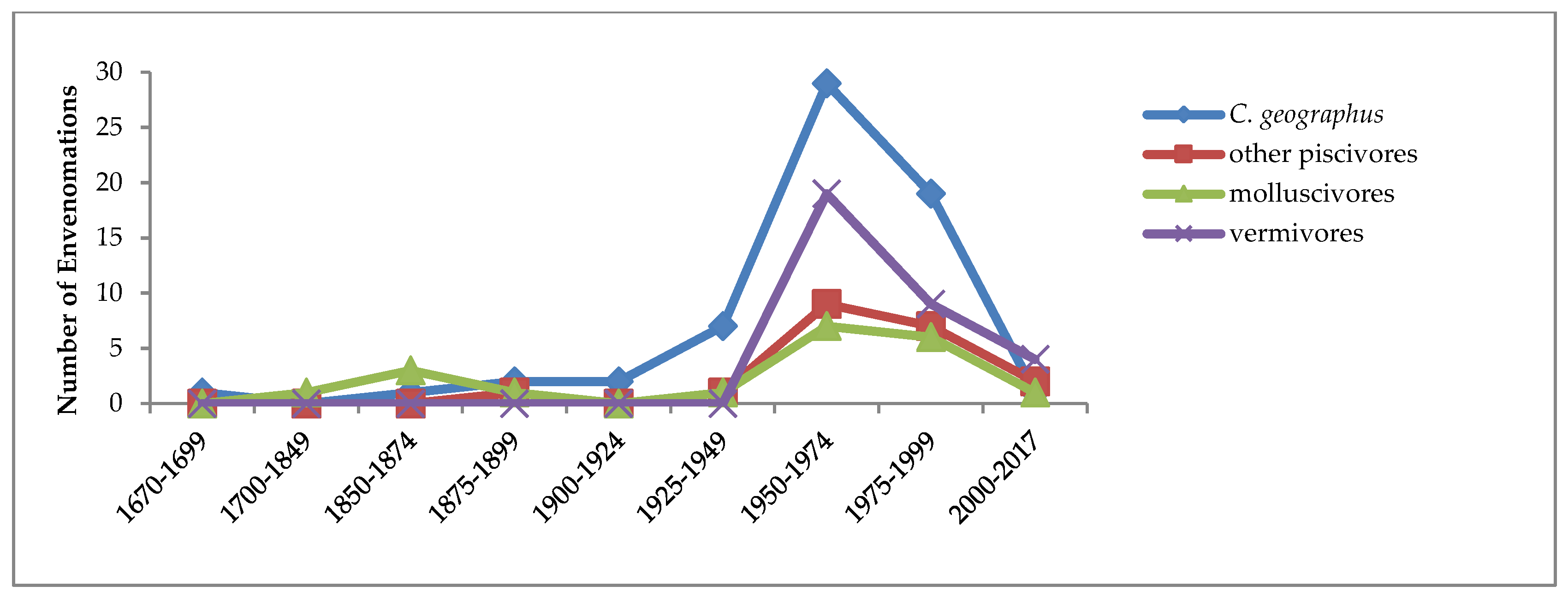
2.1. Temporal Trends of Fatal and Non-Fatal Reported Cases of Conus Envenomations of Humans, 1670–2017
Prior studies have not explicitly examined the patterns over time of severity of human responses to reported
Conus stings.
Table 1 and
Figure 1 show the trends in numbers and severity of the 141 known human injuries reported from the 34
Conus species known to be responsible, dating from the initial account of ca. 1670 [
8] through to 2017. The IJCPT website distinguishes three levels of severity of
Conus stings, fatal (designated “A”), serious symptoms followed by complete recovery (designated “B”), and minor effects only (comparable to a bee sting and designated “C”). For simplicity and clarity of the graphs in this paper, these are condensed into two categories, fatal (F, for outcome A), and non-fatal (NF, combining the two survivor outcomes B and C) (
Figure 1).
The marked change in slopes, especially of the red and green lines in
Figure 1, strongly suggest that starting from the second quarter of the 20th century, higher proportions of non-fatal cases were reported in news media than in the previous three centuries, followed by that trend leveling off in the 21st century. All points in
Figure 1 from the first case through 1925–1949 indicate fatality rates of 42–100%. Thereafter, 70–75% of all reported cases resulted in recovery (
Table 1). The lower cumulative number and proportion of cases resulting in loss of life during the later 20th century and into the 21st may be attributed mainly to increasing availability of medical treatment and of media in formerly more remote tropical regions reporting less serious cases.
2.2. Relation of Natural History of Conus Species to Severity of their Stings of Humans
Differences in the natural prey organisms of
Conus species are strongly correlated both with the likelihood that a human will be stung and with the severity of the effects of envenomation. As noted briefly above, almost all
Conus species whose diets in nature have been studied belong to one of three predatory feeding guilds, often referred to as worm-hunters, mollusc-hunters, and fish-hunters [
9]. Probably more than two-thirds of all species are specialist predators on segmented worms of the phylum Annelida. In addition to including most species in the genus, the vermivorous guild is evolutionarily the oldest; its fossils first appear in the early Eocene epoch, about 57 million years before present (mybp). The other feeding guilds evolved from it. The ages of origin of these clades are uncertain, but recent molecular phylogenetic analysis of the Conidae [
10] and systematic classification of Conoidea [
5] indicate that the fossil record of the extant piscivorous subgenus
Pionoconus first appeared in the Upper Oligocene (about 30 mybp). In contrast, the fossil record suggests that the origin of molluscivory in
Conus is much more recent. The oldest fossils of an extant molluscivorous subgenus appear to be those of
C. kanayai [
11], from the latest Miocene (about 5–7 mybp). Shuto [
11] originally placed
C. kanayai in the genus
Conolithus. Tucker and Tenorio [
5] later correctly assigned it to
Cylinder, which is now classified as a subgenus of
Conus [
10].
Like many other gastropods,
Conus of all species that have been tested recognize the presence of appropriate prey at a distance from chemical signals released in the water [
12,
13]. They move toward the prey, apparently following the concentration gradient of the chemoattractants, and extend the tubular proboscis toward the prey. Upon making contact, the proboscis musculature injects a complex, species-specific venom pumped through a disposable, hollow, hypodermic needle-like radular tooth (Figures 1 and 2 in Ref. [
3]) from the proboscis into the prey, which is rapidly immobilized and swallowed whole [
14]. Several recent reviews survey the structure, target receptors, and functions of these highly neurotoxic compounds, primarily small peptides, as well as their increasing therapeutic roles [
15,
16,
17].
2.3. Piscivorous Conus Species
As
Figure 2 shows, more than half of all known human envenomations (57%) have resulted from stings by the single large, widely distributed, exclusively piscivorous species
Conus geographus. It is also quite likely that this species has caused all of the human fatalities, although species identity of the perpetrators is still uncertain in a few cases (about 8; see SM1). They have been ascribed in the literature to other
Conus species, primarily the large, exclusively molluscivorous
C. textile, but none of these has been adequately verified (see SM7). No fatalities have been reported from the nine other piscivorous species known to have stung humans (
n = 20 cases; SM3).
Why
Conus geographus is by far the most dangerous species to humans has not been thoroughly studied, particularly with respect to how the
Conus becomes aware of the presence of an adversary worthy of attack. Chemical attractant signals are almost certainly released into the water by appropriate prey of molluscivorous and vermivorous, as well as piscivorous
Conus species, but in contrast to substantial research on the effective toxic conopeptides [
15,
16,
17], the nature of these initial attractants remain largely unknown [
12,
13,
18].
The temporal pattern of reported human attacks by other piscivorous species (the red curve in
Figure 2) is similar to that of
C. geographus. These signal molecules likely differ among the three main feeding guilds of
Conus species, but experimental data on food preference derive mainly from studies of vermivores (e.g., Ref. [
18]), and I am not aware of any recent studies. The attractant molecules released by fishes are possibly more general attributes of vertebrates.
Fish-hunting
Conus species typically share a feeding strategy that involves a very long, often curved radular tooth (often about 13% of shell length), with wide interspecific variation in prominence of blade, barbs and serrations (Figure 8 in Ref. [
19]; see also Figure 13.2 in Ref. [
17]). Prey is typically subdued by a single injection of venom in each feeding event. The tooth remains attached to the prey by its arrangement of barbs, and to the predator’s proboscis by contraction of a sphincter muscle in the proboscis in front of the basal spur when the tooth is injected. The tooth thus functions as a harpoon as well as a hypodermic needle, holding the prey until it gradually becomes paralyzed and can be completely engulfed.
Body size is also an important factor in the danger of
C. geographus envenomation of humans. It is the largest extant piscivorous species in the genus, reaching a shell length of 166 mm [
4]; very few piscivorous
Conus species attain a shell length in excess of 100 mm. Body size, usually measured as shell length, is strongly correlated with severity of the injuries that
C. geographus can inflict on people (Figures 4 and 5 in Ref. [
3]). It is highly likely, but to my knowledge undocumented, that the larger the animal, the more venom it can inject in a single sting. The shells of the eight
C. geographus individuals that are known to have killed people and whose shells have been measured ranged 80–135 mm in length (mean = 103 mm), while those that inflicted non-fatal stings ranged 50–125 mm in length (mean = 86 mm) (SM1). All of the shells of piscivores other than
C. geographus that are known to have stung people (22 individuals of 11 species) were smaller; those whose shell lengths are known ranged from 24 to 55 mm (SM3).
One case (No. 133; SM1, SM3) involving the piscivorous species
Conus fulmen deserves special mention because it involves the only known intentional human injection of
Conus venom.
C. fulmen occurs from Taiwan to the main Japanese islands, and by 1977, only one mild human injury from a sting had been reported (Case No. 78). Professor Shigeo Yoshiba, then of the Jikei University School of Medicine in Tokyo, decided to assess its danger and toxicity by injecting himself with its venom (Case No. 133). In addition, he carried out similar experiments on other mammals (mice and rabbits), two amphibians, five fishes, and members of five invertebrate phyla as well [
20].
After injecting crude venom from
Conus fulmen intracutaneously at a concentration of 0.0014 mg/kg into his forearm, Yoshiba stated that “no neurological or functional disturbance appeared; only local findings such as pain, redness, ischemia, edema, and itching appeared and lasted for 3 days” (p. 112 in Ref. [
20]).
2.4. Molluscivorous Conus Species
There are about as many extant specialist molluscivorous
Conus species as there are predators on fishes. However, in general the venom of the former guild is less toxic to humans, and members of only six different species are reported to have stung people (SM4). The piscivorous clades have a long evolutionary history of adapting their chemical weapons to overcoming vertebrate prey with a single dose of a formidable cocktail of numerous neurotoxic peptides, typically injected by a strongly barbed hypodermic radular tooth (Figure 8 in Ref. [
19]) that also functions to catch and hold the prey until it can be engulfed by the rhynchocoel (= proboscis sheath or rostrum) (Figure 5.6 in Ref. [
7]).
Successful predation on molluscs, most of which have strong protective shells that they can withdraw their bodies into when an enemy threatens or has already stung once, imposes different challenges. Almost all molluscan prey of Conus are other gastropods, and while a few species prey partly on shell-less nudibranchs, most molluscivorous Conus specialize on other shelled prosobranchiate gastropods, even including other Conus species in some cases.
These prey species often respond to being stung by withdrawing quickly (for a gastropod) into the shell, thus avoiding capture, perhaps until danger passes. However, the radular tooth form in molluscivorous
Conus species follows different functions. They are typically slender, very elongated, and armed with a single moderately strong barb. They also have a narrower, more conical base without a spur, features that facilitate leaving the tooth embedded in the victim when feeding, in contrast to teeth of the other two feeding guilds (Figures 1–7 in Ref. [
19]). Molluscivorous
Conus species also tend to be patient and prepared to sting their injured victim again (and sometimes again) when it attempts to emerge from its shell.
Schoenberg [
21] first reported the injection of multiple radular teeth by a molluscivorous
Conus in the same feeding episode, in
C. textile in Hawaii. To my knowledge, this staged encounter established the record number of radular teeth injected in a single predation episode. In this case the prey was another molluscivorous neogastropod,
Harpa amouretta. A common defense of
Harpa spp. to attempted predation is autotomy of the posterior portion of the foot. In this case, the
H. amouretta did so upon recognizing the presence of the
C. textile, but before the latter was able to inject a tooth into it. Subsequently, it injected five teeth into the
Harpa’s shell aperture. These were not immediately lethal, the
H. amouretta continued to wave its siphon, and the
C. textile proceeded to inject 12 more radular teeth into its victim. Then, as the author described it, “When the
amouretta stopped moving, Sir Textile began his meal. Fifteen minutes later, only an empty shell remained and the
textile lazily slid down into the sand for a nap” (p. 4 in Ref. [
20]).
More recently, Yoshiba studied the feeding process in
Conus bandanus [
22], and in greater detail in nearly 200 feeding events by a captive
C. textile over five years in Japan. He showed that each feeding event averaged the injection of three radular teeth, with a maximum of six [
23].
Kohn followed and videotaped an individual of
Conus victoriae, a species closely related to
C. textile, preying on the buccinid gastropod
Cantharus erythrostomus, in Western Australia [
24]. It successfully injected four teeth, after missing the target with one, over a period of about 29 min. It then completed swallowing the prey’s body within the next 6.5 h, after the venom had weakened the bond between the shell and columellar muscle.
Although Prator et al. considered the reason why molluscivorous species inject multiple venom-laden teeth in one feeding event to be unclear [
9], this strategy has likely been selected because the first one or two injections often miss the target or fail to immobilize the prey, which may extend from its shell and move away. Succeeding injections result in the victim remaining within its shell, increasingly unable to extend and more easily extracted intact from the shell and engulfed by the
Conus due to weakened shell–muscle adhesion.
The habit of molluscivorous Conus injecting more than one tooth while overcoming a single prey organism is also exemplified by the only known case of a human having been stung more than once in the same attack episode. In this case (No. 128; SM4) a C. bandanus 48 mm long stung the victim on the same finger three times in succession, near a remote, uninhabited island in the Solomons. The most severe symptoms included an immediate burning sensation on the hand, blurred vision, lightheadedness, and chest pain with some difficulty breathing, which ameliorated after 24 h, headache that persisted for two weeks, and numbness and stiffness of the finger for about one week. The victim utilized the only medications available, meat tenderizer topically and strong antihistamine tablets, and recovered fully.
2.5. Vermivorous Conus Species
Annelid worms in the class Polychaeta, segmented relatives of earthworms, are the predominant prey of the numerically predominant vermivorous Conus species. Some annelids move freely about or burrow in sandy or muddy sediments, while others secrete and dwell in tubes that they construct of organic matter or calcium carbonate. The feeding biology of vermivorous Conus species more closely resembles that of piscivores than molluscivores. Like the former, in each feeding event they inject a single radular tooth containing enough venom to overcome the prey, which is often also harpooned, then engulfed and swallowed whole.
Although the majority of
Conus species are vermivorous, reported stings of humans by them are both infrequent and limited to mild effects. Only 30 stings of humans have been reported, by 20 different vermivorous species (SM1, SM5). Two-thirds of these had only mild effects on victims, usually no more serious than a bee sting, and none have been life-threatening. The earliest reported cases occurred in 1955, and the most recent, in July 2017, from
C. regius in Key West, Florida. In
Figure 2, the purple curve shows that from the mid-20th century, the temporal pattern of stings by vermivorous species closely resembles those of the other
Conus feeding guilds, suggesting that it reflects increased media interest in even mild molluscogenic human injuries.
2.8. Cone Snails as Murder Weapons in Fiction
A few murder mystery writers have ventured to fictionalize the dangers to humans from conotoxins and their delivery to inappropriate targets. The earliest I have been able to locate is the “The Cloth-of-Gold Murders,” by the well-known 20th century American mystery writer Baynard Kendrick (1894–1977), who published “The Cloth-of-Gold Murders” in the American Magazine’s then monthly feature, “Complete American Mystery Novel” in February 1958 [
29]. A glance at
Figure 2 shows that this time period was one of increasingly frequent media reports of human injuries and fatalities due to stings by
Conus. Kendrick’s complex story of about 4000 words is subtitled “A killer prowled the peaceful island—his weapon a deadly, gleaming shell.” It involves two murders, a diverse cast including a malacologist and others with serious interests, and is set on the Florida Gulf Coast. The cast of
Conus comprises two species,
C. textile and
C. gloriamaris, for whose exotic provenance the author provides a rational explanation [
29].
A more recent (1986), small book-length mystery novel is also by a malacologically able author, Ann Kengalu, (d. 1998) who ventured to the Solomon Islands as an Anglican missionary and stayed to marry into a local family. It also involves murders and appearances of a couple of cone snails and cowries, but they have only cameo roles in the murders and are not weaponized. The book is “Murder on the Mataniko Bridge,” set and published in the Solomons and with a young woman from New Zealand as the central character [
30].
Aside from print media, a 1972 episode of the television series
Hawaii 5-0 featured an apparent murder plan involving
Conus textile, but instead resulted in the perpetrator’s suicide [
31]. Yuhas also noted that “in the recent film
Jurassic Park 2 only cone snail venom was powerful enough to fell a
Tyrannosaurus rex,” although the last dinosaurs had become extinct about seven million years before the first
Conus appeared in the fossil record [
32].