Impact of Antibiotics on Efficacy of Cry Toxins Produced in Two Different Genetically Modified Bt Maize Varieties in Two Lepidopteran Herbivore Species, Ostrinia nubilalis and Spodoptera littoralis
Abstract
1. Introduction
2. Results
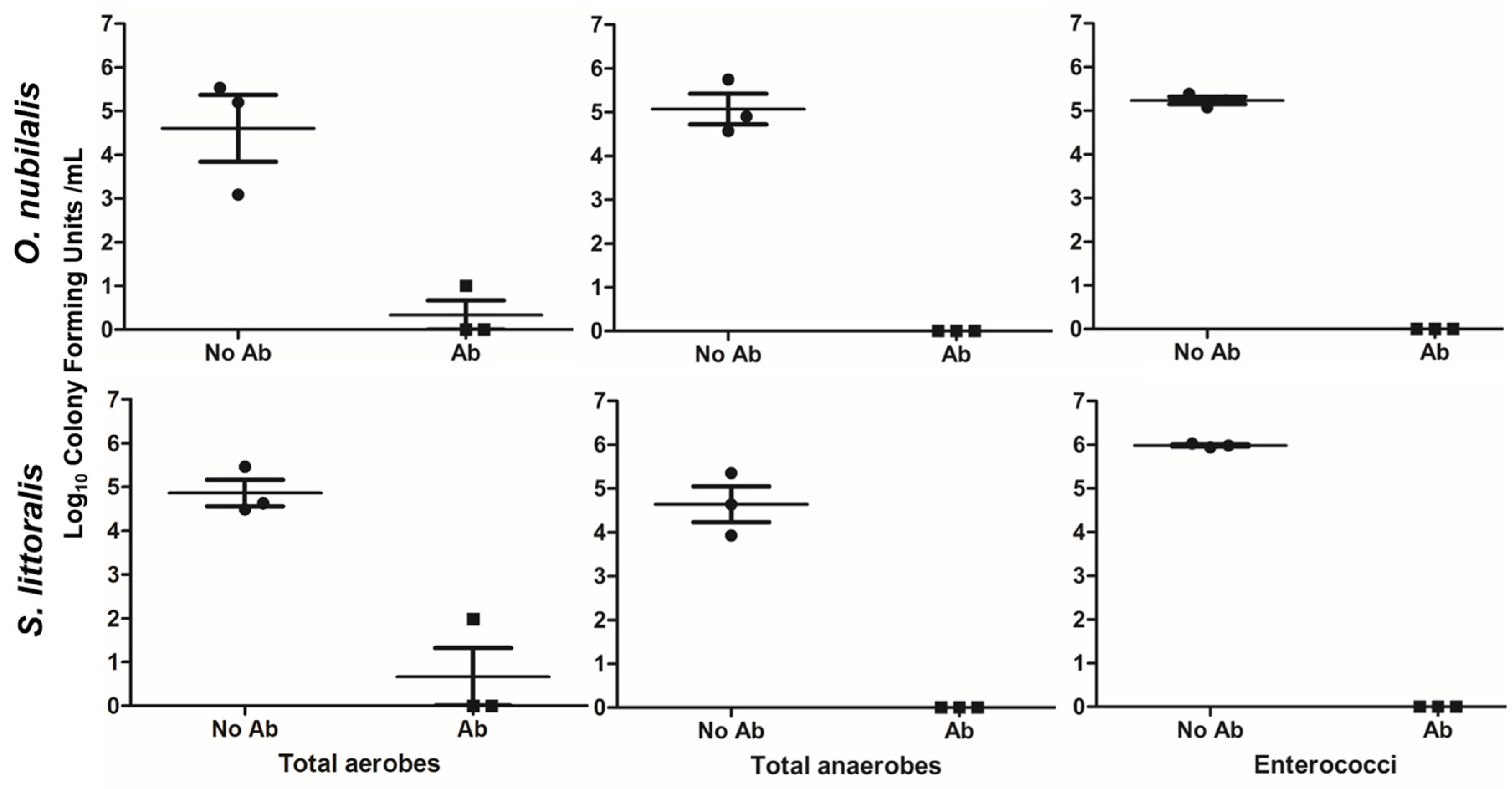
2.1. Effects of Antibiotics on Midgut Bacterial Strains
2.2. Impacts of GM Bt Maize with or without Antibiotic Treatments
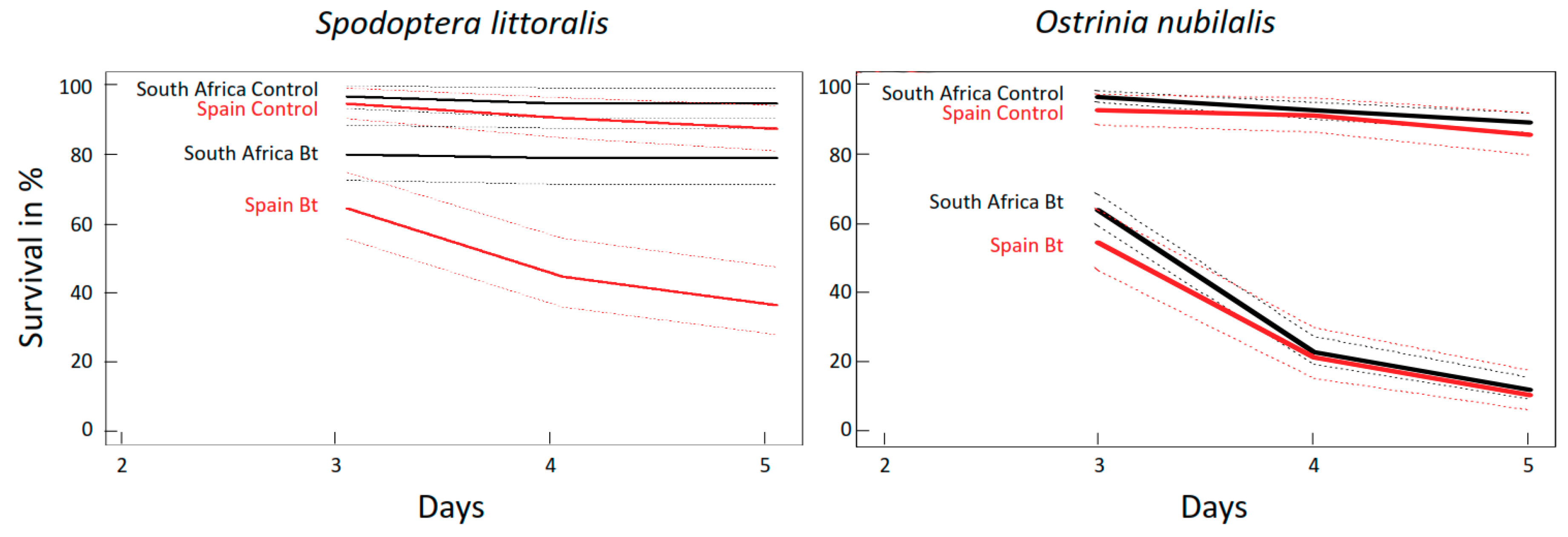
2.2.1. Mortality of O. nubilalis and S. littoralis Larvae Fed GM Bt Maize Varieties without Antibiotics
2.2.2. Effects of Antibiotics (AB) on Survival of O. nubilalis and S. littoralis Larvae Fed GM Bt Maize Varieties
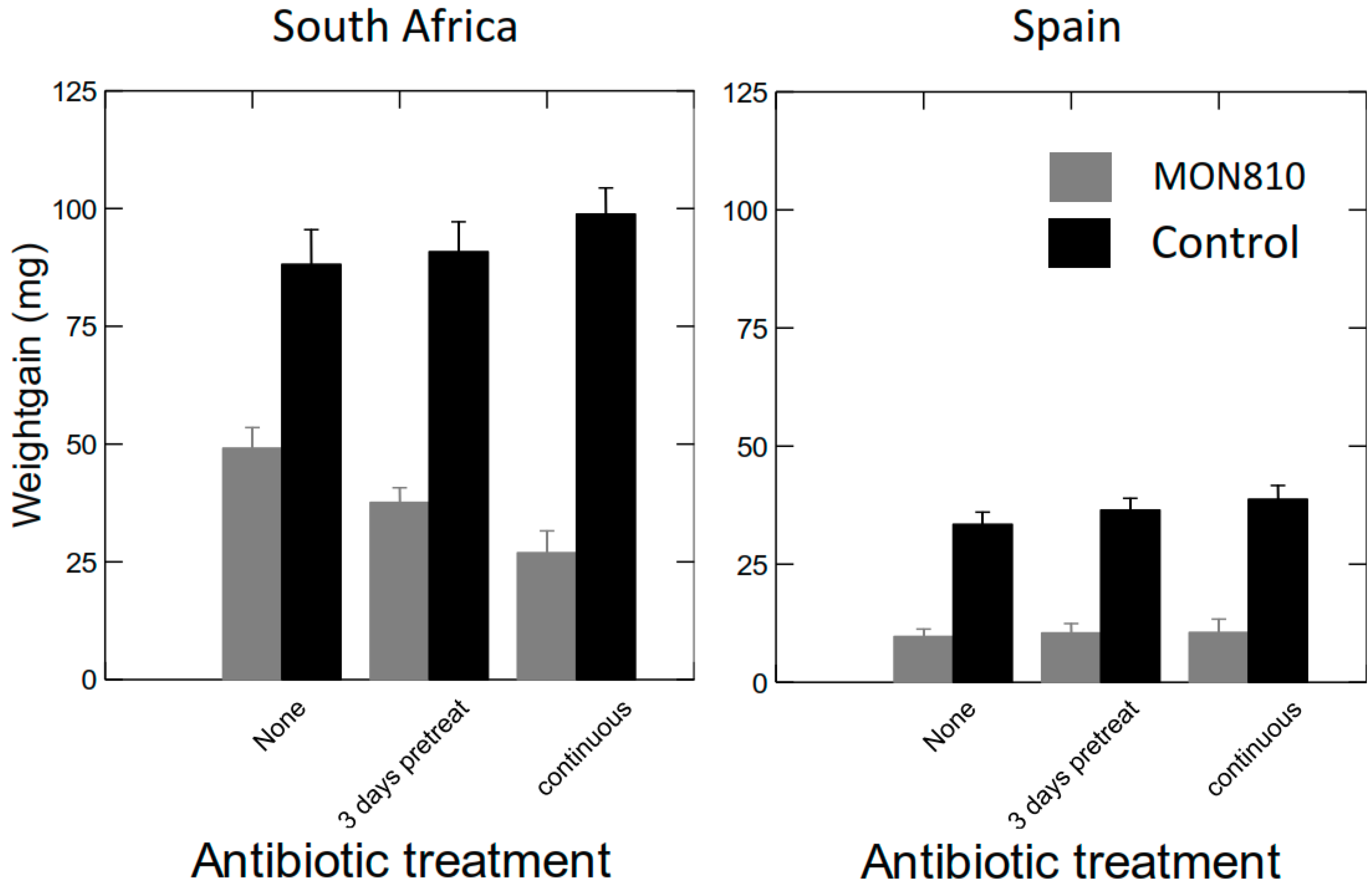
2.2.3. Effects of Antibiotics (AB) on Weight Gain of S. littoralis Larvae Fed GM Bt Maize Varieties
2.3. Effects of Antibiotics on Survival of O. nubilalis and S. littoralis Larvae Fed Bt-Sprayed Non-GM Maize
3. Discussion
3.1. Contextualizing with Previous Studies
3.2. Impact of Antibiotics on Efficacy of Cry1Ab Toxin from Bt Maize
3.3. Implications
3.3.1. For Efficacy of Bt Crops in Target Pests—Further Research Questions
3.3.2. For Ecotoxicity Testing of Bt Plants and Bt Insecticides
4. Conclusions
5. Materials and Methods
5.1. Insects
5.2. Experimental Plants
5.3. Midgut Bacterial Strains and Culture Conditions
5.4. Bioassays
5.5. Statistics
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sanchis, V. From microbial sprays to insect-resistant transgenic plants: History of the biospesticide Bacillus thuringiensis: A review. Agron. Sustain. Dev. 2011, 31, 217–231. [Google Scholar] [CrossRef]
- Schnepf, E.; Crickmore, N.; Van Rie, J.; Lereclus, D.; Baum, J.; Feitelson, J.; Zeigler, D.R.; Dean, D.H. Bacillus thuringiensis and its pesticidal crystal proteins. Microbiol. Mol. Biol. Rev. 1998, 62, 775–806. [Google Scholar] [PubMed]
- Sansinenea, E. Bacillus thuringiensis Biotechnology; Sansinenea, E., Ed.; Springer: Dordrecht, The Netherlands, 2012. [Google Scholar]
- James, C. Global Status of Commercialized Biotech/GM Crops: 2015; ISAAA Brief No. 51; ISAAA: Manila, Philippines, 2015. [Google Scholar]
- Hilbeck, A.; Otto, M. Specificity and combinatorial effects of Bacillus thuringiensis Cry toxins in the context of GMO environmental risk assessment. Front. Environ. Sci. 2015, 3. [Google Scholar] [CrossRef]
- Vachon, V.; Laprade, R.; Schwartz, J.L. Current models of the mode of action of Bacillus thuringiensis insecticidal crystal proteins: A critical review. J. Invertebr. Pathol. 2012, 111, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Latham, J.R.; Love, M.; Hilbeck, A. The distinct properties of natural and GM Cry insecticidal proteins. Biotechnol. Genet. Eng. Rev. 2017, 33, 62–96. [Google Scholar] [CrossRef] [PubMed]
- Whalon, M.E.; Wingerd, B.A. Bt: Mode of action and use. Arch. Insect Biochem. Physiol. 2003, 54, 200–211. [Google Scholar] [CrossRef] [PubMed]
- Smouse, D.; Nishiura, J. A Bacillus thuringiensis delta-endotoxin induces programmed cell death in mosquito larvae. Cell Death Differ. 1997, 4, 560–569. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Candas, M.; Griko, N.B.; Rose-Young, L.; Bulla, L.A., Jr. Cytotoxicity of Bacillus thuringiensis Cry1Ab toxin depends on specific binding of the toxin to the cadherin receptor BT-R1 expressed in insect cells. Cell Death Differ. 2005, 12, 1407–1416. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Candas, M.; Griko, N.B.; Taussig, R.; Bulla, L.A., Jr. A mechanism of cell death involving an adenylyl cyclase/PKA signaling pathway is induced by the Cry1Ab toxin of Bacillus thuringiensis. Proc. Natl. Acad. Sci. USA 2006, 103, 9897–9902. [Google Scholar] [CrossRef] [PubMed]
- Yao, J.; Buschman, L.L.; Lu, N.; Khajuria, C.; Zhu, K.Y. Changes in gene expression in the larval gut of Ostrinia nubilalis in response to Bacillus thuringiensis Cry1Ab protoxin ingestion. Toxins 2014, 6, 1274–1294. [Google Scholar] [CrossRef] [PubMed]
- Soberon, M.; Gill, S.S.; Bravo, A. Signaling versus punching hole: How do Bacillus thuringiensis toxins kill insect midgut cells? Cell. Mol. Life Sci. CMLS 2009, 66, 1337–1349. [Google Scholar] [CrossRef] [PubMed]
- Jurat-Fuentes, J.L.; Adang, M.J. Cry toxin mode of action in susceptible and resistant Heliothis virescens larvae. J. Invertebr. Pathol. 2006, 92, 166–171. [Google Scholar] [CrossRef] [PubMed]
- Pigott, C.R.; Ellar, D.J. Role of receptors in Bacillus thuringiensis crystal toxin activity. Microbiol. Mol. Biol. Rev. 2007, 71, 255–281. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Kumari, R. Occurrence of molecularly diverse Bt Crytoxin-resistant mutations in insect pests of Bt+ corn and cotton crops and remedial approaches. Curr. Sci. 2015, 108, 1–8. [Google Scholar]
- Adang, M.J.; Crickmore, N.; Jurat-Fuentes, J.L. Diversity of Bacillus thuringiensis crystal toxins and mechanism of action. Adv. Insect Physiol. 2014, 47, 39–87. [Google Scholar]
- Graf, J. Shifting paradigm on Bacillus thuringiensis toxin and a natural model for Enterococcus faecalis septicemia. mBio 2011, 2. [Google Scholar] [CrossRef] [PubMed]
- Broderick, N.A.; Raffa, K.F.; Handelsman, J. Midgut bacteria required for Bacillus thuringiensis insecticidal activity. Proc. Natl. Acad. Sci. USA 2006, 103, 15196–15199. [Google Scholar] [CrossRef] [PubMed]
- Broderick, N.A.; Robinson, C.J.; McMahon, M.D.; Holt, J.; Handelsman, J.; Raffa, K.F. Contributions of gut bacteria to Bacillus thuringiensis-induced mortality vary across a range of lepidoptera. BMC Biol. 2009, 7, 11. [Google Scholar] [CrossRef] [PubMed]
- Mason, K.L.; Stepien, T.A.; Blum, J.E.; Holt, J.F.; Labbe, N.H.; Rush, J.S.; Raffa, K.F.; Handelsman, J. From commensal to pathogen: Translocation of Enterococcus faecalis from the midgut to the hemocoel of Manduca sexta. mBio 2011, 2, e00065-00011. [Google Scholar] [CrossRef] [PubMed]
- Raymond, B.; Johnston, P.R.; Wright, D.J.; Ellis, R.J.; Crickmore, N.; Bonsall, M.B. A mid-gut microbiota is not required for the pathogenicity of Bacillus thuringiensis to diamondback moth larvae. Environ. Microbiol. 2009, 11, 2556–2563. [Google Scholar] [CrossRef] [PubMed]
- Raymond, B.; Johnston, P.R.; Nielsen-LeRoux, C.; Lereclus, D.; Crickmore, N. Bacillus thuringiensis: An impotent pathogen? Trends Microbiol. 2010, 18, 189–194. [Google Scholar] [CrossRef]
- Johnston, P.R.; Crickmore, N. Gut bacteria are not required for the insecticidal activity of Bacillus thuringiensis toward the tobacco hornworm, Manduca sexta. Appl. Environ. Microbiol. 2009, 75, 5094–5099. [Google Scholar] [CrossRef] [PubMed]
- Caccia, S.; Di Lelio, I.; La Storia, A.; Marinelli, A.; Varricchio, P.; Franzetti, E.; Banyuls, N.; Tettamanti, G.; Casartelli, M.; Giordana, B.; et al. Midgut microbiota and host immunocompetence underlie Bacillus thuringiensis killing mechanism. Proc. Natl. Acad. Sci. USA 2016, 113, 9486–9491. [Google Scholar] [CrossRef] [PubMed]
- Hilbeck, A.; Meier, M.; Trtikova, M. Underlying reasons of the controversy over adverse effects of Bt toxins on lady beetle and lacewing larvae. Environ. Sci. Eur. 2012, 24, 9. [Google Scholar] [CrossRef]
- Cohen, A.C.; Smith, L.K. A new concept in artificial diets for Chrysoperla rufilabris: The efficacy of solid diets. Biol. Control 1998, 13, 49–54. [Google Scholar] [CrossRef]
- Li, Y.; Hu, L.; Romeis, J.; Wang, Y.; Han, L.; Chen, X.; Peng, Y. Use of an artificial diet system to study the toxicity of gut-active insecticidal compounds on larvae of the green lacewing Chrysoperla sinica. Biol. Control 2014, 69, 45–51. [Google Scholar] [CrossRef]
- Ali, I.; Zhang, S.; Cui, J.J. Bio-safety evaluation of Cry1Ac, Cry2Ab, Cry1Ca, Cry1F and Vip3Aa on Harmonia axyridis larvae. J. Appl. Entomol. 2016, 141, 53–60. [Google Scholar] [CrossRef]
- Ali, I.; Zhang, S.; Luo, J.-Y.; Wang, C.-Y.; Lv, L.-M.; Cui, J.-J. Artificial diet development and its effect on the reproductive performances of Propylea japonica and Harmonia axyridis. J. Asia-Pac. Entomol. 2016, 19, 289–293. [Google Scholar] [CrossRef]
- Trtikova, M.; Wikmark, O.G.; Zemp, N.; Widmer, A.; Hilbeck, A. Transgene expression and Bt protein content in transgenic Bt maize (MON810) under optimal and stressful environmental conditions. PLoS ONE 2015, 10, e0123011. [Google Scholar] [CrossRef] [PubMed]
- Archer, T.L.; Schuster, G.; Patrick, C.; Cronholm, G.; Bynum, E.D., Jr.; Morrison, W.P. Whorl and stalk damage by european and southwestern corn borers to four events of Bacillus thuringiensis transgenic maize. Crop Prot. 2000, 19, 181–190. [Google Scholar] [CrossRef]
- Tabashnik, B.E.; Huang, F.; Ghimire, M.N.; Leonard, B.R.; Siegfried, B.D.; Rangasamy, M.; Yang, Y.; Wu, Y.; Gahan, L.J.; Heckel, D.G.; et al. Efficacy of genetically modified Bt toxins against insects with different genetic mechanisms of resistance. Nat. Biotechnol. 2011, 29, 1128. [Google Scholar] [CrossRef] [PubMed]
- van Frankenhuyzen, K.; Liu, Y.; Tonon, A. Interactions between Bacillus thuringiensis subsp. Kurstaki HD-1 and midgut bacteria in larvae of gypsy moth and spruce budworm. J. Invertebr. Pathol. 2010, 103, 124–131. [Google Scholar] [PubMed]
- Coors, J.G. Resistance to the european corn borer, Ostrinia nubilalis (Hubner), in maize, Zea mays L., as affected by soil silica, plant silica, structural carbohydrates, and lignin. In Proceedings of the Second International Symposium on Genetic Aspects of Plant Mineral Nutrition, Genetic Aspects of Plant Mineral Nutrition, Madison, MI, USA, 16–20 June 1985; Gabelman, W.H., Loughman, B.C., Eds.; Springer: Dordrecht, The Netherlands, 1987; pp. 445–456. [Google Scholar]
- Meihls, L.N.; Handrick, V.; Glauser, G.; Barbier, H.; Kaur, H.; Haribal, M.M.; Lipka, A.E.; Gershenzon, J.; Buckler, E.S.; Erb, M.; et al. Natural variation in maize aphid resistance is associated with 2,4-dihydroxy-7-methoxy-1,4-benzoxazin-3-one glucoside methyltransferase activity. Plant Cell 2013, 25, 2341–2355. [Google Scholar] [CrossRef] [PubMed]
- Chacón, J.M.; Asplen, M.K.; Heimpel, G.E. Combined effects of host-plant resistance and intraguild predation on the soybean aphid parasitoid Binodoxys communis in the field. Biol. Control 2012, 60, 16–25. [Google Scholar] [CrossRef]
- Denno, R.F.; McClure, M.S. Introduction: Variability: A key to understanding plant–herbivore interactions. In Variable Plants and Herbivores in Natural and Managed Systems; Denno, R.F., McClure, M.S., Eds.; Academic Press: Cambridge, MA, USA, 1983; pp. 1–12. [Google Scholar]
- Panda, N.; Khush, G.S. Host Plant Resistance to Insects; CAB International in Association with the International Rice Research Institute: New York, NY, USA, 1995. [Google Scholar]
- Bernays, E.A. Insect-Plant Interactions; CRC Press: Boca Raton, FL, USA, 1989; Volume 1. [Google Scholar]
- van Emden, H.F. The role of host plant resistance in insect pest mis-management. Bull. Entomol. Res. 2009, 81, 123–126. [Google Scholar] [CrossRef]
- Silbergeld, E.K.; Graham, J.; Price, L.B. Industrial food animal production, antimicrobial resistance, and human health. Annu. Rev. Public Health 2008, 29, 151–169. [Google Scholar] [CrossRef] [PubMed]
- Sarmah, A.K.; Taylor, M.T.; Boxall, A.B.A. A global perspective on the use, sales, exposure pathways, occurrence, fate and effects of veterinary antibiotics (VAs) in the environment. Chemosphere 2006, 65, 725–759. [Google Scholar] [CrossRef] [PubMed]
- Glyphosate Formulations and Their Use for the Inhibition of 5-Enolpyruvylshikimate-3-Phosphate Synthase. U.S. Patent 7771736 b2, 30 August 2002. Available online: http://www.Google.Com/patents/us7771736 (accessed on 28 May 2015).
- Lancaster, S.H.; Hollister, E.B.; Senseman, S.A.; Gentry, T.J. Effects of repeated glyphosate applications on soil microbial community composition and the mineralization of glyphosate. Pest Manag. Sci. 2010, 66, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Kruger, M.; Shehata, A.A.; Schrodl, W.; Rodloff, A. Glyphosate suppresses the antagonistic effect of Enterococcus spp. On Clostridium botulinum. Anaerobe 2013, 20, 74–78. [Google Scholar] [CrossRef] [PubMed]
- Lozano, V.L.; Defarge, N.; Rocque, L.M.; Mesnage, R.; Hennequin, D.; Cassier, R.; de Vendomois, J.S.; Panoff, J.M.; Seralini, G.E.; Amiel, C. Sex-dependent impact of Roundup on the rat gut microbiome. Toxicol. Rep. 2018, 5, 96–107. [Google Scholar] [CrossRef] [PubMed]
- Schulz, A.; Krüper, A.; Amrhein, N. Differential sensitivity of bacterial 5-enolpyruvylshikimate-3-phosphate synthases to the herbicide glyphosate. FEMS Microbiol. Lett. 1985, 28, 297–301. [Google Scholar] [CrossRef]
- Dai, P.; Yan, Z.; Ma, S.; Yang, Y.; Wang, Q.; Hou, C.; Wu, Y.; Liu, Y.; Diao, Q. The herbicide glyphosate negatively affects midgut bacterial communities and survival of honey bee during larvae reared in vitro. J. Agric. Food Chem. 2018, 66, 7786–7793. [Google Scholar] [CrossRef] [PubMed]
- Porcar, M.; Garcia-Robles, I.; Dominguez-Escriba, L.; Latorre, A. Effects of Bacillus thuringiensis Cry1Ab and Cry3Aa endotoxins on predatory coleoptera tested through artificial diet-incorporation bioassays. Bull. Entomol. Res. 2010, 100, 297–302. [Google Scholar] [CrossRef] [PubMed]
- Ivaldi-Sender, C. Simple techniques for a permanent breeding of oriental fruit moth Grapholitha molesta (Lepidoptera: Tortricidae) on artificial diet. Ann. Zool. Ecol. Anim. 1974, 6, 337–343. [Google Scholar]
- Poulsen, M.; Schroder, M.; Wilcks, A.; Kroghsbo, S.; Lindecrona, R.H.; Miller, A.; Frenzel, T.; Danier, J.; Rychlik, M.; Shu, Q.; et al. Safety testing of GM-rice expressing PHA-E lectin using a new animal test design. Food Chem. Toxicol. 2007, 45, 364–377. [Google Scholar] [CrossRef] [PubMed]
- Muñoa, F.J.; Pares, R. Selective medium for isolation and enumeration of Bifidobacterium spp. Appl. Environ. Microbiol. 1988, 54, 1715–1718. [Google Scholar] [PubMed]
- Cox, D.R. Regression models and life-tables. Journal of the Royal Statistical Society. Ser. B (Methodol.) 1972, 34, 187–220. [Google Scholar]
| Reference | Route of Administration, Type of Bt Toxin | Source of Bt Toxins | Test Species, Tested Lifestage | Treatments | Observed Effects (% Survival) | |
|---|---|---|---|---|---|---|
| Antibiotics | Bt toxins | |||||
| Broderick et al. 2006 [19] | AD | Cry1Aa, Cry1Ab, Cry1Ac, Cry2A proteins (crystals) and spores; Cry1Aa protoxins | Dipel, commercial formulation of B. thuringiensis var. kurstaki incl. other compounds for formulation; E. coli strain ECE52 producing | Lymantria dispar L3 | Pre-treatment AB prior to assay: Rifampin, Gentamicin, Penicillin, Streptomycin; Gut bacteria: Enterobacter sp. NAB3, Enterococcus casseliflavus, Staphylococcus xylosus; | Close to zero survival without antibiotics, 100% survival with no gut bacteria present (with antibiotics) after 7 days. No timeline for survival Hypothesis: Septicemia by gut bacteria Cry toxins enable gut bacteria to reach hemocoel (breaching gut wall) but does not kill the larvae. |
| Broderick et al. 2009 [20] | AD | Cry1Aa, Cry1Ab, Cry1Ac, Cry2A proteins (crystals) and spores Cry1Ac protoxins encapsulated in Pseudomonas fluorescens | Dipel, commercial formulation of B. thuringiensis var. kurstaki incl. other compounds for formulation; MPVII, commercial formulation incl. other compounds for formulation | Vanessa cardui, Manduca sexta, Heliothis virescence L3 Pieris rapae L4 Pectinophorella gossypiella, Lymantria dispar L? | Pre-treatment AB prior to assay: Rifampin, Gentamicin, Penicillin, Streptomycin; Gut bacteria: Enterobacter sp. NAB3 | For all, except P. gossypiella, survival exceeded 90% in presence of antibiotics after 7 days. For P. gossypiella another mechanism seemed at work No timeline of survival given but mentioned that “antibiotics greatly delayed time ... to kill.” Hypothesis: Except for P. gossypiella, septicemia caused by Enterobacter and not Bt—confirmed by use of MPVII free Bt and live cells |
| Mason et al. 2011 [21] | AD, injections | Cry1Ac protoxins encapsulated in Pseudomonas fluorescens | MPVII, commercial formulation incl. other compounds for formulation | Manduca sexta, L5 | Pre-treatment AB prior to assay: rifampin, gentamicin, nystatin (antifungal); Gut bacteria: Enterococcus faecalis (Ef) ± Bt; starvation | E. faecalis + Bt—0% at day 5 Bt 0% at day 8 (like starvation)—delay in mortality—although no time line presented Hypothesis: E. faecalis induced septicemia, Bt toxin caused paralysis and death due to starvation—combined effect E. faecalis + Bt |
| Raymond et al. 2009 [22] | AD | Cry 1 and Cry 2 crystals Cry1Ac crystals | HD-1 strain derived from Dipel, commercial formulation of B. thuringiensis var. kurstaki HD-73, and rifampicin resistant strains of HG-73 reifR | Plutella xylostella L2 and L3 | Pre-treatment AB prior to assay: Rifampin Gut bacteria: Enterobacter sp. Mn2 | Significant delays in mortality onset with ABs 0% survival when Bt w/o AB or AB-resistant Bt was used Cry1Ac from HD-73 survival 10% w/o AB, 20% with AB, effect annulled when AB-resistant HD-73 strain was used Hypothesis: Septicemia by Bt and toxins, no gut bacteria necessary to kill larvae. |
| Johnston and Crickmore 2009 [24] | AD | Cry 1 and Cry 2 toxins crystals Cry1Ac crystals | HD-1 strain derived from Dipel, commercial formulation of B. thuringiensis var. kurstaki HD-73, and rifampicin resistant strains of HG-73 reif R | Manduca sexta L2 and L3 | Pre-treatment AB prior to assay: first instar only or continuously | Timelines show significant delays in mortality onset with ABs Cry1Ac from HD-73: 0% survival w/o AB, 10% with AB after 6 days Dipel: 0% survival w/o AB, 20% survival with AB after 6 days Hypothesis: Septicemia by Bt and toxins, no gut bacteria |
| Ingredient | Amount | Ingredient Description | Manufacturer |
|---|---|---|---|
| Total Volume | 0.5 L | - | - |
| H2O Dest. | 390 mL | - | - |
| Agar | 10 g | - | Sigma-Aldrich (Buchs, S) |
| Maize Semolina | 25 g | M Classic Polenta 2 min, Maisgriess | Migros (Zurich, S) |
| Wheat Germ | 25 g | Qualité & Prix Weizenkeime | Coop (Zurich, S) |
| Yeast Powder | 25 g | Actilife Vitamin-Bierhefe | Migros (Zurich, S) |
| Benzoic Acid | 0.9 g | - | Sigma-Aldrich (Buchs, S) |
| Nipagin | 0.9 g | Methyl 4-hydroxybenzoate | Sigma-Aldrich (Buchs, S) |
| Ascorbic Acid | 2.25 g | L-ascorbic acid | Sigma-Aldrich (Buchs, S) |
| Ingredient | Amount | Ingredient Description | Manufacturer |
|---|---|---|---|
| Penicillin G Potassium Salt | 500 mg/L | ~1600 units/mg | Sigma-Aldrich (Buchs, S) |
| Gentamycin Sulfate | 500 mg/L | From Micromonospora purpurea, −700 U/mg dried material | Sigma-Aldrich (Buchs, S) |
| Rifampicin | 500 mg/L | ≥97% (HPLC) powder | Sigma-Aldrich (Buchs, S) |
| Streptomycin Sulfate Salt | 500 mg/L | - | Sigma-Aldrich (Buchs, S) |
| Selection | Media | Conditions |
|---|---|---|
| Agar plates | ||
| Total Aerobes | Plate Count Agar (Biokar, Beauvais, France) | 72 h Aerobically, 30 °C |
| Total Anaerobes | Plate Count Agar (Biokar, Beauvais, France) | 72 h Anaerobically (AnaeroGen 2.5 L, Sigma-Aldrich), 30 °C |
| Enterococci | Slanetz and Bartley (Biokar, Beauvais, France) | 48 h Aerobically, 37 °C |
| Maize Varieties | Antibiotic Treatment (Ab) | ||
|---|---|---|---|
| Ab 3/5 days | Ab Cont. | 0 Ab (Control) | |
| Bt South Africa | 32 larvae | 32 larvae | 32 larvae |
| non-Bt South Africa | 32 larvae | 32 larvae | 32 larvae |
| Bt Spain | 32 larvae | 32 larvae | 32 larvae |
| non-Bt Spain | 32 larvae | 32 larvae | 32 larvae |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hilbeck, A.; Defarge, N.; Bøhn, T.; Krautter, M.; Conradin, C.; Amiel, C.; Panoff, J.-M.; Trtikova, M. Impact of Antibiotics on Efficacy of Cry Toxins Produced in Two Different Genetically Modified Bt Maize Varieties in Two Lepidopteran Herbivore Species, Ostrinia nubilalis and Spodoptera littoralis. Toxins 2018, 10, 489. https://doi.org/10.3390/toxins10120489
Hilbeck A, Defarge N, Bøhn T, Krautter M, Conradin C, Amiel C, Panoff J-M, Trtikova M. Impact of Antibiotics on Efficacy of Cry Toxins Produced in Two Different Genetically Modified Bt Maize Varieties in Two Lepidopteran Herbivore Species, Ostrinia nubilalis and Spodoptera littoralis. Toxins. 2018; 10(12):489. https://doi.org/10.3390/toxins10120489
Chicago/Turabian StyleHilbeck, Angelika, Nicolas Defarge, Thomas Bøhn, Michelle Krautter, Constanze Conradin, Caroline Amiel, Jean-Michel Panoff, and Miluse Trtikova. 2018. "Impact of Antibiotics on Efficacy of Cry Toxins Produced in Two Different Genetically Modified Bt Maize Varieties in Two Lepidopteran Herbivore Species, Ostrinia nubilalis and Spodoptera littoralis" Toxins 10, no. 12: 489. https://doi.org/10.3390/toxins10120489
APA StyleHilbeck, A., Defarge, N., Bøhn, T., Krautter, M., Conradin, C., Amiel, C., Panoff, J.-M., & Trtikova, M. (2018). Impact of Antibiotics on Efficacy of Cry Toxins Produced in Two Different Genetically Modified Bt Maize Varieties in Two Lepidopteran Herbivore Species, Ostrinia nubilalis and Spodoptera littoralis. Toxins, 10(12), 489. https://doi.org/10.3390/toxins10120489





