Cyanobacterial Blooms and Microcystins in Southern Vietnam
Abstract
1. Introduction
2. Results
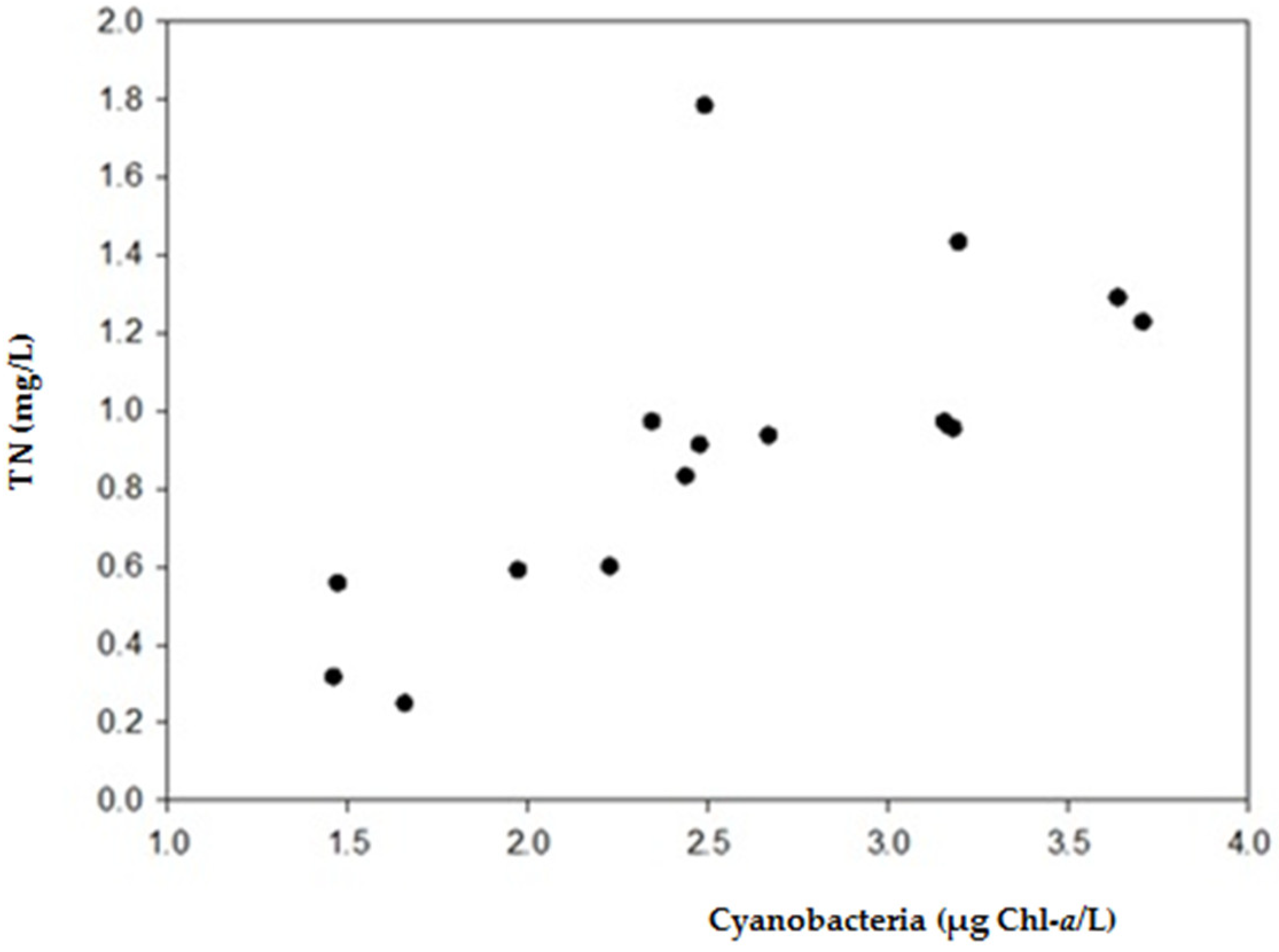
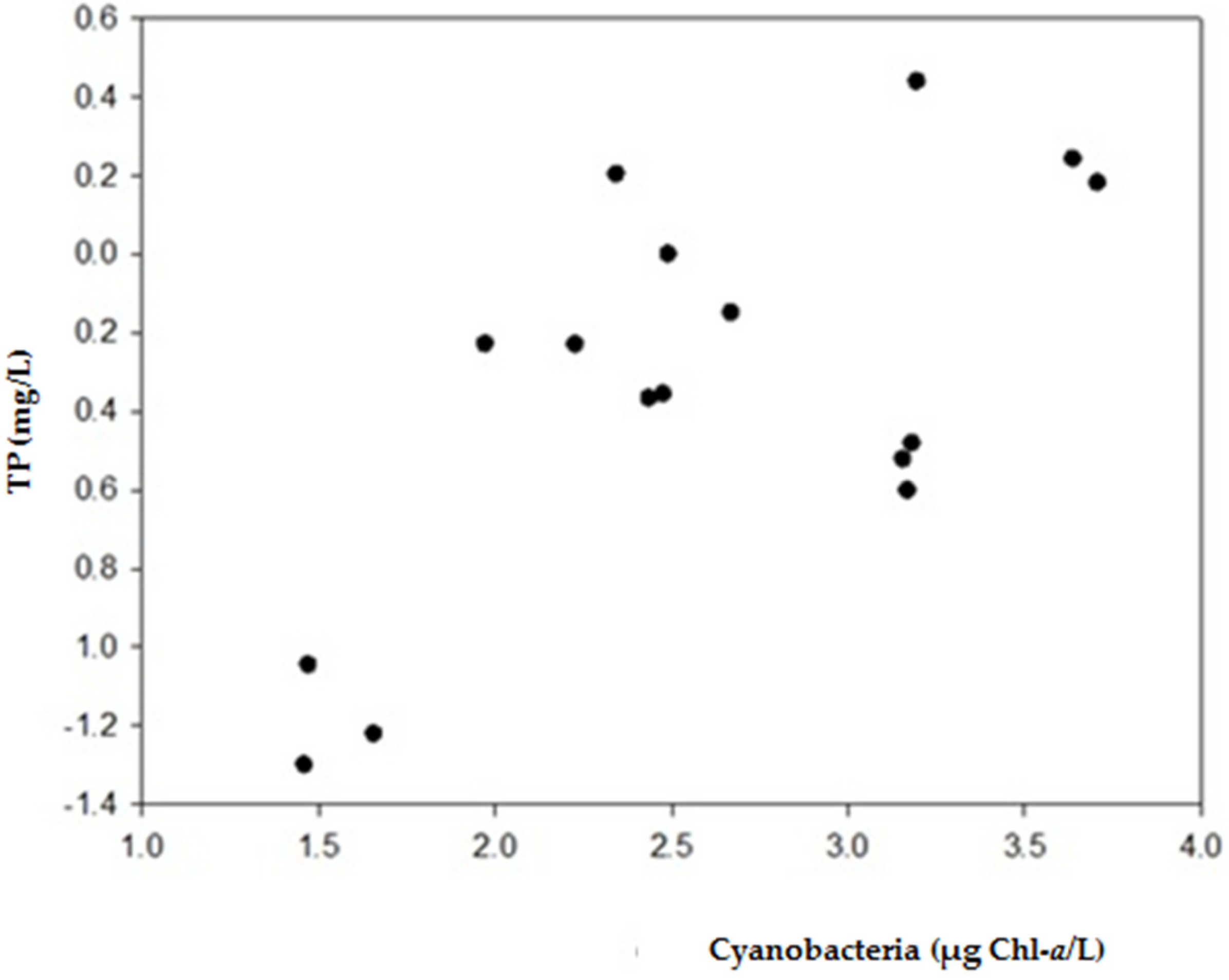
2.1. Physical and Chemical Characteristics and Cyanobacterial Blooms
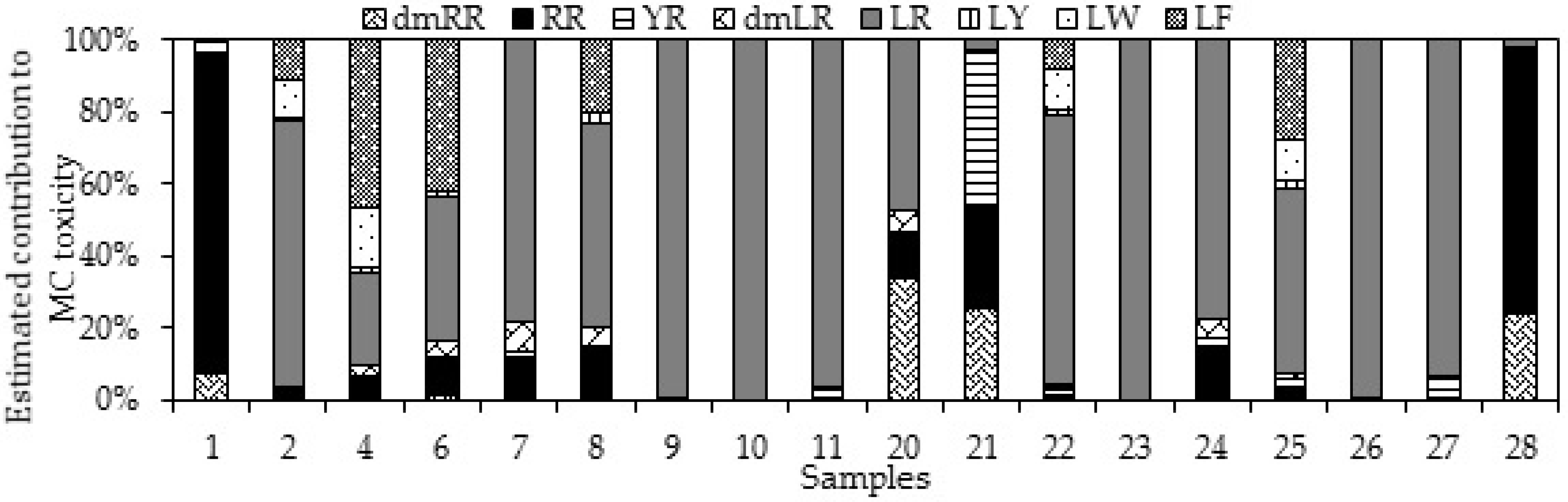
2.2. MC Content in Cyanobacterial Field Samples and Isolated Strains
2.3. MCs Accumulation in Animals
3. Discussions
3.1. Occurrence of Cyanobacterial Blooms
3.2. MC Content in Cyanobacterial Field Samples and Isolated Strains
3.3. MC Accumulation In Animals
3.4. Public Health Risk Assessment
3.4.1. Risk Assessment for Drinking Water Supplies
3.4.2. Risk Assessment for MC-Contaminated Foods
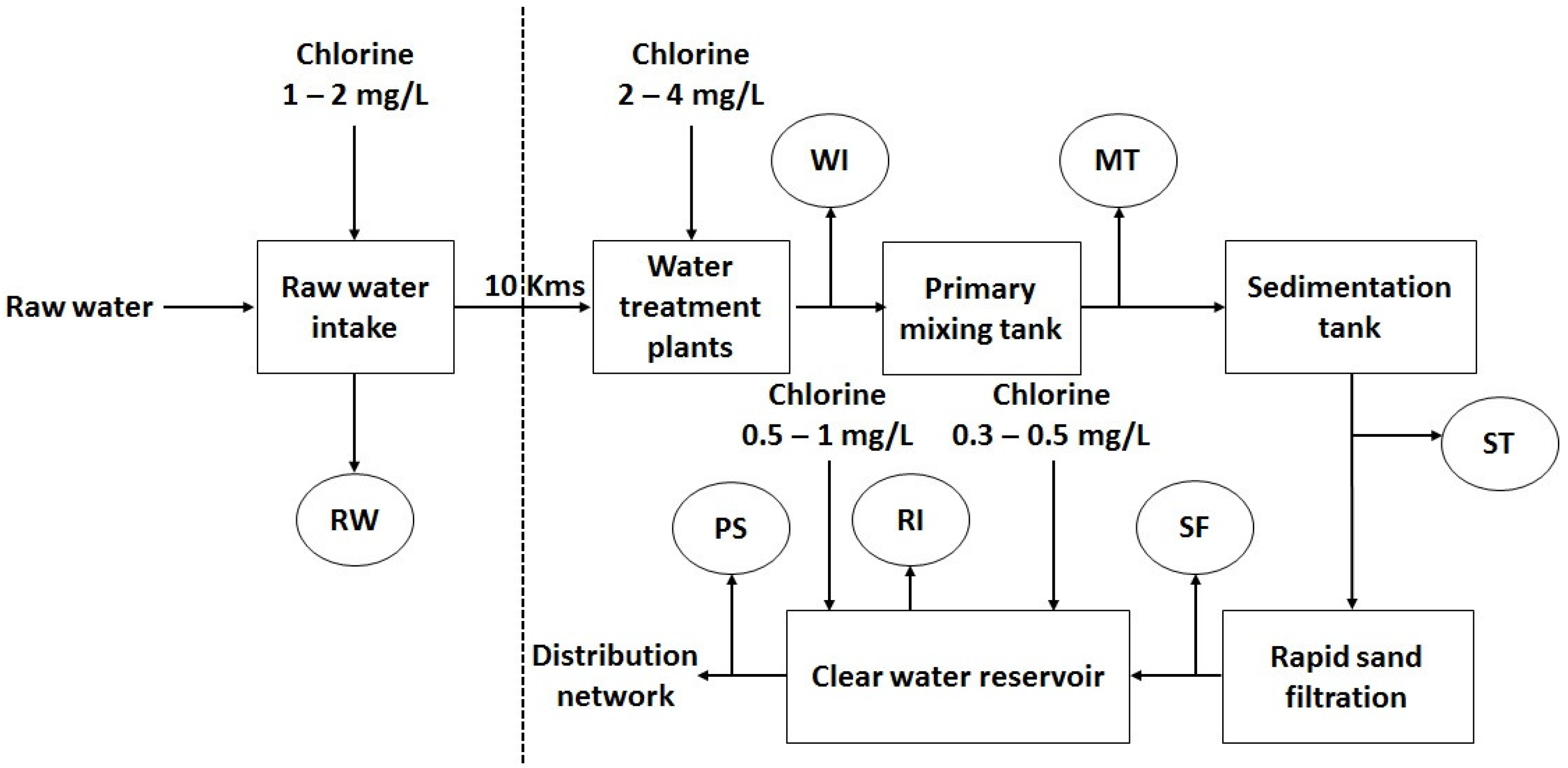
3.4.3. Cyanobacteria Control in Southern Vietnam
4. Conclusions
5. Materials and Methods
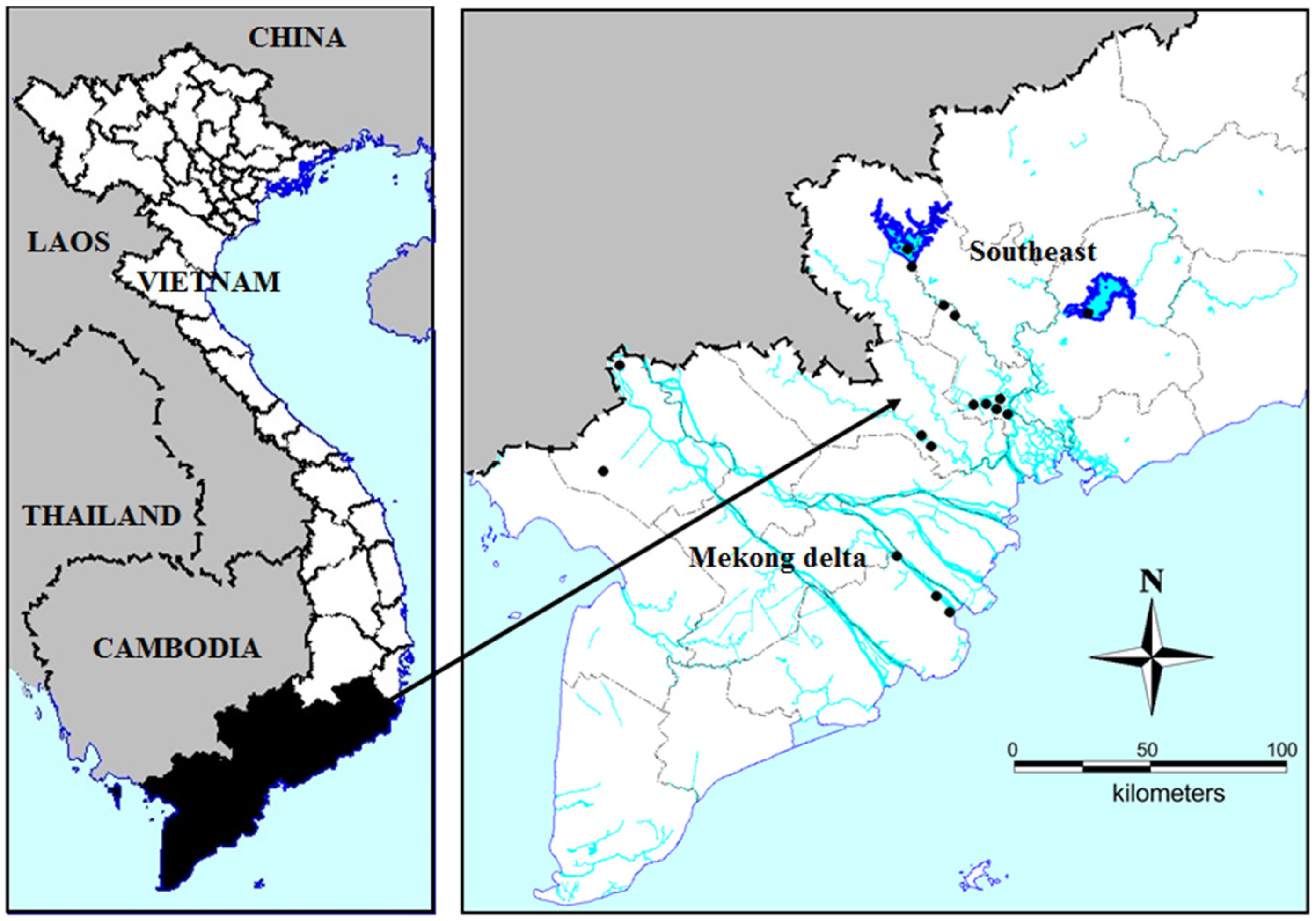
5.1. Sampling
5.2. Strains Isolation
5.3. MC Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
Appendix B
| No. | Name | dmRR | RR | NOD | YR | dmLR | LR | LY | LW | LF | Total |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Microcystis sp.1 | UDL | UDL | UDL | 0 | 19.62 | 2255.31 | UDL | UDL | UDL | 2274.94 |
| 2 | Microcystis sp.2 | UDL | UDL | UDL | 5.87 | 6.55 | 67.08 | UDL | UDL | UDL | 79.50 |
| 3 | Microcystis sp.3 | UDL | UDL | UDL | 0 | 6.12 | 1693.98 | UDL | UDL | UDL | 1700.10 |
| 4 | Microcystis sp.4 | UDL | UDL | UDL | 539.80 | 4.93 | 336.06 | UDL | UDL | UDL | 880.79 |
| 5 | Microcystis sp.5 | UDL | UDL | UDL | UDL | 0.66 | 1368.22 | UDL | UDL | UDL | 1368.88 |
| 6 | Microcystis sp.6 | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL |
| 7 | Microcystis sp.7 | UDL | UDL | UDL | UDL | UDL | 1.22 | UDL | UDL | UDL | 1.22 |
| 8 | Microcystis sp.8 | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL |
| 9 | Microcystis sp.9 | 5.10 | 19.03 | 0.03 | 0.21 | 1.32 | 9.67 | UDL | UDL | UDL | 35.36 |
| 10 | Microcystis sp.10 | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL |
| 11 | Microcystis sp.11 | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL |
| 12 | Microcystis sp.12 | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL |
| 13 | Microcystis sp.13 | 18.41 | 123.66 | UDL | 2.057 | UDL | UDL | UDL | UDL | UDL | 144.13 |
| 14 | Anabaenopsis sp.1 | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL |
| 15 | Anabaenopsis sp.2 | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL |
| 16 | Anabaenopsis sp.3 | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL |
| 17 | Anabaenopsis sp.4 | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL |
| 18 | Anabaenopsis sp.5 | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL |
| 19 | Anabaenopsis sp.6 | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL |
| 20 | Anabaena sp. 1 | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL |
| 21 | Anabaena sp. 2 | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL |
| 22 | Anabaena sp. 3 | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL |
| 23 | Anabaena sp. 4 | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL |
| 24 | Planktothrix sp.1 | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL |
| No. | Cyanobacteria | dmRR | RR | NOD | YR | dmLR | LR | LY | LW | LF | Total |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Microcystis sp.1 | UDL | UDL | UDL | 0 | 65.68 | 6773.68 | UDL | UDL | UDL | 6839.35 |
| 2 | Microcystis sp.2 | UDL | UDL | UDL | 33.65 | 30.26 | 257.98 | UDL | UDL | UDL | 321.90 |
| 3 | Microcystis sp.3 | UDL | UDL | UDL | 0 | 23.03 | 4418.81 | UDL | UDL | UDL | 4441.84 |
| 4 | Microcystis sp.4 | UDL | UDL | UDL | 1377.87 | 12.74 | 901.75 | UDL | UDL | UDL | 2292.36 |
| 5 | Microcystis sp.5 | UDL | UDL | UDL | UDL | 21.27 | 9203.45 | UDL | UDL | UDL | 9224.72 |
| 6 | Microcystis sp.6 | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL |
| 7 | Microcystis sp.7 | UDL | UDL | UDL | UDL | UDL | 4.66 | UDL | UDL | UDL | 4.66 |
| 8 | Microcystis sp.8 | UDL | UDL | UDL | UDL | UDL | 2.04 | UDL | UDL | UDL | 2.036 |
| 9 | Microcystis sp.9 | 87.30 | 307.47 | 0.32 | 6.65 | 38.95 | 155.16 | UDL | UDL | UDL | 595.84 |
| 10 | Microcystis sp.10 | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL |
| 11 | Microcystis sp.11 | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL |
| 12 | Microcystis sp.12 | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL |
| 13 | Microcystis sp.13 | 182.73 | 751.11 | 0.40 | 22.82 | UDL | UDL | 1.02 | 1.40 | UDL | 959.48 |
| 14 | Anabaenopsis sp.1 | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL |
| 15 | Anabaenopsis sp.2 | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL |
| 16 | Anabaenopsis sp.3 | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL |
| 17 | Anabaenopsis sp.4 | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL |
| 18 | Anabaenopsis sp.5 | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL |
| 19 | Anabaenopsis sp.6 | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL |
| 20 | Anabaena sp. 1 | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL |
| 21 | Anabaena sp. 2 | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL |
| 22 | Anabaena sp. 3 | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL |
| 23 | Anabaena sp. 4 | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL |
| 24 | Planktothrix sp.1 | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL | UDL |
Appendix C
| No. | Animals | Organ | dmRR | MCRR | NOD | MCYR | dmLR | MCLR | MCLY | MCLW | MCLF | Total |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Fish | ||||||||||||
| 1 | Ganges river sprat (Corica soborna) | Visceral | UDL | 1.29 | UDL | UDL | UDL | 1.09 | UDL | UDL | UDL | 2.38 |
| 2 | Tilapia (Oreochromis sp.) | Visceral | UDL | 2.87 | UDL | UDL | UDL | n.q. | UDL | UDL | UDL | 2.87 |
| 3 | Tilapia (Oreochromis sp.) | Visceral | UDL | UDL | UDL | UDL | UDL | 0.76 | UDL | UDL | UDL | 0.76 |
| 4 | Tilapia (Oreochromis sp.) | Gut | UDL | 9.91 | UDL | UDL | 0.36 | 1.29 | UDL | UDL | UDL | 11.55 |
| 5 | Tilapia (Oreochromis sp.) | Digestive tract | UDL | UDL | UDL | 4.90 | 1.22 | 109.83 | UDL | UDL | UDL | 115.95 |
| 6 | Tilapia (Oreochromis sp.) | Liver | UDL | UDL | UDL | 0.27 | UDL | 13.12 | UDL | UDL | UDL | 13.39 |
| 7 | Sknakskin gourami (Trichogaster pectoralis) | Visceral | UDL | UDL | UDL | 0.02 | UDL | 13.84 | UDL | UDL | UDL | 13.86 |
| 8 | Gold fish (Carassius sp.) | Visceral | UDL | UDL | UDL | UDL | UDL | 18.57 | UDL | UDL | UDL | 18.57 |
| 9 | Gold fish (Carassius sp.) | Digestive tract | UDL | UDL | UDL | UDL | UDL | 0.36 | UDL | UDL | UDL | 0.36 |
| 10 | Suckermouth catfish (Hypostomus plecostomus) | Digestive tract | UDL | UDL | UDL | 9.08 | 1.15 | 98.15 | UDL | UDL | UDL | 108.38 |
| 11 | Suckermouth catfish (Hypostomus plecostomus) | Liver | UDL | UDL | UDL | 0.99 | UDL | 17.99 | UDL | UDL | UDL | 18.97 |
| 12 | Suckermouth catfish (Hypostomus plecostomus) | Musscle | UDL | UDL | UDL | 0.07 | UDL | 3.66 | UDL | UDL | UDL | 3.73 |
| Mollusca | ||||||||||||
| 13 | Apple snail (Pomacea canaliculata) | Visceral | UDL | UDL | UDL | UDL | 0.21 | UDL | UDL | UDL | UDL | 0.21 |
| 14 | Apple snail (Pomacea canaliculata) | Visceral | UDL | UDL | UDL | UDL | 0.33 | UDL | UDL | UDL | UDL | 0.33 |
| 15 | Apple snail (Pomacea canaliculata) | Visceral | UDL | 1.67 | UDL | UDL | 0.22 | UDL | UDL | UDL | UDL | 1.89 |
| 16 | Apple snail (Pomacea canaliculata) | Visceral | UDL | UDL | UDL | UDL | 0.24 | UDL | UDL | UDL | UDL | 0.24 |
| 17 | Apple snail (Pomacea canaliculata) | Visceral | UDL | UDL | UDL | UDL | 0.36 | UDL | UDL | UDL | UDL | 0.36 |
| 18 | Apple snail (Pomacea canaliculata) | Visceral | UDL | 2.35 | UDL | UDL | 0.36 | UDL | UDL | UDL | UDL | 2.71 |
| 19 | Apple snail (Pomacea canaliculata) | Visceral | UDL | 2.61 | UDL | UDL | 0.37 | UDL | UDL | UDL | UDL | 2.98 |
| 20 | Apple snail (Pomacea canaliculata) | Visceral | UDL | UDL | UDL | UDL | 0.25 | UDL | UDL | UDL | UDL | 0.25 |
| 21 | Apple snail (Pomacea canaliculata) | Visceral | UDL | UDL | UDL | UDL | 0.26 | UDL | UDL | UDL | UDL | 0.26 |
| 22 | Apple snail (Pomacea canaliculata) | Visceral | UDL | UDL | UDL | UDL | 0.27 | UDL | UDL | UDL | UDL | 0.27 |
| 23 | Apple snail (Pomacea canaliculata) | Visceral | UDL | 2.75 | UDL | UDL | 0.46 | UDL | UDL | UDL | UDL | 3.21 |
| 24 | Apple snail (Pomacea canaliculata) | Visceral | UDL | 2.56 | UDL | UDL | 0.43 | UDL | UDL | UDL | UDL | 2.99 |
| 25 | Apple snail (Pomacea canaliculata) | Visceral | UDL | UDL | UDL | UDL | 0.33 | UDL | UDL | UDL | UDL | 0.33 |
| 26 | Apple snail (Pomacea canaliculata) | Visceral | UDL | UDL | UDL | UDL | 0.28 | UDL | UDL | UDL | UDL | 0.28 |
| 27 | Apple snail (Pomacea canaliculata) | Visceral | UDL | UDL | UDL | UDL | 0.28 | UDL | UDL | UDL | UDL | 0.28 |
| 28 | Apple snail (Pomacea canaliculata) | Visceral | UDL | UDL | UDL | UDL | UDL | 2.56 | UDL | UDL | UDL | 2.56 |
| 29 | Apple snail (Pomacea canaliculata) | Visceral | UDL | UDL | UDL | 0.05 | UDL | 2.86 | UDL | UDL | UDL | 2.90 |
| 30 | Apple snail (Pomacea canaliculata) | Visceral | UDL | UDL | UDL | UDL | UDL | 1.61 | UDL | UDL | UDL | 1.61 |
| 31 | Apple snail (Pomacea canaliculata) | Egg | UDL | UDL | UDL | UDL | 0.23 | UDL | UDL | UDL | UDL | 0.23 |
| 32 | Apple snail (Pomacea canaliculata) | Musscle | UDL | UDL | UDL | UDL | 0.15 | UDL | UDL | UDL | UDL | 0.15 |
| 33 | Apple snail (Pomacea canaliculata) | Musscle | UDL | UDL | UDL | UDL | 0.23 | UDL | UDL | UDL | UDL | 0.23 |
| 34 | Apple snail (Pomacea canaliculata) | Musscle | UDL | UDL | UDL | UDL | 0.30 | UDL | UDL | UDL | UDL | 0.30 |
| 35 | Apple snail (Pomacea canaliculata) | Whole neonates | UDL | UDL | UDL | UDL | 0.25 | UDL | UDL | UDL | UDL | 0.25 |
| Crustaceae | UDL | UDL | UDL | |||||||||
| 36 | White leg shrimp (Litopenaeus vannamei) | Whole head | UDL | 14.43 | UDL | UDL | UDL | 0.79 | UDL | UDL | UDL | 15.21 |
References
- Bartram, J.; Burch, M.; Falconer, I.R.; Jones, G.; Kuiper-Goodman, T. Situation Assessment, Planning and Management. In Toxic Cyanobacteria in Water: A Guide to Their Public Health Consequences, Monitoring, and Management; Chorus, I., Bartram, J., Eds.; E & FN Spon.: London, UK, 1999; pp. 183–210. [Google Scholar]
- Graham, J.L.; Loftin, K.A.; Ziegler, A.C.; Meyer, M.T. Guidelines for Design and Sampling for Cyanobac-Terial Toxin and Taste-and-Odor Studies in Lakes and Reservoirs; U.S. Geological Survey Scientific Investigations Report; U.S. Geological Survey Scientific: Reston, VA, USA, 2008; Volume 5038, 39p.
- Hudnell, H.K.; Stewart, I.; Seawright, A.; Shaw, G. Cyanobacterial poisoning in livestock, wild mammals and birds—An overview. In Cyanobacterial Harmful Algal Blooms: State of the Science and Research Needs; Springer: Berlin, Germany, 2008; pp. 613–637. [Google Scholar]
- Faassen, E.J.; Harkema, L.; Begeman, L.; Lurling, M. First report of (homo)anatoxin-a and dog neurotoxicosis after ingestion of benthic cyanobacteria in The Netherlands. Toxicon 2012, 60, 378–384. [Google Scholar] [CrossRef]
- Lürling, M.; Faassen, E. Dog Poisonings Associated with a Microcystis aeruginosa Bloom in the Netherlands. Toxins 2013, 5, 556–567. [Google Scholar] [CrossRef]
- Saker, M.L.; Thomas, A.D.; Norton, J.H. Cattle mortality attributed to the toxic cyanobacterium Cylindrospermopsis raciborskii in an outback region of north Queensland. Environ. Toxicology 1999, 14, 179–182. [Google Scholar]
- Hawkins, P.R.; Runnegar, M.T.C.; Jackson, A.R.B.; Falconer, I.R. Severe hepatotoxicity caused by the tropical cyanobacterium (blue-green alga) Cylindrospermopsis raciborskii (Woloszynska) Seenaya and Subba Raju isolated from a domestic supply reservoir. Appl. Environ. Microbiol. 1985, 50, 1292–1295. [Google Scholar] [PubMed]
- Bourke, A.T.C.; Hawes, R.B.; Neilson, A.; Stallman, N.D. An outbreak of hepato-enteritis (the Palm Island mystery disease) possibly caused by algal intoxication. Toxicon 1983, 3, 45–48. [Google Scholar] [CrossRef]
- Backer, L.; Manassaram-Baptiste, D.; LePrell, R.; Bolton, B. Cyanobacteria and Algae Blooms: Review of Health and Environmental Data from the Harmful Algal Bloom-Related Illness Surveillance System (HABISS) 2007–2011. Toxins 2015, 7, 1048. [Google Scholar] [CrossRef] [PubMed]
- Sivonen, K.; Jones, G. Cyanobacterial toxins. In Toxic Cyanobacteria in Water: A Guide to Their Public Health Consequences, Monitoring, and Management; Chorus, I., Bartram, J., Eds.; E & FN Spon.: London, UK, 1999; pp. 41–111. [Google Scholar]
- Zurawell, R.W.; Chen, H.; Burke, J.M.; Prepas, E.E. Hepatotoxic Cyanobacteria: A Review of the Biological Importance of Microcystins in Freshwater Environments. J. Toxicol. Environ. Health 2005, 8 Pt B, 1–37. [Google Scholar] [CrossRef]
- Fan, J.; Ho, L.; Hobson, P.; Brookes, J. Evaluating the effectiveness of copper sulphate, chlorine, potassium permanganate, hydrogen peroxide and ozone on cyanobacterial cell integrity. Water Res. 2013, 47, 5153–5164. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.; Hobson, P.; Ho, L.; Daly, R.; Brookes, J. The effects of various control and water treatment processes on themembrane integrity and toxin fate of cyanobacteria. J. Hazard. Mater. 2014, 264, 313–322. [Google Scholar] [CrossRef] [PubMed]
- Wilbers, G.-J.; Sebesvari, Z.; Renaud, F. Piped-Water Supplies in Rural Areas of the Mekong Delta, Vietnam: Water Quality and Household Perceptions. Water 2014, 6, 2175. [Google Scholar] [CrossRef]
- Shirota, A. The Plankton of South Vietnam. Freshwater and Marine Plankton; Overseas Technical Cooperation Agency Japan: Tokyo, Japan, 1966. [Google Scholar]
- Pham, H.H. Quelques Algues d’eau Douce de la Région de Cantho; Annals of the University of Cantho, Science and Agriculture: Cantho, Vietnam, 1969; pp. 35–59. [Google Scholar]
- Nguyen, T.T. Algae from the Central-Highland of Vietnam; Annals of Ho Chi Minh City University: Ho Chi Minh City, Vietnam, 1983; pp. 113–118. [Google Scholar]
- Nguyen, T.T. The freshwater algae of Nam Cat Tien National Park. In Bulletin of Natural Sciences, No. 1; College of Natural Sciences, Vietnam National University: Ho Chi Minh City, Vietnam, 1997; pp. 57–72. [Google Scholar]
- Thi Nguyet Hong, P.; Coute, A.; Bourrelly, P. Les cyanophycées du delta du Mékong (Viet-Nam). Nova Hedwigia 1992, 54, 403–446. [Google Scholar]
- Duong, D.T. Taxonomy of Cyanobacteria of Vietnam; Agricultural Publishing House: Ha Noi, Vietnam, 1996; p. 220. [Google Scholar]
- Hummert, C.; Dahlmann, J.; Reinhardt, K.; Dang, H.P.H.; Dang, D.K.; Luckas, B. Liquid chromatography-mass spectrometry identification of microcystins in Microcystis aeruginosa strain from lake Thanh Cong, Hanoi, Vietnam. Chromatographia 2001, 54, 569–575. [Google Scholar] [CrossRef]
- Dao, T.S.; Cronberg, G.; Nimptsch, J.; Do-Hong, L.-C.; Wiegand, C. Toxic cyanobacteria from Tri An Reservoir, Vietnam. Nova Hedwigia 2010, 90, 433–448. [Google Scholar] [CrossRef]
- Nguyen, T.T.L.; Pham, N.T.T.; Tran, T.M.H. Occurences of Microcystis spp. and microcystins in some cyanobacterial blooms in freshwater bodies in Vietnam. VNU J. Sci. Nat. Sci. Technol. 2010, 26, 172–177. [Google Scholar]
- Duong, T.; LE, T.; Dao, T.-S.; Pflugmacher, S.; Rochelle-Newall, E.; Hoang, T.; VU, T.; Ho, C.; Dang, D. Seasonal variation of cyanobacteria and microcystins in the Nui Coc Reservoir, Northern Vietnam. J. Appl. Phycol. 2013, 25, 1065–1075. [Google Scholar] [CrossRef]
- Pham, T.-L.; Shimizu, K.; Dao, T.-S.; Motoo, U. First report on free and covalently bound microcystins in fish and bivalves from Vietnam: Assessment of risks to humans. Environ. Toxicol. Chem. 2017, 36, 2953–2957. [Google Scholar] [CrossRef] [PubMed]
- NOAA National Centers for Environmental Information. State of the Climate: Global Climate Report for April 2016. Published Online May 2016. Available online: https://www.ncdc.noaa.gov/sotc/global/201604 (accessed on 21 November 2017).
- Wetzel, R.G. Limnology: Lake and River Ecosystems, 3rd ed.; Academic Press: San Diego, CA, USA, 2001; pp. 213–241. [Google Scholar]
- Nguyen, T.T.L.; Cronberg, G.; Annadotter, H.; Larsen, J. Planktic cyanobacteria from freshwater localities in Thuathien-Hue Province, Vietnam II. Algal biomass and microcystin production. Nova Hedwigia 2007, 85, 35–49. [Google Scholar] [CrossRef]
- Duong, T.T.; Jähnichen, S.; Le, T.P.Q.; Ho, C.T.; Hoang, T.K.; Nguyen, T.K.; Vu, T.N.; Dang, D.K. The occurrence of cyanobacteria and microcystins in the Hoan Kiem Lake and the Nui Coc reservoir (North Vietnam). Environ. Earth Sci. 2014, 71, 2419–2427. [Google Scholar] [CrossRef]
- Pham, T.-L.; Dao, T.-S.; Shimizu, K.; Lan-Chi, D.-H.; Utsumi, M. Isolation and characterization of microcystin-producing cyanobacteria from Dau Tieng Reservoir, Vietnam. Nova Hedwigia 2015, 101, 3–20. [Google Scholar] [CrossRef]
- Drobac, D.; Tokodi, N.; Lujić, J.; Marinović, Z.; Subakov-Simić, G.; Dulić, T.; Važić, T.; Nybom, S.; Meriluoto, J.; Codd, G.A.; et al. Cyanobacteria and cyanotoxins in fishponds and their effects on fish tissue. Harmful Algae 2016, 55, 66–76. [Google Scholar] [CrossRef] [PubMed]
- Jia, J.; Luo, W.; Lu, Y.; Giesy, J.P. Bioaccumulation of microcystins (MCs) in four fish species from Lake Taihu, China: Assessment of risks to humans. Sci. Total Environ. 2014, 487, 224–232. [Google Scholar] [CrossRef] [PubMed]
- Gurbuz, F.; Uzunmehmetoğlu, O.Y.; Diler, Ö.; Metcalf, J.S.; Codd, G.A. Occurrence of microcystins in water, bloom, sediment and fish from a public water supply. Sci. Total Environ. 2016, 562, 860–868. [Google Scholar] [CrossRef] [PubMed]
- Greer, B.; Maul, R.; Campbell, K.; Elliott, C.T. Detection of freshwater cyanotoxins and measurement of masked microcystins in tilapia from Southeast Asian aquaculture farms. Anal. Bioanal. Chem. 2017, 409, 4057–4069. [Google Scholar] [CrossRef] [PubMed]
- Deblois, C.P.; Aranda-Rodriguez, R.; Giani, A.; Bird, D.F. Microcystin accumulation in liver and muscle of tilapia in two large Brazilian hydroelectric reservoirs. Toxicon 2008, 51, 435–448. [Google Scholar] [CrossRef] [PubMed]
- Nyakairu, G.W.A.; Nagawa, C.B.; Mbabazi, J. Assessment of cyanobacteria toxins in freshwater fish: A case study of Murchison Bay (Lake Victoria) and Lake Mburo, Uganda. Toxicon 2010, 55, 939–946. [Google Scholar] [CrossRef] [PubMed]
- Ramlal, P.S.; Bugenyi, F.W.B.; Kling, G.W.; Nriagu, J.O.; Rudd, J.W.M.; Campbell, L.M. Mercury Concentrations in Water, Sediment, and Biota from Lake Victoria, East Africa. J. Great Lakes Res. 2003, 29, 283–291. [Google Scholar] [CrossRef]
- Poste, A.E.; Hecky, R.E.; Guildford, S.J. Evaluating microcystin exposure risk through fish consumption. Environ. Sci. Technol. 2011, 45, 5806–5811. [Google Scholar] [CrossRef] [PubMed]
- Fischer, A.; Hoeger, S.J.; Stemmer, K.; Feurstein, D.J.; Knobeloch, D.; Nussler, A.; Dietrich, D.R. The role of organic anion transporting polypeptides (OATPs/SLCOs) in the toxicity of different microcystin congeners in vitro: A comparison of primary human hepatocytes and OATP-transfected HEK293 cells. Toxicol. Appl. Pharmacol. 2010, 245, 9–20. [Google Scholar] [CrossRef] [PubMed]
- Vesterkvist, P.; Misiorek, J.; Spoof, L.; Toivola, D.; Meriluoto, J. Comparative Cellular Toxicity of Hydrophilic and Hydrophobic Microcystins on Caco-2 Cells. Toxins 2012, 4, 1008. [Google Scholar] [CrossRef] [PubMed]
- Faassen, E.; Lürling, M. Occurrence of the Microcystins MC-LW and MC-LF in Dutch Surface Waters and Their Contribution to Total Microcystin Toxicity. Mar. Drugs 2013, 11, 2643–2654. [Google Scholar] [CrossRef] [PubMed]
- Dao, T.-S.; Nimptsch, J.; Wiegand, C. Dynamics of cyanobacteria and cyanobacterial toxins and their correlation with environmental parameters in Tri an Reservoir, Vietnam. J. Water Health 2016, 14, 699–712. [Google Scholar] [CrossRef] [PubMed]
- Pham, T.-L.; Dao, T.-S.; Tran, N.-D.; Nimptsch, J.; Wiegand, C.; Motoo, U. Influence of environmental factors on cyanobacterial biomass and microcystin concentration in the Dau Tieng Reservoir, a tropical eutrophic water body in Vietnam. Ann. Limnol. Int. J. Limnol. 2017, 53, 89–100. [Google Scholar] [CrossRef]
- Bui, T.; Dao, T.-S.; Vo, T.-G.; Lürling, M. Warming Affects Growth Rates and Microcystin Production in Tropical Bloom-Forming Microcystis Strains. Toxins 2018, 10, 123. [Google Scholar] [CrossRef] [PubMed]
- Vietnam Water, Sanitation and Environment Company (VIWASE). Master Plan for HCMC Water Suppy System up to 2025; VIWASE: Ho Chi Minh City, Vietnam, 2008. [Google Scholar]
- Dan, N.; Khoa, L.; Bui, X.-T.; Nga, P.; Visvanathan, C. Potential of Wastewater Reclamation to Reduce Fresh Water Stress in Ho Chi Minh City-Vietnam. J. Water Sustain. 2011, 1, 279–287. [Google Scholar]
- Nha Trang, V.; Phuong, L.; Dan, N.; Bui, X.-T.; Visvanathan, C. Assessment on the trihalomethanes formation potential of Tan Hiep Water Treatment Plant. J. Water Sustain. 2012, 2, 43–53. [Google Scholar]
- Özdemir, S.; Elliott, M.; Brown, J.; Nam, P.K.; Hien, V.T.; Sobsey, M.D. Rainwater harvesting practices and attitudes in the mekong delta of Vietnam. J. Water Sanit. Hyg. Dev. 2011, 1, 171–177. [Google Scholar] [CrossRef]
- General Statistics Office of Vietnam. Result of the Vietnam Household Living Standards Survey 2006; Statistics Publishing House: Hanoi, Vietnam, 2008.
- Matsukura, K.; Tsumuki, H.; Izumi, Y.; Wada, T. Changes in chemical components in the freshwater apple snail, Pomacea canaliculata (Gastropoda: Ampullariidae), in relation to the development of its cold hardiness. Cryobiology 2008, 56, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Hayes, K.A.; Joshi, R.C.; Thiengo, S.C.; Cowie, R.H. Out of South America: Multiple Origins of Non-Native Apple Snails in Asia. Divers. Distrib. 2008, 14, 701–712. [Google Scholar] [CrossRef]
- Van Tu, D.; Nha, N.P.; Joshi, R.C. Invasive apple snails (Pomacea spp.) in Vietnam: Short review. Aquac. Asia Mag. 2018, 22, 3–8. [Google Scholar]
- Da, C.T.; Lundh, T.; Lindberg, J.E. Evaluation of local feed resources as alternatives to fish meal in terms of growth performance, feed utilisation and biological indices of striped catfish (Pangasianodon hypophthalmus) fingerlings. Aquaculture 2012, 364–365, 150–156. [Google Scholar] [CrossRef]
- Xuan, B.; Nguyen, M.V.; Cong, T. Use of Golden apple snail pests as a protein supplement in diets of meat ducks. CanTho Univ. J. Sci. 2010, 16, 199–208. [Google Scholar]
- Martínez-Alvarez, O.; Chamorro, S.; Brenes, A. Protein hydrolysates from animal processing by-products as a source of bioactive molecules with interest in animal feeding: A review. Food Res. Int. 2015, 73, 204–212. [Google Scholar] [CrossRef]
- Tuoitre NEWS. 2004. Available online: https://tuoitre.vn/ca-lau-kinh---mot-loai-ngoai-lai-xam-hai-14863.htm (accessed on 5 June 2018).
- Tuoitre NEWS. 2011. Available online: https://tuoitre.vn/ca-lau-kinh-len-ban-nhau-431194.htm (accessed on 5 June 2018).
- DanTri NEWS. 2018. Available online: https://dantri.com.vn/suc-khoe/hau-qua-an-ca-lau-kinh-de-tri-tieu-duong-2018071906492906.htm (accessed on 9 September 2018).
- APHA. Standard Methods for the Examination of Water and Wastewater, 21st ed.; American Public Health Association: Washington, DC, USA; American Water Works Foundation: Denver, CO, USA; Water Environment Federation: Alexandria, VA, USA, 2005; p. 2671. [Google Scholar]
- Hoshaw, R.; Rosowski, J.R. Methods for microscopic algae. In Handbook of Phycological Methods. Culture Methods and Growth Measurements; Stein, R., Ed.; Cambridge University Press: London, UK, 1973; pp. 53–68. [Google Scholar]
- Lürling, M.; Beekman, W. Palmelloids formation in Chlamydomonas reinhardtii: Defence against rotifer predators? Ann. Limnol. 2006, 42, 65–72. [Google Scholar] [CrossRef]
- Lürling, M.; van Oosterhout, F.; Faassen, E.J. Eutrophication and warming boost cyanobacterial biomass and microcystins. Toxins 2017, 9, 64. [Google Scholar] [CrossRef] [PubMed]


| Location | Water Body | Domi. | Temp. (°C) | Sal. (‰) | pH | TN (mg/L) | TP (mg/L) | N-NH4 (mg/L) | N-NO3 (mg/L) | P-PO4 (mg/L) | Chl-a (µg/L) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| TraVinh | Duck–fish pond | Mic | 33.1 | 0.5 | 9.58 | 19.5 | 1.75 | 1.14 | 0.28 | 0.02 | 4352 |
| TraVinh | Mekong river | Mic | 33.2 | 6.7 | 7.88 | 2.07 | 0.05 | BLD | 0.08 | 0.02 | 28.9 |
| TraVinh | Shrimp pond | Plan | 27.5 | 1.0 | 8.75 | 8.19 | 0.44 | 0.83 | 0.08 | BLD | 300 |
| LongAn | Wastewater pond | Plan | 31.5 | 0.0 | 9.22 | 27.1 | 2.75 | 2.12 | 0.31 | 0.43 | 1572 |
| LongAn | Duck pond | Plan | 31.2 | 0.0 | 9.17 | 60.7 | 1.00 | 1.06 | 0.14 | 0.06 | 310 |
| BinhChanh | Fish pond | Mic | 37.5 | 7.4 | 9.89 | 9.13 | 0.25 | 0.57 | BLD | BLD | 1480 |
| BinhChanh | Fish pond | Mic | 37.4 | 7.5 | 9.77 | 9.00 | 0.33 | 0.67 | BLD | BLD | 1520 |
| BinhChanh | Fish pond | Mic | 37.4 | 7.4 | 9.79 | 9.36 | 0.30 | 0.46 | BLD | BLD | 1437 |
| BinhChanh | Fish pond | Mic | 37.7 | 7.6 | 9.66 | 16.91 | 1.52 | 1.02 | 0.04 | 0.19 | 5100 |
| BinhChanh | Wastewater canal | Mic | 36.0 | 7.0 | 9.00 | 8.62 | 0.71 | 0.55 | BLD | BLD | 467 |
| CuChi | Rice farm | Cyl | 34.9 | 0.0 | 9.13 | 6.78 | 0.43 | 0.53 | 0.23 | BLD | 274 |
| CuChi | Fishing pond | Plan | 34.3 | 0.0 | 7.91 | 3.6 | 0.09 | 0.17 | 0.12 | BLD | 29.7 |
| An Giang | reservoir | Osc/Plan | 33.6 | 0.0 | 7.96 | 1.77 | 0.06 | 0.96 | 0.18 | 0.03 | 45.6 |
| An Giang | Catfish pond | Plan | 30.0 | 0.1 | 7.31 | 9.38 | 1.60 | 3.96 | 0.1 | 1.04 | 221 |
| DauTieng | Duck–fish pond | Mic | 33.5 | 0.0 | 9.82 | 3.98 | 0.59 | 0.39 | 0.14 | 0.10 | 169 |
| DauTieng | reservoir | Mic | 34.0 | 0.0 | 8.12 | 3.89 | 0.59 | 0.39 | 0.14 | 0.10 | 94 |
| TriAn | reservoir | Mic | 34.2 | 0.0 | 8.57 | 15.5 | 0.84 | - | 0.4 | 0.10 | 420 |
| Location | Water Body | Domi. Genera | Chl-a | MC Concentration | |
|---|---|---|---|---|---|
| µg/L | µg/L | µg/g DW | |||
| TraVinh | Duck–fish pond | Mic | 4352 | 11,039 | 4033 |
| LongAn | duck pond | Osc | 310 | 0.18 | <LOD |
| Mekong-TraVinh | river | Mic | 29 | 57 | 2591 |
| TriAn-DongNai | reservoir | Mic | 187 | 2610 | - |
| DauTieng-TayNinh | reservoir | Mic | 169 | 30 | - |
| DauTieng-TayNinh | reservoir | Mic | 94 | - | 485 |
| BinhChanh | Fish pond | Mic | 5100 | 821 | 1156 |
| BinhChanh | Fish pond | Mic | 1520 | 293 | 1477 |
| CuChi | Canal | Plank | 8 | 0.03 | - |
| CuChi | Fish pond | Plank | 30 | 0.08 | - |
| An Giang | Reservoir | Plank | 46 | 0.31 | - |
| XuanHuong-DaLat | Lake | Mic | - | - | 6 |
| TuyenLam-DaLat | Lake | Mic | - | - | 278 |
| DongNai | River | Mic | - | - | 602–664 |
| Location Water Bodies | Species/Genera Dominant | MC Variants | Dominant Variants | Microcystin Concentration | Ref. | |
|---|---|---|---|---|---|---|
| µg/L | µg/g DW | |||||
| Lake Thanh Cong, Ha Noi | M. aeruginosa | MC-RR, MC-YR, MC-WR, MC-dmRR, MC-dmWR, MC-LR | MC-WR | 4240 | [21] | |
| Pond and rivers in Hue | M. spp. | MC-RR, MC-LR | MC-RR, MC-LR | 1.02–76.20 | [28] | |
| TriAn reservoir | M. botrys M. wesenbergii | MC-LR, MC-RR, MC-LA, MC-LY, | MC-RR MC-LR | 450–640 | [22] | |
| NuiCoc resrvoir | M. aeruginosa, M. wesenbergii, M. botrys. | MC-RR MC-LR | MC-RR MC-LR | 45.4–1699 | [24,29] | |
| DauTieng reservoir | Microcystis aeruginosa | MC-LR, MC-RR, MC-YR | MC-RR | 521–669 | [30] | |
| Duck–fish pond TraVinh | Microcystis spp. | MC-dmRR, MC-RR, YR and dmLR | MC-RR | 7771–91,721 | 407–4033 | This study |
| Mekong river TraVinh | Microcystis spp. | MC-dmRR, MC-RR, YR and dmLR, MC-LR, MC-LY, MC-LW, MC-LF | MC-LR | 57.26 | 1575–2591 | This study |
| TriAn reservoir | Microcystis spp. | MC-RR, dmLR, MC-LR, MC-YR, MC-LY, MC-LW, | MC-LR | 425–3619 | This study | |
| DauTieng reservoir | Microcystis spp. | MC-dmRR, MC-RR, dmLR, MC-LR | MC-RR | 0.09–30 | 485 | This study |
| Fish pond BinhChanh | Microcystis spp. | dmLR, MC-LR, MC-YR | 293–821 | 1156–1477 | This study | |
| XuanHuong Lake | Microcystis spp. | MC-LR | MC-LR | 6 | This study | |
| TuyenLam Lake | Microcystis spp. | MC-RR, MC-YR, dmLR, MC-LR | MC-RR | 278 | This study | |
| DongNai river | Microcystis spp. | MC-dmRR, MC-RR, MC-YR, dmLR, MC-LR, MC-LY, MC-LW, MC-LF | MC-LR | 602–664 | This study | |
| Organism/Species | Organ | MC Concentration (µg/g DW) | Location | Ref. |
|---|---|---|---|---|
| Fish | ||||
| Common carp Cyprinus carpio | G, M | 0.36; UD | BinhChanh | This study |
| M | 0.060 | Fishponds, Serbia | [31] | |
| M, L, K, I, H | 0.0317; 0.0295; 0.0323; 0.133; 0.019 | Lake Taihu, China | [32] | |
| M, L | 0.69–3.45; 1.09–2.05 | Eğirdir, Turkey | [33] | |
| Golden carp Carassius auratus | VM, M | 18.57, UD | BinhChanh | This study |
| M, L, K, I, H | 0.0267, 0.0454, 0.114, 2.04, 0.0595 | Taihu, China | [32] | |
| Suckermouth catfish (Hypostomus punctatus) | G, L, M | 108.38; 18.97; 3.73 | BinhChanh | This study |
| Tilapia (Oreochromis sp.) | G; L, M | 115.95; 13.39; UD | BinhChanh | This study |
| G, VM, L, M | 11.55; 2.87; UD; UD | TriAn reservoir | This study | |
| G, M | 0.76; UD | DauTieng reservoir | This study | |
| L, I, M | 2.1; 2.6; UD | DauTieng reservoir | [25] | |
| L, M | 0.0562; 0.0134–0.0168 | Fish pond, Southeast Asian | [34] | |
| L | 6.752 *–8.682 * | Funil and Furnas Reservoirs, Brazil | [35] | |
| G, L, M | 4.756 *; 0.154 *; 0.029 * | Lake Victoria, Uganda | [36] | |
| G, L, M | 4.219 *; 0.235 *; 0.672 * | Lake Mburo, Uganda | [36] | |
| Snakeskin gourami (Trichogaster pectoralis) | VM, M | 13.86; UD | BinhChanh | This study |
| Ganges river sprat (Corica soborna) | VM, M | 2.38, UD | TriAn reservoir | This study |
| Snails | ||||
| Apple snail (Pomacea canaliculata) | VM, M | 0.21–3.21; 0.15–0.30 | TriAn reservoir | This study |
| VM | 1.61–2.90 | BinhChanh | This study | |
| Shrimp | ||||
| White leg shrimp (Litopenaeus vannamei) | VM, M | 15.21; UD | TraVinh | This study |
| EDI | Muscle Tissue | Visceral Mass | Gut | Liver | Egg | Head |
|---|---|---|---|---|---|---|
| Snails-TriAn | 0.039 | 0.107 | - | - | 0.038 | - |
| Snails BinhChanh | 0 | 1.997 | - | - | 0 | - |
| Ganges river sprat-TriAn | 0 | 0.618 | - | - | - | - |
| Sknakskin gourami fish-BinhChanh | 0 | 7.155 | - | - | - | 0 |
| Tilapia-TriAn | 0 | 0.119 | 1.111 | - | - | - |
| Tilapia-DauTieng | 0 | 0.393 | - | - | - | - |
| Tilapia-BinhChanh | 0 | - | 57.61 | 6.819 | - | - |
| Suckermouth catfish-BinhChanh | 1.903 | - | 52.19 | 9.441 | - | - |
| TDI recommended by WHO | 0.04 | 0.04 | 0.04 | 0.04 | 0.04 | 0.04 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Trung, B.; Dao, T.-S.; Faassen, E.; Lürling, M. Cyanobacterial Blooms and Microcystins in Southern Vietnam. Toxins 2018, 10, 471. https://doi.org/10.3390/toxins10110471
Trung B, Dao T-S, Faassen E, Lürling M. Cyanobacterial Blooms and Microcystins in Southern Vietnam. Toxins. 2018; 10(11):471. https://doi.org/10.3390/toxins10110471
Chicago/Turabian StyleTrung, Bui, Thanh-Son Dao, Elisabeth Faassen, and Miquel Lürling. 2018. "Cyanobacterial Blooms and Microcystins in Southern Vietnam" Toxins 10, no. 11: 471. https://doi.org/10.3390/toxins10110471
APA StyleTrung, B., Dao, T.-S., Faassen, E., & Lürling, M. (2018). Cyanobacterial Blooms and Microcystins in Southern Vietnam. Toxins, 10(11), 471. https://doi.org/10.3390/toxins10110471






