Toxic Cyanobacteria in Svalbard: Chemical Diversity of Microcystins Detected Using a Liquid Chromatography Mass Spectrometry Precursor Ion Screening Method
Abstract
1. Introduction
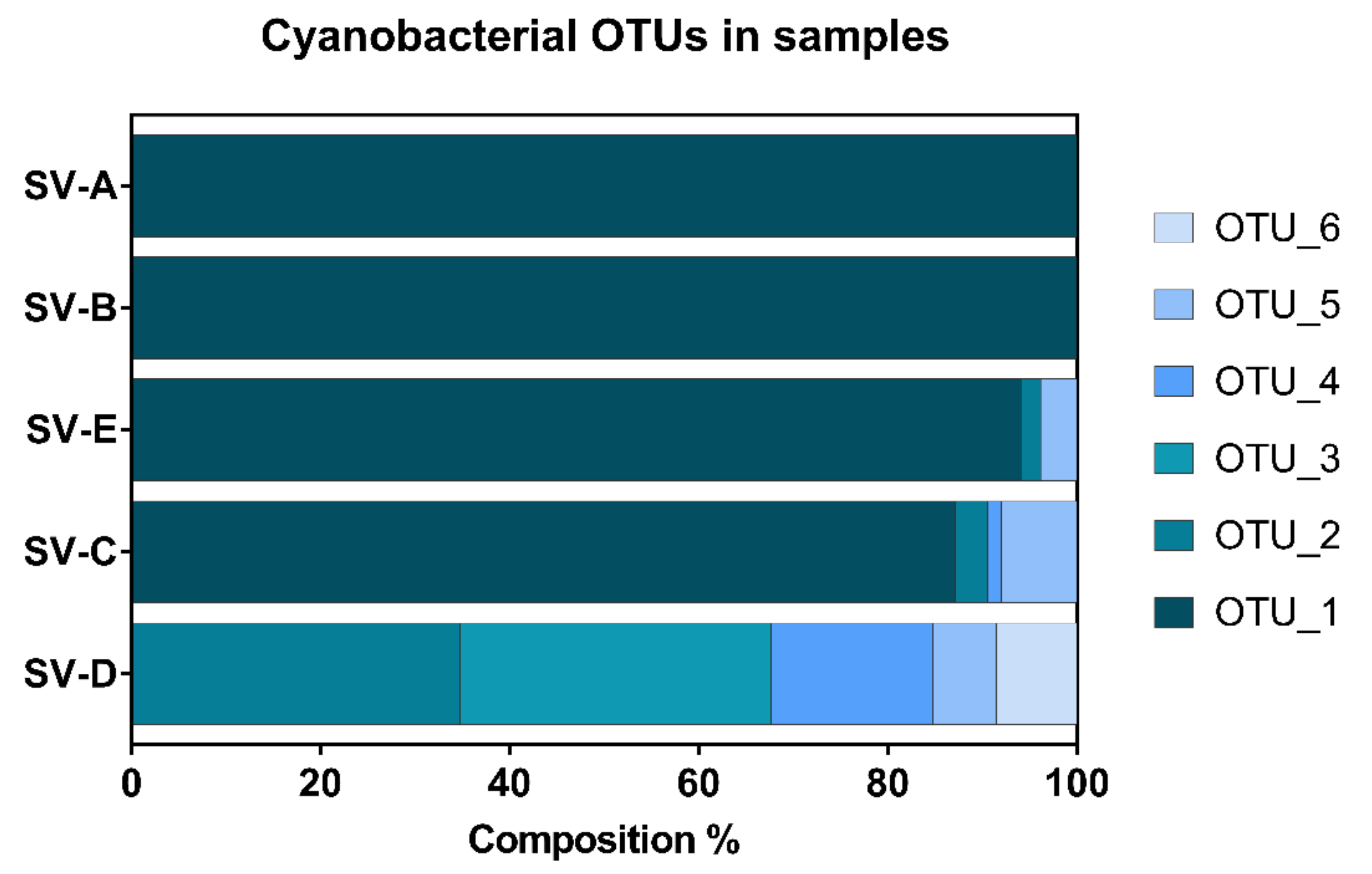
2. Results
3. Discussion
4. Materials and Methods
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Mowe, M.A.D.; Mitrovic, S.M.; Lim, R.P.; Furey, A.; Yeo, D.C.J. Tropical cyanobacterial blooms: A review of prevalence, problem taxa, toxins and influencing environmental factors. J. Limnol. 2014, 73. [Google Scholar] [CrossRef]
- O’Neil, J.M.; Davis, T.W.; Burford, M.A.; Gobler, C.J. The rise of harmful cyanobacteria blooms: The potential roles of eutrophication and climate change. Harmful Algae 2012, 14, 313–334. [Google Scholar] [CrossRef]
- Davis, T.W.; Berry, D.L.; Boyer, G.L.; Gobler, C.J. The effects of temperature and nutrients on the growth and dynamics of toxic and non-toxic strains of Microcystis during cyanobacteria blooms. Harmful Algae 2009, 8, 715–725. [Google Scholar] [CrossRef]
- Posch, T.; Köster, O.; Salcher, M.M.; Pernthaler, J. Harmful filamentous cyanobacteria favoured by reduced water turnover with lake warming. Nat. Clim. Chang. 2012, 2, 809–813. [Google Scholar] [CrossRef]
- Chorus, I.; Bartram, J. (Eds.) Toxic Cyanobacteria in Water: A Guide to Their Public Health Consequences, Monitoring and Management; E & FN Spon: London, UK; New York, NY, USA, 1999; ISBN 0-419-23930-8. [Google Scholar]
- Dietrich, D.R.; Fischer, A.; Michel, C.; Hoeger, S.J. Toxin mixture in cyanobacterial blooms—A critical comparison of reality with current procedures employed in human health risk assessment cyanobacterial metabolites: Health hazards for humans? In Cyanobacterial Harmful Algal Blooms; Hudnell, H.K., Ed.; Springer: New York, NY, USA, 2008; pp. 885–912. [Google Scholar]
- Feurstein, D.; Kleinteich, J.; Heussner, A.H.A.H.; Stemmer, K.; Dietrich, D.R. Investigation of Microcystin Congener–Dependent Uptake into Primary Murine Neurons. Environ. Health Perspect. 2010, 118, 1370–1375. [Google Scholar] [CrossRef] [PubMed]
- Feurstein, D.; Stemmer, K.; Kleinteich, J.; Speicher, T.; Dietrich, D.R. Microcystin congener- and concentration-dependent induction of murine neuron apoptosis and neurite degeneration. Toxicol. Sci. 2011, 124, 424–431. [Google Scholar] [CrossRef] [PubMed]
- Zimmer, C. Cyanobacteria Are Far from Just Toledo’s Problem. The New York Times. 2014. Available online: http://www.nytimes.com/2014/08/07/science/cyanobacteria-are-far-from-just-toledos-problem.html (accessed on 15 June 2016).
- Quiblier, C.; Wood, S.; Echenique-Subiabre, I.; Heath, M.; Villeneuve, A.; Humbert, J.-F. A review of current knowledge on toxic benthic freshwater cyanobacteria—Ecology, toxin production and risk management. Water Res. 2013, 47, 5464–5479. [Google Scholar] [CrossRef]
- Kaasalainen, U.; Fewer, D.P.; Jokela, J.; Wahlsten, M.; Sivonen, K.; Rikkinen, J. Cyanobacteria produce a high variety of hepatotoxic peptides in lichen symbiosis. Proc. Natl. Acad. Sci. USA 2012, 109, 5886–5891. [Google Scholar] [CrossRef] [PubMed]
- Puddick, J.; Prinsep, M.; Wood, S.; Cary, S.; Hamilton, D.; Holland, P. Further Characterization of Glycine-Containing Microcystins from the McMurdo Dry Valleys of Antarctica. Toxins 2015, 7, 493–515. [Google Scholar] [CrossRef] [PubMed]
- Vincent, W.F. Cyanobacterial Dominance in the Polar Regions. In The Ecology of Cyanobacteria; Whitton, B.A., Potts, M., Eds.; Springer: Dordrecht, The Netherlands, 2000; pp. 321–340. ISBN 978-0-7923-4735-4. [Google Scholar]
- Vincent, W.F.; Quesada, A. Cyanobacteria in High Latitude Lakes, Rivers and Seas. In Ecology of Cyanobacteria II Their Diversity in Space and Time; Whitton, B.A., Ed.; Springer: Dordrecht, The Netherlands, 2012; pp. 371–385. ISBN 978-94-007-3855-3. [Google Scholar]
- Hitzfeld, B.C.; Lampert, C.S.; Spaeth, N.; Mountfort, D.; Kaspar, H.; Dietrich, D.R. Toxin production in cyanobacterial mats from ponds on the McMurdo Ice Shelf, Antarctica. Toxicon 2000, 38, 1731–1748. [Google Scholar] [CrossRef]
- Jungblut, A.; Hoeger, S.; Mountfort, D. Characterization of microcystin production in an Antarctic cyanobacterial mat community. Toxicon 2006, 47, 271–278. [Google Scholar] [CrossRef] [PubMed]
- Wood, S.A.; Mountfort, D.; Selwood, A.I.; Holland, P.T.; Puddick, J.; Cary, S.C. Widespread distribution and identification of eight novel microcystins in Antarctic cyanobacterial mats. Appl. Environ. Microbiol. 2008, 74, 7243–7251. [Google Scholar] [CrossRef] [PubMed]
- Kleinteich, J.; Wood, S.A.; Puddick, J.; Schleheck, D.; Küpper, F.C.; Dietrich, D. Potent toxins in Arctic environments—Presence of saxitoxins and an unusual microcystin variant in Arctic freshwater ecosystems. Chem. Biol. Interact. 2013, 206, 423–431. [Google Scholar] [CrossRef] [PubMed]
- Kleinteich, J.; Hildebrand, F.; Wood, S.A.; Cirés, S.; Agha, R.; Quesada, A.; Pearce, D.A.; Convey, P.; Küpper, F.C.; Dietrich, D.R. Diversity of toxin and non-toxin containing cyanobacterial mats of meltwater ponds on the Antarctic Peninsula: A pyrosequencing approach. Antarct. Sci. 2014, 26, 521–532. [Google Scholar] [CrossRef]
- Cirés, S.; Casero, M.; Quesada, A. Toxicity at the Edge of Life: A Review on Cyanobacterial Toxins from Extreme Environments. Mar. Drugs 2017, 15, 233. [Google Scholar] [CrossRef] [PubMed]
- Meriluoto, J.; Spoof, L.; Codd, G.A. (Eds.) Handbook of Cyanobacterial Monitoring and Cyanotoxin Analysis; John Wiley & Sons: Chichester, UK, 2016; ISBN 9781119068761. [Google Scholar]
- Jungblut, A.D.; Neilan, B.A. Molecular identification and evolution of the cyclic peptide hepatotoxins, microcystin and nodularin, synthetase genes in three orders of cyanobacteria. Arch. Microbiol. 2006, 185, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Fischer, A.; Hoeger, S.J.; Stemmer, K.; Feurstein, D.J.; Knobeloch, D.; Nussler, A.; Dietrich, D.R. The role of organic anion transporting polypeptides (OATPs/SLCOs) in the toxicity of different microcystin congeners in vitro: A comparison of primary human hepatocytes and OATP-transfected HEK293 cells. Toxicol. Appl. Pharmacol. 2010, 245, 9–20. [Google Scholar] [CrossRef] [PubMed]
- De los Rios, A.; Ascaso, C.; Wierzchos, J.; Fernández-Valiente, E.; Quesada, A. Microstructural characterization of cyanobacterial mats from the McMurdo Ice Shelf, Antarctica. Appl. Environ. Microbiol. 2004, 70, 569–580. [Google Scholar] [CrossRef] [PubMed]
- Nagata, S.; Tsutsumi, T.; Hasegawa, A.; Yoshida, F.; Ueno, Y. Enzyme Immunoassay for Direct Determination of Microcystins in Environmental Water. J. AOAC Int. 1997, 80, 408–417. [Google Scholar]
- Rivasseau, C.; Racaud, P.; Deguin, A.; Hennion, M.C. Evaluation of an ELISA kit for the monitoring of microcystins (cyanobacterial toxins) in water and algae environmental samples. Environ. Sci. Technol. 1999, 33, 1520–1527. [Google Scholar] [CrossRef]
- Puddick, J.; Prinsep, M.; Wood, S.; Kaufononga, S.; Cary, S.; Hamilton, D. High Levels of Structural Diversity Observed in Microcystins from Microcystis CAWBG11 and Characterization of Six New Microcystin Congeners. Mar. Drugs 2014, 12, 5372–5395. [Google Scholar] [CrossRef] [PubMed]
- Puddick, J. Spectroscopic Investigations of Oligopeptides from Aquatic Cyanobacteria: Characterisation of New Oligopeptides, Development of Microcystin Quantification Tools and Investigations into Microcystin Production. Ph.D. Thesis, The University of Waikato, Hamilton, New Zealand, 2013. [Google Scholar]
- Strunecký, O.; Komárek, J.; Johansen, J.; Lukešová, A.; Elster, J. Molecular and morphological criteria for revision of the genus Microcoleus (Oscillatoriales, Cyanobacteria). J. Phycol. 2013, 49, 1167–1180. [Google Scholar] [CrossRef] [PubMed]
- Biondi, N.; Tredici, M.R.; Taton, A.; Wilmotte, A.; Hodgson, D.A.; Losi, D.; Marinelli, F. Cyanobacteria from benthic mats of Antarctic lakes as a source of new bioactivities. J. Appl. Microbiol. 2008, 105, 105–115. [Google Scholar] [CrossRef] [PubMed]
- Wynn-Williams, D.D.; Edwards, H.G.M.; Garcia-Pichel, F. Functional biomolecules of Antarctic stromatolitic and endolithic cyanobacterial communities. Eur. J. Phycol. 1999, 34, 381–391. [Google Scholar] [CrossRef]
- Asthana, R.K.; Tripathi, M.K.; Srivastava, A.; Singh, A.P.; Singh, S.P.; Nath, G.; Srivastava, R.; Srivastava, B.S. Isolation and identification of a new antibacterial entity from the Antarctic cyanobacterium Nostoc CCC 537. J. Appl. Phycol. 2009, 21, 81–88. [Google Scholar] [CrossRef]
- Chrapusta, E.; Węgrzyn, M.; Zabaglo, K.; Kaminski, A.; Adamski, M.; Wietrzyk, P.; Bialczyk, J. Microcystins and anatoxin-a in Arctic biocrust cyanobacterial communities. Toxicon 2015, 101, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Tillett, D.; Dittmann, E.; Erhard, M.; Von Döhren, H.; Börner, T.; Neilan, B.A. Structural organization of microcystin biosynthesis in Microcystis aeruginosa PCC7806: An integrated peptide-polyketide synthetase system. Chem. Biol. 2000, 7, 753–764. [Google Scholar] [CrossRef]
- Fewer, D.P.; Köykkä, M.; Halinen, K.; Jokela, J.; Lyra, C.; Sivonen, K. Culture-independent evidence for the persistent presence and genetic diversity of microcystin-producing Anabaena (Cyanobacteria) in the Gulf of Finland. Environ. Microbiol. 2009, 11, 855–866. [Google Scholar] [CrossRef] [PubMed]
- Oksanen, I.; Jokela, J.; Fewer, D.P.; Wahlsten, M.; Rikkinen, J.; Sivonen, K. Discovery of rare and highly toxic microcystins from lichen-associated cyanobacterium Nostoc sp. strain IO-102-I. Appl. Environ. Microbiol. 2004, 70, 5756–5763. [Google Scholar] [CrossRef] [PubMed]
- Solheim, B.; Endal, A.; Vigstad, H. Nitrogen fixation in Arctic vegetation and soils from Svalbard, Norway. Polar Biol. 1996, 16, 35–40. [Google Scholar] [CrossRef]
- Tedersoo, L.; Anslan, S.; Bahram, M.; Põlme, S.; Riit, T.; Liiv, I.; Kõljalg, U.; Kisand, V.; Nilsson, H.; Hildebrand, F.; et al. Shotgun metagenomes and multiple primer pair-barcode combinations of amplicons reveal biases in metabarcoding analyses of fungi. MycoKeys 2015, 10, 1–43. [Google Scholar] [CrossRef]
- Lee, C.K.; Herbold, C.W.; Polson, S.W.; Wommack, K.E.; Williamson, S.J.; McDonald, I.R.; Cary, S.C. Groundtruthing next-gen sequencing for microbial ecology-biases and errors in community structure estimates from PCR amplicon pyrosequencing. PLoS ONE 2012, 7, e44224. [Google Scholar] [CrossRef] [PubMed]
- Pessi, I.S.; de Maalouf, P.C.; Laughinghouse, H.D.; Baurain, D.; Wilmotte, A. On the use of high-throughput sequencing for the study of cyanobacterial diversity in Antarctic aquatic mats. J. Phycol. 2016, 52, 356–368. [Google Scholar] [CrossRef] [PubMed]
- Kleinteich, J.; Hildebrand, F.; Bahram, M.; Voigt, A.Y.; Wood, S.A.; Jungblut, A.D.; Küpper, F.C.; Quesada, A.; Camacho, A.; Pearce, D.A.; et al. Pole-to-Pole Connections: Similarities between Arctic and Antarctic Microbiomes and Their Vulnerability to Environmental Change. Front. Ecol. Evol. 2017, 5, 137. [Google Scholar] [CrossRef]
- Jungblut, A.D.; Lovejoy, C.; Vincent, W.F. Global distribution of cyanobacterial ecotypes in the cold biosphere. ISME J. 2010, 4, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Kleinteich, J.; Wood, S.A.; Küpper, F.C.; Camacho, A.; Quesada, A.; Frickey, T.; Dietrich, D.R. Temperature-related changes in polar cyanobacterial mat diversity and toxin production. Nat. Clim. Chang. 2012, 2, 356–360. [Google Scholar] [CrossRef]
- Fraser, C.I.; Terauds, A.; Smellie, J.; Convey, P.; Chown, S.L. Geothermal activity helps life survive glacial cycles. Proc. Natl. Acad. Sci. USA 2014, 111, 5634–5639. [Google Scholar] [CrossRef] [PubMed]
- Stocker, T.F.; Qin, D.; Plattner, G.-K.; Tignor, M.; Allen, S.K.; Boschung, J.; Nauels, A.; Xia, Y.; Bex, V.; Midgley, P.M. IPCC, 2013: Climate Change 2013: The Physical Science Basis; Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; IPCC: Cambridge, UK; New York, NY, USA, 2013. [Google Scholar]
- Niedermeyer, T.; Strohalm, M. mMass as a software tool for the annotation of cyclic peptide tandem mass spectra. PLoS ONE 2012, 7, e44913. [Google Scholar] [CrossRef] [PubMed]
- Hummert, C.; Dahlmann, J.; Reinhardt, K.; Dang, H.P.H.; Dang, D.K.; Luckas, B. Liquid chromatography-mass spectrometry identification of microcystins in Microcystis aeruginosa strain from lake Thanh Cong, Hanoi, Vietnam. Chromatographia 2001, 54, 569–575. [Google Scholar] [CrossRef]
- Welker, M.; Christiansen, G.; von Döhren, H. Diversity of coexisting Planktothrix (Cyanobacteria) chemotypes deduced by mass spectral analysis of microcystins and other oligopeptides. Arch. Microbiol. 2004, 182, 288–298. [Google Scholar] [CrossRef] [PubMed]
- Miles, C.O.; Sandvik, M.; Haande, S.; Nonga, H.; Ballot, A. LC-MS Analysis with Thiol Derivatization to Differentiate [Dhb7]- from [Mdha7]-Microcystins: Analysis of Cyanobacterial Blooms, Planktothrix Cultures and European Crayfish from Lake Steinsfjorden, Norway. Environ. Sci. Technol. 2013, 47, 4080–4087. [Google Scholar] [CrossRef] [PubMed]
- Puddick, J.; Prinsep, M.; Wood, S.; Miles, C.; Rise, F.; Cary, S.; Hamilton, D.; Wilkins, A. Structural Characterization of New Microcystins Containing Tryptophan and Oxidized Tryptophan Residues. Mar. Drugs 2013, 11, 3025–3045. [Google Scholar] [CrossRef] [PubMed]
- Cirés, S.; Wörmer, L.; Ballot, A.; Agha, R.; Wiedner, C.; Velázquez, D.; Casero, M.C.; Quesada, A. Phylogeography of cylindrospermopsin and paralytic shellfish toxin-producing Nostocales cyanobacteria from Mediterranean Europe (Spain). Appl. Environ. Microbiol. 2014, 80, 1359–1370. [Google Scholar] [CrossRef] [PubMed]
- Nübel, U.; Garcia-Pichel, F.; Muyzer, G. PCR primers to amplify 16S rRNA genes from cyanobacteria. Appl. Environ. Microbiol. 1997, 63, 3327–3332. [Google Scholar] [PubMed]
- Weisburg, W.G.; Barns, S.M.; Pelletier, D.A.; Lane, D.J. 16S ribosomal DNA amplification for phylogenetic study. J. Bacteriol. 1991, 173, 697–703. [Google Scholar] [CrossRef] [PubMed]
- Hildebrand, F.; Tadeo, R.; Voigt, A.Y.; Bork, P.; Raes, J. LotuS: An efficient and user-friendly OTU processing pipeline. Microbiome 2014, 2, 30. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.C. UPARSE: Highly accurate OTU sequences from microbial amplicon reads. Nat. Methods 2013, 10, 996–998. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.C.; Haas, B.J.; Clemente, J.C.; Quince, C.; Knight, R. UCHIME improves sensitivity and speed of chimera detection. Bioinformatics 2011, 27, 2194–2200. [Google Scholar] [CrossRef] [PubMed]
- Magoč, T.; Salzberg, S.L. FLASH: Fast length adjustment of short reads to improve genome assemblies. Bioinformatics 2011, 27, 2957–2963. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Garrity, G.M.; Tiedje, J.M.; Cole, J.R. Naive Bayesian Classifier for Rapid Assignment of rRNA Sequences into the New Bacterial Taxonomy. Appl. Environ. Microbiol. 2007, 73, 5261–5267. [Google Scholar] [CrossRef] [PubMed]
- Hildebrand, F.; Ebersbach, T.; Nielsen, H.B.; Li, X.; Sonne, S.B.; Bertalan, M.; Dimitrov, P.; Madsen, L.; Qin, J.; Wang, J.; et al. A comparative analysis of the intestinal metagenomes present in guinea pigs (Cavia porcellus) and humans (Homo sapiens). BMC Genom. 2012, 13, 514. [Google Scholar] [CrossRef] [PubMed]
- Saary, P.; Forslund, K.; Bork, P.; Hildebrand, F. RTK: Efficient rarefaction analysis of large datasets. Bioinformatics 2017, 33, 2594–2595. [Google Scholar] [CrossRef] [PubMed]
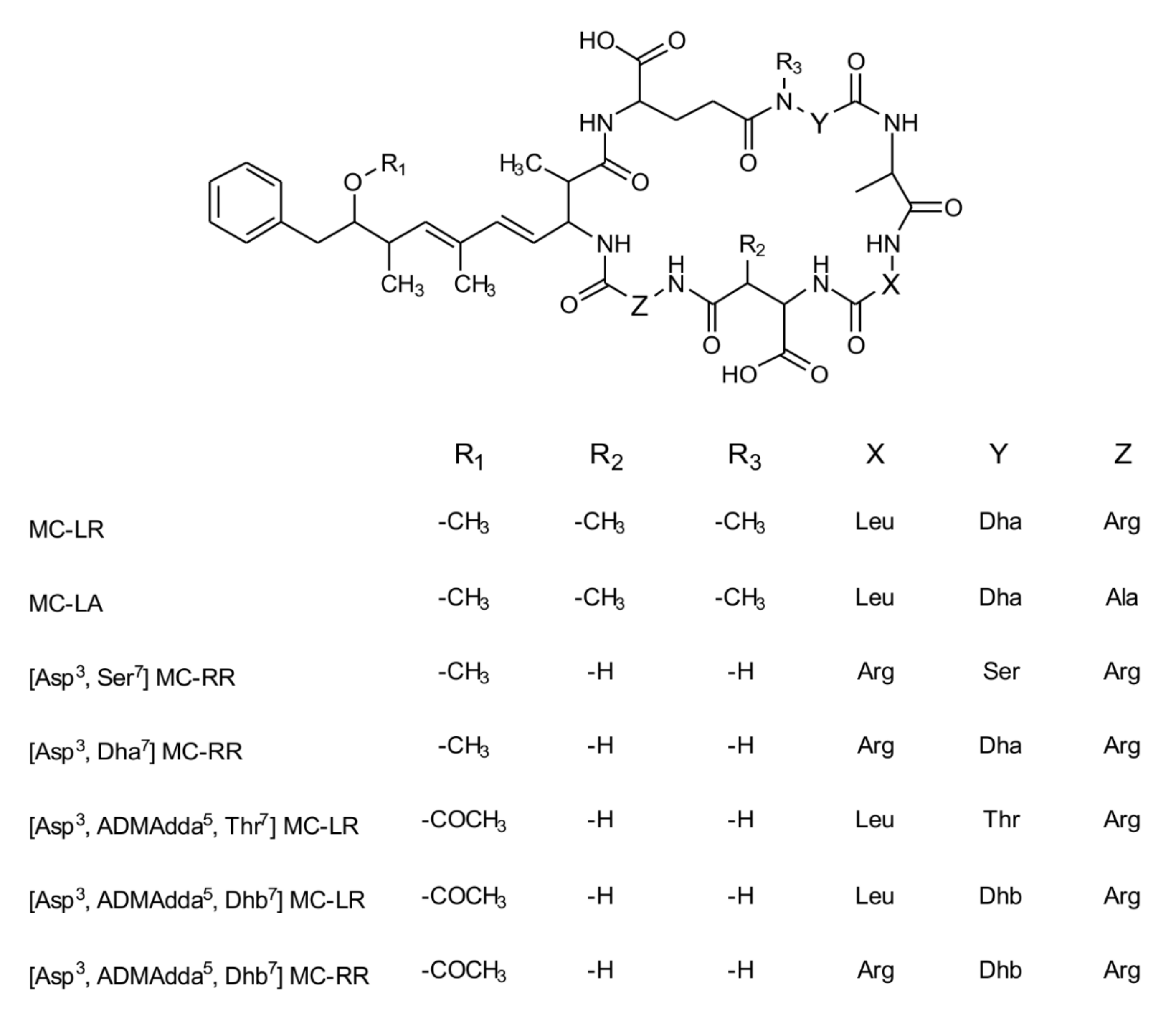
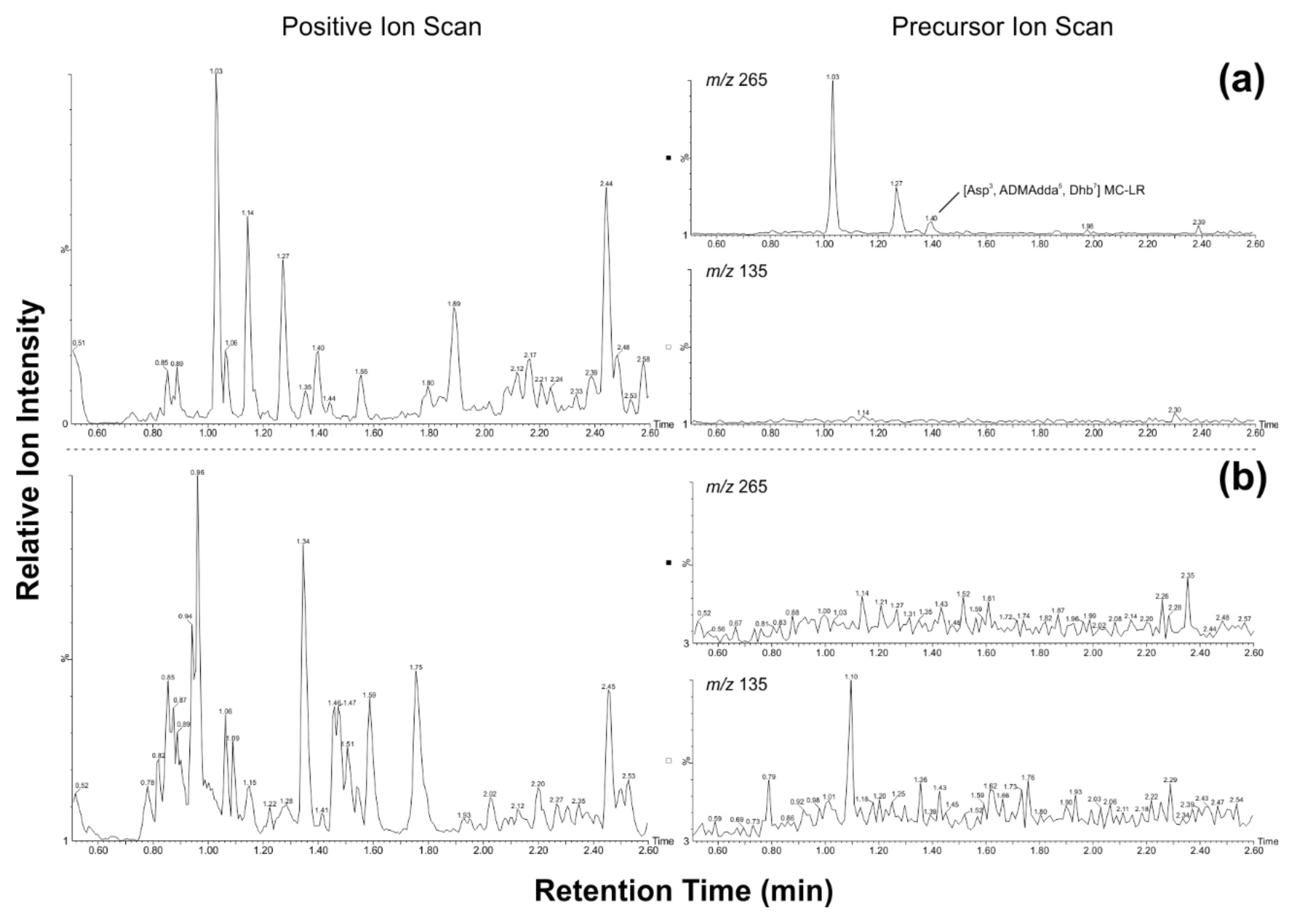
| Sample | Extracted Mass [g] | ELISA [µg/L] | LC-MS Precursor Ion Category (1–3) | LC-MS/MS Characterization | Genes |
|---|---|---|---|---|---|
| SV-A | 0.047 | n.a. | 2 | n.d. | NRPS, PKS |
| SV-B | 0.078 | n.a. | 3 | n.a. | NRPS, PKS |
| SV-C | 0.089 | n.a. | 3 | n.a. | - |
| SV-D N | 0.093 | n.a. | 1 | [Asp3, ADMAdda5, Dhb7] MC-LR | NRPS, PKS, mcyE, mcyB |
| SV-E N | 0.068 | n.a. | 3 | n.a. | NRPS, PKS, mcyE |
| SV-2 | 2.017 | 8 | 3 | n.a. | NRPS, PKS |
| SV-8 | 0.679 | 9 | 3 | n.a. | PKS |
| SV-13 | 0.109 | 14 | 3 | n.a. | - |
| SV-14 | 0.025 | 0 | n.a. | n.a. | NRPS, PKS |
| SV-16 | 0.105 | 19 | 3 | n.a. | NRPS, PKS |
| SV-17 | 0.262 | 18 | 2 | n.d. | NRPS, PKS |
| SV-24 | 1.010 | 2 | n.a. | n.a. | - |
| SV-28 | 0.893 | 8 | n.a. | n.a. | - |
| SV-39 | 0.020 | 0 | n.a. | n.a. | NRPS, PKS |
| SV-40 N | 0.815 | 2 | n.a. | n.a. | - |
| SV-46 | 2.438 | 6 | n.a. | n.a. | - |
| SV-49 N | 0.663 | 54 | 1 | [Asp3, ADMAdda5, Dhb7] MC-LR | -* |
| SV-54 | 1.267 | >STD | 3 | n.a. | - |
| SV-56 N | 0.569 | 37 | 3 | n.a. | - |
| SV-65 | 1.805 | 3 | n.a. | n.a. | - |
| SV-74 N | 0.101 | >STD | 1 | MC-LA | -* |
| SV-75 N | 0.056 | >STD | 1 | [Asp3, ADMAdda5, Dhb7] MC-RR [Asp3, ADMAdda5, Dhb7] MC-LR | NRPS, PKS, mcyE, mcyB, sxtA |
| SV-77 | 0.256 | 2 | n.a. | n.a. | - |
| SV-80 H | 0.104 | 25 | 1 | [Asp3, Ser7] MC-RR [Asp3, Dha7] MC-RR [Asp3, ADMAdda5, Dhb7] MC-RR [Asp3, ADMAdda5, Thr7] MC-LR [Asp3, ADMAdda5, Dhb7] MC-LR | NRPS, PKS, mcyE, mcyB |
| SV-81 H,E | 11.309 | n.a. | 1 | [Asp3, ADMAdda5, Dhb7] MC-LR | NRPS, PKS, mcyE, mcyB |
| SV-83 H | 0.126 | 2 | 1 | [Asp3, ADMAdda5, Dhb7] MC-RR [Asp3, ADMAdda5, Dhb7] MC-LR Unidentified microcystin | -* |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kleinteich, J.; Puddick, J.; Wood, S.A.; Hildebrand, F.; Laughinghouse IV, H.D.; Pearce, D.A.; Dietrich, D.R.; Wilmotte, A. Toxic Cyanobacteria in Svalbard: Chemical Diversity of Microcystins Detected Using a Liquid Chromatography Mass Spectrometry Precursor Ion Screening Method. Toxins 2018, 10, 147. https://doi.org/10.3390/toxins10040147
Kleinteich J, Puddick J, Wood SA, Hildebrand F, Laughinghouse IV HD, Pearce DA, Dietrich DR, Wilmotte A. Toxic Cyanobacteria in Svalbard: Chemical Diversity of Microcystins Detected Using a Liquid Chromatography Mass Spectrometry Precursor Ion Screening Method. Toxins. 2018; 10(4):147. https://doi.org/10.3390/toxins10040147
Chicago/Turabian StyleKleinteich, Julia, Jonathan Puddick, Susanna A. Wood, Falk Hildebrand, H. Dail Laughinghouse IV, David A. Pearce, Daniel R. Dietrich, and Annick Wilmotte. 2018. "Toxic Cyanobacteria in Svalbard: Chemical Diversity of Microcystins Detected Using a Liquid Chromatography Mass Spectrometry Precursor Ion Screening Method" Toxins 10, no. 4: 147. https://doi.org/10.3390/toxins10040147
APA StyleKleinteich, J., Puddick, J., Wood, S. A., Hildebrand, F., Laughinghouse IV, H. D., Pearce, D. A., Dietrich, D. R., & Wilmotte, A. (2018). Toxic Cyanobacteria in Svalbard: Chemical Diversity of Microcystins Detected Using a Liquid Chromatography Mass Spectrometry Precursor Ion Screening Method. Toxins, 10(4), 147. https://doi.org/10.3390/toxins10040147







