Zearalenone Promotes Cell Proliferation or Causes Cell Death?
Abstract
1. Introduction
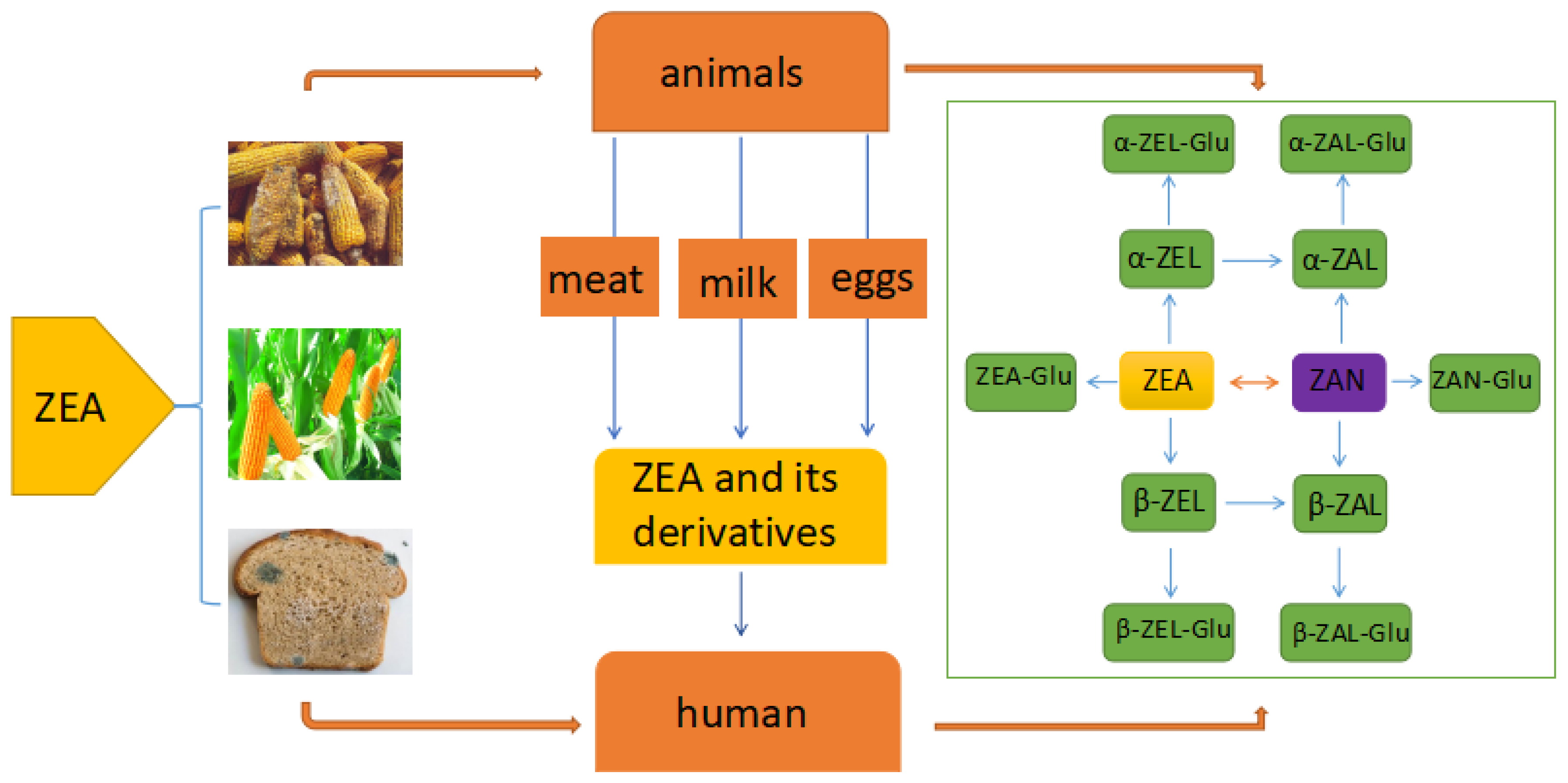
2. The Metabolic Process of ZEA
3. The Molecular Mechanisms of ZEA Promoted the Cell Proliferation
3.1. ZEA and Its Derivatives Can Exert the Estrogen-Like Effects Which Can Stimulate the Cell Proliferation
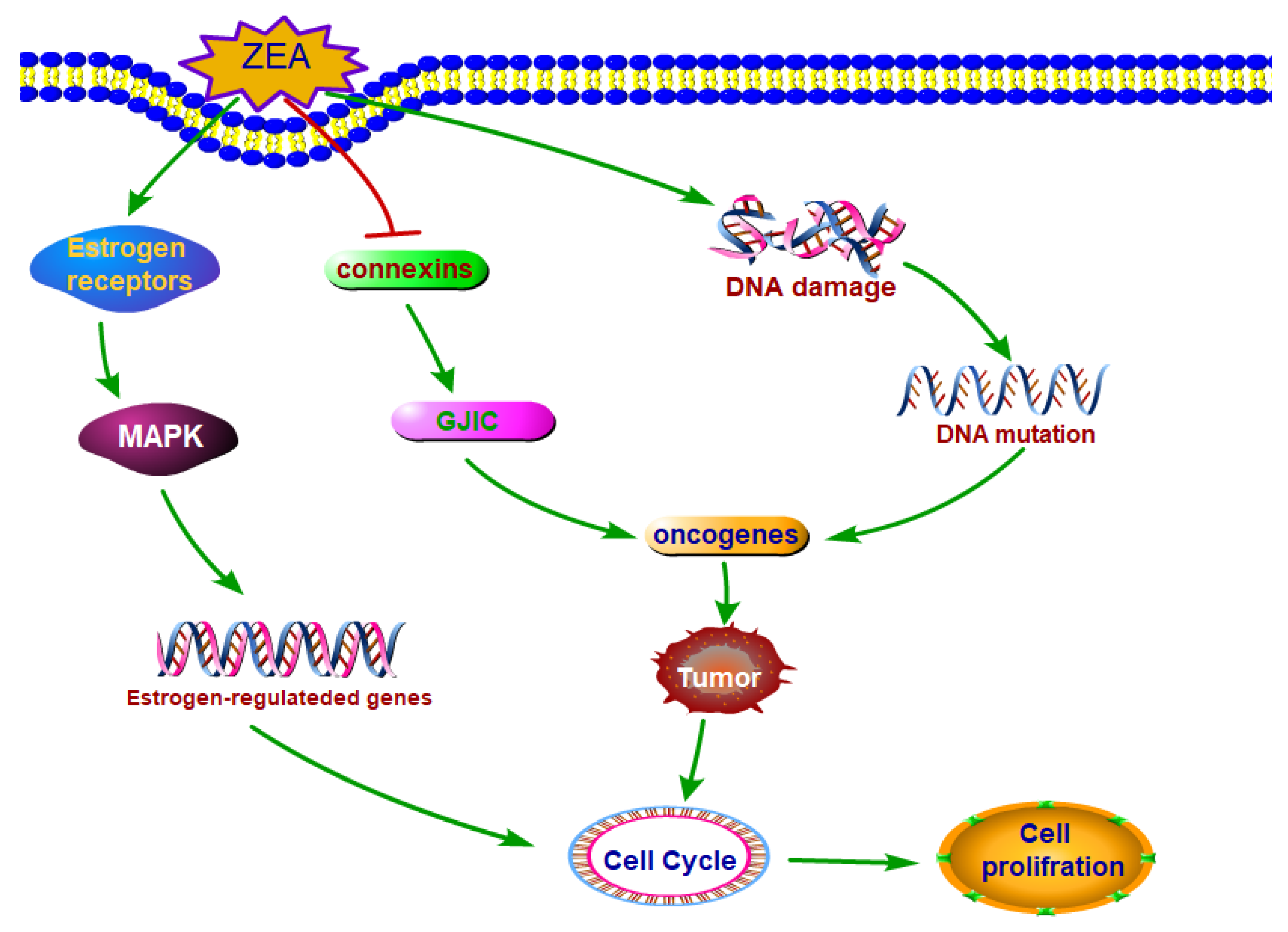
3.2. ZEA Exhibited the Carcinogenic Property
The Mechanisms of ZEA Exhibited the Carcinogenic Property
4. The Molecular Mechanisms of ZEA Caused Cell Death
4.1. ZEA Causes Cell Death through Disturbing the Distribution of Cell Cycle and the Expressions of Cell Cycle Regulating Proteins
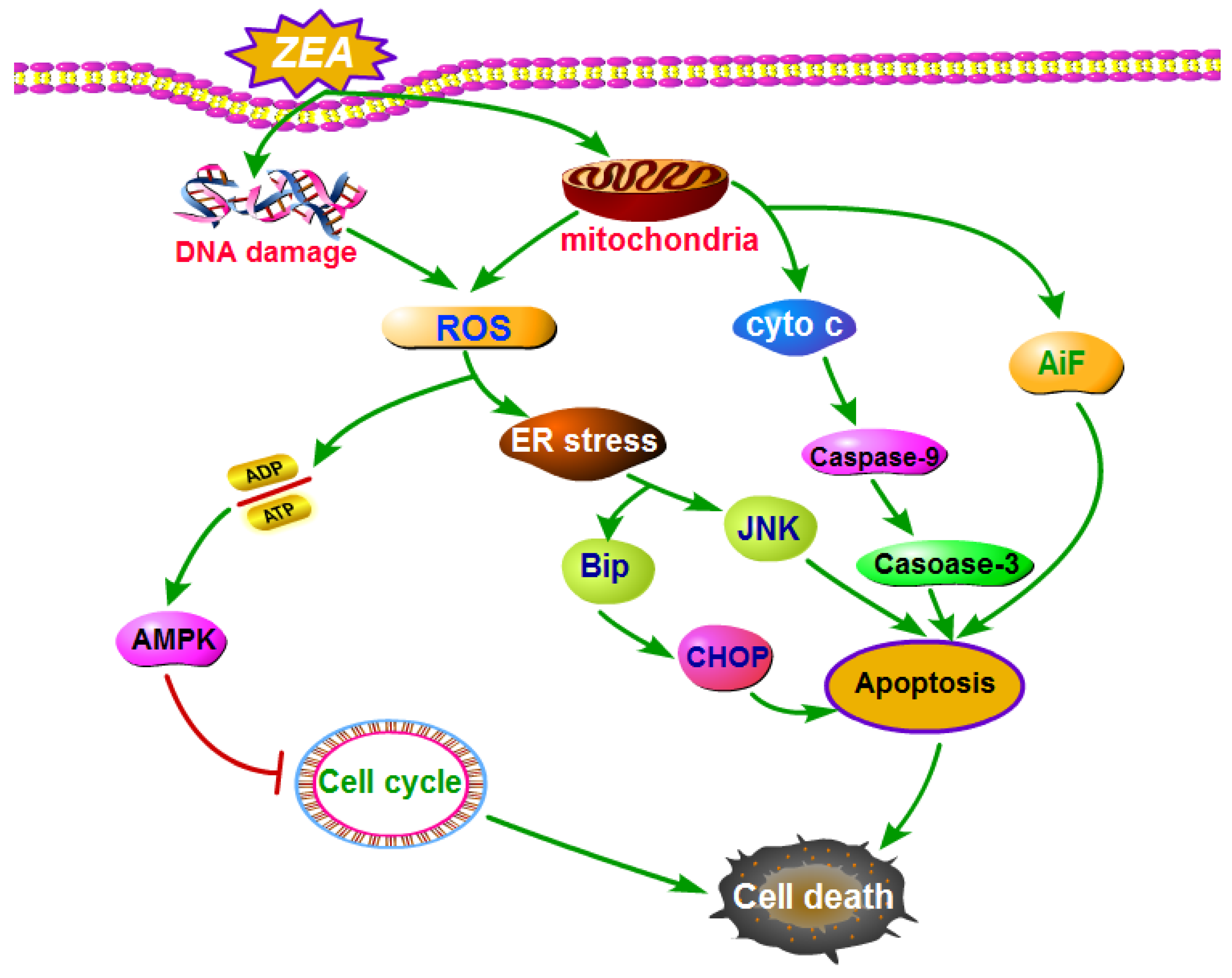
4.2. ZEA Caused Cell Death through Inducing Cell Oxidative Stress
4.3. ZEA Caused Cell Death through Inducing Cell Apoptosis
4.3.1. ZEA Induced Cell Apoptosis through ER Stress
4.3.2. ZEA Induced Cell Apoptosis through Producing of ROS
4.3.3. ZEA Induced Cell Apoptosis through Mitochondrial Signaling Pathway
5. What Can Protect the Damage or Cell Death Induced by ZEA
5.1. ZEA Can Induce Autophagy Which Can Exert a Protective Function Against the Cytotoxicity
5.2. Some Antioxidants Which Can Alleviate the Oxidative Damage May Partially Prevent the Cytotoxicity Induced by ZEA
6. Conclusions and Future Perspective
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Chang, H.; Kim, W.; Park, J.H.; Kim, D.; Kim, C.R.; Chung, S.; Lee, C. The occurrence of zearalenone in South Korean feedstuffs between 2009 and 2016. Toxins 2017, 9. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhang, B.; He, X.; Cheng, W.H.; Xu, W.; Luo, Y.; Liang, R.; Luo, H.; Huang, K. Analysis of individual and combined effects of ochratoxin A and zearalenone on HepG2 and KK-1 cells with mathematical models. Toxins 2014, 6, 1177–1192. [Google Scholar] [CrossRef] [PubMed]
- Kuiper-Goodman, T.; Scott, P.M.; Watanabe, H. Risk assessment of the mycotoxin zearalenone. Regul. Toxicol. Pharmacol. RTP 1987, 7, 253–306. [Google Scholar] [CrossRef]
- Ma, R.; Zhang, L.; Liu, M.; Su, Y.T.; Xie, W.M.; Zhang, N.Y.; Dai, J.F.; Wang, Y.; Rajput, S.A.; Qi, D.S.; et al. Individual and combined occurrence of mycotoxins in feed ingredients and complete feeds in China. Toxins 2018, 10. [Google Scholar] [CrossRef] [PubMed]
- Zinedine, A.; Soriano, J.M.; Molto, J.C.; Manes, J. Review on the toxicity, occurrence, metabolism, detoxification, regulations and intake of zearalenone: An oestrogenic mycotoxin. Food Chem. Toxicol. 2007, 45, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Makowska, K.; Obremski, K.; Zielonka, L.; Gonkowski, S. The influence of low doses of zearalenone and T-2 toxin on calcitonin gene related peptide-like immunoreactive (CGRP-LI) neurons in the ENS of the porcine descending colon. Toxins 2017, 9. [Google Scholar] [CrossRef] [PubMed]
- Yip, K.Y.; Wan, M.L.Y.; Wong, A.S.T.; Korach, K.S.; El-Nezami, H. Combined low-dose zearalenone and aflatoxin B1 on cell growth and cell-cycle progression in breast cancer MCF-7 cells. Toxicol. Lett. 2017, 281, 139–151. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.; Huang, Q.; Pan, S.; Fan, W.; Wang, G.; Yuan, Y.; Gu, J.; Liu, X.; Liu, Z.; Bian, J. Regulation of oncogenes and gap junction intercellular communication during the proliferative response of zearalenone in TM3 cells. Hum. Exp. Toxicol. 2017, 36, 701–708. [Google Scholar] [CrossRef] [PubMed]
- Kowalska, K.; Habrowska-Gorczynska, D.E.; Dominska, K.; Piastowska-Ciesielska, A.W. The dose-dependent effect of zearalenone on mitochondrial metabolism, plasma membrane permeabilization and cell cycle in human prostate cancer cell lines. Chemosphere 2017, 180, 455–466. [Google Scholar] [CrossRef] [PubMed]
- Lecomte, S.; Lelong, M.; Bourgine, G.; Efstathiou, T.; Saligaut, C.; Pakdel, F. Assessment of the potential activity of major dietary compounds as selective estrogen receptor modulators in two distinct cell models for proliferation and differentiation. Toxicol. Appl. Pharm. 2017, 325, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Khosrokhavar, R.; Rahimifard, N.; Shoeibi, S.; Hamedani, M.P.; Hosseini, M.J. Effects of zearalenone and alpha-Zearalenol in comparison with Raloxifene on T47D cells. Toxicol. Mech. Methods 2009, 19, 246–250. [Google Scholar] [CrossRef] [PubMed]
- Zong, S.; Zeng, G.; Fang, Y.; Peng, J.; Zou, B.; Gao, T.; Zhao, J. The effects of alpha-zearalanol on the proliferation of bone-marrow-derived mesenchymal stem cells and their differentiation into osteoblasts. J. Bone Miner. Metab. 2016, 34, 151–160. [Google Scholar] [CrossRef] [PubMed]
- Cortinovis, C.; Caloni, F.; Schreiber, N.B.; Spicer, L.J. Effects of fumonisin B1 alone and combined with deoxynivalenol or zearalenone on porcine granulosa cell proliferation and steroid production. Theriogenology 2014, 81, 1042–1049. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.; Pan, S.; Wang, G.; Wang, Y.J.; Liu, Q.; Gu, J.; Yuan, Y.; Liu, X.Z.; Liu, Z.P.; Bian, J.C. Zearalenone impairs the male reproductive system functions via inducing structural and functional alterations of Sertoli cells. Environ. Toxicol. Pharmacol. 2016, 42, 146–155. [Google Scholar] [CrossRef] [PubMed]
- Sang, Y.; Li, W.; Zhang, G. The protective effect of resveratrol against cytotoxicity induced by mycotoxin, zearalenone. Food Funct. 2016, 7, 3703–3715. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.Y.; Zheng, Z.H.; Son, Y.O.; Shi, X.; Jang, Y.O.; Lee, J.C. Mycotoxin zearalenone induces AIF- and ROS-mediated cell death through p53- and MAPK-dependent signaling pathways in RAW264.7 macrophages. Toxicol In Vitro 2011, 25, 1654–1663. [Google Scholar] [CrossRef] [PubMed]
- Vlata, Z.; Porichis, F.; Tzanakakis, G.; Tsatsakis, A.; Krambovitis, E. A study of zearalenone cytotoxicity on human peripheral blood mononuclear cells. Toxicol. Lett. 2006, 165, 274–281. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Yuan, H.; Guo, C.; Lu, Y.; Deng, S.; Yang, Y.; Wei, Q.; Wen, L.; He, Z. Zearalenone induces apoptosis and necrosis in porcine granulosa cells via a caspase-3- and caspase-9-dependent mitochondrial signaling pathway. J. Cell. Physiol. 2012, 227, 1814–1820. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.L.; Wang, B.J.; Wang, L.; Shan, Y.P.; Zou, H.; Song, R.L.; Wang, T.; Gu, J.H.; Yuan, Y.; Liu, X.Z.; et al. ROS-mediated cell cycle arrest and apoptosis induced by zearalenone in mouse Sertoli cells via ER stress and the ATP/AMPK pathway. Toxins 2018, 10. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Zhang, S.; Zhang, M.; Yang, L.; Cheng, B.; Li, J.; Shan, A. Individual and combined effects of Fusarium toxins on apoptosis in PK15 cells and the protective role of N-acetylcysteine. Food Chem. Toxicol. 2018, 111, 27–43. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Xu, M.; Dai, Y.; Ding, X.; Xiao, C.; Ji, H.; Xu, Y. Exploration of Bcl-2 family and caspases-dependent apoptotic signaling pathway in Zearalenone-treated mouse endometrial stromal cells. Biochem. Biophys. Res. Commun. 2016, 476, 553–559. [Google Scholar] [CrossRef] [PubMed]
- Lin, P.; Chen, F.; Sun, J.; Zhou, J.; Wang, X.; Wang, N.; Li, X.; Zhang, Z.; Wang, A.; Jin, Y. Mycotoxin zearalenone induces apoptosis in mouse Leydig cells via an endoplasmic reticulum stress-dependent signalling pathway. Reprod. Toxicol. 2015, 52, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zheng, W.; Bian, X.; Yuan, Y.; Gu, J.; Liu, X.; Liu, Z.; Bian, J. Zearalenone induces apoptosis and cytoprotective autophagy in primary Leydig cells. Toxicol. Lett. 2014, 226, 182–191. [Google Scholar] [CrossRef] [PubMed]
- Ismaiel, A.A.; Papenbrock, J. Mycotoxins: Producing fungi and mechanisms of phytotoxicity. Agriculture 2015, 5, 492–537. [Google Scholar] [CrossRef]
- Ren, G.X.; Hu, Y.C.; Zhang, J.M.; Zou, L.; Zhao, G. Determination of multi-class mycotoxins in tartary buckwheat by ultra-fast liquid chromatography coupled with triple quadrupole mass spectrometry. Toxins 2018, 10. [Google Scholar] [CrossRef] [PubMed]
- Belhassen, H.; Jimenez-Diaz, I.; Arrebola, J.P.; Ghali, R.; Ghorbel, H.; Olea, N.; Hedili, A. Zearalenone and its metabolites in urine and breast cancer risk: A case-control study in Tunisia. Chemosphere 2015, 128, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Jeong, S.H.; Kang, D.; Lim, M.W.; Kang, C.S.; Sung, H.J. Risk assessment of growth hormones and antimicrobial residues in meat. Toxicol. Res. 2010, 26, 301–313. [Google Scholar] [CrossRef] [PubMed]
- Signorini, M.L.; Gaggiotti, M.; Molineri, A.; Chiericatti, C.A.; de Basilico, M.L.Z.; Basilico, J.C.; Pisani, M. Exposure assessment of mycotoxins in cow’s milk in Argentina. Food Chem. Toxicol. 2012, 50, 250–257. [Google Scholar] [CrossRef] [PubMed]
- Hao, G.; Liang, H.; Li, Y.; Li, H.; Gao, H.; Liu, G.; Liu, Z. Simple, sensitive and rapid HPLC-MS/MS method for the determination of cepharanthine in human plasma. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2010, 878, 2923–2927. [Google Scholar] [CrossRef] [PubMed]
- Filannino, A.; Stout, T.A.; Gadella, B.M.; Sostaric, E.; Pizzi, F.; Colenbrander, B.; Dell’Aquila, M.E.; Minervini, F. Dose-response effects of estrogenic mycotoxins (zearalenone, alpha- and beta-zearalenol) on motility, hyperactivation and the acrosome reaction of stallion sperm. Reprod. Boil. Endocrinol. 2011, 9, 134. [Google Scholar] [CrossRef] [PubMed]
- Malekinejad, H.; Maas-Bakker, R.F.; Fink-Gremmels, J. Bioactivation of zearalenone by porcine hepatic biotransformation. Vet. Res. 2005, 36, 799–810. [Google Scholar] [CrossRef] [PubMed]
- D’Mello, J.P.F.; Placinta, C.M.; Macdonald, A.M.C. Fusarium mycotoxins: A review of global implications for animal health, welfare and productivity. Anim. Feed Sci. Technol. 1999, 80, 183–205. [Google Scholar] [CrossRef]
- Gromadzka, K.; Waskiewicz, A.; Chelkowski, J.; Golinski, P. Zearalenone and its metabolites: Occurrence, detection, toxicity and guidelines. World Mycotoxin J. 2008, 1, 209–220. [Google Scholar] [CrossRef]
- Tatay, E.; Espin, S.; Garcia-Fernandez, A.J.; Ruiz, M.J. Estrogenic activity of zearalenone, alpha-zearalenol and beta-zearalenol assessed using the E-screen assay in MCF-7 cells. Toxicol. Mech. Methods 2018, 28, 239–242. [Google Scholar] [CrossRef] [PubMed]
- Kunishige, K.; Kawate, N.; Inaba, T.; Tamada, H. Exposure to zearalenone during early pregnancy causes estrogenic multitoxic effects in mice. Reprod. Sci. 2017, 24, 421–427. [Google Scholar] [CrossRef] [PubMed]
- Parveen, M.; Zhu, Y.; Kiyama, R. Expression profiling of the genes responding to zearalenone and its analogues using estrogen-responsive genes. FEBS Lett. 2009, 583, 2377–2384. [Google Scholar] [CrossRef] [PubMed]
- Tsai, M.J.; O’Malley, B.W. Molecular mechanisms of action of steroid/thyroid receptor superfamily members. Ann. Rev. Biochem. 1994, 63, 451–486. [Google Scholar] [CrossRef] [PubMed]
- Edwards, D.P. Regulation of signal transduction pathways by estrogen and progesterone. Ann. Rev. Physiol. 2005, 67, 335–376. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.Z.; Zhao, B.C.; Zhou, Y.L.; Zhou, Y.T.; Ma, K.G.; Zhang, J.H. 17 beta-estradiol regulates cultured immature boar Sertoli cell proliferation via the cAMP-ERK1/2 pathway and the estrogen receptor beta. Agric. Sci. China 2010, 9, 1201–1210. [Google Scholar] [CrossRef]
- Lau, W.S.; Chan, R.Y.; Guo, D.A.; Wong, M.S. Ginsenoside Rg1 exerts estrogen-like activities via ligand-independent activation of ERalpha pathway. J. Steroid Biochem. Mol. Boil. 2008, 108, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Razandi, M.; Pedram, A.; Merchenthaler, I.; Greene, G.L.; Levin, E.R. Plasma membrane estrogen receptors exist and functions as dimers. Mol. Endocrinol. 2004, 18, 2854–2865. [Google Scholar] [CrossRef] [PubMed]
- Pedram, A.; Razandi, M.; Levin, E.R. Nature of functional estrogen receptors at the plasma membrane. Mol. Endocrinol. 2006, 20, 1996–2009. [Google Scholar] [CrossRef] [PubMed]
- Darbre, P.D.; Byford, J.R.; Shaw, L.E.; Horton, R.A.; Pope, G.S.; Sauer, M.J. Oestrogenic activity of isobutylparaben in vitro and in vivo. J. Appl. Toxicol. 2002, 22, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Pistol, G.C.; Braicu, C.; Motiu, M.; Gras, M.A.; Marin, D.E.; Stancu, M.; Calin, L.; Israel-Roming, F.; Berindan-Neagoe, I.; Taranu, I. Zearalenone mycotoxin affects immune mediators, MAPK signalling molecules, nuclear receptors and genome-wide gene expression in pig spleen. PLoS ONE 2015, 10. [Google Scholar] [CrossRef] [PubMed]
- Abassi, H.; Ayed-Boussema, I.; Shirley, S.; Abid, S.; Bacha, H.; Micheau, O. The mycotoxin zearalenone enhances cell proliferation, colony formation and promotes cell migration in the human colon carcinoma cell line HCT116. Toxicol. Lett. 2016, 254, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Abid-Essefi, S.; Baudrimont, I.; Hassen, W.; Ouanes, Z.; Mobio, T.A.; Anane, R.; Creppy, E.E.; Bacha, H. DNA fragmentation, apoptosis and cell cycle arrest induced by zearalenone in cultured DOK, Vero and Caco-2 cells: Prevention by Vitamin E. Toxicology 2003, 192, 237–248. [Google Scholar] [CrossRef]
- Yuri, T.; Tsukamoto, R.; Miki, K.; Uehara, N.; Matsuoka, Y.; Tsubura, A. Biphasic effects of zeranol on the growth of estrogen receptor-positive human breast carcinoma cells. Oncol. Rep. 2006, 16, 1307–1312. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tomaszewski, J.; Miturski, R.; Semczuk, A.; Kotarski, J.; Jakowicki, J. Tissue zearalenone concentration in normal, hyperplastic and neoplastic human endometrium. Ginekol. Polska 1998, 69, 363–366. [Google Scholar]
- Yu, Z.; Zhang, L.; Wu, D.; Liu, F. Anti-apoptotic action of zearalenone in MCF-7 cells. Ecotoxicol. Environ. Saf. 2005, 62, 441–446. [Google Scholar] [CrossRef] [PubMed]
- Belli, P.; Bellaton, C.; Durand, J.; Balleydier, S.; Milhau, N.; Mure, M.; Mornex, J.F.; Benahmed, M.; Le Jan, C. Fetal and neonatal exposure to the mycotoxin zearalenone induces phenotypic alterations in adult rat mammary gland. Food Chem. Toxicol. 2010, 48, 2818–2826. [Google Scholar] [CrossRef] [PubMed]
- Becci, P.J.; Johnson, W.D.; Hess, F.G.; Gallo, M.A.; Parent, R.A.; Taylor, J.M. Combined two-generation reproduction-teratogenesis study of zearalenone in the rat. J. Appl. Toxicol. 1982, 2, 201–206. [Google Scholar] [CrossRef] [PubMed]
- Program, N.T. Carcinogenesis bioassay of zearalenone (CAS No. 17924-92-4) in F344/N rats and B6C3F1 mice (feed study). Natl. Toxicol. Program Tech. Rep. 1982, 235, 1–155. [Google Scholar]
- Othmen, O.B.; Essefi, S.A.; Bacha, H. Mutagenic and epigenetic mechanisms of zearalenone: Prevention by vitamin E. World Mycotoxin J. 2008, 1, 369–374. [Google Scholar] [CrossRef]
- Ghedira-Chekir, L.; Maaroufi, K.; Zakhama, A.; Ellouz, F.; Dhouib, S.; Creppy, E.E.; Bacha, H. Induction of a SOS repair system in lysogenic bacteria by zearalenone and its prevention by vitamin E. Chem.-Biol. Interact. 1998, 113, 15–25. [Google Scholar] [CrossRef]
- Liu, K.H.; Sun, X.F.; Feng, Y.Z.; Cheng, S.F.; Li, B.; Li, Y.P.; Shen, W.; Li, L. The impact of zearalenone on the meiotic progression and primordial follicle assembly during early oogenesis. Toxicol. Appl. Pharm. 2017, 329, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Ueno, Y.; Kubota, K. DNA-attacking ability of carcinogenic mycotoxins in recombination-deficient mutant cells of Bacillus subtilis. Cancer Res. 1976, 36, 445–451. [Google Scholar] [PubMed]
- Ouanes, Z.; Ayed-Boussema, I.; Baati, T.; Creppy, E.E.; Bacha, H. Zearalenone induces chromosome aberrations in mouse bone marrow: Preventive effect of 17beta-estradiol, progesterone and vitamin E. Mutat. Res. 2005, 565, 139–149. [Google Scholar] [CrossRef] [PubMed]
- Demple, B.; Harrison, L. Repair of oxidative damage to DNA: Enzymology and biology. Annu. Rev. Biochem. 1994, 63, 915–948. [Google Scholar] [CrossRef] [PubMed]
- Dizdaroglu, M. Oxidative damage to DNA in mammalian chromatin. Mutat. Res. 1992, 275, 331–342. [Google Scholar] [CrossRef]
- Von Sonntag, C. New aspects in the free-radical chemistry of pyrimidine nucleobases. Free Radic. Res. Commun. 1987, 2, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Mesnil, M.; Crespin, S.; Avanzo, J.L.; Zaidan-Dagli, M.L. Defective gap junctional intercellular communication in the carcinogenic process. BBA Biomembr. 2005, 1719, 125–145. [Google Scholar] [CrossRef] [PubMed]
- Zou, H.; Zhuo, L.L.; Han, T.; Hu, D.; Yang, X.K.; Wang, Y.; Yuan, Y.; Gu, J.H.; Bian, J.C.; Liu, X.Z.; et al. Autophagy and gap junctional intercellular communication inhibition are involved in cadmium-induced apoptosis in rat liver cells. Biochem. Biophys. Res. Commun. 2015, 459, 713–719. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.B.; Kinoshita, C.; Kinoshita, Y.; Morrison, R.S. P53 and mitochondrial function in neurons. BBA Mol. Basis Dis. 2014, 1842, 1186–1197. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Tan, X.; Li, Y.; Liang, G.; Ning, Z.; Ma, Y.; Li, Y. Transcriptional profiling analysis of Zearalenone-induced inhibition proliferation on mouse thymic epithelial cell line 1. Ecotoxicol. Environ. Saf. 2018, 153, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Tobey, R.A. Different drugs arrest cells at a number of distinct stages in G2. Nature 1975, 254, 245–247. [Google Scholar] [CrossRef] [PubMed]
- Gao, F.; Jiang, L.P.; Chen, M.; Geng, C.Y.; Yang, G.; Ji, F.; Zhong, L.F.; Liu, X.F. Genotoxic effects induced by zearalenone in a human embryonic kidney cell line. Mut. Res. 2013, 755, 6–10. [Google Scholar] [CrossRef] [PubMed]
- Abid-Essefi, S.; Ouanes, Z.; Hassen, W.; Baudrimont, I.; Creppy, E.; Bacha, H. Cytotoxicity, inhibition of DNA and protein syntheses and oxidative damage in cultured cells exposed to zearalenone. Toxicol. In Vitro 2004, 18, 467–474. [Google Scholar] [CrossRef] [PubMed]
- Tatay, E.; Font, G.; Ruiz, M.J. Cytotoxic effects of zearalenone and its metabolites and antioxidant cell defense in CHO-K1 cells. Food Chem. Toxicol. 2016, 96, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.; Chen, L.K.; Peng, Z.; Wang, D.; Song, Y.D.; Wang, H.Y.; Yao, P.; Yan, H.; Nussler, A.K.; Liu, L.G.; et al. Embryotoxicity caused by DON-induced oxidative stress mediated by Nrf2/HO-1 pathway. Toxins 2017, 9. [Google Scholar] [CrossRef] [PubMed]
- Tatay, E.; Espin, S.; Garcia-Fernandez, A.J.; Ruiz, M.J. Oxidative damage and disturbance of antioxidant capacity by zearalenone and its metabolites in human cells. Toxicol. In Vitro 2017, 45, 334–339. [Google Scholar] [CrossRef] [PubMed]
- So, M.Y.; Tian, Z.P.; Phoon, Y.S.; Sha, S.; Antoniou, M.N.; Zhang, J.W.; Wu, R.S.S.; Tan-Un, K.C. Gene expression profile and toxic effects in human bronchial epithelial cells exposed to zearalenone. PLoS ONE 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- Minervini, F.; Dell’Aquila, M.E. Zearalenone and reproductive function in farm animals. Int. J. Mol. Sci. 2008, 9, 2570–2584. [Google Scholar] [CrossRef] [PubMed]
- Pfeiffer, E.; Hildebrand, A.; Damm, G.; Rapp, A.; Cramer, B.; Humpf, H.U.; Metzler, M. Aromatic hydroxylation is a major metabolic pathway of the mycotoxin zearalenone in vitro. Mol. Nutr. Food Res. 2009, 53, 1123–1133. [Google Scholar] [CrossRef] [PubMed]
- Pfeiffer, E.; Wefers, D.; Hildebrand, A.A.; Fleck, S.C.; Metzler, M. Catechol metabolites of the mycotoxin zearalenone are poor substrates but potent inhibitors of catechol-O-methyltransferase. Mycotoxin Res. 2013, 29, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.; Wang, Z.H.; Liu, Y.; Liu, Y.Y.; Li, H.Y.; Di, C.X.; Wu, Z.H.; Gan, L.; Zhang, H. Carbon ion beams induce hepatoma cell death by NADPH oxidase-mediated mitochondrial damage. J. Cell. Physiol. 2014, 229, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Ray, P.D.; Huang, B.W.; Tsuji, Y. Reactive oxygen species (ROS) homeostasis and redox regulation in cellular signaling. Cell Signal. 2012, 24, 981–990. [Google Scholar] [CrossRef] [PubMed]
- Murphy, M.P. How mitochondria produce reactive oxygen species. Biochem. J. 2009, 417, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Ott, M.; Gogvadze, V.; Orrenius, S.; Zhivotovsky, B. Mitochondria, oxidative stress and cell death. Apoptosis 2007, 12, 913–922. [Google Scholar] [CrossRef] [PubMed]
- Zorov, D.B.; Juhaszova, M.; Sollott, S.J. Mitochondrial ROS-induced ROS release: An update and review. BBA Bioenerg. 2006, 1757, 509–517. [Google Scholar] [CrossRef] [PubMed]
- Tharaux, P.L.; Huber, T.B. How many ways can a podocyte die? Semin. Nephrol. 2012, 32, 394–404. [Google Scholar] [CrossRef] [PubMed]
- Erikstein, B.S.; Hagland, H.R.; Nikolaisen, J.; Kulawiec, M.; Singh, K.K.; Gjertsen, B.T.; Tronstad, K.J. Cellular stress induced by resazurin leads to autophagy and cell death via production of reactive oxygen species and mitochondrial impairment. J. Cell. Biochem. 2010, 111, 574–584. [Google Scholar] [CrossRef] [PubMed]
- Fan, W.T.; Shen, T.T.; Ding, Q.Q.; Lv, Y.A.; Li, L.; Huang, K.H.; Yan, L.P.; Song, S.Q. Zearalenone induces ROS-mediated mitochondrial damage in porcine IPEC-J2 cells. J. Biochem. Mol. Toxicol. 2017, 31. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Li, Q.; Zhang, Z.; Lin, P.; Lei, L.; Wang, A.; Jin, Y. Endoplasmic reticulum stress cooperates in zearalenone-induced cell death of RAW 264.7 macrophages. Int. J. Mol. Sci. 2015, 16, 19780–19795. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.; Jiang, T.; Lin, P.; Chen, H.; Wang, L.; Wang, N.; Zhao, F.; Tang, K.; Zhou, D.; Wang, A.; et al. Apoptosis inducing factor gene depletion inhibits zearalenone-induced cell death in a goat Leydig cell line. Reprod. Toxicol. 2017, 67, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.Y.; Wang, G.X.; Liu, J.L.; Fan, J.J.; Cui, S. Toxic effects of zearalenone and its derivatives alpha-zearalenol on male reproductive system in mice. Reprod. Toxicol. 2007, 24, 381–387. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Lin, P.; Wang, N.; Yang, D.; Wen, X.; Zhou, D.; Wang, A.; Jin, Y. Herp depletion inhibits zearalenone-induced cell death in raw 264.7 macrophages. Toxicol. In Vitro 2016, 32, 115–122. [Google Scholar] [CrossRef] [PubMed]
- Iurlaro, R.; Munoz-Pinedo, C. Cell death induced by endoplasmic reticulum stress. FEBS J. 2016, 283, 2640–2652. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.; Wang, B.; Si, M.; Zou, H.; Song, R.; Gu, J.; Yuan, Y.; Liu, X.; Zhu, G.; Bai, J.; et al. Zearalenone altered the cytoskeletal structure via ER stress-autophagy-oxidative stress pathway in mouse TM4 Sertoli cells. Sci. Rep. 2018, 8, 3320. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Yu, J.Y.; Lim, S.S.; Son, Y.O.; Kim, D.H.; Lee, S.A.; Shi, X.L.; Lee, J.C. Cellular mechanisms of the cytotoxic effects of the zearalenone metabolites alpha-zearalenol and beta-zearalenol on RAW264.7 macrophages. Toxicol. In Vitro 2013, 27, 1007–1017. [Google Scholar] [CrossRef] [PubMed]
- Banjerdpongchai, R.; Kongtawelert, P.; Khantamat, O.; Srisomsap, C.; Chokchaichamnankit, D.; Subhasitanont, P.; Svasti, J. Mitochondrial and endoplasmic reticulum stress pathways cooperate in zearalenone-induced apoptosis of human leukemic cells. J. Hematol. Oncol. 2010, 3. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.L.; Hu, J.; Guo, B.P.; Niu, Y.R.; Xiao, C.; Xu, Y.X. Exploration of intrinsic and extrinsic apoptotic pathways in zearalenone-treated rat sertoli cells. Environ Toxicol 2016, 31, 1731–1739. [Google Scholar] [CrossRef] [PubMed]
- Ayed-Boussema, I.; Bouaziz, C.; Rjiba, K.; Valenti, K.; Laporte, F.; Bacha, H.; Hassen, W. The mycotoxin zearalenone induces apoptosis in human hepatocytes (HepG2) via p53-dependent mitochondrial signaling pathway. Toxicol. In Vitro 2008, 22, 1671–1680. [Google Scholar] [CrossRef] [PubMed]
- Ren, Z.H.; Deng, H.D.; Deng, Y.T.; Liang, Z.; Deng, J.H.; Zuo, Z.C.; Hu, Y.C.; Shen, L.H.; Yu, S.M.; Cao, S.Z. Combined effects of deoxynivalenol and zearalenone on oxidative injury and apoptosis in porcine splenic lymphocytes in vitro. Exp. Toxicol. Pathol. 2017, 69, 612–617. [Google Scholar] [CrossRef] [PubMed]
- Ahamed, S.; Foster, J.S.; Bukovsky, A.; Wimalasena, J. Signal transduction through the Ras/Erk pathway is essential for the mycoestrogen zearalenone-induced cell-cycle progression in MCF-7 cells. Mol. Carcinog. 2001, 30, 88–98. [Google Scholar] [CrossRef]
- Zheng, J.; Xiao, X.; Liu, J.; Zheng, S.; Yin, Q.; Yu, Y. Growth-promoting effect of environmental endocrine disruptors on human neuroblastoma SK-N-SH cells. Environ. Toxicol. Pharmacol. 2007, 24, 189–193. [Google Scholar] [CrossRef] [PubMed]
- Yan, W.J.; Dong, H.L.; Xiong, L.Z. The protective roles of autophagy in ischemic preconditioning. Acta Pharmacol. Sin. 2013, 34, 636–643. [Google Scholar] [CrossRef] [PubMed]
- Kiriyama, Y.; Nochi, H. The function of autophagy in neurodegenerative diseases. Int. J. Mol. Sci. 2015, 16, 26797–26812. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.J.; Yang, W.; Wang, C.; He, W.S.; Deng, H.Y.; Yan, Y.G.; Zhang, J.; Xiang, Y.X.; Wang, W.J. Autophagy: A double-edged sword in intervertebral disk degeneration. Clin. Chim. Acta 2016, 457, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Ben Salem, I.; Boussabbeh, M.; Da Silva, J.P.; Guilbert, A.; Bacha, H.; Abid-Essefi, S.; Lemaire, C. Sirt1 protects cardiac cells against apoptosis induced by zearalenone or its metabolites alpha- and beta-zearalenol through an autophagy-dependent pathway. Toxicol. Appl. Pharm. 2017, 314, 82–90. [Google Scholar] [CrossRef] [PubMed]
- Duan, P.; Hu, C.H.; Quan, C.; Yu, T.T.; Huang, W.T.; Chen, W.; Tang, S.; Shi, Y.Q.; Martin, F.L.; Yang, K.D. 4-nonylphenol induces autophagy and attenuates mTOR-p70S6K/4EBP1 signaling by modulating AMPK activation in Sertoli cells. Toxicol. Lett. 2017, 267, 21–31. [Google Scholar] [CrossRef] [PubMed]
- Ozcan, L.; Tabas, I. Role of endoplasmic reticulum stress in metabolic disease and other disorders. Annu. Rev. Med. 2012, 63, 317–328. [Google Scholar] [CrossRef] [PubMed]
- Audenaert, K.; Vanheule, A.; Hofte, M.; Haesaert, G. Deoxynivalenol: A major player in the multifaceted response of fusarium to its environment. Toxins 2013, 6, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Zhang, A.; Shi, Z.; He, C.; Ding, J.; Wang, X.; Ma, J.; Zhang, H. A mitochondria-mediated apoptotic pathway induced by deoxynivalenol in human colon cancer cells. Toxicol. In Vitro 2012, 26, 414–420. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Wang, Q.C.; Zhu, C.C.; Liu, J.; Zhang, Y.; Cui, X.S.; Kim, N.H.; Sun, S.C. Deoxynivalenol exposure induces autophagy/apoptosis and epigenetic modification changes during porcine oocyte maturation. Toxicol. Appl. Pharmacol. 2016, 300, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Li, J.; Li, F.; Hu, C.A.; Liao, P.; Tan, K.; Tan, B.; Xiong, X.; Liu, G.; Li, T.; et al. Autophagy protects intestinal epithelial cells against deoxynivalenol toxicity by alleviating oxidative stress via IKK signaling pathway. Free Radic. Biol. Med. 2015, 89, 944–951. [Google Scholar] [CrossRef] [PubMed]
- Qi, Y.; Zhang, H.; Wu, G.; Zhang, H.; Gu, L.; Wang, L.; Qian, H.; Qi, X. Mitigation effects of proanthocyanidins with different structures on acrylamide formation in chemical and fried potato crisp models. Food Chem. 2018, 250, 98–104. [Google Scholar] [CrossRef] [PubMed]
- Long, M.; Yang, S.; Zhang, Y.; Li, P.; Han, J.; Dong, S.; Chen, X.; He, J. Proanthocyanidin protects against acute zearalenone-induced testicular oxidative damage in male mice. Environ. Sci. Pollut. Res. Int. 2017, 24, 938–946. [Google Scholar] [CrossRef] [PubMed]
- Samuni, Y.; Goldstein, S.; Dean, O.M.; Berk, M. The chemistry and biological activities of N-acetylcysteine. Biochim. Biophys. Acta 2013, 1830, 4117–4129. [Google Scholar] [CrossRef] [PubMed]
- Seng, D.; Hoell, A.; Mussler, B.; Eisenbrand, G. Interaction of estrogenic chemicals and phytoestrogens with estrogen receptor alpha and beta. Naunyn-Schmiedebergs Arch. Pharmacol. 2001, 363, R144. [Google Scholar]
| Mycotoxin | Exposure Dose | Exposure Period | Cell Line or Tissues | Effects | Ref |
|---|---|---|---|---|---|
| ZEA | 10−10–10−8 M | 6 d | T47D cells | Cell proliferation  | [10] |
| ZEA | 10−10–10−8 M | 6 d | MCF-7 cells | Cell proliferation  | [10] |
| ZEA | 10−11–10−6 M | 24 h | KK-1 cells | Cell proliferation  | [2] |
| ZEA | 10−7 M | 36 h | MCF-7 cells | Cell proliferation  | [94] |
| ZEA | 10−8–10−9 M | 7d | T47D cells | Cell proliferation  | [11] |
| α-ZEL | 10−9–10−6 M | 7d | T47D cells | Cell proliferation  | [11] |
| α-ZAL | 10−7–10−5 M | 5 d | BMS Cells | Cell proliferation  | [12] |
| ZEA | 10−11–10−5 M | 72h | MCF-7 cells | Cell proliferation  | [36] |
| ZEA | 10−8–10−5 M | 6d | MCF-7 cells, T47D cells, | Cell proliferation  | [10] |
| ZEA, FB1 | ZEA 9.4 µM | 48 h | Granulosa cells | Cell proliferation  | [13] |
| FB1 3.4 µM | |||||
| ZEA | 10−9–10−6 M | 0-120 h | HCT116 | Cell proliferation  | [45] |
| ZEA | 10−8 M | 72 h | TM3 cells | Cell proliferation  | [8] |
Cell apoptosis  | |||||
| ZEA | 10−8–10−6 | 72 h | SK-N-SH cells | Cell proliferation  | [95] |
| ZEA | 0.1, 1.0 and 10.0 mg per kg body weight per day | 10 months | Liver, uterine | The weights of liver and uterine  | [51] |
| ZEA | 60, 90, and 120 µM | 24 h | Porcine granulosa cells | Cell apoptosis  | [18] |
| ZEA | 0–600 µM | 24 h | Mouse Leydig cells | Cell apoptosis  | [22] |
| ZEA | 50–300 µM | 24 h | HEK293 cells | Cell viability  | [15] |
| ZEA | 25, 50, 75, and 100 μM | 24 h | Mouse ES cells | Cell apoptosis  | [21] |
| ZEA | 20 μM | 24 h | PK15 cells | Cell viability  | [20] |
| ZEA | 1, 10, 20, 40, 60, 80 and 100 μM | 24 h | TM4 cells | Cell apoptosis  | [19] |
Cell viability  | |||||
| ZEA | 15–60 μM | 24 h | Sertoli cells | Cell death  | [14] |
Cell viability  | |||||
| ZEA | 3–300 μM | 24 h | HEK 293 cells | Cell apoptosisthe  | [15] |
cell viability  | |||||
| ZEA | 10, 20 and 40 μM | 24 h | Caco-2, DOK and Verocells | Cell apoptosis  | [46] |
| ZEA | 10−5 M | 6 d | T47D cells | Cell death  | [10] |
| ZEA | 10−5 M | 6 d | MCF-7 cells | Cell death  | [10] |
| ZEA | 10−6–10−4 M | 24 h | KK-1 cells | Cell death  | [2] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zheng, W.; Wang, B.; Li, X.; Wang, T.; Zou, H.; Gu, J.; Yuan, Y.; Liu, X.; Bai, J.; Bian, J.; et al. Zearalenone Promotes Cell Proliferation or Causes Cell Death? Toxins 2018, 10, 184. https://doi.org/10.3390/toxins10050184
Zheng W, Wang B, Li X, Wang T, Zou H, Gu J, Yuan Y, Liu X, Bai J, Bian J, et al. Zearalenone Promotes Cell Proliferation or Causes Cell Death? Toxins. 2018; 10(5):184. https://doi.org/10.3390/toxins10050184
Chicago/Turabian StyleZheng, Wanglong, Bingjie Wang, Xi Li, Tao Wang, Hui Zou, Jianhong Gu, Yan Yuan, Xuezhong Liu, Jianfa Bai, Jianchun Bian, and et al. 2018. "Zearalenone Promotes Cell Proliferation or Causes Cell Death?" Toxins 10, no. 5: 184. https://doi.org/10.3390/toxins10050184
APA StyleZheng, W., Wang, B., Li, X., Wang, T., Zou, H., Gu, J., Yuan, Y., Liu, X., Bai, J., Bian, J., & Liu, Z. (2018). Zearalenone Promotes Cell Proliferation or Causes Cell Death? Toxins, 10(5), 184. https://doi.org/10.3390/toxins10050184





