Fatal Canine Intoxications Linked to the Presence of Saxitoxins in Stranded Marine Organisms Following Winter Storm Activity
Abstract
1. Introduction
2. Results
2.1. Canine Observations
2.2. Veterinary Results and Treatment
2.3. Canine Post Mortem Assessment
2.4. Toxin Analysis
2.4.1. LC-FLD
2.4.2. LC–MS/MS
2.4.3. Pathology Samples
3. Discussion
4. Materials and Methods
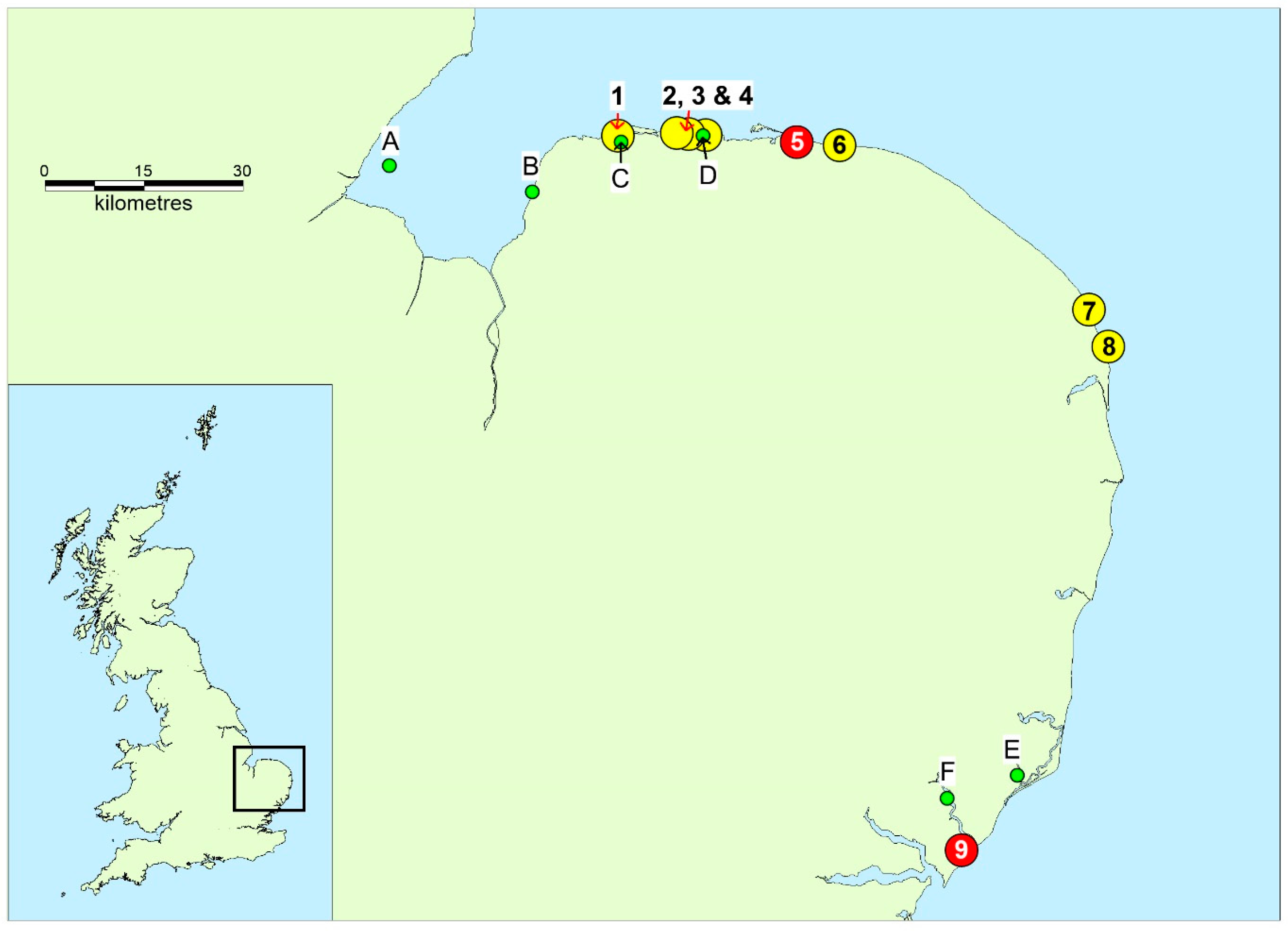
4.1. Samples
4.2. Reagents and Chemicals
4.3. Sample Extraction
4.4. Extract Clean-Up and Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hallegraef, G.M. Harmful Algal Blooms: A Global Overview. In Manual on Harmful Marine Microalgae; Hallegraef, G.M., Anderson, D.M., Cembella, A.D., Eds.; UNESCO: Paris, France, 2003; pp. 25–49. [Google Scholar]
- Llewellyn, L.E.; Negri, A.; Robertson, A. Paralytic shellfish toxins in tropical oceans. Toxin Rev. 2006, 25, 159–196. [Google Scholar] [CrossRef]
- Anon. Regulation (EC) No 854/2004 of the European Parliament and of the Council of 29th April 2004 laying down specific rules for the organisation of official controls on products of animal origin intended for human consumption. Off. J. Eur. Union 2004, L226, 83–127. [Google Scholar]
- Shumway, S.E. Phycotoxin-related shellfiish posoning: Bivalve molluscs are not the only vectors. Rev. Fish. Sci. 1995, 3, 1–31. [Google Scholar] [CrossRef]
- Rodriguez, P.; Alfonso, A.; Vale, C.; Alfonso, C.; Vale, P.; Tellez, A.; Botana, L.M. First toxicity report of tetrodotoxin and 5,6,11-trideoxyTTX in the trumpet shell Charonia lampas lampas in Europe. Anal. Chem. 2008, 80, 5622–5629. [Google Scholar] [CrossRef] [PubMed]
- Silva, M.; Barreiro, A.; Rodriguez, P.; Azevedo, J.; Alfonso, A.; Botana, L.M.; Vasconcelos, V. New intertebrate vectors for PST, spirolides and okadaic acid in the North Atlantic. Mar. Drugs 2013, 11, 1936–1960. [Google Scholar] [CrossRef] [PubMed]
- Costa, P.R.; Costa, S.T.; Braga, A.C.; Rodrigues, S.M.; Vale, P. Relevance and challenges in monitoring marine biotoxins in non-bivalve vectors. Food Control 2017, 76, 24–33. [Google Scholar] [CrossRef]
- Jensen, S.-K.; Lacaze, J.-P.; Hermann, G.; Kershaw, J.; Brownlow, A.; Turner, A.D.; Hall, A. Detection and effects of harmful algal toxins in Scottish harbour seals and potential links to population decline. Toxicon 2015, 97, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Asakawa, M.; Nishimura, F.; Miyazawa, K.; Noguchi, T. Occurrence of paralytic shellfish poison in the starfish Asterias amurensis in Kure Bay, Hiroshima prefecture, Japan. Toxicon 1997, 35, 1081–1087. [Google Scholar] [CrossRef]
- Lin, S.J.; Tsai, Y.H.; Lin, H.P.; Hwang, D.F. Paralytic toxins in Taiwanese starfish Astropecten scoparius. Toxicon 1998, 36, 799–803. [Google Scholar] [CrossRef]
- Ito, K.; Asakawa, M.; Sida, Y.; Miyazawa, K. Occurrence of paralytic shellfish poison (PSP) in the starfish Asterina pectinifera collected from the Kure Bay, Hiroshima Prefecture, Japan. Toxicon 2003, 41, 291–295. [Google Scholar] [CrossRef]
- Terrazas, J.O.; Contreras, H.R.; Carcia, C. Prevalence, variability and bioconcentration of saxitoxin-group in different marine species present in the food chain. Toxins 2017, 9, 190. [Google Scholar] [CrossRef] [PubMed]
- Met Office. Historical Weather Data for Lowestoft, Suffolk, England. 2018. Available online: https://www.metoffice.gov.uk/pub/data/weather/uk/climate/stationdata/lowestoftdata.txt (accessed on 12 January 2018).
- Eastern Daily Press. Reports of Dog Dying after Eating Contaminated Fish on Norfolk Beach Spark Warnings; Eastern Daily Press: Norwich, UK, 2018; Available online: http://www.edp24.co.uk/news/reports-dog-dying-eating-contaminated-fish-cley-beach-1-5338806 (accessed on 11 January 2018).
- Veterinary Poisons Information Service (VPIS) (London, UK). Personal communication, 2018.
- Alexander, J.; Benford, D.; Cockburn, A.; Cravedi, J.P.; Dogliotti, E.; Di Domenico, A. Scientific opinion of the panel on contaminants in the food chain on a request from the European Commission on marine biotoxins in shellfish—Saxitoxin group. EFSA J. 2009, 1019, 1–76. [Google Scholar]
- Turner, A.D.; Stubbs, B.; Coates, L.; Dhanji-Rapkova, M.; Hatfield, R.G.; Lewis, A.M.; Rowland-Pilgrim, S.; O’Neil, A.; Stubbs, P.; Ross, S.; et al. Variability of paralytic shellfish toxin occurrence and profiles in bivalve molluscs from Great Britain from official control monitoring as determined by pre-column oxidation liquid chromatography and implications for applying immunochemical tests. Harmful Algae 2014, 31, 87–99. [Google Scholar] [CrossRef] [PubMed]
- Deeds, J.R.; Landsberg, J.H.; Etheridge, S.M.; Pitcher, G.C.; Longan, S.W. Non-traditional vectors for paralytic shellfish poisoning. Mar. Drugs 2008, 6, 308–348. [Google Scholar] [CrossRef] [PubMed]
- Coulson, J.C.; Potts, G.R.; Deans, I.R.; Fraser, S.M. Mortality of shags and other sea birds caused by paralytic shellfish poison. Nature 1968, 220, 23–24. [Google Scholar] [CrossRef] [PubMed]
- Coulson, J.C.; Potts, G.R.; Deans, I.R.; Fraser, S.M. Exceptional mortality of shags and other seabirds caused by paralytic shellfish poison. Br. Birds 1968, 61, 381–404. [Google Scholar]
- Landsberg, J.H. The effects of harmful algal blooms on marine and aquatic organisms. Rev. Fish. Sci. 2002, 10, 113–390. [Google Scholar] [CrossRef]
- Noguchi, T.; Daigo, K.; Arakawa, O.; Hashimoto, K. Release of paralytic shellfish poison from the exoskeleton of a xanthid crab Zosimus aeneus. In Toxic Dinoflagellates; Anderson, D.M., White, A.W., Baden, D.G., Eds.; Elsevier: New York, NY, USA, 1985; pp. 495–500. [Google Scholar]
- Asakawa, M.; Toyoshima, T.; Shida, Y.; Noguchi, T.; Miyazawa, K. Paralytic toxins in a ribbon work Cephalothrix species (Nemertean) adherent to cultured oysters in Hiroshima Bay, Hiroshima Prefecture, Japan. Toixon 2000, 38, 763–773. [Google Scholar]
- Asakawa, M.; Itol, K.; Kajhara, H. Highly toxic Ribbon Worm Cephalothrix simula containing tetrodotoxin in Hiroshima Bay, Hiroshima Prefecture, Japan. Toxins 2013, 5, 376–395. [Google Scholar] [CrossRef] [PubMed]
- Kajihara, H.; Sun, S.-C.; Chernyshev, A.V.; Chen, H.-X.; Ito, K.; Asakawa, M.; Maslakova, S.A.; Norenburg, J.L.; Strand, M.; Sundberg, P.; et al. Taxonomic identity of a tetrodotoxin-accumulating ribbon worm Cephalothrix simula (Nemertea: Palaeonemertea): A species artificially introduced from the Pacific to Europe. Zool. Sci. 2013, 30, 985–997. [Google Scholar] [CrossRef] [PubMed]
- Ueda, H.; Itoli, S.; Sugita, H. TTX-bearing planocerid flatworm (Platyhelminthes: Acotylea) in the Ryukyu Islands, Japan. Mar. Drugs 2018, 16, 37. [Google Scholar] [CrossRef] [PubMed]
- Braid, H.E.; Deeds, J.; DeGrasse, S.L.; Wilson, J.J.; Osborne, J.; Hanner, R.H. Preying on commercial fisheries and accumulating paralytic shellfish toxins: A dietary analysis of invasive Dosidicus gigas (Cephalopoda Ommastrephidae) stranded in Pacific Canada. Mar. Biol. 2012, 159, 25–31. [Google Scholar] [CrossRef]
- Robertson, A.; Stirling, D.; Robillot, C.; Llewellyn, L.; Negri, A. First report of saxitoxin in octopi. Toxicon 2004, 44, 765–771. [Google Scholar] [CrossRef] [PubMed]
- Beitler, M.K.; Liston, J. Uptake and tissue distribution of PSP toxin in butter clams. In Toxic Marine Phytoplankton; Graneli, E., Sundstrom, B., Edler, L., Anderson, D.M., Eds.; Elsevier Science Publications: New York, NY, USA, 1990; pp. 257–263. [Google Scholar]
- Hall, S. Toxins and Toxicity of Protogonyaulax from the Northeast Pacific. Ph.D. Thesis, University of Alaska Fairbanks, Fairbanks, AK, USA, 1982. [Google Scholar]
- Oshima, Y.; Bolch, C.J.; Hallegraeff, G.M. Toxin composition of resting cysts of Alexandrium tamarense (Dinophyceae). Toxicon 1992, 30, 1539–1544. [Google Scholar] [CrossRef]
- Kwong, R.W.M.; Wang, W.-X.; Lam, P.K.S.; Yu, P.K.N. The uptake distribution and elimination of paralytic shellfish toxins in mussels and fish exposed to toxic dinoflagellates. Aquat. Toxicol. 2006, 80, 82–91. [Google Scholar] [CrossRef] [PubMed]
- Sephton, D.H.; Haya, K.; Martin, J.L.; LeGresley, M.M.; Page, F.H. Paralytic shellfish toxins in zooplankton, Salmo salar, during a bloom of Alexandrium fundyense off Grand Mana Island, in the Bay of Fundy. Harmful Algae 2007, 6, 745–758. [Google Scholar] [CrossRef]
- Jester, R.J.; Baugh, K.A.; Lefebvre, K.A. Presence of Alexandrium catenealla and paralytic shellfish toxins in finfish, shellfish and rock crabs in Monterey Bay, Californica, USA. Mar. Biol. 2009, 156, 493–504. [Google Scholar] [CrossRef]
- Jester, R.; Rhodes, L.; Beuzenberg, V. Uptake of paralytic shellfish poisoning and spirolides toxins by paddle crab (Ovalipes catharus) via a bivalve vector. Harmful Algae 2009, 8, 369–376. [Google Scholar] [CrossRef]
- Llewellyn, L.E. Haemolymph protein in xanthid crabs: Its selective binding of saxitoxin and possible role in toxin bioaccumulation. Mar. Biol. 1997, 128, 599–606. [Google Scholar] [CrossRef]
- Oikawa, H.; Fujita, T.; Saito, K.; Watabe, S.; Satomi, M.; Yano, Y. Comparison of paralytic shellfish poisoning toxin between carnivorous crabs (Telmessus acutidens and Charybdis japonica) and their prey mussel (Mytilus galloprovincialis) in an inshore food chain. Toxicon 2004, 43, 713–719. [Google Scholar] [CrossRef] [PubMed]
- Negri, A.P.; Llewellyn, L. Comparative analyses by HPLC and the sodium channel and saxiphilin 3H-saxitoxin receptor assays for paralytic shellfish toxins in crustaceans and molluscs from tropical north west Australia. Toxicon 1998, 36, 283–298. [Google Scholar] [CrossRef]
- McCollum, J.P.K.; Pearson, R.C.M.; Ingham, H.R.; Wood, P.C.; Dewar, H.A. An epidemic of mussel poisoning in north-east England. Lancet 1968, 292, 767–770. [Google Scholar] [CrossRef]
- Ayres, P.A.; Cullum, M. Paralytic Shellfish Poisoning. An Account of Investigations into Mussel Toxicity in England 1968–77; Fisheries Research Technical Report Number 40; MAFF Directorate Fisheries Research: Lowestoft, UK, 1978; Volume 40, pp. 1–23. Available online: https://www.cefas.co.uk/publications/techrep/tech40.pdf (accessed on 19 January 2018).
- Alderman, D.J. Monitoring and Surveillance of Biological Contaminants and Disease in the Aquatic Environment 1990; Aquatic Environment Monitoring Report; MAFF Directorate Fisheries Research: Lowestoft, UK, 1993; Volume 35, pp. 1–35. [Google Scholar]
- Haya, K.; Martin, J.L.; Waiwood, B.A.; Burridge, L.E.; Hungerford, J.M.; Zitko, V. Identification of paralytic shellfish toxins in mackerel from southwest Bay of Fundy, Canada. In Toxic Marine Phytoplankton; Graneli, E., Sundstrom, B., Edler, L., Anderson, D.M., Eds.; Academic Press: New York, NY, USA, 1990; pp. 350–355. [Google Scholar]
- Oikawa, H.; Fujita, T.; Satomi, M.; Suzuki, T.; Kotani, Y.; Yano, Y. Accumulation of paralytic shellfish poisoning toxins in the edibile shore crab Telmessus acutidens. Toxion 2002, 40, 1593–1599. [Google Scholar]
- Garcia, C.; Bravo, M.C.; Lagos, M.; Lagos, N. Paralytic shellfish poisoning: Post-mortem analysis of tissue and body fluid samples from human victims in the Patagonia fjords. Toxicon 2004, 43, 149–158. [Google Scholar] [CrossRef] [PubMed]
- Garcia, C.; Barriga, A.; Diaz, J.C.; Lagos, M.; Lagos, N. Route of metabolisation and detoxification of paralytic shellfish toxins in humans. Toxicon 2010, 55, 135–144. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, A.; Turner, A.D. Performance characteristics of AOAC method 2005.06 for the determination of paralytic shellfish toxins in Manilla clams, European Otter Clams, Grooved Carpet Shell Clams, Surf Clams and Processed King Scallops. J. AOAC Int. 2015, 98, 628–635. [Google Scholar] [CrossRef] [PubMed]
- Tidholm, A.; Jönsson, L. Histologic characterisation of canine dilated cardiomyopathy. Vet. Pathol. 2005, 42, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Mail, Graveyard on the Shore for Thousands of Starfish as Storm Throws Them from the Sea. 2009. Available online: http://www.dailymail.co.uk/news/article-1234177/Thousands-starfish-carpet-Norfolk-beach-storm-throws-sea.html (accessed on 19 January 2018).
- Mercury. Hundreds of Starfish Wash Up on Gorleston Beach. 2016. Available online: http://www.greatyarmouthmercury.co.uk/news/hundreds-of-starfish-wash-up-on-gorleston-beach-1-4452527 (accessed on 19 January 2018).
- Geraci, J.R.; Anderson, D.M.; Timperi, R.J.; St. Aubin, D.J.; Early, G.A.; Prescott, J.H.; Mayo, C.A. Humpback Whales (Megaptera novaeangliae) fatally poisoned by dinoflagellate toxin. Can. J. Fish. Aquat. Sci. 1989, 46, 1895–1898. [Google Scholar] [CrossRef]
- Hernandez, M.; Robinson, I.; Aguilar, A.; Gonzalez, L.M.; Lopez-Jurado, L.F.; Reyero, M.I.; Cacho, E.; Franco, J.; Eduardo-Costas, V.L.-R. Did algal toxins cause monk seal mortality? Nature 1998, 393, 28–29. [Google Scholar] [CrossRef] [PubMed]
- Trainer, V.L.; Baden, D.G. High affinity binding of red tide neurotoxins to marine mammal brain. Aquat. Toxicol. 1999, 46, 139–148. [Google Scholar] [CrossRef]
- Reyero, M.; Cacho, E.; Martinez, A.; Vazquez, J.; Marina, A.; Fraga, S.; Franco, J.M. Evidence of saxitoxin derivatives as causative agents in the 1997 mass mortality of Monk seals in the Cape Blanc Peninsula. Nat. Toxins 1999, 7, 311–315. [Google Scholar] [CrossRef]
- Fire, S.E.; Wang, Z.; Byrd, M.; Whitehead, H.R.; Paternoster, J.; Morton, S.L. Co-occurrence of multiple classes of harmful algal toxins in bottlenose dolphins (Tursiops truncatus) stranding during an unusual mortality event in Texas, USA. Harmful Algae 2011, 10, 330–336. [Google Scholar] [CrossRef]
- Zeidberg, L.D.; Robison, B.H. Invasive range expansion by the Humbolt squid, Dosidicus gigas in the eastern North Pacific. Proc. Natl. Acad. Sci. USA 2007, 104, 12948–12950. [Google Scholar] [CrossRef] [PubMed]
- Desbiens, M.; Cembella, A.D. Occurrence and elimination kinetics of PSP toxins in the American lobster (Homarus americanus). In Harmful Marine Algal Blooms; Lassus, P., Arzul, G., Denn, E.E., Gentien, P., Baut, C.M., Eds.; Lavoisier Publishing: Paris, France, 1995; pp. 433–438. [Google Scholar]
- White, A.W. Dinoflagellate toxins as a probable cause of an Atlantic herring (Clupea harengus harengus) kill and pteropods as apparent vector. J. Fish. Res. Bd. Can. 1977, 34, 2421–2424. [Google Scholar] [CrossRef]
- Mills, L.J.; Klein-MacPhee, G.K. Toxicity of the New England red tide dinoflagellate to winter flounder larvae. In Toxic Dinoflagellates; Anderson, D.M., White, A., Baden, D.G., Eds.; Elsevier: New York, NY, USA, 1985; pp. 389–394. [Google Scholar]
- Huntley, M.E.; Sykes, P.; Rohan, S.; Marin, V. Chemically mediated rejection of dinoflagellate prey by the copepods Calanus pacificus and Paracalanus parvus: Mechanism, occurrence and significance. Mar. Ecol. Prog. Ser. 1986, 28, 105–120. [Google Scholar] [CrossRef]
- Gosselin, S.; Fortier, L.; Gagne, J.A. Vulnerability of marine fish larvae to the toxic dinoflagellate Protogonyaulax tamarensis. Mar. Ecol. Prog. Ser. 1989, 57, 1–10. [Google Scholar] [CrossRef]
- Robineau, B.; Fortier, L.; Gagne, J.A.; Cembella, A.D. Comparison of the response of five larval fish species to the toxic dinoflagellate Alexandrium excavatum (Braarud) Balech. J. Exp. Mar. Biol. Ecol. 1991, 152, 225–242. [Google Scholar] [CrossRef]
- Robineau, B.; Gagne, J.A.; Fortier, L.; Cembella, A.D. Potential impact of a toxic dinoflagellate (Alexandrium excavatum) bloom on survival of fish and crustacean larvae. Mar. Biol. 1991, 108, 293–301. [Google Scholar] [CrossRef]
- Montoya, R.; Akselman, J.; Franco, J.I.; Carreto, J. Paralytic shellfish toxins and mackerel (Scomber japonicus) mortality in the Argentine Sea. In Harmful and Toxic Algal Blooms; Yasumoto, T., Oshima, Y., Fukuyo, Y., Eds.; IOC of UNESCO: Paris, France, 1996; pp. 417–420. [Google Scholar]
- White, A.W. Recurrence of kills of Atlantic herring (Clupea harengus harengus) caused by dinoflagellate toxins transferred through herbivorous zooplankton. Can. J. Fish. Aquat. Sci. 1980, 37, 2262–2265. [Google Scholar] [CrossRef]
- Gonzalez, C.L.; Ordonez, J.A.; Maala, A.M. Red tide: The Philippine experience. In Red Tides, Biology, Environmental Science and Toxicology; Okaichi, T., Anderson, D.M., Nemoto, T., Eds.; Elsevier: New York, NY, USA, 1989; pp. 97–100. [Google Scholar]
- White, A.W. Paralytic shellfish toxins and finfish. In Seafood Toxins; Ragelis, E.P., Ed.; American Chemical Society Symposium Series: Washington, DC, USA, 1984; pp. 257–269. [Google Scholar]
- Lawrence, J.F.; Maher, M.; Watson-Wright, W. Effect of cooking on the concentration of toxins associated with paralytic shellfish poison in lobster hepatopancreas. Toxicon 1994, 32, 57–64. [Google Scholar] [CrossRef]
- Jonas-Davies, J.; Liston, J. The occurrence of PSP toxins in intertidal organisms. In Toxic Dinoflagellates; Anderson, D.M., White, A., Baden, D.G., Eds.; Elsevier: New York, NY, USA, 1985; pp. 467–472. [Google Scholar]
- Turner, A.D.; McNabb, P.S.; Harwood, D.T.; Selwood, A.I.; Boundy, M.J. Single laboratory validation of a multitoxin LC-hydrophilic interaction LC-MS/MS method for quantitation of Paralytic Shellfish Toxins in bivalve shellfish. J. AOAC Int. 2015, 98, 609–621. [Google Scholar] [CrossRef] [PubMed]
- Quilliam, M.A.; Xie, M.; Hardstaff, W.R. A rapid extraction and cleanup procedure for the liquid chromatographic determination of domoic acid in unsalted seafood. J. AOAC Int. 1995, 78, 543–554. [Google Scholar]
- Turner, A.D.; Algoet, M.; Hatfield, R.; Norton, D.; Morris, S.; Reese, A.; Lees, D.N. Single laboratory validation of the AOAC HPLC method (2005.06) for mussels: Refinement and extension of the method to additional toxins. J. AOAC 2009, 92, 190–207. [Google Scholar]
- Boundy, M.J.; Selwood, A.I.; Harwood, D.T.; McNabb, P.S.; Turner, A.D. Development of a sensitive and selective liquid chromatography-mass spectrometry method for high throughput analysis of paralytic shellfish toxins using graphitic carbon SPE. J. Chrom. A 2015, 1387, 1–12. [Google Scholar] [CrossRef] [PubMed]
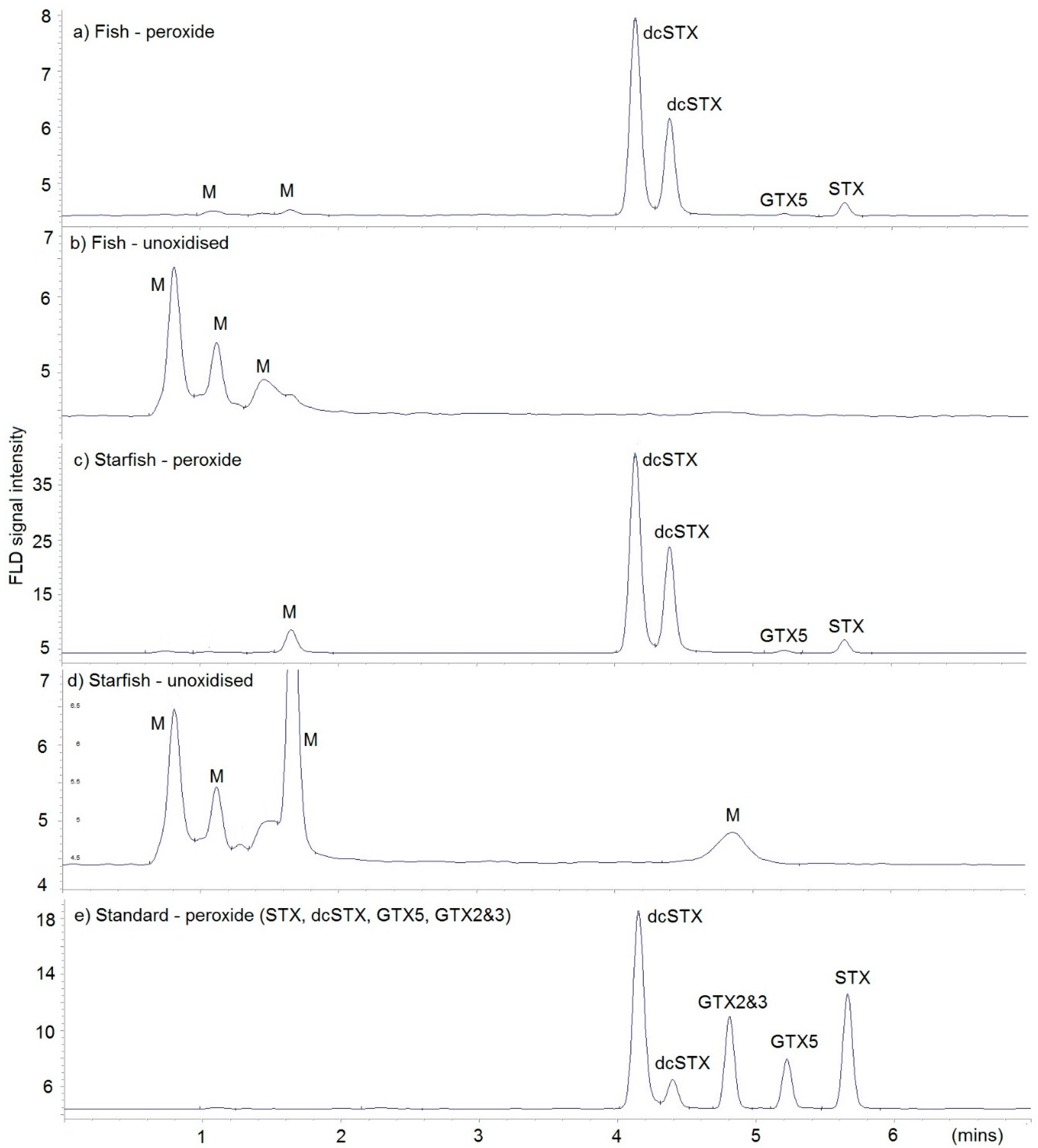
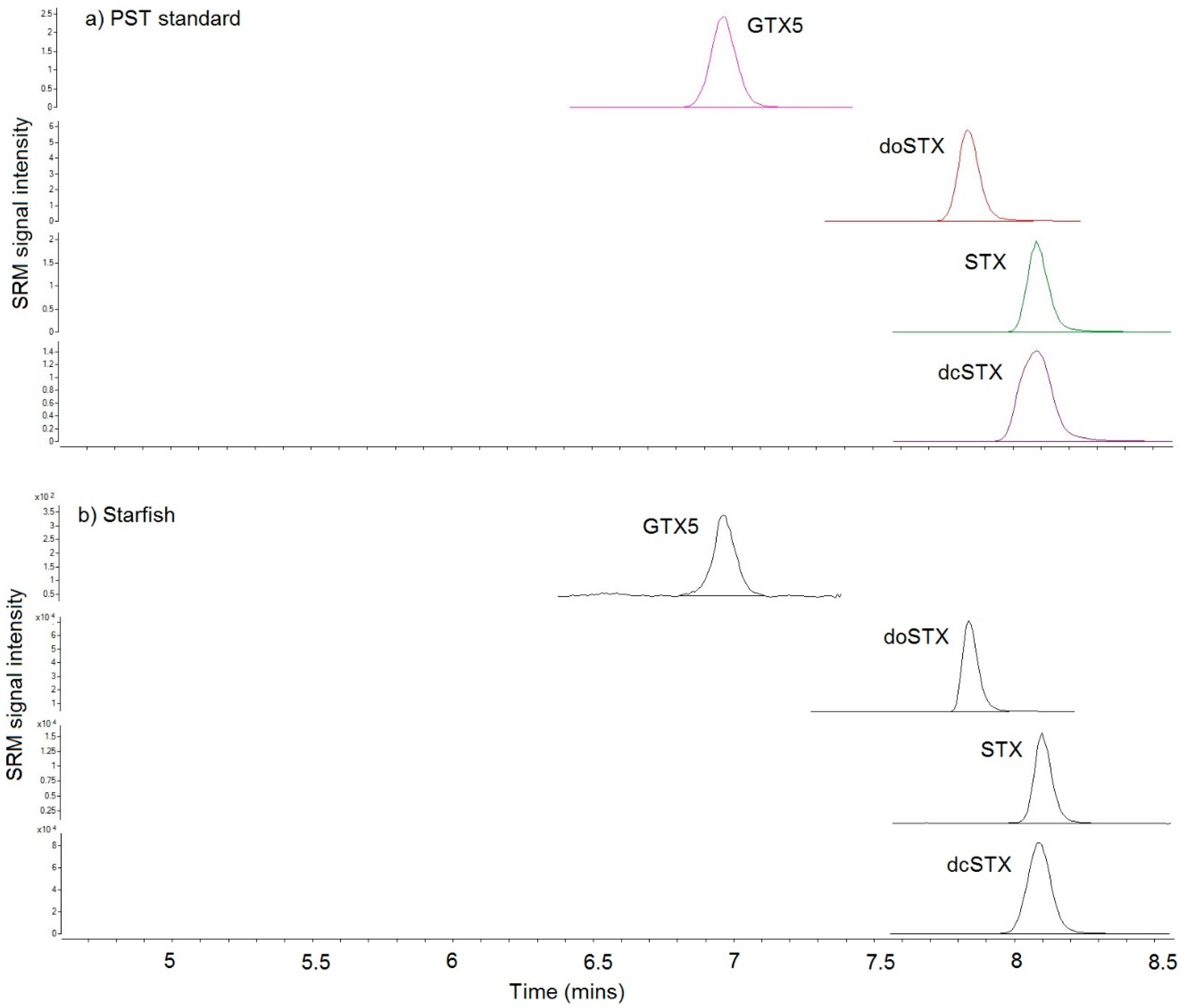
| LC-MS/MS | LC-FLD | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Toxin | Starfish 1 | Dab 1 | Dab 2 | Starfish 2 | Crab | Starfish 1 | Dab 1 | Dab 2 | Starfish 2 | Crab |
| dcSTX | 913 | 115 | 320 | 13,730 | 2363 | 1361 | 132 | 506 | 21,680 | 3175 |
| doSTX | 37 | 1.2 | 3.7 | 34 | 13 | na | na | na | na | na |
| GTX5 | 7 | 1.6 | 4.5 | 96 | 8.6 | 6.2 | nd | nd | nd | nd |
| STX | 126 | 46 | 70 | 579 | 153 | 149 | 16 | 60 | 759 | 154 |
| Total | 1083 | 164 | 398 | 14,439 | 2537 | 1516 | 148 | 566 | 22,440 | 3329 |
| Sample | GTX5 | doSTX | dcSTX | STX | Total |
|---|---|---|---|---|---|
| Small intestine contents—dog 1 | 3.6 | 1.3 | 107.3 | 51.3 | 164 |
| Spleen—dog 1 | nd | nd | 33.0 | nd | 33 |
| Liver—dog 1 | nd | nd | 15.1 | nd | 15 |
| Stomach contents—dog 1 | 1.6 | 0.4 | 50.0 | 37.3 | 89 |
| Kidney—dog 1 | nd | nd | 15.5 | nd | 16 |
| Brain (formalin fixed tissue)—dog 1 | nd | nd | nd | nd | nd |
| Blood—dog 2 | nd | nd | nd | nd | nd |
| Sample | Matrix | Date Collected | Location |
|---|---|---|---|
| Starfish 1 | Starfish (A. rubens) | 2/1/18 | Cley Beach † |
| Dab 1 | Fish (L. limanda) | 2/1/18 | Cley Beach |
| Dab 2 | Fish (L. limanda) | 2/1/18 | Cley Beach |
| Post mortem samples—dog 1 | Organs (various) and gastrointestinal contents | 2/1/18 | Cley Beach |
| Blood—dog 2 | Blood | 1/1/18 | Cley Beach |
| Starfish 2 | Starfish (C. papposus) | 15/1/18 | Felixstowe Ferry Beach ‡ |
| Crab/vomit—dog 3 | Crab—partially digested | 13/1/18 | Felixstowe Ferry Beach |
| Shore crabs | Crab (C. maenas) | 17/1/18 | Felixstowe Ferry Beach |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Turner, A.D.; Dhanji-Rapkova, M.; Dean, K.; Milligan, S.; Hamilton, M.; Thomas, J.; Poole, C.; Haycock, J.; Spelman-Marriott, J.; Watson, A.; et al. Fatal Canine Intoxications Linked to the Presence of Saxitoxins in Stranded Marine Organisms Following Winter Storm Activity. Toxins 2018, 10, 94. https://doi.org/10.3390/toxins10030094
Turner AD, Dhanji-Rapkova M, Dean K, Milligan S, Hamilton M, Thomas J, Poole C, Haycock J, Spelman-Marriott J, Watson A, et al. Fatal Canine Intoxications Linked to the Presence of Saxitoxins in Stranded Marine Organisms Following Winter Storm Activity. Toxins. 2018; 10(3):94. https://doi.org/10.3390/toxins10030094
Chicago/Turabian StyleTurner, Andrew D., Monika Dhanji-Rapkova, Karl Dean, Steven Milligan, Mike Hamilton, Julie Thomas, Chris Poole, Jo Haycock, Jo Spelman-Marriott, Alice Watson, and et al. 2018. "Fatal Canine Intoxications Linked to the Presence of Saxitoxins in Stranded Marine Organisms Following Winter Storm Activity" Toxins 10, no. 3: 94. https://doi.org/10.3390/toxins10030094
APA StyleTurner, A. D., Dhanji-Rapkova, M., Dean, K., Milligan, S., Hamilton, M., Thomas, J., Poole, C., Haycock, J., Spelman-Marriott, J., Watson, A., Hughes, K., Marr, B., Dixon, A., & Coates, L. (2018). Fatal Canine Intoxications Linked to the Presence of Saxitoxins in Stranded Marine Organisms Following Winter Storm Activity. Toxins, 10(3), 94. https://doi.org/10.3390/toxins10030094







